Struvite Grown in Gel, Its Crystal Structure at 90 K and Thermoanalytical Study
Abstract
:1. Introduction
2. Materials and Methods
2.1. Struvite Crystals Growth from Gel Medium
2.2. Instrumentation
2.3. Data Collection, Structure Determination and Refinement
3. Results and Discussion
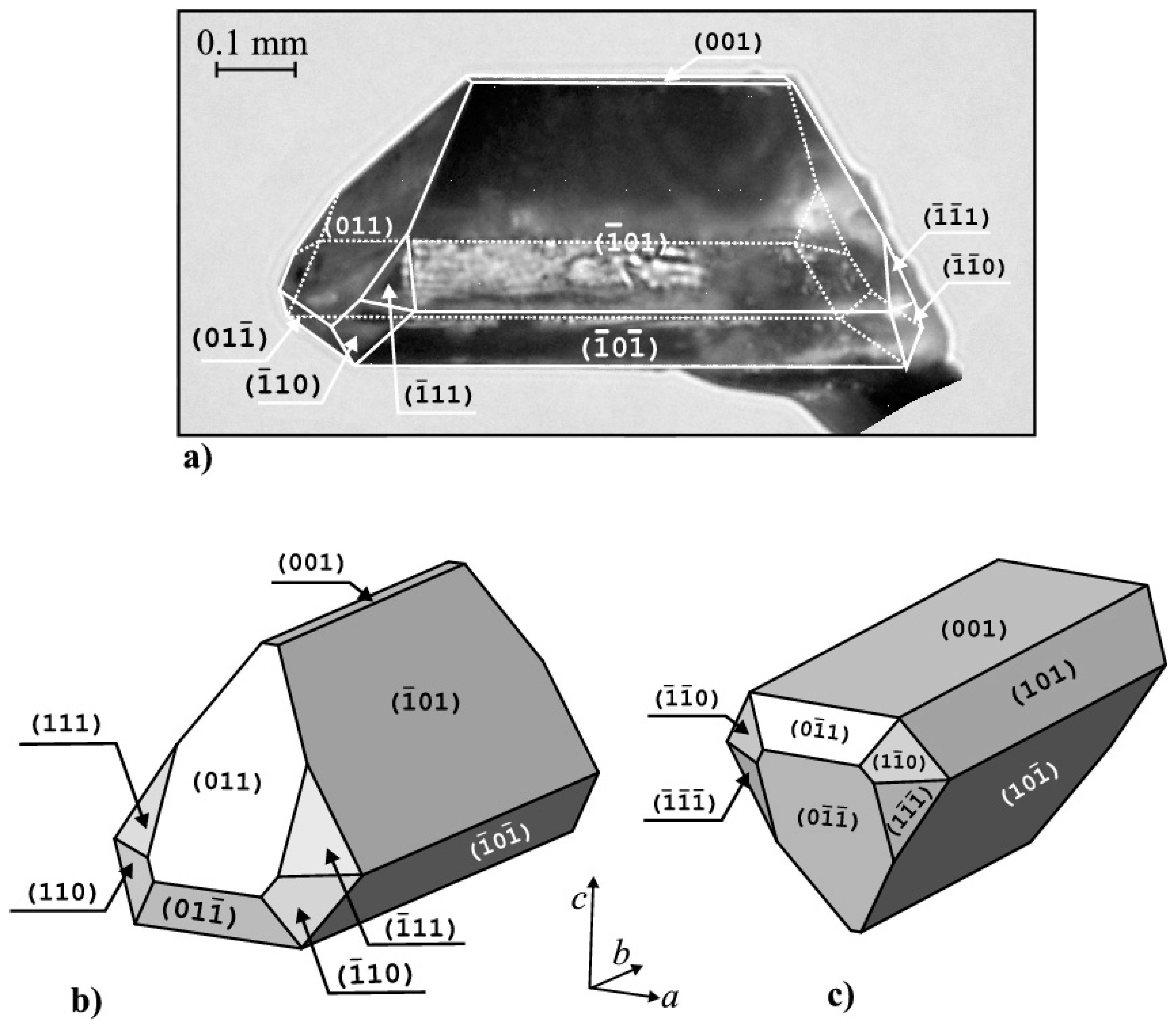
3.1. Struvite Synthesis, Morphology and Habit
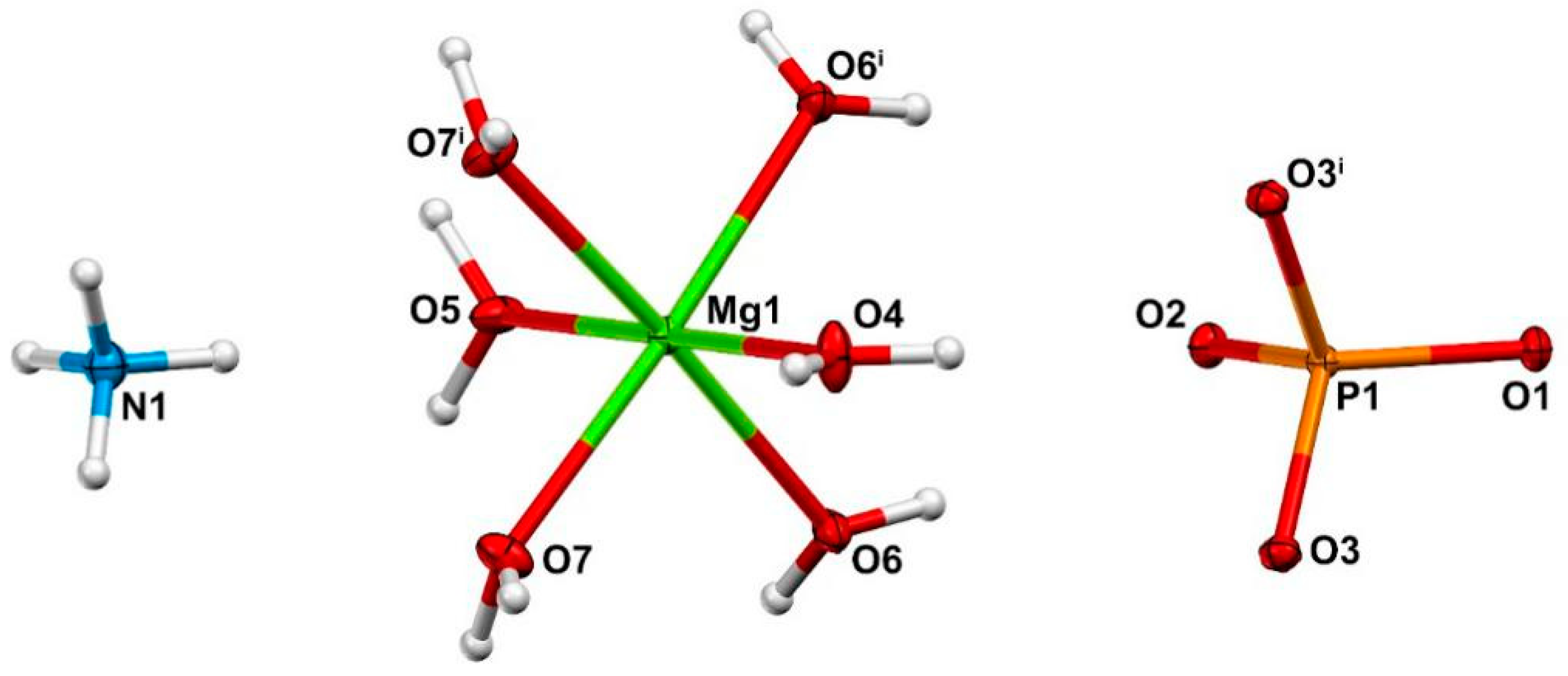
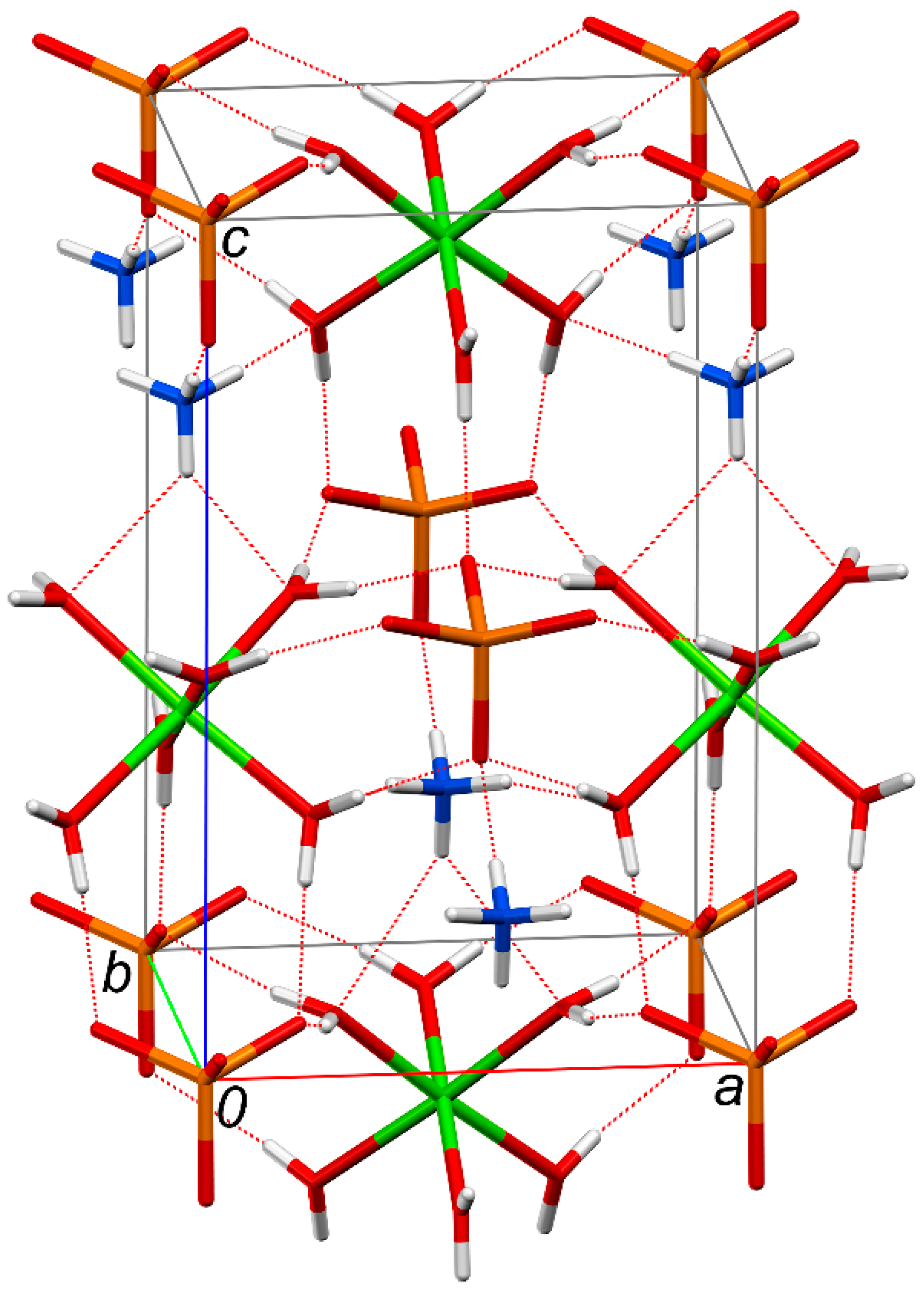
3.2. Crystal Structure
3.3. Thermoanalytical Study
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Henisch, H.K. Crystal Growth in Gels; The Pennsylvania State University Press: University Park, PA, USA; London, UK, 1970. [Google Scholar]
