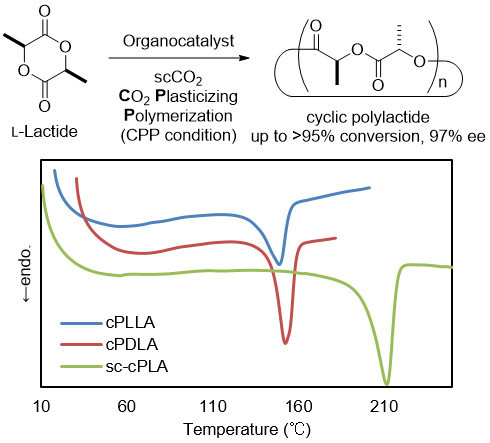
Organocatalytic Stereoselective Cyclic Polylactide Synthesis in Supercritical Carbon Dioxide under Plasticizing Conditions
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Synthesis
2.3. Characterization
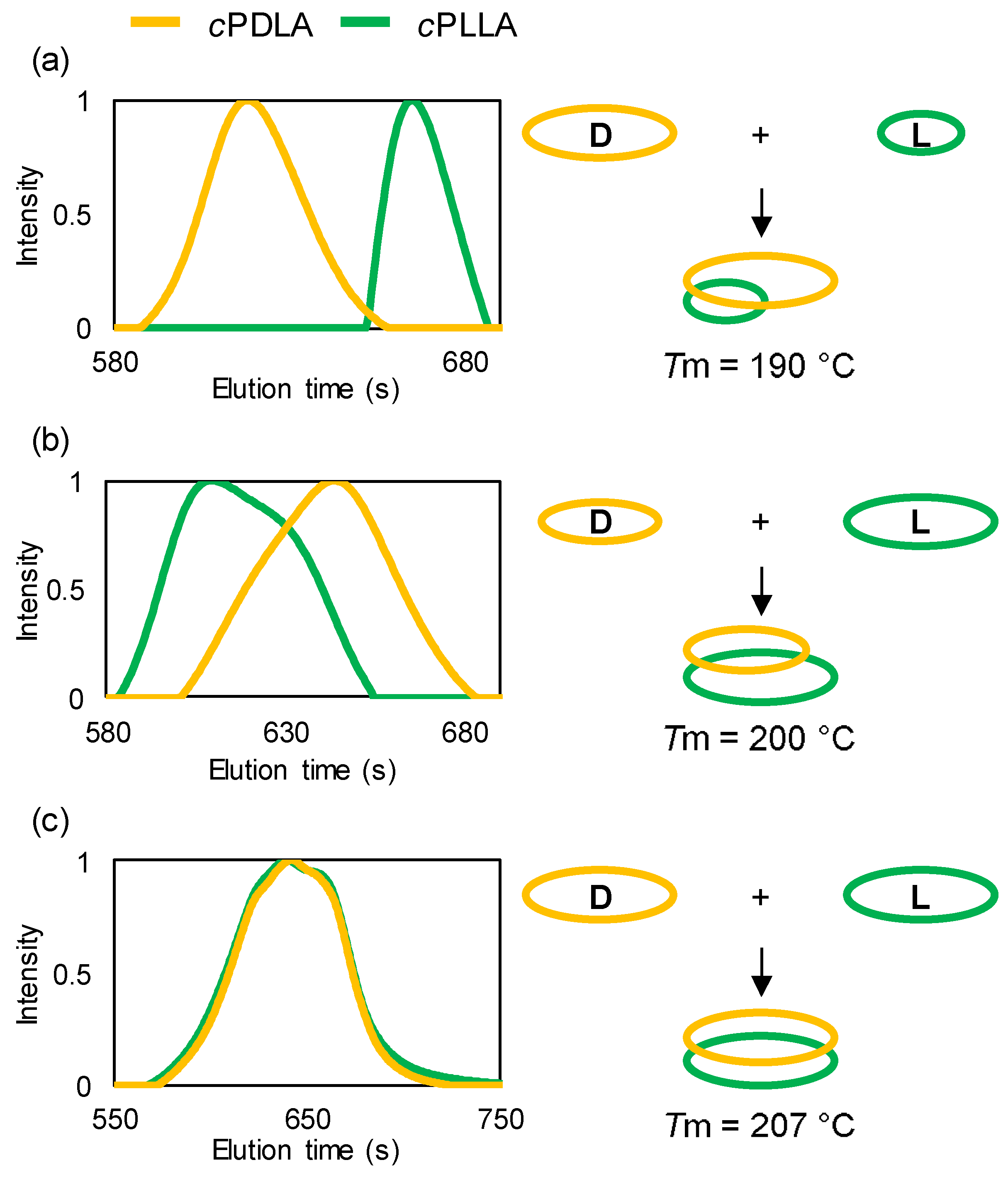
3. Results and Discussion
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Thakur, V.K.; Thakur, M.K. Handbook of Polymers for Pharmaceutical Technologies: Biodegradable Polymers; Scrivener Publishing LLC: New York, NY, USA, 2015; Volume 3, pp. 1–583. [Google Scholar] [CrossRef]
- Rieger, B.; Kunkel, A.; Coates, G.W.; Reichardt, R.; Dinjus, E.; Zevaco, T.A. Synthetic Biodegradable Polymers; Springer: Berlin/Heidelberg, Germany, 2012; Volume 245, pp. 1–364. [Google Scholar]
- Lendlein, A.; Sisson, A. Handbook of Biodegradable Polymers: Isolation, Synthesis, Characterization and Applications; Wiley-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2011; pp. 1–405. [Google Scholar]
- Nomura, K.; Ohara, H. Synthesis and function of cyclic poly(lactic acid) and oligo(lactic acid). Mini-Rev. Org. Chem. 2017, 14, 35–43. [Google Scholar] [CrossRef]
- Tezuka, Y. Topological Polymer Chemistry: Progress of Cyclic Polymers in Syntheses, Properties and Functions; World Scientific Publishing Co., Pte., Ltd.: Singapore, 2013; pp. 1–352. [Google Scholar]
- Yamamoto, T.; Tezuka, Y. Cyclic and multicyclic topological polymers. In Complex Macromolecular Architectures: Synthesis, Characterization, and Self-Assembly; Hadjichristidis, N., Hirao, A., Tezuka, Y., DuPrez, F., Eds.; John Wiley & Sons (Asia) Pte. Ltd.: Singapore, 2011; pp. 3–19. [Google Scholar]
