Epoxy Vitrimers: The Effect of Transesterification Reactions on the Network Structure
Abstract
1. Introduction
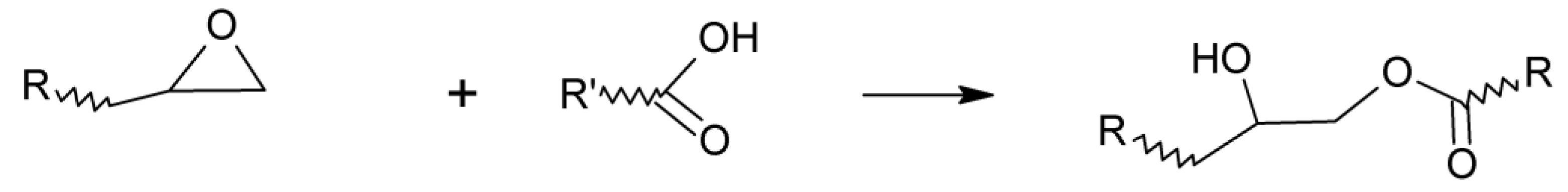
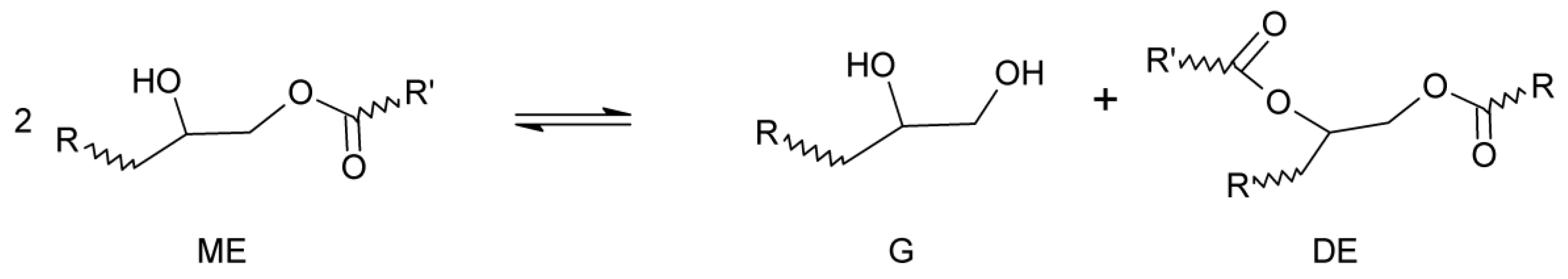
2. Equilibrium in Transesterification Reactions
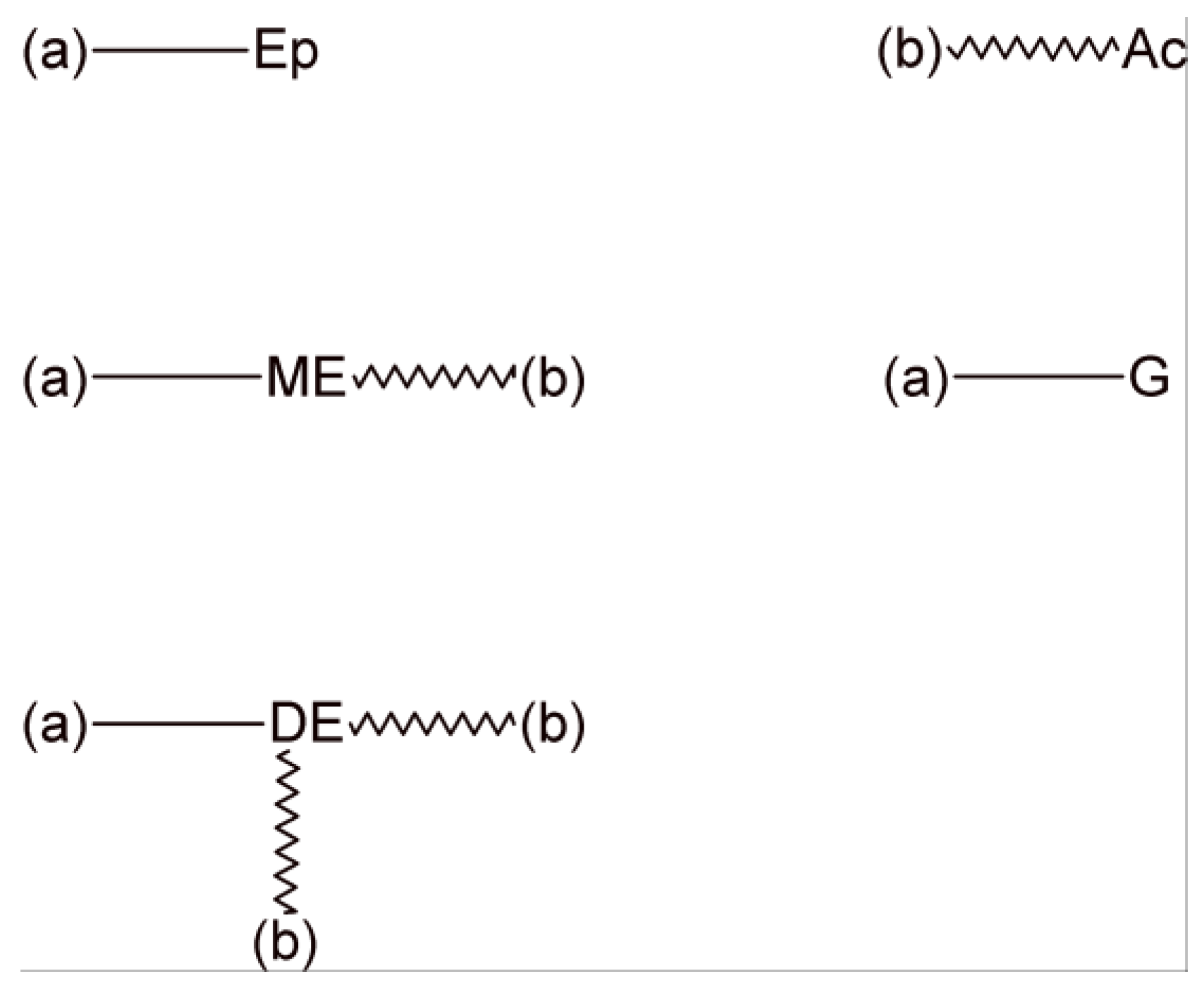
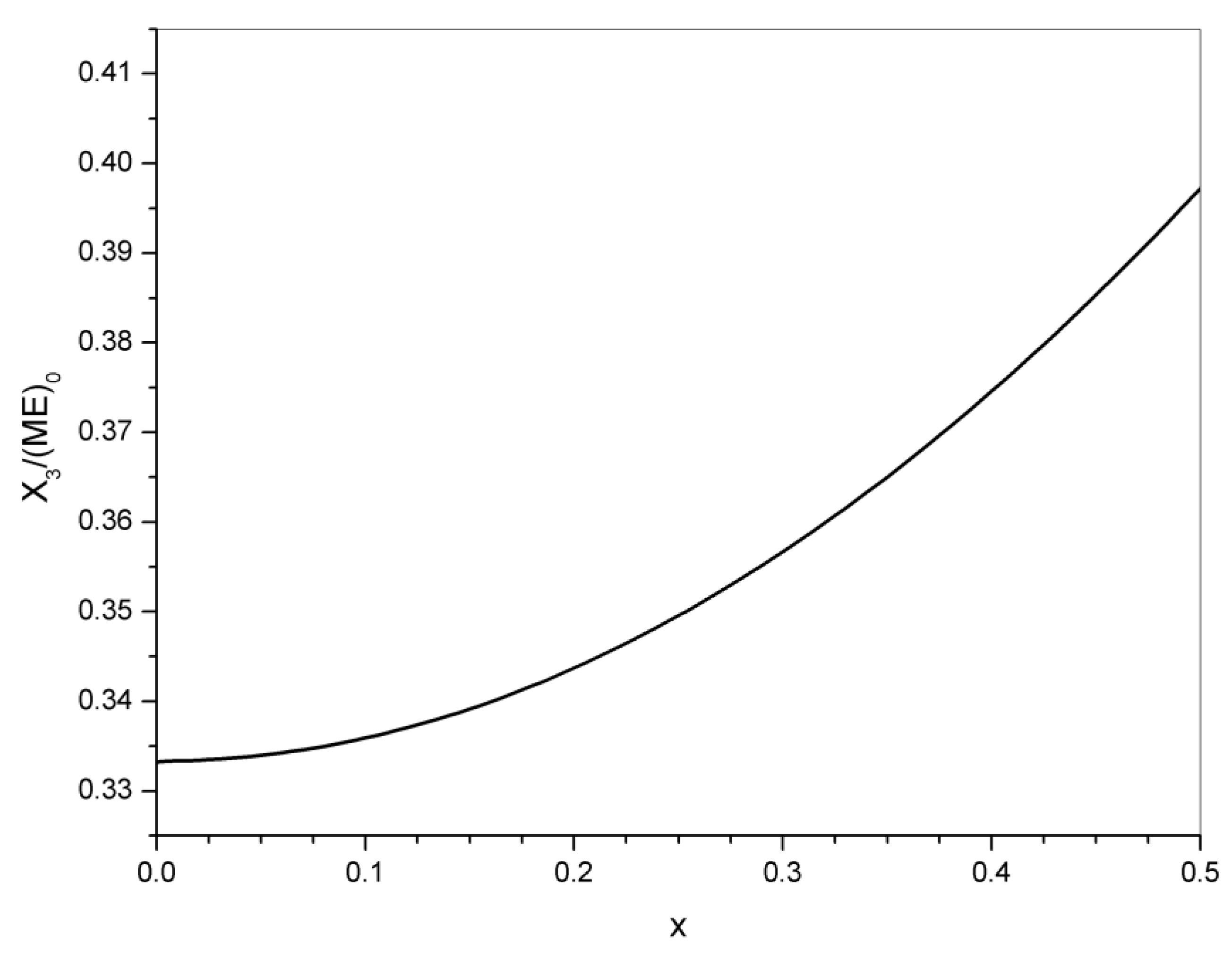
3. Statistical Analysis of Transesterification in a Stoichiometric A2 + B2 (Dicarboxylic Acid + Diepoxide) Formulation
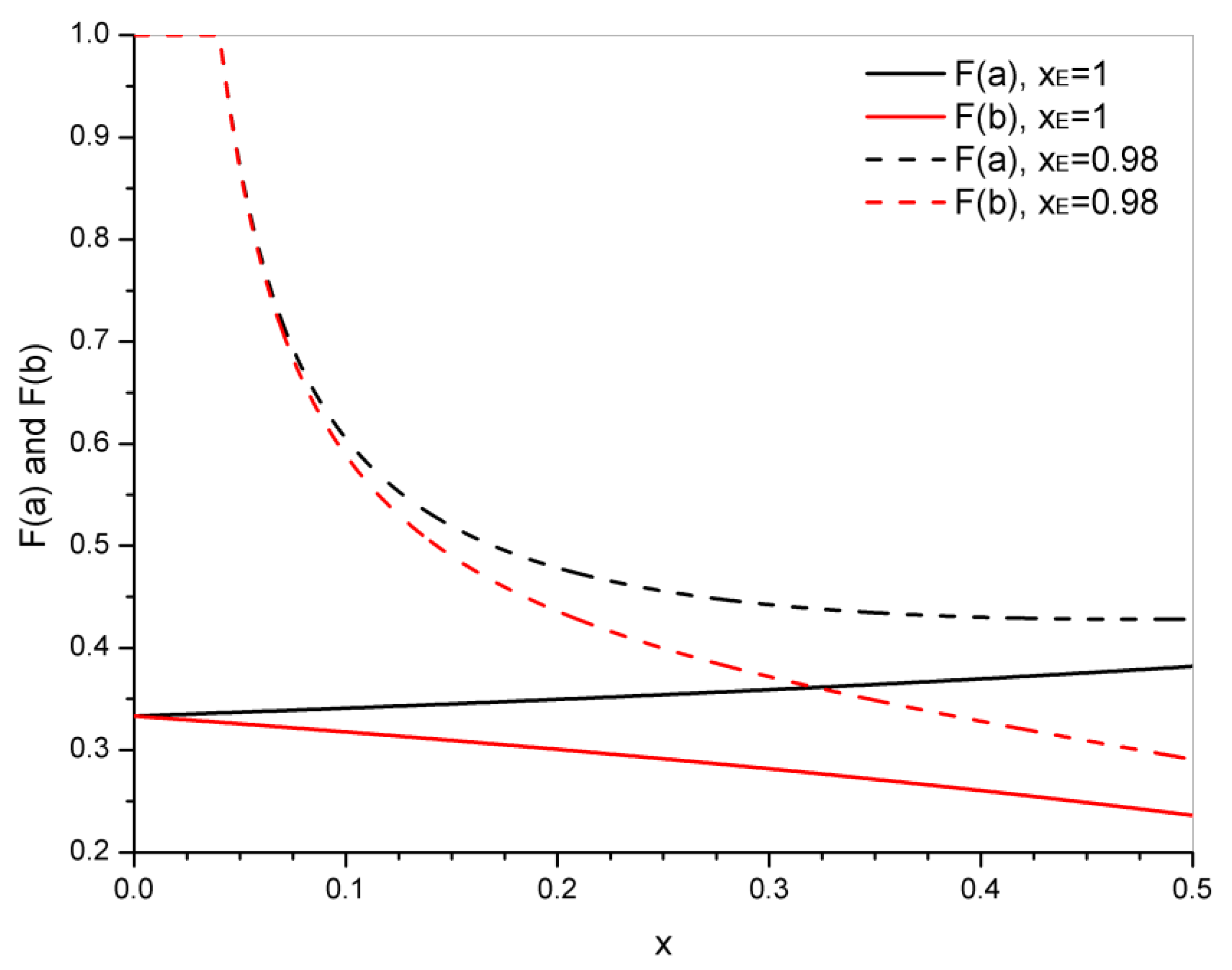
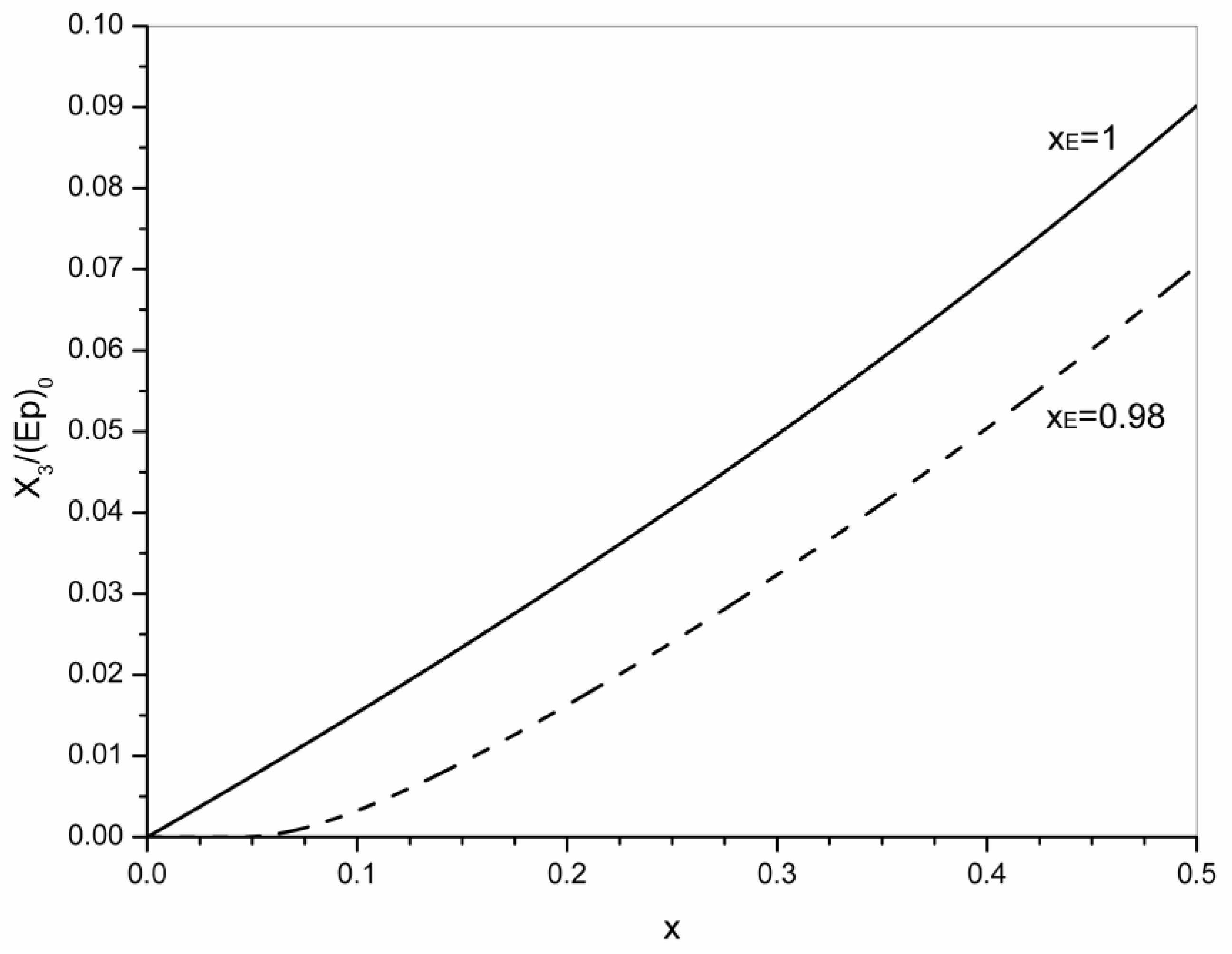
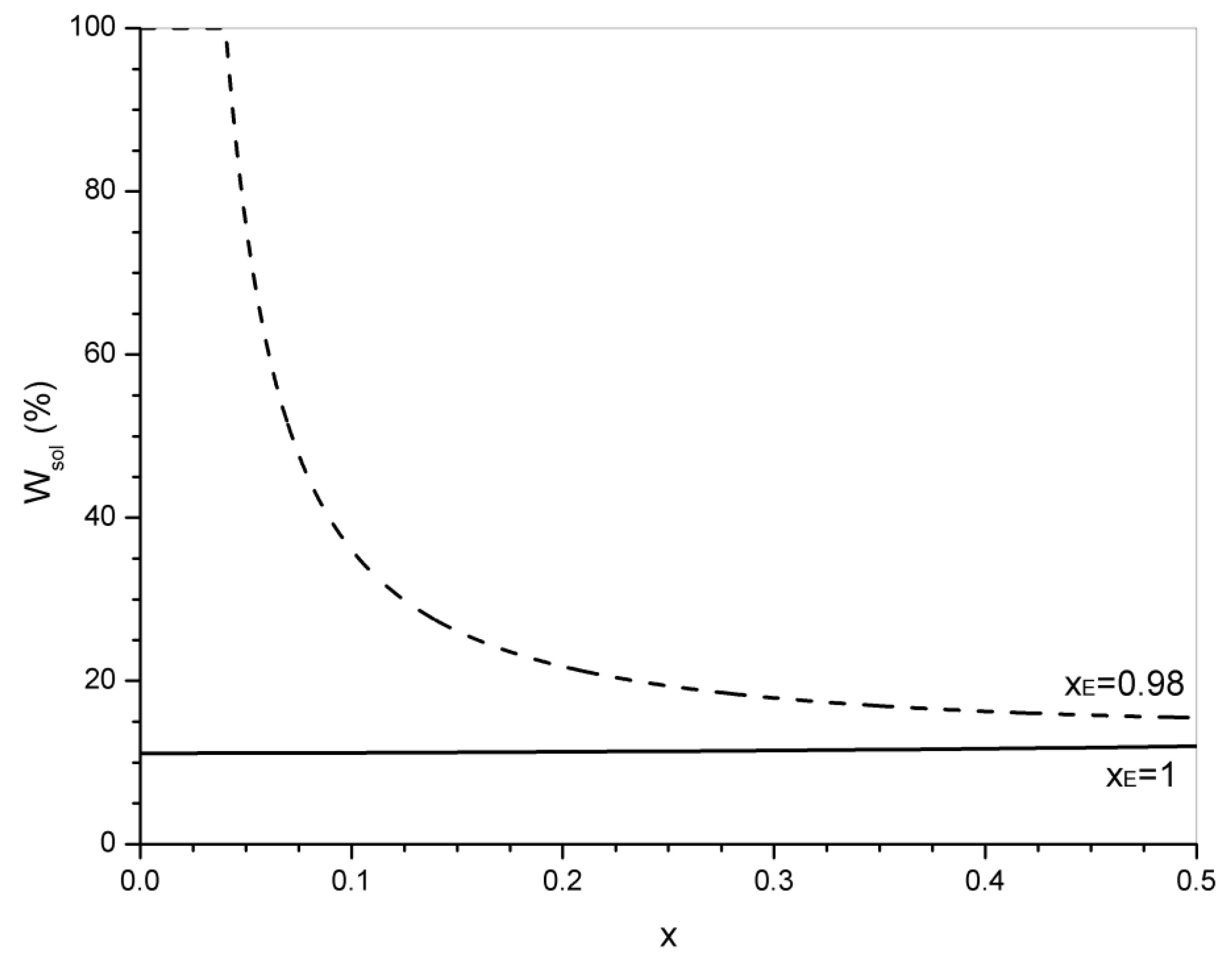
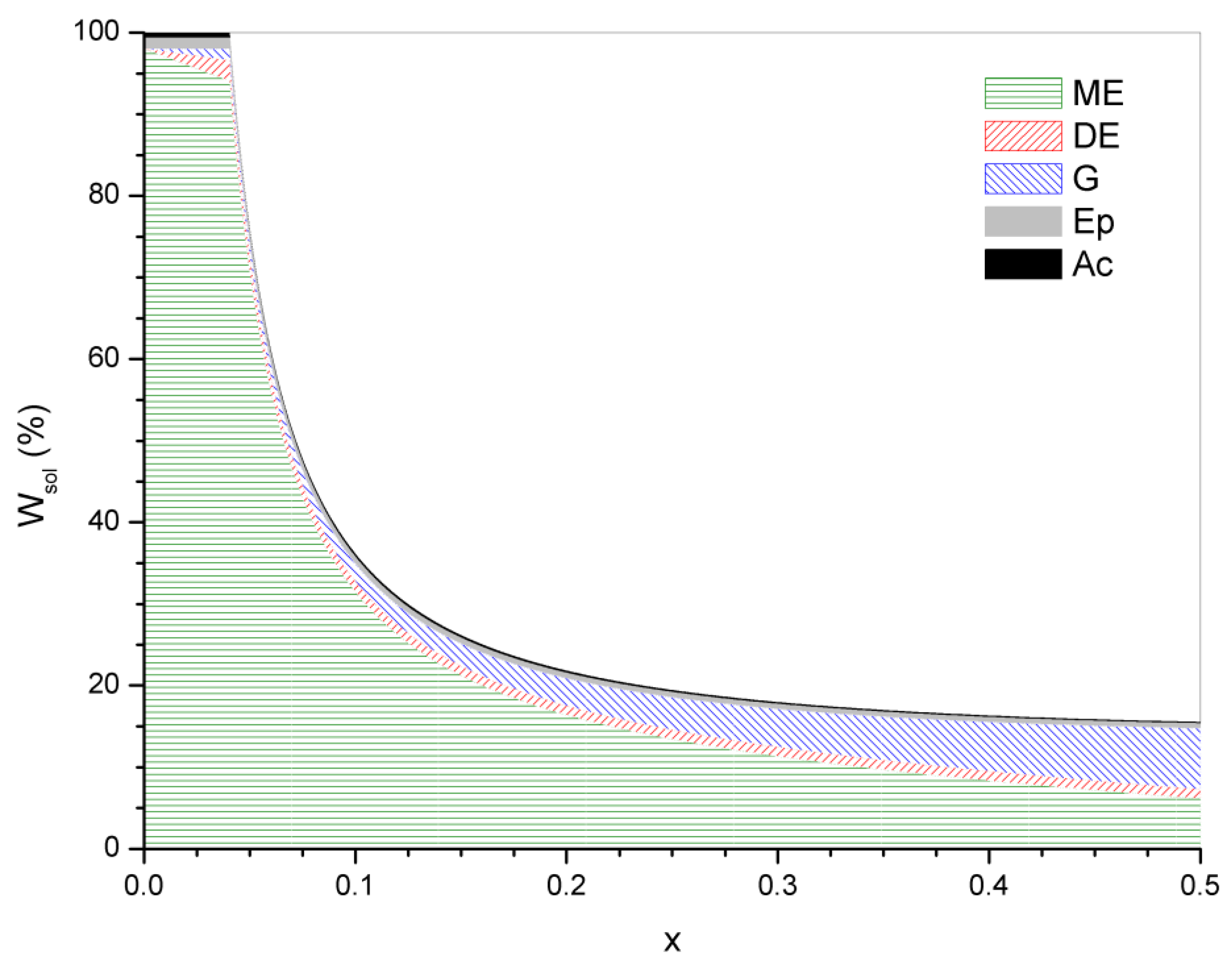
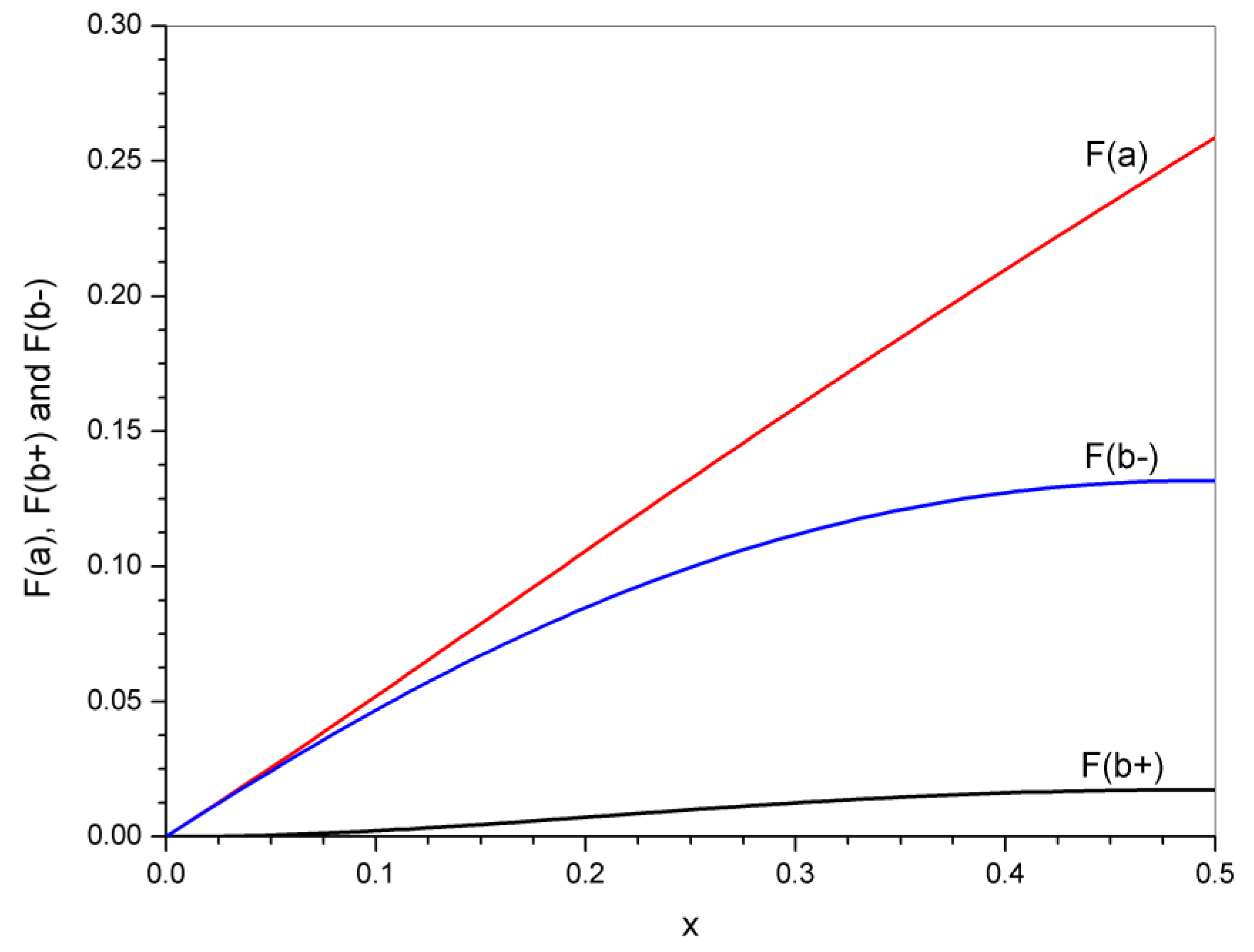
4. Statistical Analysis of Transesterification in Stoichiometric A3 + B2 (Tricarboxylic Acid + Diepoxide) Formulations
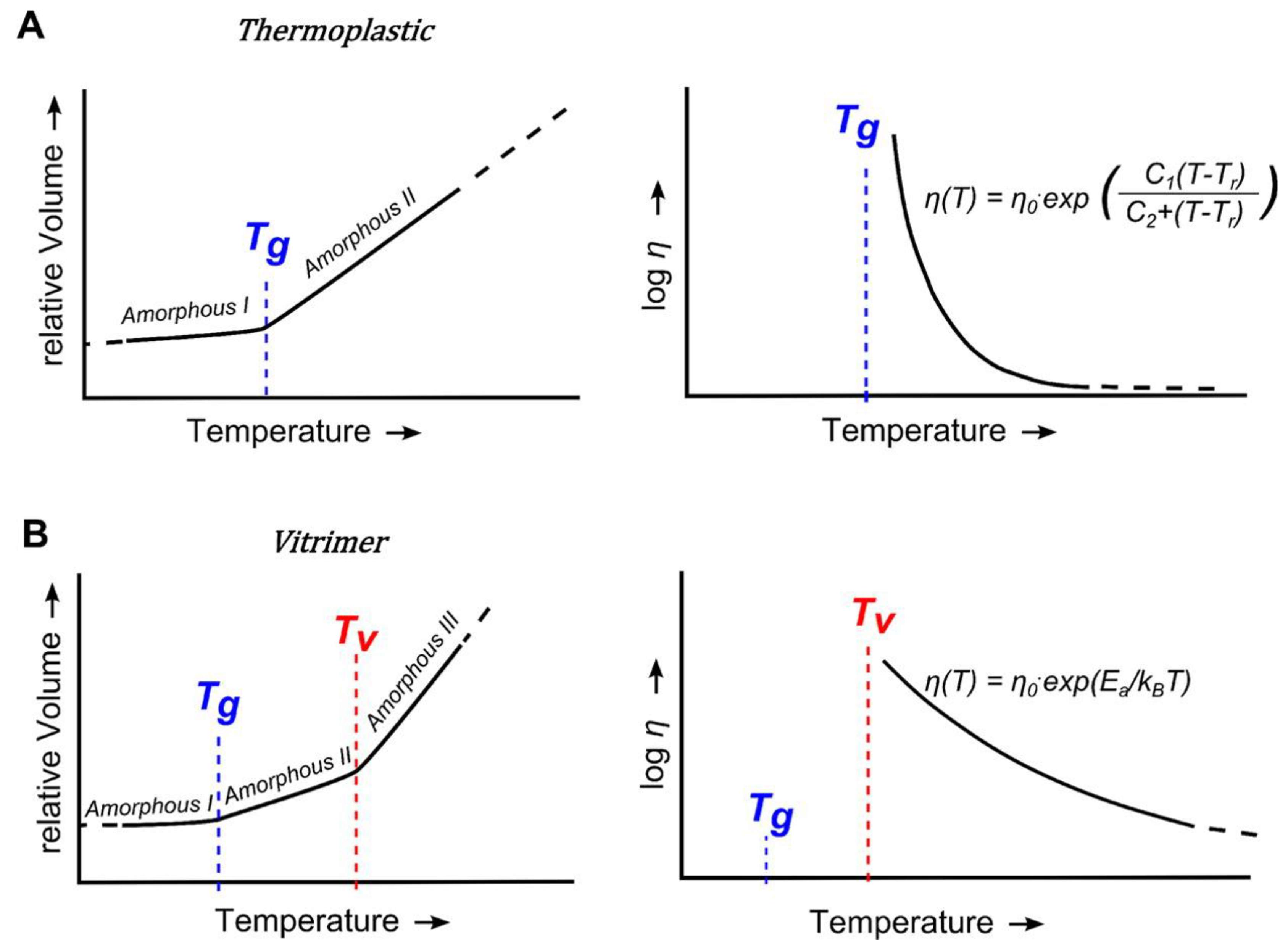
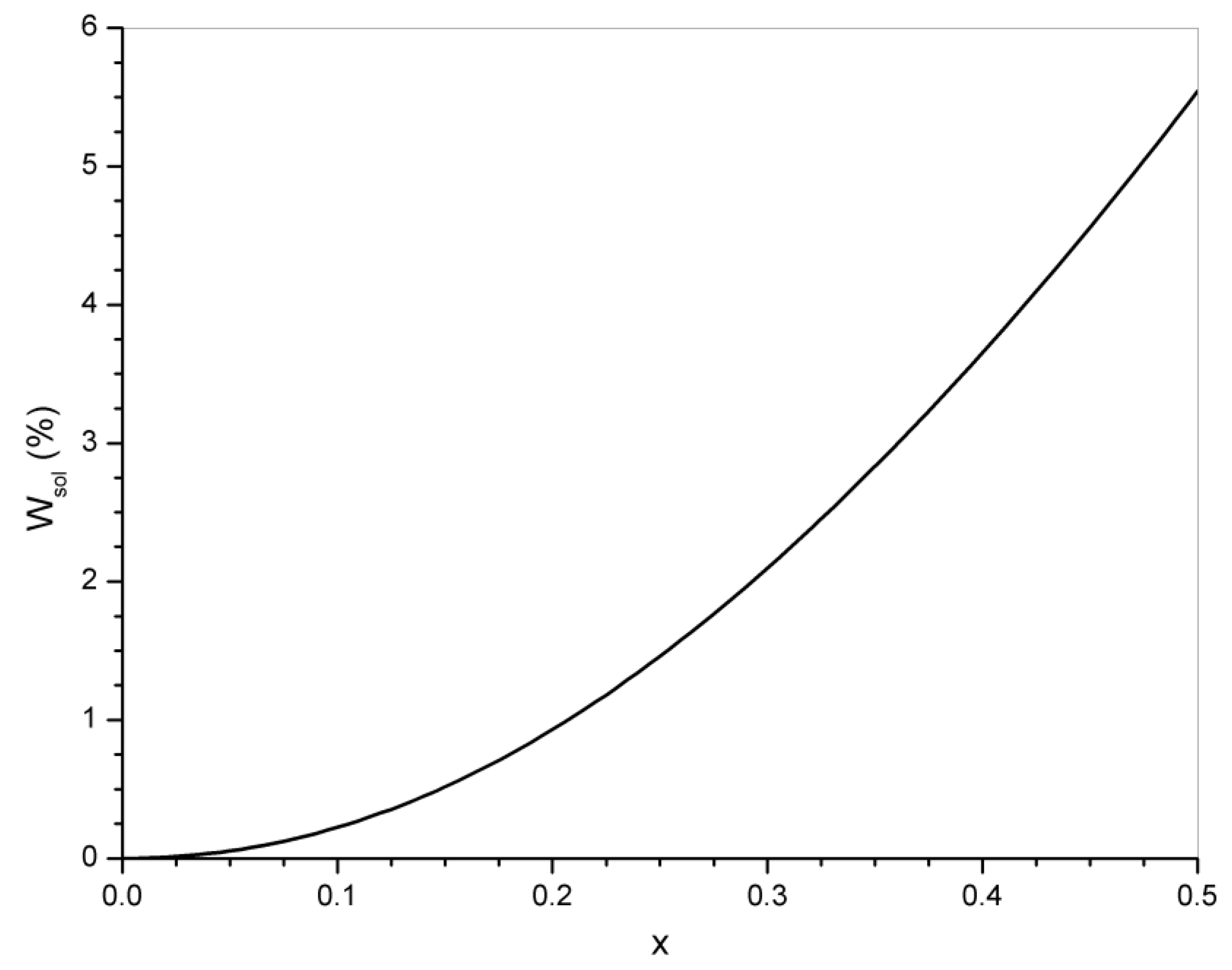
5. Significance of the Statistical Analysis Regarding the Synthesis and Use of Epoxy Vitrimers
6. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Flosbach, C.; Fugier, R. Epoxy functional acrylic polymers for high performance coating applications. In Epoxy Polymers: New Materials and Innovations; Pascault, J.-P., Williams, R.J.J., Eds.; Wiley-VCH: Weinheim, Germany, 2010; pp. 39–54. ISBN 978-3-527-32480-4. [Google Scholar]
- Matějka, L.; Pokomý, S.; Dušek, K. Network formation involving epoxide and carboxyl groups: Course of the model reaction monoepoxide-monocarbonic acid. Polym. Bull. 1982, 7, 123–128. [Google Scholar] [CrossRef]
- Montarnal, D.; Capelot, M.; Tournilhac, F.; Leibler, L. Silica-Like Malleable Materials from Permanent Organic Networks. Science 2011, 334, 965–968. [Google Scholar] [CrossRef] [PubMed]
