Synthesis and Characterization of Renewable Polyester Coil Coatings from Biomass-Derived Isosorbide, FDCA, 1,5-Pentanediol, Succinic Acid, and 1,3-Propanediol
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
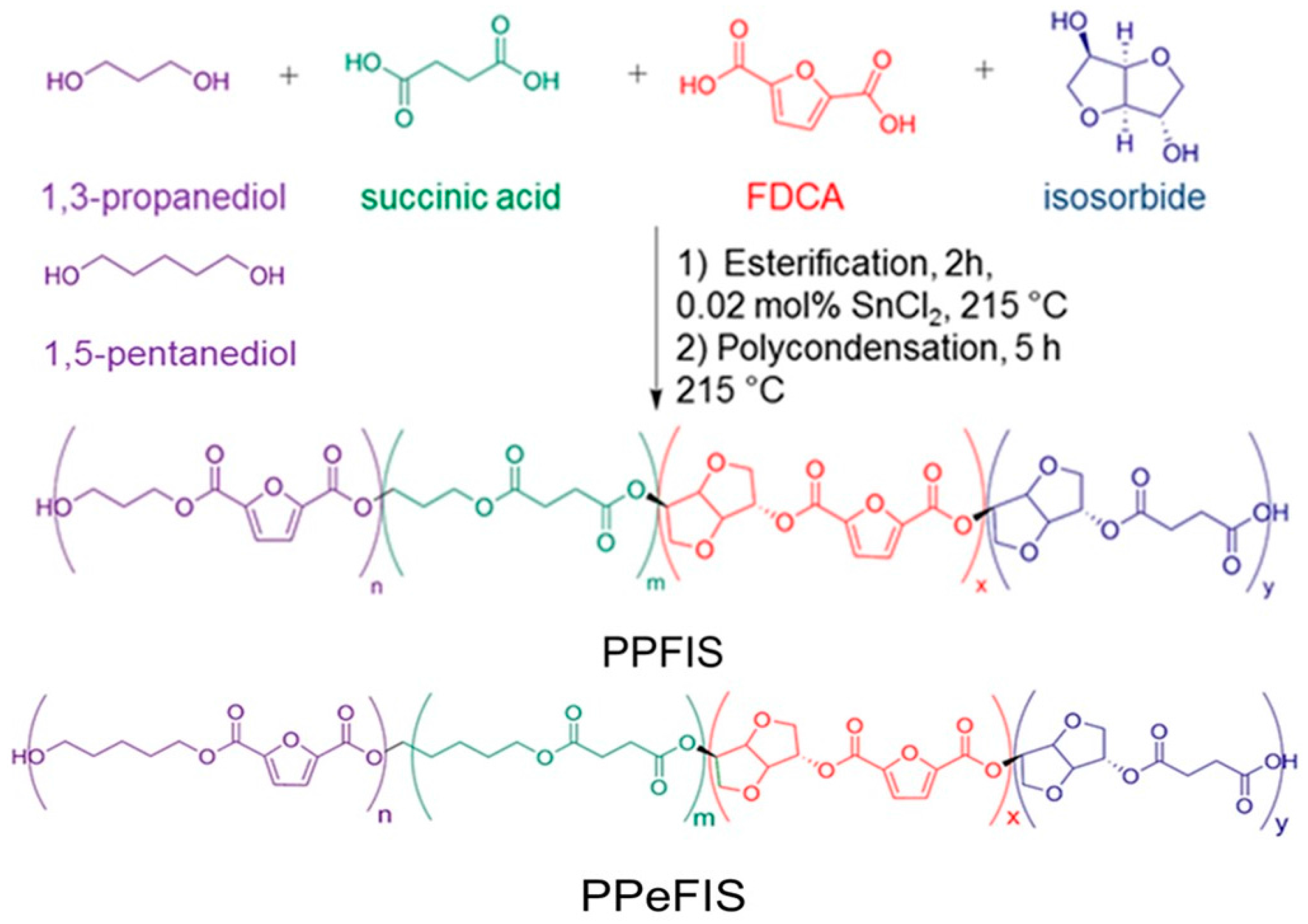
2.2. Synthesis of Isosorbide-Based Renewable Polyesters
2.3. Characterization Methods
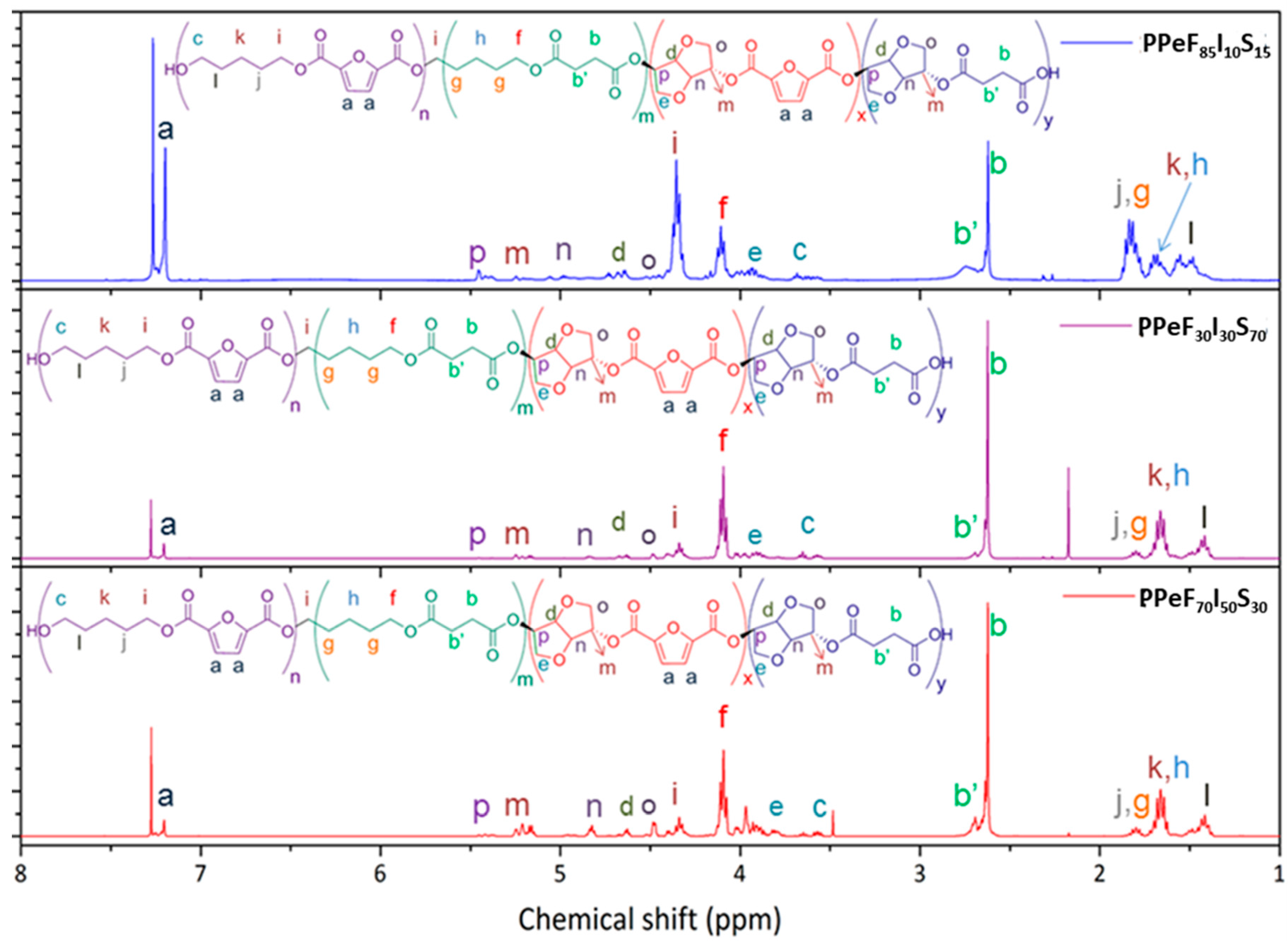
2.3.1. Nuclear Magnetic Resonance Spectroscopy (1H NMR)
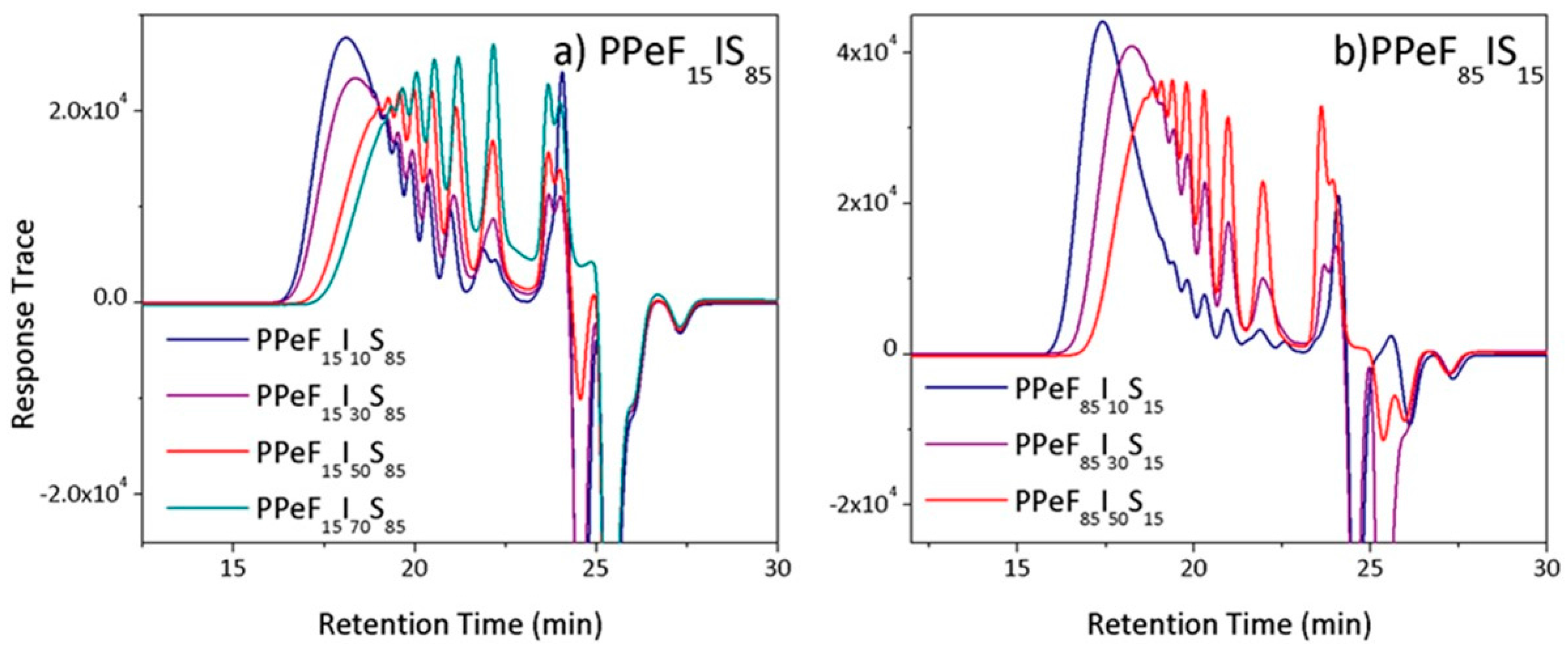
2.3.2. Gel Permeation Chromatography
2.3.3. Differential Scanning Calorimetry (DSC)
2.3.4. Thermal Gravimetric Analysis (TGA)
2.3.5. Coatings Characterization
3. Results and Discussion
3.1. 1H NMR
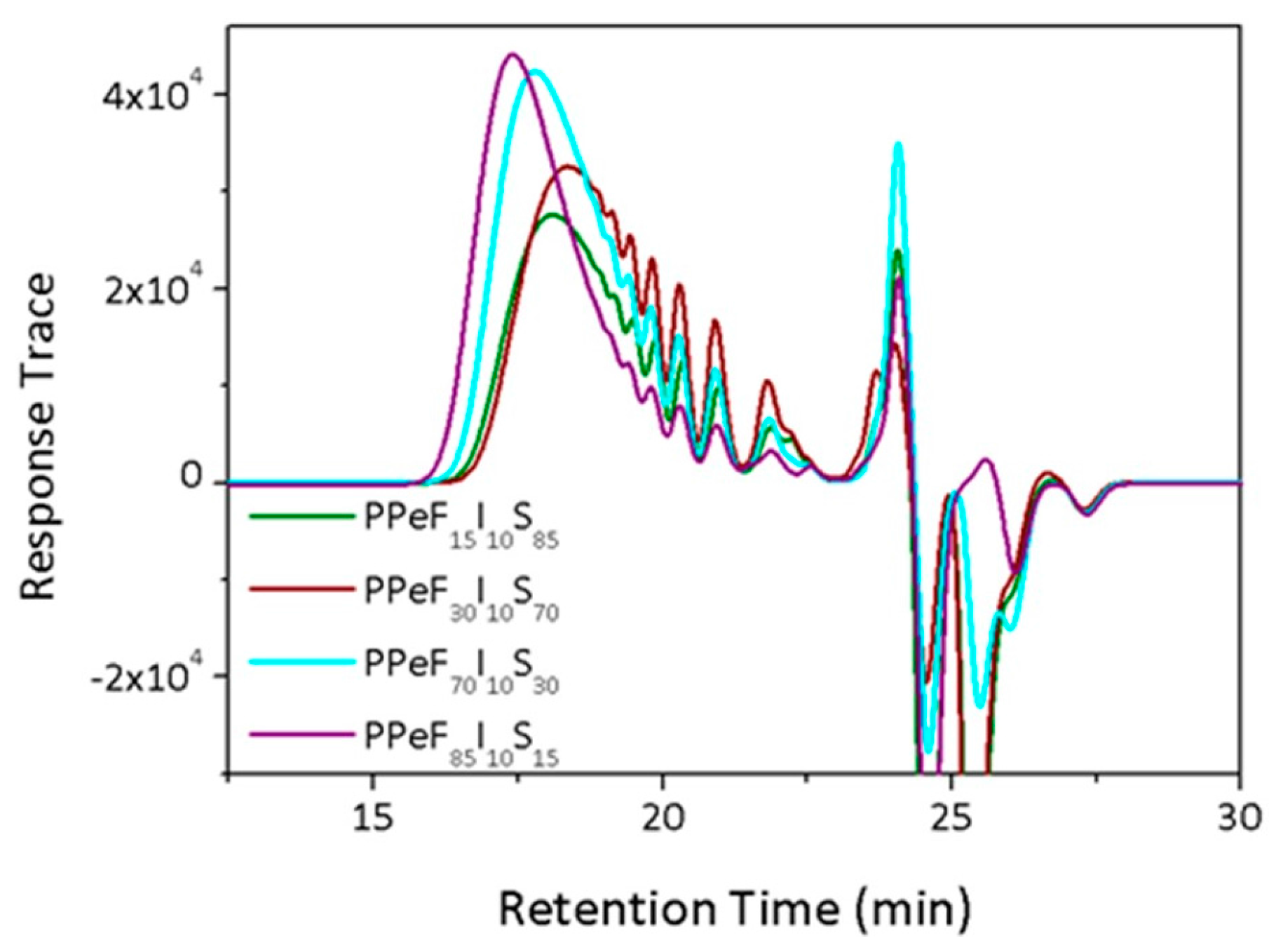
3.2. GPC
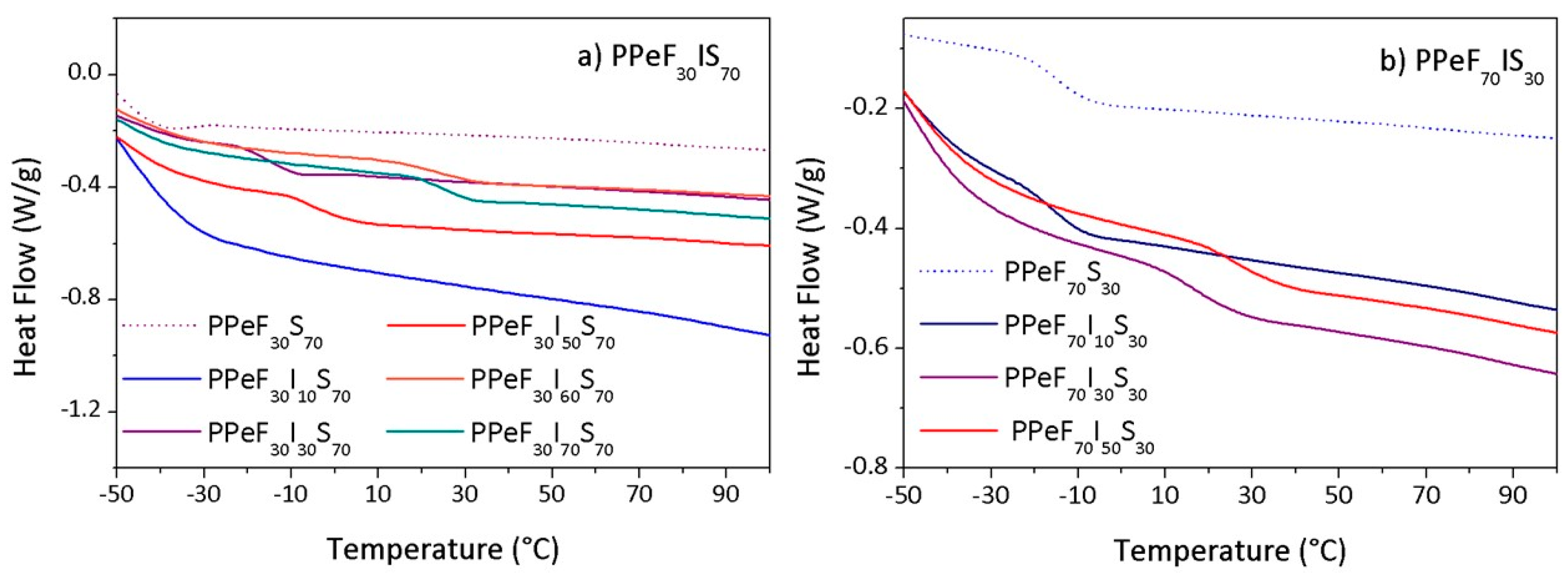
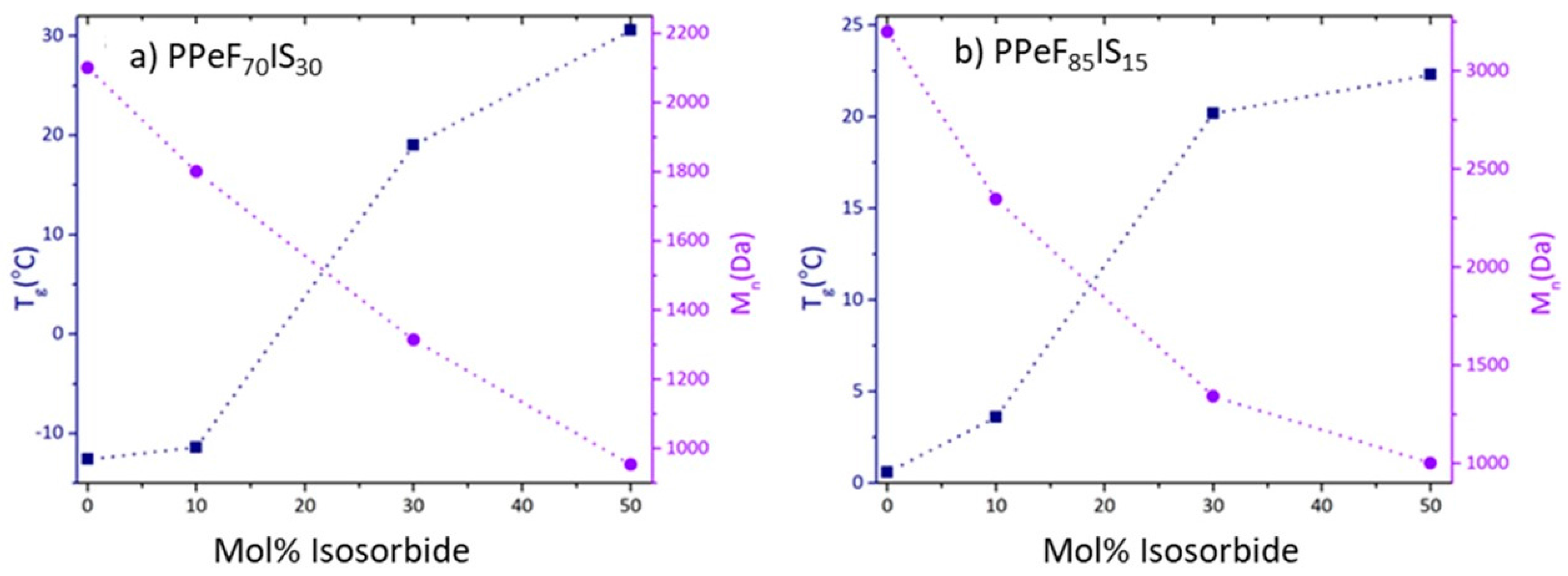
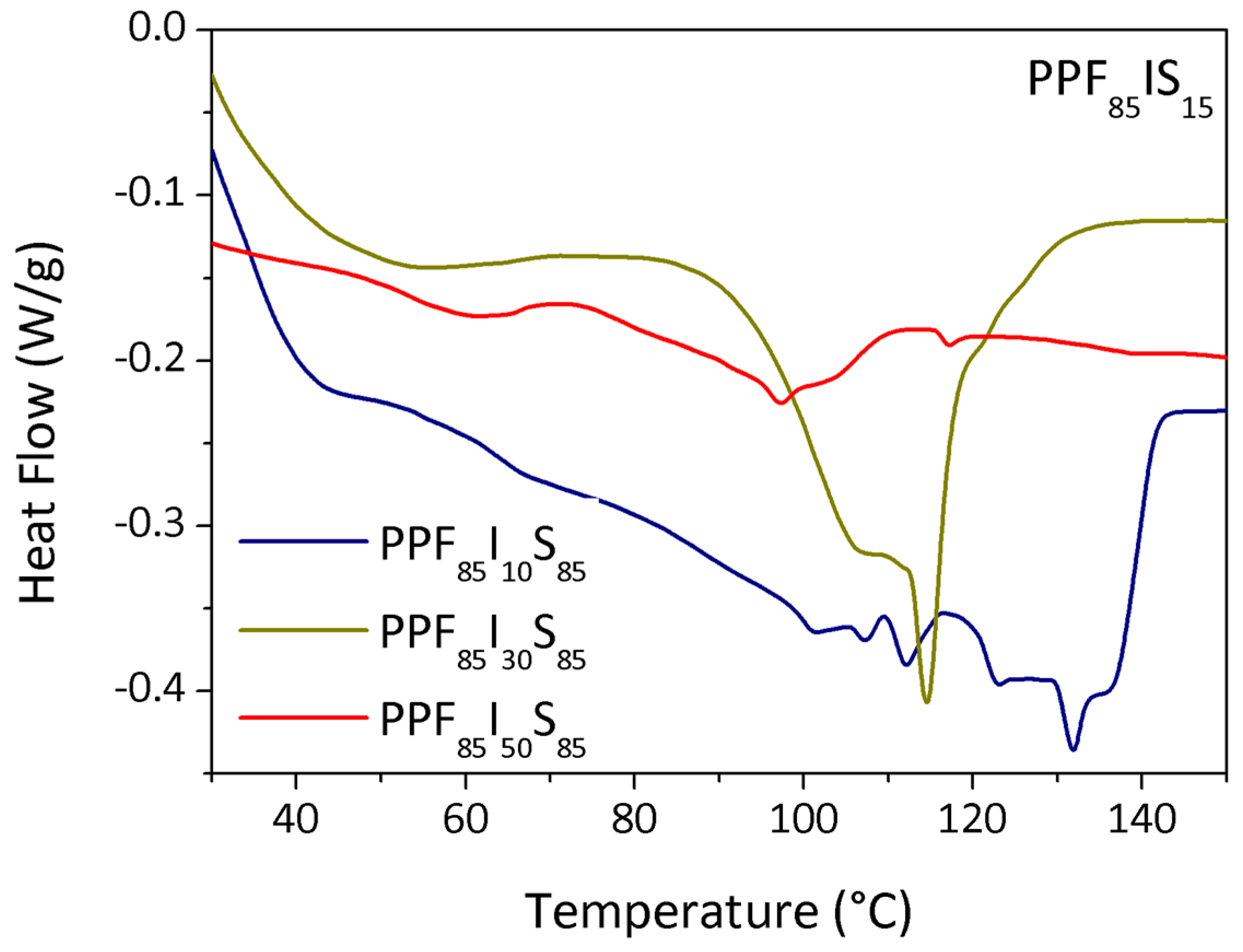
3.3. DSC
3.4. TGA
3.5. Coatings
4. Conclusions
Supplementary Materials
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Siyab, N.; Tenbusch, S.; Willis, S.; Lowe, C.; Maxted, J. Going Green: Making reality match ambition for sustainable coil coatings. J. Coat. Technol. Res. 2016, 13, 629–643. [Google Scholar] [CrossRef]
- Lomelí-Rodríguez, M.; Martín-Molina, M.; Jiménez-Pardo, M.; Nasim-Afzal, Z.; Cauët, S.I.; Davies, T.E.; Rivera-Toledo, M.; Lopez-Sanchez, J.A. Synthesis and kinetic modeling of biomass-derived renewable polyesters. J. Polym. Sci. Part A Polym. Chem. 2016, 54, 2876–2887. [Google Scholar] [CrossRef]
- Lomelí-Rodríguez, M.; Rivera-Toledo, M.; López-Sánchez, J.A. Optimum Batch-Reactor Operation for the Synthesis of Biomass-Derived Renewable Polyesters. Ind. Eng. Chem. Res. 2017, 56, 549–559. [Google Scholar] [CrossRef]
- Lomelí-Rodríguez, M.; Rivera-Toledo, M.; López-Sánchez, J.A. Process Intensification of the Synthesis of Biomass-Derived Renewable Polyesters: Reactive Distillation and Divided Wall Column Polyesterification. Ind. Eng. Chem. Res. 2017, 56, 3017–3032. [Google Scholar] [CrossRef]
- Gubbels, E.; Drijfhout, J.P.; Posthuma-van Tent, C.; Jasinska-Walc, L.; Noordover, B.A.J.; Koning, C.E. Bio-based semi-aromatic polyesters for coating applications. Prog. Organ. Coat. 2014, 77, 277–284. [Google Scholar] [CrossRef]
- Naves, A.F.; Fernandes, H.T.C.; Immich, A.P.S.; Catalani, L.H. Enzymatic syntheses of unsaturated polyesters based on isosorbide and isomannide. J. Polym. Sci. Part A Polym. Chem. 2013, 51, 3881–3891. [Google Scholar] [CrossRef]