- Endo, K. Synthesis and properties of cyclic polymers. In New Frontiers in Polymer Synthesis; Kobayashi, S., Ed.; Springer: Berlin/Heidelberg, Germany, 2008; Volume 217, pp. 121–183. [Google Scholar]
- Roovers, J. Organic cyclic polymers. In Cyclic Polymers, 2nd ed.; Semlyen, J.A., Ed.; Kluwer Academic Publishers: Dordrecht, The Netherlands, 2000; pp. 347–384. [Google Scholar]
- Takada, S.; Nagato, Y.; Yamamura, M. Effect of cyclic polylactates on tumor cells and tumor bearing mice. Biochem. Mol. Biol. Int. 1997, 43, 9–17. [Google Scholar] [CrossRef] [PubMed]
- Prasad, A.V.; Biying, A.O.; Ling, W.Y.; Stubbs, L.P.; Zhu, Y. Synthesis and new application of green and recyclable cyclic poly(l-lactide)-clay hybrid. J. Polym. Sci. Part A Polym. Chem. 2013, 51, 4167–4174. [Google Scholar] [CrossRef]
- Kato, H.; Naganushi, Y. Preparation of l-Lactic Acid Condensation Products for Cancer Treatment. JPH06336427A, 6 December 1994. [Google Scholar]
- Osaka, I.; Watanabe, M.; Takama, M.; Murakami, M.; Arakawa, R. Characterization of linear and cyclic polylactic acids and their solvolysis products by electrospray ionization mass spectrometry. J. Mass Spectrom. 2006, 41, 1369–1377. [Google Scholar] [CrossRef] [PubMed]
- Osaka, I.; Yoshimoto, A.; Watanabe, M.; Takama, M.; Murakami, M.; Kawasaki, H.; Arakawa, R. Quantitative determination of cyclic polylactic acid oligomers in serum by direct injection liquid chromatography tandem mass spectrometry. J. Chromatogr. B 2008, 870, 247–250. [Google Scholar] [CrossRef] [PubMed]
