Morphologically Controlled Synthesis of Cs2SnCl6 Perovskite Crystals and Their Photoluminescence Activity
Abstract
:1. Introduction
2. Experiments
3. Result and Discussion
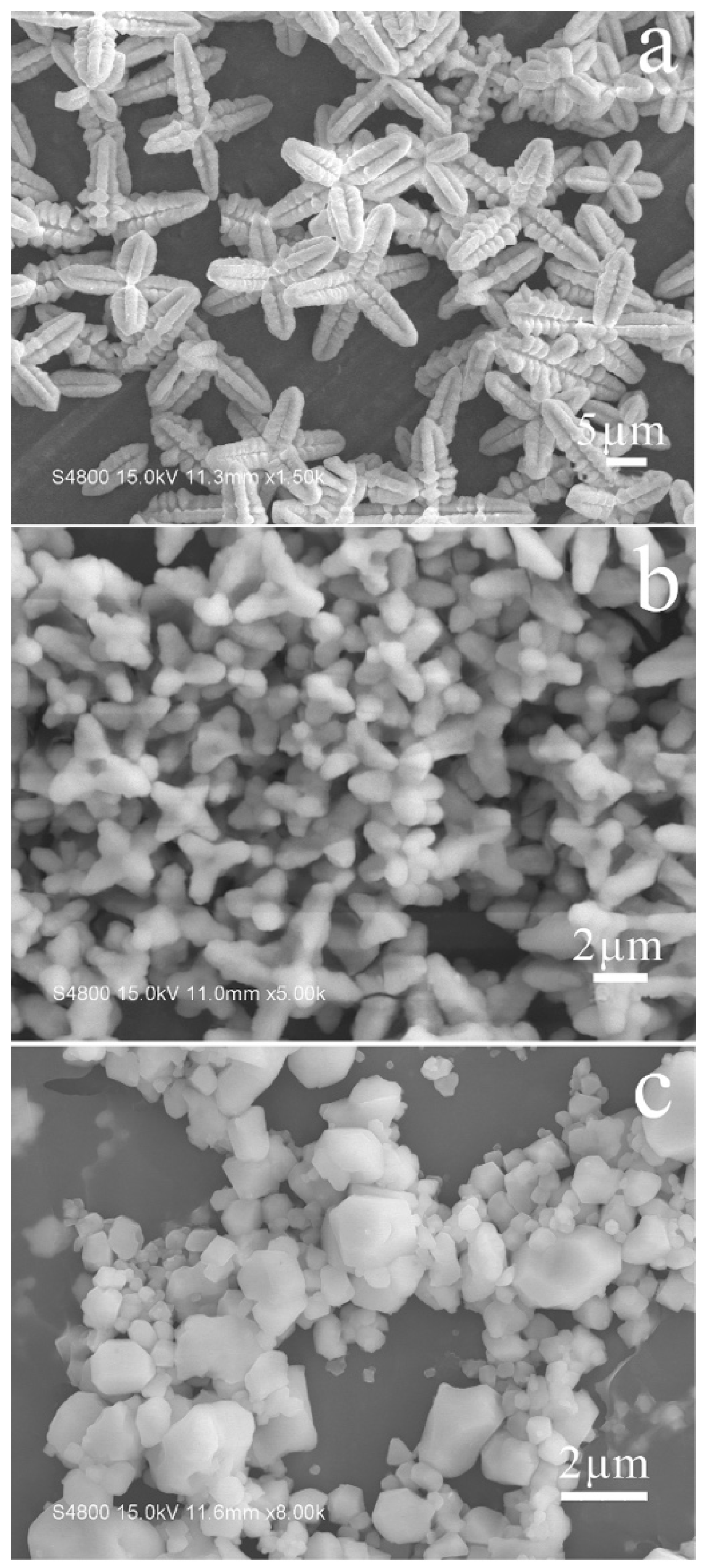
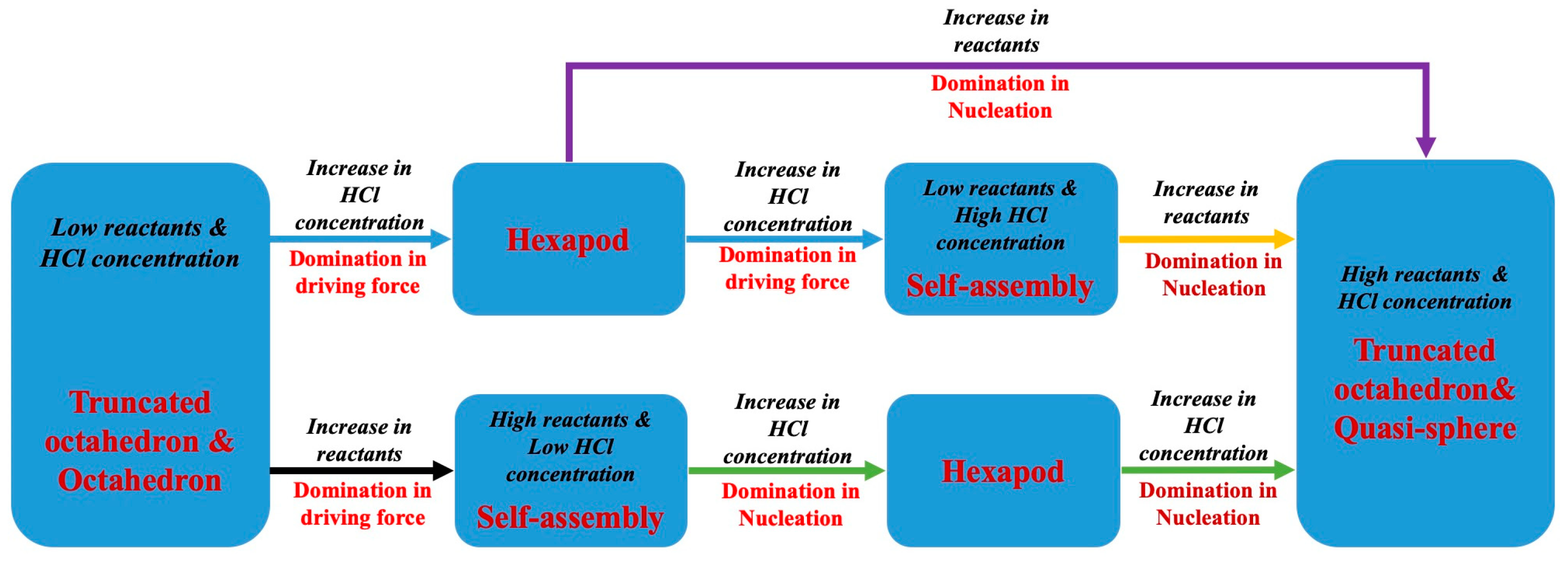
3.1. The Crystal Growth and Morphologies of the Cs2SnCl6 Crystals in Different Synthesis Conditions
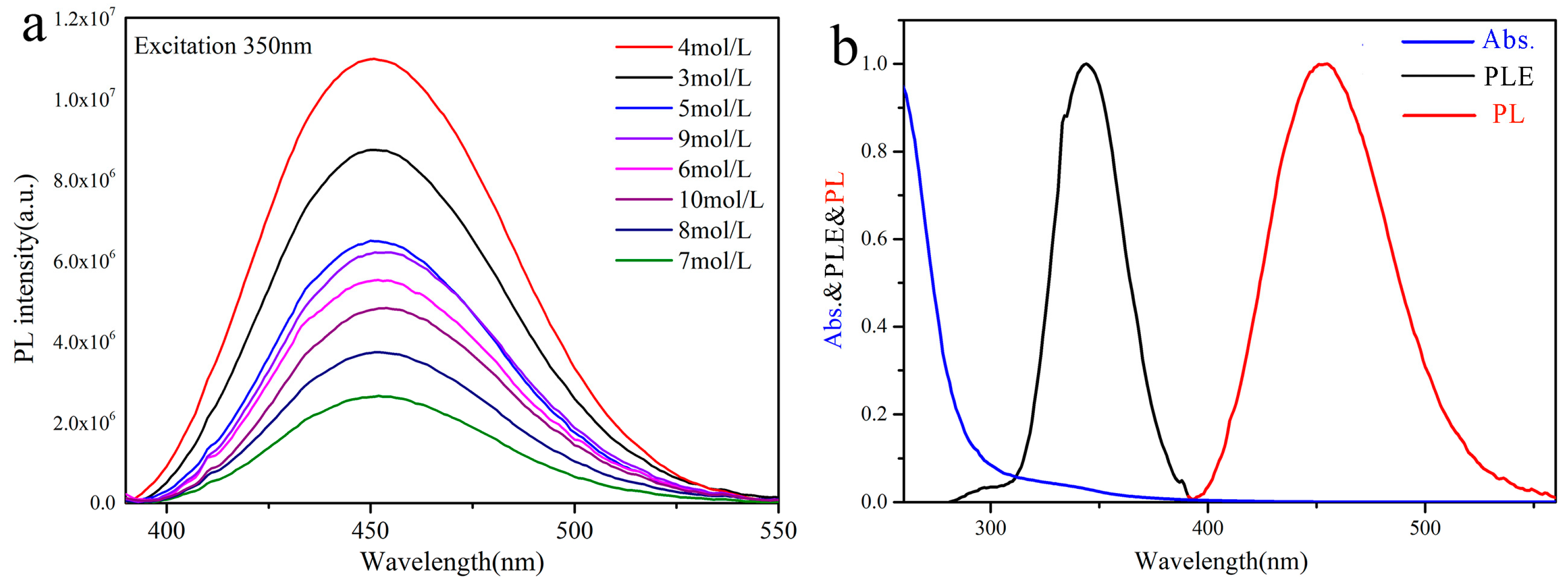
3.2. Photoluminescence Characteristics
4. Conclusions
Supplementary Materials
Author Contributions
Conflicts of Interest
References
- Palazon, F.; Di Stasio, F.; Akkerman, Q.A.; Krahne, R.; Prato, M.; Manna, L. Polymer-free Films of Inorganic Halide Perovskite Nanocrystals as UV-to-White Color-conversion Layers in LEDs. Chem. Mater. 2016, 28, 2902–2906. [Google Scholar] [CrossRef]
- Wang, Y.; Li, X.; Song, J.; Xiao, L.; Zeng, H.; Sun, H. All-inorganic Colloidal Perovskite Quantum Dots: A New Class of Lasing Materials with Favorable Characteristics. Adv. Mater. 2015, 27, 7101–7108. [Google Scholar] [CrossRef]
- Ramasamy, P.; Lim, D.H.; Kim, B.; Lee, S.H.; Lee, M.S.; Lee, J.S. All-inorganic cesium lead halide perovskite nanocrystals for photodetector applications. Chem. Commun. 2016, 52, 2067–2070. [Google Scholar] [CrossRef] [PubMed]
- Saparov, B.; Sun, J.P.; Meng, W.; Xiao, Z.; Duan, H.S.; Gunawan, O.; Shin, D.; Hill, I.G.; Yan, Y.; Mitzi, D.B. Thin-film Deposition and Characterization of a Sn-deficient Perovskite Derivative Cs2SnI6. Chem. Mater. 2016, 28, 2315–2322. [Google Scholar] [CrossRef]
