Two-Dimensional Hydrogen-Bonded Crystal Structure, Hirshfeld Surface Analysis and Morphology Prediction of a New Polymorph of 1H-Nicotineamidium Chloride Salt
Abstract
1. Introduction
2. Experimental
2.1. Synthesis
2.2. The SEM Technique
2.3. X-ray Crystallography and Data Collection
2.4. Hirshsfeld Surface Analysis
3. Results and Discussion
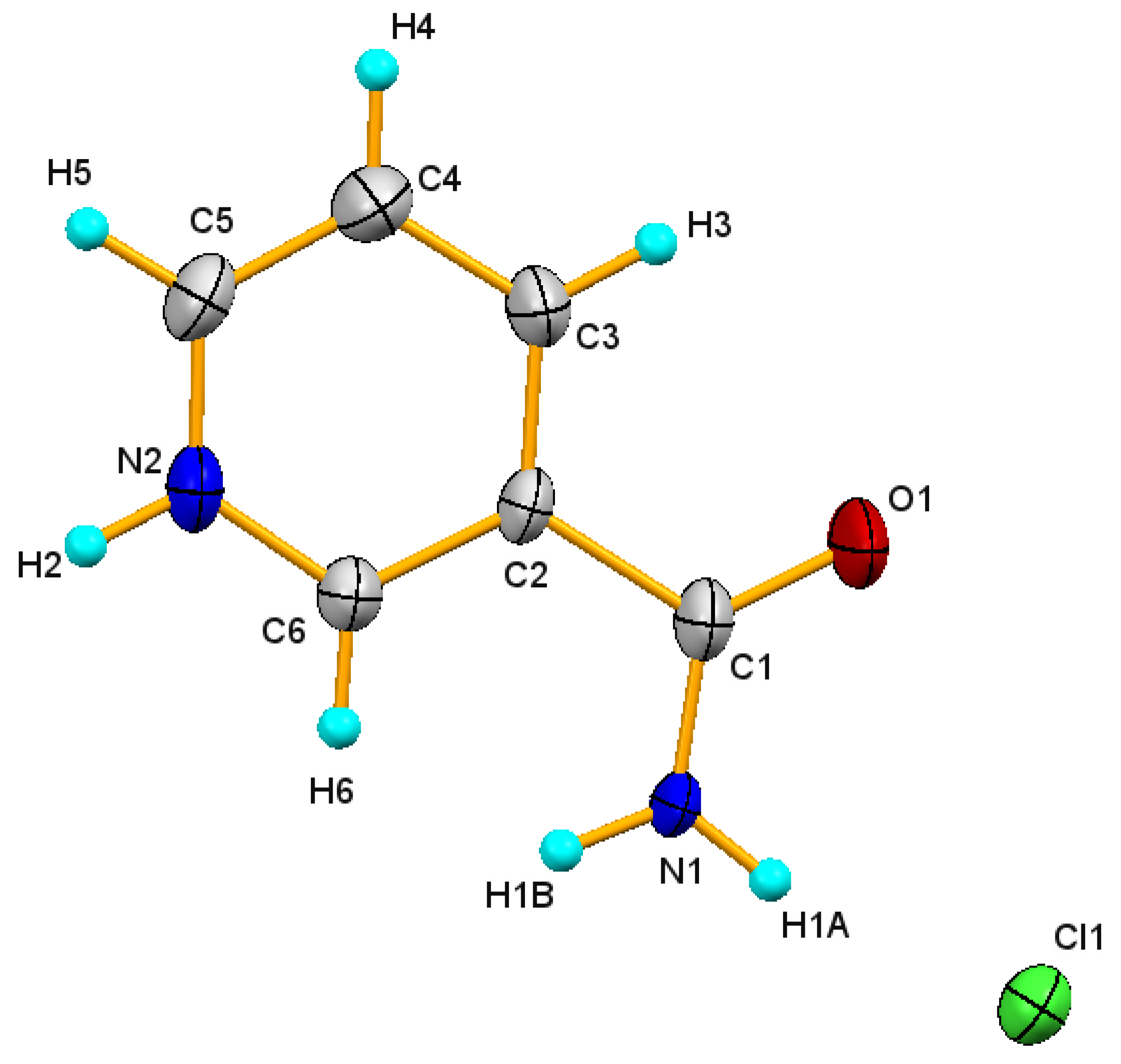
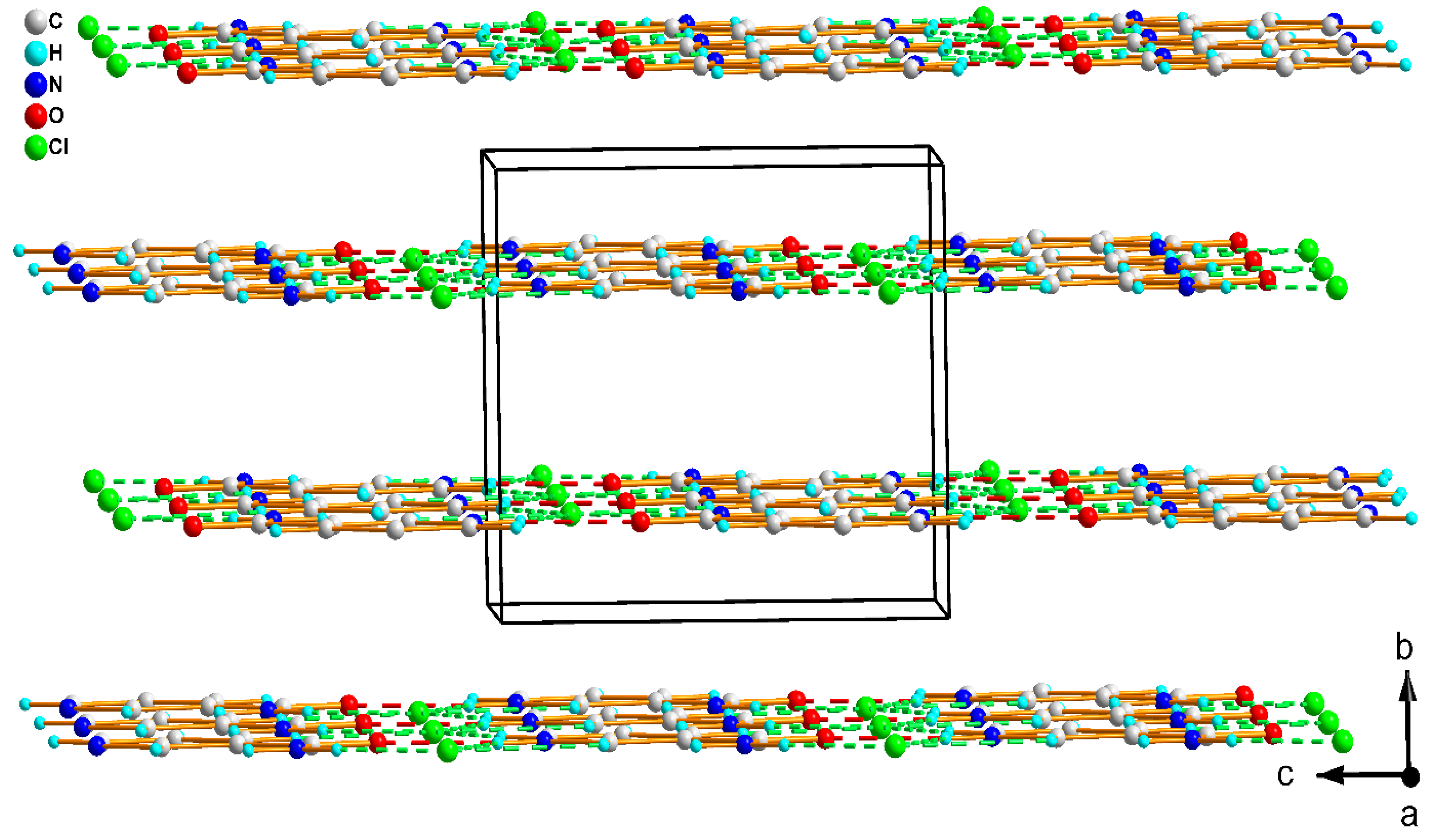
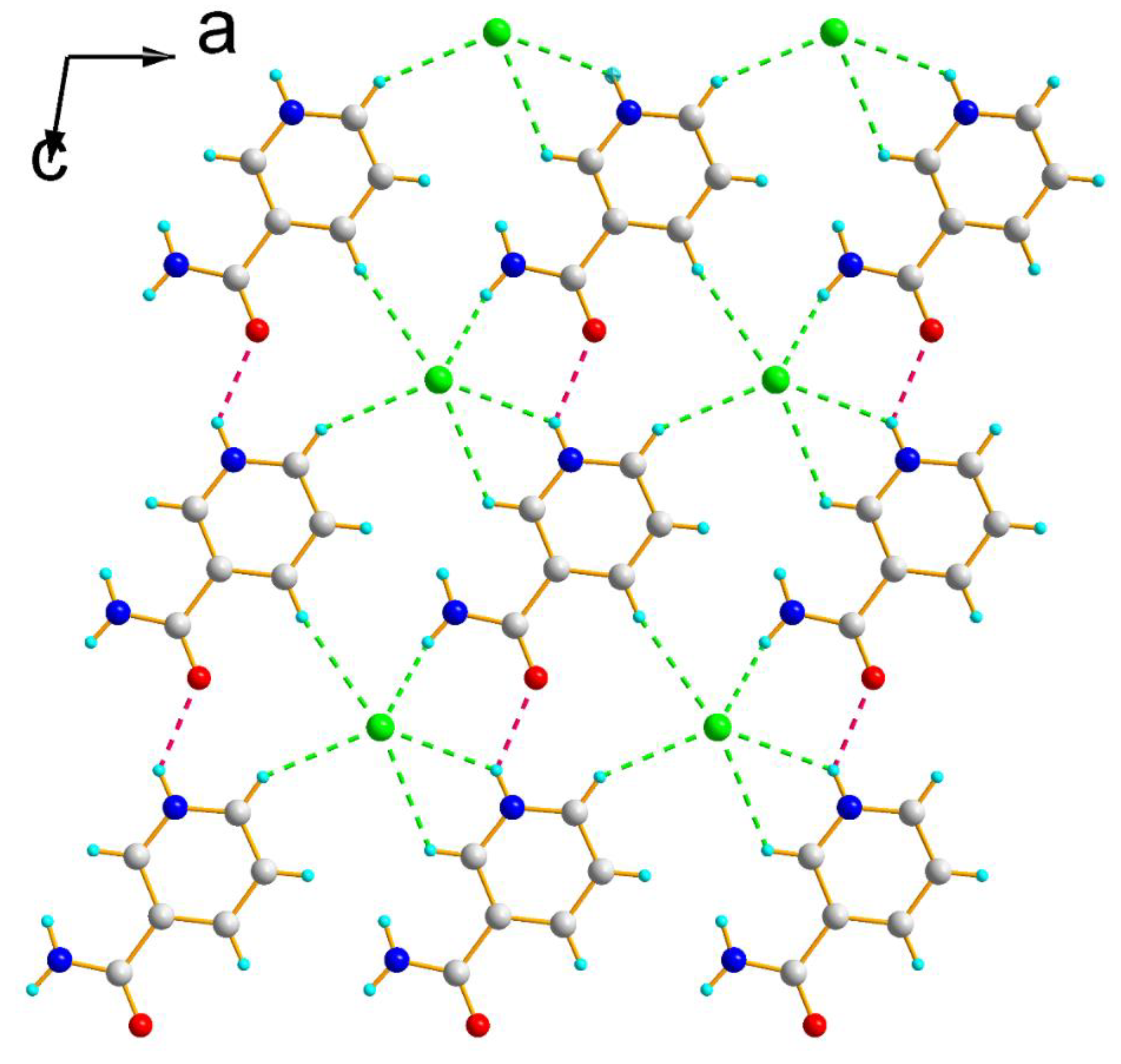
3.1. Structure Description
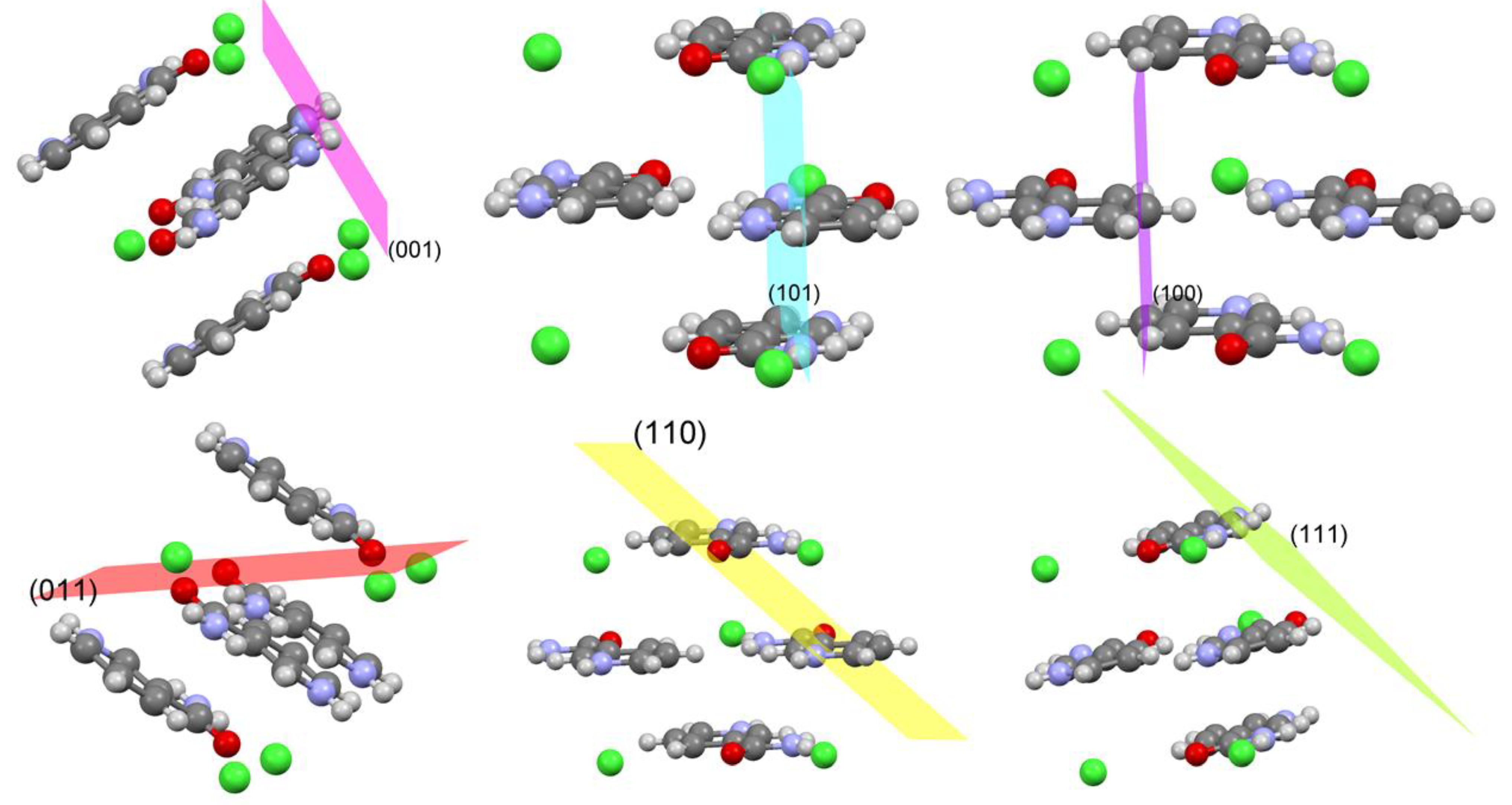
3.2. Crystal Morphology and Packing
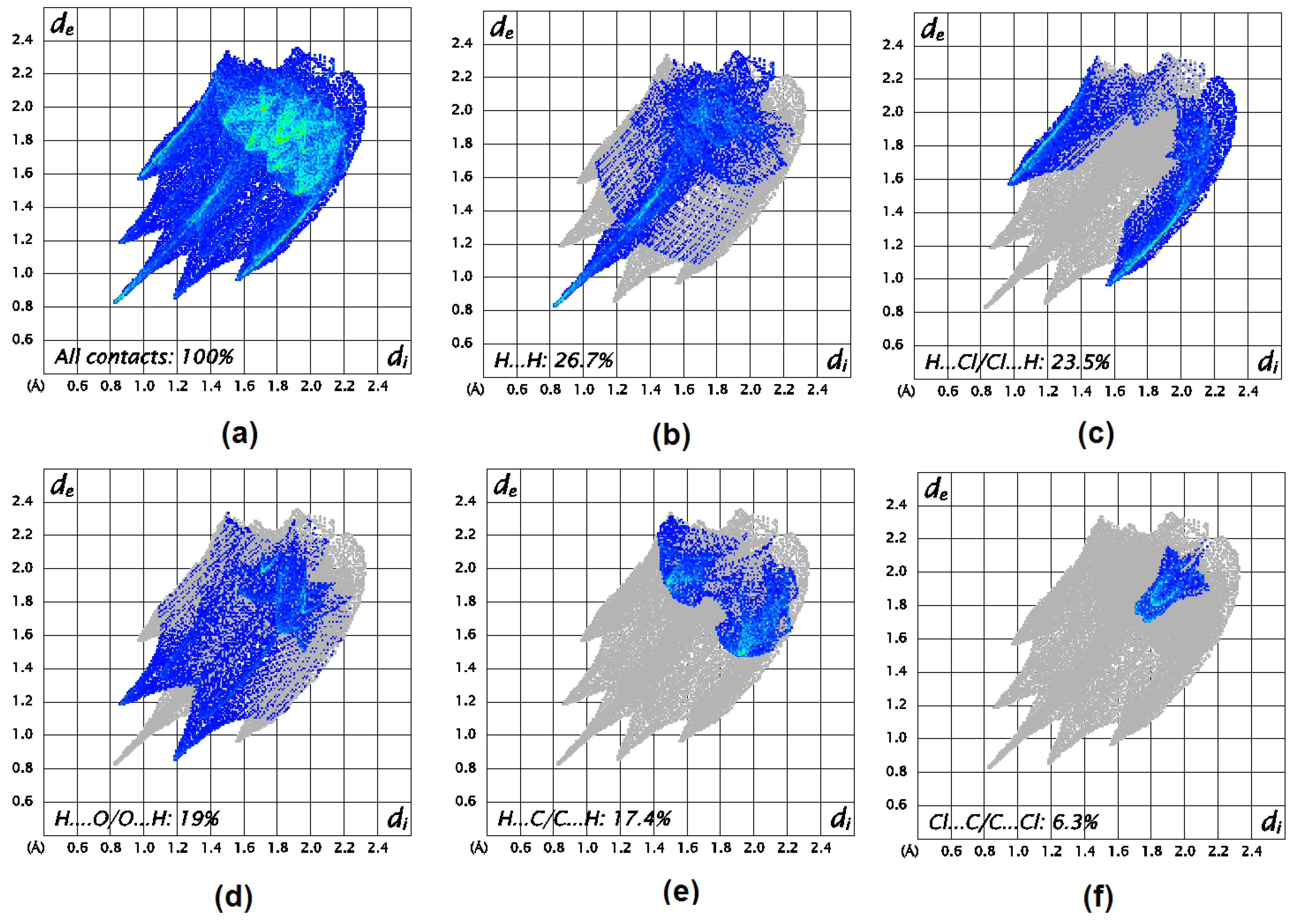
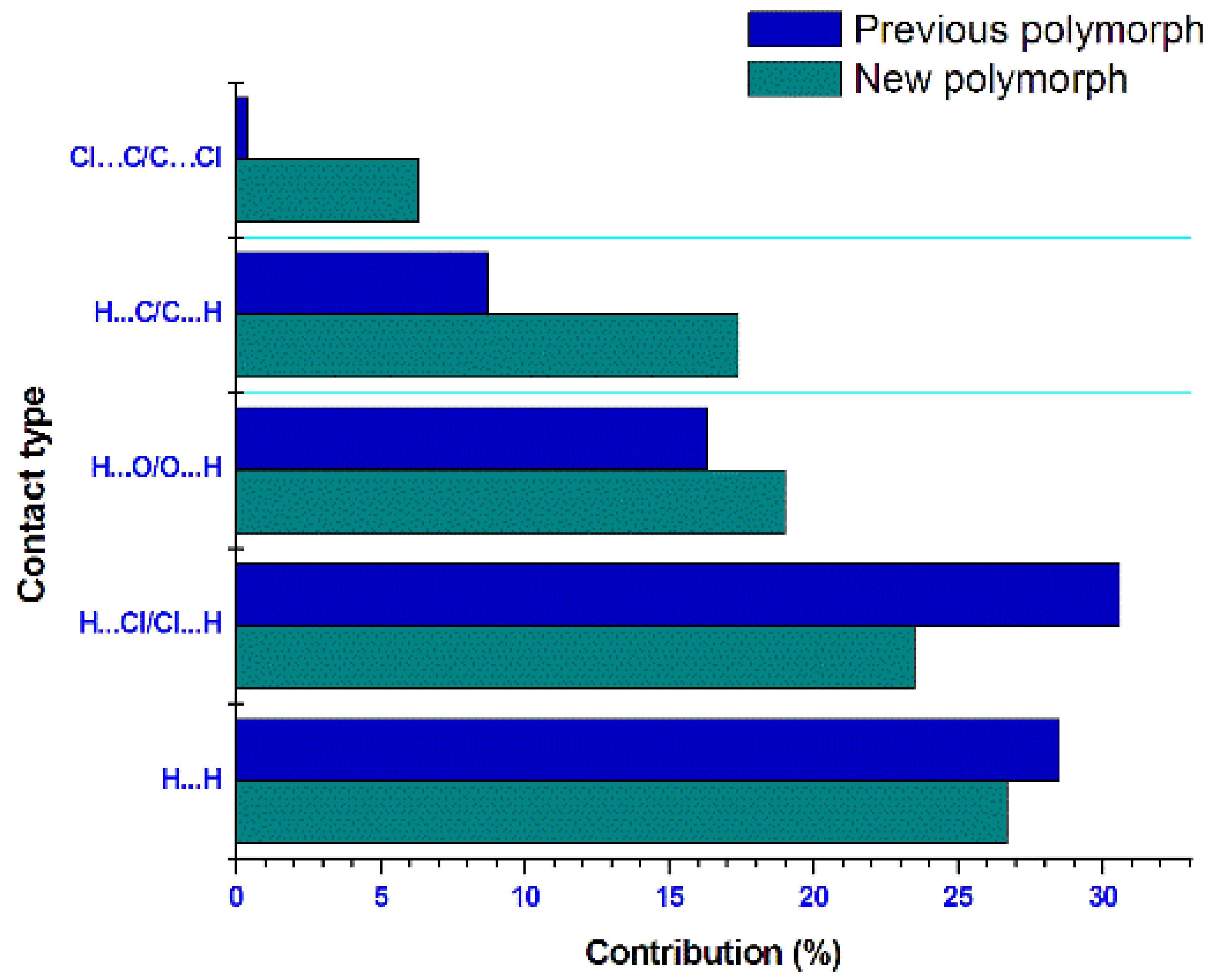