- Sugai, N.; Yamamoto, T.; Tezuka, Y. Synthesis of orientationally isomeric cyclic stereoblock polylactides with head-to-head and head-to-tail linkages of the enantiomeric segments. ACS Macro Lett. 2012, 1, 902–906. [Google Scholar] [CrossRef]
- Josse, T.; De Winter, J.; Dubois, P.; Coulembier, O.; Gerbaux, P.; Memboeuf, A. A tandem mass spectrometry-based method to assess the architectural purity of synthetic polymers: A case of a cyclic polylactide obtained by click chemistry. Polym. Chem. 2015, 6, 64–69. [Google Scholar] [CrossRef]
- Stanford, M.J.; Pflughaupt, R.L.; Dove, A.P. Synthesis of stereoregular cyclic poly(lactide)s via “thiol-ene” click chemistry. Macromolecules 2010, 43, 6538–6541. [Google Scholar] [CrossRef]
- Culkin, D.A.; Jeong, W.H.; Csihony, S.; Gomez, E.D.; Balsara, N.R.; Hedrick, J.L.; Waymouth, R.M. Zwitterionic polymerization of lactide to cyclic poly(lactide) by using N-heterocyclic carbene organocatalysts. Angew. Chem. Int. Ed. 2007, 46, 2627–2630. [Google Scholar] [CrossRef] [PubMed]
- Jeong, W.; Shin, E.J.; Culkin, D.A.; Hedrick, J.L.; Waymouth, R.M. Zwitterionic polymerization: A kinetic strategy for the controlled synthesis of cyclic polylactide. J. Am. Chem. Soc. 2009, 131, 4884–4891. [Google Scholar] [CrossRef] [PubMed]
- Shin, E.J.; Jones, A.E.; Waymouth, R.M. Stereocomplexation in cyclic and linear polylactide blends. Macromolecules 2012, 45, 595–598. [Google Scholar] [CrossRef]
