Abstract
In this study, four additives—montmorillonite, activated carbon, and the layered double hydroxides (LDHs), Mg2Fe–LDH and Mg2Al–LDH—were tested for their ability to promote surfactin production in a Bacillus subtilis ATCC 21332 culture. Among these tested materials, the addition of 4 g/L of the Mg-Fe LDH, which featured an Mg/Fe molar ratio of 2:1, produced the highest surfactin yield of 5280 mg/L. During the time course of B. subtilis cultivation with the added LDH, two phases of cell growth were evident: Growth and decay. In the growth phase, the cells grew slowly and secreted a high amount of surfactin; in the decay phase, the cells degraded rapidly. The production in the presence of the Mg2Fe–LDH had three characteristics: (i) High surfactin production at low biomass, indicating a high specific surfactin yield of 3.19 g/g DCW; (ii) rapid surfactin production within 24 h, inferring remarkably high productivity (4660 mg/L/d); and (iii) a lower carbon source flux to biomass, suggesting an efficient carbon flux to surfactin, giving a high carbon yield of 52.8%. The addition of Mg2Fe–LDH is an effective means of enhancing surfactin production, with many potential applications and future industrial scale-up.
1. Introduction
Biosurfactants are amphipathic molecules produced by microorganisms [1,2,3] with the capability of decreasing surface and interfacial tension [4]. Depending on their chemical composition and their producing organism, biosurfactants can possess high biodegradability, low toxicity, ecological acceptability, and high efficiency. Accordingly, they have been investigated as possible alternatives to chemical surfactants [5,6]. Bacillus spp., bacterial strains of complicated physiological diversity, can be used to produce many bioactive peptides with potential biotechnological and biopharmaceutical applications. Among these peptides, the lipopeptides that feature an alkyl group and a circular peptide group are the most popular biosurfactants [7]; these materials include surfactins [8,9,10], iturins [11,12], and fengycins [13].
The surfactin produced by B. subtilis is one of the strongest biosurfactants available [7]. Its chemical composition is that of a cyclic lipopeptide (comprising seven amino acids) with a 12 to 19-carbon atom hydrophobic fatty acid chain [14]. Surfactin can lower the surface tension of water to 27 mN/m even when its concentration is as low as 0.005% [7,10,15,16], suggesting its great potential applicability. Nevertheless, the high expense and low yield of surfactin production have limited its commercial use. Yeh et al. found that limiting the concentration of the carbon source (glucose) affected the surfactin production mediated by B. subtilis [17]. Davis et al. observed the highest production of surfactin when ammonium nitrate was the nitrogen source during B. subtilis cultivation in a defined medium [18]. Sen et al. noted that the ratio of Mn and Fe mineral salts in the medium was a factor affecting the production of surfactin [19]. Wei and Chu found that the yield of surfactin increased dramatically, over those obtained using genetic strains, when employing 0.01 mM Mn2+ [20]. Furthermore, Wei et al. employed an iron-enriched (4 mM Fe2+) minimal salt medium to produce 3000 mg/L of surfactin [21]. Moreover, some of these studies revealed that the addition of solid additives (e.g., activated carbon (AC) or expanded clay) could increase surfactin production significantly. For example, Yeh et al. added AC and increased the yield of surfactin to 3600 mg/L [17].
Layered double hydroxides (LDHs)—also known as anionic clays—comprise cationic brucite-like layers with exchangeable interlayer anions [22]. Because a positive ionic charge appears on the surface layer, many types of molecules can be intercalated into LDHs [23,24,25,26,27]. Several methods have been developed to widen the layered gallery, with globular macromolecules as intercalating agents [28,29]. Conterosito et al. intercalated various pharmaceutics drugs and cosmetic sunscreen into Mg-Al_LDH and Zn-Al_LDH. They revealed that different bioactive molecules could interact with inorganic LDH and demonstrated the relationship between the molecular length and an enlarged interlayer spacing [30]. Toson et al. showed the intercalation of organic molecules into the LDH interlayer by the liquid-assisted grinding method. The intercalation mechanism for layer widening with intercalated organic molecules was investigated [31]. Choy et al. employed supramolecular inorganic species (e.g., nanoscale Mg-Al LDH) as biomolecule reservoirs that could be used for gene and drug delivery [23,32]. Nevertheless, few studies have focused on using LDHs as additives for microbial cultivation. Kan et al. prepared Mg2Al–LDH and investigated its effect as an additive on surfactin production and surfactin intercalation [33]. When considering the application of the surfactin-intercalated LDH as a slow release bio-pesticide, however, this aluminum salt was prohibited from field tests [34]. For agricultural applications, iron salts are generally considered less toxic. Therefore, in this present study, we prepared several Mg-Fe LDH derivatives with potentially greater practicality. We tested the effects of their addition on the production of surfactin from a B. subtilis culture. To our surprise, replacing the additive to Mg2Fe–LDH had an extraordinary effect on the surfactin production. Accordingly, we examined various MgnFe–LDH (n = 1, 2, 3) compositions and concentrations to determine the optimal conditions for surfactin production. In addition, we examined the time course of the production in the optimal culture. An extra low biomass of cells yielded the highest surfactin production. This result was quite different from that obtained after the addition of Mg2Al–LDH. Furthermore, we compared the effects of the LDHs with those of other additives (e.g., montmorillonite (MMT), AC), and determined the conditions for the highest production of surfactin through quantitative analysis. Herein, we also suggest possible reasons for the enhancement of surfactin production mediated by LDHs.