- Lee, B.; Stoumpos, C.C.; Zhou, N.; Hao, F.; Malliakas, C.; Yeh, C.Y.; Marks, T.J.; Kanatzidis, M.G.; Chang, R.P. Air-stable Molecular Semiconducting Iodosalts for Solar Cell Applications: Cs2SnI6 as a Hole Conductor. J. Am. Chem. Soc. 2014, 136, 15379–15385. [Google Scholar] [CrossRef]
- Qiu, X.; Cao, B.; Yuan, S.; Chen, X.; Qiu, Z.; Jiang, Y.; Ye, Q.; Wang, H.; Zeng, H.; Liu, J.; et al. From Unstable CsSnI3 to Air-stable Cs2SnI6: A Lead-free Perovskite Solar Cell Light Absorber with Bandgap of 1.48eV and High Absorption Coefficient. Sol. Energ. Mat. Sol. C. 2017, 159, 227–234. [Google Scholar] [CrossRef]
- Noel, N.K.; Stranks, S.D.; Abate, A.; Wehrenfennig, C.; Guarnera, S.; Haghighirad, A.A.; Sadhanala, A.; Eperon, G.E.; Pathak, S.K.; Johnston, M.B.; et al. Lead-free Organic–inorganic Tin Halide Perovskites for Photovoltaic Applications. Energ. Environ. Sci. 2014, 7, 3061–3068. [Google Scholar] [CrossRef]
- Xing, G.; Kumar, M.H.; Chong, W.K.; Liu, X.; Cai, Y.; Ding, H.; Asta, M.; Gratzel, M.; Mhaisalkar, S.; Mathews, N.; et al. Solution-processed Tin-based Perovskite for Near-infrared Lasing. Adv. Mater. 2016, 28, 8191–8196. [Google Scholar] [CrossRef]
- Lai, M.L.; Tay, T.Y.; Sadhanala, A.; Dutton, S.E.; Li, G.; Friend, R.H.; Tan, Z.K. Tunable Near-infrared Luminescence in Tin Halide Perovskite Devices. J. Phys. Chem. Lett. 2016, 7, 2653–2658. [Google Scholar] [CrossRef]
