Some Key Factors Influencing the Flame Retardancy of EDA-DOPO Containing Flexible Polyurethane Foams
Abstract
:1. Introduction
2. Experimental
2.1. Materials and Methods
2.2. PU Foam Manufacturing
2.3. UL 94 HB Tests
2.4. Thermal Analysis
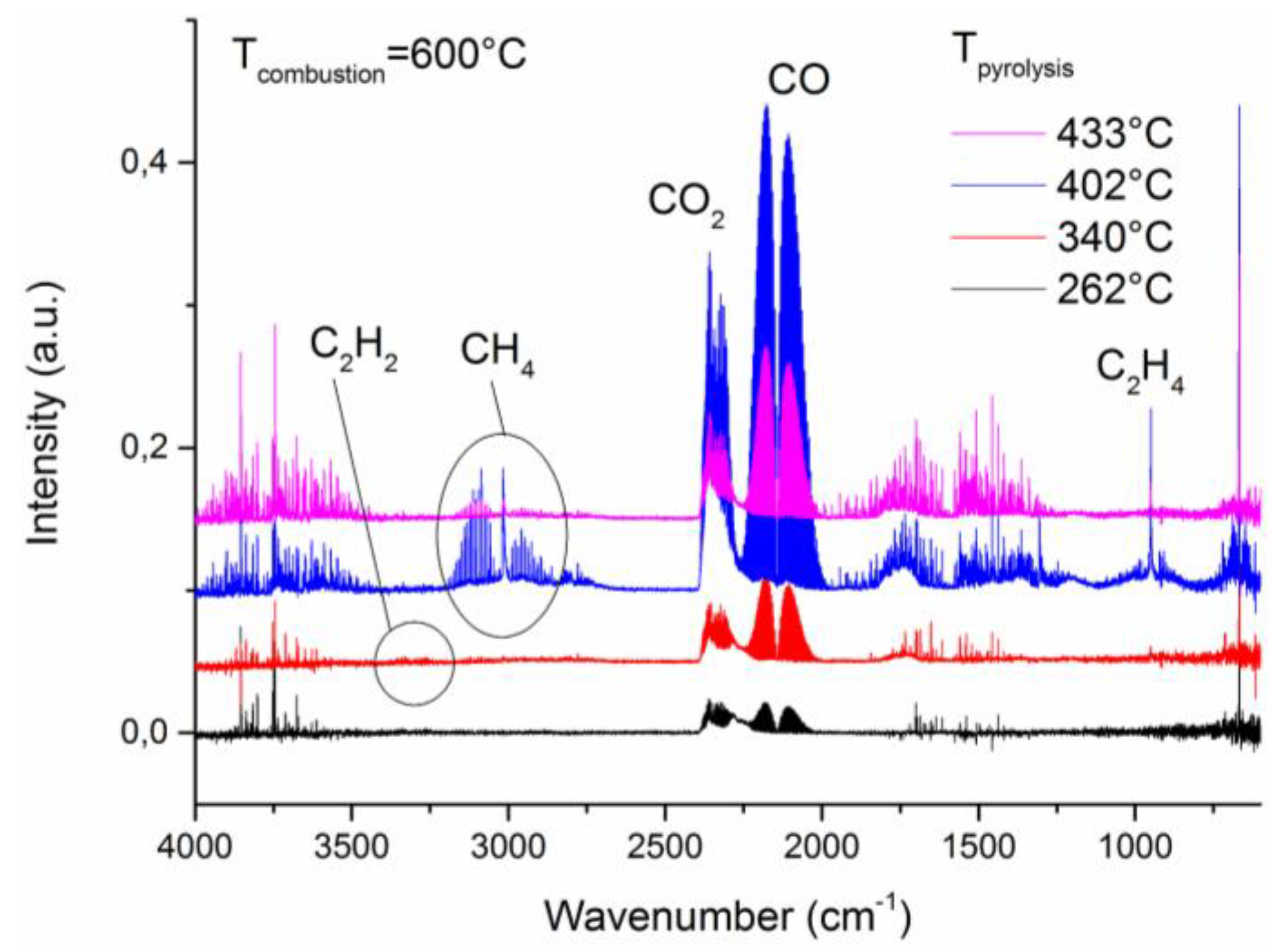
2.5. MCC/FTIR Coupling
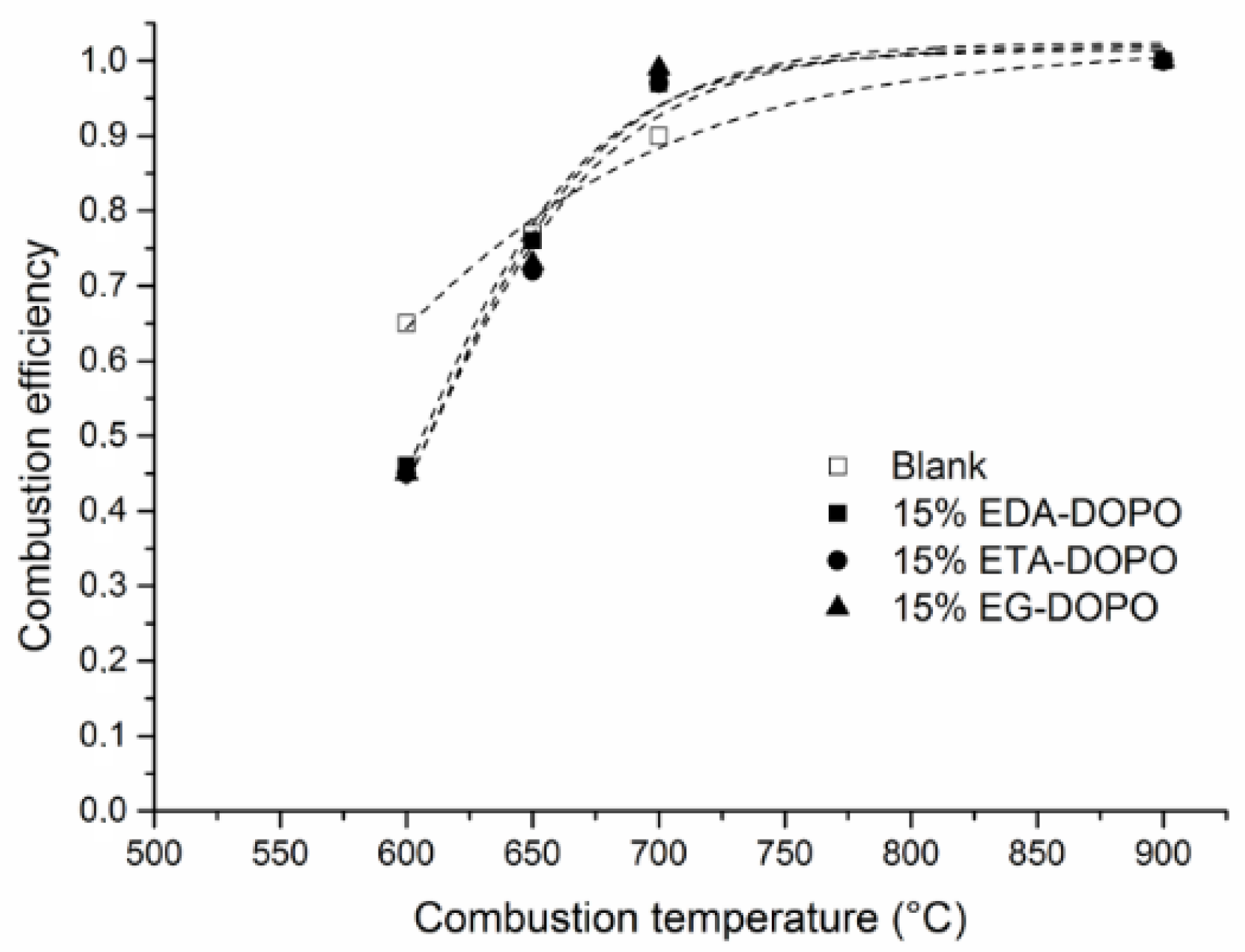
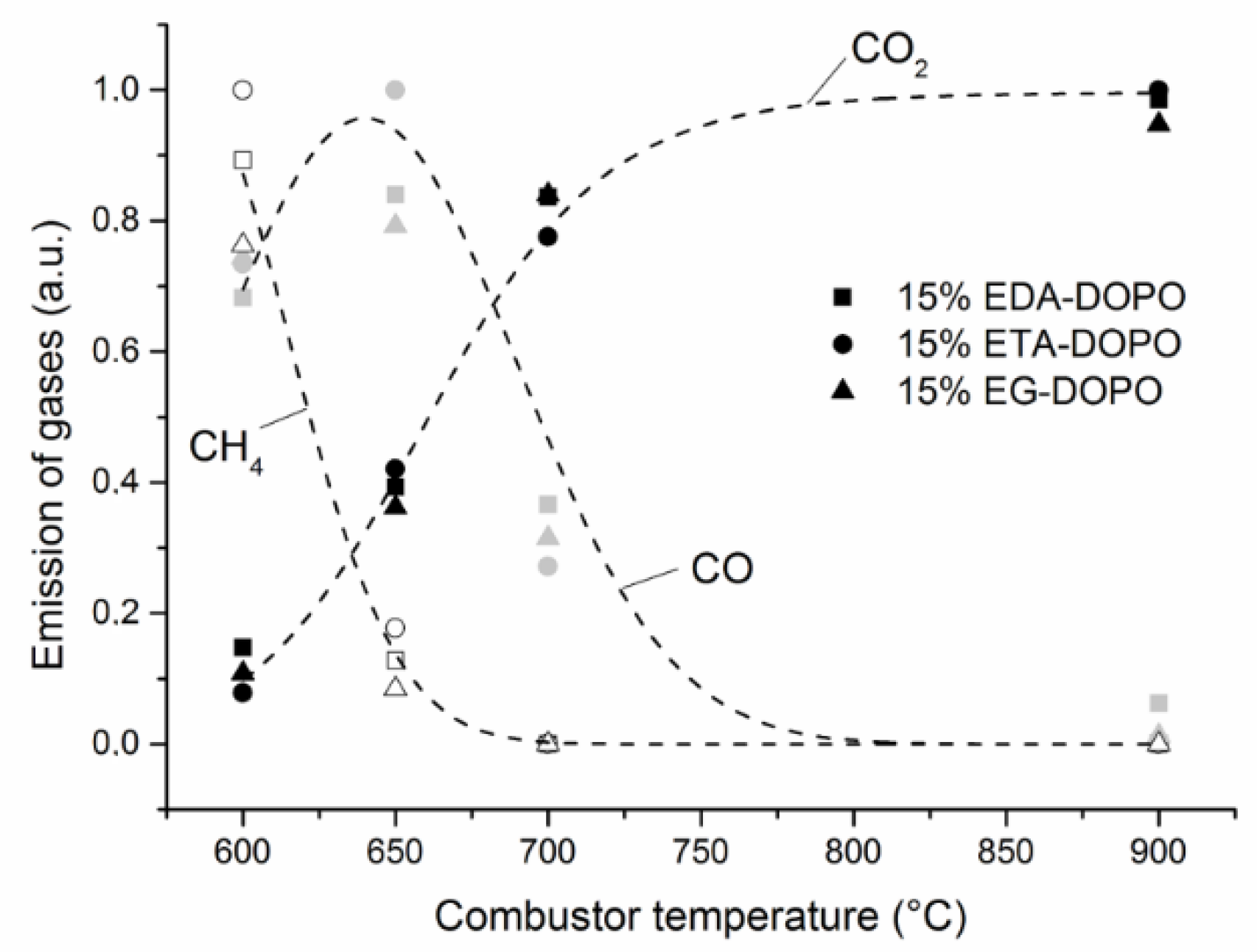
3. Results and Discussion
3.1. PU Foam Formulations
3.2. Properties of Bridged DOPO Compounds
3.3. Effect of PU Formulation on Fire Performance
3.4. Modification of Formulation B and Fire Performance
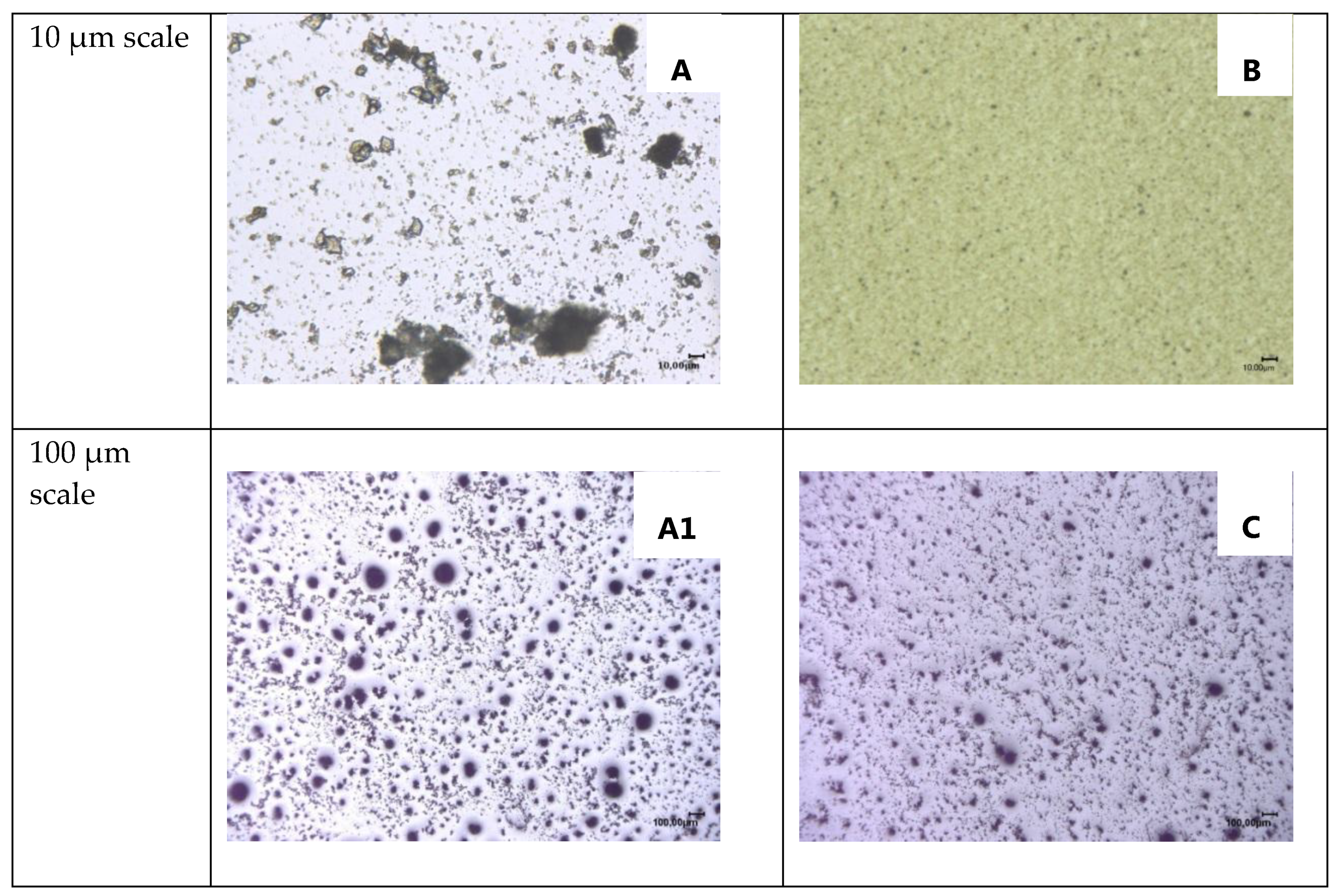
3.5. Dripping Behavior of PU Foams during UL 94 HB Tests
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Levchik, S.V.; Weil, E.D. Thermal decomposition, combustion and fire-retardancy of polyurethanes—A review of the recent literature. Polym. Int. 2004, 53, 1585–1610. [Google Scholar] [CrossRef]
- Chattopadhyay, D.K.; Webster, D.C. Thermal stability and flame retardancy of polyurethanes. Prog. Polym. Sci. 2009, 34, 1068–1133. [Google Scholar] [CrossRef]
- Neisius, N.M.; Liang, S.; Mispreuve, H.; Gaan, S. Recent developments in flame retardancy of flexible polyurethane foams. In Fire and Polymers vi: New Advances in Flame Retardant Chemistry and Science; American Chemical Society: Washington, DC, USA, 2012; pp. 251–270. [Google Scholar]
- Salmeia, K.; Flaig, F.; Rentsch, D.; Gaan, S. One-pot synthesis of P(O)-N containing compounds using N-chlorosuccinimide and their influence in thermal decomposition of PU foams. Polymers 2018, 10, 740. [Google Scholar] [CrossRef]
