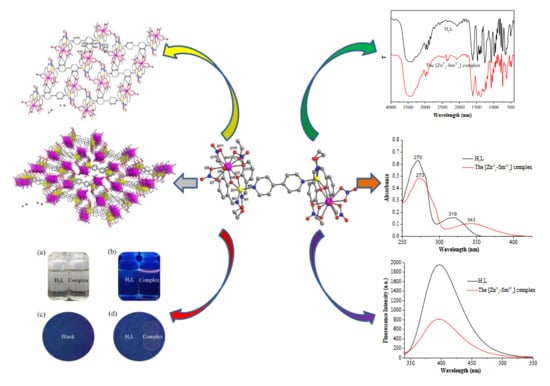
Fluorescence Properties and Density Functional Theory Calculation of a Structurally Characterized Heterotetranuclear [ZnII2–SmIII2] 4,4′-Bipy-Salamo-Constructed Complex
Abstract
1. Introduction
2. Experimental
2.1. General Details
2.2. Synthesis of H2L
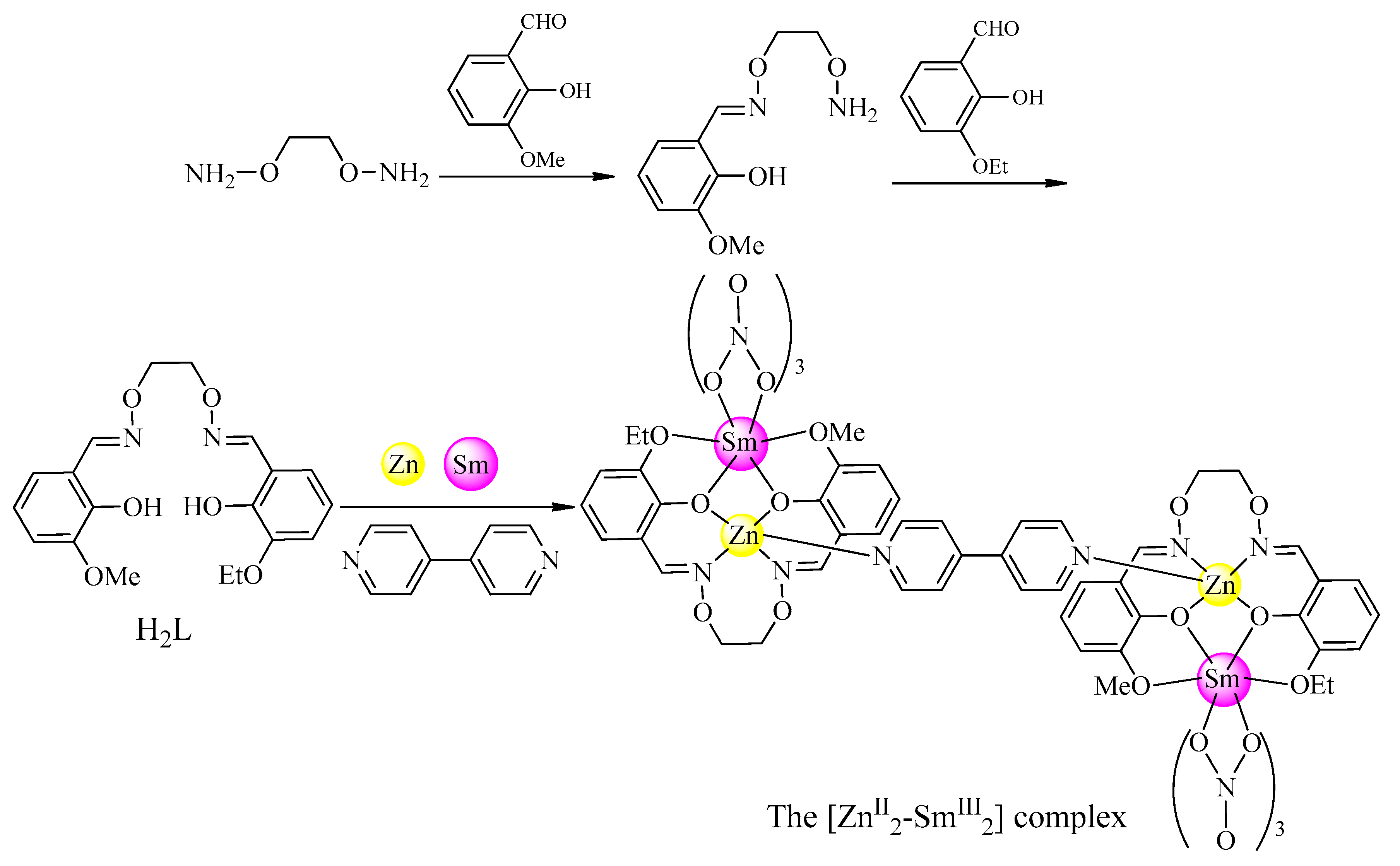
2.3. Synthesis of the [ZnII2–SmIII2] Complex
2.4. X-ray Crystallography Analysis
3. Results and Discussion
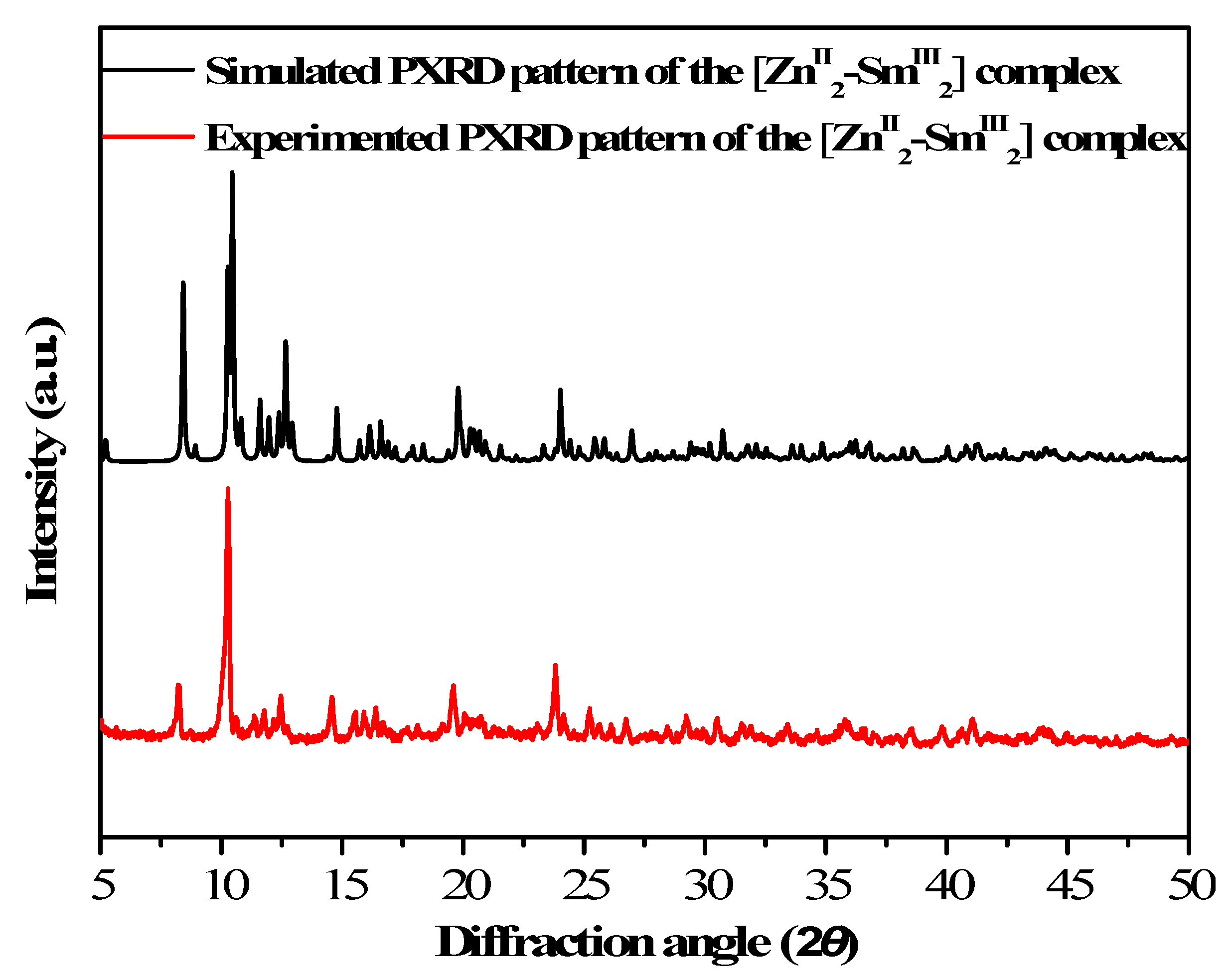
3.1. PXRD Analysis
3.2. IR Spectra
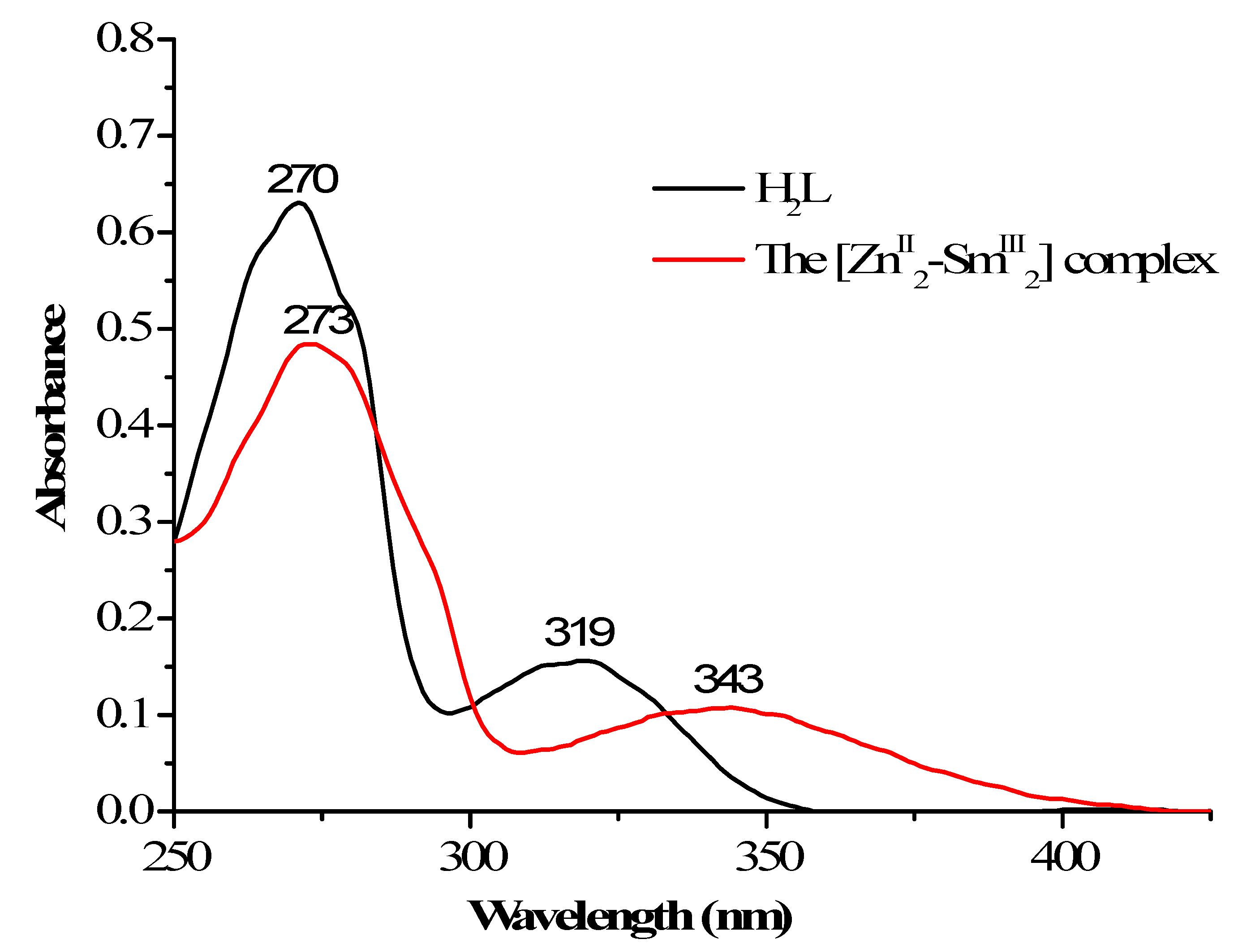
3.3. UV–Vis Spectra
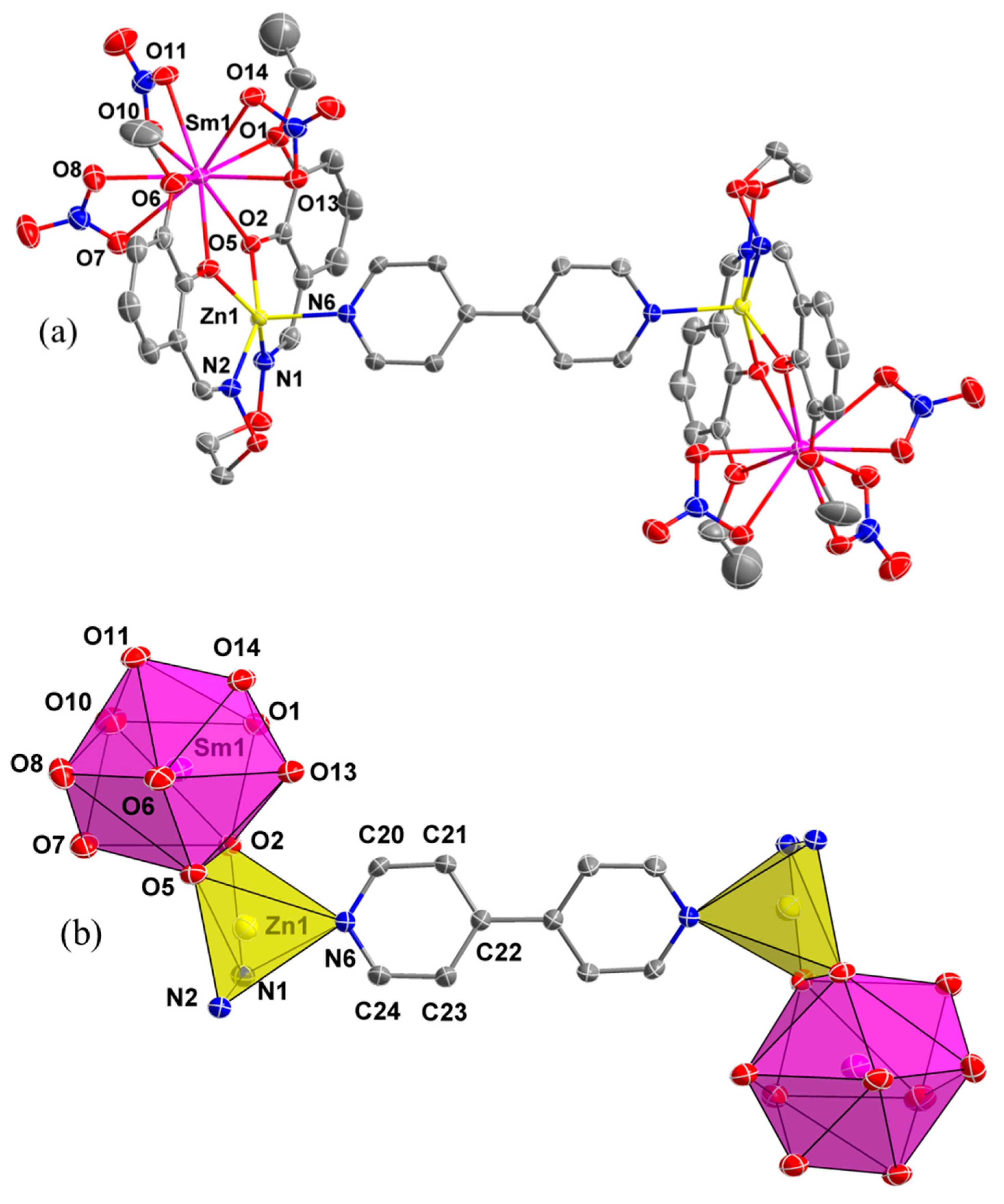
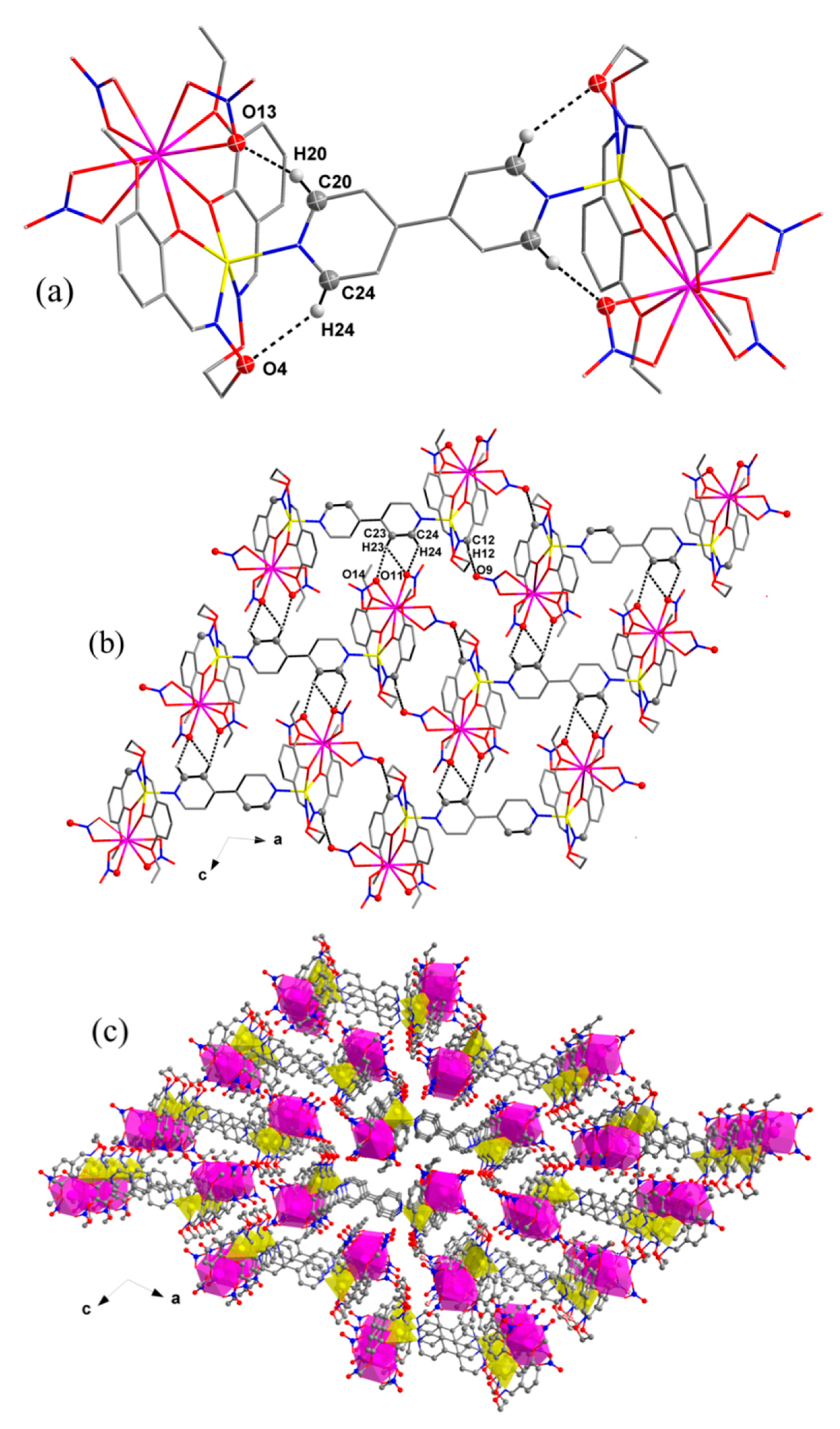
3.4. Crystal Structure and Supramolecular Interactions
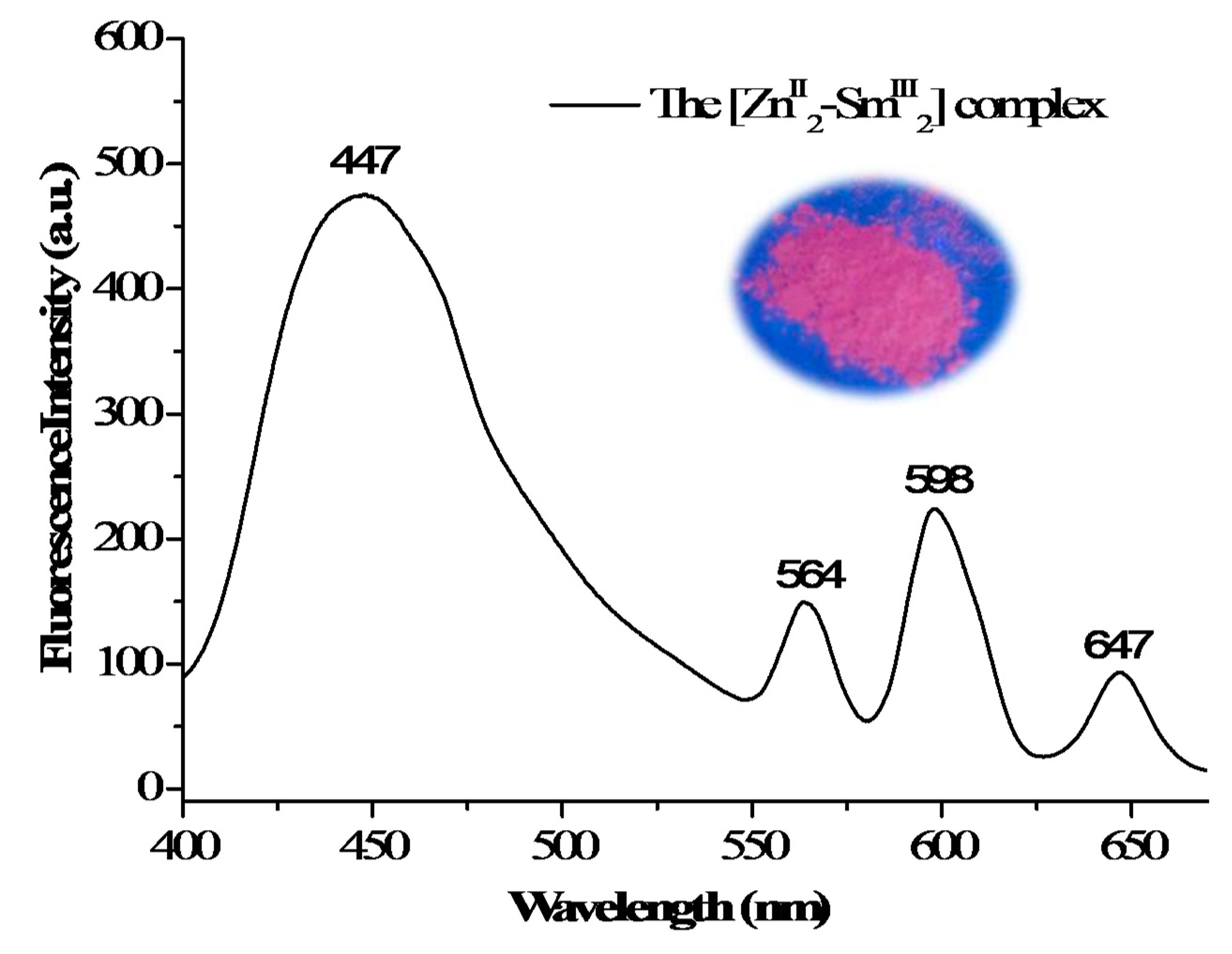
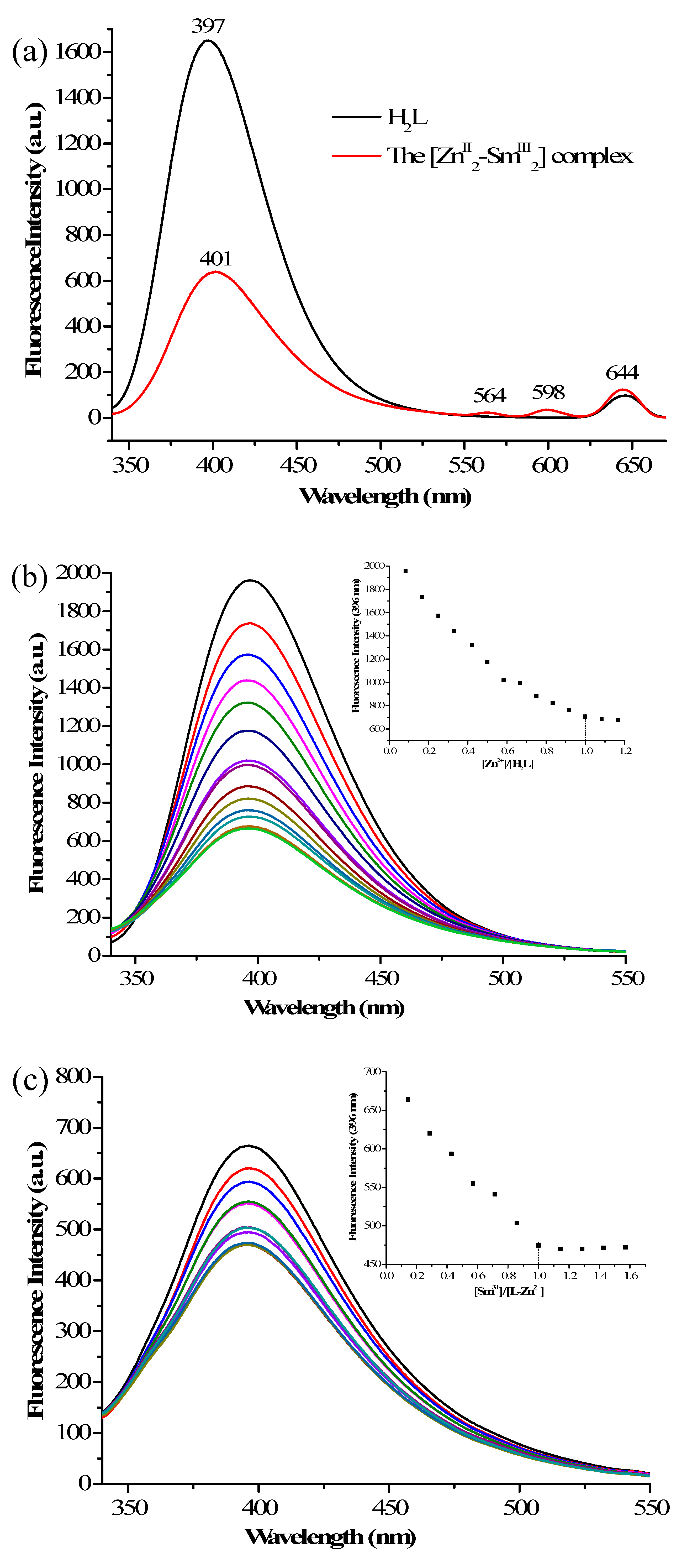
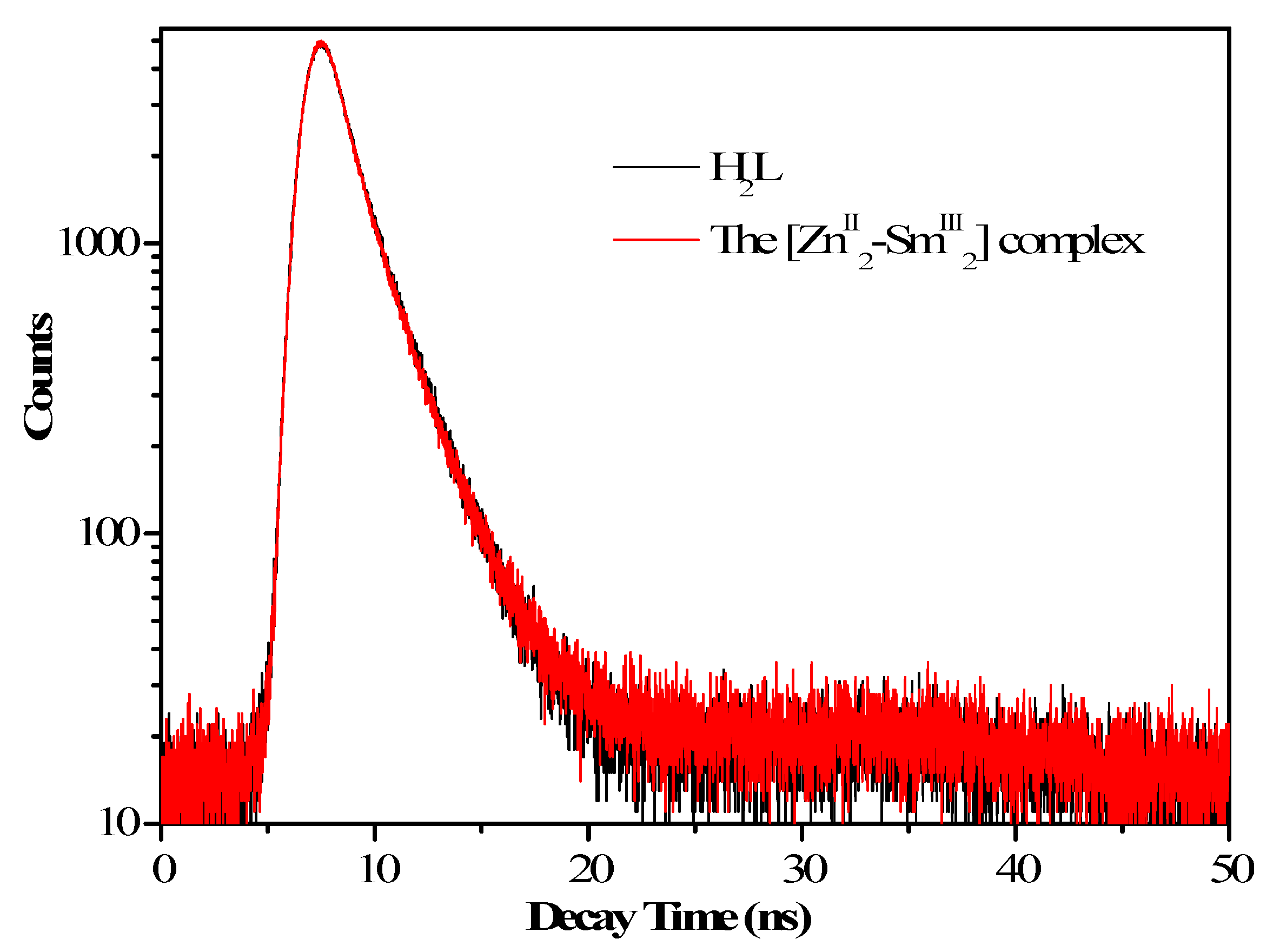
3.5. Fluorescent Properties
3.6. Fluorescence Print Imaging
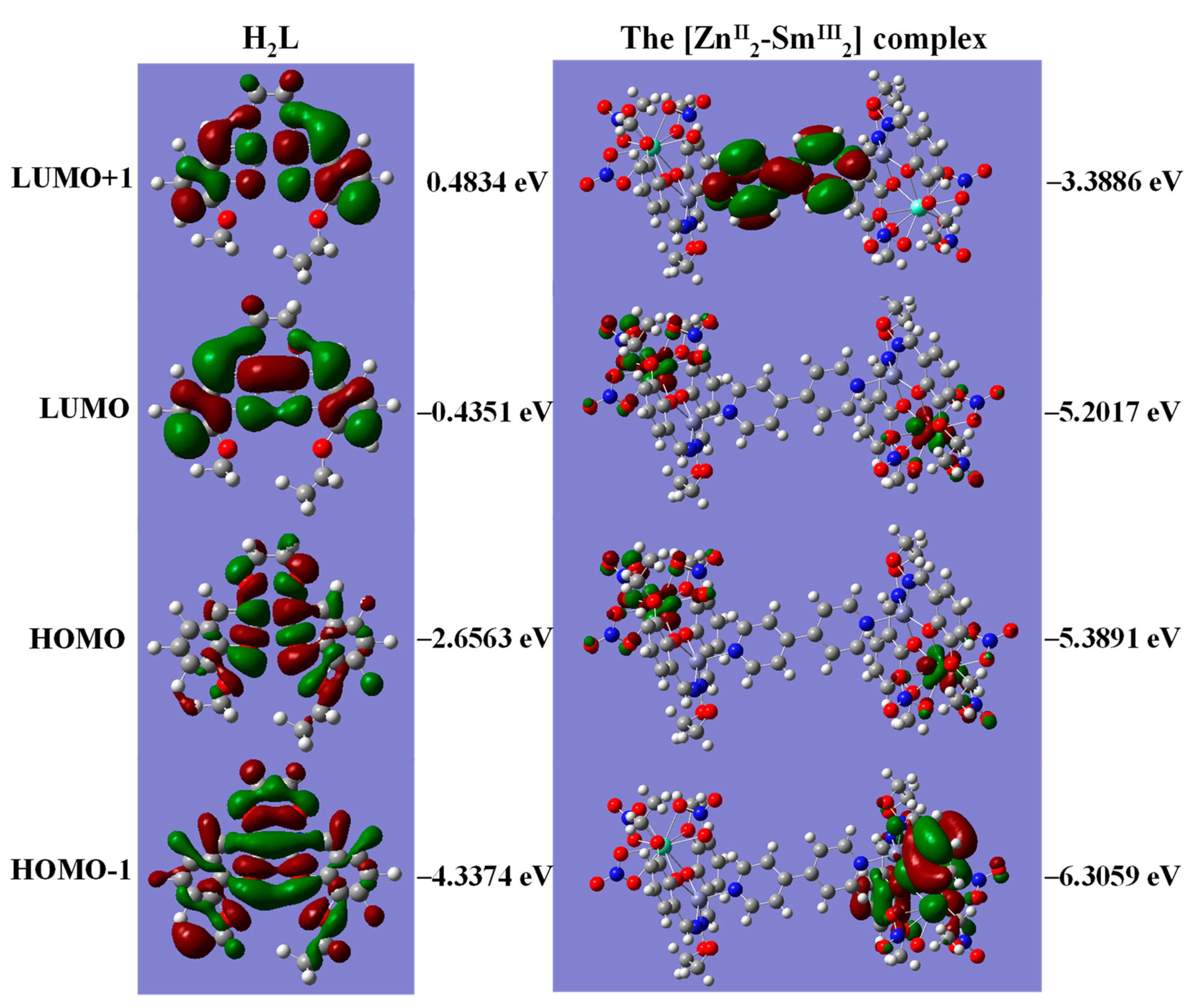
3.7. DFT Calculation
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Akine, S.; Nabeshima, T. Cyclic and acyclic oligo(N2O2) ligands for cooperative multi-metal complexation. Dalton Trans. 2009, 47, 10395–10408. [Google Scholar] [CrossRef]
- Amirkhanov, O.V.; Moroz, O.V.; Znovjyak, K.O.; Sliva, T.Y.; Penkova, L.V.; Yushchenko, T.; Szyrwiel, L.; Konovalova, I.S.; Dyakonenko, V.V.; Shishkin, O.V.; et al. Heterobinuclear Zn-Ln and Ni-Ln complexes with Schiff-base and carbacylamidophosphate ligands: Synthesis, crystal structures, and catalytic activity. Eur. J. Inorg. Chem. 2014, 23, 3720–3730. [Google Scholar] [CrossRef]
- Zhang, H.J.; Chang, J.; Jia, H.R.; Sun, Y.X. Syntheses, supramolecular structures and spectroscopic properties of Cu(II) and Ni(II) complexes with Schiff base containing oxime group. Chin. J. Inorg. Chem. 2018, 34, 2261–2270. [Google Scholar]
- Akine, S.; Taniguchi, T.; Matsumoto, T.; Nabeshima, T. Guest-dependent inversion rate of a tetranuclear single metallohelicate. Chem. Commun. 2006, 47, 4961–4963. [Google Scholar] [CrossRef] [PubMed]
- Chang, J.; Zhang, H.J.; Jia, H.R.; Sun, Y.X. Binuclear nickel(II) and zinc(II) complexes based on 2-amino-3-hydroxy-pyridine Schiff base: Syntheses, supramolecular structures and spectral properties. Chin. J. Inorg. Chem. 2018, 34, 2097–2107. [Google Scholar]
- Costes, J.P.; Donnadieu, B.; Gheorghe, R.; Novitchi, G.; Tuchagues, J.P.; Vendier, L. Di- or trinuclear 3d-4f Schiff base complexes: The role of anions. Eur. J. Inorg. Chem. 2008, 33, 5235–5244. [Google Scholar] [CrossRef]
- Guo, J.Q.; Sun, Y.X.; Yu, B.; Li, J.; Jia, H.R. Syntheses, crystal structures and spectroscopic properties of copper(II) and nickel(II) complexes with oxime-type Schiff base ligands. Chin. J. Inorg. Chem. 2017, 33, 1481–1488. [Google Scholar]
