3.1. Point Defects, Clusters, and Domains in Real LN Crystals
General thermodynamic concepts allow us to consider the formation of self-similar micro- and fractal-type nanostructures in LN crystals. These processes relate to different types of defective structure regardless of the specific physical mechanisms that generate it. Self-organization of a defective structure can occur at different scale levels and stages as a result of of the technological process of obtaining a crystalline material and an intense high-energy effect on the already grown LN crystals. The growth of LN crystals by Czochralski always takes place at a greater or lesser distance from the thermodynamic equilibrium. The formation of nano- and microstructures of the fractal type is especially characteristic of heavily doped LN crystals. Analysis of crystal growth processes and further study of their growth structure allows us to describe the ability of the system to self-organize. For example, complex non-equilibrium micro- and nanostructures of the fractal type are formed in an LN crystal during a non-equilibrium melt crystallization [
6,
13,
32]. They are usually difficult to reproduce (
Figure 2a). At the same time, the formation of micro- and nanostructures in crystals in the region of relatively low temperatures (T
Room < T < 140 °C) can occur under high-energy effects, for example, under an intense flow of external energy. This effect can be in the form of large doses of ionizing radiation (γ radiation) high power. The domain microstructures of the fractal type (
Figure 2a) were found in a LiNbO
3:Tm crystal. The crystal is grown under conditions that are far from thermodynamic equilibrium. Self-similar domain microstructures of the fractal type, spatially oriented along the direction of the Y axis of the hexagonal crystallographic symmetry, were also found in nominally pure LiNbO
3 crystals. They are irradiated with large doses of γ-radiation from the Co
60 source,
Figure 2b.
Domain microstructures presented on
Figure 2a, have properties of self-similarity at different scale levels and can be identified as fractals. Self-similar domain microstructures of the fractal type in the LiNbO
3:Tm crystal are spatially oriented along the three Y-directions of the hexagonal crystallographic symmetry,
Figure 2a. Such spatial self-organization of domains is associated with the pinning of domain walls on defects arising during non-equilibrium crystallization of a doped melt—gradients of a dopant distribution, as well as dislocations. Their arrangement follows the symmetry of the unit cell of a LN crystal, in accordance with Curie’s principle. Judging by the crystallographic orientation of self-similar structures in the LiNbO
3:Tm crystal, they were formed during the growth process. Probably, the fluctuations of the crystal growth rate occurred at a relatively high concentration of the dopant. At the same time, doping ions diffusing from the melt to the interfacial surface occupied the most favorable areas for growth—the edges of the steps, which coincided with the direction of the rhombohedral faces, and the latter had the highest growth rate.
Single microdomains with a size of about ~1 to 2 μm are observed in both doped and nominally pure CLN crystals. Single microdomains have a hexagonal shape in a plane perpendicular to the polar Z axis (
Figure 3a). Hexagonal microdomains are complex formations. They have their own fine structure. The splitting of a hexagonal microdomain ~1.6 μm in size into separate nanostructured layers is clearly seen on
Figure 3a. The splitting occurs in the direction of the polar axis Z. Microdomains of a triangular shape (
Figure 3b), wedge-shaped microdomains (
Figure 4) and “finger” domains (
Figure 2) are observed in LN crystals. “Finger” domains are oriented along the Y directions and form self-similar domain microstructures of a fractal type.
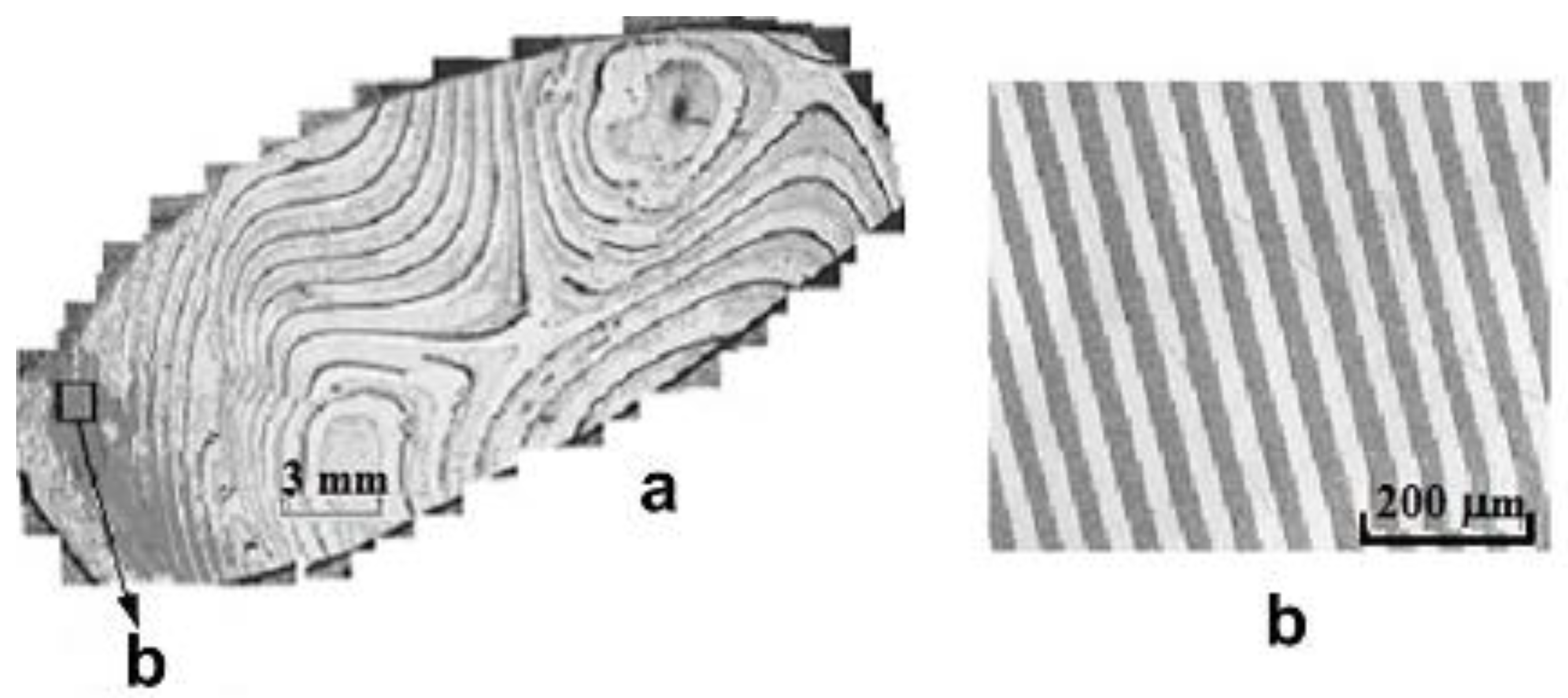
CLN crystals with a regular domain structure (RDS) doped with REE can be obtained under substantially unequilibrium thermal conditions. A RDS is formed along the polar axis. Its step is determined by the ratio of the speed of extrusion and rotation of the crystal and ranged from ~100 to 1 μm,
Figure 5.
Periodic nanoscale structures of the fractal type with a step from 10 to 100 nm were detected by atomic force microscopy for these crystals in the region of periodic regular domain structures (on negative domain walls). Periodic splitting occurs both in the direction parallel and perpendicular to the polar axis of the crystal. Periodic structural formations with a volume of several hundred unit cells appear in the crystal,
Figure 6,
Table 2. The detected structures have clear signs of spatial self-organization. They possess self-similarity properties at different scale levels and can be identified as fractals.
