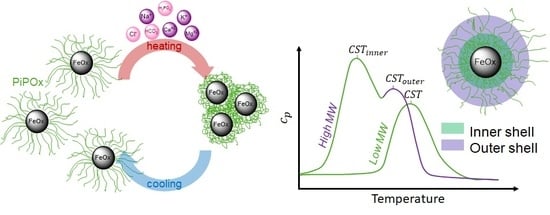
The Role of Chain Molecular Weight and Hofmeister Series Ions in Thermal Aggregation of Poly(2-Isopropyl-2-Oxazoline) Grafted Nanoparticles
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Methods
3. Results and Discussion
3.1. Synthesis of PiPOx-NDA Grafted Superparamagnetic Iron Oxide Nanoparticles (SPION)
3.2. Temperature-Induced Aggregation of Free Linear PiPOx Coils
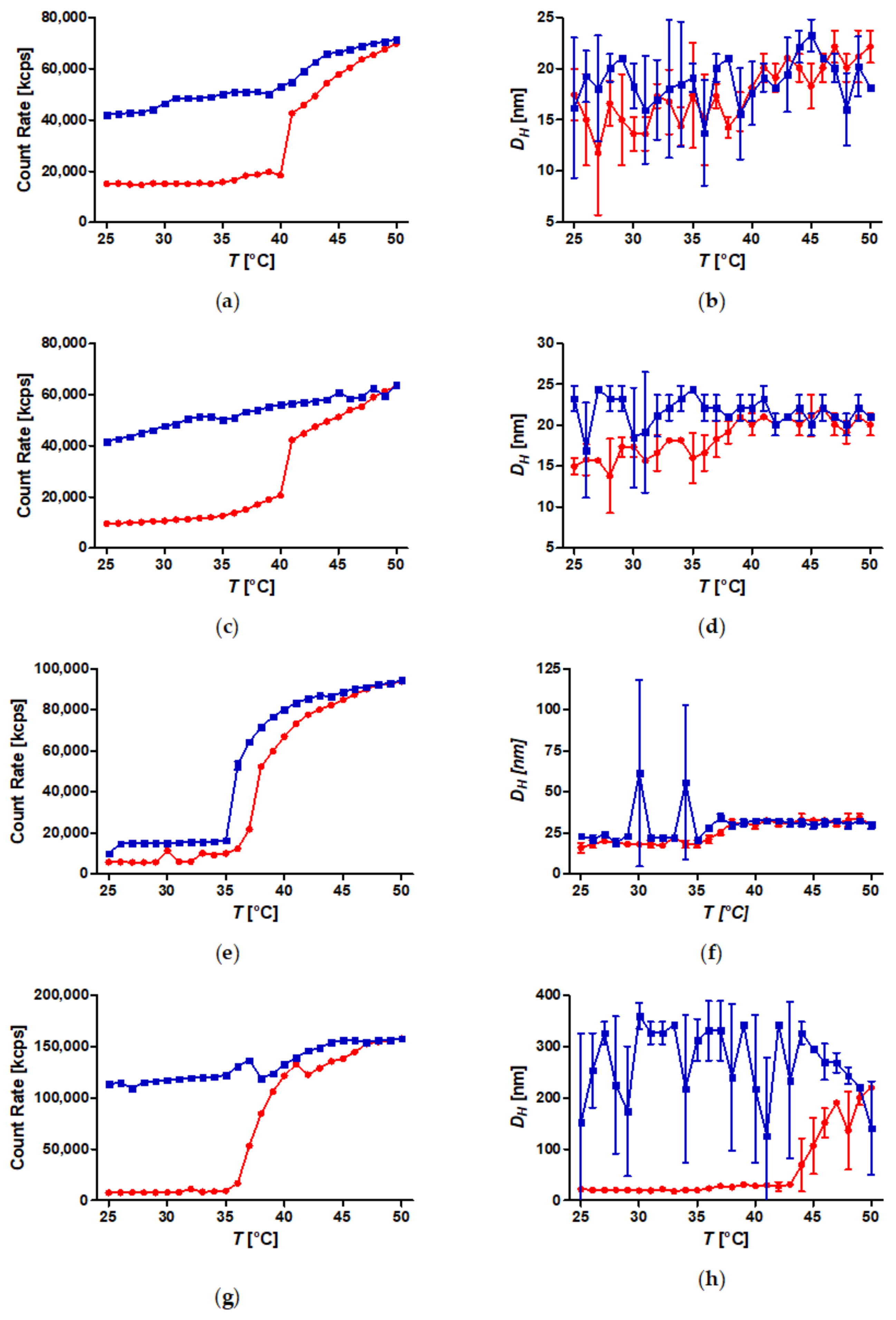
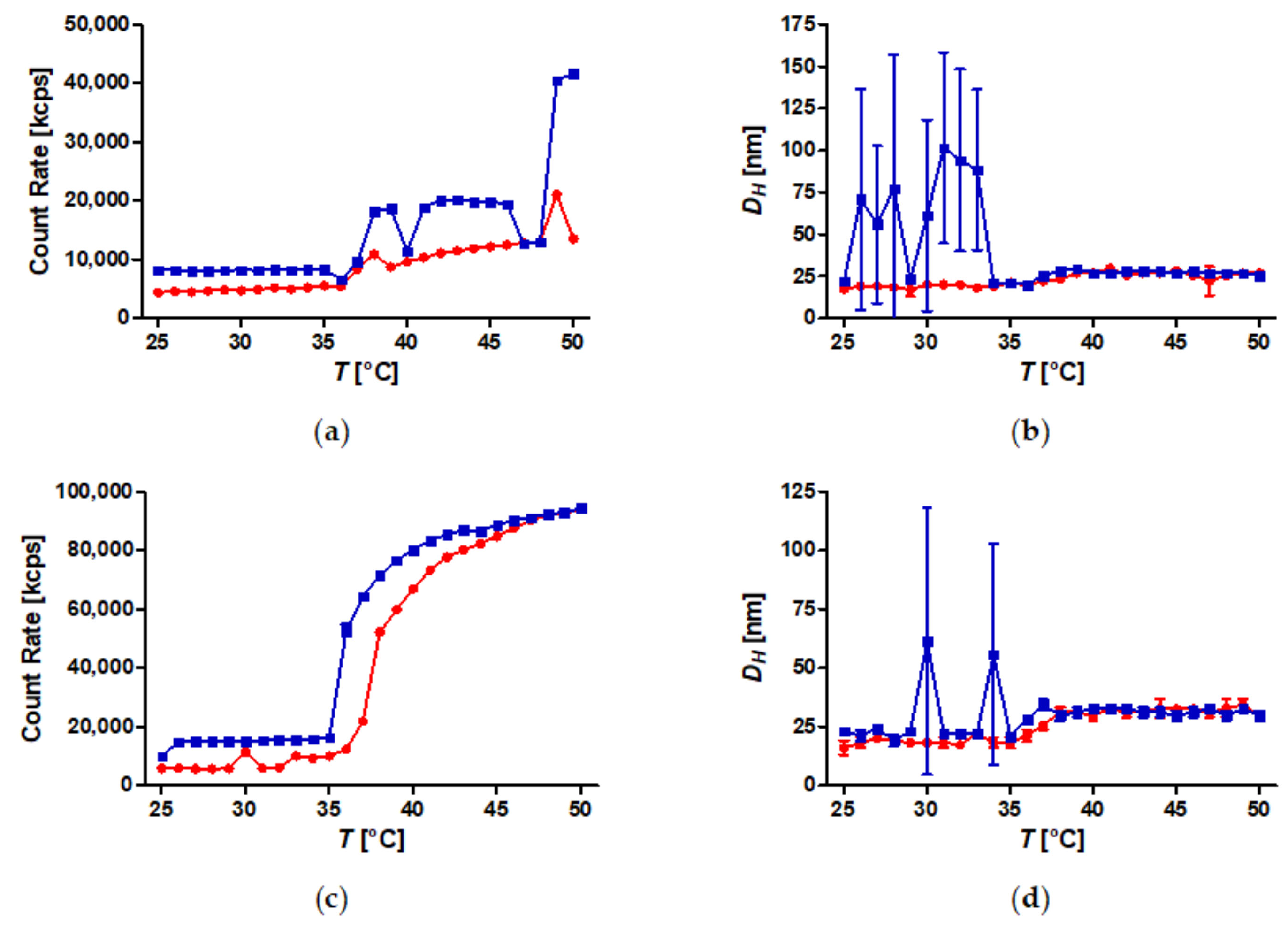
3.3. Temperature-Induced Aggregation of Dispersed SPION Grafted with PiPOx at Constant Mass Concentration
3.4. Temperature-Induced Aggregation of Dispersed SPION Grafted with PiPOx at Constant Molarity
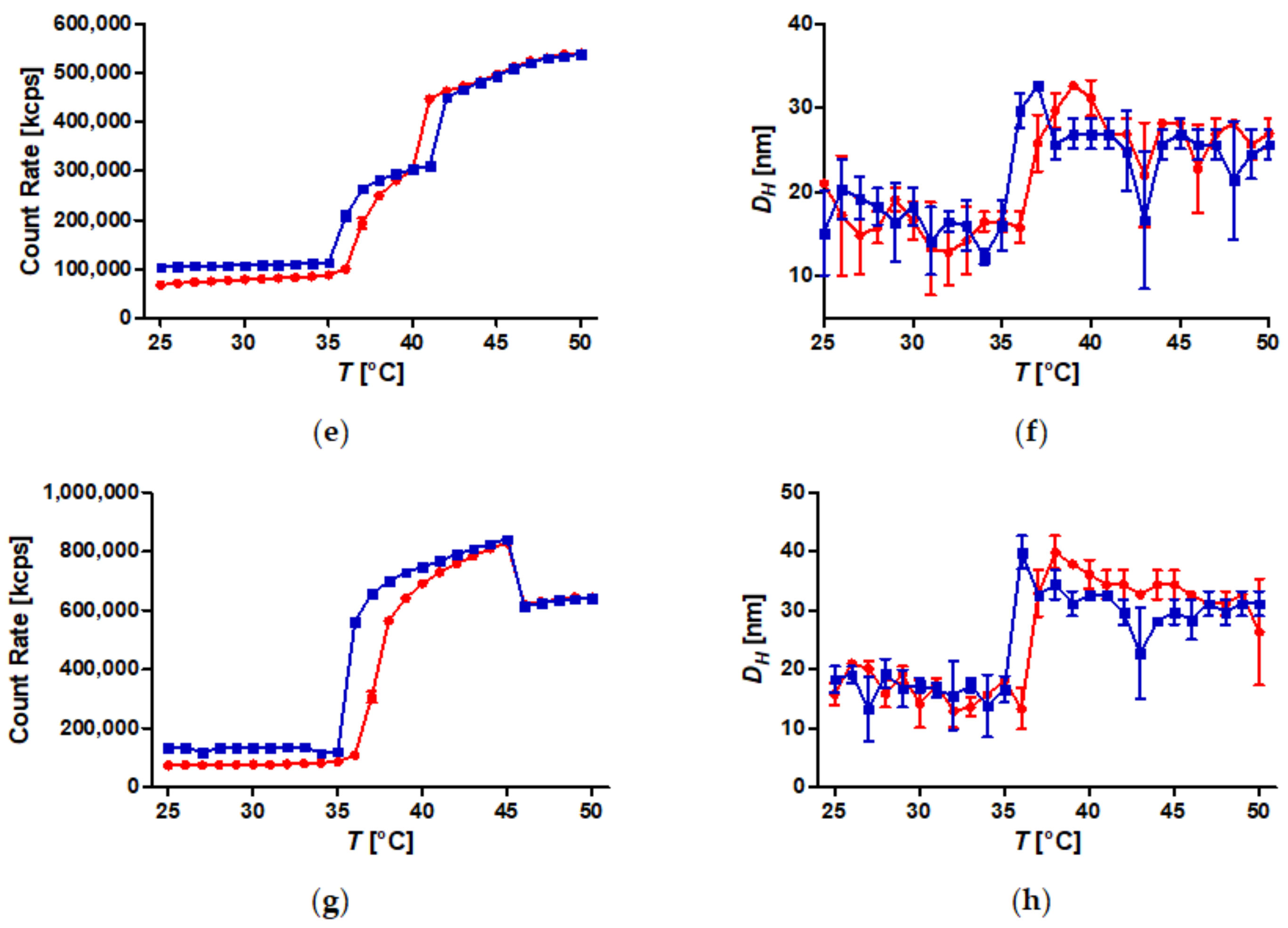
3.5. Temperature-Induced Aggregation of FeOx-21 at Different Concentrations
3.6. Comparison of Enthalpic Transition Measured by DSC and Colloidal Aggregation of PiPOx-Grafted SPION
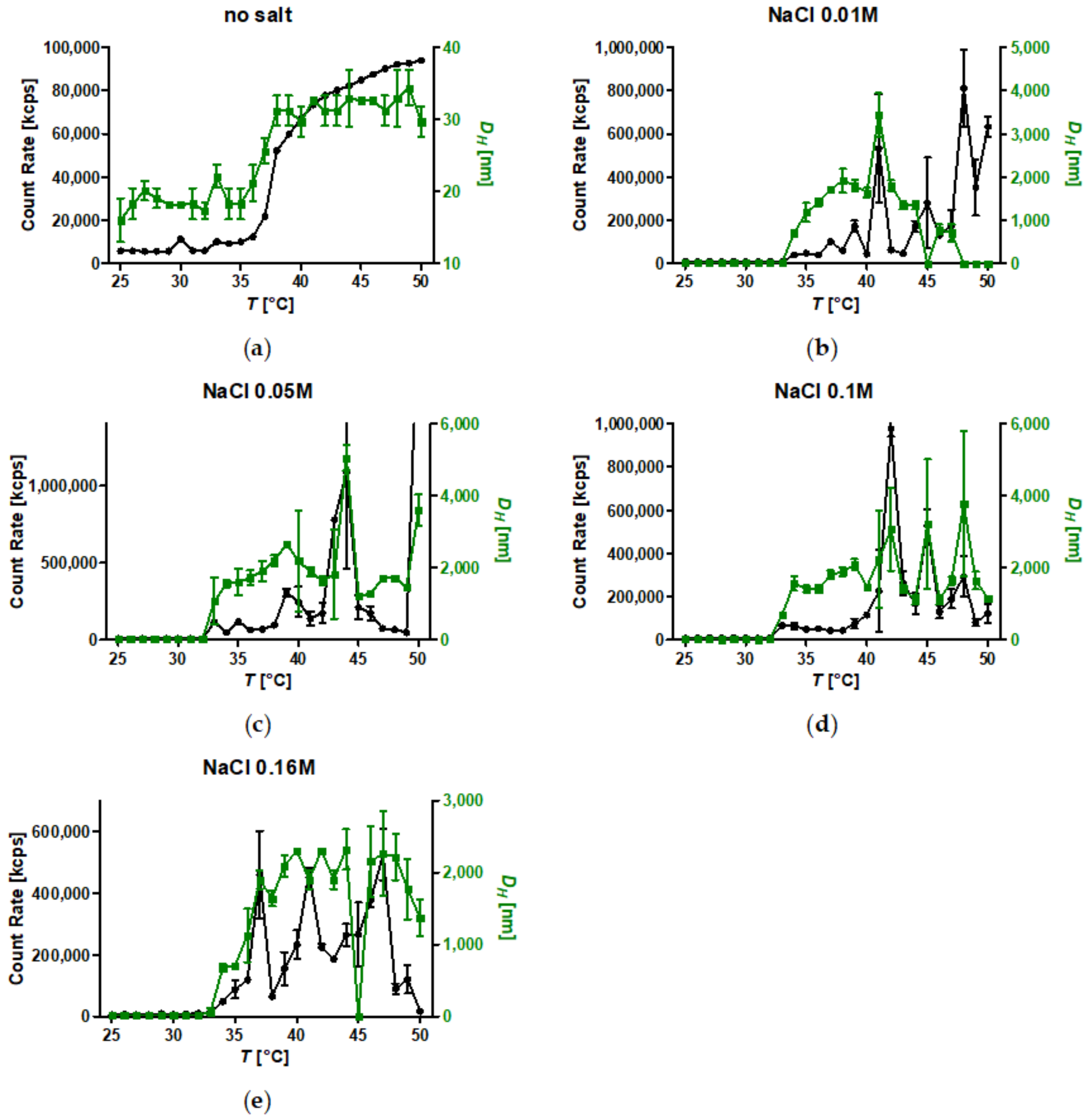
3.7. Temperature-Induced Aggregation of Dispersed SPION Grafted with PiPOx at Different Salt Concentrations
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Kakwere, H.; Leal, M.P.; Materia, M.E.; Curcio, A.; Guardia, P.; Niculaes, D.; Marotta, R.; Falqui, A.; Pellegrino, T. Functionalization of strongly interacting magnetic nanocubes with (thermo)responsive coating and their application in hyperthermia and heat-triggered drug delivery. ACS Appl. Mater. Interfaces 2015, 7, 10132–10145. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Guo, J.; Yang, W.; Hu, J.; Wang, C.; Fu, S. Magnetic mesoporous silica microspheres with thermo-sensitive polymer shell for controlled drug release. J. Mater. Chem. 2009, 19, 4764–4770. [Google Scholar] [CrossRef]
- Zhang, J.; Misra, R.D.K. Magnetic drug-targeting carrier encapsulated with thermosensitive smart polymer: Core-shell nanoparticle carrier and drug release response. Acta Biomater. 2007, 3, 838–850. [Google Scholar] [CrossRef] [PubMed]
- Yildiz, I.; Sizirici Yildiz, B. Applications of Thermoresponsive Magnetic Nanoparticles. J. Nanomater. 2015, 2015, 1–12. [Google Scholar] [CrossRef]