2. Materials and Methods
2.1. Chemicals and Reagents
Al(NO3)3·9H2O, Fe(NO3)3·9H2O, and Mg(NO3)2·6H2O were purchased from SHOWA, USA. MMT was obtained from Alfa Aesar, USA. The AC was obtained from China Activated Carbon (Taipei, Taiwan); it had a diameter of 3 to 4 mm, a height of 9 mm, and a specific surface area of 1200 m2/g, and was prepared from bituminous coal with an iodine number of 1150 mg/g. Surfactin (≥98%, Sigma–Aldrich, Missouri, MO, USA) was used as the standard. All solvents and other chemicals were of analytical grade.
2.2. Microorganisms and Culture Conditions
The strain, B. subtilis ATCC 21332, was obtained from Professor Wei Yu-Hong of Yuan Ze University. This strain was kept on a nutrient-agar plate at 30 °C. For cultivation, its seed medium comprised 1% glucose, 0.5% yeast extract, 1% peptone, and 1% NaCl. The seed culture was performed in Erlenmeyer flasks (500 mL) containing the seed medium (100 mL) inoculated with two loops of cells. The cultivation was conducted at 200 rpm and 30 °C for 12 h. The main shake-flask culture was conducted in an Erlenmeyer flask (500 mL) containing the main medium (100 mL) comprising 10 g/L sucrose, 5 g/L (NH4)2SO4, 5.67 g/L Na2HPO4, 4.08 g/L KH2PO4, 0.2 g/L MgSO4·7H2O, and 0.57 g/L FeSO4·7H2O. The media were sterilized (121 °C, 20 min); the carbon source was autoclaved separately. The medium (90 mL) was inoculated with the seed broth (10 mL). The flasks were incubated on a rotary shaker (200 rpm, 30 °C, 5 days). When testing additives, MMT, AC, and the prepared LDHs were added (2 g/L) to the culture medium at the beginning of the culture process.
2.3. Mg2Al–LDH and Mg2Fe–LDH
Mg2Al-NO3–LDH and Mg2Fe-NO3–LDH were prepared through co-precipitation, as described previously [27]. To prepare the Mg2Al–LDH sample, Mg(NO3)2·6H2O (120 g) and Al(NO3)3·9H2O (90 g) were dissolved in deionized H2O (1 L). To prepare the Mg2Fe–LDH sample, Mg(NO3)2·6H2O (169 g) and Fe(NO3)3·9H2O (134 g) were mixed in deionized H2O (1 L). To prepare samples with Mg/Fe molar ratios of 1.0 and 3.0, appropriate amounts of Mg(NO3)2·6H2O and Fe(NO3)3·9H2O were used. Each aqueous solution was stirred vigorously at 80 °C while purging with nitrogen gas. When preparing the Mg2Al–LDH sample, the pH was maintained at 10 ± 0.2 by adding 4 N NaOH in portions. For the Mg2Fe-NO3–LDH sample, the pH was adjusted to 9.5 ± 0.2 by using a mixture of NaOH and K3[Fe(CN)6], prepared based on the following compositions: [OH–]/([Mg2+] + [Fe3+]) = 1.6 and [[Fe(CN)6]3–]/[Fe3+] = 3. The suspension that formed was stirred at 80 °C for 24 h. The obtained precipitates—white Mg2Al–LDH and dark-red Mg2Fe–LDH—were filtered off and washed (deionized H2O). The filtered cakes were lyophilized (freeze-drying). The dried LDHs were characterized using x-ray diffraction (XRD; PANalytical, X’Pert PRO MRD, Almelo, Netherlands) and attenuated total reflectance Fourier transform infrared (ATR-FTIR) spectroscopy (Thermo Scientific, Nicolet iS50 FTIR, Madison, WI, USA).
2.4. Quantitative Analysis
To study the effects of LDH addition, the following quantitative terms are defined. The surfactin yield (mg/L) is expressed by the volumetric concentration. The carbon source yield is defined as:
The productivity is defined as:
The specific yield is defined as:
where P represents the surfactin concentration, S is the carbon source concentration, X is the concentration of biomass dried cells, and t is the duration of cultivation. All concentrations are expressed herein on a volumetric basis.