3.3. Hirshfeld Surface Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Ganguly, P.; Desiraju, G.R. Van der Waals and Polar Intermolecular Contact Distances: Quantifying Supramolecular Synthons. Chem. Asian J. 2008, 3, 868–880. [Google Scholar] [CrossRef]
- Nangia, A. Conformational Polymorphism in Organic Crystals. Acc. Chem. Res. 2008, 41, 595–604. [Google Scholar] [CrossRef]
- Hulme, A.T.; Johnston, A.; Florence, A.J.; Fernandes, P.; Shankland, K.; Bedford, C.T.; Welch, G.W.; Sadiq, G.; Haynes, D.A.; Motherwell, W.D.S.; et al. Search for a Predicted Hydrogen Bonding Motif—A Multidisciplinary Investigation into the Polymorphism of 3-Azabicyclo[3.3.1]nonane-2,4-dione. J. Am. Chem. Soc. 2007, 129, 3649–3657. [Google Scholar] [CrossRef]
- Price, S.L. From Crystal Structure Prediction to Polymorph Prediction: Interpreting the Crystal Energy Landscape. Phys. Chem. Chem. Phys. 2008, 10, 1996–2009. [Google Scholar] [CrossRef]
- Vallejos, M.M.; Angelina, E.L.; Peruchena, N.M. Bifunctional Hydrogen Bonds in Monohydrated Cycloether Complexes. J. Phys. Chem. A. 2010, 114, 2855–2863. [Google Scholar] [CrossRef]
- Jin, S.; Wang, D.J. Hydrogen Bonded 3D Supramolecular Architectures of Three Saccharinate Salts. Chem. Crystallogr. 2011, 41, 1085–1092. [Google Scholar] [CrossRef]
- Korotkovaa, E.I.; Kratochvíl, B. Pharmaceutical Cocrystals. Procedia Chem. 2014, 10, 473–476. [Google Scholar] [CrossRef]
- Lu, J.; Rohani, S. Preparation and Characterization of Theophylline−Nicotinamide Cocrystal. Org. Process Res. Dev. 2009, 13, 1269–1275. [Google Scholar] [CrossRef]