Regular domain structures can also be formed in a plane that is perpendicular to the polar Z axis.
Figure 7 and
Figure 8 show that the boundaries of such domains are blurred and cut up. At the same time, there are areas with a strictly regular domain structure (
Figure 7b and
Figure 8b) on the periphery of the crystal plates. The period of RDS Λ decreases from the center of the plates to the edge: for crystals grown in highly unsteady conditions, from 23 to 14 μm over the average length of the RDS region of ~2 mm, and for crystals grown under conditions close to equilibrium, from 46 to 38 μm over the average length of the RDS region ~1.5 mm. The presence of RDS in the edge part of the plates, cut perpendicular to the growth axis, is typical of most heavily doped LN crystals, thus it is natural.
The structural defects of the macro-, meso-, micro-, and nanolevels can affect the Raman scattering in nominally pure and doped LN crystals. This influence is noticeable, however, difficult to interpret. Domains that we have revealed on
Figure 2,
Figure 3,
Figure 4,
Figure 5,
Figure 6,
Figure 7,
Figure 8,
Figure 9,
Figure 10,
Figure 11,
Figure 12,
Figure 13 and
Figure 14 are only bigger part of the hierarchy of different defects forming in LN crystal during crystallization under highly non-equilibrium conditions and energy dissipating. At this, macro-domains (sizes of which are comparable with sizes of the samples that we have used in our research) in general usually have the structure similar to the structure of a crystal matrix. However, such macro-domains have microstructure that consists of fractal-like micro-domains. They themselves obviously cannot cause appearance of extra Raman bands. However, periodically poled structures have their own substructure. We have observed it by SPM to the size scale 5–10 nm. For example, on LN:REE crystals we have observed the following: On negative domain walls (
Figure 5) we have detected periodic nano-size structures with a step 10–100 nm,
Figure 6.
Figure 6 clearly shows that periods are directed both along and perpendicular to the crystal polar axis. Thus, LN crystals contain periodic structures several hundred unit cells in volume. Formation of such structures is not connected with growth processes that cause growth strips. These periodic nano-size structures are not domains in the common sense. Formation of these defects is probably caused by ordering of clusters that form around intrinsic and doping defects during crystallization in non-equilibrium state. Boundaries between such nano-structures probably have some charge (at least in non-equilibrium state, for example, during heating or leaching). Otherwise, leaching could not have been able to reveal them. Such separate nano-domains (
Figure 3) could possibly have even smaller intrinsic structure. The more unordered the structure is (as in non-stoichiometric LN crystals), the more substructures are formed in the materials.
3.2. Raman Spectra, Conoscopic Patterns, and Photoinduced Light Scattering in Nominal Pure and Heavily Doped LiNbO3 Crystals
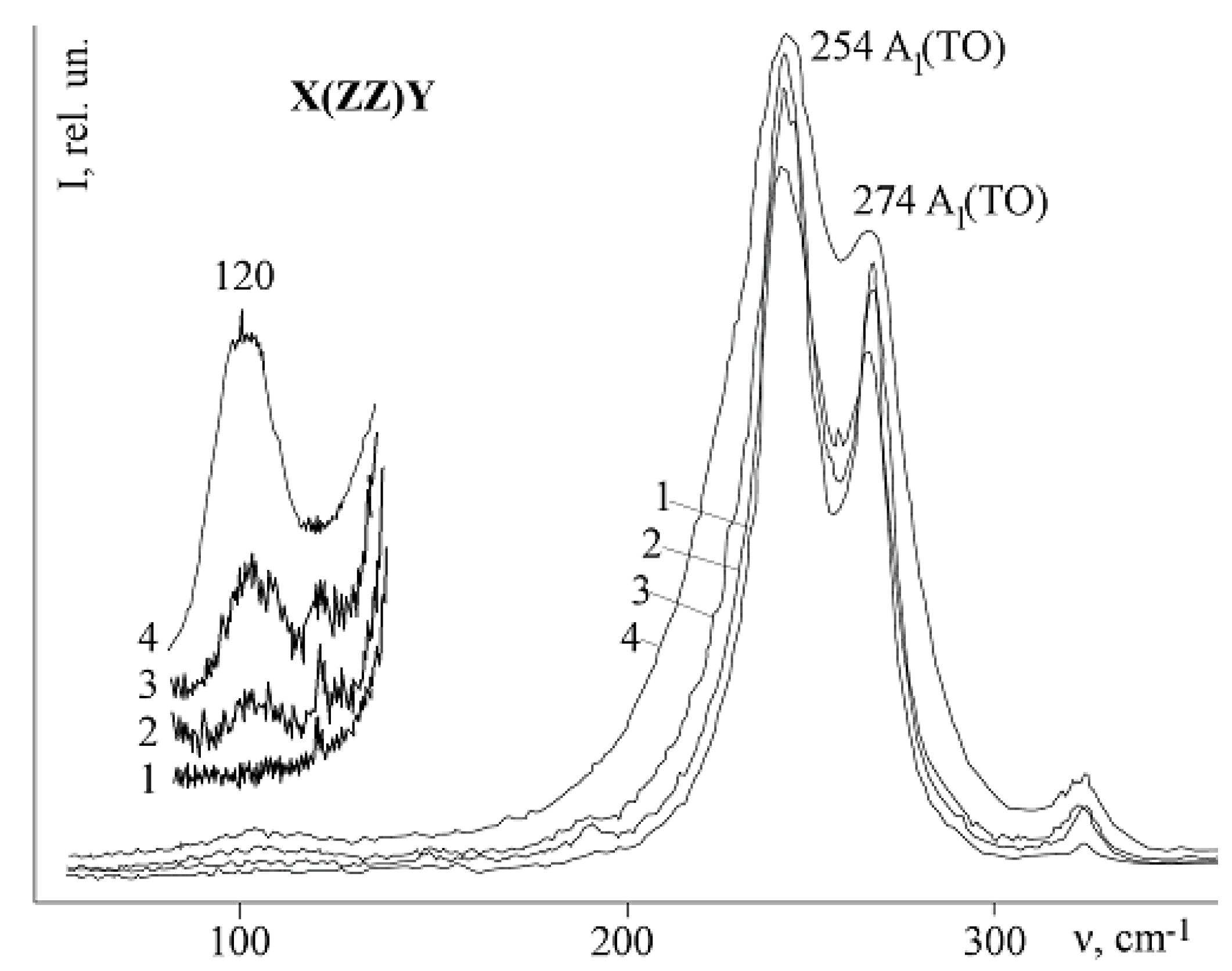
Figure 14 and
Figure 15 show fragments of Raman spectra of a nominally pure CLN crystal and heavily doped (with a dopant concentration near the main concentration threshold) LiNbO
3:Zn (4.5 mol.% ZnO), LiNbO
3:Mg (5.01):Fe (0.005), LiNbO
3:Mg (5.1), LiNbO
3:Mg (5.3 mol.% MgO). The spectra geometries are Y(ZZ)Ȳ and Y(ZX)Ȳ; vibrations of the A
1(TO) and E(TO) symmetry types are active.
Figure 16 and
Figure 17 demonstrate conoscopic and PILS patterns of of the investigated crystals.
Conoscopic patterns of crystals correspond to patterns of a highly perfect single axis crystals. An increase in the laser radiation power density p from 0.039 to 3.54 W/cm
2 (
Figure 16) leads to a certain increase in contrast and image clarity, but does not introduce significant interference anomalies into conoscopic patterns.