2.5. Assays
The surfactin concentration was measured using a modified approach called salt-assisted homogeneous liquid-liquid extraction via high-performance liquid chromatography (HPLC) [33,35]. The culture broth (1 mL) was subjected to a centrifugation (3200 g, 3 min, 4 °C) to remove the solid pellets. The supernatant was mixed with MeCN (0.5 mL) and ammonium sulfate (0.8 g) and subjected to vigorous stirring for 1 min, and then centrifuged (3200 g, 3 min). The supernatant was filtered (0.22 μm) to obtain the sample for injection. HPLC analysis was performed under the following conditions: A reversed-phase C-18A column (5 mm, 18 mm × 100 mm BDS-Hypersil, Thermo Fisher Scientific, Waltham, MA, USA); a mobile phase comprising CF3CO2H, MeCN, and deionized H2O (0.1:400:100); a flow rate of 1.0 mL/min; an injection sample volume of 20 μL; and a UV–Vis detector (JASCO, Tokyo, Japan) operated at 220 nm. A standard curve was constructed using a freshly prepared solution of surfactin (Sigma). The chromatogram of the standard (supplemental Figure S1) revealed various ratios of the surfactin isoforms A–F. The surfactin produced using B. subtilis ATCC 21332 featured the same surfactin isoforms A–F at various ratios. In the surfactin assay, the whole isoforms were measured and added up for quantitative calculation. To analyze the cells’ dried weight (CDW), 5 mL of the broth sample was subjected to centrifugation (12,000 g, 10 min) to obtain a pellet. Distilled H2O (5 mL) was added to the pellet; after adjusting to pH 2.0, the sample was vigorously stirred (1 min). The mixture was centrifuged (12,000 g, 5 min). The pellet obtained was dissolved in distilled H2O (5 mL); the pH was adjusted to 7.0 and the mixture was again subjected to centrifugation. The obtained pellet was washed with distilled H2O (2 × 5 mL), dried (80 °C, 12 h), and then weighed. The basal spacing of the LDH was determined using a Shimadzu SD-D1 X-ray diffractometer with a Cu target (scanning rate: 1°/min). The basal spacing was estimated using the Bragg equation (nλ = 2dsinθ).
2.6. Statistical Analysis
Multiple flasks were run concurrently. Three flasks were employed each time for daily sampling. Each data point is expressed as a mean plus standard deviation. The Tukey test was applied for the comparison of results (p ≤ 0.05).
3. Results and Discussion
3.1. Preparation of MgFe–LDH
The addition of a small quantity of a solid carrier (AC or expanded clay) has been claimed as an effective approach toward increasing surfactin production [17]. LDHs are layered anionic exchange substances that have been intercalated with various macromolecules for the purpose of their slow release [36,37]. The addition of Mg2Al–LDH LDH to a surfactin production fermentation system involving B. subtilis incubation revealed that surfactin could indeed intercalate into the LDH layer gallery to form a surfactin–LDH complex; this phenomenon occurred with a significant increase in the production of surfactin [33]. In consideration of a slow-release composite for agricultural use, Mg2Al–LDH would be inappropriate for field trials. For this study, therefore, we prepared Mg2Fe–LDH instead. We examined the effect of adding this iron salt LDH to B. subtilis cultivation to study whether it, too, would promote surfactin production. The prepared Mg2Fe–LDH was subjected to XRD and ATR-FTIR spectroscopic analysis. These analyses revealed an Mg2Fe–LDH layer spacing of 7.8 Å at a value of 2θ of 11.3°, derived from the calculation of Bragg’s equation (Figure 1a), and a typical adsorption peak (1381 cm−1) for NO3– anions within the prepared LDH (data not shown). In addition, to confirm the interaction between LDH and bacterial cells, the LDH after the cultivation was collected and subjected to XRD analysis. The result in Figure 1b shows that the collected LDH did vary its 2θ from the original 11.3° to 8.3°, indicating a d-spacing of 10.8 Å. The original XRD peak with a d-spacing of 7.8 Å completely disappeared. The enlarged spacing was likely due to the LDH interaction with surfactin molecules. The isoelectric point (IEP) of surfactin is around pH 5, and the fermentation process while applying LDH to the cultivation was around pH 7.4. The pH higher than the IEP would allow the surfactin to possess a negative charge, giving the chance of anion exchange for LDH intercalation. Besides, the interlayer spacing expansion of LDH might be ascribed not only to the surface interaction of surfactin intercalation but also the combination of water and other anion molecules in the culture medium into the Mg2Fe–LDH interlayer.
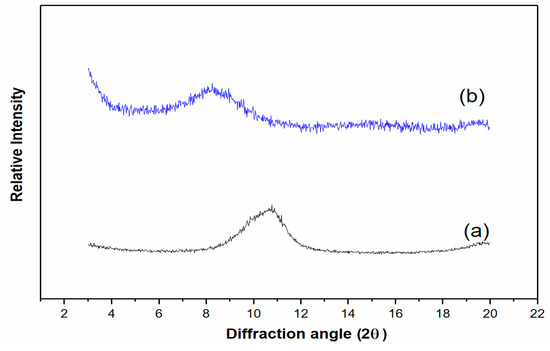
Figure 1.
XRD pattern of (a) pristine Mg2Fe–LDH and (b) Mg2Fe–LDH collected after fermentation.
