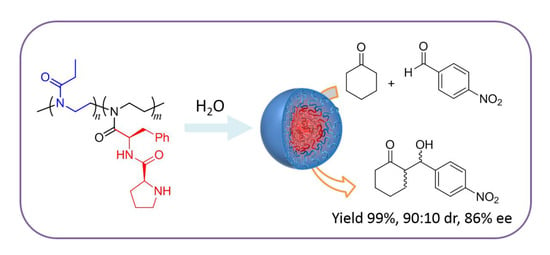
A Pseudopeptide Polymer Micelle Used for Asymmetric Catalysis of the Aldol Reaction in Water
Abstract
1. Introduction
2. Materials and Methods
2.1. Raw Materials and Instruments
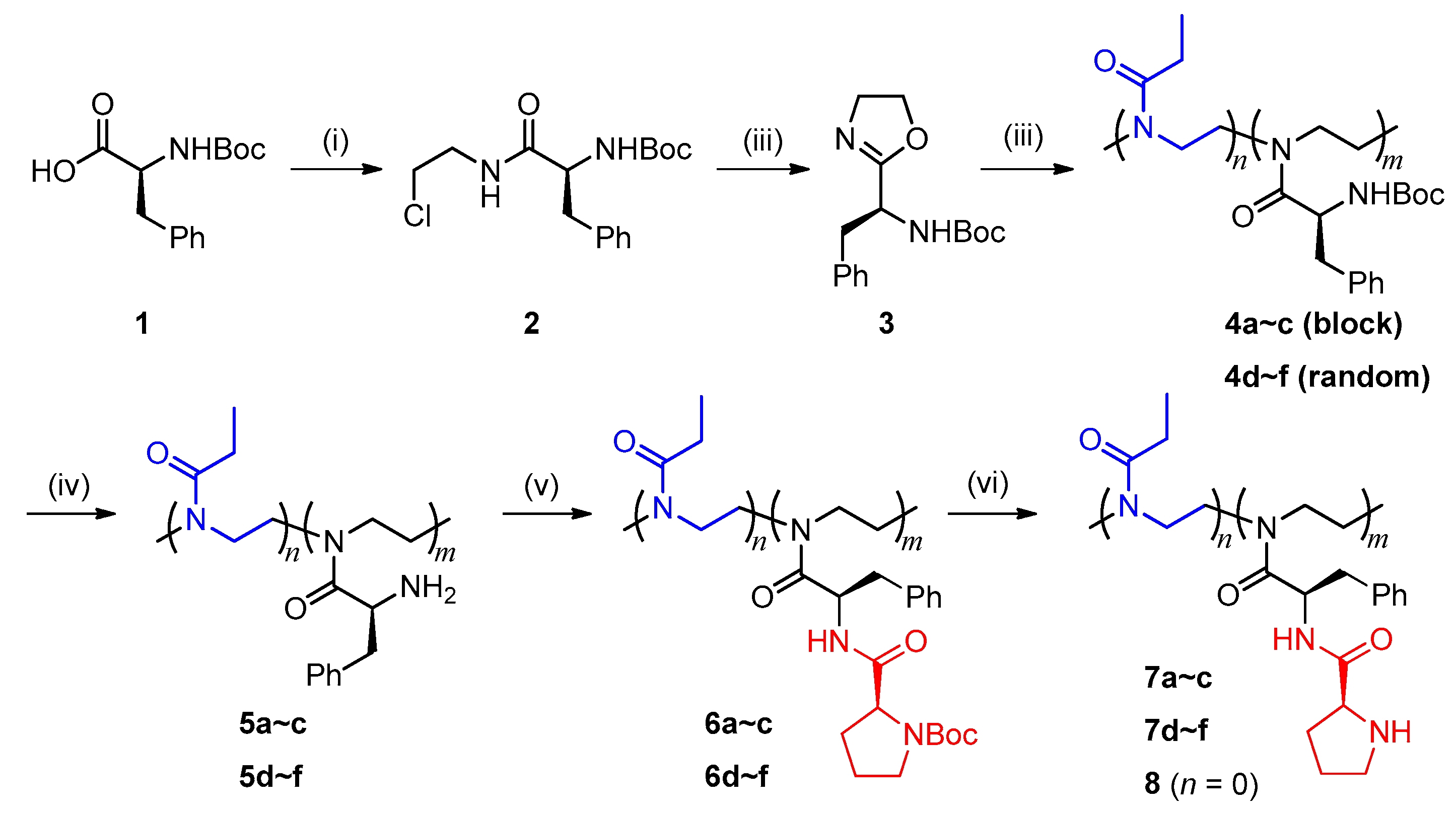
2.2. Synthesis of Polymeric Catalysts
2.2.1. (S)-2-(1-Boc-amino-2-phenyl)ethyl-2-oxazoline (3)
2.2.2. Cationic Ring-Opening Copolymerization of Monomer 3 and 2-ethyl-2-oxazoline (EtOx)
2.2.3. Postmodification of Precursor Polymers
2.3. Micellization of Amphiphilic Copolymers
2.4. General Procedure for Aldol Reaction
3. Results and Discussion
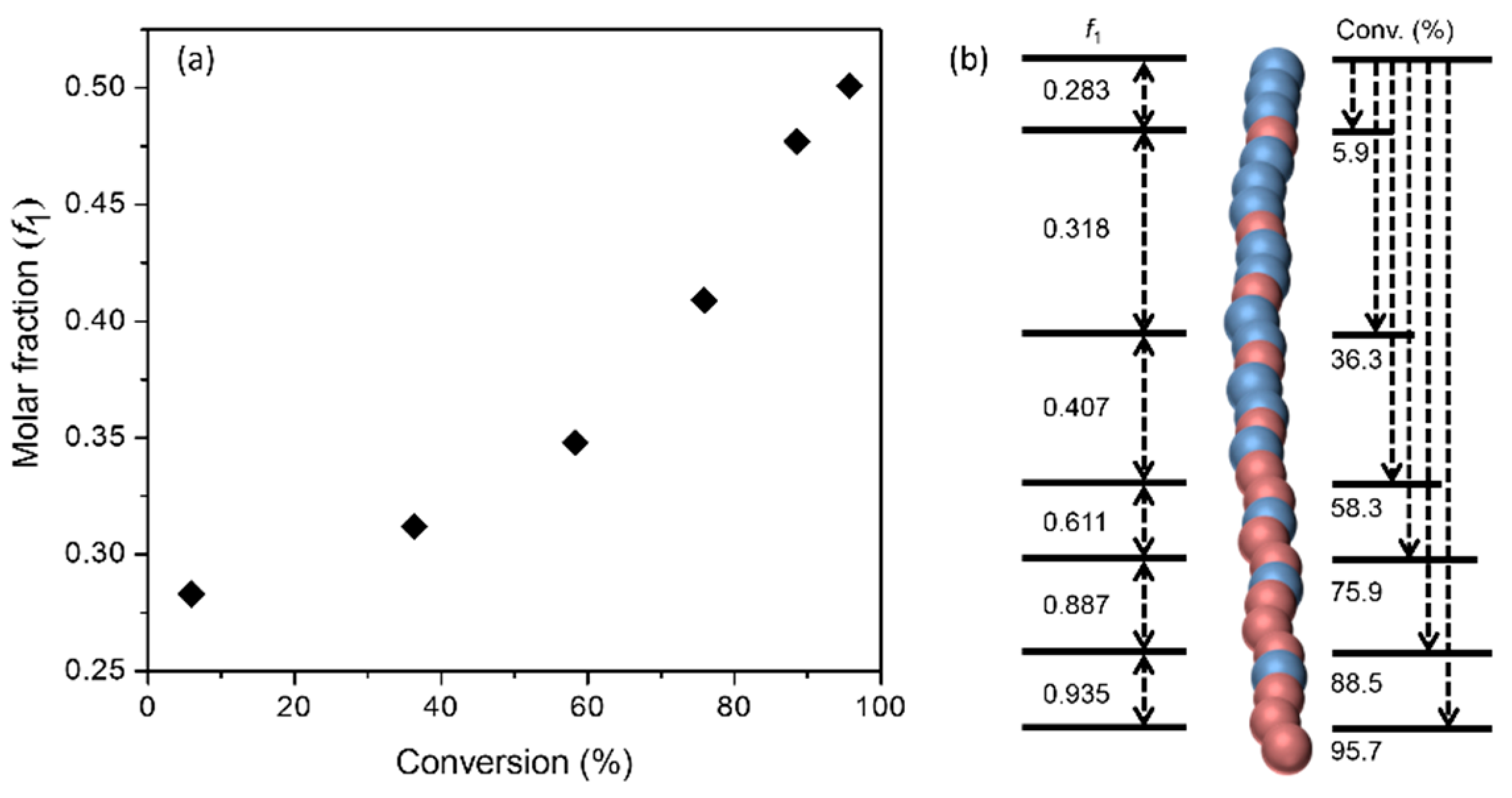
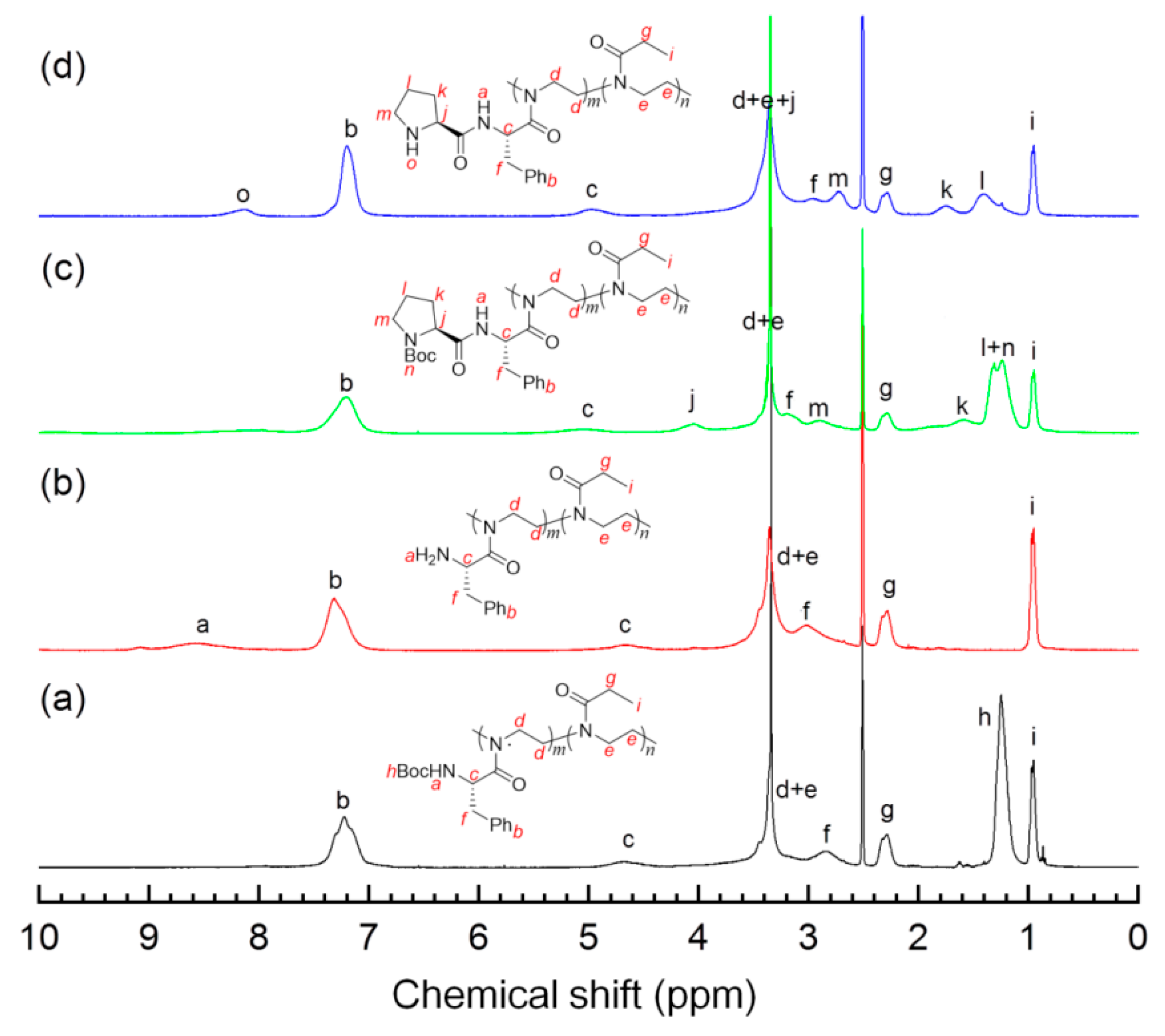
3.1. Polymerization and Structural Characterization of Polymers
3.2. Postmodification of Copolymer 4
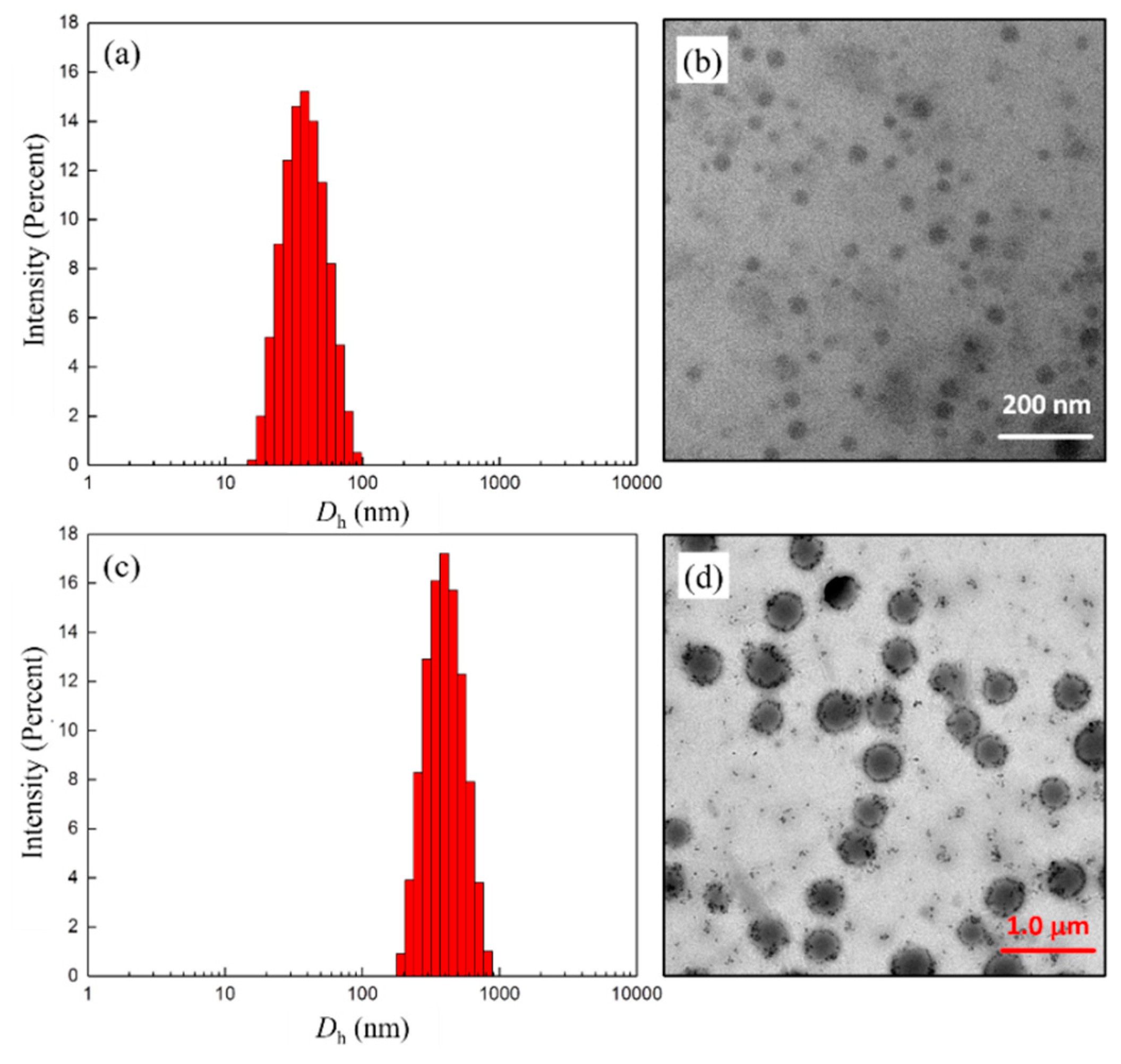
3.3. Self-Assembly Behavior of Polymer 7 in Water
3.4. Evaluation of Catalytic Activity
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Benaglia, M.; Puglisi, A.; Cozzi, F. Polymer-supported organic catalysts. Chem. Rev. 2003, 103, 3401–3430. [Google Scholar] [CrossRef] [PubMed]
- Cozzi, F. Immobilization of organic catalysts: when, why, and how. Adv. Synth. Catal. 2006, 348, 1367–1390. [Google Scholar] [CrossRef]
- Gruttadauria, M.; Giacalone, F.; Noto, R. Supported proline and proline-derivatives as recyclable organocatalysts. Chem. Soc. Rev. 2008, 37, 1666–1688. [Google Scholar] [CrossRef] [PubMed]
- Yin, Y.; Dong, Z.; Luo, Q.; Liu, J. Biomimetic catalysts designed on macromolecular scaffolds. Prog. Polym. Sci. 2012, 37, 1476–1509. [Google Scholar] [CrossRef]