Figure 16 and
Figure 17 show that doping with magnesium cations suppresses the effect of photorefraction in an LN crystal. In a double doped crystal (Fe: Mg), concentrations of magnesium are close to the second concentration threshold (5.5 mol.% MgO). However, photorefractive effect is almost absent in the crystal; the “photorefractive” Fe
3+–Fe
2+ have a minor effect on the photorefraction, contrary to the effects caused by iron in singly doped crystals. The PILS indicatrix is impossible to excite even at relatively high powers density of laser radiation (6.29 W/cm
2). Only a insignificant circular scattering appears. This indicates the smallness of the photorefractive effect.
Crystal LiNbO
3:Zn (4.5 mol.% ZnO) deserves special attention. Studies of zinc-doped LN crystals with a small concentration step (0.2 mol.%) showed that heavily doped LiNbO
3:Zn crystals are the most optically and compositionally uniform in the concentration range ≈4.0–6.5 mol.% ZnO, i.e., between concentration thresholds at ≈3.0 and ≈6.76 mol.% ZnO [
6]. At this, the most complete suppression of photorefraction occurs at concentrations above the second concentration threshold at ≈6.8 mol.% ZnO in the crystal. However, LiNbO
3:Zn crystals with concentrations greater than ≈6.8 mol.% ZnO are characterized by low optical and compositional uniformity and high probability of precipitation of the Li
6ZnNb
4O
14 mixed phase [
6]. Thereby they are not suitable for practical applications as high-quality non-linear optical materials. It is better to use crystals with a concentration of ≈4.0–6.5 mol.% ZnO, i.e., crystals with a zinc concentration between the main concentration thresholds at ≈3.0 and ≈6.76 mol.% ZnO as non-linear optical materials with the lowest photorefraction effect. According to PILS data, LiNbO
3:Zn crystal (4.5 mol.% ZnO) has the lowest photorefractive effect,
Figure 17. A significant shift of the optical transmission edge to the shortwave region was detected in an LiNbO
3:Zn crystal (4.5 mol.% ZnO), when compared with other Zn-doped crystals. This shift indicates a higher structural uniformity of the crystal compared to other LiNbO
3: Zn crystals.
Table 3 shows frequencies of experimentally observed bands and calculated data for the LiNbO
3 crystal. The data were obtained by ab initio calculations [
57,
58] and in the approximation of the valence-force field [
68].
Table 3 shows that frequencies of bands corresponding to fundamental oscillations coincide well within the experimental errors in Raman spectra of SLN and CLN crystals, but SLN Raman bands are significantly narrower than that of the other crystals [
5,
36,
37,
38,
39,
59].
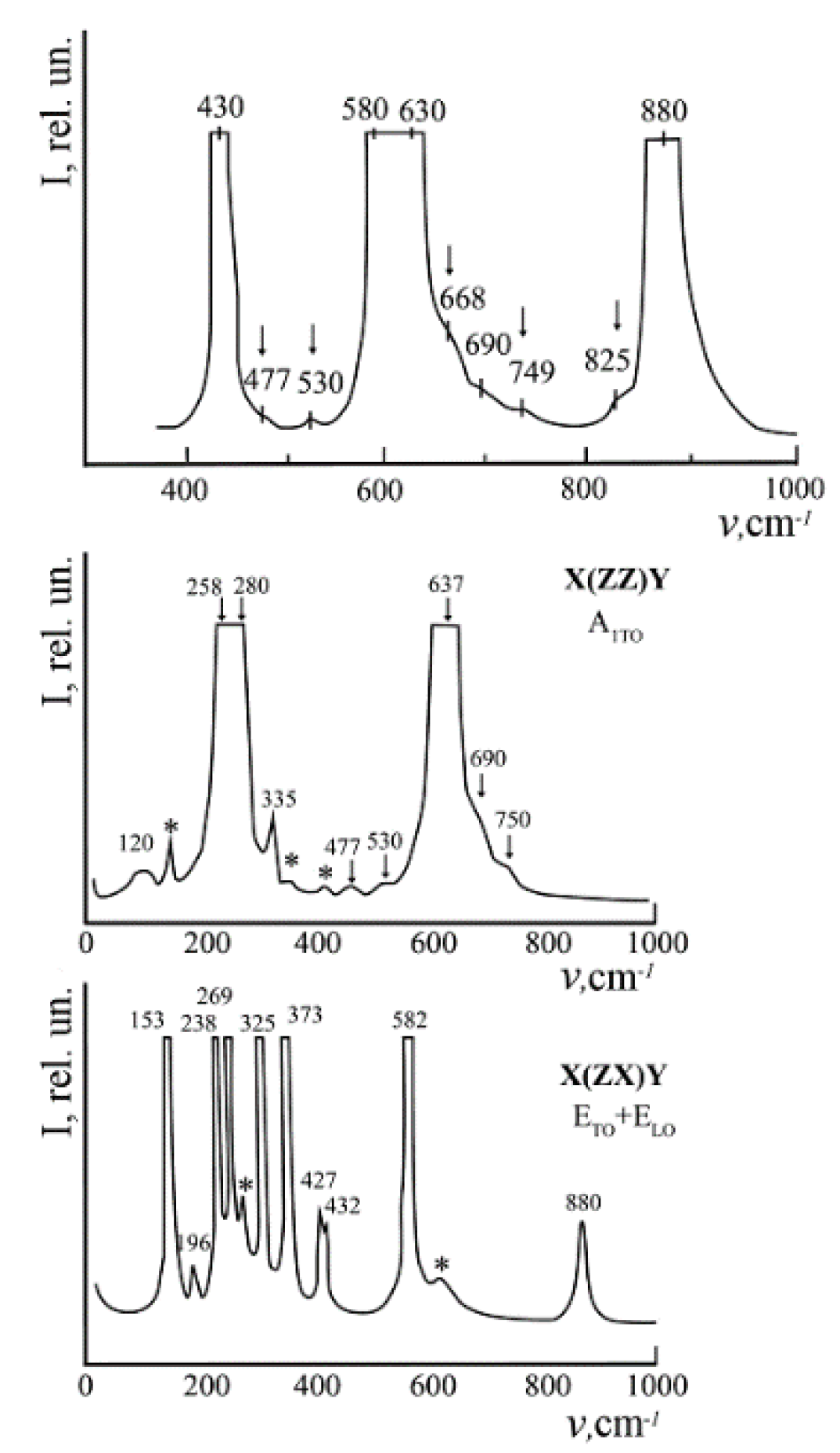