- Feng, X.; East, A.J.; Hammond, W.B.; Zhang, Y.; Jaffe, M. Overview of advances in sugar-based polymers. Polym. Adv. Technol. 2011, 22, 139–150. [Google Scholar] [CrossRef]
- Fenouillot, F.; Rousseau, A.; Colomines, G.; Saint-Loup, R.; Pascault, J.-P. Polymers from renewable 1, 4: 3, 6-dianhydrohexitols (isosorbide, isomannide and isoidide): A review. Prog. Polym. Sci. 2010, 35, 578–622. [Google Scholar] [CrossRef]
- Rose, M.; Palkovits, R. Isosorbide as a renewable platform chemical for versatile applications—Quo Vadis? ChemSusChem 2012, 5, 167–176. [Google Scholar] [CrossRef] [PubMed]
- Flèche, G.; Huchette, M. Isosorbide. Preparation, Properties and Chemistry. Starch Stärke 1986, 38, 26–30. [Google Scholar] [CrossRef]
- Fertier, L.; Ibert, M.; Buffe, C.; Saint-Loup, R.; Joly-Duhamel, C.; Robin, J.J.; Giani, O. New biosourced UV curable coatings based on isosorbide. Prog. Organ. Coat. 2016, 99, 393–399. [Google Scholar] [CrossRef]
- Zhou, C.; Wei, Z.; Yu, Y.; Wang, Y.; Li, Y. Biobased copolyesters from renewable resources: Synthesis and crystallization kinetics of poly(propylene sebacate-co-isosorbide sebacate). RSC Adv. 2015, 5, 68688–68699. [Google Scholar] [CrossRef]
- Wang, G.; Jiang, M.; Zhang, Q.; Wang, R.; Zhou, G. Biobased copolyesters: Synthesis, crystallization behavior, thermal and mechanical properties of poly(ethylene glycol sebacate-co-ethylene glycol 2,5-furan dicarboxylate). RSC Adv. 2017, 7, 13798–13807. [Google Scholar] [CrossRef]
- Noordover, B.A.; Duchateau, R.; van Benthem, R.A.; Ming, W.; Koning, C.E. Enhancing the functionality of biobased polyester coating resins through modification with citric acid. Biomacromolecules 2007, 8, 3860–3870. [Google Scholar] [CrossRef] [PubMed]
- Kricheldorf, H.R.; Weidner, S.M. High T g copolyesters of lactide, isosorbide and isophthalic acid. Eur. Polym. J. 2013, 49, 2293–2302. [Google Scholar] [CrossRef]
- Garaleh, M.; Yashiro, T.; Kricheldorf, H.R.; Simon, P.; Chatti, S. (Co-) Polyesters Derived from Isosorbide and 1, 4-Cyclohexane Dicarboxylic Acid and Succinic Acid. Macromol. Chem. Phys. 2010, 211, 1206–1214. [Google Scholar] [CrossRef]
- Park, H.-S.; Gong, M.-S.; Knowles, J.C. Synthesis and biocompatibility properties of polyester containing various diacid based on isosorbide. J. Biomater. Appl. 2012, 27, 99–109. [Google Scholar] [CrossRef] [PubMed]
- Goerz, O.; Ritter, H. Polymers with shape memory effect from renewable resources: Crosslinking of polyesters based on isosorbide, itaconic acid and succinic acid. Polym. Int. 2013, 62, 709–712. [Google Scholar] [CrossRef]