- Breslow, R.; Bandyopadhyay, S.; Levine, M.; Zhou, W. Water exclusion and enantioselectivity in catalysis. ChemBioChem 2006, 7, 1491–1496. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Liu, K.; Xing, R.R.; Yan, X.H. Peptide self-assembly: thermodynamics and kinetics. Chem. Soc. Rev. 2016, 45, 5589–5604. [Google Scholar] [CrossRef] [PubMed]
- Torchilin, V.P. Structure and design of polymeric surfactant-based drug delivery systems. J. Controlled Release 2001, 73, 137–172. [Google Scholar] [CrossRef]
- Wong, C.H.; Halcomb, R.L.; Ichikawa, Y.; Kajimoto, T. Enzymes in organic synthesis: application to the problems of carbohydrate recognition (part 1). Angew. Chem. Int. Ed. 1995, 34, 412–432. [Google Scholar] [CrossRef]
- Gruttadauria, M.; Giacalone, F.; Marculescu, A.M.; Lo Meo, P.; Riela, S.; Noto, R. Hydrophobically directed aldol reactions: polystyrene-supported L-proline as a recyclable catalyst for direct asymmetric aldol reactions in the presence of water. Eur. J. Org. Chem. 2007, 28, 4688–4698. [Google Scholar] [CrossRef]
- Benaglia, M.; Celentano, G.; Cozzi, F. Enantioselective aldol condensations catalyzed by poly(ethylene glycol)-supported proline. Adv. Synth. Catal. 2001, 343, 171–173. [Google Scholar] [CrossRef]
- Benaglia, M.; Cinquini, M.; Cozzi, F.; Puglisi, A.; Celentano, G. Poly(ethylene glycol)-supported proline: a versatile catalyst for the enantioselective aldol and iminoaldol reactions. Adv. Synth. Catal. 2002, 344, 533–542. [Google Scholar] [CrossRef]
- Huerta, E.; Stals, P.J.; Meijer, E.W.; Palmans, A.R.A. Consequences of folding a water-soluble polymer around an organocatalyst. Angew. Chem. Int. Ed. 2013, 52, 2906–2910. [Google Scholar] [CrossRef] [PubMed]