- Lorber, B.; Sauter, C.; Theobald-Dietrich, A.; Moreno, A.; Schellenberger, P.; Robert, M.C.; Capelle, B.; Sanglier, S.; Potier, N.; Giege, R. Crystal growth of proteins, nucleic acids, and viruses in gels. Prog. Biophys. Mol. Biol. 2009, 101, 13–25. [Google Scholar] [CrossRef] [PubMed]
- Otalora, F.; Gavira, J.A.; Ng, J.D.; Garcia-Ruiz, J.M. Counter diffusion methods applied to protein crystallization. Prog. Biophys. Mol. Biol. 2009, 101, 26–37. [Google Scholar] [CrossRef] [PubMed]
- Bogorodskiy, A.; Frolov, F.; Mishin, A.; Round, E.; Polovinkin, V.; Cherezov, V.; Gordeliy, V.; Büldt, G.; Gensch, T.; Borshchevskiy, V. Nucleation and Growth of Membrane Protein Crystals in Meso—A Fluorescence Microscopy Study. Cryst. Growth Des. 2015, 15, 5656–5660. [Google Scholar] [CrossRef]
- Cherezov, V.; Clogston, J.; Papiz, M.Z.; Caffrey, M. Room to Move: Crystallizing Membrane Proteins in Swollen Lipidic Mesophases. J. Mol. Biol. 2006, 357, 1605–1618. [Google Scholar] [CrossRef] [PubMed]
- Foster, J.A.; Damodaran, K.K.; Maurin, A.; Day, G.M.; Thompson, H.P.G.; Cameron, G.J.; Bernal, J.C.; Steed, J.W. Pharmaceutical polymorph control in a drugmimetic supramolecular gel. Chem. Sci. 2017, 8, 78–84. [Google Scholar] [CrossRef] [PubMed]
- Moreno, A.; Rosales-Hoz, M.J. Crystal growth of inorganic, organic, and biological macromolecules in gels. Prog. Cryst. Growth Charact. Mater. 2017, 3, 63–71. [Google Scholar] [CrossRef]
- Chauhan, C.K.; Joshi, M.J. Growth inhibition of Struvite crystals in the presence of juice of Citrus medica Linn. Urol. Res. 2008, 36, 265–273. [Google Scholar] [CrossRef]
- Manuel Bravo-Arredondo, J.; Moreno, A.; Mendoza, M.E. Crystal growth of cholesterol in hydrogels and its characterization. J. Cryst. Growth 2014, 401, 242–247. [Google Scholar] [CrossRef]
- Ulex, G.L. On struvite, a new mineral. Mem. Proc. Chem. Soc. 1845, 3, 106–110. [Google Scholar] [CrossRef]
- Lafuente, B.; Downs, R.T.; Yang, H.; Stone, N. The power of databases: The RRUFF project. In Highlights in Mineralogical Crystallography; Armbruster, T., Danisi, R.M., Eds.; W. De Gruyter: Berlin, Germany, 2015; pp. 1–30. Available online: http://rruff.geo.arizona.edu/doclib/hom/struvite.pdf (accessed on 20 January 2019).
