Mechanical Properties of Geopolymers Synthesized from Fly Ash and Red Mud under Ambient Conditions
Abstract
1. Introduction
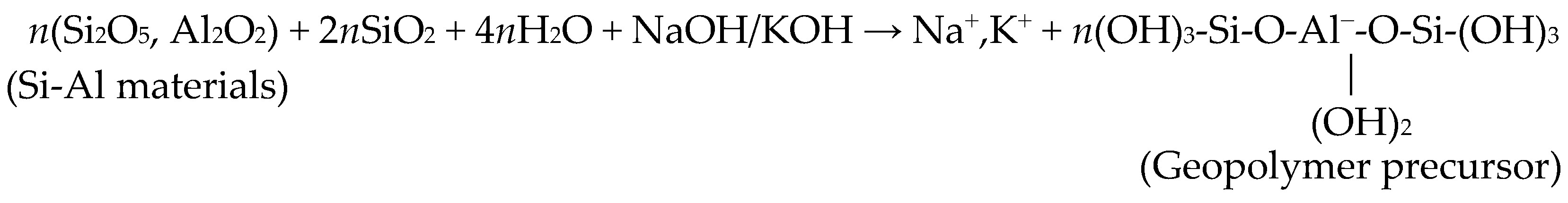
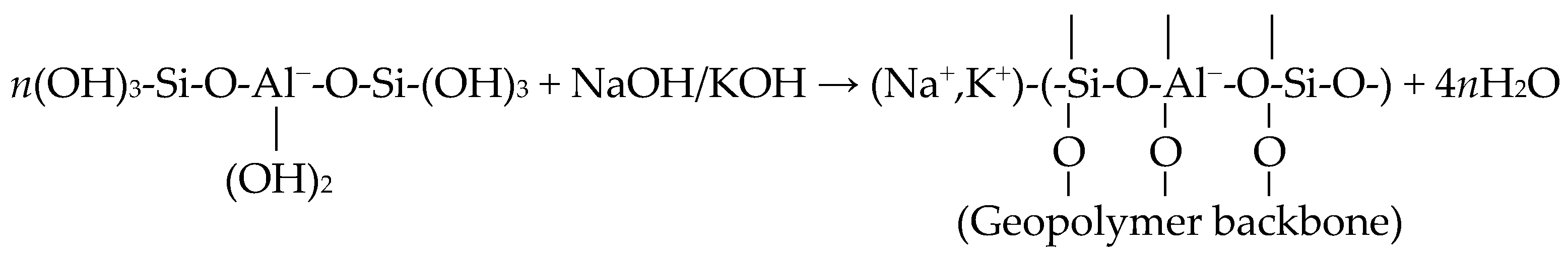
2. Materials and Methods
2.1. Materials Used
2.2. Experimental Procedure
2.3. Characterization
3. Results and Discussion
3.1. Characterization of Fly Ash and Red Mud
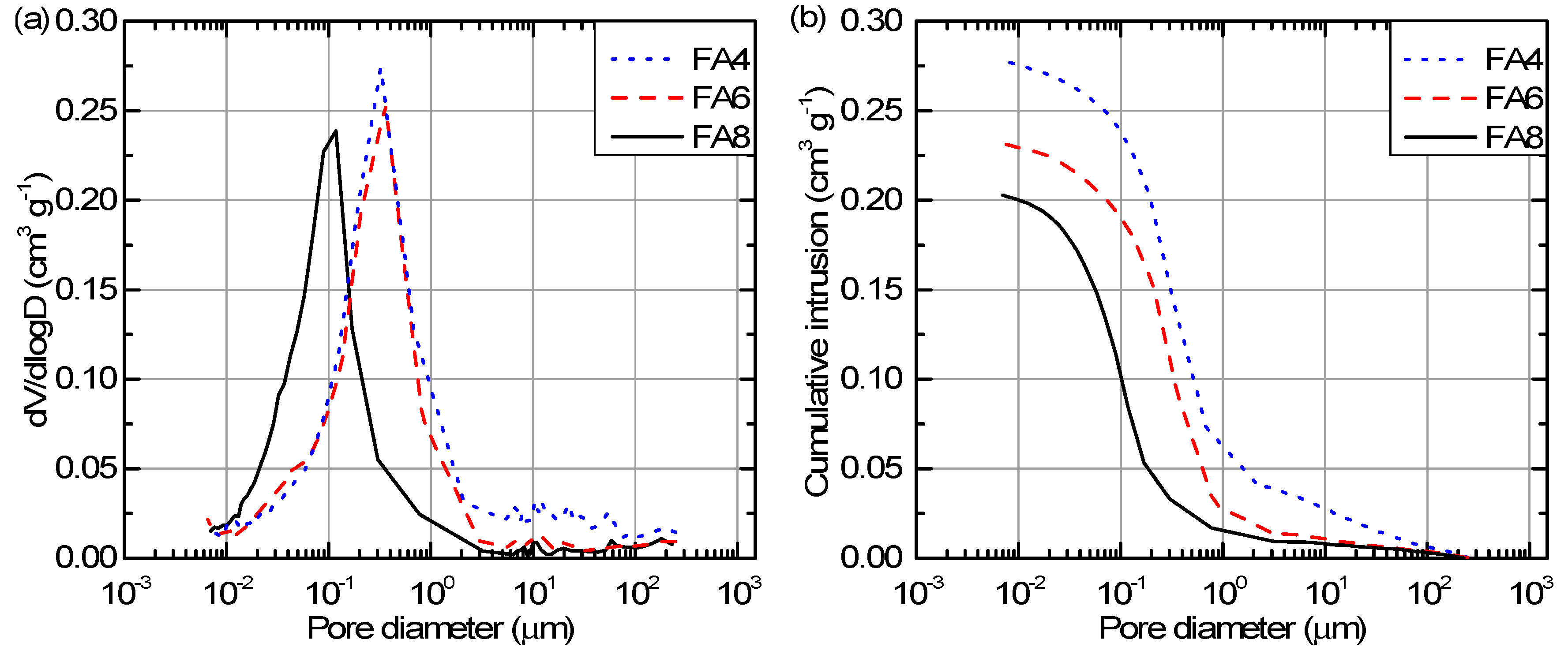
3.2. Pore Characteristics
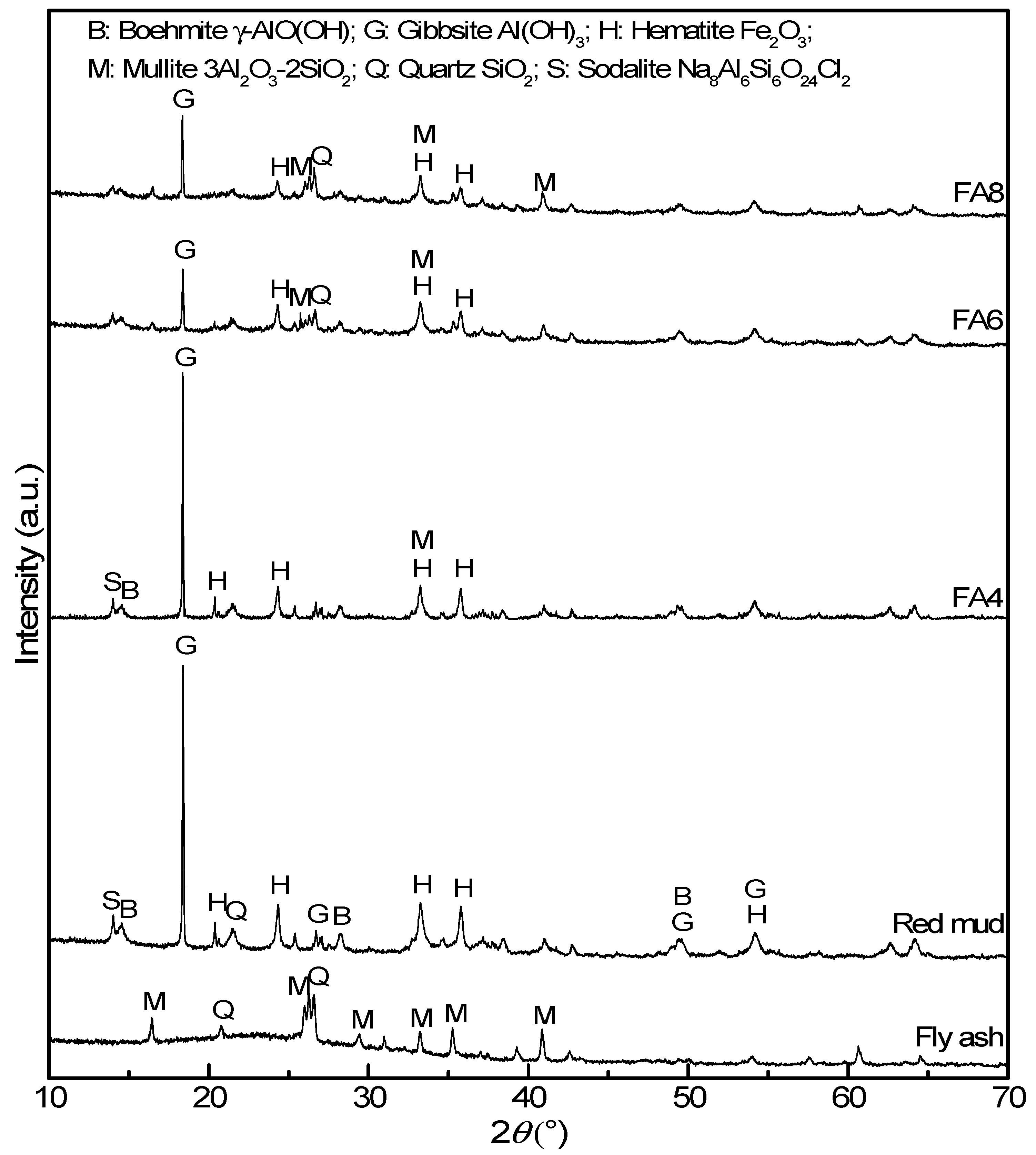
3.3. Microstructural and Mineralogical Characteristics
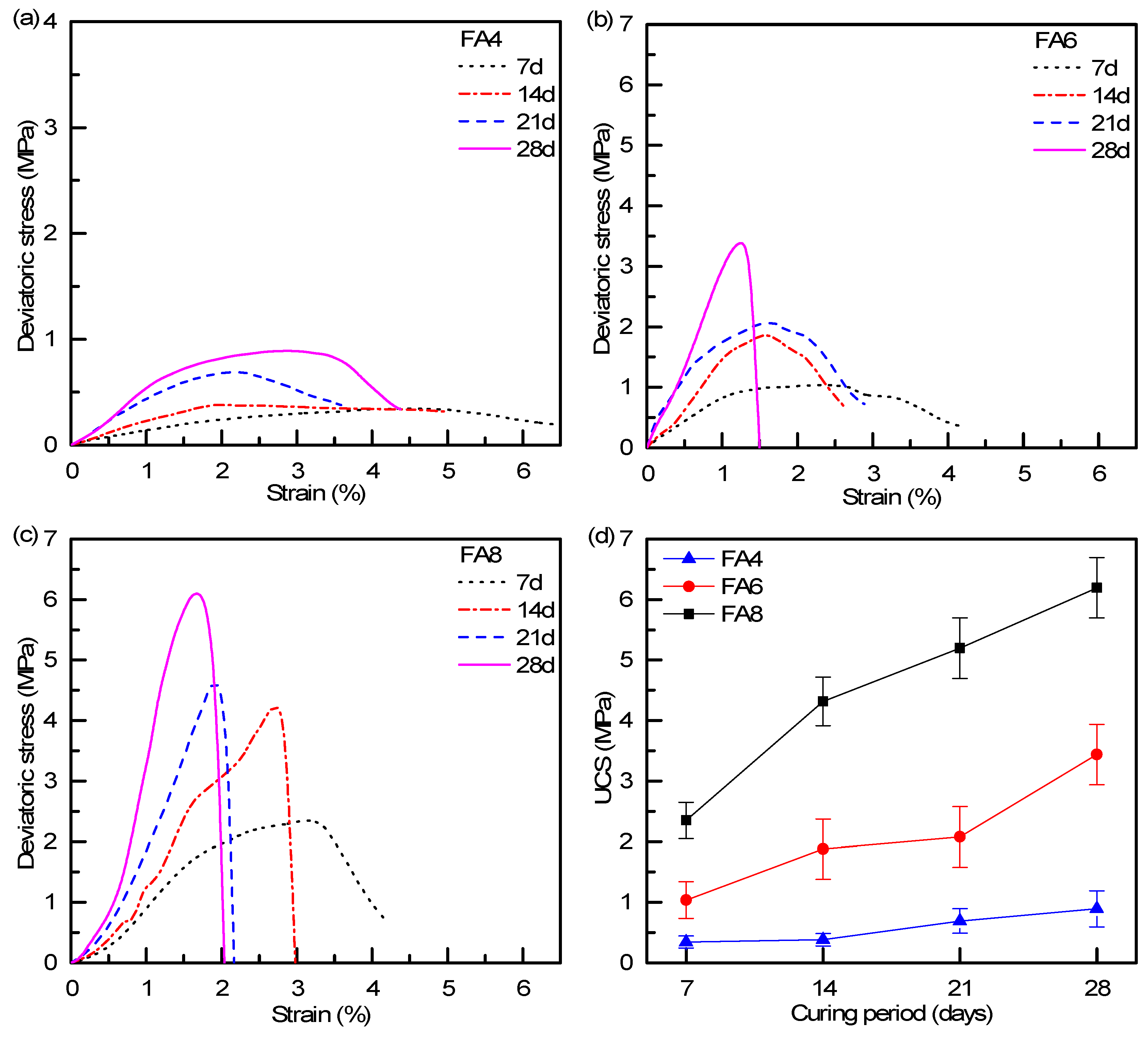
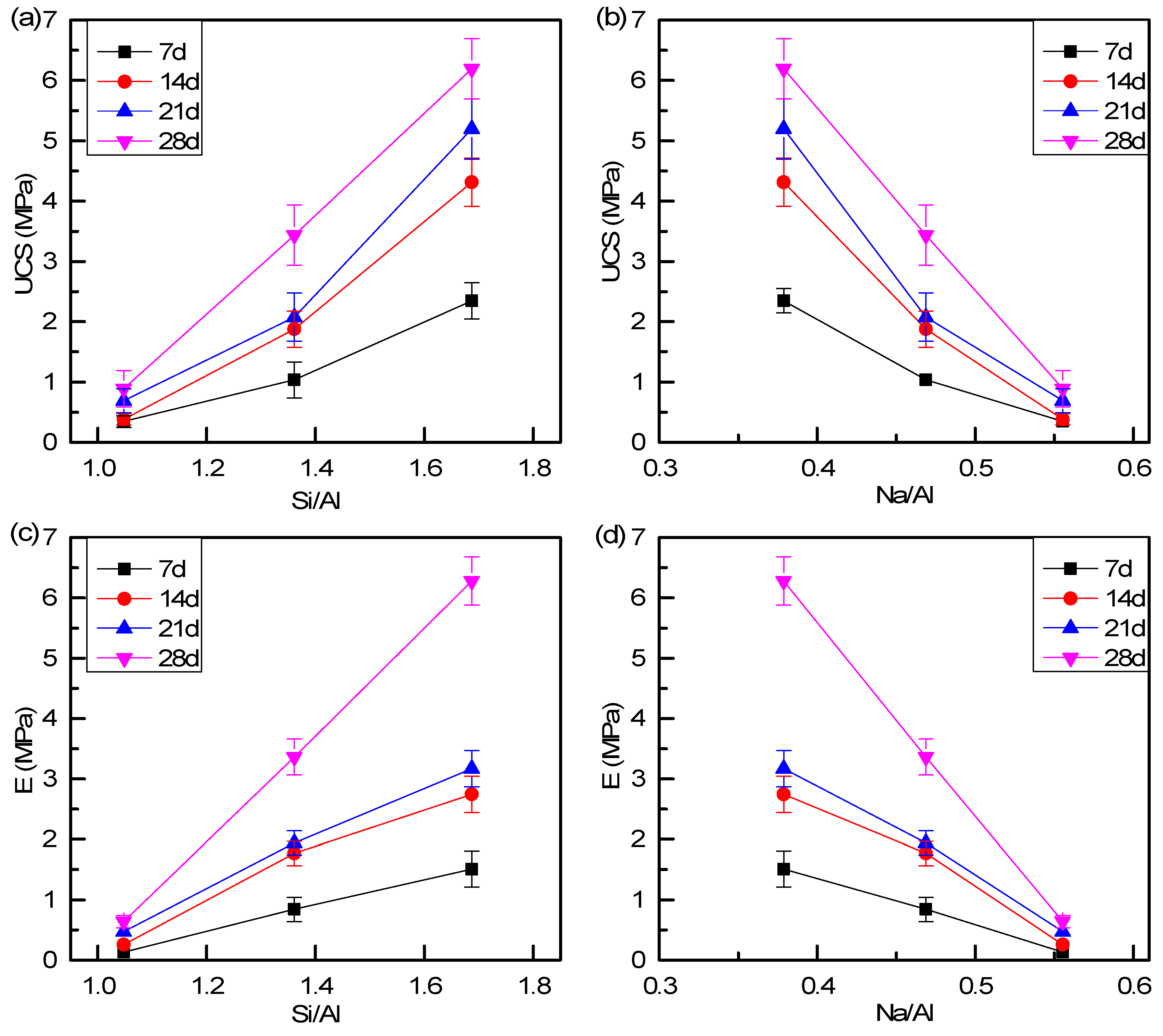
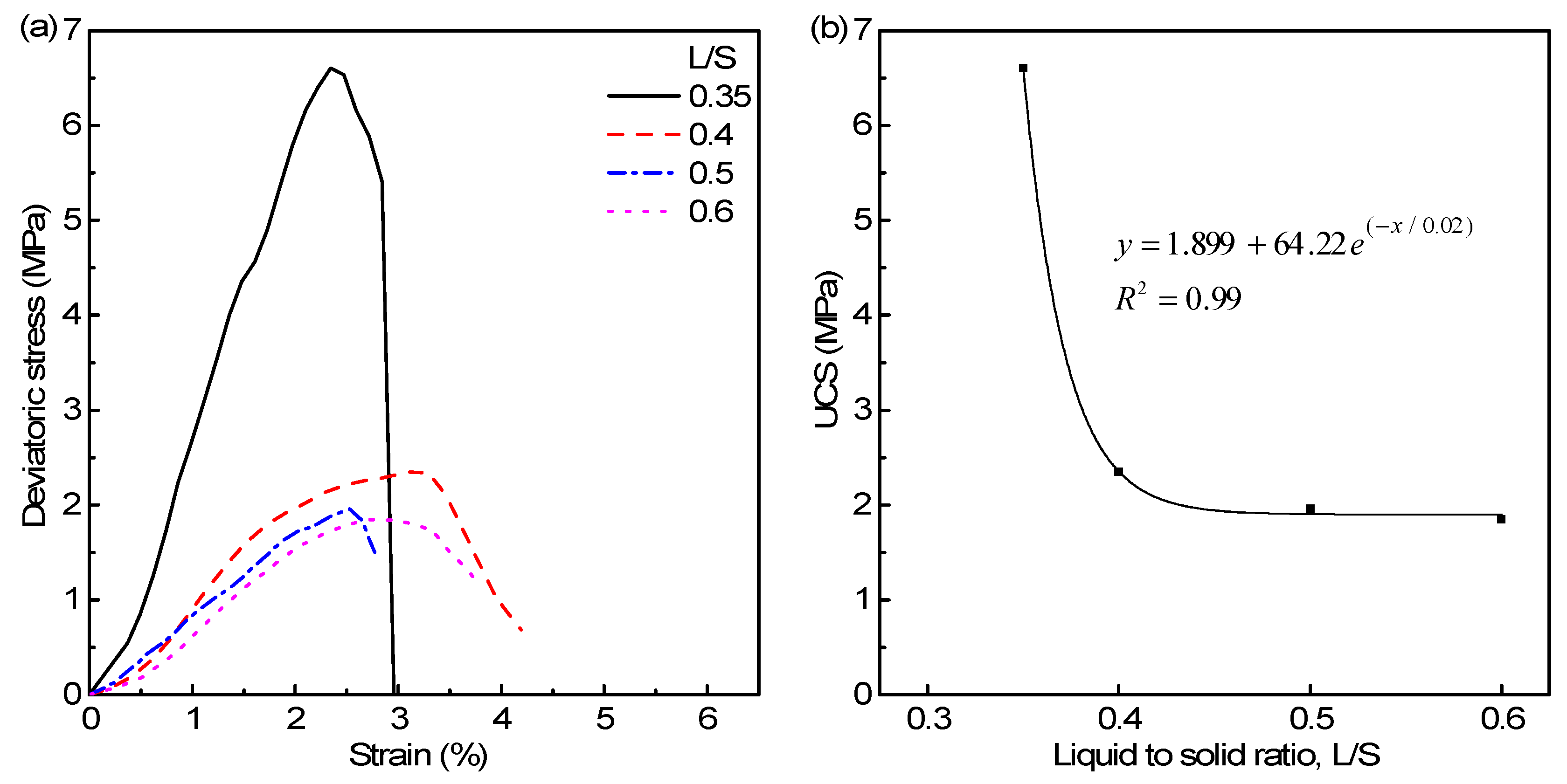
3.4. Parametric Influences on Mechanical Properties
4. Conclusions
- Geopolymers can be sustainably synthesized by utilizing the high alkalinity of red mud without the introduction of commercial hydroxides.
- The inherent alkalinity of the red mud results in partial dissolution of the aluminosilicates in the raw fly ash and subsequent formation of aluminosilicate gels.
- The strength of the end products synthesized under ambient conditions using fly ash and red mud, without the addition of alkali such as NaOH, continues to increase significantly from 7 to 28 days with UCS = 6.19 MPa for FA8 after 28 days.
- The stiffness increases with the increase in the curing time and there is a transition from ductile to brittle behavior with the increase in both fly ash content as well as curing time. For the same synthesis conditions, a higher quantity of fly ash with same amount of red mud yields better strength and stiffness values as seen by E = 0.63 GPa at 28 days for FA8.
- The amount of fly ash affects the formation and distribution of various types of pores in the geopolymeric matrix, wherein a high starting Si/Al ratio gives rise to interstitial pores in the final geopolymeric matrix. For a curing period of 7 days, the end product showed 6.6 MPa at L/S of 0.35.
