Phase Stability and Morphology of Gel Grown Crystals: The Case of CdCl2-bpp Polymeric System
Abstract
1. Introduction
2. Materials and Methods
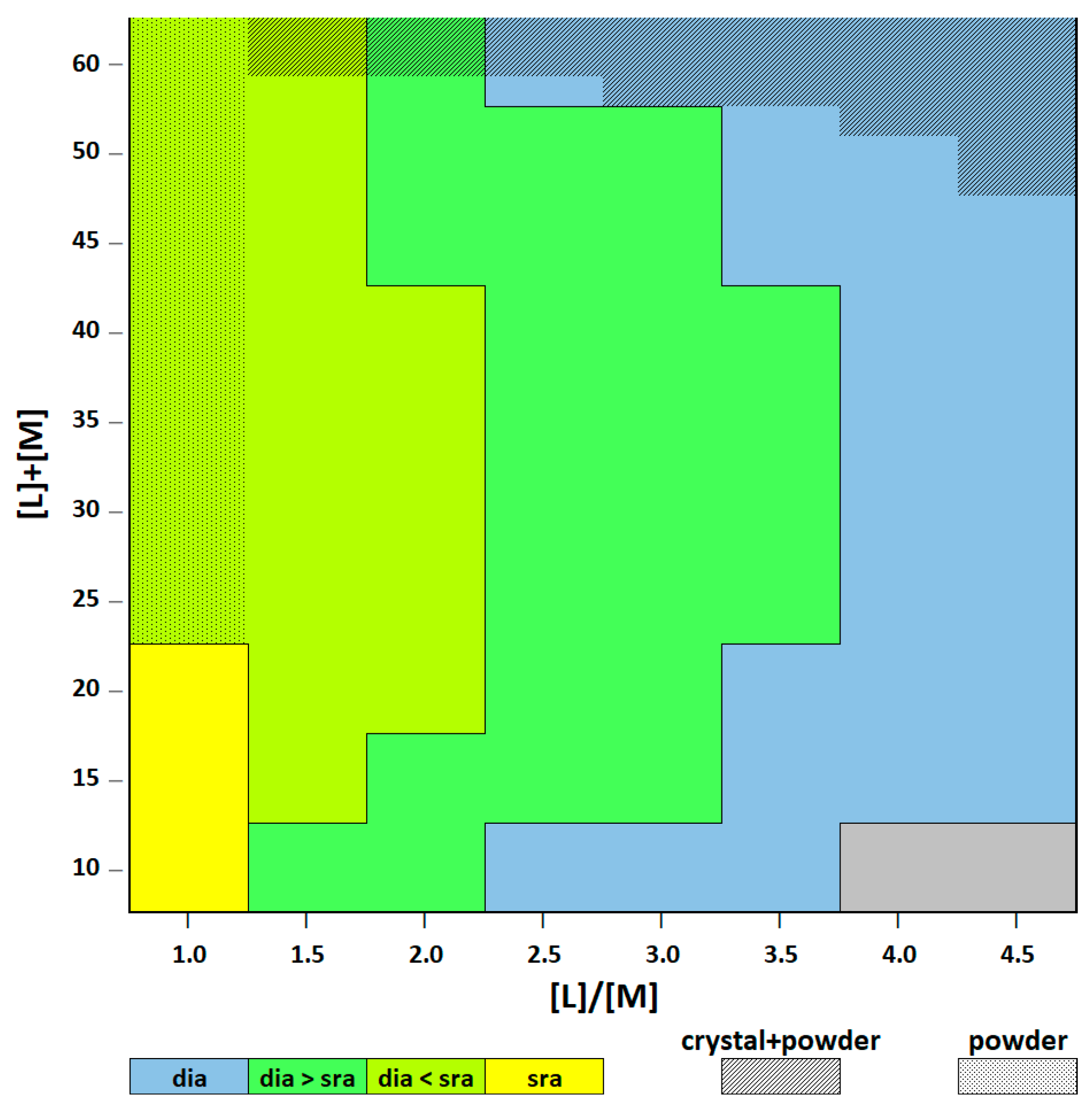
2.1. Determination of Crystal Phase Diagram (CPD) in Solution
2.2. Crystallization Screening in Gel (Single Diffusion Technique)
2.2.1. Two-Layers Method (Water-Ethanol Solvent System)
2.2.2. Three-Layers Method (Water-Ethanol Solvent System)
2.2.3. Two-Layers Method (Water-Water Solvent System)
2.3. Crystallization Screening in Gel (Double Diffusion Technique)
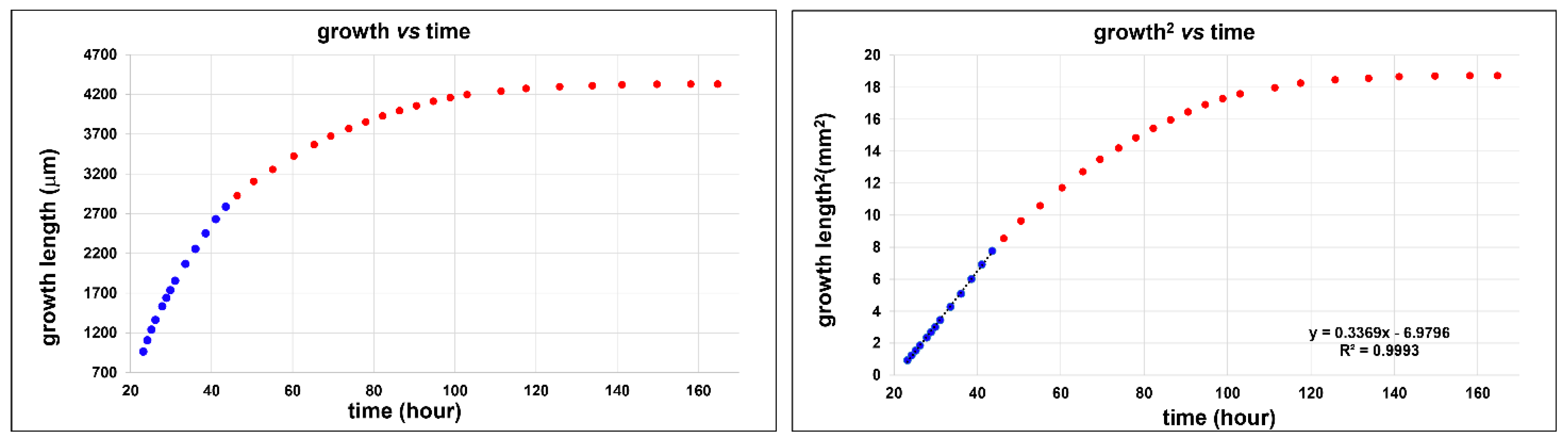
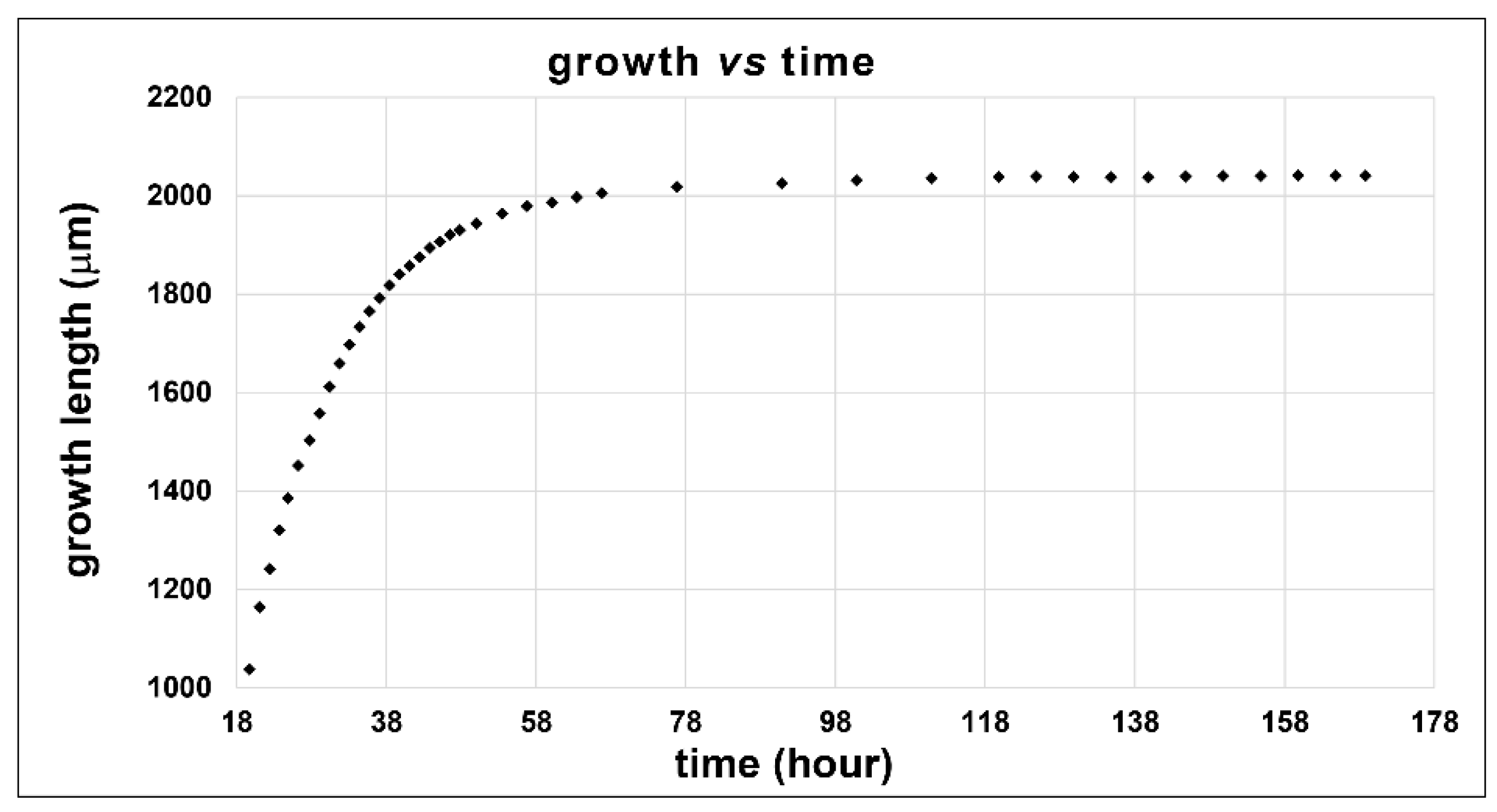
2.4. Kinetic Growth Study
3. Results and Discussion
3.1. Crystallization in Solution
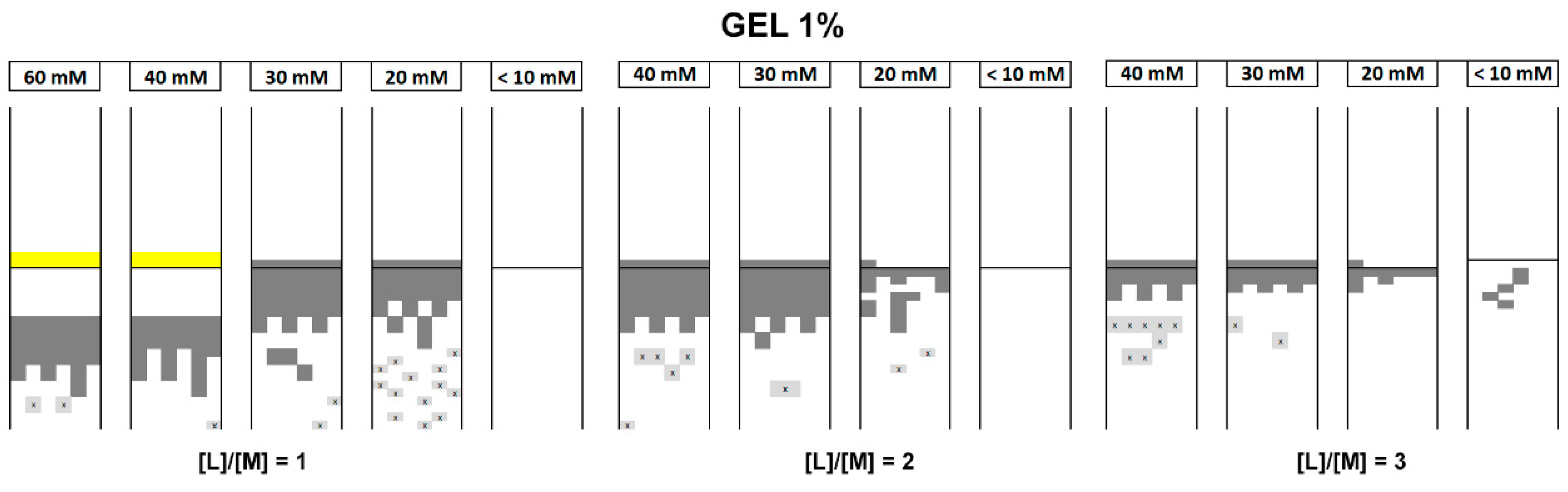
3.2. Crystallization in Gel
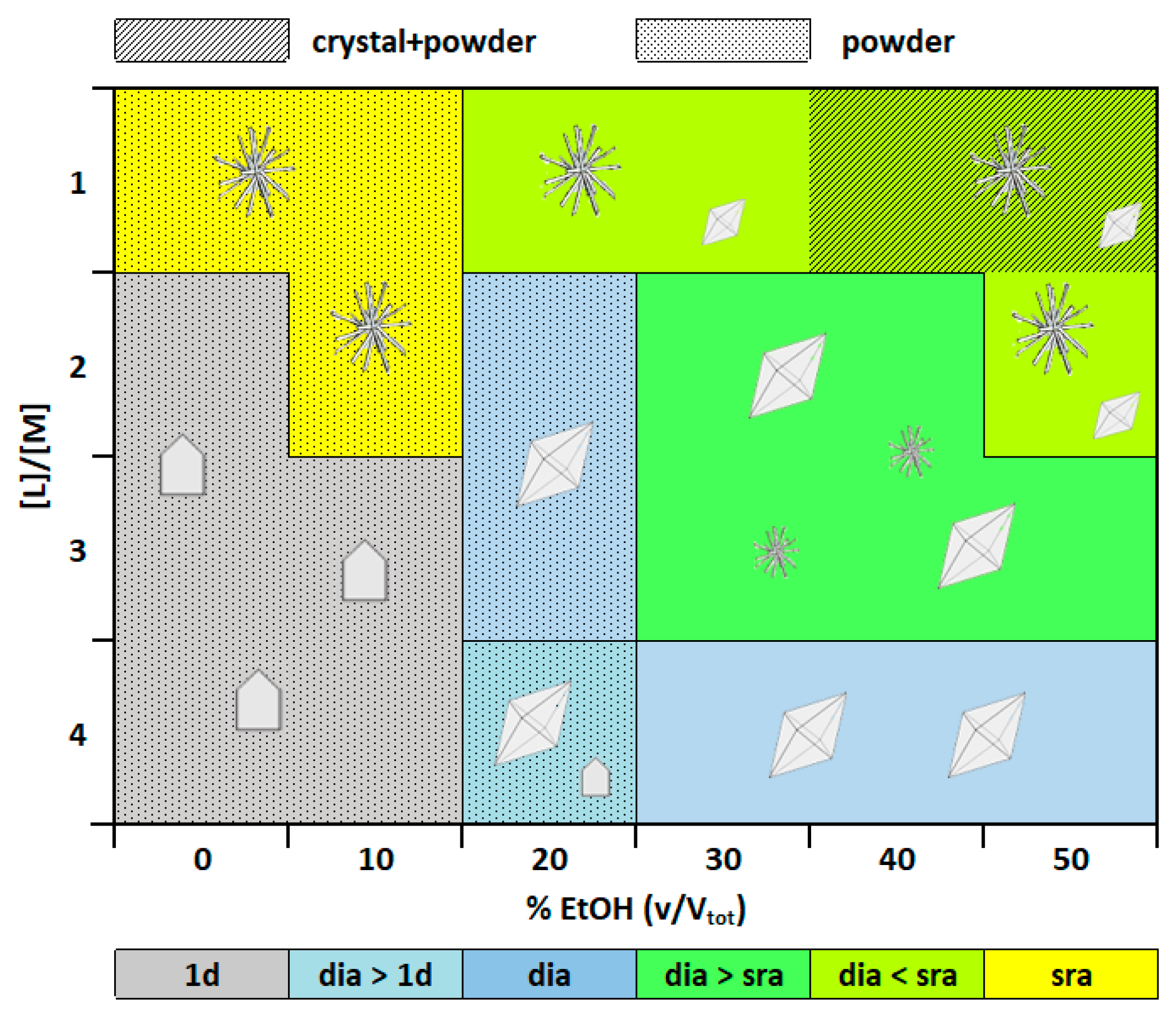
3.2.1. Crystallization in Water-Ethanol Solvent System
3.2.2. Addition of an Intermediate Layer of Pure Gel
3.2.3. Crystallization in Water-Water Solvent System
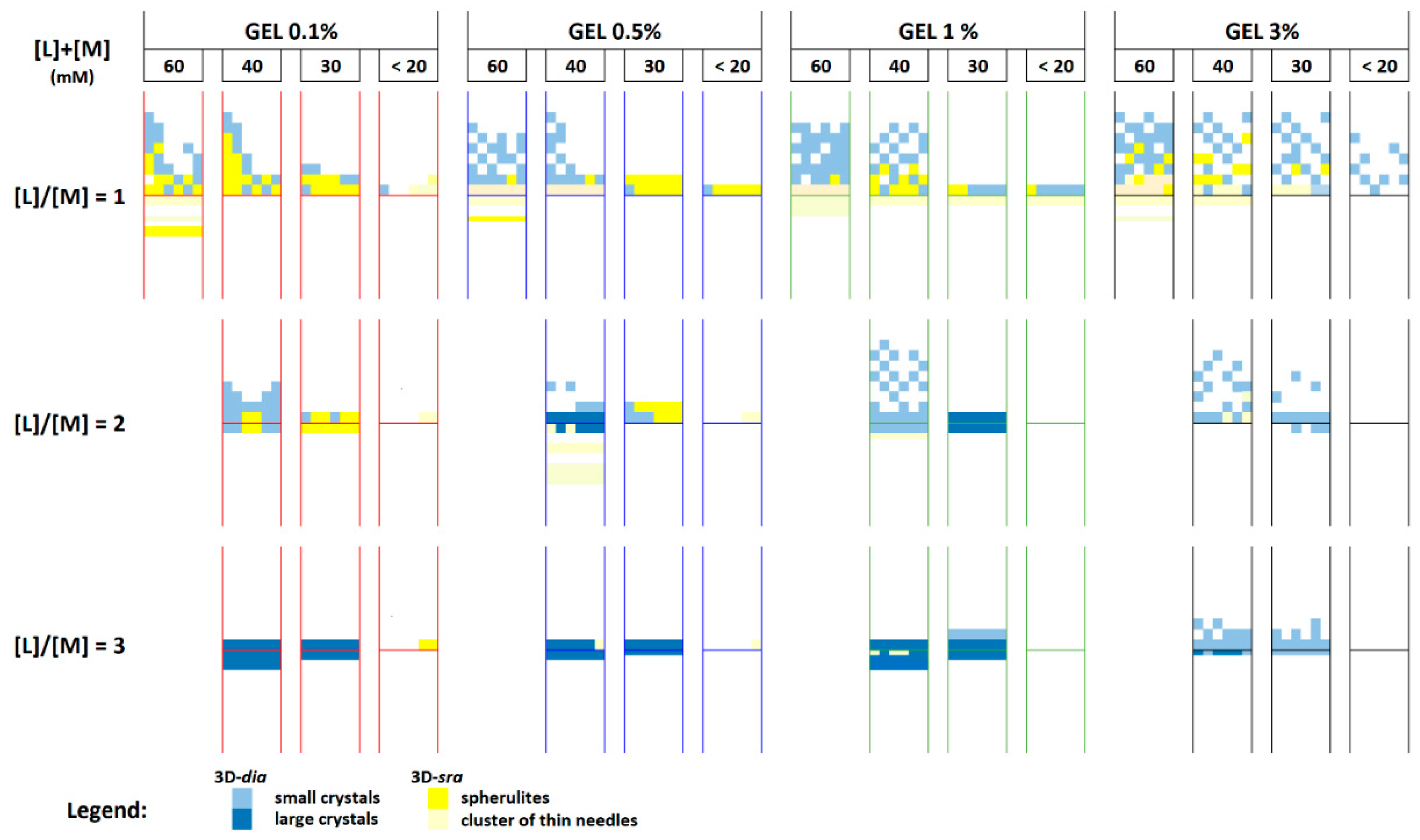
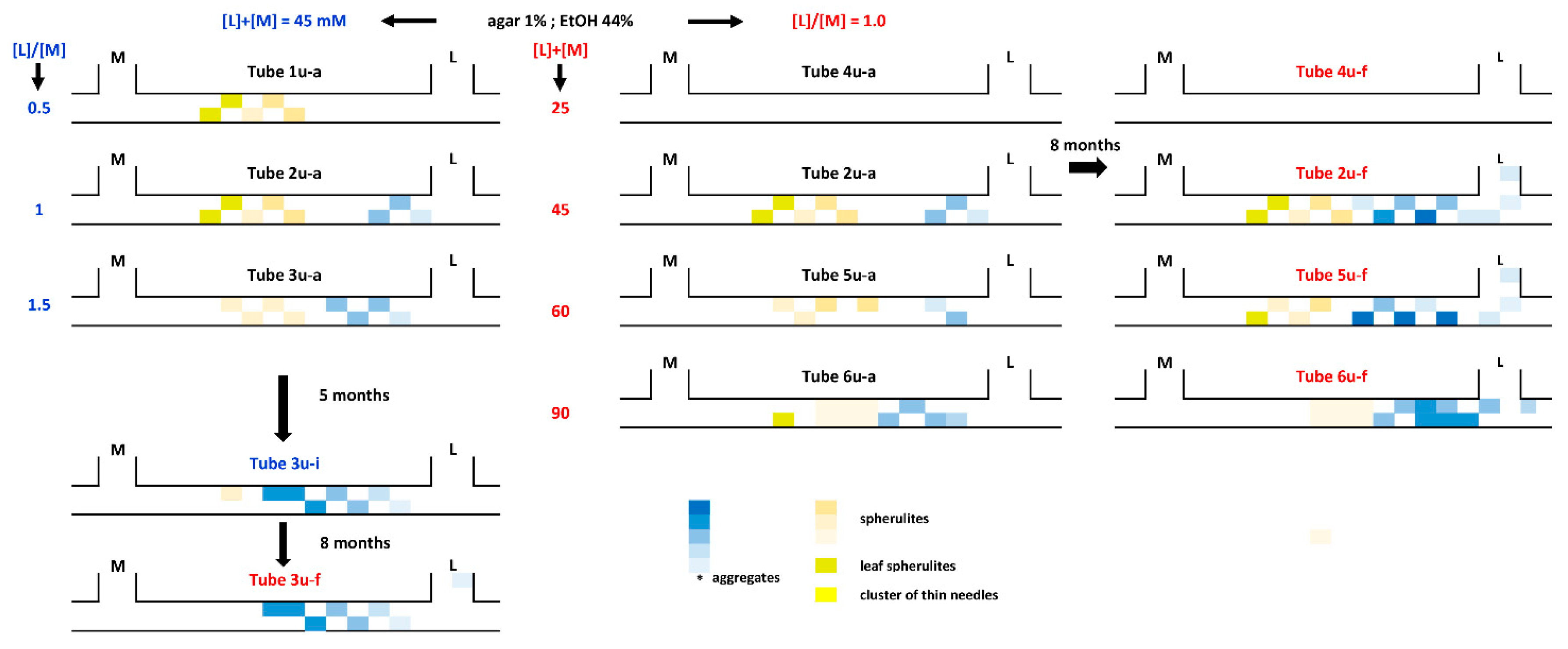
3.3. Crystallization by Double-Diffusion Method in U-Tube
3.3.1. Concentration of Reactants
3.3.2. Chemical Environment
3.3.3. Overall Parameters Analysis
3.4. Kinetics of Growth
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Gavezzotti, A. Are Crystal Structures Predictable? Acc. Chem. Res. 1994, 27, 309–314. [Google Scholar] [CrossRef]
- Neumann, M.A.; Leusen, F.J.J.; Kendrick, J. A Major Advance in Crystal Structure Prediction. Angew. Chemie Int. Ed. 2008, 47, 2427–2430. [Google Scholar] [CrossRef] [PubMed]
- Desiraju, G.R. Crystal Engineering: A Holistic View. Angew. Chemie Int. Ed. 2007, 46, 8342–8356. [Google Scholar] [CrossRef] [PubMed]