- Prasad, A.V.; Stubbs, L.P.; Zhun, M.; Zhu, Y.H. Zwitterionic ring opening polymerization of lactide by metal free catalysts: Production of cyclic polymers. J. Appl. Polym. Sci. 2012, 123, 1568–1575. [Google Scholar] [CrossRef]
- Kricheldorf, H.R.; Lomadze, N.; Schwarz, G. Cyclic polylactides by imidazole-catalyzed polymerization of l-lactide. Macromolecules 2008, 41, 7812–7816. [Google Scholar] [CrossRef]
- Brown, H.A.; De Crisci, A.G.; Hedrick, J.L.; Waymouth, R.M. Amidine-mediated zwitterionic polymerization of lactide. ACS Macro Lett. 2012, 1, 1113–1115. [Google Scholar] [CrossRef]
- Zhang, X.Y.; Waymouth, R.M. Zwitterionic ring opening polymerization with isothioureas. ACS Macro Lett. 2014, 3, 1024–1028. [Google Scholar] [CrossRef]
- Yao, L.; Wang, L.; Pan, X.; Tang, N.; Wu, J. Synthesis, characterization and catalytic activity of salen-(sodium)2 and (salen)2-lanthanum-sodium complexes. Inorg. Chim. Acta 2011, 373, 219–225. [Google Scholar] [CrossRef]
- Weil, J.; Mathers, R.T.; Getzler, Y.D.Y.L. Lactide cyclopolymerization by an alumatrane-inspired catalyst. Macromolecules 2012, 45, 1118–1121. [Google Scholar] [CrossRef]
- Piedra-Arroni, E.; Ladavière, C.; Amgoune, A.; Bourissou, D. Ring-opening polymerization with Zn(C6F5)2-based lewis pairs: Original and efficient approach to cyclic polyesters. J. Am. Chem. Soc. 2013, 135, 13306–13309. [Google Scholar] [CrossRef] [PubMed]