- Capelot, M.; Montarnal, D.; Tournilhac, F.; Leibler, L. Metal-Catalyzed Transesterification for Healing and Assembling of Thermosets. J. Am. Chem. Soc. 2012, 134, 7664–7667. [Google Scholar] [CrossRef] [PubMed]
- Capelot, M.; Unterlass, M.M.; Tournilhac, F.; Leibler, L. Catalytic Control of the Vitrimer Glass Transition. ACS Macro Lett. 2012, 789–792. [Google Scholar] [CrossRef]
- Altuna, F.I.; Pettarin, V.; Williams, R.J.J. Self-healable polymer networks based on the cross-linking of epoxidised soybean oil by an aqueous citric acid solution. Green Chem. 2013, 15, 3360–3366. [Google Scholar] [CrossRef]
- Demongeot, A.; Mougnier, S.J.; Okada, S.; Soulié-Ziakovic, C.; Tournilhac, F. Coordination and catalysis of Zn 2+ in epoxy-based vitrimers. Polym. Chem. 2016, 7, 4486–4493. [Google Scholar] [CrossRef]
- Long, R.; Qi, H.J.; Dunn, M.L. Modeling the mechanics of covalently adaptable polymer networks with temperature-dependent bond exchange reactions. Soft Matter 2013, 9, 4083. [Google Scholar] [CrossRef]
- Smallenburg, F.; Leibler, L.; Sciortino, F. Patchy Particle Model for Vitrimers. Phys. Rev. Lett. 2013, 111. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.; Yu, K.; Mu, X.; Shi, X.; Wei, Y.; Guo, Y.; Qi, H.J. A molecular dynamics study of bond exchange reactions in covalent adaptable networks. Soft Matter 2015, 11, 6305–6317. [Google Scholar] [CrossRef] [PubMed]
- Sun, X.; Wu, H.; Long, R. Thermomechanics of a temperature sensitive covalent adaptable polymer with bond exchange reactions. Soft Matter 2016, 12, 8847–8860. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.; Yu, K.; Mu, X.; Wei, Y.; Guo, Y.; Qi, H.J. Molecular dynamics studying on welding behavior in thermosetting polymers due to bond exchange reactions. RSC Adv. 2016, 6, 22476–22487. [Google Scholar] [CrossRef]