- Wang, C.; Irudayaraj, J. Multifunctional magnetic-optical nanoparticle probes for simultaneous detection, separation, and thermal ablation of multiple pathogens. Small 2010, 6, 283–289. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Q.; Chen, N.; Zhao, D.; Lu, X. Thermoresponsive magnetic nanoparticles for seawater desalination. ACS Appl. Mater. Interfaces 2013, 5, 11453–11461. [Google Scholar] [CrossRef] [PubMed]
- Crassous, J.J.; Mihut, A.M.; Dietsch, H.; Pravaz, O.; Ackermann-Hirschi, L.; Hirt, A.M.; Schurtenberger, P. Advanced multiresponsive comploids: From design to possible applications. Nanoscale 2014, 6, 8726–8735. [Google Scholar] [CrossRef] [PubMed]
- Luxenhofer, R.; Sahay, G.; Schulz, A.; Alakhova, D.; Bronich, T.K.; Jordan, R.; Kabanov, A.V. Structure-property relationship in cytotoxicity and cell uptake of poly(2-oxazoline) amphiphiles. J. Control. Release 2011, 153, 73–82. [Google Scholar] [CrossRef] [PubMed]
- Schellekens, H.; Hennink, W.E.; Brinks, V. The immunogenicity of polyethylene glycol: Facts and fiction. Pharm. Res. 2013, 30, 1729–1734. [Google Scholar] [CrossRef] [PubMed]
- Luxenhofer, R.; Han, Y.; Schulz, A.; Tong, J.; He, Z.; Kabanov, A.V.; Jordan, R. Poly(2-oxazoline)s as Polymer Therapeutics. Macromol. Rapid Commun. 2012, 33, 1613–1631. [Google Scholar] [CrossRef] [PubMed]
- Glassner, M.; Lava, K.; De La Rosa, V.R.; Hoogenboom, R. Tuning the LCST of poly(2-cyclopropyl-2-oxazoline) via gradient copolymerization with 2-ethyl-2-oxazoline. J. Polym. Sci. Part A Polym. Chem. 2014, 52, 3118–3122. [Google Scholar] [CrossRef]
- Park, J.S.; Kataoka, K. Precise control of lower critical solution temperature of thermosensitive poly(2-isopropyl-2-oxazoline) via gradient copolymerization with 2-ethyl-2-oxazoline as a hydrophilic comonomer. Macromolecules 2006, 39, 6622–6630. [Google Scholar] [CrossRef]
- Hoogenboom, R.; Thijs, H.M.L.; Jochems, M.J.H.C.; van Lankvelt, B.M.; Fijten, M.W.M.; Schubert, U.S. Tuning the LCST of poly(2-oxazoline)s by varying composition and molecular weight: Alternatives to poly(N-isopropylacrylamide)? Chem. Commun. 2008, 5758–5760. [Google Scholar] [CrossRef] [PubMed]