- Yu, B.; Sun, Y.X.; Yang, C.J.; Guo, J.Q.; Li, J. Synthesis and crystal structures of an unexpected tetranuclear zinc(II) complex and a benzoquinone compound derived from ZnII- and CdII-promoted reactivity of Schiff base ligands. Z. Anorg. Allg. Chem. 2017, 643, 689–698. [Google Scholar] [CrossRef]
- Song, X.Q.; Liu, P.P.; Liu, Y.A.; Zhou, J.J.; Wang, X.L. Two dodecanuclear heterometallic [Zn6Ln6] clusters constructed by a multidentate salicylamide salen-like ligand: Synthesis, structure, luminescence and magnetic properties. Dalton Trans. 2016, 45, 8154–8163. [Google Scholar] [CrossRef]
- Chin, T.K.; Endud, S.; Jamil, S.; Budagumpi, S.; Lintang, H.O. Oxidative dimerization of o-aminophenol by heterogeneous mesoporous material modified with biomimetic salen-type copper(II) complex. Catal. Lett. 2013, 143, 282–288. [Google Scholar] [CrossRef]
- Yang, X.P.; Schipper, D.; Liao, A.; Stanley, J.M.; Jones, R.A.; Holliday, B.J. Anion dependent self-assembly of luminescent Zn–Ln (Eu and Tb) salen complexes. Polyhedron 2013, 52, 165–169. [Google Scholar] [CrossRef]
- Akine, S.; Piao, S.J.; Miyashita, M.; Nabeshima, T. Cage-like tris(salen)-type metallocryptand for cooperative guest recognition. Tetrahedron Lett. 2013, 54, 6541–6544. [Google Scholar] [CrossRef]
- Li, X.Y.; Kang, Q.P.; Liu, L.Z.; Ma, J.C.; Dong, W.K. Trinuclear Co(II) and mononuclear Ni(II) salamo-type bisoxime coordination compounds. Crystals 2018, 8, 43. [Google Scholar] [CrossRef]
- Wang, F.; Liu, L.Z.; Gao, L.; Dong, W.K. Unusual constructions of two salamo-based copper(II) complexes. Spectrochim. Acta A 2018, 203, 56–64. [Google Scholar] [CrossRef] [PubMed]
- Peng, Y.D.; Wang, F.; Gao, L.; Dong, W.K. Structurally characterized dinuclear zinc(II) bis(salamo)-type tetraoxime complex possessing square pyramidal and trigonal bipyramidal geometries. J. Chin. Chem. Soc. 2018, 65, 893–899. [Google Scholar] [CrossRef]
- Kang, Q.P.; Li, X.Y.; Zhao, Q.; Ma, J.C.; Dong, W.K. Structurally characterized homotrinuclear salamo-type nickel(II) complexes: Synthesis, solvent effect and fluorescence properties. Appl. Organomet. Chem. 2018, 32, e4379. [Google Scholar] [CrossRef]
- Zhang, L.W.; Liu, L.Z.; Wang, F.; Dong, W.K. Unprecedented fluorescent dinuclear CoII and ZnII coordination compounds with a symmetric bis(salamo)-like tetraoxime. Molecules 2018, 23, 1141. [Google Scholar] [CrossRef]
- Andruh, M. Oligonuclear complexes as tectons in crystal engineering: Structural diversity and magnetic properties. Chem. Commun. 2007, 25, 2565–2577. [Google Scholar] [CrossRef]
- Zhang, L.W.; Li, X.Y.; Kang, Q.P.; Liu, L.Z.; Ma, J.C.; Dong, W.K. Structures and fluorescent and magnetic behaviors of newly synthesized NiII and CuII coordination compounds. Crystals 2018, 8, 173. [Google Scholar] [CrossRef]