- Gavezzotti, A.; Presti, L. Lo Building Blocks of Crystal Engineering: A Large-Database Study of the Intermolecular Approach between C–H Donor Groups and O, N, Cl, or F Acceptors in Organic Crystals. Cryst. Growth Des. 2016, 16, 2952–2962. [Google Scholar] [CrossRef]
- Gavezzotti, A.; Lo Presti, L. Dynamic simulation of liquid molecular nanoclusters: Structure, stability and quantification of internal (pseudo)symmetries. New J. Chem. 2019, 43, 2077–2084. [Google Scholar] [CrossRef]
- Destro, R.; Sartirana, E.; Loconte, L.; Soave, R.; Colombo, P.; Destro, C.; Lo Presti, L. Competing C=O…C=O, C-H…O, Cl…O, and Cl…Cl interactions governing the structural phase transition of 2,6-dichloro-p- benzoquinone at T c = 122.6 K. Cryst. Growth Des. 2013, 13, 4571–4582. [Google Scholar] [CrossRef]
- Sacchi, P.; Loconte, L.; Macetti, G.; Rizzato, S.; Lo Presti, L. Correlations of Crystal Structure and Solubility in Organic Salts: The Case of the Antiplasmodial Drug Piperaquine. Cryst. Growth Des. 2019, 19, 1399–1410. [Google Scholar] [CrossRef]
- Nakamura, A.; Ishida, T.; Fushinobu, S.; Kusaka, K.; Tanaka, I.; Inaka, K.; Higuchi, Y.; Masaki, M.; Ohta, K.; Kaneko, S.; et al. Phase-diagram-guided method for growth of a large crystal of glycoside hydrolase family 45 inverting cellulase suitable for neutron structural analysis. J. Synchrotron Radiat. 2013, 20, 859–863. [Google Scholar] [CrossRef]
- Haas, C.; Drenth, J. Understanding protein crystallization on the basis of the phase diagram. J. Cryst. Growth 1999, 196, 388–394. [Google Scholar] [CrossRef]
- Govada, L.; Chayen, N. Choosing the Method of Crystallization to Obtain Optimal Results. Crystals 2019, 9, 106. [Google Scholar] [CrossRef]