- Lu, A.; Moatsuo, D.; Longbottom, D.A.; O’Reilly, R.K. Tuning the catalytic activity of L-proline functionalized hydrophobic nanogel particles in water. Chem. Sci. 2013, 4, 965–969. [Google Scholar] [CrossRef]
- Kehat, T.; Goren, K.; Portnoy, M. Effects of dendritic interface on enantioselective catalysis by polymer- bound prolines. New J. Chem. 2012, 36, 394–401. [Google Scholar] [CrossRef]
- Lu, A.; Cotanda, P.; Patterson, J.P.; Longbottom, D.A.; O’Reilly, R.K. Aldol reactions catalyzed by L-proline functionalized polymeric nanoreactors in water. Chem. Commun. 2012, 48, 9699–9701. [Google Scholar] [CrossRef] [PubMed]
- Zayas, H.A.; Lu, A.; Valade, D.; Amir, F.; Jia, Z.; O’Reilly, R.K.; Monteiro, M.J. Thermoresponsive polymer-supported L-proline micelle catalysts for the direct asymmetric aldol reaction in water. ACS Macro Lett. 2013, 2, 327–331. [Google Scholar] [CrossRef]
- Guo, G.; Wu, Y.; Zhao, X.; Wang, J.; Zhang, L.; Cui, Y. Polymerization of L-proline functionalized styrene and its catalytic performance as a supported organocatalyst for direct enantioselective aldol reaction. Tetrahedron Asymmetry 2016, 27, 740–746. [Google Scholar] [CrossRef]
- Arakawa, Y.; Wennemers, H. Enamine catalysis in flow with an immobilized peptidic catalyst. ChemSusChem. 2013, 6, 242–245. [Google Scholar] [CrossRef] [PubMed]
- Doyagüez, E.G.; Parra, F.; Corrales, G.; Fernández-Mayoralas, A.; Gallardo, A. New hydroxyproline based methacrylic polybetaines: Synthesis, pH sensitivity and catalytic activity. Polymer 2009, 50, 4438–4446. [Google Scholar] [CrossRef]
- Uemukai, T.; Ishifune, M. Synthesis and catalytic activity of the thermoresponsive polymers having pyrrolidine side chains as base functionalities. J. Appl. Polym. Sci. 2013, 127, 2554–2560. [Google Scholar] [CrossRef]