- Carl-Christoph, H.; Ronny, H.; Edwin, K. Intrinsic flame resistance of polyurethane flexible foams: Unexpectedly low flammability without any flame retardant. Fire Mater. 2018, 42, 394–402. [Google Scholar] [Green Version]
- Salmeia, K.A.; Gaan, S. An overview of some recent advances in DOPO-derivatives: Chemistry and flame retardant applications. Polym. Degrad. Stab. 2015, 113, 119–134. [Google Scholar] [CrossRef]
- Buczko, A.; Stelzig, T.; Bommer, L.; Rentsch, D.; Heneczkowski, M.; Gaan, S. Bridged DOPO derivatives as flame retardants for PA6. Polym. Degrad. Stab. 2014, 107, 158–165. [Google Scholar] [CrossRef]
- Stelzig, T.; Bommer, L.; Gaan, S.; Buczko, A. Dopo-based Hybrid Flame Retardants. WO2015140105A1, 23 March 2015. [Google Scholar]
- Butnaru, I.; Fernandez-Ronco, M.P.; Czech-Polak, J.; Heneczkowski, M.; Bruma, M.; Gaan, S. Effect of meltable triazine-DOPO additive on rheological, mechanical, and flammability properties of PA6. Polymers 2015, 7, 1541–1563. [Google Scholar] [CrossRef]
- Gaan, S.; Neisius, M.; Mercoli, P.; Liang, S.; Mispreuve, H.; Naescher, R. Phosphonamidates, Production Methods and Flame Retardant Applications. WO2013020696A2, 14 February 2013. [Google Scholar]
- Neisius, N.M.; Lutz, M.; Rentsch, D.; Hemberger, P.; Gaan, S. Synthesis of DOPO-based phosphonamidates and their thermal properties. Ind. Eng. Chem. Res. 2014, 53, 2889–2896. [Google Scholar] [CrossRef]