3.2. Effect of Solid Additives on Surfactin Production
As reported previously, the addition of some solid additives can enhance surfactin production [17]. For this present study, four solid additives—MMT, AC, and two LDHs—were prepared and added respectively to the B. subtilis culture medium; the medium prepared without any additives was used as the control during the five-day fermentation. The surfactin production increased when the culture medium contained each of these solid additives, relative to the control. The addition of MMT, AC, and the two LDHs (2 g/L) resulted in surfactin yields that had increased by 2.0-, 3.0-, 3.8-, and 4.5-fold, respectively, when compared with the control (Figure 2). It is noteworthy that the AC with the alkaline characterization might lead to surfactin linearization and surfactin binding on the AC surface, which may be an underestimation of the actual production. Thus, the LDHs were the most effective carriers for enhanced surfactin production in a culture of B. subtilis ATCC 21332. Furthermore, the amount of surfactin produced in the presence of Mg2Fe–LDH was more than that produced in the presence of Mg2Al–LDH. Indeed, Mg2Fe–LDH had an extraordinary stimulatory effect on promoting surfactin production.
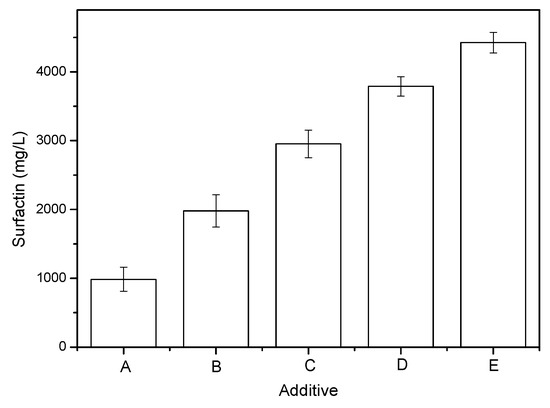
Figure 2.
Effects of solid additives on surfactin yield in B. subtilis ATCC 21332 cultivation in a 5-day fermentation: (A) none; (B) MMT; (C) AC; (D) Mg2Al–LDH; (E) Mg2Fe–LDH. The surfactin level in the supernatant of the broth was determined. Error bars indicate the standard deviations from three tests.
3.3. Effect of MgFe–LDH Composition on Surfactin Production
To study the effect of the Mg/Fe molar ratio on surfactin production, LDHs were prepared with Mg:Fe molar ratios of 1:1, 2:1, and 3:1 and added into B. subtilis cultivation. The concentrations of the additive ranged from 1 to 6 g/L in the fermentation medium. The cultivation was performed for 5 days. Figure 3 reveals that the LDH prepared with a Mg:Fe molar ratio of 2:1 had the greatest effect at promoting surfactin production. In general, LDHs possessing different ratios of divalent and trivalent metal ions possess different types of positively charged sheets and different layer dimensions in their resulting layered structures [38,39,40,41]. In the brucite-like layers of an LDH, a fraction of the divalent metal ions is replaced by trivalent metal ions, with the molar ratio of M3+:(M3+ + M2+) (x) normally positioned between 0.2 and 0.4 [24,42]. In this present study, an Mg:Fe ratio of 2:1 (x = 0.33) had the best effect on improving surfactin production. Thus, it appears that the layer size associated with the positively charged sheets of the Mg2Fe–LDH structure had the strongest stimulatory effect on the cells.
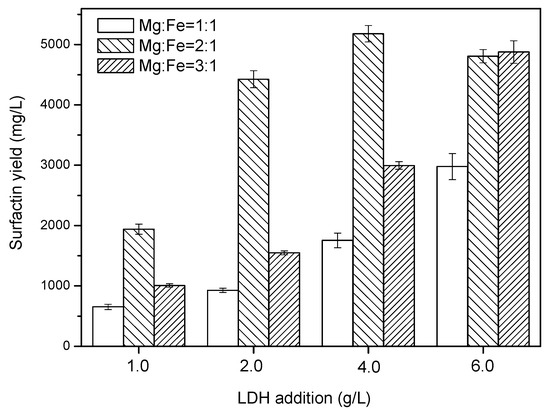
Figure 3.
Effect of Mg2Fe–LDH concentration (1, 2, 4, and 6 g/L) on the specific surfactin yield, where bars with different patterns represent various Mg2+:Fe3+ ratios: blank, 1:1; back slash, 2:1; slash, 3:1. Error bars indicate standard deviations from three tests.
3.4. Time Course of Cultivation with LDH Addition
To study the cell growth after adding LDH, the time courses of the cultivation events performed with and without added LDH were recorded (Figure 4). Although the addition of Mg2Fe–LDH promoted surfactin production, relative to that of the control, it was interesting to observe that the cell growth ended on the first day, where the amount of surfactin reached 4.8 g/L. In terms of product formation kinetics, this behavior was a clear growth-associated pattern: The cells grew and surfactin was produced. After day 1, the cells began to degrade in a decay phase, with the surfactin production decelerating. In contrast, the growth of cells was very rapid in the culture medium prepared without LDH, but the level of surfactin production was very low. Thus, a slight inhibition of cell growth appeared to trigger the cells to secrete more surfactin. We suspect that the surfactin secreted by the cells performed a role as a protecting agent that kept the cells from coming into direct contact with the LDH.