- Yamashita, A.; Watanabe, A.; Akine, S.; Nabeshima, T.; Nakano, M.; Yamamura, T.; Kajiwara, T. Wheel-shaped ErIIIZnII3 single-molecule magnet: A macrocyclic approach to designing magnetic anisotropy. Angew. Chem. Int. Ed. 2011, 50, 4016–4019. [Google Scholar] [CrossRef]
- Song, X.Q.; Wang, C.Y.; Meng, H.H.; Shamshoom, A.A.A.; Liu, W.S. Coordination-driven self-assembled ZnII6-LnIII3 metallocycles based on a salicylamide imine ligand: Synthesis, structure, and selective luminescence enhancement induced by OAc−. Inorg. Chem. 2018, 57, 10873–10880. [Google Scholar] [CrossRef] [PubMed]
- Song, X.Q.; Liu, P.P.; Xiao, Z.R.; Li, X.; Liu, Y.A. Four polynuclear complexes a versatile salicylamide salen-like ligand: Synthesis, structural variations and magnetic properties. Inorg. Chim. Acta 2015, 438, 232–244. [Google Scholar] [CrossRef]
- Costes, J.P.; Yamaguchi, T.; Kojima, M.; Vendier, L. Experimental evidence for the participation of 5d GdIII orbitals in the magnetic interaction in Ni-Gd complexes. Inorg. Chem. 2009, 48, 5555–5561. [Google Scholar] [CrossRef] [PubMed]
- Yamaguchi, T.; Costes, J.P.; Kishima, Y.; Kojima, M.; Sunatsuki, Y.; Bréfuel, N.; Tuchagues, J.P.; Vendier, L.; Wernsdorfer, W. Face-sharing heterotrinuclear M(II)-Ln(III)-M(II) (M = Mn, Fe, Co, Zn; Ln = La, Gd, Tb, Dy) complexes: Synthesis, structures, and magnetic properties. Inorg. Chem. 2010, 49, 9125–9135. [Google Scholar] [CrossRef]
- Li, J.; Zhang, H.J.; Chang, J.; Jia, H.R.; Sun, Y.X.; Huang, Y.Q. Solvent-induced unsymmetric Salamo-like trinuclear NiII complexes: Syntheses, crystal structures, fluorescent and magnetic properties. Crystals 2018, 8, 176. [Google Scholar] [CrossRef]
- Li, L.H.; Dong, W.K.; Zhang, Y.; Akogun, S.F.; Xu, L. Syntheses, structures and catecholase activities of homo-and hetero-trinuclear cobalt(II) complexes constructed from an acyclic naphthalenediol-based bis(salamo)-type ligand. Appl. Organomet. Chem. 2017, 31, e3818. [Google Scholar] [CrossRef]
- Li, X.Y.; Kang, Q.P.; Liu, C.; Zhang, Y.; Dong, W.K. Structurally characterized homo-trinuclear ZnII and hetero-pentanuclear [ZnII4LnIII] complexes constructed from an octadentate bis(Salamo)-based ligand: Hirshfeld surfaces, fluorescence and catalytic properties. New J. Chem. 2019, 43, 4605–4619. [Google Scholar] [CrossRef]
- Chen, C.Y.; Zhang, J.W.; Zhang, Y.H.; Yang, Z.H.; Wu, H.L.; Pan, G.L.; Bai, Y.C. Gadolinium(III) and dysprosium(III) complexes with a Schiff base bis(N-salicylidene)-3-oxapentane-1,5-diamine: Synthesis, characterization, antioxidation, and DNA-binding studies. J. Coord. Chem. 2015, 68, 1054–1071. [Google Scholar] [CrossRef]
- Wu, H.L.; Wang, H.; Wang, X.L.; Pan, G.L.; Shi, F.R.; Zhang, Y.H.; Bai, Y.C.; Kong, J. V-shaped ligand bis(2-benzimidazolylmethyl)amine containing three copper(II) ternary complexes: Synthesis, structure, DNA binding properties and antioxidant activity. New J. Chem. 2014, 38, 1052–1061. [Google Scholar] [CrossRef]