- Yu, K.; Shi, Q.; Li, H.; Jabour, J.; Yang, H.; Dunn, M.L.; Wang, T.; Qi, H.J. Interfacial welding of dynamic covalent network polymers. J. Mech. Phys. Solids 2016, 94, 1–17. [Google Scholar] [CrossRef]
- Snijkers, F.; Pasquino, R.; Maffezzoli, A. Curing and viscoelasticity of vitrimers. Soft Matter 2017, 13, 258–268. [Google Scholar] [CrossRef] [PubMed]
- Yu, K.; Taynton, P.; Zhang, W.; Dunn, M.L.; Qi, H.J. Reprocessing and recycling of thermosetting polymers based on bond exchange reactions. RSC Adv. 2014, 4, 10108. [Google Scholar] [CrossRef]
- Lu, L.; Fan, J.; Li, G. Intrinsic healable and recyclable thermoset epoxy based on shape memory effect and transesterification reaction. Polymer 2016, 105, 10–18. [Google Scholar] [CrossRef]
- Yu, K.; Shi, Q.; Dunn, M.L.; Wang, T.; Qi, H.J. Carbon Fiber Reinforced Thermoset Composite with Near 100% Recyclability. Adv. Funct. Mater. 2016, 26, 6098–6106. [Google Scholar] [CrossRef]
- Shi, Q.; Yu, K.; Dunn, M.L.; Wang, T.; Qi, H.J. Solvent Assisted Pressure-Free Surface Welding and Reprocessing of Malleable Epoxy Polymers. Macromolecules 2016, 49, 5527–5537. [Google Scholar] [CrossRef]
- Yang, Y.; Pei, Z.; Zhang, X.; Tao, L.; Wei, Y.; Ji, Y. Carbon nanotube–vitrimer composite for facile and efficient photo-welding of epoxy. Chem. Sci. 2014, 5, 3486. [Google Scholar] [CrossRef]