- Chayen, N. Methods for separating nucleation and growth in protein crystallisation. Prog. Biophys. Mol. Biol. 2005, 88, 329–337. [Google Scholar] [CrossRef] [PubMed]
- Threlfall, T. Crystallisation of Polymorphs: Thermodynamic Insight into the Role of Solvent. Org. Process Res. Dev. 2000, 4, 384–390. [Google Scholar] [CrossRef]
- Kee, N.C.S.; Tan, R.B.H.; Braatz, R.D. Semiautomated Identification of the Phase Diagram for Enantiotropic Crystallizations using ATR-FTIR Spectroscopy and Laser Backscattering. Ind. Eng. Chem. Res. 2011, 50, 1488–1495. [Google Scholar] [CrossRef]
- Li, H.; Wang, K.; Sun, Y.; Lollar, C.T.; Li, J.; Zhou, H.-C. Recent advances in gas storage and separation using metal–organic frameworks. Mater. Today 2018, 21, 108–121. [Google Scholar] [CrossRef]
- Dhakshinamoorthy, A.; Li, Z.; Garcia, H. Catalysis and photocatalysis by metal organic frameworks. Chem. Soc. Rev. 2018, 47, 8134–8172. [Google Scholar] [CrossRef]
- Kreno, L.E.; Leong, K.; Farha, O.K.; Allendorf, M.; Van Duyne, R.P.; Hupp, J.T. Metal–Organic Framework Materials as Chemical Sensors. Chem. Rev. 2012, 112, 1105–1125. [Google Scholar] [CrossRef] [PubMed]
- Keskin, S.; Kızılel, S. Biomedical Applications of Metal Organic Frameworks. Ind. Eng. Chem. Res. 2011, 50, 1799–1812. [Google Scholar] [CrossRef]
- Northrop, B.H.; Zheng, Y.-R.; Chi, K.-W.; Stang, P.J. Self-Organization in Coordination-Driven Self-Assembly. Acc. Chem. Res. 2009, 42, 1554–1563. [Google Scholar] [CrossRef]
- Chakrabarty, R.; Mukherjee, P.S.; Stang, P.J. Supramolecular Coordination: Self-Assembly of Finite Two- and Three-Dimensional Ensembles. Chem. Rev. 2011, 111, 6810–6918. [Google Scholar] [CrossRef]