- Zhang, H.; Xu, Y.L.; Wu, H.L.; Aderinto, S.O.; Fan, X.Y. Mono-, bi- and multi-nuclear silver complexes constructed from bis(benzimidazole)-2-oxapropane ligands and methacrylate: Syntheses, crystal structures, DNA-binding properties and antioxidant activities. RSC Adv. 2016, 6, 83697–83708. [Google Scholar] [CrossRef]
- Gao, L.; Liu, C.; Wang, F.; Dong, W.K. Tetra-, penta- and hexa-coordinated transition metal complexes constructed from coumarin-containing N2O2 ligand. Crystals 2018, 8, 77. [Google Scholar] [CrossRef]
- Chai, L.Q.; Zhang, K.Y.; Tang, L.J.; Zhang, J.Y.; Zhang, H.S. Two mono- and dinuclear Ni(II) complexes constructed from quinazoline-type ligands: Synthesis, X-ray structures, spectroscopic, electrochemical, thermal, and antimicrobial studies. Polyhedron 2017, 130, 100–107. [Google Scholar] [CrossRef]
- Chai, L.Q.; Li, Y.X.; Chen, L.C.; Zhang, J.Y.; Huang, J.J. Synthesis, X-ray structure, spectroscopic, electrochemical properties and DFT calculation of a bridged dinuclear copper(II) complex. Inorg. Chim. Acta 2016, 444, 193–201. [Google Scholar] [CrossRef]
- Ren, Z.L.; Hao, J.; Hao, P.; Dong, X.Y.; Bai, Y.; Dong, W.K. Synthesis, crystal structure, luminescence and electrochemical properties of a salamo-type trinuclear cobalt(II) complex. Z. Nat. B 2018, 73, 203–210. [Google Scholar] [CrossRef]
- Chai, L.Q.; Tang, L.J.; Chen, L.C.; Huang, J.J. Structural, spectral, electrochemical and DFT studies of two mononuclear manganese(II) and zinc(II) complexes. Polyhedron 2017, 122, 228–240. [Google Scholar] [CrossRef]
- Chai, L.Q.; Hu, Q.; Zhang, K.Y.; Chen, L.C.; Li, Y.X.; Zhang, H.S. X-ray structures, spectroscopic, electrochemical, thermal, antibacterial, and DFT studies of two nickel(II) and cobalt(III) complexes constructed from a new quinazoline-type ligand. Appl. Organomet. Chem. 2018, 32, e4426. [Google Scholar] [CrossRef]
- Chai, L.Q.; Zhou, L.; Zhang, K.Y.; Zhang, H.S. Structural characterizations, spectroscopic, electrochemical properties, and antibacterial activities of copper (II) and cobalt (II) complexes containing imidazole ring. Appl. Organomet. Chem. 2018, 32, e4576. [Google Scholar] [CrossRef]
- Nabeshima, T.; Yamamura, M. Cooperative formation and functions of multimetal supramolecular systems. Pure Appl. Chem. 2013, 85, 763–776. [Google Scholar] [CrossRef]
- Akine, S.; Hotate, S.; Nabeshima, T.A. Molecular leverage for helicity control and helix inversion. J. Am. Chem. Soc. 2011, 133, 13868–13871. [Google Scholar] [CrossRef]
- Akine, S.; Matsumoto, T.; Sairenji, S.; Nabeshima, T. Synthesis of acyclic tetrakis- and pentakis(N2O2) ligands for single-helical heterometallic complexes with a greater number of winding turns. Supramol. Chem. 2011, 23, 106–112. [Google Scholar] [CrossRef]