- Chen, Q.; Yu, X.; Pei, Z.; Yang, Y.; Wei, Y.; Ji, Y. Multi-stimuli responsive and multi-functional oligoaniline-modified vitrimers. Chem. Sci. 2017, 8, 724–733. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z.; Wang, Q.; Wang, T. Dual-Triggered and Thermally Reconfigurable Shape Memory Graphene-Vitrimer Composites. ACS Appl. Mater. Interfaces 2016, 8, 21691–21699. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Pei, Z.; Li, Z.; Wei, Y.; Ji, Y. Making and Remaking Dynamic 3D Structures by Shining Light on Flat Liquid Crystalline Vitrimer Films without a Mold. J. Am. Chem. Soc. 2016, 138, 2118–2121. [Google Scholar] [CrossRef] [PubMed]
- Altuna, F.I.; Antonacci, J.; Arenas, G.F.; Pettarin, V.; Hoppe, C.E.; Williams, R.J.J. Photothermal triggering of self-healing processes applied to the reparation of bio-based polymer networks. Mater. Res. Express 2016, 3, 045003. [Google Scholar] [CrossRef]
- Pei, Z.; Yang, Y.; Chen, Q.; Wei, Y.; Ji, Y. Regional Shape Control of Strategically Assembled Multishape Memory Vitrimers. Adv. Mater. 2016, 28, 156–160. [Google Scholar] [CrossRef] [PubMed]
- Legrand, A.; Soulié-Ziakovic, C. Silica–Epoxy VitrimerNanocomposites. Macromolecules 2016, 49, 5893–5902. [Google Scholar] [CrossRef]
- Pei, Z.; Yang, Y.; Chen, Q.; Terentjev, E.M.; Wei, Y.; Ji, Y. Mouldable liquid-crystalline elastomer actuators with exchangeable covalent bonds. Nat. Mater. 2013, 13, 36–41. [Google Scholar] [CrossRef] [PubMed]
- Imbernon, L.; Norvez, S.; Leibler, L. Stress Relaxation and Self-Adhesion of Rubbers with Exchangeable Links. Macromolecules 2016, 49, 2172–2178. [Google Scholar] [CrossRef]
- Pritchard, R.H.; Redmann, A.-L.; Pei, Z.; Ji, Y.; Terentjev, E.M. Vitrification and plastic flow in transient elastomer networks. Polymer 2016, 95, 45–51. [Google Scholar] [CrossRef]