- Hirsch, C.; Striegl, B.; Mathes, S.; Adlhart, C.; Edelmann, M.; Bono, E.; Gaan, S.; Salmeia, K.A.; Hoelting, L.; Krebs, A.; et al. Multiparameter toxicity assessment of novel DOPO-derived organophosphorus flame retardants. Arch. Toxicol. 2017, 91, 407–425. [Google Scholar] [CrossRef] [PubMed]
- Neisius, M.; Liang, S.; Mispreuve, H.; Gaan, S. Phosphoramidate-containing flame-retardant flexible polyurethane foams. Ind. Eng. Chem. Res. 2013, 52, 9752–9762. [Google Scholar] [CrossRef]
- Liang, S.; Neisius, M.; Mispreuve, H.; Naescher, R.; Gaan, S. Flame retardancy and thermal decomposition of flexible polyurethane foams: Structural influence of organophosphorus compounds. Polym. Degrad. Stab. 2012, 97, 2428–2440. [Google Scholar] [CrossRef]
- Walters, R.N.; Safronava, N.; Lyon, R.E. A microscale combustion calorimeter study of gas phase combustion of polymers. Combust. Flame 2015, 162, 855–863. [Google Scholar] [CrossRef]
- Stoliarov, S.I.; Raffan-Montoya, F.; Walters, R.N.; Lyon, R.E. Measurement of the global kinetics of combustion for gaseous pyrolyzates of polymeric solids containing flame retardants. Combust. Flame 2016, 173, 65–76. [Google Scholar] [CrossRef]
- Sonnier, R.; Vahabi, H.; Ferry, L.; Lopez-Cuesta, J.M. Pyrolysis-combustion flow calorimetry: A powerful tool to evaluate the flame retardancy of polymers. In Fire and Polymers vi: New Advances in Flame Retardant Chemistry and Science; American Chemical Society: Washington, DC, USA, 2012; pp. 361–390. [Google Scholar]
- Sonnier, R.; Otazaghine, B.; Ferry, L.; Lopez-Cuesta, J.-M. Study of the combustion efficiency of polymers using a pyrolysis-combustion flow calorimeter. Combust. Flame 2013, 160, 2182–2193. [Google Scholar] [CrossRef]