- Verbraeken, B.; Monnery, B.D.; Lava, K.; Hoogenboom, R. The chemistry of poly(2-oxazoline)s. Eur. Polym. J. 2017, 88, 451–469. [Google Scholar] [CrossRef]
- Uyama, H.; Kobayashi, S. A Novel Thermo-Sensitive Polymer. Poly(2-iso-propyl-2-oxazoline). Chem. Lett. 1992, 21, 1643–1646. [Google Scholar] [CrossRef]
- Hoogenboom, R.; Schlaad, H. Thermoresponsive poly(2-oxazoline)s, polypeptoids, and polypeptides. Polym. Chem. 2017, 8, 24–40. [Google Scholar] [CrossRef]
- Zhao, J.; Hoogenboom, R.; Van Assche, G.; Van Mele, B. Demixing and remixing kinetics of poly(2-isopropyl-2-oxazoline) (PIPOZ) aqueous solutions studied by modulated temperature differential scanning calorimetry. Macromolecules 2010, 43, 6853–6860. [Google Scholar] [CrossRef]
- Aseyev, V.; Tenhu, H.; Winnik, F.M. Non-ionic Thermoresponsive Polymers in Water. In Romanian Reports of Physics; Springer: Berlin, Germany, 2010; Volume 54, pp. 29–89. ISBN 9783319248608. [Google Scholar]
- Park, J.S.; Akiyama, Y.; Winnik, F.M.; Kataoka, K. Versatile synthesis of end-functionalized thermosensitive poly(2-isopropyl-2-oxazolines). Macromolecules 2004, 37, 6786–6792. [Google Scholar] [CrossRef]
- Salzinger, S.; Huber, S.; Jaksch, S.; Busch, P.; Jordan, R.; Papadakis, C.M. Aggregation behavior of thermo-responsive poly(2-oxazoline)s at the cloud point investigated by FCS and SANS. Colloid Polym. Sci. 2012, 290, 385–400. [Google Scholar] [CrossRef]
- Bloksma, M.M.; Bakker, D.J.; Weber, C.; Hoogenboom, R.; Schubert, U.S. The effect of hofmeister salts on the LCST transition of poly(2-oxazoline)s with varying hydrophilicity. Macromol. Rapid Commun. 2010, 31, 724–728. [Google Scholar] [CrossRef] [PubMed]
- Huber, S.; Hutter, N.; Jordan, R. Effect of end group polarity upon the lower critical solution temperature of poly(2-isopropyl-2-oxazoline). Colloid Polym. Sci. 2008, 286, 1653–1661. [Google Scholar] [CrossRef]
- Diab, C.; Akiyama, Y.; Kataoka, K.; Winnik, F.M. Microcalorimetric Study of the Temperature-Induced Phase Separation in Aqueous Solutions of Poly(2-isopropyl-2-oxazolines). Macromolecules 2004, 37, 2556–2562. [Google Scholar] [CrossRef]