- The unconfined compressive strength of the end products and the L/S ratio follow an inverse exponential relationship similar to the porosity characteristics, and the minimum L/S ratio of 0.35 was found to be optimum for obtaining higher strength fly ash–red mud based aluminosiliceous materials with lesser porosity.
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Koshy, N.; Singh, D.N. Fly ash zeolites for water treatment applications. J. Environ. Chem. Eng. 2016, 4, 1460–1472. [Google Scholar] [CrossRef]
- Toniolo, N.; Boccaccini, A.R. Fly ash-based geopolymers containing added silicate waste. A review. Ceram. Int. 2017, 43, 14545–14551. [Google Scholar] [CrossRef]
- Evans, K. The History, Challenges, and New Developments in the Management and Use of Bauxite Residue. J. Sustain. Metall. 2016, 2, 316–331. [Google Scholar] [CrossRef]
- Koshy, N.; Jha, B.; Kadali, S.; Singh, D.N. Synthesis and Characterization of Ca and Na Zeolites (Non-Pozzolanic Materials) Obtained From Fly Ash–Ca(OH)2 Interaction. Mater. Perform. Charact. 2015, 4, 87–102. [Google Scholar]
- Ascensão, G.; Seabra, M.P.; Aguiar, J.B.; Labrincha, J.A. Red mud-based geopolymers with tailored alkali diffusion properties and pH buffering ability. J. Clean. Prod. 2017, 148, 23–30. [Google Scholar] [CrossRef]
- He, J.; Jie, Y.; Zhang, J.; Yu, Y.; Zhang, G. Synthesis and characterization of red mud and rice husk ash-based geopolymer composites. Cem. Concr. Compos. 2013, 37, 108–118. [Google Scholar] [CrossRef]
- Provis, J.L.; Lukey, G.C.; Van Deventer, J.S.J. Do geopolymers actually contain nanocrystalline zeolites? A reexamination of existing results. Chem. Mater. 2005, 17, 3075–3085. [Google Scholar] [CrossRef]
- Xu, H.; Van Deventer, J.S.J. Geopolymerisation of multiple minerals. Miner. Eng. 2002, 15, 1131–1139. [Google Scholar] [CrossRef]