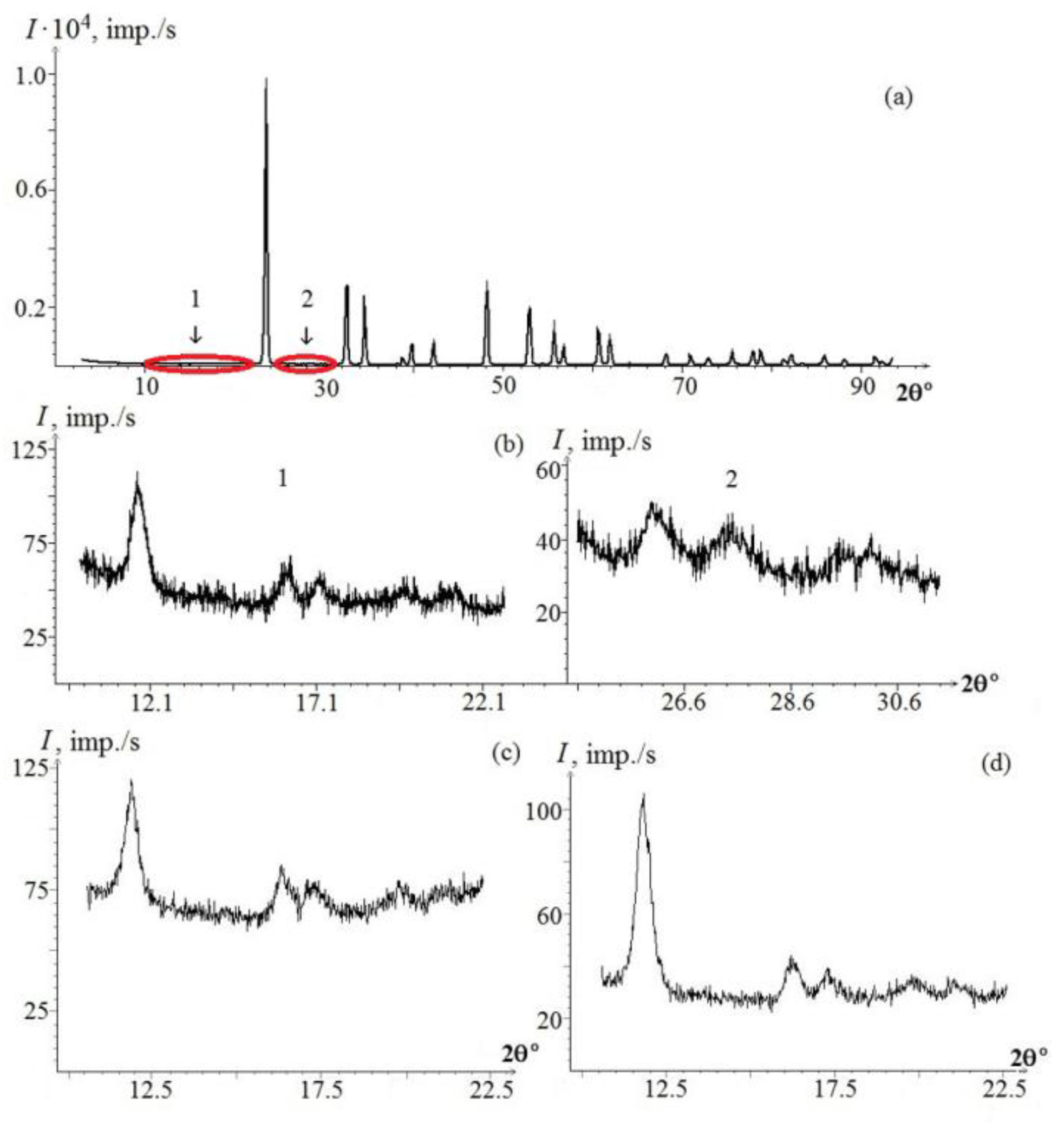
We observed only the bands corresponding to the fundamental lattice vibrations allowed by the selection rules for the space group C
3V6 (R3c) in Raman spectrum of the SLN crystal. Low-intensity bands, which characterize the spectra of non-stoichiometric crystals (
Figure 1) [
5,
11,
36,
37,
38,
39,
40,
41,
42,
43,
44,
45,
46,
47,
56,
59,
60,
61,
62,
63,
64,
65,
66,
67], were not found (
Table 3). We observed only bands corresponding to the fundamental oscillations of the E(TO) symmetry type (
Table 3) in the scattering geometry Y(ZX)Ȳ for all the studied crystals. Extra bands were not detected. The band with a frequency of 630 cm
−1 is clearly manifested in the geometry Y(ZX)Ȳ on the shoulder of the intensive band 580 cm
−1 (8E(TO),
Figure 14). The 630 cm
−1 band corresponds to the 4A
1 (TO) oscillation and is manifested due to the photorefractive effect [
5,
36]; 180 and 610 cm
−1 bands correspond to the E(TO) fundamental vibrations (
Table 3). These two low intensity bands are located on the shoulders of high-intensity bands and are observed only in the spectrum of highly perfect SLN crystal. 180 and 610 cm
−1 bands are masked with effects of structural disordering and are not observed in spectra of non-stoichiometric crystals [
38,
39,
59].
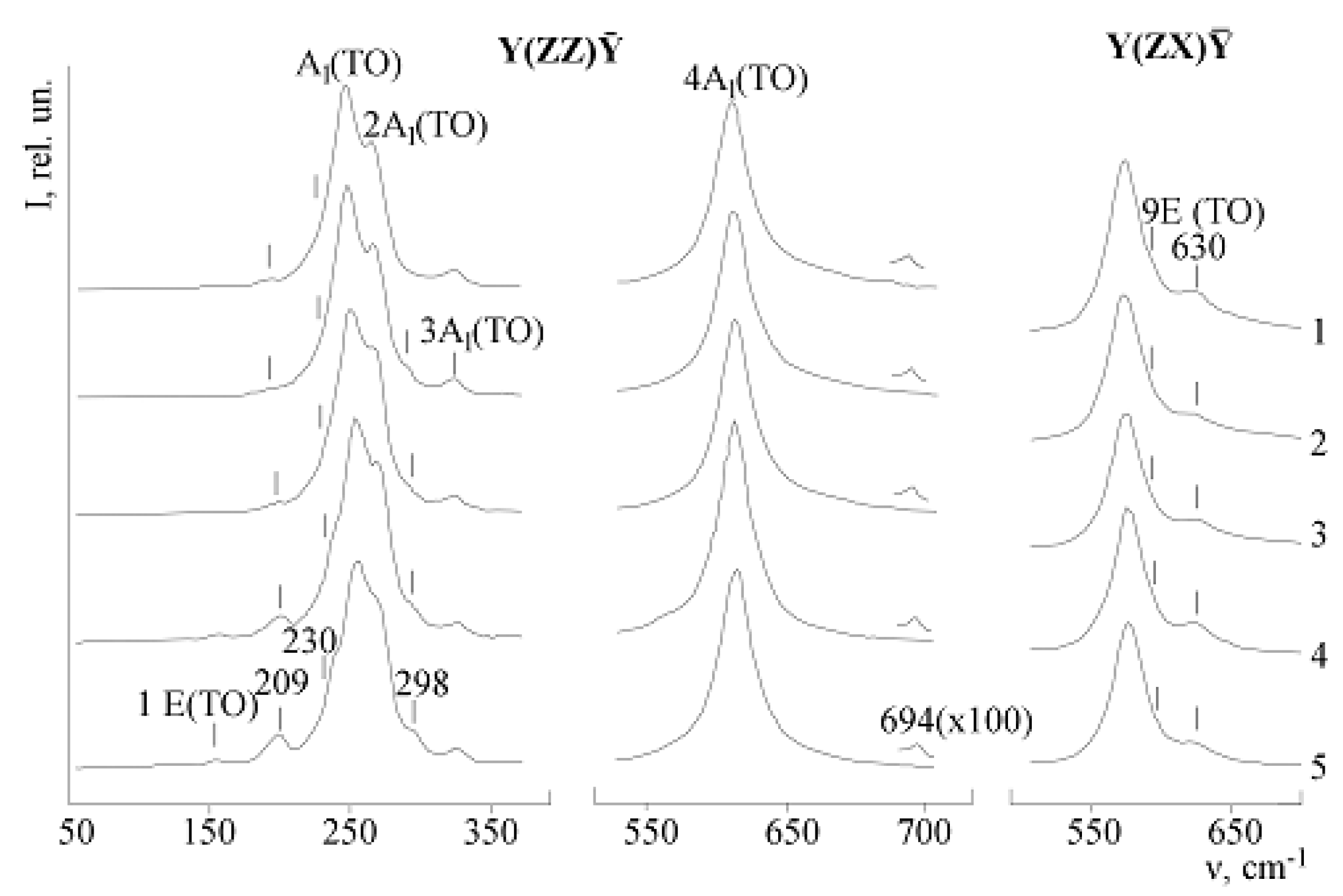
LN Raman spectra in the Y(ZZ)Ȳ polarization geometry must contain only fundamental A
1 (TO) vibrations. At the same time, we discovered much more bands than is allowed by the selection rules in the spectrum of crystals CLN, LiNbO
3:Zn (4.5 mol.% ZnO), LiNbO
3:Mg: Fe (5.01; 0.005), LiNbO
3:Mg (5.1), LiNbO
3:Mg (5.3 mol.% MgO) in this geometry (
Figure 14 and
Figure 15,
Table 3). In addition to bands corresponding to the fundamental oscillations for the studied crystals at 100 K, extra bands with frequencies of 104, 120, 209, 230, 298, 337, 694, 880 cm
–1 were observed. Their intensity is much less than the intensity of fundamental bands. 209, 230, 298 cm
−1 bands are observed only in the Raman spectrum of heavily doped crystals when the concentration of the doping element is close to the threshold. Earlier, the manifestation of low-intensity extra bands in different polarization scattering geometries in Raman spectrum of the SLN crystal was reported in detail in [
5,
11,
36,
37,
38,
39,
40,
41,
42,
43,
44,
45,
46,
47,
56,
59,
60,
61,
62,
63,
64,
65,
66,
67]. A review of research is given in [
5,
11]. Since the frequencies of these extra bands (85, 92, 103, 117, 187, 305, 331, 412, 477, 535, 605, 668, 690, 739, 743, 825 cm
−1, T = 293K) are well known in the literature (
Figure 8) [
5,
11,
36,
37,
38,
39,
40,
41,
42,
43,
44,
45,