- Doyle, J.D.; Parsons, S.A. Struvite formation, control and recovery. Water Res. 2002, 36, 3925–3940. [Google Scholar] [CrossRef]
- Brett, S.; Guy, J.; Morse, G.K.; Lester, J.N. Phosphorous Removal and Recovery Technologies; Selper Publications: London, UK, 1997. [Google Scholar]
- Ye, Z.; Shen, Y.; Ye, X.; Zhang, Z.; Chen, S.; Shi, J. Phosphorus recovery from wastewater by struvite crystallization: Property of aggregates. J. Environ. Sci. 2014, 26, 991–1000. [Google Scholar] [CrossRef]
- Booker, N.A.; Pristley, A.J.; Fraser, I.H. Struvite formation in wasterwater treatment plants: Opportunities for nutrient recovery. Environ. Technol. 1999, 20, 777–782. [Google Scholar] [CrossRef]
- Gaterell, M.R.; Gay, R.; Wilson, R.; Gochin, R.J.; Lester, J.N. An economic and environmental evaluation of the opportunities for substituting phosphorus recovered from wastewater treatment works in existing UK fertiliser markets. Environ. Technol. 2000, 21, 1067–1084. [Google Scholar] [CrossRef]
- Jaffera, Y.; Clarkb, T.A.; Pearceb, P.; Parsons, S.A. Potential phosphorous recovery by struvite formation. Water Res. 2002, 36, 1834–1842. [Google Scholar] [CrossRef]
- Maqueda, C.; Perez Rodriguez, J.L.; Lebrato, J. Study of struvite precipitation in anaerobic digesters. Water Res. 1994, 28, 411–416. [Google Scholar] [CrossRef]
- Ghos, G.; Mohan, K.; Sarkar, A. Struvite proves a good fertilizer. CEEP Scope Newsl. 2002, 37, 3–4. [Google Scholar]
- Capdevielle, A.; Sýkorová, E.; Béline, F.; Daumer, M.L. Kinetics of struvite precipitation in synthetic biologically treated swine wastewaters. Environ. Technol. 2014, 35, 1250–1262. [Google Scholar] [CrossRef]
- Weil, M. The struvite-type compounds M[Mg(H2O)6](XO4), where M = Rb, Tl and X = P, As. Cryst. Res. Technol. 2008, 43, 1286–1291. [Google Scholar] [CrossRef]
- Fang, C.; Zhang, T.; Jiang, R.; Ohtake, H. Phosphate enhance recovery from wastewater by mechanism analysis and optimization of struvite settle ability in fluidized bed reactor. Sci. Rep. 2016, 6, 32215. [Google Scholar] [CrossRef]
- Strickland, J. Perspectives for phosphorous recovery offered by enhanced biological removal. Environ. Technol. 1999, 20, 721–725. [Google Scholar] [CrossRef]
- Abbona, F. Crystallization of calcium and magnesium phosphates from solution of low concentration. J. Cryst. Growth 1997, 104, 661–671. [Google Scholar] [CrossRef]
- Bridger, G. Fertilizer value of struvite. CEEP Scope Newsl. 2001, 43, 3–4. [Google Scholar]
- Bichler, K.H.; Eipper, E.; Naber, K.; Braun, V.; Zimmermann, R.; Lahme, S. Urinary infection stones. Int. J. Antimicrob. Agents 2002, 19, 488–498. [Google Scholar] [CrossRef]
- Gleeson, M.J.; Griffith, D.P. Struvite calculi. Br. J. Urol. 1993, 71, 503–511. [Google Scholar] [CrossRef] [PubMed]
- Griffith, D.P.; Musher, D.M.; Itin, C. Urease. The primary cause of infection-induced urinary stones. Investig. Urol. 1976, 13, 346–350. [Google Scholar]
- McLean, R.J.C.; Nickel, J.C.; Cheng, K.J.; Costerton, J.W. The ecology and pathogenicity of urease-producing bacteria in the urinary tract. CRC Crit. Rev. Microbiol. 1988, 16, 37–79. [Google Scholar] [CrossRef] [PubMed]
- Leusmann, D.B.; Sabinski, F. Potential contribution of optional urease-positive bacteria to idiopathic urinary calcium stone formation. Urol. Res. 1996, 24, 73–78. [Google Scholar] [CrossRef] [PubMed]
- Chauhan, C.K.; Joshi, M.J. In vitro crystallization, characterization and growth inhibition study of urinary type struvite crystals. J. Cryst. Growth 2013, 362, 330–337. [Google Scholar] [CrossRef]
- Website of e-Medicine (Medscape): Struvite and Staghorn Calculi. Available online: http://emedicine.medscape.com/article/439127 (accessed on 18 December 2018).