- Zhang, Y.; Liu, L.Z.; Peng, Y.D.; Li, N.; Dong, W.K. Structurally characterized trinuclear nickel(II) and copper(II) salamo type complexes: Syntheses, Hirshfeld analyses and fuorescent properties. Transit. Met. Chem. 2019, 44, 627–639. [Google Scholar] [CrossRef]
- Kang, Q.P.; Li, X.Y.; Wang, L.; Zhang, Y.; Dong, W.K. Containing-PMBP N2O2-donors transition metal(II) complexes: Synthesis, crystal structure, Hirshfeld surface analyses and fluorescence properties. Appl. Organomet. Chem. 2019, 33, e5013. [Google Scholar] [CrossRef]
- Liu, L.Z.; Yu, M.; Li, X.Y.; Kang, Q.P.; Dong, W.K. Syntheses, structures, Hirshfeld analyses and fluorescent properties of two Ni(II) and Zn(II) complexes constructed from a bis(salamo)-like ligand. Chin. J. Inorg. Chem. 2019, 35, 1283–1294. [Google Scholar]
- Kang, Q.P.; Li, X.Y.; Wei, Z.L.; Zhang, Y.; Dong, W.K. Symmetric containing-PMBP N2O2-donors nickel(II) complexes: Syntheses, structures, Hirshfeld analyses and fluorescent properties. Polyhedron 2019, 165, 38–50. [Google Scholar] [CrossRef]
- Chai, L.Q.; Mao, K.H.; Zhang, J.Y.; Zhang, K.Y.; Zhang, H.S. Synthesis, X-ray crystal structure, spectroscopic, electrochemical and antimicrobial studies of a new dinuclear cobalt(III) complex. Inorg. Chim. Acta 2017, 457, 34–40. [Google Scholar] [CrossRef]
- Jia, H.R.; Chang, J.; Zhang, H.J.; Li, J.; Sun, Y.X. Three polyhydroxyl-bridged defective dicubane tetranuclear MnIII complexes: Synthesis, crystal structures, and spectroscopic properties. Crystals 2018, 8, 272. [Google Scholar] [CrossRef]
- Chai, L.Q.; Hu, Q.; Zhang, K.Y.; Zhou, L.; Huang, J.J. Synthesis, structural characterization, spectroscopic, and DFT studies of two pentacoordinated zinc(II) complexes containing quinazoline and 1, 10-phenanthroline as mixed ligands. J. Lumin. 2018, 203, 234–246. [Google Scholar] [CrossRef]
- Jia, H.R.; Li, J.; Sun, Y.X.; Guo, J.Q.; Yu, B.; Wen, N.; Xu, L. Two supramolecular cobalt(II) complexes: Syntheses, crystal structures, spectroscopic behaviors, and counter anion effects. Crystals 2017, 7, 247. [Google Scholar]
- Zhou, L.; Hu, Q.; Chai, L.Q.; Mao, K.H.; Zhang, H.S. X-ray characterization, spectroscopic, DFT calculations and Hirshfeld surface analysis of two 3-D supramolecular mononuclear zinc(II) and trinuclear copper(II) complexes. Polyhedron 2019, 158, 102–116. [Google Scholar] [CrossRef]
- Sun, Y.X.; Zhao, Y.Y.; Li, C.Y.; Yu, B.; Guo, J.Q.; Li, J. Supramolecular cobalt(II) and copper(II) complexes with Schiff base ligand: Syntheses, characterizations and crystal structures. Chin. J. Inorg. Chem. 2016, 32, 913–920. [Google Scholar]
- Chai, L.Q.; Tang, L.J.; Zhang, K.Y.; Zhang, J.Y.; Zhang, H.S. Two two-dimensional supramolecular copper(II) and cobalt(III) complexes derived from a new quinazoline-type ligand: Syntheses, structures, and spectral, thermal, electrochemical and antimicrobial activity studies. Appl. Organomet. Chem. 2017, 31, e3786. [Google Scholar] [CrossRef]