46,
47,
56,
59,
60,
61,
62,
63,
64,
65,
66,
67] for nominally pure CLN crystals of different origin, then in the
Table 3 they are not given. Experimental data show that Zn
2+ cations affect the LN structure less rigid than Mg
2+: zinc cations cause significant local distortions of the structure which manifest themselves in Raman spectrum at higher concentrations than Mg
2+ cations,
Figure 14.
The most likely reason for the appearance of extra bands is errors of polarization measurements and effect of photorefraction. These phenomena are a possible cause of the manifestation of forbidden fundamental oscillations of A
1 (LO) and E (TO, LO) symmetry types in the scattering geometry Y(ZZ)Ȳ where only A
1 (TO) vibrations are active. Mixed phonons may also appear due to the photorefractive effect [
5,
11,
36,
56]. Effect of photorefraction in studied heavily doped crystals is substantially less than in nominally pure crystals [
6,
39,
42,
43,
44,
59]. Thereby effect of photorefraction cannot serve as an explanation for this phenomenon. Of course, we can try to attribute the 230 cm
−1 band to forbidden fundamental oscillations of the E (TO) symmetry type, which manifest themselves in geometry Y(ZZ)Ȳ; 298 and 880 cm
−1—to forbidden fundamental oscillations of A
1 (LO) and E (LO) symmetry types. The 235, 298, and 880 cm
−1 bands are intense in the spectrum of nominally pure LN crystals in scattering geometries, where oscillations of the E (TO), E (LO), and A
1 (LO) symmetry types should manifest themselves. These bands were observed in many works and are reliably attributed to the fundamental oscillations of E (TO), E (LO), and A
1 (LO) symmetry types [
5,
11,
36,
37,
38,
39,
55,
56,
57,
58,
59,
60,
61,
62]. Actually, there are two bands with frequencies near 880 cm
−1 that correspond to A
1 (LO) and E (LO) phonons [
5,
36,
37,
55,
56,
57,
58]. A low-intensity 694 cm
−1 band was previously observed in the Raman spectrum of a nominally pure CLN crystal [
5,
36,
37,
63,
64]. Its attribution is still discussed [
5,
63,
64]. Besides, we found a low-intensity extra band with a frequency of 327 cm
−1 at 100 K in Raman spectrum of a CLN crystal near the 340 cm
–1 band (3A
1 (TO)). It is important that the detected band does not relate to a fundamental oscillation. The 327 cm
−1 low-intensity extra band disappears in effects of structural disordering and is not observed in the spectrum at higher temperatures and of doped crystals,
Figure 15.
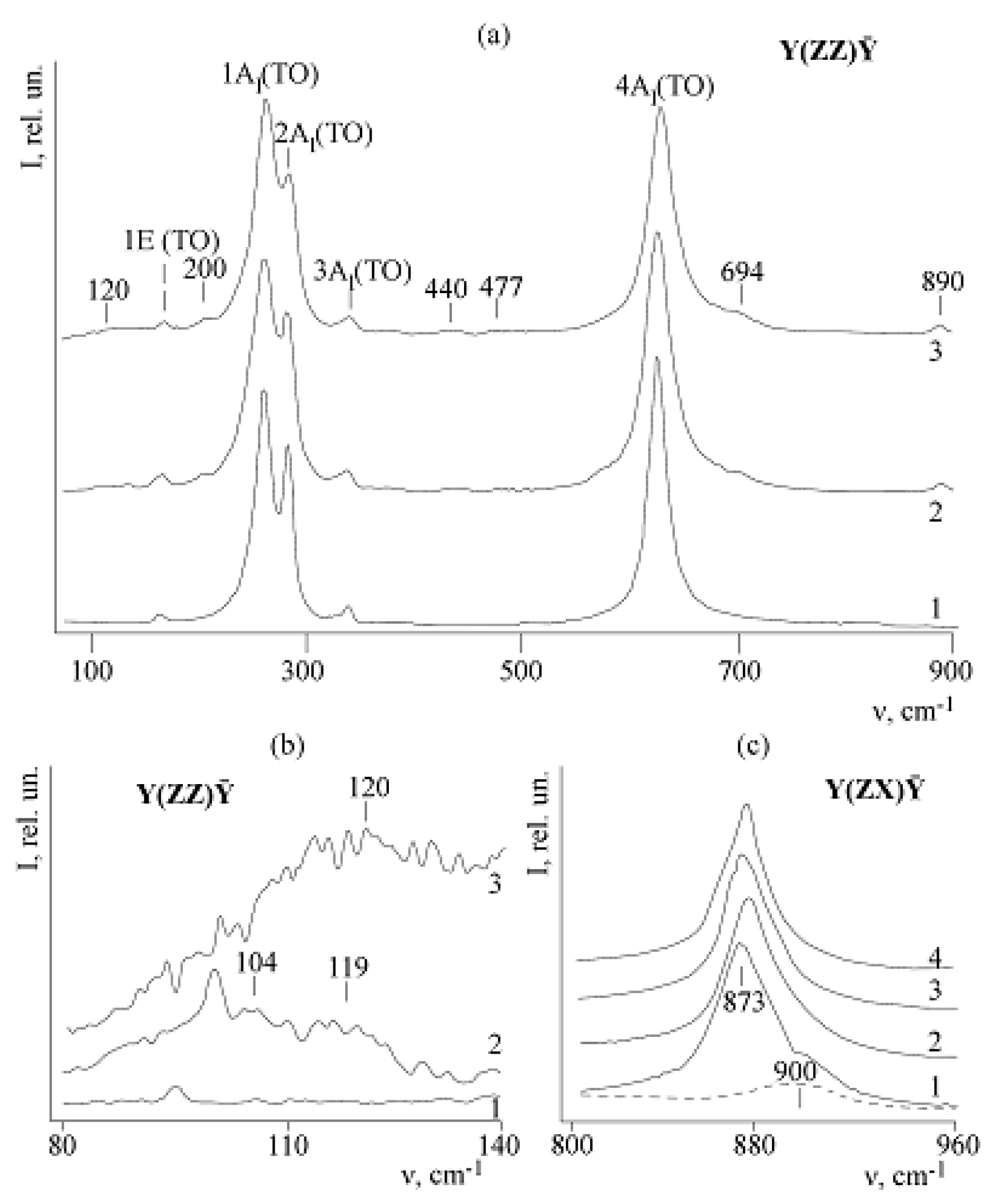
Figure 18 shows Raman spectra in scattering geometries Y(ZZ)Ȳ and Y(ZX)Ȳ of nominally pure SLN and CLN, as well as doped LiNbO
3:Zn (0.07, 5.84 mol.% ZnO) and LiNbO
3:Er (2.67 mol.% Er
2O
3).