- Huber, S.; Jordan, R. Modulation of the lower critical solution temperature of 2-Alkyl-2-oxazoline copolymers. Colloid Polym. Sci. 2008, 286, 395–402. [Google Scholar] [CrossRef]
- Tatar Güner, P.; Demirel, A.L. Effect of Anions on the Cloud Point Temperature of Aqueous Poly(2-ethyl-2-oxazoline) Solutions. J. Phys. Chem. B 2012, 116, 14510–14514. [Google Scholar] [CrossRef] [PubMed]
- Amirova, A.I.; Dudkina, M.M.; Tenkovtsev, A.V.; Filippov, A.P. Self-assembly of star-shaped poly(2-isopropyl-2-oxazoline) in aqueous solutions. Colloid Polym. Sci. 2014, 293, 239–248. [Google Scholar] [CrossRef]
- Zhang, N.; Luxenhofer, R.; Jordan, R. Thermoresponsive Poly(2-Oxazoline) Molecular Brushes by Living Ionic Polymerization: Modulation of the Cloud Point by Random and Block Copolymer Pendant Chains. Macromol. Chem. Phys. 2012, 213, 1963–1969. [Google Scholar] [CrossRef]
- Zhang, N.; Huber, S.; Schulz, A.; Luxenhofer, R.; Jordan, R. Cylindrical Molecular Brushes of Poly(2-oxazoline)s from 2-Isopropenyl-2-oxazoline. Macromolecules 2009, 42, 2215–2221. [Google Scholar] [CrossRef]
- Zhang, N.; Luxenhofer, R.; Jordan, R. Thermoresponsive Poly(2-oxazoline) Molecular Brushes by Living Ionic Polymerization: Kinetic Investigations of Pendant Chain Grafting and Cloud Point Modulation by Backbone and Side Chain Length Variation. Macromol. Chem. Phys. 2012, 213, 973–981. [Google Scholar] [CrossRef]
- Grünewald, T.A.; Lassenberger, A.; van Oostrum, P.D.J.; Rennhofer, H.; Zirbs, R.; Capone, B.; Vonderhaid, I.; Amenitsch, H.; Lichtenegger, H.C.; Reimhult, E. Core–Shell Structure of Monodisperse Poly(ethylene glycol)-Grafted Iron Oxide Nanoparticles Studied by Small-Angle X-ray Scattering. Chem. Mater. 2015, 27, 4763–4771. [Google Scholar] [CrossRef] [PubMed]
- Kurzhals, S.; Gal, N.; Zirbs, R.; Reimhult, E. Controlled aggregation and cell uptake of thermoresponsive polyoxazoline-grafted superparamagnetic iron oxide nanoparticles. Nanoscale 2017, 9, 2793–2805. [Google Scholar] [CrossRef] [PubMed]