- Hu, J.H.; Sun, Y.; Qi, J.; Li, Q.; Wei, T.B. A new unsymmetrical azine derivative based on coumarin group as dual-modal sensor for CN− and fluorescent “OFF-ON” for Zn2+. Spectrochim. Acta A 2017, 175, 125–133. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Hu, J.H.; Qi, J.; Li, J.B. A highly selective colorimetric and “turn-on” fluorimetric chemosensor for detecting CN− based on unsymmetrical azine derivatives in aqueous media. Spectrochim. Acta A 2016, 167, 101–105. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.Z.; Wang, L.; Yu, M.; Zhao, Q.; Zhang, Y.; Sun, Y.X.; Dong, W.K. A highly sensitive and selective fluorescent “off-on-off” relay chemosensor based on a new bis(salamo)-type tetraoxime for detecting Zn2+ and CN−. Spectrochim. Acta A 2019, 222, 117209. [Google Scholar] [CrossRef] [PubMed]
- Hao, J.; Li, X.Y.; Zhang, Y.; Dong, W.K. A reversible bis(salamo)-based fluorescence sensor for selective detection of Cd2+ in water-containing systems and food samples. Materials 2018, 11, 523. [Google Scholar] [CrossRef] [PubMed]
- Yu, B.; Li, C.Y.; Sun, Y.X.; Jia, H.R.; Guo, J.Q.; Li, J. A new azine derivative colorimetric and fluorescent dual-channel probe for cyanide detection. Spectrochim. Acta A 2017, 184, 249–254. [Google Scholar] [CrossRef] [PubMed]
- Liao, S.; Yang, X.P.; Jones, R.A. Self-assembly of luminescent hexanuclear lanthanide salen complexes. Cryst. Growth Des. 2012, 12, 970–974. [Google Scholar] [CrossRef]
- Wang, B.; Zang, Z.P.; Wang, H.H.; Dou, W.; Tang, X.L.; Liu, W.S.; Shao, Y.L.; Ma, J.X.; Li, Y.Z.; Zhou, J. Multiple lanthanide helicate clusters and the effects of anions on their configuration. Angew. Chem. 2013, 125, 3844–3847. [Google Scholar] [CrossRef]
- Feng, W.X.; Zhang, Y.; Zhang, Z.; Lü, X.Q.; Liu, H.; Shi, G.X.; Zou, D.; Song, J.R.; Fan, D.D.; Wong, W.K.; et al. Anion-induced self-assembly of luminescent and magnetic homoleptic cyclic tetranuclear Ln4(Salen)4 and Ln4(Salen)2 complexes (Ln = Nd, Yb, Er, or Gd). Inorg. Chem. 2012, 51, 11377–11386. [Google Scholar] [CrossRef]
- Zhao, Q.; An, X.X.; Liu, L.Z.; Dong, W.K. Syntheses, luminescences and Hirshfeld surfaces analyses of structurally characterized homo-trinuclear ZnII and hetero-pentanuclear ZnII-LnIII (Ln=Eu, Nd) bis(salamo)-like complexes. Inorg. Chim. Acta 2019, 490, 6–15. [Google Scholar] [CrossRef]
- Akine, S.; Utsuno, F.; Taniguchi, T.; Nabeshima, T. Dinuclear complexes of the N2O2 oxime chelate ligand with Zinc(II)–Lanthanide(III) as a selective sensitization system for Sm3+. Eur. J. Inorg. Chem. 2010, 20, 3143–3152. [Google Scholar] [CrossRef]
- Song, X.Q.; Liu, P.P.; Wang, C.Y.; Liu, Y.A.; Liu, W.S.; Zhang, M. Three sandwich-type zinc(II)–lanthanide(III) clusters: Structures, luminescence and magnetic properties. RSC Adv. 2017, 7, 22692–22698. [Google Scholar] [CrossRef]