- Wilson, P.; Ke, P.C.; Davis, T.P.; Kempe, K. Poly(2-oxazoline)-based micro-and nanoparticles: A review. Eur. Polym. J. 2017, 88, 486–515. [Google Scholar] [CrossRef]
- Hoogenboom, R. Poly(2-oxazoline)s: A polymer class with numerous potential applications. Angew. Chem. Int. Ed. 2009, 48, 7978–7994. [Google Scholar] [CrossRef] [PubMed]
- Luxenhofer, R.; Han, Y.; Schulz, A.; Tong, J.; He, Z.; Kabanov, A.V.; Jordan, R. Poly(2-oxazoline)s as polymer therapeutics. Macromol. Rapid. Commun. 2012, 33, 1613–1631. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Shen, H.; Zhou, L.; Hu, F.; Xie, S.; Jiang, L. Novel poly(2-oxazoline)s with pendant L-prolinamide moieties as efficient organocatalysts for direct asymmetric aldol reaction. Catal. Sci. Technol. 2016, 6, 6739–6749. [Google Scholar] [CrossRef]
- Hu, F.; Du, G.; Ye, L.; Zhu, Y.; Wang, Y.; Jiang, L. Novel amphiphilic poly(2-oxazoline)s bearing L-prolinamide moieties as the pendants: Synthesis, micellization and catalytic activity in aqueous aldol reaction. Polymer 2016, 102, 33–42. [Google Scholar] [CrossRef]
- Kobayashi, S.; Hachiya, I. Lanthanide triflates as water-tolerant Lewis acids. Activation of commercial formaldehyde solution and use in the aldol reaction of silyl enol ethers with aldehydes in aqueous media. J. Org. Chem. 1994, 59, 3590–3596. [Google Scholar] [CrossRef]
- Engelhardt, N.; Weberskirch, R. Synthesis and characterization of surface functional polymer nanoparticles by a bottom-up approach from tailor-made amphiphilic block copolymers. Macromol. Chem. Phys. 2013, 214, 2783–2791. [Google Scholar] [CrossRef]