- Qiu, X.; Jiang, Y.; Zhang, H.; Qiu, Z.; Yuan, S.; Wang, P.; Cao, B. Lead-free Mesoscopic Cs2SnI6 Perovskite Solar Cells Using Different Nanostructured ZnO Nanorods As Electron Transport Layers. Phy. Status Solidi-RRL. 2016, 10, 587–591. [Google Scholar] [CrossRef]
- Xiao, Z.; Zhou, Y.; Hosono, H.; Kamiya, T. Intrinsic Defects in a Photovoltaic Perovskite Variant Cs2SnI6. Phys. Chem. Chem. Phys. 2015, 17, 18900–18903. [Google Scholar] [CrossRef] [PubMed]
- Kaltzoglou, A.; Antoniadou, M.; Kontos, A.G.; Stoumpos, C.C.; Perganti, D.; Siranidi, E.; Raptis, V.; Trohidou, K.; Psycharis, V.; Kanatzidis, M.G.; et al. Optical-vibrational Properties of the Cs2SnX6(X = Cl, Br, I) Defect Perovskites and Hole-transport Efficiency in Dye-sensitized Solar Cells. J. Phys. Chem. C 2016, 120, 11777–11785. [Google Scholar] [CrossRef]
- Wang, A.; Yan, X.; Zhang, M.; Sun, S.; Yang, M.; Shen, W.; Pan, X.; Wang, P.; Deng, Z. Controlled Synthesis of Lead-free and Stable Perovskite Derivative Cs2SnI6 Nanocrystals via a Facile Hot-injection Process. Chem. Mater. 2016, 28, 8132–8140. [Google Scholar] [CrossRef]
- Dolzhnikov, D.S.; Wang, C.; Xu, Y.; Kanatzidis, M.G.; Weiss, E.A. Ligand-free, Quantum-confined Cs2SnI6 Perovskite Nanocrystals. Chem. Mater. 2017, 29, 7901–7907. [Google Scholar] [CrossRef]