- Hyeon, T.; Lee, S.S.; Park, J.; Chung, Y.; Na, H.B. Synthesis of highly crystalline and monodisperse maghemite nanocrystallites without a size-selection process. J. Am. Chem. Soc. 2001, 123, 12798–12801. [Google Scholar] [CrossRef] [PubMed]
- Zirbs, R.; Lassenberger, A.; Vonderhaid, I.; Kurzhals, S.; Reimhult, E. Melt-grafting for the synthesis of core–shell nanoparticles with ultra-high dispersant density. Nanoscale 2015, 7, 11216–11225. [Google Scholar] [CrossRef] [PubMed]
- Witte, H.; Seeliger, W. Cyclische Imidsäureester aus Nitrilen und Aminoalkoholen. Justus Liebigs Ann. Chem. 1974, 1974, 996–1009. [Google Scholar] [CrossRef]
- Monnery, B.D.; Shaunak, S.; Thanou, M.; Steinke, J.H.G. Improved synthesis of linear poly(ethylenimine) via low-temperature polymerization of 2-isopropyl-2-oxazoline in chlorobenzene. Macromolecules 2015, 48, 3197–3206. [Google Scholar] [CrossRef]
- Mondini, S.; Ferretti, A.M.; Puglisi, A.; Ponti, A. Pebbles and PebbleJuggler: Software for accurate, unbiased, and fast measurement and analysis of nanoparticle morphology from transmission electron microscopy (TEM) micrographs. Nanoscale 2012, 4, 5356. [Google Scholar] [CrossRef] [PubMed]
- Amstad, E.; Gillich, T.; Bilecka, I.; Textor, M.; Reimhult, E. Ultrastable Iron Oxide Nanoparticle Colloidal Suspensions Using Dispersants with Catechol-Derived Anchor Groups. Nano Lett. 2009, 9, 4042–4048. [Google Scholar] [CrossRef] [PubMed]
- Yuen, A.K.L.; Hutton, G.A.; Masters, A.F.; Maschmeyer, T. The interplay of catechol ligands with nanoparticulate iron oxides. Dalton Trans. 2012, 41, 2545–2559. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Wei, Q.; Qu, S. Synthesis and characterization of uniform and crystalline magnetite nanoparticles via oxidation-precipitation and modified co-precipitation methods. Int. J. Electrochem. Sci. 2013, 8, 3786–3793. [Google Scholar] [CrossRef]
- Amstad, E.; Gehring, A.U.; Fischer, H.; Nagaiyanallur, V.V.; Hähner, G.; Textor, M.; Reimhult, E. Influence of Electronegative Substituents on the Binding Affinity of Catechol-Derived Anchors to Fe3O4 Nanoparticles. J. Phys. Chem. C 2011, 115, 683–691. [Google Scholar] [CrossRef]