- Pasatoiu, T.D.; Tiseanu, C.; Madalan, A.M.; Jurca, B.; Duhayon, C.; Sutter, J.P.; Andruh, M. Study of the luminescent and magnetic properties of a series of heterodinuclear [Zn(II)Ln(III)] complexes. Inorg. Chem. 2011, 50, 5879–5889. [Google Scholar] [CrossRef] [PubMed]
- Pasatoiu, T.D.; Madalan, A.M.; Kumke, M.U.; Tiseanu, C.; Andruh, M. Study of the luminescent and magnetic properties of a series of heterodinuclear [ZnIILnIII] complexes. Inorg. Chem. 2010, 49, 2310–2315. [Google Scholar] [CrossRef] [PubMed]
- Dong, W.K.; Ma, J.C.; Zhu, L.C.; Sun, Y.X.; Akogun, S.F.; Zhang, Y. A series of heteromultinuclear Zinc(II)-Lanthanide(III) complexes based on 3-MeOsalamo: Syntheses, structural characterizations, and luminescent properties. Cryst. Growth Des. 2016, 16, 6903–6914. [Google Scholar] [CrossRef]
- An, X.X.; Zhao, Q.; Mu, H.R.; Dong, W.K. A new half-salamo-based homo-trinuclear nickel(II) complex: Crystal structure, Hirshfeld surface analysis, and fluorescence properties. Crystals 2019, 9, 101. [Google Scholar] [CrossRef]
- Peng, Y.D.; Li, X.Y.; Kang, Q.P.; An, G.X.; Zhang, Y.; Dong, W.K. Synthesis and fluorescence properties of asymmetrical Salamo-type tetranuclear zinc(II) complex. Crystals 2018, 8, 107. [Google Scholar] [CrossRef]
- Hu, J.H.; Li, J.B.; Qi, J.; Sun, Y. Acylhydrazone based fluorescent chemosensor for zinc in aqueous solution with high selectivity and sensitivity. Sens. Actuators B 2015, 208, 581–587. [Google Scholar] [CrossRef]
- Hao, J.; Li, X.Y.; Wang, L.; Zhang, Y.; Dong, W.K. Luminescent and electrochemical properties of four novel butterfly-shaped hetero-pentanuclear [Zn4Ln] complexes constructed from a bis(salamo)-type ligand. Spectrochim. Acta A 2018, 204, 388–402. [Google Scholar] [CrossRef]
- Dong, X.Y.; Kang, Q.P.; Li, X.Y.; Ma, J.C.; Dong, W.K. Structurally characterized solvent-induced homotrinuclear cobalt(II) N2O2-donor bisoxime-type complexes. Crystals 2018, 8, 139. [Google Scholar] [CrossRef]
- Brubaker, C.D.; Frecker, T.M.; McBride, J.R.; Reid, K.R.; Jennings, G.K.; Rosenthal, S.J.; Adams, D.E. Incorporation of fluorescent quantum dots for 3D printing and additive manufacturing applications. J. Mater. Chem. C 2018, 6, 7584–7593. [Google Scholar] [CrossRef]
- Kim, B.H.; Onses, M.S.; Lim, J.B.; Nam, S.; Oh, N.; Kim, H.; Yu, K.J.; Lee, J.W.; Kim, J.H.; Kang, S.K.; et al. High-resolution patterns of quantum dots formed by electrohydrodynamic jet printing for light-emitting diodes. Nano Lett. 2015, 15, 969–973. [Google Scholar] [CrossRef] [PubMed]
- Kumar, P.; Singh, S.; Gupta, B.K. Future prospects of luminescent nanomaterial based security inks: From synthesis to anti-counterfeiting applications. Nanoscale 2016, 8, 14297–14340. [Google Scholar] [CrossRef] [PubMed]