- Juenger, M.C.G.; Winnefeld, F.; Provis, J.L.; Ideker, J.H. Advances in alternative cementitious binders. Cem. Concr. Res. 2011, 41, 1232–1243. [Google Scholar] [CrossRef]
- Koshy, N.; Dondrob, K.; Hu, L.; Wen, Q.; Meegoda, J.N. Synthesis and characterization of geopolymers derived from coal gangue, fly ash and red mud. Constr. Build. Mater. 2019, 206, 287–296. [Google Scholar] [CrossRef]
- Kamseu, E.; Beleuk à Moungam, L.M.; Cannio, M.; Billong, N.; Chaysuwan, D.; Melo, U.C.; Leonelli, C. Substitution of sodium silicate with rice husk ash-NaOH solution in metakaolin based geopolymer cement concerning reduction in global warming. J. Clean. Prod. 2017, 142, 3050–3060. [Google Scholar] [CrossRef]
- Yang, T.; Zhu, H.; Zhang, Z. Influence of fly ash on the pore structure and shrinkage characteristics of metakaolin-based geopolymer pastes and mortars. Constr. Build. Mater. 2017, 153, 284–293. [Google Scholar] [CrossRef]
- Ducman, V.; Korat, L. Characterization of geopolymer fly-ash based foams obtained with the addition of Al powder or H2O2 as foaming agents. Mater. Charact. 2016, 113, 207–213. [Google Scholar] [CrossRef]
- Guo, X.; Zhang, L.; Huang, J.; Shi, H. Detoxification and solidification of heavy metal of chromium using fly ash-based geopolymer with chemical agents. Constr. Build. Mater. 2017, 151, 394–404. [Google Scholar] [CrossRef]
- Suksiripattanapong, C.; Horpibulsuk, S.; Boongrasan, S.; Udomchai, A. Unit weight, strength and microstructure of a water treatment sludge—Fly ash lightweight cellular geopolymer. Constr. Build. Mater. 2015, 94, 807–816. [Google Scholar] [CrossRef]
- Arulrajah, A.; Kua, T.-A.; Horpibulsuk, S.; Phetchuay, C. Strength and microstructure evaluation of recycled glass-fly ash geopolymer as low-carbon masonry units. Constr. Build. Mater. 2016, 114, 400–406. [Google Scholar] [CrossRef]