- Sonnier, R.; Dorez, G.; Vahabi, H.; Longuet, C.; Ferry, L. Ftir-pcfc coupling: A new method for studying the combustion of polymers. Combust. Flame 2014, 161, 1398–1407. [Google Scholar] [CrossRef]
- Neisius, N.M.; Liang, S.; Gaan, S.; Mispreuve, H.; Nascher, R.; Rentsch, D. Flame Retardants for Flexible Polyurethane Foams: Structure-property-relationship Studies; American Chemical Society: Washington, DC, USA, 2012. [Google Scholar]
- Malewska, E.; Prociak, A. The effect of nanosilica filler on the foaming process and properties of flexible polyurethane foams obtained with rapeseed oil-based polyol. Polimery 2015, 60, 472–479. [Google Scholar] [CrossRef]
- Rao, J.; Fernandez-Ronco, M.P.; Vong, M.; Gaan, S. Enhanced flame-retardancy and controlled physical properties of flexible polyurethane foams based on a shear-responsive internal network. RSC Adv. 2017, 7, 44013–44020. [Google Scholar] [CrossRef] [Green Version]
- Karrasch, A.; Wawrzyn, E.; Schartel, B.; Jäger, C. Solid-state NMR on thermal and fire residues of bisphenol a polycarbonate/silicone acrylate rubber/bisphenol a bis(diphenyl-phosphate)/(PC/SiR/BDP) and PC/SiR/BDP/zinc borate (PC/SiR/BDP/ZnB)—part I;: PC charring and the impact of BDP and ZnB. Polym. Degrad. Stab. 2010, 95, 2525–2533. [Google Scholar] [CrossRef]
- Levchik, S.V.; Weil, E.D. Flame retardants in commercial use or in advanced development in polycarbonates and polycarbonate blends. J. Fire Sci. 2006, 24, 137–151. [Google Scholar] [CrossRef]