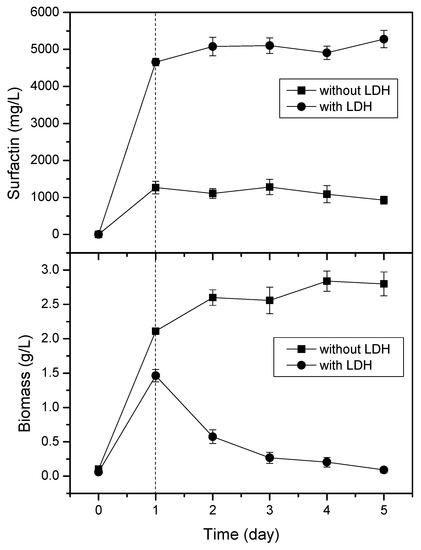
Figure 4.
Time courses of surfactin and biomass production in the presence and absence of Mg2Fe–LDH (4 g/L). Error bars indicate the standard deviations from three tests.
Figure 5A,B present microscopy images of the morphologies of the cells grown in the presence of the LDH. In the culture medium lacking the LDH, the cells had a short and rod-like morphology from day 1 to day 3 of culturing. By the fifth day, some cells became slenderer than the original short-rod cells. In contrast, in the culture medium incorporating the LDH, the cells grew in a short-rod shape on the first day, but, by the third day, most of the cells had decayed and shrunk, with many endospores present. By the fifth day, almost none of the cells were evident in the broth, with only some spores remaining in the culture. This observation is consistent with the amounts of cells measured in the study. Therefore, the addition of LDH did inhibit the growth of cells during the cell growth phase, but it also enhanced the production of surfactin. Accordingly, in addition to the high surfactin yield of the culture incorporating the LDH, an extremely high productivity also ensued. Because of the lower number of cells, not only was the specific production elevated, the carbon source conversion to surfactin was also enhanced and provided a high carbon yield.
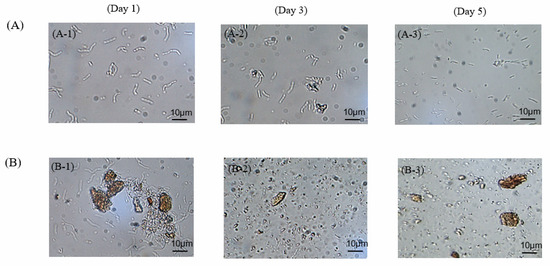
Figure 5.
Optical microscopy images of B. subtilis ATCC 21332 growth, taken after various numbers of cultivation days; (A) in the absence of any LDH; (B) in the presence of Mg2Fe–LDH.
3.5. Comparison of Surfactin Production
Table 1 compares the surfactin production in this present study with those reported previously in the literature. Four factors characterize surfactin production in these bioprocesses in terms of their efficiency for fermentation on industrial scale: the surfactin yield, the carbon yield, the productivity, and the specific production. Due to the variation on surfactin quantification, the surfactin assays, such as HPLC, surface tensions, and acid precipitation, were also listed. As evident in Table 1, the addition of Mg2Fe–LDH had a unique effect in promoting surfactin production. Historically, surfactin production has improved gradually from an original yield of less than 1000 mg/L two decades ago to approximately 2000 to 3000 mg/L recently. When using this present approach, the yield of surfactin after the addition of Mg2Fe–LDH was enhanced significantly, to greater than 5000 mg/L. Furthermore, the addition of Mg2Fe–LDH ensured that the carbon source mostly flowed to surfactin production. Indeed, the carbon source yield was approximately 52.8%. This high carbon yield characterizes a surfactin production process with a highly efficient use of the raw material. In addition, the presence of Mg2Fe–LDH caused the surfactin yield to reach 4660 mg/L after one day of culturing; that is, the productivity was 4660 mg/L, a remarkably high value as compared in the literature. In addition, because the number of cells decreased in the presence of Mg2Fe–LDH, the smaller amount of biomass and the higher surfactin yield led to a specific yield of 3.19 g/g DCW. The addition of Mg2Fe–LDH in B. subtilis submerged cultivation provided a high carbon yield, high productivity, and high specific production of surfactin; such a high efficiency appears well suited to industrial applications.

Table 1.
Various approaches used for surfactin production.