- Kaltzoglou, A.; Antoniadou, M.; Perganti, D.; Siranidi, E.; Raptis, V.; Trohidou, K.; Psycharis, V.; Kontos, A.G.; Falaras, P. Mixed-halide Cs2SnI3Br3 Perovskite as Low Resistance Hole-transporting Material in Dye-sensitized Solar Cells. Electrochim. Acta 2015, 184, 466–474. [Google Scholar] [CrossRef]
- Tan, Z.; Li, J.; Zhang, C.; Li, Z.; Hu, Q.; Xiao, Z.; Kamiya, T.; Hosono, H.; Niu, G.; Lifshitz, E.; et al. Highly Efficient Blue-emitting Bi-doped Cs2SnCl6 Perovskite Variant: Photoluminescence Induced by Impurity Doping. Adv. Funct. Mater. 2018, 28, 1801131. [Google Scholar] [CrossRef]
- Donnay, D.J.D.; Harker, D. A New Law of Crystal Morphology Extending the Law of Bravais. Am. Mineral. 1937, 22, 446–467. [Google Scholar]
- Sunagawa, I. Growth and Morphology of Crystals. Forma-Tokyo 1999, 14, 147–166. [Google Scholar]
- Giuseppe, A.; Nuccioo, B.; John, R.F.; Antta, F.; Giovanna, I.; Giovanni, P.; Maria, V.R. Mössbauer, Far-infrared, and XPS Investigations of SnCl2 and SnCl4 Introduced in Polyconjugated Monosubstituted Acetylene Matrices. Appl. Spectrosc. 1995, 49, 237–240. [Google Scholar]
- Zhang, H.; Zhu, L.; Cheng, J.; Chen, L.; Liu, C.; Yuan, S. Photoluminescence Characteristics of Sn2+ and Ce3+-doped Cs2SnCl6 Double-perovskite Crystals. Materials 2019, 12, 1501. [Google Scholar] [CrossRef] [PubMed]