- Roma, P.; Camino, G.; Luda, M.P. Mechanistic studies on fire retardant action of fluorinated additives in ABS. Fire Mater. 1997, 21, 199–204. [Google Scholar] [CrossRef]
- Roma, P.; Luda, M.P.; Camino, G. Synergistic action of fluorine-containing additives in bromine/antimony fire retardant ABS. Polym. Degrad. Stab. 1999, 64, 497–500. [Google Scholar] [CrossRef]
- Zhang, X.; Zhang, F. Application of novel P-N intumescent flame retardant in polypropylene. Zhongguo Suliao 2012, 26, 92–96. [Google Scholar] [CrossRef]
- Matzen, M.; Kandola, B.; Huth, C.; Schartel, B. Influence of flame retardants on the melt dripping behaviour of thermoplastic polymers. Materials 2015, 8, 5267. [Google Scholar] [CrossRef] [PubMed]
- Levchik, S.V.; Bright, D.A.; Alessio, G.R.; Dashevsky, S. New halogen-free fire retardant for engineering plastic applications. J. Vinyl Addit. Technol. 2001, 7, 98–103. [Google Scholar] [CrossRef]
| Properties | EDA-DOPO | ETA-DOPO | EG-DOPO |
|---|---|---|---|
| Structures |  | 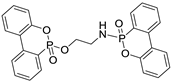 |  |
| Appearance | White powder | White powder | White powder |
| Bulk density | 2.10 g/mL | 2.01 g/mL | 2.02 g/mL |
| Melting point | 272–275 °C | 177–178 °C | 130–147 °C |
| Td 5% * | 357 °C | 321 °C | 341 °C |
| %P | 12.68 | 12.66 | 12.63 |
| Diastereoisomers ratio | 1.2:0.8 | 1:1 | 1:1 |
| Ingredients | Formulation A B | |
|---|---|---|
| Part (grams) | ||
| PO 56 (Polyol) | 97 | 100 |
| Soft 38 (Polyol) | 3 | - |
| Sodium dodecyl sulfonate | 0.8 | - |