- Altuna, F.I.; Hoppe, C.E.; Williams, R.J.J. Shape memory epoxy vitrimers based on DGEBA crosslinked with dicarboxylic acids and their blends with citric acid. RSC Adv. 2016, 6, 88647–88655. [Google Scholar] [CrossRef]
- Lu, X.; Guo, S.; Tong, X.; Xia, H.; Zhao, Y. Tunable Photocontrolled Motions Using Stored Strain Energy in Malleable Azobenzene Liquid Crystalline Polymer Actuators. Adv. Mater. 2017, 29, 1606467. [Google Scholar] [CrossRef] [PubMed]
- Shi, Q.; Yu, K.; Kuang, X.; Mu, X.; Dunn, C.K.; Dunn, M.L.; Wang, T.; Jerry Qi, H. Recyclable 3D printing of vitrimer epoxy. Mater. Horiz. 2017, 4, 598–607. [Google Scholar] [CrossRef]
- Zhang, H.; Xu, X. Improving the transesterification and electrical conductivity of vitrimers by doping with conductive polymer wrapped carbon nanotubes. Compos. Part Appl. Sci. Manuf. 2017, 99, 15–22. [Google Scholar] [CrossRef]
- Chabert, E.; Vial, J.; Cauchois, J.-P.; Mihaluta, M.; Tournilhac, F. Multiple welding of long fiber epoxy vitrimer composites. Soft Matter 2016, 12, 4838–4845. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Schmidt, D.F.; Reynaud, E. Catalyst Selection, Creep, and Stress Relaxation in High-Performance Epoxy Vitrimers. Ind. Eng. Chem. Res. 2017, 56, 2667–2672. [Google Scholar] [CrossRef]
- Dell’Erba, I.E.; Williams, R.J.J. Homopolymerization of epoxy monomers initiated by 4-(dimethylamino)pyridine. Polym. Eng. Sci. 2006, 46, 351–359. [Google Scholar] [CrossRef]
- Puig, J.; Hoppe, C.E.; Fasce, L.A.; Pérez, C.J.; Piñeiro-Redondo, Y.; Bañobre-López, M.; López-Quintela, M.A.; Rivas, J.; Williams, R.J.J. SuperparamagneticNanocomposites Based on the Dispersion of Oleic Acid-Stabilized Magnetite Nanoparticles in a Diglycidylether of Bisphenol A-Based Epoxy Matrix: Magnetic Hyperthermia and Shape Memory. J. Phys. Chem. C 2012, 116, 13421–13428. [Google Scholar] [CrossRef]