- Vlassi, E.; Papagiannopoulos, A.; Pispas, S. Amphiphilic poly(2-oxazoline) copolymers as self-assembled carriers for drug delivery applications. Eur. Polym. J. 2017, 88, 516–523. [Google Scholar] [CrossRef]
- Hu, F.; Xie, S.; Jiang, L.; Shen, Z. Living cationic ring-opening polymerization of 2-oxazolines initiated by rare-earth metal triflates. RSC Adv. 2014, 4, 59917–59926. [Google Scholar] [CrossRef]
- Fineman, M.; Ross, S.D. Linear method for determining monomer reactivity ratios in copolymerization. J. Polym. Sci. 1950, 5, 259–262. [Google Scholar] [CrossRef]
- Lee, J.S.; Regatte, R.R.; Jerschow, A. Bloch equations for proton exchange reactions in an aqueous solution. Concepts Magn. Reson. Part A 2016, 45A, e21397. [Google Scholar] [CrossRef] [PubMed]
- Craven, C.J.; Derix, N.M.; Hendriks, J.; Boelens, R.; Hellingwerf, K.J.; Kaptein, R. Probing the nature of the blue-shifted intermediate of photoactive yellowprotein in solution by NMR: Hydrogen−deuterium exchange data and pH studies. Biochemistry 2000, 39, 14392–14399. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Liu, L.; Du, J. Probing into homopolymer self-assembly: how does hydrogen bonding influence morphology? Macromolecules 2012, 46, 194–203. [Google Scholar] [CrossRef]
- Feng, C.; Lu, G.; Li, Y.; Huang, X. Self-assembly of amphiphilic homopolymers bearing ferrocene and carboxyl functionalities: Effect of polymer concentration, β-cyclodextrin, and length of alkyl linker. Langmuir 2013, 29, 10922–10931. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.; Ren, C.; Yang, W.; Deng, J. Helical polymer as mimetic enzyme catalyzing asymmetric aldol reaction. Macromol. Rapid Commun. 2012, 33, 652–657. [Google Scholar] [CrossRef] [PubMed]