- Dey, C.; Kundu, T.; Biswal, B.P.; Mallick, A.; Banerjee, R. Crystalline metal-organic frameworks (MOFs): Synthesis, structure and function. Acta Crystallogr. Sect. B Struct. Sci. Cryst. Eng. Mater. 2014, 70, 3–10. [Google Scholar] [CrossRef]
- Zhang, B.; Zhang, J.; Liu, C.; Sang, X.; Peng, L.; Ma, X.; Wu, T.; Han, B.; Yang, G. Solvent determines the formation and properties of metal–organic frameworks. RSC Adv. 2015, 5, 37691–37696. [Google Scholar] [CrossRef]
- Moreno, A.; Rosales-Hoz, M.J. Crystal growth of inorganic, organic, and biological macromolecules in gels. Prog. Cryst. Growth Charact. Mater. 2017, 63, 63–71. [Google Scholar] [CrossRef]
- Prieto, M.; Fernández-Diaz, L.; López-Andrés, L.; López-Andrés, S. Supersaturation evolution and first precipitate location in crystal growth in gels; application to barium and strontium carbonates. J. Cryst. Growth 1989, 98, 447–460. [Google Scholar] [CrossRef]
- Garcia-Ruiz, J.M. The uses of crystal growth in gels and other diffusing-reacting systems. Key Eng. Mater. 1991, 58, 87–106. [Google Scholar] [CrossRef]
- Rizzato, S.; Moret, M.; Merlini, M.; Albinati, A.; Beghi, F. Crystal growth in gelled solution: Applications to coordination polymers. CrystEngComm 2016, 18, 2455–2462. [Google Scholar] [CrossRef]
- Carlucci, L.; Ciani, G.; Garcia-Ruiz, J.M.; Moret, M.; Proserpio, D.M.; Rizzato, S. Crystallization Behavior of Coordination Polymers. 1: Kinetic and Thermodynamic Features of 1,3-Bis(4-pyridyl)propane/MCl2 Systems. Cryst. Growth Des. 2009, 9, 5024–5034. [Google Scholar] [CrossRef]
- Robert, M.-C.; Vidal, O.; Garcia-Ruiz, J.-M.; Otalora, F. Crystallization in gels and related methods; Oxford University Press: New York, NY, USA, 1999; pp. 149–175. [Google Scholar]