The 880 cm
−1 band of is reliably attributed to forbidden fundamental vibration of A
1(LO) symmetry type—stretching bridge vibration of oxygen atoms Nb-O-Me (Me-Li
+ or dopant) along the polar axis in the scattering geometry Y(ZX)Ȳ. However, this band also appears as an extra band in other scattering geometries, for example, in scattering geometry Y(ZZ)Ȳ (
Figure 18a).
Figure 18c shows the transformation of the shape of 880 cm
−1 Raman band with a change in the composition of LN crystal and its complex contour. The contour separation program analysis revealed only one band in spectra of SLN, CLN, and LiNbO
3:Zn (0.07 mol.% ZnO) in the region of stretching bridge vibrations of oxygen atoms along the polar axis (
Figure 18, curves 2–4). Note that concentration of Zn-doped crystal in this case is relatively small. Two 873 and ≈900 cm
−1 bands are clearly observed in the spectrum of congruent crystals doped with relatively large amounts of impurity ions (in our case, Er) in the region of the stretching bridge vibrations of oxygen atoms along the polar axis (
Figure 18c, curve 1). A 900 cm
−1 band is highlighted separately with a dotted line on the
Figure 18c.
Thereby, nominally pure and lightly doped LN crystals demonstrate single-mode behavior within the homogeneity region. LN crystals show signs of two-mode behavior in the case of relatively high concentrations of dopants. Such a situation is possible when cations of the same type are located in different sites in oxygen octahedra due to the clusterization of the main and doping ions in the cation sublattice. This fact confirms the existence of superstructure ordered sublattices of cluster-like defects of different types; otherwise, it would be not a splitting but only a broadening of the corresponding band.
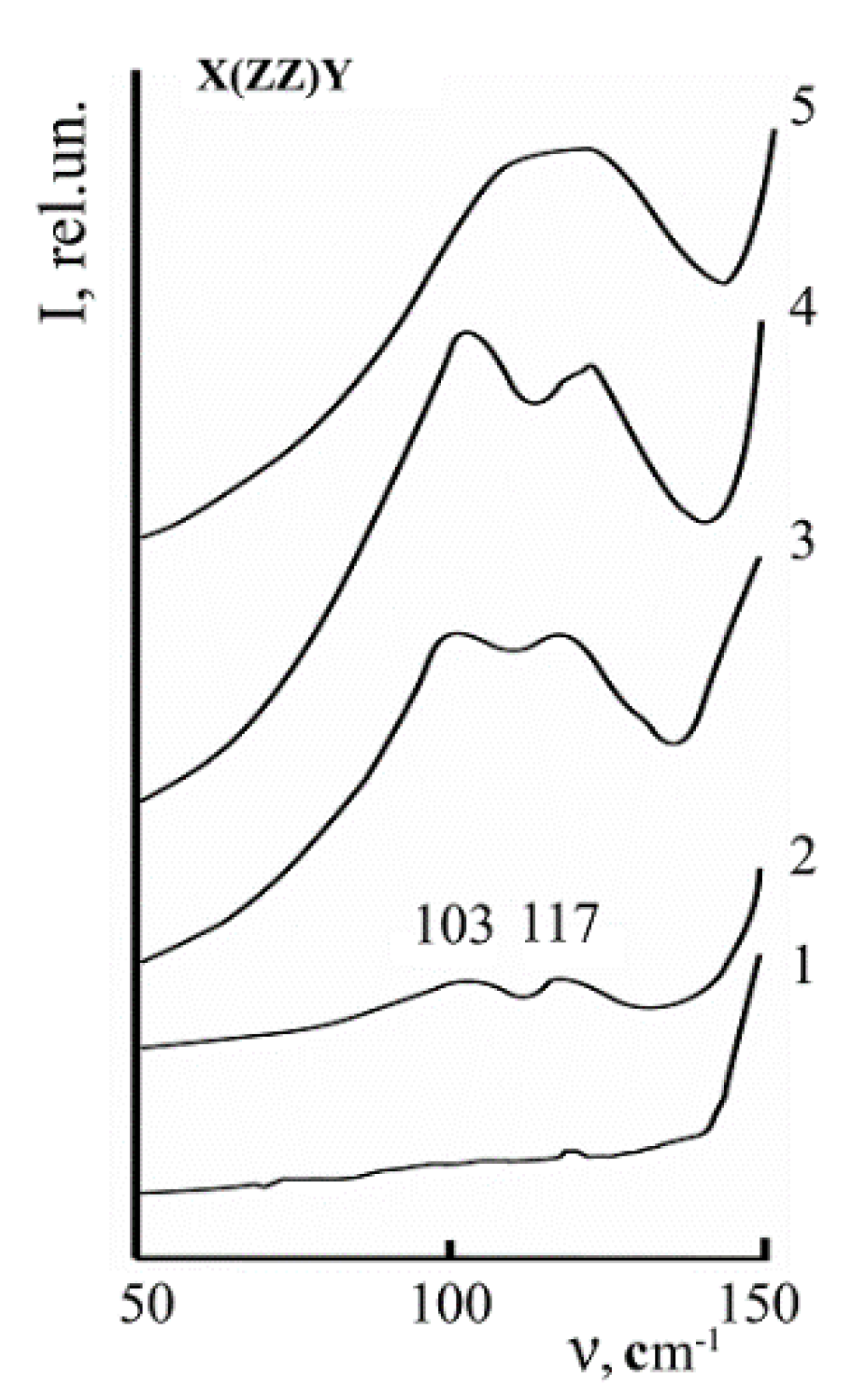
A low-intence 152 cm
−1 band corresponds to fundamental vibration of 1E(TO) symmetry type (
Table 3,
Figure 15). It is forbidden for the Y(ZZ)Ȳ scattering geometry and reveals due to polarization measurement errors. A low-intensity 120 cm
–1 band is observed in the low-frequency region of the Raman spectrum (
Figure 15). The band intensity increases with an increase in temperature [
5,
36,
37,
39,
55,
56]. This band corresponds to quasi-particle oscillations—two-particle states of acoustic phonons with a zero total wave vector [
5]. This band is split into two with frequencies of ~104 and 117 cm
−1 in spectra of highly ordered LN crystals (
Figure 15 and
Figure 19) [
5,
37]. The 120 cm
−1 band nature is discussed in [
69]. The intensity of this band is sensitive to very insignificant changes in the composition (including the number of Nb
Li defects) and fine features of the LN crystal structure. This feature allows the band to be an accurate experimental criterion stoichiometry of a LN single crystal composition [
5,
37,
39].