- Katsumoto, Y.; Tsuchiizu, A.; Qiu, X.; Winnik, F.M. Dissecting the mechanism of the heat-induced phase separation and crystallization of poly(2-isopropyl-2-oxazoline) in water through vibrational spectroscopy and molecular orbital calculations. Macromolecules 2012, 45, 3531–3541. [Google Scholar] [CrossRef]
- Furuncuoğlu Özaltın, T.; Aviyente, V.; Atılgan, C.; Demirel, L. Multiscale modeling of poly(2-isopropyl-2-oxazoline) chains in aqueous solution. Eur. Polym. J. 2017, 88, 594–604. [Google Scholar] [CrossRef]
- Li, T.; Tang, H.; Wu, P. Molecular Evolution of Poly(2-isopropyl-2-oxazoline) Aqueous Solution during the Liquid–Liquid Phase Separation and Phase Transition Process. Langmuir 2015, 31, 6870–6878. [Google Scholar] [CrossRef] [PubMed]
- Sun, S.; Wu, P. From globule to crystal: A spectral study of poly(2-isopropyl-2-oxazoline) crystallization in hot water. Phys. Chem. Chem. Phys. 2015, 17, 32232–32240. [Google Scholar] [CrossRef] [PubMed]
- Kurzhals, S.; Gal, N.; Zirbs, R.; Reimhult, E. Aggregation of thermoresponsive core-shell nanoparticles: Influence of particle concentration, dispersant molecular weight and grafting. J. Colloid Interface Sci. 2017, 500, 321–332. [Google Scholar] [CrossRef] [PubMed]
- Vasicek, T.W.; Jenkins, S.V.; Vaz, L.; Chen, J.; Stenken, J.A. Thermoresponsive nanoparticle agglomeration/aggregation in salt solutions: Dependence on graft density. J. Colloid Interface Sci. 2017, 506, 338–345. [Google Scholar] [CrossRef] [PubMed]
- Koshkina, O.; Lang, T.; Thiermann, R.; Docter, D.; Stauber, R.H.; Secker, C.; Schlaad, H.; Weidner, S.; Mohr, B.; Maskos, M.; et al. Temperature-Triggered Protein Adsorption on Polymer-Coated Nanoparticles in Serum. Langmuir 2015, 31, 8873–8881. [Google Scholar] [CrossRef] [PubMed]
- Chen, N.; Xiang, X.; Heiden, P.A. Tuning thermoresponsive behavior of diblock copolymers and their gold core hybrids. Part 2. How properties change depending on block attachment to gold nanoparticles. J. Colloid Interface Sci. 2013, 396, 39–46. [Google Scholar] [CrossRef] [PubMed]
- Kurzhals, S.; Pretzner, B.; Reimhult, E.; Zirbs, R. Thermoresponsive Polypeptoid-Coated Superparamagnetic Iron Oxide Nanoparticles by Surface-Initiated Polymerization. Macromol. Chem. Phys. 2017, 218, 1700116. [Google Scholar] [CrossRef]