- Luft, J.R.; Newman, J.; Snell, E.H. Crystallization screening: The influence of history on current practice. Acta Crystallogr. Sect. F Struct. Biol. Commun. 2014, 70, 835–853. [Google Scholar] [CrossRef]
- Sinder, M.I. Crystallization in gels. In Proceedings of the Fiz. Krist.; Kalinin. Gos. Univ.: Moscow, Russia, 1984; pp. 112–113. [Google Scholar]
- Carlucci, L.; Ciani, G.; Moret, M.; Proserpio, D.M.; Rizzato, S. Monitoring the Crystal Growth and Interconversion of New Coordination Networks in the Self-assembly of MCl2 Salts (M = Co, Ni, Cu, Cd) and 1,3-Bis(4-pyridyl)propane. Chem. Mater. 2002, 14, 12–16. [Google Scholar] [CrossRef]
- Luo, F.; Che, Y.; Zheng, J. Synthesis, structure and characteristics of one 3D 2-fold interpenetrating net with rare 42638 topology. Inorg. Chem. Commun. 2006, 9, 856–858. [Google Scholar] [CrossRef]
- Oaki, Y.; Imai, H. Experimental Demonstration for the Morphological Evolution of Crystals Grown in Gel Media. Cryst. Growth Des. 2003, 3, 711–716. [Google Scholar] [CrossRef]
- Petrova, R.I.; Swift, J.A. Habit Changes of Sodium Bromate Crystals Grown from Gel Media. Cryst. Growth Des. 2002, 2, 573–578. [Google Scholar] [CrossRef]
- Pauchet, M.; Morelli, T.; Coste, S.; Malandain, J.-J.; Coquerel, G. Crystallization of (±)-Modafinil in Gel: Access to Form I, Form III, and Twins. Cryst. Growth Des. 2006, 6, 1881–1889. [Google Scholar] [CrossRef]
- Diao, Y.; Whaley, K.E.; Helgeson, M.E.; Woldeyes, M.A.; Doyle, P.S.; Myerson, A.S.; Hatton, T.A.; Trout, B.L. Gel-induced selective crystallization of polymorphs. J. Am. Chem. Soc. 2012, 134, 673–684. [Google Scholar] [CrossRef] [PubMed]