The other spectroscopic criterion for stoichiometry of LN composition is only one band in OH-groups stretching vibrations area of its vibrational spectrum [
10]. Appearance of complex defects with OH groups leads to splitting of this band into several components even at strictly stoichiomertic (Li/Nb = 1) composition [
10,
26,
27,
28,
70]. The number of components depends on particularities of protons localization in LN crystal structure.
There is no certain opinion in the literature about 120 cm
−1 band attribution. Authors of studies [
60,
65] associate it with phonons of E symmetry. The latter are forbidden in the scattering geometry X(ZZ)Y but manifest in it due to internal stresses in the crystal. The authors of the study [
61] associated the origin of the band with the scattering of radiation on the different optical phonons of A
1 (soft mode) and E symmetry type. Authors of [
61] believe that the interaction of phonons of different symmetry is possible due to violation of the selection rules for E-type phonons. Violation of the selection rules is caused by intrinsic and impurity defects characteristic of a LN crystal. At the same time, they interpret the band in the region of 120 cm
−1 as a zone of different tones of three low-frequency branches of A
1 and E symmetry type optical oscillations. Thus, the region of 120 cm
−1 should not contain a band at low temperatures, since the intensity of the difference tones in this case is zero. This opinion was taken as the main one until recent times. Later detailed measurements of the temperature evolution of the spectra in polarized radiation with different scattering geometries, performed in [
55], suggested that a band in the 120 cm
−1 region corresponds to two-phonon acoustic excitations: the pattern of the Raman spectral intensity distribution in the low-frequency region is characteristic of Fermi phonon resonance. The manifestation of two-phonon acoustic excitations in the Raman spectrum is explained by the effective attraction between acoustic phonons. The attraction takes place considering the anharmonic interaction of the lowest-frequency optical A
1 symmetry type phonon (254 cm
−1, quasi-soft mode) with a two-phonon acoustic continuum [
61].
We used the intensity of the 120 cm
−1 band when developing the obtaining technology of SLN(K
2O) by HTTSSG method. Potassium does not incorporate into the LN structure due to the large ionic radius.
Figure 20 shows the approach of a LN crystal to a stoichiometric composition with an increas in K
2O concentration in the melt. When the content of K
2O in the melt is 6.0 wt. %, ratio Li/Nb = 1 and the crystal composition corresponds to the stoichiometric. The stoichiometric crystal is characterized by a zero intensity of the 120 cm
−1 band and minimal values of the Raman bands widths,
Figure 20.
Stoichiometric crystals grown by the HTTSSG method with the addition of K2O flux are characterized by a uniform refractive index along the growth axis. At the same time, crystals grown due to a conventional technology from the melt with 58.6 mol.% Li2O are characterized by ununiformity of the refractive index along the growth axis. Such crystals are unsuitable for practical applications.
The periodic (discontinuous) structure of the laser beam along the direction of its propagation is observed in SLN crystal,
Figure 21. At the same time, LN crystals are not gyrotropic. The discontinuous structure of the laser beam is not observed in non-stoichiometric crystals or in a stoichiometric crystal grown by the HTTSSG method. The high intensity of laser radiation in the region of the focal waist can lead to the formation of bound states of two photons (baundtons) [
71]. The latter move in a crystal and break up into two photons. Thus, the discontinuous trajectory of the laser beam can be realized not only in gyrotropic crystals. This effect may be due to the formation of bound states of two photons as a result of various nonlinear-optical processes: two-photon absorption, stimulated Raman scattering, etc. [
71]. The periodic structure of a laser beam in a crystal does not affect the Raman spectrum corresponding to the fundamental oscillations of the LNcrystal lattice.
Extra bands with frequencies 209, 230, 298 cm
−1 are observed only in Raman spectra of heavily doped crystals LiNbO
3: Zn (4.5 mol.% ZnO), LiNbO
3:Mg:Fe (5.01; 0.005), LiNbO
3: Mg (5.1), LiNbO
3: Mg (5.3 mol.% MgO), (
Figure 14 and
Figure 18a). Their intensity depends on the crystal composition and increases as the concentration of dopants approaches threshold. These bands are not observed in the spectrum of nominally pure crystals. At the same time, various authors observed a sufficiently large number of other extra bands in the Raman spectrum of congruent crystals (85, 92, 100, 120, 187, 305, 331, 412, 477, 535, 605, 668, 690, 739, 743, 825 cm
−1 (
Figure 1). These extra bands did not correspond to the fundamental lattice vibrations [
5,
11,
36,
37,
38,
39,
40,
41,
42,
43,
44,
45,
46,
47,
56,
59,
60,
61,
62,
63,
64,
65,
66,
67]. There may be several reasons for the appearance of extra bands in the Raman spectrum of nonstoichiometric (nominally pure and doped) LN crystals. Polarization measurement errors are not the only reason. Strong phonon-phonon interaction is characteristic of defective and anharmonic crystals, such as LN. This can lead not only to the broadening of bands corresponding to the fundamental lattice vibrations but also to the appearance of new bands in the spectrum. This happens because quasiparticles are formed from interacting phonons (optical and acoustic) and mix one-phonon with two-phonon states due to Fermi resonance [
5,
72]. First of all, quasiparticles with a zero total wave vector should manifest themselves in the processes of Raman and IR absorption, [
5,
36,
55,
56]. Regulation of the conditions of anharmonic interaction can be carried out either by varying the order of cations in the cation sublattice or by changing the temperature [
5,
55]. With an increase in temperature (as well as an increase in the disordering of the structural units of the cation sublattice), maxima of 104 and 119 cm
−1 merge into a wide maximum with a frequency of 120 cm
−1. [
5,
39,
55]. This is a confirmation of the anharmonic nature of these Raman bands.
The phases of other lithium niobates are likely to precipitate as microstructures, and the crystals are multiphase in heavily doped crystals,
Figure 9,
Figure 10,
Figure 11,
Figure 12 and
Figure 13. However, none of the possible phases of other lithium niobates (LiNb
3O
8 or Li
3NbO
4) that may precipitate during crystallization of LiNbO
3 (lithium metaniobate) [
5] does not have bands with frequencies 209, 230, 298 cm
−1 or close to it in its Raman spectrum [
4,
5,
37,
73]. At the same time, according to the data of study [
51], the precipitation of the Li
6ZnNb
4O
14 phase is possible in the form of microinclusions at dopant concentrations above the threshold.