- Lemmerer, A.; Esterhuysen, C.; Bernstein, J. Synthesis, Characterization, and Molecular Modeling of A Pharmaceutical Co-Crystal: (2-Chloro-4-Nitrobenzoic Acid):(Nicotinamide). J. Pharm. Sci. 2010, 99, 4054–4071. [Google Scholar] [CrossRef]
- Bathori, N.B.; Lemmerer, A.; Venter, G.A.; Bourne, S.A.; Caira, M.R. Pharmaceutical Co-crystals with Isonicotinamide-Vitamin B3, Clofibric Acid, and Diclofenac-and Two Isonicotinamide Hydrates. Cryst. Growth Des. 2011, 11, 75–87. [Google Scholar] [CrossRef]
- Fábián, L.; Hamill, N.; Eccles, K.S.; Moynihan, H.A.; Maguire, A.R.; McCausland, L.; Lawrence, S.E. Cocrystals of Fenamic Acids with Nicotinamide. Cryst. Growth Des. 2011, 11, 3522–3528. [Google Scholar] [CrossRef]
- Sheldrick, G.M. SHELXT—Integrated space-group and crystal-structure determination. Acta Crystallogr. A 2015, A71, 3–8. [Google Scholar] [CrossRef]
- Sheldrick, G.M. Crystal structure refinement with SHELXL. Acta Crystallogr. C 2015, C71, 3–8. [Google Scholar] [CrossRef]
- Brandenburg, K. DIAMOND; Crystal Impact GbR: Bonn, Germany, 2006. [Google Scholar]
- Hirshfeld, H.L. Bonded-atom fragments for describing molecular charge densities. Theor. Chim. Acta 1977, 44, 129–138. [Google Scholar] [CrossRef]
- Spackman, M.A.; Jayatilaka, D. Hirshfeld surface analysis. Cryst. Eng. Comm. 2009, 11, 19–32. [Google Scholar] [CrossRef]