- Estroff, L.A.; Hamilton, A.D. At the Interface of Organic and Inorganic Chemistry: Bioinspired Synthesis of Composite Materials. Chem. Mater. 2001, 13, 3227–3235. [Google Scholar] [CrossRef]
- Rizzato, S.; Moret, M.; Beghi, F.; Lo Presti, L. Crystallization and structural properties of a family of isotopological 3D-networks: The case of a 4,4′-bipy ligand–M2+ triflate system. CrystEngComm 2018, 20, 3784–3795. [Google Scholar] [CrossRef]
- Garcia-Ruiz, J.M.; Gavira, J.A.; Otalora, F.; Guasch, A.; Coll, M. Reinforced protein crystals. Mater. Res. Bull. 1998, 33, 1593–1598. [Google Scholar] [CrossRef]
- Yang, T.; Han, Y. Quantitatively Relating Diffusion and Reaction for Shaping Particles. Cryst. Growth Des. 2016, 16, 2850–2859. [Google Scholar] [CrossRef]
- Lagzi, I.; Ueyama, D. Pattern transition between periodic Liesegang pattern and crystal growth regime in reaction-diffusion systems. Chem. Phys. Lett. 2009. [Google Scholar] [CrossRef]
- Dirany, N.; Arab, M.; Moreau, A.; Valmalette, J.C.; Gavarri, J.R. Hierarchical design and control of NaCe(WO 4 ) 2 crystals: Structural and optical properties. CrystEngComm 2016, 18, 6579–6593. [Google Scholar] [CrossRef]
- Srivastava, R.; Srivastava, P.K. Nanostructured diffusion-limited-aggregation crystal pattern formation in a reactive microemulsion system. Adv. Nat. Sci. Nanosci. Nanotechnol. 2014, 5, 015018. [Google Scholar] [CrossRef][Green Version]
- Morphogenesis and Pattern Formation in Biological Systems; Sekimura, T., Noji, S., Ueno, N., Maini, P.K., Eds.; Springer Japan: Tokyo, Japan, 2003; ISBN 978-4-431-65960-0. [Google Scholar]