- Kumar, A.; Kumar, S. Development of paving blocks from synergistic use of red mud and fly ash using geopolymerization. Constr. Build. Mater. 2013, 38, 865–871. [Google Scholar] [CrossRef]
- Zhang, M.; Zhao, M.; Zhang, G.; Nowak, P.; Coen, A.; Tao, M. Calcium-free geopolymer as a stabilizer for sulfate-rich soils. Appl. Clay Sci. 2015, 108, 199–207. [Google Scholar] [CrossRef]
- Rowles, M.; O’Connor, B. Chemical optimisation of the compressive strength of aluminosilicate geopolymers synthesised by sodium silicate activation of metakaolinite. J. Mater. Chem. 2003, 13, 1161–1165. [Google Scholar] [CrossRef]
- He, J.; Zhang, J.; Yu, Y.; Zhang, G. The strength and microstructure of two geopolymers derived from metakaolin and red mud-fly ash admixture: A comparative study. Constr. Build. Mater. 2012, 30, 80–91. [Google Scholar] [CrossRef]
- Hajjaji, W.; Andrejkovičová, S.; Zanelli, C.; Alshaaer, M.; Dondi, M.; Labrincha, J.A.; Rocha, F. Composition and technological properties of geopolymers based on metakaolin and red mud. Mater. Des. 2013, 52, 648–654. [Google Scholar] [CrossRef]
- Izquierdo, M.; Querol, X.; Davidovits, J.; Antenucci, D.; Nugteren, H.; Fernández-Pereira, C. Coal fly ash-slag-based geopolymers: Microstructure and metal leaching. J. Hazard. Mater. 2009, 166, 561–566. [Google Scholar] [CrossRef] [PubMed]
- Zhuang, X.Y.; Chen, L.; Komarneni, S.; Zhou, C.H.; Tong, D.S.; Yang, H.M.; Yu, W.H.; Wang, H. Fly ash-based geopolymer: Clean production, properties and applications. J. Clean. Prod. 2016, 125, 253–267. [Google Scholar] [CrossRef]
- Zhang, M.; Zhao, M.; Zhang, G.; Mann, D.; Lumsden, K.; Tao, M. Durability of red mud-fly ash based geopolymer and leaching behavior of heavy metals in sulfuric acid solutions and deionized water. Constr. Build. Mater. 2016, 124, 373–382. [Google Scholar] [CrossRef]