- Wolff, S.K.; Grimwood, D.J.; McKinnon, J.J.; Turner, M.J.; Jayatilaka, D.; Spackman, M.A. Crystal Explorer version 3.1; University of Western Australia: Perth, Australia, 2012. [Google Scholar]
- Venkatesan, P.; Thamotharan, S.; Ilangovan, A.; Liang, H.; Sundius, T. Crystal structure, Hirshfeld surfaces and DFT computation of NLO active (2E)-2-(ethoxycarbonyl)-3-[(1-methoxy-1-oxo-3-phenylpropan-2-yl)amino] prop-2-enoic acid. Spectrochim. Acta Part A 2016, 153, 625–636. [Google Scholar] [CrossRef]
- Gubin, A.I.; Nurakhmetov, N.N.; Buranbaev, M.Z.; Mulkina, R.I.; Erkasov, R.S. Crystal and molecular structure of nicotinamide hydrochloride. Donor-acceptor properties of protonated nicotinamide. Kristallografiya (Russ.) (Crystallogr. Rep.) 1989, 34, 238–239. [Google Scholar]
- Delgado, G.E.K.; Varela, N.; Mora, A.J.; Colmenárez, J.B.; Atencio, R. Synthesis, Thermal Studies, Spectroscopy Characterization, and Crystal Structure of Nicotinamidium Oxamate. Mol. Cryst. Liq. Cryst. 2015, 609, 218–227. [Google Scholar] [CrossRef]
- Oswald, I.D.H.; Motherwell, W.D.S.; Parsons, S. A 1:2 co-crystal of isonicotinamide and propionic acid. Acta Crystallogr. E 2004, 60, 2380–2383. [Google Scholar] [CrossRef]
- Perumalla, S.R.; Sun, C.C. Design and Synthesis of Solid State Structures with Conjugate Acid-Base Pair Interactions. Cryst. Eng. Comm. 2012, 14, 3851–3853. [Google Scholar] [CrossRef]
- Materials Studio, Version 7.0; Accelrys Software Inc.: San Diego, CA, USA, 2013.