- Pina, C.M.; Fernández-Díaz, L.; Astilleros, J.M. Nucleation and Growth of Scheelite in a Diffusing-Reacting System. Cryst. Res. Technol. 2000, 35, 1015–1022. [Google Scholar] [CrossRef]
- Prieto, M.; Fernández-Diaz, L.; López-Andrés, S. Spatial and evolutionary aspects of nucleation in diffusing-reacting systems. J. Cryst. Growth 1991, 108, 770–778. [Google Scholar] [CrossRef]
- Desai, C.C.; Rai, J.L. Growth kinetics of tin iodide single crystals in silica gels. Surf. Technol. 1982, 15, 25–32. [Google Scholar] [CrossRef]
- Cubillas, P.; Anderson, M.W. Synthesis Mechanism: Crystal Growth and Nucleation. In Zeolites and Catalysis; Wiley-VCH Verlag GmbH & Co. KGaA: Hoboken, NJ, USA, 2010; pp. 1–55. ISBN 9783527630295. [Google Scholar]
- Raj, A.M.E.; Jayanthi, D.D.; Jothy, V.B.; Jayachandran, M.; Sanjeeviraja, C. Growth aspects of barium oxalate monohydrate single crystals in gel medium. Cryst. Res. Technol. 2008, 43, 1307–1313. [Google Scholar] [CrossRef]
- Garcia-Ruiz, J.M.; Moreno, A. Growth kinetics of protein single crystals in the gel acupuncture technique. J. Cryst. Growth 1997, 178, 393–401. [Google Scholar] [CrossRef][Green Version]
- Pyankova, L.A.; Punin, Y.O.; Bocharov, S.N.; Shtukenberg, A.G. Growth kinetics and micromorphology of NH4Cl:Mn2+ crystals formed in the NH4Cl-MnCl2-H2O-CONH3 system. Crystallogr. Rep. 2012, 57, 317–326. [Google Scholar] [CrossRef]
- Gao, P.; Gu, M.; Xiao, L.-L. Understanding the growth mechanism of CuI crystals during gel growth experiments. Cryst. Res. Technol. 2008, 43, 496–501. [Google Scholar] [CrossRef]
- Al-Ghoul, M.; Issa, R.; Hmadeh, M. Synthesis, size and structural evolution of metal–organic framework-199 via a reaction–diffusion process at room temperature. CrystEngComm 2017, 19, 608–612. [Google Scholar] [CrossRef]