- Huerta, E.; van Genabeek, B.; Stals, P.J.M.; Meijer, E.W.; Palmans, A.R.A. A modular approach to introduce function into single-chain polymeric nanoparticles. Macromol. Rapid Commun. 2014, 35, 1320–1325. [Google Scholar] [CrossRef] [PubMed]
- Yeh, C.; Sun, Y.R.; Huang, S.J.; Tsai, Y.M.; Cheng, S. Alternating chiral selectivity of aldol reactions under the confined space of mesoporous silica. Chem. Commun. 2015, 51, 17116–17119. [Google Scholar] [CrossRef] [PubMed]
- Deng, J.; Deng, J. Optically active microspheres from helical substituted polyacetylene with pendent ferrocenyl amino-acid derivative. Preparation and recycling use for direct asymmetric aldol reaction in water. Polymer 2017, 125, 200–207. [Google Scholar] [CrossRef]
- Chen, F.; Huang, S.; Zhang, H.; Liu, F.; Peng, Y. Proline-based dipeptides with two amide units as organocatalyst for the asymmetric aldol reaction of cyclohexanone with aldehydes. Tetrahedron 2008, 64, 9585–9591. [Google Scholar] [CrossRef]
- Miura, T.; Imai, K.; Ina, M.; Tada, N.; Imai, N.; Itoh, A. Direct asymmetric aldol reaction with recyclable fluorous organocatalyst. Org. Lett. 2010, 12, 1620–1623. [Google Scholar] [CrossRef] [PubMed]
- de Arriba, Á.L.F.; Simon, L.; Raposo, C.; Alcazar, V.; Sanz, F.; Muniz, F.M.; Moran, J.R. Imidazolidinone intermediates in prolinamide-catalyzed aldol reactions. Org. Biomol. Chem. 2010, 8, 2979–2985. [Google Scholar] [CrossRef] [PubMed]
- Duss, M.; Manni, L.S.; Moser, L.; Handschin, S.; Mezzenga, R.; Jessen, H.J.; Landau, E.M. Lipidic mesophases as novel nanoreactor scaffolds for organocatalysts: Heterogeneously catalyzed asymmetric aldol reactions in confined water. ACS Appl. Mater. Inter. 2018, 10, 5114–5124. [Google Scholar] [CrossRef] [PubMed]