- Vázquez Barreiro, E.C.; Fraga López, F.; Jover, A.; Meijide, F.; Rodríguez, E.; Vázquez Tato, J. Paramagneticepoxyresin. Express Polym. Lett. 2017, 11, 60–72. [Google Scholar] [CrossRef]
- Dušek, K.; Matějka, L. Transesterification and Gelation of Polyhydroxy Esters Formed from Diepoxides and Dicarboxylic Acids. In Rubber-Modified Thermoset Resins; Riew, C.K., Gillham, J.K., Eds.; Advances in Chemistry Series; American Chemical Society: Washington, DC, USA, 1984; Volume 208, pp. 15–26. ISBN 978-0-8412-0828-5. [Google Scholar]
- Pascault, J.-P.; Sautereau, H.; Verdu, J.; Williams, R.J.J. Thermosetting Polymers, 1st ed.; Marcel Dekker: New York, NY, USA, 2002; ISBN 0-8247-0670-6. [Google Scholar]
- Riccardi, C.C.; Williams, R.J.J. Statistical structural model for the build-up of epoxy-amine networks with simultaneous etherification. Polymer 1986, 27, 913–920. [Google Scholar] [CrossRef]
- Williams, R.J.J.; Riccardi, C.C.; Dušek, K. Build-up of polymer networks by initiated polyreactions: 5. Comparison of fragment vs. cluster approaches to derive postgel parameters for postetherification in diamine-diepoxide curing. Polym. Bull. 1991, 25, 231–237. [Google Scholar] [CrossRef]
- Fernàndez-Francos, X. Theoretical modeling of the effect of proton donors and regeneration reactions in the network build-up of epoxy thermosets using tertiary amines as initiators. Eur. Polym. J. 2014, 55, 35–47. [Google Scholar] [CrossRef]