- Whitaker, A.; Jeffery, J.W. The Crystal Structure of Struvite, MgNH4PO4.6H2O. Acta Crystallogr. Sect. B 1970, 26, 1429–1440. [Google Scholar] [CrossRef]
- Abbona, F.; Calleri, M.; Ivaldi, G. Synthetic Struvite, MgNH4PO4.6H2O: Correct Polarity and Surface Features of Some Complementary Forms. Acta Crystallogr. Sect. B 1984, 40, 223–227. [Google Scholar] [CrossRef]
- Tansman, G.F.; Kindstedt, P.S.; Hughes, J.M. Minerals in Food: Crystal Structures of Ikaite and Struvite from Bacterial Smears on Washed-Rind Cheese. Can. Mineral. 2017, 55, 89–100. [Google Scholar] [CrossRef]
- Ferraris, G.; Fuess, H.; Joswig, W. Neutron Diffraction Study of MgNH4PO4.6H2O (Struvite) and Survey of Water Molecules Donating Short Hydrogen Bonds. Acta Crystallogr. Sect. B 1986, 42, 253–258. [Google Scholar] [CrossRef]
- Ramlogan, M.V.; Rouff, A.A. An investigation of the thermal behavior of magnesium ammonium phosphate hexahydrate. J. Therm. Anal. Calorim. 2016, 123, 145–152. [Google Scholar] [CrossRef]
- Chen, Y.; Tang, J.; Li, We.; Zhong, Z.; Yin, J. Thermal decomposition of magnesium ammonium phosphate and adsorption properties of its pyrolysis products toward ammonia nitrogen. Trans. Nonferrous Met. Soc. China 2015, 25, 497–503. [Google Scholar] [CrossRef]
- Yu, R.; Ren, H.; Wang, Y.; Ding, L.; Geng, J.; Xu, K.; Zhang, Y. A kinetic study of struvite precipitation recycling technology with NaOH/Mg(OH)2 addition. Bioresour. Technol. 2013, 143, 519–524. [Google Scholar] [CrossRef] [PubMed]
- Ma, N.; Rouff, A.A.; Phillips, B.L. A 31P NMR and TG/DSC-FTIR Investigation of the Influence of Initial pH on Phosphorus Recovery as Struvite. ACS Sustain. Chem. Eng. 2014, 4, 816–822. [Google Scholar] [CrossRef]
- Rigaku, O.D. CrysAlis PRO; Rigaku Oxford Diffraction Ltd.: Yarnton, UK, 2016. [Google Scholar]
- Sheldrick, G.M. SHELXT—Integrated space-group and crystal-structure determination. Acta Crystallogr. Sect. A 2015, 71, 3–8. [Google Scholar] [CrossRef]
- Sheldrick, G.M. Crystal structure refinement with SHELXL. Acta Crystallogr. Sect. C 2015, 71, 3–8. [Google Scholar] [CrossRef]
- Wierzbicki, A.; Sallis, J.D.; Stevens, E.D.; Smith, M.; Sikes, C.S. Crystal Growth and Molecular Modeling Studies of Inhibition of Struvite by Phosphocitrate. Calcif. Tissue Int. 1997, 61, 216–222. [Google Scholar] [CrossRef]
- Prywer, J.; Torzewska, A. Bacterially Induced Struvite Growth from Synthetic Urine: Experimental and Theoretical Characterization of Crystal Morphology. Cryst. Growth Des. 2009, 9, 3538–3543. [Google Scholar] [CrossRef]