- Noordover, B.A.; van Staalduinen, V.G.; Duchateau, R.; Koning, C.E.; van Benthem, R.A.; Mak, M.; Heise, A.; Frissen, A.E.; van Haveren, J. Co-and terpolyesters based on isosorbide and succinic acid for coating applications: Synthesis and characterization. Biomacromolecules 2006, 7, 3406–3416. [Google Scholar] [CrossRef] [PubMed]
- Wei, Z.; Zhou, C.; Yu, Y.; Li, Y. Biobased copolyesters from renewable resources: Synthesis and crystallization behavior of poly (decamethylene sebacate-co-isosorbide sebacate). RSC Adv. 2015, 5, 42777–42788. [Google Scholar] [CrossRef]
- Kasmi, N.; Majdoub, M.; Papageorgiou, G.Z.; Bikiaris, D.N. Synthesis and crystallization of new fully renewable resources-based copolyesters: Poly(1,4-cyclohexanedimethanol-co-isosorbide 2,5-furandicarboxylate). Polym. Degrad. Stab. 2018, 152, 177–190. [Google Scholar] [CrossRef]
- Terzopoulou, Z.; Kasmi, N.; Tsanaktsis, V.; Doulakas, N.; Bikiaris, D.N.; Achilias, D.S.; Papageorgiou, G.Z. Synthesis and Characterization of Bio-Based Polyesters: Poly(2-methyl-1,3-propylene-2,5-furanoate), Poly(isosorbide-2,5-furanoate), Poly(1,4-cyclohexanedimethylene-2,5-furanoate). Materials 2017, 10, 801. [Google Scholar] [CrossRef] [PubMed]
- Van Haveren, J.; Oostveen, E.A.; Miccichè, F.; Noordover, B.A.J.; Koning, C.E.; van Benthem, R.A.T.M.; Frissen, A.E.; Weijnen, J.G.J. Resins and additives for powder coatings and alkyd paints, based on renewable resources. J. Coat. Technol. Res. 2007, 4, 177–186. [Google Scholar] [CrossRef]
- Gioia, C.; Vannini, M.; Marchese, P.; Minesso, A.; Cavalieri, R.; Colonna, M.; Celli, A. Sustainable polyesters for powder coating applications from recycled PET, isosorbide and succinic acid. Green Chem. 2014, 16, 1807–1815. [Google Scholar] [CrossRef]
- Jacquel, N.; Saint-Loup, R.; Pascault, J.-P.; Rousseau, A.; Fenouillot, F. Bio-based alternatives in the synthesis of aliphatic–aromatic polyesters dedicated to biodegradable film applications. Polymer 2015, 59, 234–242. [Google Scholar] [CrossRef]
- Bersot, J.C.; Jacquel, N.; Saint-Loup, R.; Fuertes, P.; Rousseau, A.; Pascault, J.P.; Spitz, R.; Fenouillot, F.; Monteil, V. Efficiency Increase of Poly (ethylene terephthalate-co-isosorbide terephthalate) Synthesis using Bimetallic Catalytic Systems. Macromol. Chem. Phys. 2011, 212, 2114–2120. [Google Scholar] [CrossRef]
- Gandini, A.; Silvestre, A.J.; Neto, C.P.; Sousa, A.F.; Gomes, M. The furan counterpart of poly (ethylene terephthalate): An alternative material based on renewable resources. J. Polym. Sci. Part A Polym. Chem. 2009, 47, 295–298. [Google Scholar] [CrossRef]