- Williams, R.J.J.; Riccardi, C.C.; Dušek, K. Build-up of polymer networks by initiated polyreactions: 3. Analysis of the fragment approach to the living polymerization type of build-up. Polym. Bull. 1987, 17, 515–521. [Google Scholar] [CrossRef]
- Moschiar, S.M.; Piacentini, C.A.L.; Williams, R.J.J. Analysis of the build-up of polyurethane networks from toluenediisocyanate and castor oil considering intra-molecular reactions. Polymer 1988, 29, 914–918. [Google Scholar] [CrossRef]
- Williams, R.J.J. Statistics of free-radical polymerizations revisited using a fragment approach. 1. Bifunctional monomers. Macromolecules 1988, 21, 2568–2571. [Google Scholar] [CrossRef]
- Williams, R.J.J.; Vallo, C.I. Statistics of free-radical polymerizations revisited using a fragment approach. 2. Polyfunctional monomers. Macromolecules 1988, 21, 2571–2575. [Google Scholar] [CrossRef]
- Williams, R.J.J.; Vázquez, A.; Pascault, J.P. Gelation in the cyclotrimerization of dicyanates considering substitution effects. Polym. Bull. 1992, 28, 219–225. [Google Scholar] [CrossRef]
- Georjon, O.; Galy, J.; Pascault, J.P.; Williams, R.J.J. Statistical calculations of the network build-up in the postgel stage of A2-cyclotrimerization. Polym. Bull. 1993, 31, 503–509. [Google Scholar] [CrossRef]
- Riccardi, C.C.; Williams, R.J.J. Modeling strategy for systems with both stepwise and chainwise chemistry revisited. The directionality effect on the build-up of the network structure. J. Polym. Sci. Part B Polym. Phys. 1993, 31, 389–393. [Google Scholar] [CrossRef]
- Fernàndez-Francos, X.; Ramis, X.; Serra, À. From curing kinetics to network structure: A novel approach to the modeling of the network buildup of epoxy-anhydride thermosets. J. Polym. Sci. Part Polym. Chem. 2014, 52, 61–75. [Google Scholar] [CrossRef]