- Piromjitpong, P.; Ratanapanee, P.; Thumrongpatanaraks, W.; Kongsaeree, P.; Phomphrai, K. Synthesis of cyclic polylactide catalysed by bis(salicylaldiminato)tin(II) complexes. Dalton Trans. 2012, 41, 12704–12710. [Google Scholar] [CrossRef] [PubMed]
- Wongmahasirikun, P.; Prom-On, P.; Sangtrirutnugul, P.; Kongsaeree, P.; Phomphrai, K. Synthesis of cyclic polyesters: Effects of alkoxy side chains in salicylaldiminato tin(II) complexes. Dalton Trans. 2015, 44, 12357–12364. [Google Scholar] [CrossRef] [PubMed]
- Kricheldorf, H.R.; Weidner, S.M.; Scheliga, F. Cyclic poly(l-lactide)s via ring-expansion polymerizations catalysed by 2,2-dibutyl-2-stanna-1,3-dithiolane. Polym. Chem. 2017, 8, 1589–1596. [Google Scholar] [CrossRef]
- Farah, S.; Anderson, D.G.; Langer, R. Physical and mechanical properties of PLA, and their functions in widespread applications—A comprehensive review. Adv. Drug Deliv. Rev. 2016, 107, 367–392. [Google Scholar] [CrossRef] [PubMed]
- Martin, O.; Avérous, L. Poly(lactic acid): Plasticization and properties of biodegradable multiphase systems. Polymer 2001, 42, 6209–6219. [Google Scholar] [CrossRef]
- Mase, N.; Nakaya, N. Organocatalytic synthesis of highly pure linear- and cyclic-polylactide in supercritical carbon dioxide. In Proceedings of the Symposium on Molecular Chirality Asia 2016, Osaka, Japan, 22 April 2016. No. PB-33. [Google Scholar]
- Pratt, R.C.; Lohmeijer, B.G.G.; Long, D.A.; Lundberg, P.N.P.; Dove, A.P.; Li, H.; Wade, C.G.; Waymouth, R.M.; Hedrick, J.L. Exploration, optimization, and application of supramolecular thiourea-amine catalysts for the synthesis of lactide (co)polymers. Macromolecules 2006, 39, 7863–7871. [Google Scholar] [CrossRef]