- Geng, J.J.; Zhou, M.; Li, Y.; Chen, Y.; Han, Y.; Wan, S.; Zhou, X.; Hou, H. Comparison of red mud and coal gangue blended geopolymers synthesized through thermal activation and mechanical grinding preactivation. Constr. Build. Mater. 2017, 153, 185–192. [Google Scholar] [CrossRef]
- Dondrob, K.; Koshy, N.; Wen, Q.; Hu, L. Synthesis and Characterization of Geopolymers from Coal Gangue, Fly Ash and Red Mud. In The International Congress on Environmental Geotechnics; Springer: Berlin/Heidelberg, Germany, 2018; pp. 420–427. [Google Scholar]
- Ye, N.; Yang, J.; Liang, S.; Hu, Y.; Hu, J.; Xiao, B.; Huang, Q. Synthesis and strength optimization of one-part geopolymer based on red mud. Constr. Build. Mater. 2016, 111, 317–325. [Google Scholar] [CrossRef]
- Hoy, M.; Horpibulsuk, S.; Rachan, R.; Chinkulkijniwat, A.; Arulrajah, A. Recycled asphalt pavement—Fly ash geopolymers as a sustainable pavement base material: Strength and toxic leaching investigations. Sci. Total Environ. 2016, 573, 19–26. [Google Scholar] [CrossRef]
- Zhang, G.; He, J.; Gambrell, R. Synthesis, Characterization, and Mechanical Properties of Red Mud-Based Geopolymers. Transp. Res. Rec. 2010, 2167, 1–9. [Google Scholar] [CrossRef]
- Davidovits, J. Geopolymers: Inorganic polymeric new materials. J. Therm. Anal. 1991, 37, 1633–1656. [Google Scholar] [CrossRef]
- Zhang, M.; El-Korchi, T.; Zhang, G.; Liang, J.; Tao, M. Synthesis factors affecting mechanical properties, microstructure, and chemical composition of red mud-fly ash based geopolymers. Fuel 2014, 134, 315–325. [Google Scholar] [CrossRef]
- Hu, W.; Nie, Q.; Huang, B.; Shu, X.; He, Q. Mechanical and Microstructural Characterization of Geopolymers Derived from Red Mud and Fly Ashes. J. Clean. Prod. 2018, 186, 799–806. [Google Scholar] [CrossRef]
- ASTM. Standard Test Methods for Loss on Ignition (LOI) of Solid Combustion. In Annual Book of ASTM Standards; ASTM International: West Conshohocken, PA, USA, 2008; Volume D7348-08. [Google Scholar]