- Jiang, M.; Liu, Q.; Zhang, Q.; Ye, C.; Zhou, G. A series of furan-aromatic polyesters synthesized via direct esterification method based on renewable resources. J. Polym. Sci. Part A Polym. Chem. 2012, 50, 1026–1036. [Google Scholar] [CrossRef]
- Papageorgiou, G.Z.; Tsanaktsis, V.; Papageorgiou, D.G.; Exarhopoulos, S.; Papageorgiou, M.; Bikiaris, D.N. Evaluation of polyesters from renewable resources as alternatives to the current fossil-based polymers. Phase transitions of poly (butylene 2, 5-furan-dicarboxylate). Polymer 2014, 55, 3846–3858. [Google Scholar] [CrossRef]
- Sousa, A.; Fonseca, A.; Serra, A.; Freire, C.; Silvestre, A.; Coelho, J. New unsaturated copolyesters based on 2, 5-furandicarboxylic acid and their crosslinked derivatives. Polym. Chem. 2016, 7, 1049–1058. [Google Scholar] [CrossRef]
- Vannini, M.; Marchese, P.; Celli, A.; Lorenzetti, C. Fully biobased poly (propylene 2, 5-furandicarboxylate) for packaging applications: Excellent barrier properties as a function of crystallinity. Green Chem. 2015, 17, 4162–4166. [Google Scholar] [CrossRef]
- Tsanaktsis, V.; Terzopoulou, Z.; Nerantzaki, M.; Papageorgiou, G.Z.; Bikiaris, D.N. New poly (pentylene furanoate) and poly (heptylene furanoate) sustainable polyesters from diols with odd methylene groups. Mater. Lett. 2016, 178, 64–67. [Google Scholar] [CrossRef]
- Terzopoulou, Z.; Tsanaktsis, V.; Bikiaris, D.N.; Exarhopoulos, S.; Papageorgiou, D.G.; Papageorgiou, G.Z. Biobased poly (ethylene furanoate-co-ethylene succinate) copolyesters: Solid state structure, melting point depression and biodegradability. RSC Adv. 2016, 6, 84003–84015. [Google Scholar] [CrossRef]
- Burgess, S.K.; Leisen, J.E.; Kraftschik, B.E.; Mubarak, C.R.; Kriegel, R.M.; Koros, W.J. Chain mobility, thermal, and mechanical properties of poly (ethylene furanoate) compared to poly (ethylene terephthalate). Macromolecules 2014, 47, 1383–1391. [Google Scholar] [CrossRef]
- Terzopoulou, Z.; Karakatsianopoulou, E.; Kasmi, N.; Tsanaktsis, V.; Nikolaidis, N.; Kostoglou, M.; Papageorgiou, G.Z.; Lambropoulou, D.A.; Bikiaris, D.N. Effect of catalyst type on molecular weight increase and coloration of poly(ethylene furanoate) biobased polyester during melt polycondensation. Polym. Chem. 2017, 8, 6895–6908. [Google Scholar] [CrossRef]
- Wu, L.; Mincheva, R.; Xu, Y.; Raquez, J.-M.; Dubois, P. High molecular weight poly (butylene succinate-co-butylene furandicarboxylate) copolyesters: From catalyzed polycondensation reaction to thermomechanical properties. Biomacromolecules 2012, 13, 2973–2981. [Google Scholar] [CrossRef] [PubMed]