- García-Ruiz, J.M.; Otálora, F.; Novella, M.L.; Gavira, J.A.; Sauter, C.; Vidal, O. A supersaturation wave of protein crystallization. J. Cryst. Growth 2001, 232, 149–155. [Google Scholar] [CrossRef]
- Arend, H.; Perison, J. The gel growth of Ag2H3IO6 crystals. Mater. Res. Bull. 1971, 6, 1205–1210. [Google Scholar] [CrossRef]
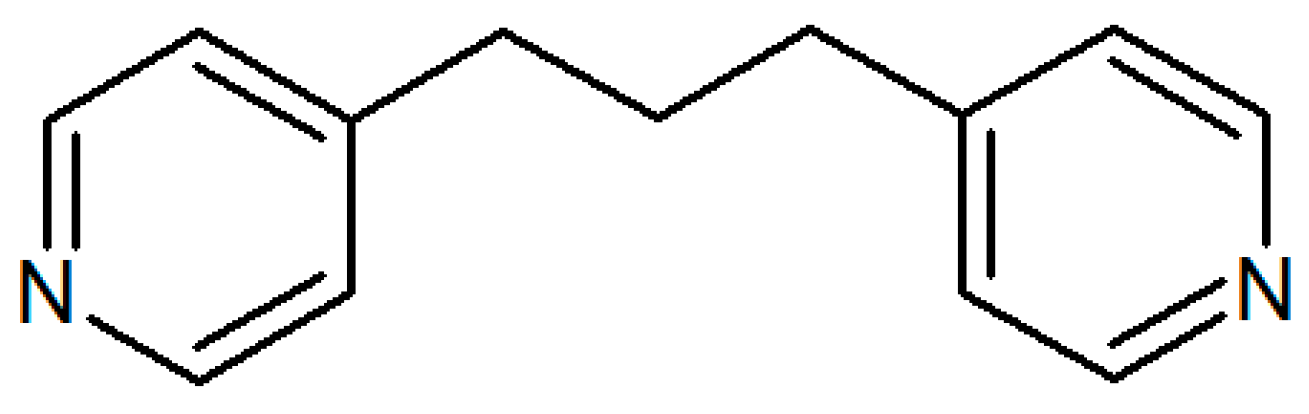




| Label | Formula | CCDC Number | DimensionAlity | Topology | Morphology | |
|---|---|---|---|---|---|---|
| 3D-dia | [Cd(bpp)2Cl2]·solv | 761818 CUPLIJ | 3D, 4-fold | dia (66) | bipyramid |  |
| 3D-sra | [Cd2(bpp)3Cl4] | 604092 REMMIG [31] | 3D, 2-fold | sra (42.63.8) | needle (aggregate) |  |
| 1D-cha | [Cd(bpp)3Cl2]·solv | 761814 CUPKOO | 1D | zig-zag chain | plate |  |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lo Presti, L.; Moret, M.; Rizzato, S. Phase Stability and Morphology of Gel Grown Crystals: The Case of CdCl2-bpp Polymeric System. Crystals 2019, 9, 363. https://doi.org/10.3390/cryst9070363
Lo Presti L, Moret M, Rizzato S. Phase Stability and Morphology of Gel Grown Crystals: The Case of CdCl2-bpp Polymeric System. Crystals. 2019; 9(7):363. https://doi.org/10.3390/cryst9070363
Chicago/Turabian StyleLo Presti, Leonardo, Massimo Moret, and Silvia Rizzato. 2019. "Phase Stability and Morphology of Gel Grown Crystals: The Case of CdCl2-bpp Polymeric System" Crystals 9, no. 7: 363. https://doi.org/10.3390/cryst9070363
APA StyleLo Presti, L., Moret, M., & Rizzato, S. (2019). Phase Stability and Morphology of Gel Grown Crystals: The Case of CdCl2-bpp Polymeric System. Crystals, 9(7), 363. https://doi.org/10.3390/cryst9070363