| Stannous catalyst | 0.25 | 0.45 |
| Triethylenediamine | 0.1 | 0.4 |
| Silicone surfactant | 0.5 | 0.5 |
| Water (total) | 1.85 | 1.6 |
| TDI | 29.8 | 26.2 |
| FR a | X b | X b |
| Ingredients | Modified B Formulations | ||
|---|---|---|---|
| BDisp a | BE | BP b | |
| Part (grams) | |||
| PO 56 | 100 | 100 | 100 |
| Sodium dodecyl sulfonate | - | 0.8 | |
| PTFE | - | - | 0.5 c |
| Stannous catalyst | 0.45 | 0.45 | 0.45 |
| Triethylenediamine | 0.4 | 0.4 | 0.4 |
| Silicone surfactant | 0.5 | 0.5 | 0.5 |
| Water (total) | 1.6 | 1.15 | 1.6 |
| TDI | 26.2 | 26.2 | 26,2 |
| EDA-DOPO | 0–7.5 d | 0–7.5 d | 0–7.5 d |
| Foam Samples | Formulation A | Formulation B |
|---|---|---|
| Conc. and FR Type | UL-94 HB | UL-94 HB |
| Blank | No Rating | No Rating |
| 2.5% EDA-DOPO | HF-1 | HF-2 |
| 5% EDA-DOPO | HF-1 | HF-2/HF1 * (14/6) |
| 7.5% EDA-DOPO | HF-1 | HF-2/HF1 ** (11/14) |
| 2.5% ETA-DOPO | HF-1 | HF-2 |
| 5% ETA-DOPO | - | HF-1 |
| 7.5% ETA-DOPO | - | HF-1 |
| 2.5% EG-DOPO | HF-1 | HF-2 |
| 5% EG-DOPO | - | HF-1 |
| 7.5% EG-DOPO | - | HF-1 |
| Type of Modified Formulations | FR Concentration | UL-94 HB | |
|---|---|---|---|
| Dispersion via ball mill (BDisp) | 2.5% EDA-DOPO | HF-2/HF1 * (7/3) | |
| 5% EDA-DOPO | HF-2/HF1 ** (4/6) | ||
| 6% EDA-DOPO | HF-1 | ||
| 7.5% EDA-DOPO | HF-1 | ||
| Use of surfactant sodium alkane Sulfonate (BE) | 2.5% EDA-DOPO | HF-2/HF1 ***(4/6) | |
| 5% EDA-DOPO | HF-1 | ||
| 7.5% EDA-DOPO | HF-1 | ||
| 0.5% PTFE (1 µm size) Normal dispersion 1 | 2.5% EDA-DOPO | HF-2 | |
| 5% EDA-DOPO | HF-2 | ||
| 7.5% EDA-DOPO | HF-1 | ||