- Xu, J.; Liu, S. Synthesis of well-defined 7-arm and 21-arm poly(N-isopropylacrylamide) star polymers with β-cyclodextrin cores via click chemistry and their thermal phase transition behavior in aqueous solution. J. Polym. Sci. Part A Polym. Chem. 2009, 47, 404–419. [Google Scholar] [CrossRef]
- Kurzhals, S.; Schroffenegger, M.; Gal, N.; Zirbs, R.; Reimhult, E. Influence of Grafted Block Copolymer Structure on Thermoresponsiveness of Superparamagnetic Core–Shell Nanoparticles. Biomacromolecules 2017. [Google Scholar] [CrossRef] [PubMed]
- Lo Verso, F.; Egorov, S.A.; Milchev, A.; Binder, K. Spherical polymer brushes under good solvent conditions: Molecular dynamics results compared to density functional theory. J. Chem. Phys. 2010, 133. [Google Scholar] [CrossRef] [PubMed]
- Shan, J.; Chen, J.; Nuopponen, M.; Tenhu, H. Two Phase Transitions of Poly(N-isopropylacrylamide) Brushes Bound to Gold Nanoparticles. Langmuir 2004, 20, 4671–4676. [Google Scholar] [CrossRef] [PubMed]






| Sample | Core-Diameter [nm] | Molecular Weight (GPC) [kg mol−1] | TGA | DLS [nm] * | ||
|---|---|---|---|---|---|---|
| Weight Loss [wt %] | Residue [wt %] | σ [Molecules nm−²] | ||||
| FeOx-6 | 9.1 ± 0.3 | 6 | 56 | 44 | 1.0 | 16.0 ± 2.9 |
| FeOx-14 | 9.1 ± 0.3 | 14 | 70 | 30 | 0.8 | 17.2 ± 2.2 |
| FeOx-21 | 9.1 ± 0.3 | 21 | 74 | 26 | 1.0 | 18.6 ± 2.3 |
| FeOx-33 | 9.1 ± 0.3 | 33 | 87 | 13 | 1.0 | 20.2 ± 2.4 |
| Sample | Molecular Weight(GPC) [kg mol−1] | PDI | CST (DLS) [°C] * | CST (DSC) [°C] * | * [kJ mol−1] | |
|---|---|---|---|---|---|---|
| Onset | Maximum | |||||
| PiPOx-6 | 6 | 1.08 | 46 | 51 | 65 | 2.1 |
| PiPOx-14 | 14 | 1.05 | 43 | 43 | 49 | 4.2 |
| PiPOx-21 | 21 | 1.10 | 39 | 40 | 43 | 4.3 |
| PiPOx-33 | 33 | 1.15 | 37 | 38 | 39 | 4.2 |
| Sample | [nm] | [nm] | CST [°C] |
|---|---|---|---|
| FeOx-6 | 16.0 ± 2.9 | 19.9 ± 2.3 | 40 |
| FeOx-14 | 17.2 ± 2.2 | 20.7 ± 1.6 | 38 |
| FeOx-21 | 18.6 ± 2.3 | 31.8 ± 2.6 | 36 |
| FeOx-33 | 20.2 ± 2.4 | 29.5 ± 4.7 * | 35 |