- Hbaieb, S.; Kammoun, W.; Delaite, C.; Abid, M.; Abid, S.; El Gharbi, R. New copolyesters containing aliphatic and bio-based furanic units by bulk copolycondensation. J. Macromol. Sci. Part A 2015, 52, 365–373. [Google Scholar] [CrossRef]
- Yu, Z.; Zhou, J.; Cao, F.; Wen, B.; Zhu, X.; Wei, P. Chemosynthesis and characterization of fully biomass-based copolymers of ethylene glycol, 2, 5-furandicarboxylic acid, and succinic acid. J. Appl. Polym. Sci. 2013, 130, 1415–1420. [Google Scholar] [CrossRef]
- Papageorgiou, G.Z.; Papageorgiou, D.G.; Tsanaktsis, V.; Bikiaris, D.N. Synthesis of the bio-based polyester poly (propylene 2, 5-furan dicarboxylate). Comparison of thermal behavior and solid state structure with its terephthalate and naphthalate homologues. Polymer 2015, 62, 28–38. [Google Scholar] [CrossRef]
- Matos, M.; Sousa, A.F.; Fonseca, A.C.; Freire, C.S.; Coelho, J.F.; Silvestre, A.J. A New Generation of Furanic Copolyesters with Enhanced Degradability: Poly (ethylene 2, 5-furandicarboxylate)-co-poly (lactic acid) Copolyesters. Macromol. Chem. Phys. 2014, 215, 2175–2184. [Google Scholar] [CrossRef]
- Storbeck, R.; Ballauff, M. Synthesis and properties of polyesters based on 2, 5-furandicarboxylic acid and 1, 4: 3, 6-dianhydrohexitols. Polymer 1993, 34, 5003–5006. [Google Scholar] [CrossRef]
- Wu, J.; Eduard, P.; Thiyagarajan, S.; Noordover, B.A.; van Es, D.S.; Koning, C.E. Semi-Aromatic Polyesters Based on a Carbohydrate-Derived Rigid Diol for Engineering Plastics. ChemSusChem 2015, 8, 67–72. [Google Scholar] [CrossRef] [PubMed]
- Terzopoulou, Z.; Tsanaktsis, V.; Nerantzaki, M.; Papageorgiou, G.Z.; Bikiaris, D.N. Decomposition mechanism of polyesters based on 2,5-furandicarboxylic acid and aliphatic diols with medium and long chain methylene groups. Polym. Degrad. Stab. 2016, 132, 127–136. [Google Scholar] [CrossRef]
- Papageorgiou, G.Z.; Guigo, N.; Tsanaktsis, V.; Papageorgiou, D.G.; Exarhopoulos, S.; Sbirrazzuoli, N.; Bikiaris, D.N. On the bio-based furanic polyesters: Synthesis and thermal behavior study of Poly (octylene furanoate) using Fast and Temperature Modulated Scanning Calorimetry. Eur. Polym. J. 2015, 68, 115–127. [Google Scholar] [CrossRef]
- Tsanaktsis, V.; Vouvoudi, E.; Papageorgiou, G.Z.; Papageorgiou, D.G.; Chrissafis, K.; Bikiaris, D.N. Thermal degradation kinetics and decomposition mechanism of polyesters based on 2, 5-furandicarboxylic acid and low molecular weight aliphatic diols. J. Anal. Appl. Pyrolysis 2015, 112, 369–378. [Google Scholar] [CrossRef]