- ASTM. Standard Test Method for Young’s Modulus, Tangent Modulus, and Chord Modulus. In Annual Book of ASTM Standards; ASTM International: West Conshohocken, PA, USA, 2017; Volume E111-17, pp. 1–7. [Google Scholar]
- ASTM. Standard Specification for Coal Fly Ash and Raw or Calcined Natural Pozzolan for Use in Concrete. In Annual Book of ASTM Standards; ASTM International: West Conshohocken, PA, USA, 2015; Volume C618-15, pp. 3–6. [Google Scholar]
- Koshy, N.; Singh, D.N.; Jha, B.; Kadali, S.; Patil, J. Characterization of Na and Ca zeolites synthesized by various hydrothermal treatments of fly ash. Adv. Civ. Eng. Mater. 2015, 4, 131–143. [Google Scholar] [CrossRef]
- Van Deventer, J.S.J.; Provis, J.L.; Duxson, P.; Brice, D.G. Chemical research and climate change as drivers in the commercial adoption of alkali activated materials. Waste Biomass Valorization 2010, 1, 145–155. [Google Scholar] [CrossRef]
- Koshy, N.; Singh, D.N. Textural Alterations in Coal Fly Ash due to Alkali Activation. J. Mater. Civ. Eng. 2016, 28, 04016126. [Google Scholar] [CrossRef]
- Wan, Q.; Rao, F.; Song, S.; García, R.E.; Estrella, R.M.; Patino, C.L.; Zhang, Y. Geopolymerization reaction, microstructure and simulation of metakaolin-based geopolymers at extended Si/Al ratios. Cem. Concr. Compos. 2017, 79, 45–52. [Google Scholar] [CrossRef]
- Lee, B.; Kim, G.; Kim, R.; Cho, B.; Lee, S.; Chon, C.M. Strength development properties of geopolymer paste and mortar with respect to amorphous Si/Al ratio of fly ash. Constr. Build. Mater. 2017, 151, 512–519. [Google Scholar] [CrossRef]
- Sore, S.O.; Messan, A.; Prud’homme, E.; Escadeillas, G.; Tsobnang, F. Synthesis and characterization of geopolymer binders based on local materials from Burkina Faso—Metakaolin and rice husk ash. Constr. Build. Mater. 2016, 124, 301–311. [Google Scholar] [CrossRef]