| Sample | Concentration [g L−1] | [nm] | [nm] | CST [°C] |
|---|---|---|---|---|
| FeOx-6 | 0.23 | 15.8 ± 2.9 | 17.8 ± 3.2 | 44 |
| FeOx-14 | 0.34 | 18.9 ± 1.6 | 97 ± 44 | 42 |
| FeOx-21 | 0.54 | 19.0 ± 2.1 | 26.3 ± 3.4 | 37 |
| FeOx-33 | 0.78 | 18.8 ± 3.5 | 27.2 ± 5.6 * | 36 |
| Sample | CST [°C] | [kJ mol−1] | * [kJ mol−1] | Degree of Polymerisation | |
|---|---|---|---|---|---|
| Onset | Maximum | ||||
| FeOx-6 | 43 | 49 | 19 | 0.4 | 51 |
| FeOx-14 | 31 | 45 | 93 | 0.8 | 119 |
| FeOx-21 | 30 | 40 | 448 | 2.5 | 181 |
| FeOx-33 | 31 | 39 | 639 | 2.2 | 288 |
| Sample | [°C] | [°C] | [°C] | [kJ/mol] | [kJ/mol] | [%] |
|---|---|---|---|---|---|---|
| PiPOx-6 | 65 | 56 | 9 | 111 | 95 | 86 |
| PiPOx-14 | 49 | 45 | 4 | 498 | 579 | 116 |
| PiPOx-21 | 43 | 41 | 2 | 776 | 948 | 122 |
| PiPOx-33 | 39 | 39 | 0 | 1220 | 1150 | 94 |
| FeOx-6 | 49 | 45 | 4 | 19 | 4 | 21 |
| FeOx-14 | 45 | 37 | 8 | 93 | 44 | 47 |
| FeOx-21 | 40 | 40 | 0 | 448 | 373 | 83 |
| FeOx-33 | 39 | 39 | 0 | 639 | 250 | 39 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Schroffenegger, M.; Zirbs, R.; Kurzhals, S.; Reimhult, E. The Role of Chain Molecular Weight and Hofmeister Series Ions in Thermal Aggregation of Poly(2-Isopropyl-2-Oxazoline) Grafted Nanoparticles. Polymers 2018, 10, 451. https://doi.org/10.3390/polym10040451
Schroffenegger M, Zirbs R, Kurzhals S, Reimhult E. The Role of Chain Molecular Weight and Hofmeister Series Ions in Thermal Aggregation of Poly(2-Isopropyl-2-Oxazoline) Grafted Nanoparticles. Polymers. 2018; 10(4):451. https://doi.org/10.3390/polym10040451
Chicago/Turabian StyleSchroffenegger, Martina, Ronald Zirbs, Steffen Kurzhals, and Erik Reimhult. 2018. "The Role of Chain Molecular Weight and Hofmeister Series Ions in Thermal Aggregation of Poly(2-Isopropyl-2-Oxazoline) Grafted Nanoparticles" Polymers 10, no. 4: 451. https://doi.org/10.3390/polym10040451
APA StyleSchroffenegger, M., Zirbs, R., Kurzhals, S., & Reimhult, E. (2018). The Role of Chain Molecular Weight and Hofmeister Series Ions in Thermal Aggregation of Poly(2-Isopropyl-2-Oxazoline) Grafted Nanoparticles. Polymers, 10(4), 451. https://doi.org/10.3390/polym10040451