- Lei, M.; Shi, L.; Li, G.; Chen, S.; Fang, W.; Ge, Z.; Cheng, T.; Li, R. Dipeptide-catalyzed direct asymmetric aldol reactions in the presence of water. Tetrahedron 2007, 63, 7892–7898. [Google Scholar] [CrossRef]
- Hernández, J.G.; Juaristi, E. Asymmetric aldol reaction organocatalyzed by (S)-proline-containing dipeptides: improved stereoinduction under solvent-free conditions. J. Org. Chem. 2011, 76, 1464–1467. [Google Scholar] [CrossRef] [PubMed]
- Bayat, S.; Tejo, B.A.; Salleh, A.B.; Abdulmalek, E.; Yahya, N.M.; Rahman, M.B.A. Various polar tripeptides as asymmetric organocatalyst in direct aldol reactions in aqueous media. Chirality 2013, 25, 726–734. [Google Scholar] [CrossRef] [PubMed]
| Entry | Feed Ratio [3]/[EtOx]/[I] | Poly-3 | Copolymer 4 | |||||
|---|---|---|---|---|---|---|---|---|
| Mn2 | Đ2 | Code | Mn2 | Đ2 | Yield (%) 3 | 3 (mol%) 4 | ||
| 1 | 50/150/1 | 4900 | 1.19 | 4a | 8700 | 1.33 | 90 | 25 |
| 2 | 100/100/1 | 7800 | 1.25 | 4b | 11,400 | 1.38 | 83 | 50 |
| 3 | 150/50/1 | 11,600 | 1.34 | 4c | 14,000 | 1.45 | 60 | 80 |
| 4 | 50/150/1 | - | - | 4d | 9900 | 1.34 | 96 | 25 |
| 5 | 100/100/1 | - | - | 4e | 10,700 | 1.37 | 91 | 47 |
| 6 | 150/50/1 | - | - | 4f | 10,800 | 1.33 | 80 | 71 |
| Entry | Code | Mn1 | Đ1 | Ef (%) 2 |
|---|---|---|---|---|
| 1 | 6a | 6600 | 1.30 | 98 |
| 2 | 6b | 8700 | 1.39 | 97 |
| 3 | 6c | 10,000 | 1.54 | 90 |
| 4 | 6d | 8200 | 1.23 | 97 |
| 5 | 6e | 9600 | 1.27 | 95 |
| 6 | 6f | 11,900 | 1.30 | 91 |
| Entry | Code | DLS | TEM | |
|---|---|---|---|---|
| Dh (nm) 1 | PDI 1 | Size (nm) 2 | ||