- Terzopoulou, Z.; Tsanaktsis, V.; Nerantzaki, M.; Achilias, D.S.; Vaimakis, T.; Papageorgiou, G.Z.; Bikiaris, D.N. Thermal degradation of biobased polyesters: Kinetics and decomposition mechanism of polyesters from 2, 5-furandicarboxylic acid and long-chain aliphatic diols. J. Anal. Appl. Pyrolysis 2016, 117, 162–175. [Google Scholar] [CrossRef]
- Papageorgiou, G.Z.; Papageorgiou, D.G.; Terzopoulou, Z.; Bikiaris, D.N. Production of bio-based 2, 5-furan dicarboxylate polyesters: Recent progress and critical aspects in their synthesis and thermal properties. Eur. Polym. J. 2016, 83, 202–229. [Google Scholar] [CrossRef]
- Oldring, P.K.; Tuck, N. Resins for Surface Coatings, Alkyds & Polyesters; John Wiley & Sons: Hoboken, NJ, USA, 2000; Volume 2. [Google Scholar]
- Brentzel, Z.J.; Barnett, K.J.; Huang, K.; Maravelias, C.T.; Dumesic, J.A.; Huber, G.W. Chemicals from Biomass: Combining Ring-Opening Tautomerization and Hydrogenation Reactions to Produce 1,5-Pentanediol from Furfural. ChemSusChem 2017, 10, 1351–1355. [Google Scholar] [CrossRef] [PubMed]
- Sun, D.; Sato, S.; Ueda, W.; Primo, A.; Garcia, H.; Corma, A. Production of C4 and C5 alcohols from biomass-derived materials. Green Chem. 2016, 18, 2579–2597. [Google Scholar] [CrossRef]
- Huang, K.; Brentzel, Z.J.; Barnett, K.J.; Dumesic, J.A.; Huber, G.W.; Maravelias, C.T. Conversion of Furfural to 1,5-Pentanediol: Process Synthesis and Analysis. ACS Sust. Chem. Eng. 2017, 5, 4699–4706. [Google Scholar] [CrossRef]
- Chatterjee, M.; Ishizaka, T.; Kawanami, H. Hydrogenation of 5-hydroxymethylfurfural in supercritical carbon dioxide-water: A tunable approach to dimethylfuran selectivity. Green Chem. 2014, 16, 1543–1551. [Google Scholar] [CrossRef]
- Koso, S.; Furikado, I.; Shimao, A.; Miyazawa, T.; Kunimori, K.; Tomishige, K. Chemoselective hydrogenolysis of tetrahydrofurfuryl alcohol to 1, 5-pentanediol. Chem. Commun. 2009, 2035–2037. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Amada, Y.; Tamura, M.; Nakagawa, Y.; Tomishige, K. One-pot selective conversion of furfural into 1, 5-pentanediol over a Pd-added Ir–ReO x/SiO 2 bifunctional catalyst. Green Chem. 2014, 16, 617–626. [Google Scholar] [CrossRef]
- Standard Test Methods for Indentation Hardness of Organic Coatings. Available online: https://compass.astm.org/download/D1474D1474M.4895.pdf (accessed on 15 February 2017).