- Fernàndez-Francos, X.; Serra, À.; Ramis, X. Comparative analysis of stochastic network build-up methods for the curing of epoxy–anhydride thermosets. Eur. Polym. J. 2014, 53, 22–36. [Google Scholar] [CrossRef]
- Fernández-Francos, X.; Ramis, X. Structural analysis of the curing of epoxy thermosets crosslinked with hyperbranchedpoly(ethyleneimine)s. Eur. Polym. J. 2015, 70, 286–305. [Google Scholar] [CrossRef]
- Fernández-Francos, X.; Konuray, A.-O.; Belmonte, A.; De la Flor, S.; Serra, À.; Ramis, X. Sequential curing of off-stoichiometric thiol–epoxy thermosets with a custom-tailored structure. Polym. Chem. 2016, 7, 2280–2290. [Google Scholar] [CrossRef]
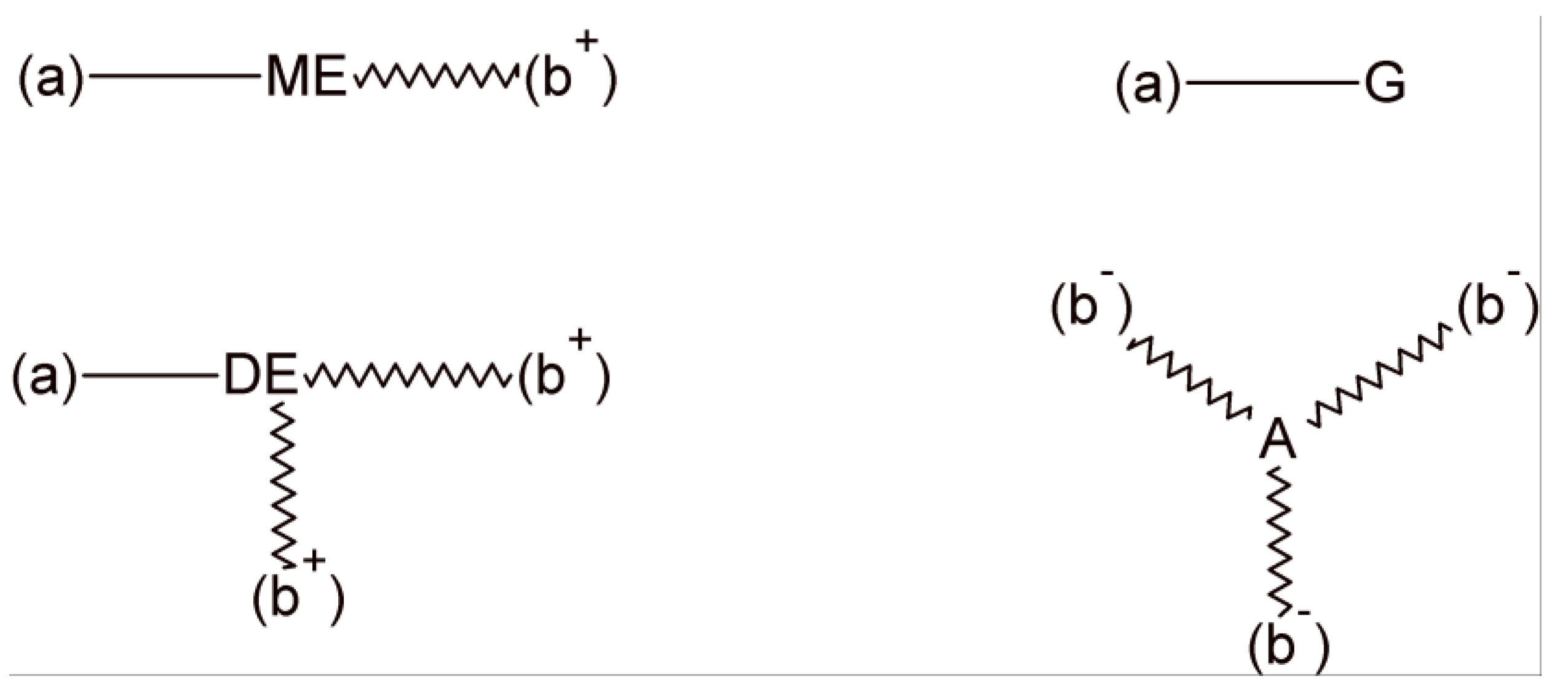
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Altuna, F.I.; Hoppe, C.E.; Williams, R.J.J. Epoxy Vitrimers: The Effect of Transesterification Reactions on the Network Structure. Polymers 2018, 10, 43. https://doi.org/10.3390/polym10010043
Altuna FI, Hoppe CE, Williams RJJ. Epoxy Vitrimers: The Effect of Transesterification Reactions on the Network Structure. Polymers. 2018; 10(1):43. https://doi.org/10.3390/polym10010043
Chicago/Turabian StyleAltuna, Facundo Ignacio, Cristina Elena Hoppe, and Roberto Juan José Williams. 2018. "Epoxy Vitrimers: The Effect of Transesterification Reactions on the Network Structure" Polymers 10, no. 1: 43. https://doi.org/10.3390/polym10010043
APA StyleAltuna, F. I., Hoppe, C. E., & Williams, R. J. J. (2018). Epoxy Vitrimers: The Effect of Transesterification Reactions on the Network Structure. Polymers, 10(1), 43. https://doi.org/10.3390/polym10010043