At the beginning of our approach, the change of Mg2Al–LDH to Mg2Fe–LDH was due to the practical need in agricultural applications, where the aluminum salt is prohibited from field tests. However, to our surprise, the replacement of additive to Mg2Fe–LDH did give an extraordinarily high surfactin production. Due to this effect, the three critical characteristics affecting surfactin production were evaluated. It was found that a high specific surfactin yield, a high productivity, and a high carbon yield could be obtained in the presence of the Mg2Fe–LDH. To explain the difference between Mg2Fe–LDH and Mg2Al–LDH additions, the effect of the Mg2Fe–LDH addition with the leaking iron trace element in the culture was the possible reason for this highly efficient surfactin production. To decipher the cause of the extraordinarily high stimulatory effect of Mg2Fe–LDH, the following considerations might be taken into account. In some previous studies, ferric ions have been found to serve as trace element stimulators, with an excellent ability to promote surfactin production [21,43,44]. In addition, the use of pristine Mg2Al–LDH has been claimed to enhance surfactin production as a result of its toxicity toward the cells [33]. Accordingly, the presence of Mg2Fe–LDH was expected to not only inhibit cell growth and promote surfactin production (similar to the behavior of Mg2Al–LDH) but also to slowly release some iron salts to serve as trace elements in the medium, thereby also improving the surfactin production. The higher production obtained using Mg2Fe–LDH, compared with that of Mg2Al–LDH, might be due to the synergistic effect of the Mg2Fe–LDH crystalline structure and the trace iron salts in the medium, with both combining to promote surfactin production to such a high level.
4. Conclusions
We investigated the effects of LDHs on the production of biomass and surfactin in a B. subtilis ATCC 21332 culture. The highest yield of surfactin (5280 mg/L) was obtained after 5 days of cultivation in the presence of 4 g/L Mg2Fe–LDH. This study demonstrated that LDHs have potential for use as additives to enhance the production of surfactin in B. subtilis ATCC 21332. Furthermore, microscopy revealed the inhibition of cell growth in the presence of the LDH, suggesting an efficient process for the production of surfactin through greater conversion of the carbon source.
Supplementary Materials
The following are available online at https://www.mdpi.com/2073-4352/9/7/355/s1, Figure S1: Typical surfactin standard chromatogram in HPLC showing surfactin isoform A–F.
Author Contributions
Data curation and methodology, P.-H.C. and S.-Y.L., conceptualization, writing—original draft preparation; writing—review and editing, project administration, T.-Y.J. and Y.-C.L.
Funding
This study was supported by research grants from the National Science Council of Taiwan, R.O.C. (grant nos. NSC 101-2221-E-005-061 and 106-2113-M-039-007), and China Medical University (grant no. CMU 107-N-22).
Acknowledgments
We thank Wei Yu-Hong of Yuan Ze University for sharing the strain B. subtilis ATCC 21332.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Maier, R.M.; Soberon-Chavez, G. Pseudomonas aeruginosa rhamnolipids: Biosynthesis and potential applications. Appl. Microbiol. Biotechnol. 2000, 54, 625–633. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.S.; Yoon, B.D.; Choung, D.H.; Oh, H.M.; Katsuragi, T.; Tani, Y. Characterization of a biosurfactant, mannosylerythritol lipid produced from Candida sp. SY16. Appl. Microbiol. Biotechnol. 1999, 52, 713–721. [Google Scholar] [CrossRef] [PubMed]
- Banat, I.M. Biosurfactants production and possible uses in microbial enhanced oil recovery and oil pollution remediation: A review. Bioresour. Technol. 1995, 51, 1–12. [Google Scholar] [CrossRef]
- Desai, J.D.; Banat, I.M. Microbial production of surfactants and their commercial potential. Microbiol. Mol. Biol. Rev. 1997, 61, 47–64. [Google Scholar] [PubMed]
- Mukherjee, A.K.; Das, K. Microbial surfactants and their potential applications: An overview. Adv. Exp. Med. Biol. 2010, 672, 54–64. [Google Scholar] [PubMed]
- Mulligan, C.N. Environmental applications for biosurfactants. Environ. Pollut. 2005, 133, 183–198. [Google Scholar] [CrossRef] [PubMed]
- Shaligram, N.S.; Singhal, R.S. Surfactin—A review on biosynthesis, fermentation, purification and applications. Food Technol. Biotechnol. 2010, 48, 119–134. [Google Scholar]