- Feng, D.; Provis, J.L.; Van Deventer, J.S.J. Thermal activation of albite for the synthesis of one-part mix geopolymers. J. Am. Ceram. Soc. 2012, 95, 565–572. [Google Scholar] [CrossRef]
- Zhang, J.; Li, C. Experimental Study on Lime and Fly Ash–Stabilized Sintered Red Mud in Road Base. J. Test. Eval. 2018, 46, 1539–1547. [Google Scholar] [CrossRef]
- Park, Y.; Park, J.; Hinoki, T.; Kohyama, A. Thermal shock behavior of NITE-porous SiC ceramics. In Proceedings of the 31st International Conference on Advanced Ceramics and Composites, Daytona Beach, FL, USA, 21–26 January 2007; Brito, M., Case, E., Kriven, W.M., Eds.; pp. 89–94. [Google Scholar]
- Steveson, M.; Sagoe-Crentsil, K. Relationships between composition, structure and strength of inorganic polymers: Part 2 Fly ash-derived inorganic polymers. J. Mater. Sci. 2005, 40, 4247–4259. [Google Scholar] [CrossRef]


| Composition | Fly Ash | Red Mud |
|---|---|---|
| SiO2 | 45.79 | 8.88 |
| Al2O3 | 21.69 | 25.34 |
| Na2O | 0.46 | 8.75 |
| CaO | 7.07 | 0.92 |
| Fe2O3 | 6.68 | 40.03 |
| TiO2 | 1.47 | 6.49 |
| SO3 | 0.47 | 0.22 |
| P2O5 | 0.33 | 0.08 |
| MnO | 0.12 | 0.05 |
| ZrO2 | 0.10 | 0.35 |
| MgO | 0.80 | - |
| SrO | 0.31 | - |
| K2O | - | 0.06 |
| Cr2O3 | - | 0.12 |
| Loss on ignition | 14.7 | 8.7 |
| Label | Curing Time (days) | FA/RM | L/S | Na2SiO3·5H2O (%wt) | Si/Al | Na/Al |
|---|---|---|---|---|---|---|
| FA4 | 7, 14, 21, 28 | 4:6 | 0.4 | 40% | 1.04 | 0.59 |
| FA6 | 7, 14, 21, 28 | 6:4 | 0.4 | 40% | 1.36 | 0.51 |
| FA8 | 7, 14, 21, 28 | 8:2 | 0.4 | 40% | 1.69 | 0.42 |
| FA8-30 | 7 | 8:2 | 0.4 | 30% | 1.65 | 0.35 |
| FA8-40 | 7 | 8:2 | 0.4 | 40% | 1.69 | 0.42 |
| FA8-50 | 7 | 8:2 | 0.4 | 50% | 1.74 | 0.50 |
| FA8-0.35 | 7 | 8:2 | 0.35 | 40% | 1.67 | 0.39 |
| FA8-0.4 | 7 | 8:2 | 0.4 | 40% | 1.69 | 0.42 |
| FA8-0.5 | 7 | 8:2 | 0.5 | 40% | 1.74 | 0.50 |
| FA8-0.6 | 7 | 8:2 | 0.6 | 40% | 1.78 | 0.57 |