| Use of PTFE and Ball mill dispersion (BP) | 1 µm, 0.5% | 5% EDA-DOPO | HF-1 |
| 4 µm, 0.5% | 5% EDA-DOPO | HF-1 | |
| 8 µm, 0.5% | 5% EDA-DOPO | HF-1 | |
| 40 µm, 0.5% | 5% EDA-DOPO | HF-2 | |
| Foams | Total Drops | Burning Drops | UL 94 HB Rating |
|---|---|---|---|
| Blank A | 132 ± 36 | 96 ± 37 | NA |
| Blank B | 106 ± 16 | 35 ± 35 | NA |
| 5% EDA-DOPO A | 56 ± 5 | 3 ± 2 | HF1 |
| 5% EDA-DOPO B | 101 ± 6 | 32 ± 7 | HF2 |
| 5% ETA-DOPO B | 61 ± 6 | 1 ± 0.3 | HF1 |
| 5% EDA-DOPO BE | 68 ± 13 | 7 ± 5 | HF1 |
| 5% EDA-DOPO BDisp | 60 ± 8 | 9 ± 5 | HF2/HF1(4/6) * |
| 5% EDA-DOPO BDisp 0.5% P1 | 38 ± 2 | 14 ± 7 | HF1 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Przystas, A.; Jovic, M.; Salmeia, K.A.; Rentsch, D.; Ferry, L.; Mispreuve, H.; Perler, H.; Gaan, S. Some Key Factors Influencing the Flame Retardancy of EDA-DOPO Containing Flexible Polyurethane Foams. Polymers 2018, 10, 1115. https://doi.org/10.3390/polym10101115
Przystas A, Jovic M, Salmeia KA, Rentsch D, Ferry L, Mispreuve H, Perler H, Gaan S. Some Key Factors Influencing the Flame Retardancy of EDA-DOPO Containing Flexible Polyurethane Foams. Polymers. 2018; 10(10):1115. https://doi.org/10.3390/polym10101115
Chicago/Turabian StylePrzystas, Agnieszka, Milijana Jovic, Khalifah A. Salmeia, Daniel Rentsch, Laurent Ferry, Henri Mispreuve, Heribert Perler, and Sabyasachi Gaan. 2018. "Some Key Factors Influencing the Flame Retardancy of EDA-DOPO Containing Flexible Polyurethane Foams" Polymers 10, no. 10: 1115. https://doi.org/10.3390/polym10101115