| Compound | The [ZnII2–SmIII2] Complex |
|---|---|
| Formula | C50H56N12O32Sm2Zn2 |
| Formula weight | 1768.50 |
| Temperature (K) | 273.15 |
| Wavelength (Å) | 0.71073 |
| Crystal system | monoclinic |
| Space group | C 2/c |
| a (Å) | 36.6271(14) |
| b (Å) | 8.8968(3) |
| c (Å) | 21.4485(8) |
| α (°) | 90.00 |
| β (°) | 112.6940(10) |
| γ (°) | 90.00 |
| V (Å3) | 6448.2(4) |
| Z | 4 |
| Dcalc (g∙cm–3) | 2.631 |
| µ (mm–1) | 3.325 |
| F (000) | 3520.0 |
| Crystal size (mm) | 0.15 × 0.11× 0.09 |
| θ Range (°) | 2.5–26.0 |
| Index ranges | –44 ≤ h ≤ 44, |
| –10 ≤ k ≤10, | |
| –26 ≤ l ≤ 26 | |
| Reflections collected | 25,740 |
| Independent reflections | 6310 |
| Rint | 0.0291 |
| Completeness to θ | 99.7 % (θ = 26.0) |
| Data/restraints/parameters | 6310/100/473 |
| GOF | 1.036 |
| R [I > 2σ(I)] | R1 = 0.0333, wR2 = 0.0747 |
| Largest differences peak and hole (e Å−3) | 0.92/−0.64 |
| Compound | ν(O-H) | ν(C=N) | ν(Ar–O) | ν(C=C) |
|---|---|---|---|---|
| H2L | 3427 | 1613 | 1250 | 1468 |
| The [ZnII2–SmIII2] complex | 3400 | 1607 | 1216 | 1461 |
| D–H···A | d(D–H) | d(H···A) | d(D···A) | ∠D–H···A | Symmetry Code |
|---|---|---|---|---|---|
| C20–H9B···O13 | 0.93 | 2.38 | 2.996(6) | 123 | |
| C24–H24···O4 | 0.93 | 2.49 | 3.298(6) | 146 | |
| C12–H12···O9 | 0.93 | 0.93 | 3.291(6) | 167 | 1/2 − x, −1/2 − y, 1/2 − z |
| C23–H23···O11 C23–H23···O14 C24–H24···O11 | 0.93 0.93 0.93 | 2.59 2.53 2.47 | 3.162(6) 3.354(5) 3.112(5) | 120 148 127 | x, 1 − y, −1/2 + z x, 1 − y, −1/2 + z x, 1 − y, −1/2 + z |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
An, X.-X.; Liu, C.; Chen, Z.-Z.; Xie, K.-F.; Zhang, Y. Fluorescence Properties and Density Functional Theory Calculation of a Structurally Characterized Heterotetranuclear [ZnII2–SmIII2] 4,4′-Bipy-Salamo-Constructed Complex. Crystals 2019, 9, 602. https://doi.org/10.3390/cryst9110602
An X-X, Liu C, Chen Z-Z, Xie K-F, Zhang Y. Fluorescence Properties and Density Functional Theory Calculation of a Structurally Characterized Heterotetranuclear [ZnII2–SmIII2] 4,4′-Bipy-Salamo-Constructed Complex. Crystals. 2019; 9(11):602. https://doi.org/10.3390/cryst9110602
Chicago/Turabian StyleAn, Xiao-Xin, Chang Liu, Zhuang-Zhuang Chen, Ke-Feng Xie, and Yang Zhang. 2019. "Fluorescence Properties and Density Functional Theory Calculation of a Structurally Characterized Heterotetranuclear [ZnII2–SmIII2] 4,4′-Bipy-Salamo-Constructed Complex" Crystals 9, no. 11: 602. https://doi.org/10.3390/cryst9110602
APA StyleAn, X.-X., Liu, C., Chen, Z.-Z., Xie, K.-F., & Zhang, Y. (2019). Fluorescence Properties and Density Functional Theory Calculation of a Structurally Characterized Heterotetranuclear [ZnII2–SmIII2] 4,4′-Bipy-Salamo-Constructed Complex. Crystals, 9(11), 602. https://doi.org/10.3390/cryst9110602