- Wesch, A.; Dahmen, N.; Ebert, K.H. Measuring the static dielectric constants of pure carbon dioxide and carbon dioxide mixed with ethanol and toluene at elevated pressures. Ber. Bunsengesellsch. Phys. Chem. 1996, 100, 1368–1371. [Google Scholar] [CrossRef]
- Moriyoshi, T.; Kita, T.; Uosaki, Y. Static relative permittivity of carbon dioxide and nitrous oxide up to 30 MPa. Ber. Bunsengesellsch. Phys. Chem. 1993, 97, 589–596. [Google Scholar] [CrossRef]
- Tsuji, H. Poly(lactide) stereocomplexes: Formation, structure, properties, degradation, and applications. Macromol. Biosci. 2005, 5, 569–597. [Google Scholar] [CrossRef] [PubMed]
- Ikada, Y.; Jamshidi, K.; Tsuji, H.; Hyon, S.H. Stereocomplex formation between enantiomeric poly(lactides). Macromolecules 1987, 20, 904–906. [Google Scholar] [CrossRef]

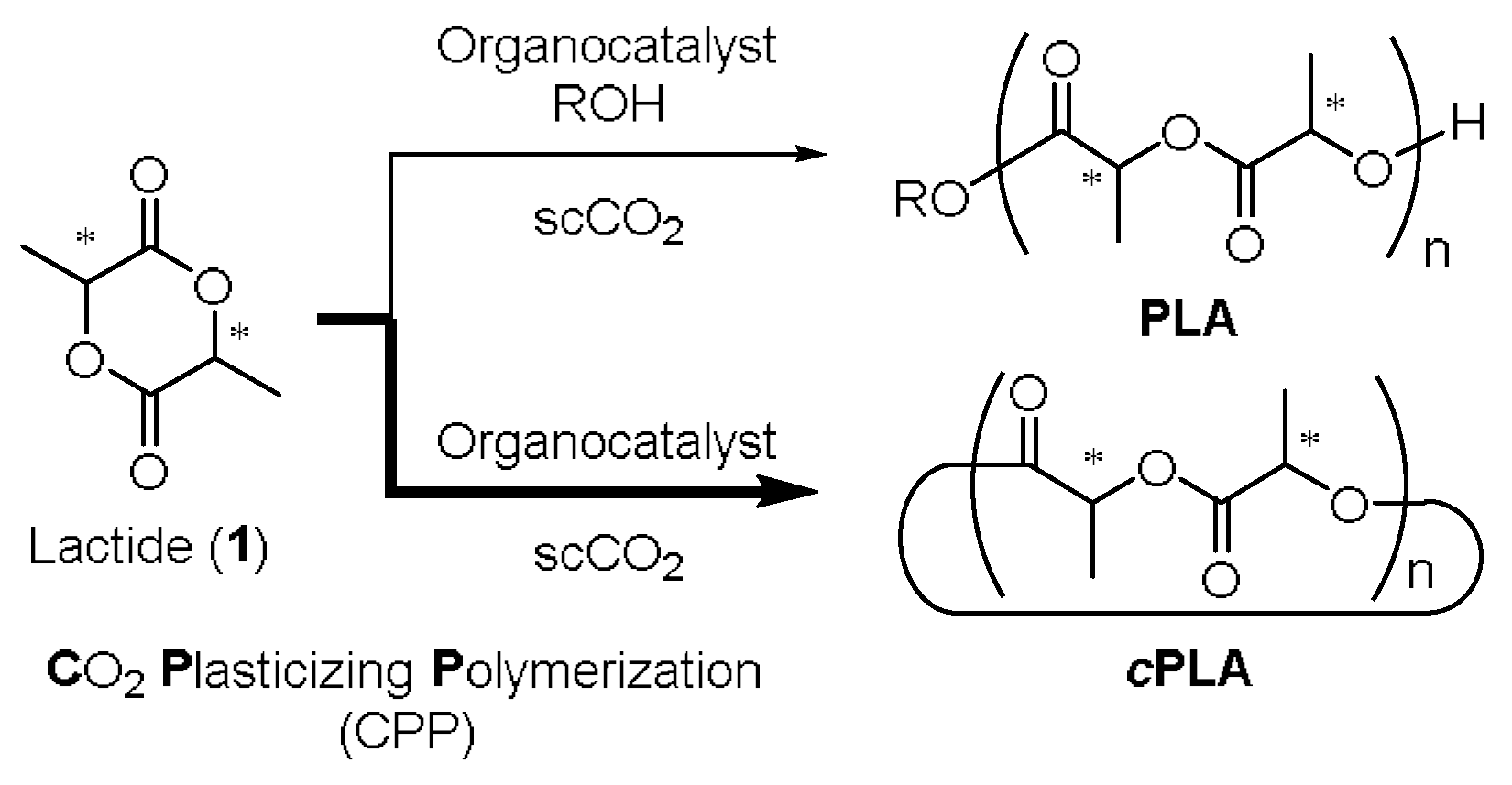
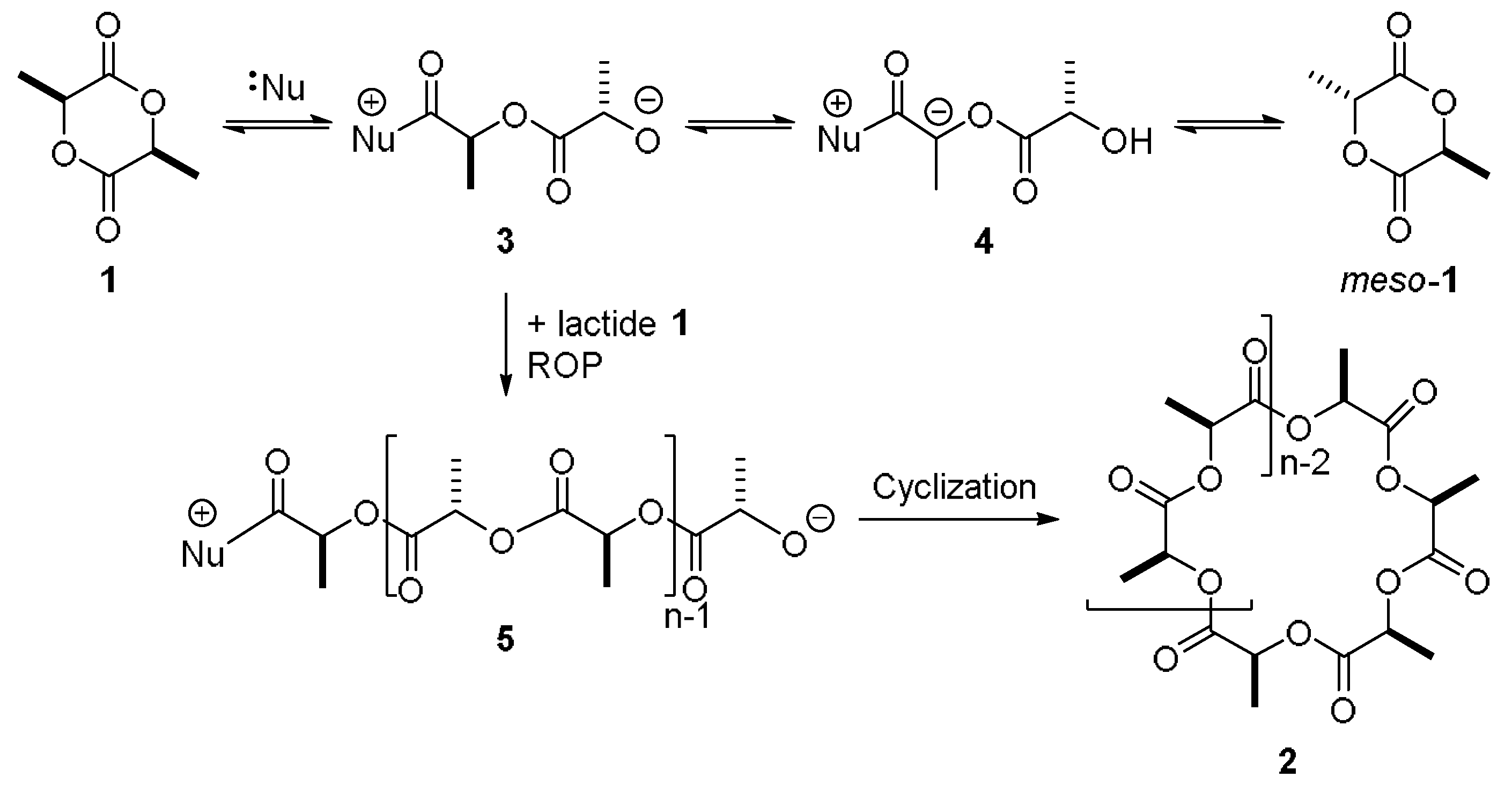

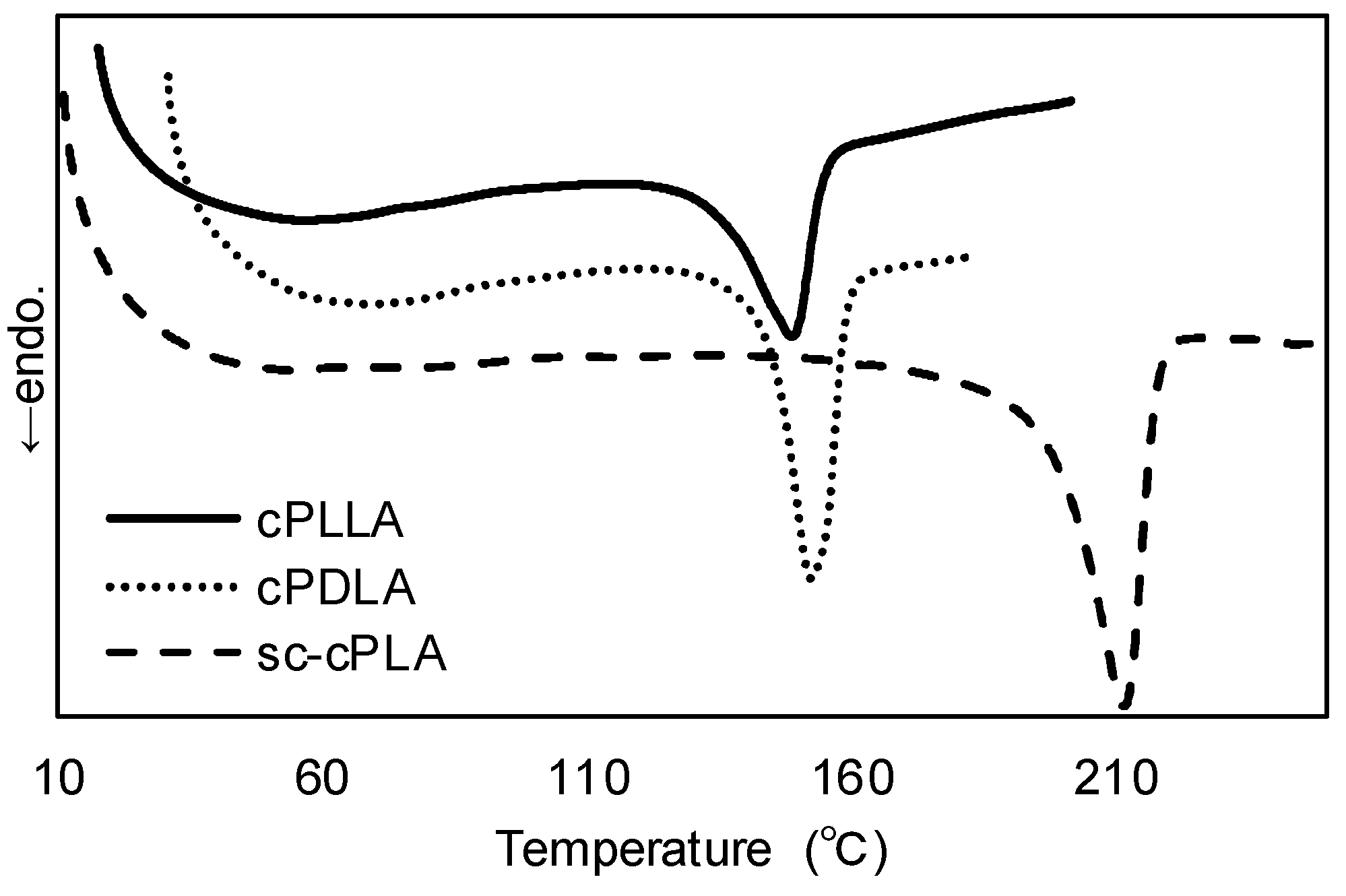


| Entry | Solvent | Time (h) | Mn (a) | PDI (b) | ee (%) (c) |
|---|---|---|---|---|---|
| 1 | CHCl3 | 120 | 12,700 | 3.11 | 39.0 |
| 2 | scCO2 | 5 | 5500 | 1.40 | 90.5 |
| 3 (d) | scCO2 | 2.5 | 11,000 | 1.60 | 93.5 |
| 4 (e) | scCO2 | 5 | 6000 | 1.31 | 91.0 |
| 5 (d,e) | scCO2 | 2.5 | 6800 | 1.20 | 97.0 |
| Entry | cPLA | Mn | PDI | Ee (%) | Tm (°C) | Tm (sc-cPLA) (°C) |
|---|---|---|---|---|---|---|
| 1 | cPDLA | 6800 | 1.20 | 97.0 | 152 | 212 |
| cPLLA | 11,000 | 1.60 | 93.5 | 149 | ||
| 2 | cPDLA | 6000 | 1.31 | 91.0 | 143 | 207 |