- Coil Coated Metals. Test Methods. Pencil Hardness. Available online: http://shop.bsigroup.com/ProductDetail/?pid=000000000030268977 (accessed on 14 February 2017).
- Standard Test Method for Specular Gloss. Available online: https://compass.astm.org/Standards/HISTORICAL/D523-89R99.htm (accessed on 14 February 2017).
- Standard Practice for Assessing the Solvent Resistance of Organic Coatings Using Solvent Rubs. Available online: https://compass.astm.org/download/D5402-93R99.15287.pdf (accessed on 15 February 2015).
- ISO 14577-1:2015 Metallic Materials—Instrumented Indentation Test for Hardness and Materials parameters—Part 1: Test Method. Available online: https://www.iso.org/obp/ui/#iso:std:iso:14577:-1:ed-2:v1:en (accessed on 15 February 2017).
- Standard Test Method for Assignment of the Glass Transition Temperatures by Differential Scanning Calorimetry or Differential Thermal Analysis. Available online: https://compass.astm.org/download/E1356-98.33431.pdf (accessed on 15 February 2017).
- Standard Test Method for Coating Flexibility of Prepainted Sheet. Available online: https://compass.astm.org/download/D4145.28079.pdf (accessed on 15 February 2017).
- Allcock, H.R.; Lampe, F.W.; Mark, J.E.; Allcock, H. Contemporary Polymer Chemistry; Pearson/Prentice Hall: Upper Saddle River, NJ, USA, 2003. [Google Scholar]
- Sadler, J.M.; Toulan, F.R.; Palmese, G.R.; La Scala, J.J. Unsaturated polyester resins for thermoset applications using renewable isosorbide as a component for property improvement. J. Appl. Polym. Sci. 2015, 132, 42315. [Google Scholar] [CrossRef]
- Koo, J.M.; Hwang, S.Y.; Yoon, W.J.; Lee, Y.G.; Kim, S.H.; Im, S.S. Structural and thermal properties of poly(1,4-cyclohexane dimethylene terephthalate) containing isosorbide. Polym. Chem. 2015, 6, 6973–6986. [Google Scholar] [CrossRef]
- Łukaszczyk, J.; Janicki, B.; Kaczmarek, M. Synthesis and properties of isosorbide based epoxy resin. Eur. Polym. J. 2011, 47, 1601–1606. [Google Scholar] [CrossRef]
- Zhu, Y.; Molinier, V.; Durand, M.; Lavergne, A.; Aubry, J.-M. Amphiphilic Properties of Hydrotropes Derived from Isosorbide: Endo/Exo Isomeric Effects and Temperature Dependence. Langmuir 2009, 25, 13419–13425. [Google Scholar] [CrossRef] [PubMed]
- Papageorgiou, G.Z.; Achilias, D.S.; Bikiaris, D.N. Crystallization Kinetics and Melting Behaviour of the Novel Biodegradable Polyesters Poly(propylene azelate) and Poly(propylene sebacate). Macromol. Chem. Phys. 2009, 210, 90–107. [Google Scholar] [CrossRef]
- Papageorgiou, G.Z.; Bikiaris, D.N. Crystallization and melting behavior of three biodegradable poly(alkylene succinates). A comparative study. Polymer 2005, 46, 12081–12092. [Google Scholar] [CrossRef]
- Wang, X.; Zhou, J.; Li, L. Multiple melting behavior of poly(butylene succinate). Eur. Polym. J. 2007, 43, 3163–3170. [Google Scholar] [CrossRef]
- Gunaratne, L.M.W.K.; Shanks, R.A. Multiple melting behaviour of poly(3-hydroxybutyrate-co-hydroxyvalerate) using step-scan DSC. Eur. Polym. J. 2005, 41, 2980–2988. [Google Scholar] [CrossRef]
- Song, P.; Chen, G.; Wei, Z.; Zhang, W.; Liang, J. Calorimetric analysis of the multiple melting behavior of melt-crystallized poly(l-lactic acid) with a low optical purity. J. Therm. Anal. Calorim. 2013, 111, 1507–1514. [Google Scholar] [CrossRef]
- Chatti, S.; Weidner, S.M.; Fildier, A.; Kricheldorf, H.R. Copolyesters of isosorbide, succinic acid, and isophthalic acid: Biodegradable, high Tg engineering plastics. J. Polym. Sci. Part A Polym. Chem. 2013, 51, 2464–2471. [Google Scholar] [CrossRef]
- Beyler, C.L.; Hirschler, M.M. Thermal decomposition of polymers. In SFPE Handbook of Fire Protection Engineering; Springer: Berlin, Germany, 2002; p. 32. [Google Scholar]
- Montaudo, G.; Puglisi, C.; Samperi, F. Primary thermal degradation mechanisms of PET and PBT. Polym. Degrad. Stab. 1993, 42, 13–28. [Google Scholar] [CrossRef]
- Goldfarb, I.J.; McGuchan, R. Thermal Degradation of Polyesters. 1. Aliphatic Polymers; DTIC Document; DTIC: Fort Belvoir, VA, USA, 1968. [Google Scholar]

| Polymer a | Temperature, °C | Mol% FDCA | Mol% Isosorbide | R b | Mn, Da | Mw, Da | Đ |
|---|---|---|---|---|---|---|---|
| PPF15I30S85 | 30 | 700 | 1100 | 1.6 | |||
| PPF15I60S85 | 215 | 15 | 60 | 1.5 | 650 | 1000 | 1.5 |
| PPF15I70S85 | 70 | 600 | 1000 | 1.5 | |||
| PPF30I30S70 | 30 | 900 | 1500 | 1.7 | |||
| PPF30I60S70 | 215 | 30 | 60 | 1.5 | 600 | 1000 | 1.6 |
| PPF30I70S70 | 70 | 500 | 700 | 1.3 | |||
| PPF70I10S30 | 10 | - c | |||||
| PPF70I30S30 | 215 | 70 | 30 | 1.5 | - c | ||
| PPF70I50S30 | 50 | - | |||||
| PPF85I10S15 | 10 | - c | |||||
| PPF85I30S15 | 215 | 85 | 30 | 1.5 | - | ||
| PPF85I30S15 | 50 | - |
| Polymer a | Temperature, C | mol % FDCA | mol % Isosorbide | r b | Mn, Da | Mw, Da | Đ |
|---|---|---|---|---|---|---|---|
| PPeF15I10S85 | 10 | 1500 | 3100 | 2.1 | |||
| PPeF15I30S85 | 30 | 1200 | 2500 | 2.2 | |||
| PPeF15I50S85 | 215 | 15 | 50 | 1.5 | 800 | 1500 | 1.8 |
| PPeF15I60S85 | 60 | 500 | 1100 | 2.1 | |||
| PPeF15I70S85 | 70 | 700 | 1100 | 1.7 | |||