| 1 | 7a | 212 | 0.259 | 190 ± 60 |
| 2 | 7b | 85 | 0.330 | 60 ± 20 |
| 3 | 7c | 35 | 0.112 | 30 ± 10 |
| 4 | 7d | 426 | 0.198 | 380 ± 60 |
| 5 | 7e | 376 | 0.182 | 330 ± 60 |
| 6 | 7f | 304 | 0.085 | 310 ± 70 |

| Entry | Catalyst | TFA (μL) | Yield (%) 2 | anti/syn 3 | ee (%) 4 |
|---|---|---|---|---|---|
| 1 | 7a | 0 | 85 | 86/14 | 58 |
| 2 | 7a | 1 | 90 | 83/17 | 71 |
| 3 | 7b | 0 | 96 | 87/13 | 63 |
| 4 | 7b | 1 | 99 | 88/12 | 77 |
| 5 | 7c | 0 | 93 | 87/13 | 67 |
| 6 | 7c | 1 | 96 | 90/10 | 86 |
| 7 | 7d | 0 | 94 | 71/29 | 31 |
| 8 | 7d | 1 | 91 | 81/19 | 82 |
| 9 | 7e | 0 | 89 | 86/14 | 70 |
| 10 | 7e | 1 | 93 | 87/13 | 82 |
| 11 | 7f | 0 | 91 | 77/23 | 65 |
| 12 | 7f | 1 | 96 | 83/17 | 80 |
| 13 | 8 5 | 0 | 97 | 77/23 | 52 |
| 14 | 85 | 1 | 96 | 81/19 | 80 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, K.; Ye, L.; Wang, Y.; Du, G.; Jiang, L. A Pseudopeptide Polymer Micelle Used for Asymmetric Catalysis of the Aldol Reaction in Water. Polymers 2018, 10, 1004. https://doi.org/10.3390/polym10091004
Liu K, Ye L, Wang Y, Du G, Jiang L. A Pseudopeptide Polymer Micelle Used for Asymmetric Catalysis of the Aldol Reaction in Water. Polymers. 2018; 10(9):1004. https://doi.org/10.3390/polym10091004
Chicago/Turabian StyleLiu, Keyuan, Long Ye, Yao Wang, Ganhong Du, and Liming Jiang. 2018. "A Pseudopeptide Polymer Micelle Used for Asymmetric Catalysis of the Aldol Reaction in Water" Polymers 10, no. 9: 1004. https://doi.org/10.3390/polym10091004
APA StyleLiu, K., Ye, L., Wang, Y., Du, G., & Jiang, L. (2018). A Pseudopeptide Polymer Micelle Used for Asymmetric Catalysis of the Aldol Reaction in Water. Polymers, 10(9), 1004. https://doi.org/10.3390/polym10091004