| cPLLA | 5500 | 1.40 | 90.5 | 145 | ||
| 3 (a) | cPDLA | 26,000 | 1.40 | (0.81) (b) | 132 | 179 |
| cPLLA | 30,000 | 1.30 | (0.83) (b) | 135 |
| Entry | cPLA | Mn | PDI | Tm (sc-cPLA) |
|---|---|---|---|---|
| 1 | cPDLA | 7900 | 1.18 | 190 |
| cPLLA | 2600 | 1.20 | ||
| 2 | cPDLA | 5100 | 1.23 | 200 |
| cPLLA | 7400 | 1.23 | ||
| 3 | cPDLA | 6000 | 1.31 | 207 |
| cPLLA | 5500 | 1.40 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mase, N.; Moniruzzaman; Yamamoto, S.; Nakaya, Y.; Sato, K.; Narumi, T. Organocatalytic Stereoselective Cyclic Polylactide Synthesis in Supercritical Carbon Dioxide under Plasticizing Conditions. Polymers 2018, 10, 713. https://doi.org/10.3390/polym10070713
Mase N, Moniruzzaman, Yamamoto S, Nakaya Y, Sato K, Narumi T. Organocatalytic Stereoselective Cyclic Polylactide Synthesis in Supercritical Carbon Dioxide under Plasticizing Conditions. Polymers. 2018; 10(7):713. https://doi.org/10.3390/polym10070713
Chicago/Turabian StyleMase, Nobuyuki, Moniruzzaman, Shoji Yamamoto, Yoshitaka Nakaya, Kohei Sato, and Tetsuo Narumi. 2018. "Organocatalytic Stereoselective Cyclic Polylactide Synthesis in Supercritical Carbon Dioxide under Plasticizing Conditions" Polymers 10, no. 7: 713. https://doi.org/10.3390/polym10070713
APA StyleMase, N., Moniruzzaman, Yamamoto, S., Nakaya, Y., Sato, K., & Narumi, T. (2018). Organocatalytic Stereoselective Cyclic Polylactide Synthesis in Supercritical Carbon Dioxide under Plasticizing Conditions. Polymers, 10(7), 713. https://doi.org/10.3390/polym10070713