| PPeF30I10S70 | 10 | 1300 | 2700 | 2.0 | |||
| PPeF30I30S70 | 30 | 1100 | 2800 | 2.5 | |||
| PPeF30I50S70 | 215 | 30 | 50 | 1.5 | 900 | 1600 | 1.9 |
| PPeF30I60S70 | 60 | 500 | 700 | 1.5 | |||
| PPeF30I70S70 | 70 | 600 | 1000 | 1.6 | |||
| PPeF70I10S30 | 10 | 1800 | 3800 | 2.1 | |||
| PPeF70I30S30 | 215 | 70 | 30 | 1.5 | 1300 | 3200 | 2.4 |
| PPeF70I50S30 | 50 | 1000 | 1900 | 2.0 | |||
| PPeF85I10S15 | 10 | 2300 | 5400 | 2.3 | |||
| PPeF85I30S15 | 215 | 85 | 30 | 1.5 | 1300 | 2800 | 2.1 |
| PPeF85I30S15 | 50 | 1000 | 1800 | 1.8 |
| Polyester | Assignment of Chemical Shifts (CDCl3, δ/ppm) | ||||||
|---|---|---|---|---|---|---|---|
| PPeFIS | a | b,b’ | c | d | e | f | g,j |
| 7.20 | 2.62, 2.69 | 3.68 | 4.62–4.67 | 3.80–3.97 | 4.09 | 1.80 | |
| h,k | i | l | m | n | o | p | |
| 1.66 | 4.34 | 1.42–1.48 | 5.21–5.25 | 4.84–4.97 | 4.48 | 5.40–5.46 | |
| Integrations for PPeF85I10S15 | |||||||
| a | b,b’ | c | d | e | f | g,j | |
| 2.03 | 2.00, 1.13 | 0.47 | 0.53 | 1.17 | 2.24 | 4.05 | |
| h,k | i | l | m | n | o | p | |
| 1.97 | 4.57 | 2.95 | 0.14 | 0.21 | 0.21 | 0.47 | |
| Polyester | Mol% Isosorbide | Mw, Da | Tg, °C | Tm, °C | ΔHm, J/g |
|---|---|---|---|---|---|
| PPeF15S85 | 0 | 2700 | −46 | - | - |
| PPeF15I10S85 | 10 | 3100 | −43 | - | - |
| PPeF15I30S85 | 30 | 2500 | −26 | - | - |
| PPeF15I50S85 | 50 | 1500 | −12 | - | - |
| PPeF15I60S85 | 60 | 1100 | 7 | - | - |
| PPeF15I70S85 | 70 | 1100 | 35 | 117 | 2.6 |
| PPeF30S70 | 0 | 2800 | −39 | - | - |
| PPeF30I10S70 | 10 | 2600 | −39 | - | - |
| PPeF30I30S70 | 30 | 2800 | −12 | 155 | 3.5 |
| PPeF30I50S70 | 50 | 1600 | 0.7 | 139 | 1.3 |
| PPeF30I60S70 | 60 | 700 | 23 | 105 | 0.1 |
| PPeF30I70S70 | 70 | 1000 | 24 | 77 | - |
| PPeF70S30 | 0 | 5900 | −10 | - | - |
| PPeF70I10S30 | 10 | 3800 | −11 | - | - |
| PPeF70I30S30 | 30 | 3200 | 19 | 94 | 0.4 |
| PPeF70I50S30 | 50 | 1900 | 31 | 105 | 1.2 |
| PPeF85S15 | 0 | 4100 | 1 | 50 | - |
| PPeF85I10S15 | 10 | 5400 | 4 | 119 | 0.1 |
| PPeF85I30S15 | 30 | 2800 | 20 | 101 | 1.0 |
| PPeF85I50S15 | 50 | 1800 | 22 | 139 | 1.4 |
| Test | Standard | R | PPeF15I70S85 | PPeF30I70S70 | PPeF70I30S30 | PPeF70I50S30 |
|---|---|---|---|---|---|---|
| Pencil hardness | EN13523-4 | H | 2H | 3H | H | H |
| Gloss top coat | 13523-2 | 35 | 39 | 35 | 40 | 39 |
| Reverse impact 80” lb | 13523-5 | Moderate cracking | Moderate cracking | Moderate cracking | Slight cracking | Moderate cracking |
| Erichsen 7.5 mm | 13523-6 | Moderate cracking | Severe cracking | Severe cracking | Slight cracking | No cracking |
| T-bend (no tape pick off) | 13523-7 | 0.5T | 1T | 1.5T | 0T | 1T |
| T-Bend no cracking | 13523-7 | 3T | 5T | 6T | 2.5T | 5T |
| MEKa rubs primer | BSSP 3.522.11 | 110 | 110 | 110 | 110 | 110 |
| Tg, °C (onset/midpoint) | 28/35 | 53/66 | 51/53 | 27/34 | 53/67 | |
| Microhardness, N∙m−2 | 216 | 237 | 287 | 190 | 270 |
| Test | Standard | R | PPeF85I10S15 | PPeF85I30S15 | PPeF85I50S15 | PPF85I30S15 |
|---|---|---|---|---|---|---|
| Pencil hardness | EN13523-4 | H | H | F | 2H | H |
| Gloss top coat | 13523-2 | 35 | 40 | 43 | 38 | 39 |
| Reverse impact 80” lb | 13523-5 | Moderate cracking | No cracking | Moderate cracking | Severe cracking | Slight cracking |
| Erichsen 7.5 mm | 13523-6 | Moderate cracking | No cracking | No cracking | Severe cracking | Slight cracking |
| T-bend (no tape pick off) | 13523-7 | 0.5T | 1.5T | 1.5T | 1T | 1.5T |
| T-Bend no cracking | 13523-7 | 3T | 1T | 2T | 5.5T | 2.5T |
| MEK rubs primer | BSSP 3.522.11 | 110 | 110 | 110 | 110 | 110 |
| Tg, °C (onset/midpoint) | 28/35 | 26/32 | 34/42 | 58/69 | 29/34 | |
| Microhardness, N∙m−2 | 216 | 174 | 287 | 299 | 169 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lomelí-Rodríguez, M.; Corpas-Martínez, J.R.; Willis, S.; Mulholland, R.; Lopez-Sanchez, J.A. Synthesis and Characterization of Renewable Polyester Coil Coatings from Biomass-Derived Isosorbide, FDCA, 1,5-Pentanediol, Succinic Acid, and 1,3-Propanediol. Polymers 2018, 10, 600. https://doi.org/10.3390/polym10060600
Lomelí-Rodríguez M, Corpas-Martínez JR, Willis S, Mulholland R, Lopez-Sanchez JA. Synthesis and Characterization of Renewable Polyester Coil Coatings from Biomass-Derived Isosorbide, FDCA, 1,5-Pentanediol, Succinic Acid, and 1,3-Propanediol. Polymers. 2018; 10(6):600. https://doi.org/10.3390/polym10060600
Chicago/Turabian StyleLomelí-Rodríguez, Mónica, José Raúl Corpas-Martínez, Susan Willis, Robert Mulholland, and Jose Antonio Lopez-Sanchez. 2018. "Synthesis and Characterization of Renewable Polyester Coil Coatings from Biomass-Derived Isosorbide, FDCA, 1,5-Pentanediol, Succinic Acid, and 1,3-Propanediol" Polymers 10, no. 6: 600. https://doi.org/10.3390/polym10060600
APA StyleLomelí-Rodríguez, M., Corpas-Martínez, J. R., Willis, S., Mulholland, R., & Lopez-Sanchez, J. A. (2018). Synthesis and Characterization of Renewable Polyester Coil Coatings from Biomass-Derived Isosorbide, FDCA, 1,5-Pentanediol, Succinic Acid, and 1,3-Propanediol. Polymers, 10(6), 600. https://doi.org/10.3390/polym10060600