- Hosono, K.; Suzuki, H. Acylpeptides, the inhibitors of cyclic adenosine 3′,5′-monophosphate phosphodiesterase. I. Purification, physicochemical properties and structures of fatty acid residues. J. Antibiot. 1983, 36, 667–673. [Google Scholar] [CrossRef] [PubMed]
- Kakinuma, A.; Hori, M.; Isono, M.; Tamura, G.; Arima, K. Determination of amino acid aequence in surfactin a crystalline peptidelipid surfactant produced by Bacillus subtilis. Agric. Biol. Chem. 1969, 33, 971–972. [Google Scholar] [CrossRef]
- Arima, K.; Kakinuma, A.; Tamura, G. Surfactin, a crystalline peptide lipid surfactant produced by Bacillus subtilis: Isolation, characterization and its inhibition of fibrin clot formation. Biochem. Biophys. Res. Commun. 1968, 31, 488–494. [Google Scholar] [CrossRef]
- Besson, F.; Peypoux, F.; Michel, G.; Delcambe, L. Identification of antibiotics of iturin group in various strains of Bacillus subtilis. J. Antibiot. 1978, 31, 284–288. [Google Scholar] [CrossRef] [PubMed]
- Peypoux, F.; Guinand, M.; Michel, G.; Delcambe, L.; Das, B.C.; Lederer, E. Structure of iturine A, a peptidolipid antibiotic from Bacillus subtilis. Biochemistry 1978, 17, 3992–3996. [Google Scholar] [CrossRef]
- Vanittanakom, N.; Loeffler, W.; Koch, U.; Jung, G. Fengycin—A novel antifungal lipopeptide antibiotic produced by Bacillus subtilis F-29-3. J. Antibiot. 1986, 39, 888–901. [Google Scholar] [CrossRef]
- Liao, J.H.; Chen, P.Y.; Yang, Y.L.; Kan, S.C.; Hsieh, F.C.; Liu, Y.C. Clarification of the antagonistic effect of the lipopeptides produced by Bacillus amyloliquefaciens BPD1 against Pyricularia oryzae via in situ MALDI-TOF IMS analysis. Molecules 2016, 21, 1670. [Google Scholar] [CrossRef] [PubMed]
- Abdel-Mawgoud, A.M.; Aboulwafa, M.M.; Hassouna, N.A. Characterization of surfactin produced by Bacillus subtilis isolate BS5. Appl. Biochem. Biotechnol. 2008, 150, 289–303. [Google Scholar] [CrossRef] [PubMed]
- Mulligan, C.N.; Chow, T.Y.-K.; Gibbs, B.F. Enhanced biosurfactant production by a mutant Bacillus subtilis strain. Appl. Microbiol. Biotechnol. 1989, 31, 486–489. [Google Scholar] [CrossRef]
- Yeh, M.S.; Wei, Y.H.; Chang, J.S. Enhanced production of surfactin from Bacillus subtilis by addition of solid carriers. Biotechnol. Prog. 2005, 21, 1329–1334. [Google Scholar] [CrossRef]
- Davis, D.A.; Lynch, H.C.; Varley, J. The production of surfactin in batch culture by Bacillus subtilis ATCC 21332 is strongly influenced by the conditions of nitrogen metabolism. Enzyme Microb. Technol. 1999, 25, 322–329. [Google Scholar] [CrossRef]
- Sen, R. Response surface optimization of the critical media components for the production of surfactin. J. Chem. Technol. Biotechnol. 1997, 68, 263–270. [Google Scholar] [CrossRef]
- Wei, Y.-H.; Chu, I.-M. Mn2+ improves surfactin production by Bacillus subtilis. Biotechnol. Lett. 2002, 24, 479–482. [Google Scholar] [CrossRef]
- Wei, Y.-H.; Wang, L.-F.; Chang, J.-S. Optimizing iron supplement strategies for enhanced surfactin production with Bacillus subtilis. Biotechnol. Prog. 2004, 20, 979–983. [Google Scholar] [CrossRef] [PubMed]
- Cavani, F.; Trifirò, F.; Vaccari, A. Hydrotalcite-type anionic clays: Preparation, properties and applications. Catal. Today 1991, 11, 173–301. [Google Scholar] [CrossRef]
- Choy, J.H.; Kwak, S.Y.; Jeong, Y.J.; Park, J.S. Inorganic layered double hydroxides as nonviral vector. Angew. Chem. Int. Ed. 2000, 39, 4041–4045. [Google Scholar] [CrossRef]
- Wang, Q.; O’Hare, D. Recent advances in the synthesis and application of layered double hydroxide (LDH) nanosheets. Chem. Rev. 2012, 112, 4124–4155. [Google Scholar] [CrossRef] [PubMed]
- Williams, G.R.; O’Hare, D. Towards understanding, control and application of layered double hydroxide chemistry. J. Mater. Chem. 2006, 16, 3065–3074. [Google Scholar] [CrossRef]
- Evans, D.G.; Duan, X. Preparation of layered double hydroxides and their applications as additives in polymers, as precursors to magnetic materials and in biology and medicine. Chem. Commun. 2006, 5, 485–496. [Google Scholar] [CrossRef] [PubMed]
- Lin, J.-J.; Juang, T.-Y. Intercalation of layered double hydroxides by poly(oxyalkylene)-amidocarboxylates: Tailoring layered basal spacing. Polymer 2004, 45, 7887–7893. [Google Scholar] [CrossRef]
- Shau, S.-M.; Juang, T.-Y.; Lin, H.-S.; Huang, C.-L.; Hsieh, C.-F.; Wu, J.-Y.; Jeng, R.-J. Individual graphene oxide platelets through direct molecular exfoliation with globular amphiphilic hyperbranched polymers. Polym. Chem. 2012, 3, 1249–1259. [Google Scholar] [CrossRef]