- Sadowski, R.R.; Prywer, J.; Torzewska, A. Morphology of struvite crystals as an evidence of bacteria mediated growth. Cryst. Res. Technol. 2014, 49, 478–489. [Google Scholar] [CrossRef]
- Dominick, M.A.; White, M.R.; Sanderson, T.P.; Van Vleet, T.; Cohen, S.M.; Arnold, L.E.; Cano, M.; Tannehill-Gregg, S.; Moehlenkamp, J.D.; Waites, C.R.; et al. Urothelial carcinogenesis in the urinary bladder of male rats treated with muraglitazar, a PPAR alpha/gamma agonist: Evidence for urolithiasis as the inciting event in the mode of action. Toxicol. Pathol. 2006, 34, 903–920. [Google Scholar] [CrossRef] [PubMed]
- Abbona, F.; Boistelle, R. Growth morphology and crystal habit of struvite crystals (MgNH4PO4∙6 H2O). J. Cryst. Growth 1979, 46, 339–354. [Google Scholar] [CrossRef]
- Parsons, S.; Flack, H.D.; Wagner, T. Use of intensity quotients and differences in absolute structure refinement. Acta Crystallogr. Sect. B 2013, 69, 249–259. [Google Scholar] [CrossRef] [PubMed]
- Groom, C.R.; Bruno, I.J.; Lightfoot, M.P.; Ward, S.C. The Cambridge Structural Database. Acta Crystallogr. Sect. B 2016, 72, 171–179. [Google Scholar] [CrossRef] [PubMed]
- Brown, I.D. Bond-Length-Bond-Valence Relationships in Inorganic Solids. In Structure Correlation, Part III; Burgi, H.-B., Dunitz, J.D., Eds.; VCH Publishers: Weinheim, Germany; New York, NY, USA, 1994; pp. 405–429. [Google Scholar]
- Brown, I.D. Chemical and steric constraints in inorganic solids. Acta Crystallogr. Sect. B 1992, 48, 553–572. [Google Scholar] [CrossRef]
- Brown, I.D. Influence of Chemical and Spatial Constraints on the Structures of Inorganic Compounds. Acta Crystallogr. Sect. B 1997, 53, 381–393. [Google Scholar] [CrossRef]
- Gagnéa, O.C.; Hawthorne, F.C. Bond-length distributions for ions bonded to oxygen: Metalloids and post-transition metals. Acta Crystallogr. Sect. B 2015, 71, 562–578. [Google Scholar] [CrossRef]


| Formula | MgNH4PO4·6H2O |
| Formula weight | 245.42 |
| Crystal system | Orthorhombic |
| Space group | Pmn21 (No. 31) |
| a (Å) | 6.9650(2) |
| b (Å) | 6.1165(2) |
| c (Å) | 11.2056(3) |
| V (Å3) | 477.38(2) |
| Z | 2 |
| ρ (calculated) (g/cm3) | 1.707 |
| F (000) | 260 |
| μ (mm−1) | 0.389 |
| Absorption correction | Analytical numeric |
| Temperature (K) | 90 |
| Wavelength (Å) | 0.71073 |
| θ (deg) | 3.44–26.36 |
| Index range | −8 ≤ h ≤ 8, −7≤ k ≤ 7, −14≤ l ≤ 14 |
| Rint | 0.019 |
| Reflections collected | 12286 |
| Independent reflections | 1061 |
| Reflections (I > 2σ(I)) | 1060 |
| Completeness | 99.8% |