- Ho, J.Y.; Huang, M.H. Synthesis of Submicrometer-sized Cu2O Crystals with Morphological Evolution from Cubic to Hexapod Structures and Their Comparative Photocatalytic Activity. J. Phys. Chem. C 2009, 113, 14159–14164. [Google Scholar] [CrossRef]
- Zhang, B.; Guo, F.; Yang, L.; Jia, X.; Liu, B.; Xie, Z.; Chen, D.; Lu, H.; Zhang, R.; Zheng, Y. Shape-evolution Control of Hybrid Perovskite CH3NH3PbI3 Crystals via Solvothermal Synthesis. J. Cryst. Growth 2017, 459, 167–172. [Google Scholar] [CrossRef]



© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, H.; Zhu, L.; Cheng, J.; Chen, L.; Liu, C.; Yuan, S. Morphologically Controlled Synthesis of Cs2SnCl6 Perovskite Crystals and Their Photoluminescence Activity. Crystals 2019, 9, 258. https://doi.org/10.3390/cryst9050258
Zhang H, Zhu L, Cheng J, Chen L, Liu C, Yuan S. Morphologically Controlled Synthesis of Cs2SnCl6 Perovskite Crystals and Their Photoluminescence Activity. Crystals. 2019; 9(5):258. https://doi.org/10.3390/cryst9050258
Chicago/Turabian StyleZhang, Hongdan, Ludan Zhu, Jun Cheng, Long Chen, Chuanqi Liu, and Shuanglong Yuan. 2019. "Morphologically Controlled Synthesis of Cs2SnCl6 Perovskite Crystals and Their Photoluminescence Activity" Crystals 9, no. 5: 258. https://doi.org/10.3390/cryst9050258
APA StyleZhang, H., Zhu, L., Cheng, J., Chen, L., Liu, C., & Yuan, S. (2019). Morphologically Controlled Synthesis of Cs2SnCl6 Perovskite Crystals and Their Photoluminescence Activity. Crystals, 9(5), 258. https://doi.org/10.3390/cryst9050258