- Juang, T.-Y.; Chen, Y.-C.; Tsai, C.-C.; Dai, S.A.; Wu, T.-M.; Jeng, R.-J. Nanoscale organic/inorganic hybrids based on self-organized dendritic macromolecules on montmorillonites. Appl. Clay Sci. 2010, 48, 103–110. [Google Scholar] [CrossRef]
- Conterosito, E.; Croce, G.; Palin, L.; Pagano, C.; Perioli, L.; Viterbo, D.; Boccaleri, E.; Paul, G.; Milanesio, M. Structural characterization and thermal and chemical stability of bioactive molecule-hydrotalcite (LDH) nanocomposites. Phys. Chem. Chem. Phys. 2013, 15, 13418–13433. [Google Scholar] [CrossRef]
- Toson, V.; Conterosito, E.; Palin, L.; Boccaleri, E.; Milanesio, M.; Gianotti, V. Facile intercalation of organic molecules into hydrotalcites by liquid-assisted grinding: Yield optimization by a chemometric approach. Cryst. Growth Des. 2015, 15, 5368–5374. [Google Scholar] [CrossRef]
- Choy, J.-H.; Choi, S.-J.; Oh, J.-M.; Park, T. Clay minerals and layered double hydroxides for novel biological applications. Appl. Clay Sci. 2007, 36, 122–132. [Google Scholar] [CrossRef]
- Kan, S.-C.; Lee, C.-C.; Hsu, Y.-C.; Peng, Y.-H.; Chen, C.-C.; Huang, J.-J.; Huang, J.-W.; Shieh, C.-J.; Juang, T.-Y.; Liu, Y.-C. Enhanced surfactin production via the addition of layered double hydroxides. J. Taiwan Inst. Chem. Eng. 2017, 80, 10–15. [Google Scholar] [CrossRef]
- Delhaize, E.; Ryan, P.R. Aluminum toxicity and tolerance in plants. Plant Physiol. 1995, 107, 315–321. [Google Scholar] [CrossRef] [PubMed]
- Pourhossein, A.; Alizadeh, K. Salt-assisted liquid-liquid extraction followed by high performance liquid chromatography for determination of carvedilol in human plasma. J. Rep. Pharm. Sci. 2018, 7, 79–87. [Google Scholar]
- Kuthati, Y.; Kankala, R.K.; Lee, C.-H. Layered double hydroxide nanoparticles for biomedical applications: Current status and recent prospects. Appl. Clay Sci. 2015, 112–113, 100–116. [Google Scholar] [CrossRef]
- Mishra, G.; Dash, B.; Pandey, S. Layered double hydroxides: A brief review from fundamentals to application as evolving biomaterials. Appl. Clay Sci. 2018, 153, 172–186. [Google Scholar] [CrossRef]
- Aisawa, S.; Takahashi, S.; Ogasawara, W.; Umetsu, Y.; Narita, E. Direct intercalation of amino acids into layered double hydroxides by coprecipitation. J. Solid State Chem. 2001, 162, 52–62. [Google Scholar] [CrossRef]
- Khan, A.I.; O’Hare, D. Intercalation chemistry of layered double hydroxides: Recent developments and applications. J. Mater. Chem. 2002, 12, 3191–3198. [Google Scholar] [CrossRef]
- Rives, V. Characterisation of layered double hydroxides and their decomposition products. Mater. Chem. Phys. 2002, 75, 19–25. [Google Scholar] [CrossRef]
- Rives, V.; Ulibarri, M.A. Layered double hydroxides (LDH) intercalated with metal coordination compounds and oxometalates. Coord. Chem. Rev. 1999, 181, 61–120. [Google Scholar] [CrossRef]
- Long, X.; Wang, Z.; Xiao, S.; An, Y.; Yang, S. Transition metal based layered double hydroxides tailored for energy conversion and storage. Mater. Today 2016, 19, 213–226. [Google Scholar] [CrossRef]
- Wei, Y.-H.; Chu, I.-M. Enhancement of surfactin production in iron-enriched media by Bacillus subtilis ATCC 21332. Enzyme Microb. Technol. 1998, 22, 724–728. [Google Scholar] [CrossRef]
- Wei, Y.-H.; Wang, L.-F.; Chang, J.-S.; Kung, S.-S. Identification of induced acidification in iron-enriched cultures of Bacillus subtilis during biosurfactant fermentation. J. Biosci. Bioeng. 2003, 96, 174–178. [Google Scholar] [CrossRef]
- Cooper, D.G.; Macdonald, C.R.; Duff, S.J.; Kosaric, N. Enhanced production of surfactin from Bacillus subtilis by continuous product removal and metal cation additions. Appl. Environ. Microbiol. 1981, 42, 408–412. [Google Scholar] [PubMed]
- Sheppard, J.D.; Mulligan, C.N. The production of surfactin by Bacillus subtilis grown on peat hydrolysate. Appl. Microbiol. Biotechnol. 1987, 27, 110–116. [Google Scholar] [CrossRef]
- Drouin, C.M.; Cooper, D.G. Biosurfactants and aqueous two-phase fermentation. Biotechnol. Bioeng. 1992, 40, 86–90. [Google Scholar] [CrossRef] [PubMed]
- Ohno, A.; Ano, T.; Shoda, M. Production of a lipopeptide antibiotic surfactin with recombinant Bacillus subtilis. Biotechnol. Lett. 1992, 14, 1165–1168. [Google Scholar] [CrossRef]
- Wei, Y.-H.; Lai, C.-C.; Chang, J.-S. Using Taguchi experimental design methods to optimize trace element composition for enhanced surfactin production by Bacillus subtilis ATCC 21332. Process Biochem. 2007, 42, 40–45. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).