| Sample | Pore Volume Contribution (%) of Various Sizes (nm) | Micro/Mesopores | Macro Pores | UCS (MPa) | |||||
|---|---|---|---|---|---|---|---|---|---|
| <10 | 10–50 | 50–100 | 100–200 | 200–1000 | >1000 | ||||
| FA4 | 0.91 | 7.86 | 9.01 | 30.05 | 37.09 | 15.08 | 8.77 | 91.23 | 0.89 |
| FA6 | 1.27 | 8.87 | 12.93 | 10.92 | 59.89 | 6.12 | 10.14 | 89.86 | 3.44 |
| FA8 | 1.56 | 25.3 | 31.6 | 25.33 | 11.68 | 4.53 | 26.86 | 73.14 | 6.19 |
| Sample | UCS (MPa) | E (GPa) |
|---|---|---|
| FA4–7d | 0.34 | 0.01 |
| FA4–14d | 0.38 | 0.03 |
| FA4–21d | 0.69 | 0.05 |
| FA4–28d | 0.89 | 0.06 |
| FA6–7d | 1.03 | 0.08 |
| FA6–14d | 1.88 | 0.18 |
| FA6–21d | 2.08 | 0.19 |
| FA6–28d | 3.44 | 0.34 |
| FA8–7d | 2.35 | 0.15 |
| FA8–14d | 4.32 | 0.27 |
| FA8–21d | 5.20 | 0.32 |
| FA8–28d | 6.19 | 0.63 |
| FA30 | 0.53 | 0.07 |
| FA40 | 2.35 | 0.15 |
| FA50 | 3.61 | 0.24 |
| FA0.35 | 6.61 | 0.38 |
| FA0.4 | 6.19 | 0.15 |
| FA0.5 | 1.96 | 0.10 |
| FA0.6 | 1.84 | 0.10 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Koshy, N.; Dondrob, K.; Hu, L.; Wen, Q.; Meegoda, J.N. Mechanical Properties of Geopolymers Synthesized from Fly Ash and Red Mud under Ambient Conditions. Crystals 2019, 9, 572. https://doi.org/10.3390/cryst9110572
Koshy N, Dondrob K, Hu L, Wen Q, Meegoda JN. Mechanical Properties of Geopolymers Synthesized from Fly Ash and Red Mud under Ambient Conditions. Crystals. 2019; 9(11):572. https://doi.org/10.3390/cryst9110572
Chicago/Turabian StyleKoshy, Nevin, Kunga Dondrob, Liming Hu, Qingbo Wen, and Jay N. Meegoda. 2019. "Mechanical Properties of Geopolymers Synthesized from Fly Ash and Red Mud under Ambient Conditions" Crystals 9, no. 11: 572. https://doi.org/10.3390/cryst9110572
APA StyleKoshy, N., Dondrob, K., Hu, L., Wen, Q., & Meegoda, J. N. (2019). Mechanical Properties of Geopolymers Synthesized from Fly Ash and Red Mud under Ambient Conditions. Crystals, 9(11), 572. https://doi.org/10.3390/cryst9110572