| Data/Restraints/Parameters | 1061/1/107 |
| R1/wR2 (I > 2σ(I)) | 0.0121/0.0333 |
| R1/wR2 (all data) | 0.0121/0.0333 |
| GOF on F2 | 1.14 |
| Largest diff. peak and hole (e/Å−3) | 0.24 and −0.19 |
| Weighting: w = 1/[σ2(Fo2)+(0.0229P)2+0.0413P] where P = (Fo2+ 2Fc2)/3 | |
| Bond Lengths (Å) | v.u. | Bond Angles (°) | ||
|---|---|---|---|---|
| P1-O1 | 1.5508(15) | O1-P1-O2 | 109.55(7) | |
| P1-O2 | 1.5468(13) | O1-P1-O3 | 109.55(4) | |
| P1-O3 | 1.5441(9) | O2-P1-O3 | 108.74(4) | |
| Mg1-O4 | 2.1071(14) | 0.324 | O3-P1-O3 i | 110.69(5) |
| Mg1-O5 | 2.1090(14) | 0.323 | O4-Mg1-O5 | 179.82(6) |
| Mg1-O6 | 2.0751(11) | 0.348 | O4-Mg1-O6 | 91.17(4) |
| Mg1-O7 | 2.0511(11) | 0.368 | O4-Mg1-O7 | 90.44(4) |
| O5-Mg1-O6 | 88.95(4) | |||
| O5-Mg1-O7 | 89.44(4) | |||
| O6-Mg1-O6 i | 95.28(4) | |||
| O6-Mg1-O7 | 87.66(4) | |||
| O6-Mg1-O7 i | 176.61(5) | |||
| O7-Mg1-O7 i | 89.34(5) | |||
| D-H···A | D-H (Å) | H···A (Å) | D···A (Å) | D-H···A (°) | A Symmetry Code |
|---|---|---|---|---|---|
| O4-H1···O2 | 0.81(3) | 1.87(3) | 2.676(2) | 179(3) | |
| O4-H2···O5 | 0.78(4) | 2.23(4) | 2.9986(19) | 167(3) | x,y+1,z |
| O5-H3···O3 | 0.87(2) | 1.79(2) | 2.6588(11) | 174(2) | 1/2-x,1-y,1/2+z |
| O6-H4···O3 | 0.82(2) | 1.81(2) | 2.6336(13) | 174(2) | x,y-1,z |
| O6-H5···O1 | 0.78(2) | 1.92(2) | 2.7017(13) | 173(2) | 1/2-x,1-y,1/2+z |
| O7-H6···O2 | 0.84(2) | 1.80(2) | 2.6264(12) | 172(2) | 1/2-x,1-y,1/2+z |
| O7-H7···O3 | 0.79(2) | 1.88(2) | 2.6544(12) | 168(2) | 1/2-x,3-y,1/2+z |
| N1-H8···O1 | 0.90(3) | 1.90(3) | 2.792(2) | 179(3) | x,y-1,z+1 |
| N1-H9···O7 | 0.89(4) | 2.49(4) | 3.1527(19) | 131.3(19) | |
| N1-H10···O6 | 0.88(2) | 2.137(19) | 2.9726(17) | 158.2(18) | 1/2-x,1-y,1/2+z |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Prywer, J.; Sieroń, L.; Czylkowska, A. Struvite Grown in Gel, Its Crystal Structure at 90 K and Thermoanalytical Study. Crystals 2019, 9, 89. https://doi.org/10.3390/cryst9020089
Prywer J, Sieroń L, Czylkowska A. Struvite Grown in Gel, Its Crystal Structure at 90 K and Thermoanalytical Study. Crystals. 2019; 9(2):89. https://doi.org/10.3390/cryst9020089
Chicago/Turabian StylePrywer, Jolanta, Lesław Sieroń, and Agnieszka Czylkowska. 2019. "Struvite Grown in Gel, Its Crystal Structure at 90 K and Thermoanalytical Study" Crystals 9, no. 2: 89. https://doi.org/10.3390/cryst9020089
APA StylePrywer, J., Sieroń, L., & Czylkowska, A. (2019). Struvite Grown in Gel, Its Crystal Structure at 90 K and Thermoanalytical Study. Crystals, 9(2), 89. https://doi.org/10.3390/cryst9020089