- Bravais, A. Etudes Crystallographiques; Academi des Sciences: Paris, France, 2010. [Google Scholar]
- Donnay, J.D.H.; Harker, D. A new law of crystal morphology extending the Law of Bravais. Am. Mineral. 1937, 22, 446–467. [Google Scholar]
- Prywer, J. Morphological importance of crystal faces in connection with growth rates and crystallographic structure of crystal. Cryst. Growth Des. 2002, 2, 281–286. [Google Scholar] [CrossRef]
- McKinnon, J.J.; Jayatilaka, D.; Spackman, M.A. Towards quantitative analysis of intermolecular interactions with Hirshfeld surfaces. Chem. Commun. 2007, 37, 3814–3816. [Google Scholar] [CrossRef] [PubMed]

| Crystal Data | |
|---|---|
| Empirical Formula | C6H7ClN2O |
| Formula Weight (g mol−1) | 158.59 |
| Crystal System, Space Group | Monoclinic, P21/m |
| a (Å) | 7.1671(4) |
| b (Å) | 6.6664(3) |
| c (Å) | 7.4883(4) |
| V (Å3) | 352.84(3) |
| β | 99.531(2) |
| Z | 2 |
| µ (mm−1) | 0.467 |
| Dx (Mg m−3) | 1.493 |
| F(000) | 164 |
| Crystal Size (mm) | 0.32 × 0.21 × 0.12 |
| Crystal Habit | Colorless block crystal |
| Data Collection | |
| Diffractometer | Bruker APEXII D8 venture, φ and ω-scans |
| Monochromater | Graphite |
| Radiation Type, λ(Å) | Mo Kα, 0.71073 |
| T (K) | 298 |
| Indexes Range | −9 ≤ h ≤ 9; −8 ≤ k ≤ 8; −9 ≤ l ≤ 9 |
| Measured Reflections | 16573 |
| Independent Reflections | 880 |
| Observed Refl. with I> 2σ(I) | 821 |
| Rint | 0.086 |
| Refinement | |
| Refinement on | F2 |
| Data/Restraints/Parameters | 880/0/61 |
| R[F2> 2σ(F2)] | 0.054 |
| wR(F2) | 0.160 |
| GooF = S | 0.93 |
| Δρmax/Δρmin (e Å−3) | 0.79/−0.85 |
| D-H···A | H···A (Å) | D···A (Å) | D-H···A (°) | Symmetry Operations |
|---|---|---|---|---|
| N1-H1A···Cl1 | 2.07 | 2.913 (8) | 167 | - |
| N2-H2···Cl1 | 2.63 | 3.282 (8) | 134 | x, y, z−1 |
| N2-H2···O1 | 2.14 | 2.799 (2) | 133 | x, y, z−1 |
| C5-H5···Cl1 | 2.71 | 3.537 (8) | 148 | x+1, y, z−1 |
| C6-H6···Cl1 | 2.83 | 3.389 (8) | 120 | x, y, z−1 |
| C5-H5···O1 | 2.51 | 2.982 (4) | 112 | x,y z−1 |
| hkl | Multiplicity | Dhkl (Ǻ) | % of Total Facet Area |
|---|---|---|---|
| 001 | 2 | 7.39 | 27.67 |
| 100 | 2 | 7.07 | 26.02 |
| 101 | 2 | 5.59 | 6.89 |
| 011 | 4 | 4.95 | 20.58 |
| 110 | 4 | 4.85 | 17.57 |
| 111 | 4 | 4.28 | 1.27 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ferjani, H.; Chebbi, H.; Guesmi, A.; AlRuqi, O.S.; Al-Hussain, S.A. Two-Dimensional Hydrogen-Bonded Crystal Structure, Hirshfeld Surface Analysis and Morphology Prediction of a New Polymorph of 1H-Nicotineamidium Chloride Salt. Crystals 2019, 9, 571. https://doi.org/10.3390/cryst9110571
Ferjani H, Chebbi H, Guesmi A, AlRuqi OS, Al-Hussain SA. Two-Dimensional Hydrogen-Bonded Crystal Structure, Hirshfeld Surface Analysis and Morphology Prediction of a New Polymorph of 1H-Nicotineamidium Chloride Salt. Crystals. 2019; 9(11):571. https://doi.org/10.3390/cryst9110571
Chicago/Turabian StyleFerjani, Hela, Hammouda Chebbi, Abderrahmen Guesmi, Obaid S. AlRuqi, and Sami A. Al-Hussain. 2019. "Two-Dimensional Hydrogen-Bonded Crystal Structure, Hirshfeld Surface Analysis and Morphology Prediction of a New Polymorph of 1H-Nicotineamidium Chloride Salt" Crystals 9, no. 11: 571. https://doi.org/10.3390/cryst9110571
APA StyleFerjani, H., Chebbi, H., Guesmi, A., AlRuqi, O. S., & Al-Hussain, S. A. (2019). Two-Dimensional Hydrogen-Bonded Crystal Structure, Hirshfeld Surface Analysis and Morphology Prediction of a New Polymorph of 1H-Nicotineamidium Chloride Salt. Crystals, 9(11), 571. https://doi.org/10.3390/cryst9110571