The appearance of extra bands in the Raman spectrum can be caused by chaotic disturbances of perfect cation sublattice, such as point defects Nb
Li, Li
□, Nb
□, and other. Such chaotic violations should lead to the dephasing of oscillations in the crystal [
5,
74,
75,
76,
77]. At this corresponding atoms of each unit cell of the crystal cease to oscillate in phase. The “opening” of the Brillouin zone is possible [
74,
77] at high dopant concentrations in the structure when point defects Nb
Li, Li
□, Nb
□ and other are located randomly. Then, the observation has access not only to the limiting (
kp = 0) frequencies of the optical branches, but also to other frequencies in the Brillouin zone in the Raman spectrum with an intensity proportional to the concentration of defects. Frequencies in the Brillouin zone are determined by the wave vector spread |Δk|~2π/l [
74,
77]. Taking into account the small dispersion of optical branches [
5], this can lead to the appearance in a LN crystal spectrum of rather narrow additional (extra) bands that are not allowed by the selection rules for a given spatial group. Due to numerous literature data (the review is given in [
5]), when R = Li/Nb decreases, several changes occur in Raman spectra: widths of bands corresponding to fundamental lattice vibrations of nominally pure crystals increase; widths of extra bands in the 120 cm
−1 region also increase (this band corresponds to the two-particle states of acoustic phonons with a zero total wave vector). At the same time, the spectrum fully retains its individuality [
5,
11,
36,
39].
Factors listed above and the increase in the number of spatial defects in the form of clusters and microstructures with a structure distorted compared to the crystal matrix (i.e., an increase in the microheterogeneity of the crystal) can lead to significant local changes in the crystal symmetry. Bands may appear that correspond to forbidden acoustic optical vibrations of (A
1 and E) and A
2 symmetry type in the Raman spectrum. The distortion of the perfect structure of a LN crystal is especially significant at high concentrations of doping metals close to threshold values. The whole spectrum of fundamental oscillations of a crystal should be radically broadened and significantly deformed for such strongly deformed structures. This is observed experimentally,
Figure 14. Thus, it is important to compare the frequencies of experimentally observed 104, 119, 209, 230, 298, 694, 880 cm
−1 bands in the Raman spectrum of non-stoichiometric LN crystals (
Table 2) with calculated data.
The LN crystal lattice dynamics was analyzed both by ab initio methods [
57,
58] and in the valence-force field approximation [
68]. Results of ab initio calculations of frequencies, vibration modes, atomic coordinates, and unit cell parameters turned out to be very close to the experiment [
57,
58]. Ab initio calculations give the unit cell parameters: a = 5.1378 Å и c = 13.4984 Å. This is only 2% to 3% less than the experimental values. The discrepancy between the frequencies calculated in [
57,
58] and those experimentally observed by us of A
1 (TO) and E (TO) of types symmetry does not exceed 5%. This indicates a high accuracy of ab initio calculations. Fundamental oscillations of A
2 symmetry type calculated ab initio in [
57,
58] in the center of the Brillouin zone (k = 0) and prohibited by the selection rules for the space group C
3V6 (R3c) are listed in
Table 3 and labeled by 2.
Table 3 shows that there is a satisfactory coincidence of the frequencies of the experimentally observed low-intensity extra bands with frequencies of 209, 230 and 880 cm
−1 and vibration frequencies of 1A
2, 2A
2, 5A
2 symmetry type calculated ab initio. At the same time, there is a serious discrepancy between the calculated frequencies of 3A
2 and 4A
2 and the experimentally observed bands with frequencies of 298 and 694 cm
−1,
Table 2. Unfortunately, it is impossible to calculate the frequency of A
2 the symmetry type oscillations prohibited by the selection rules in the approximation of the valence-force field [
68].
Raman spectra that we studied clearly show that doped LN crystals have super-structure lattice of cluster defects several unit cell in diameter (which are probably the structure elements revealed on
Figure 6). Thus we conclude that LN crystals grown in conditions far form thermodynamic equilibrium have periodic fractal-like structures ~1 nm to 1000 μm. Thus, when we write that extra Raman bands appear because of domains we mean not classic ferroelectric domains that have spontaneous polarization and structure of the domains is identical to the structure of matrix lattice. We mean micro- and nano-structures and clusters connected with classic domains (as on
Figure 2,
Figure 3,
Figure 4,
Figure 5,
Figure 6,
Figure 7,
Figure 8,
Figure 9,
Figure 10,
Figure 11,
Figure 12,
Figure 13 and
Figure 14). The exact nature of these structures is unfortunately yet unclear. Intrinsic structure of such clusters is different from that of a crystal matrix. We believe that it is these clusters and these nano-structures that cause appearance of low-intensity extra Raman bands.
3.4. Effects of Ordering of Structural Units of Cation Sublattice in LiNbO3:Zn Crystals and Their Manifestation in Raman Spectra
When doping a LN crystal with “non-photorefractive” Me cations (Me = Zn
2+, Mg
2+, Gd
3+, In
3+, etc.) following processes change non-monotonously: ordering of the structural units of the cation sublattice along the polar axis, deformation of the oxygen octahedra NbO
6, the state of defectiveness of the crystal as a whole [
3,
4,
5,
6,
14,
15,
16,
37,
73,
78]. At the same time, pronounced anomalies are observed, indicating a “threshold” nature of the impurity cations incorporation into the structure on concentration dependences of physical characteristics at certain dopant concentrations [
5,
6,
14,
15]. For example, doping a congruent LiNbO
3 crystal with Zn
2+ cations leads to a change in the polarizability of the oxygen octahedra NbO
6, parameters of the unit cell of the crystal and electro-optical characteristics [
5,
6,
14,
15,
16]. The impurity incorporation mechanism has a “threshold” character and is determined by the concentration of Zn
2+ [
5,
6,
14,
15]. The coefficients of a linear electro-optical effect in a LiNbO
3:Zn single crystal are less than in a CLN crystal and show a minimum at concentrations of ~2–3 mol.% ZnO and a maximum at ≈7 mol.% ZnO [
5,
14]. At a concentration above 7 mol.% ZnO, the photorefractive effect is small and with a further increase in zinc concentration remains almost unchanged [
5,
6,
14]. At a concentration of Zn
2+ above 7 mol.%, point defects Nb
Li are completely absent, and Zn
2+ cations in certain proportions occupy the main sites of Li
+ and Nb
5+ cations in an LiNbO
3:Zn crystal [
6,
14].
It is important to investigate fine features of the concentration restructuring of the LiNbO
3:Zn crystal structure before the first threshold concentration of Zn
2+, i.e., in the concentration range 0–2 mol.% ZnO [
15]. The greatest decrease in the photorefractive effect occurs in this area [
5]. The photorefractive effect decreases from 3.1 × 10
2 to 6.6 × 10
2 W/cm
2 in the concentration range 0–3 mol.% ZnO, and in the concentration range 5–7 mol.% Zn
2+ photorefractive effect decreases from 7.1 × 10
2 to 9.8 × 10
2 W/cm
2. The greatest change in the photorefractive effect is observed in the region of the first concentration threshold, and the smallest photorefractive effect is characteristic of the region of the second concentration threshold.
Figure 23 shows Raman spectra of SLN, CLN, LiNbO
3:Zn (0–1.59 mol.% ZnO) single crystals in scattering geometries Y(ZX)Ȳ and Y(ZZ)Ȳ (the fundamental phonons of the E (TO) and A
1 (TO) symmetry types are active, respectively, [
5]). Changes in the main parameters of the bands (frequencies, widths, and intensities) depending on the composition of the crystal are presented in
Table 7.
Table 7 shows that frequencies of most bands practically do not change with a change in the composition of the crystal. This indicates the invariance of quasi-elastic oscillation constants. However, widths of bands change noticeably. Widths of all bands are minimal in the spectrum of a SLN crystal due to the most ordered cation sublattice.
In
Table 7 v (cm
−1) is a frequency,
s (cm
−1) is a bandwidth, I (%) is a relative spectral intensity. It calculates as an intencity of a studied band, relates to an intencity of 580 cm
−1 band, [Zn] (mol.%)—concentration of Zn
2+ cations in a crystal.
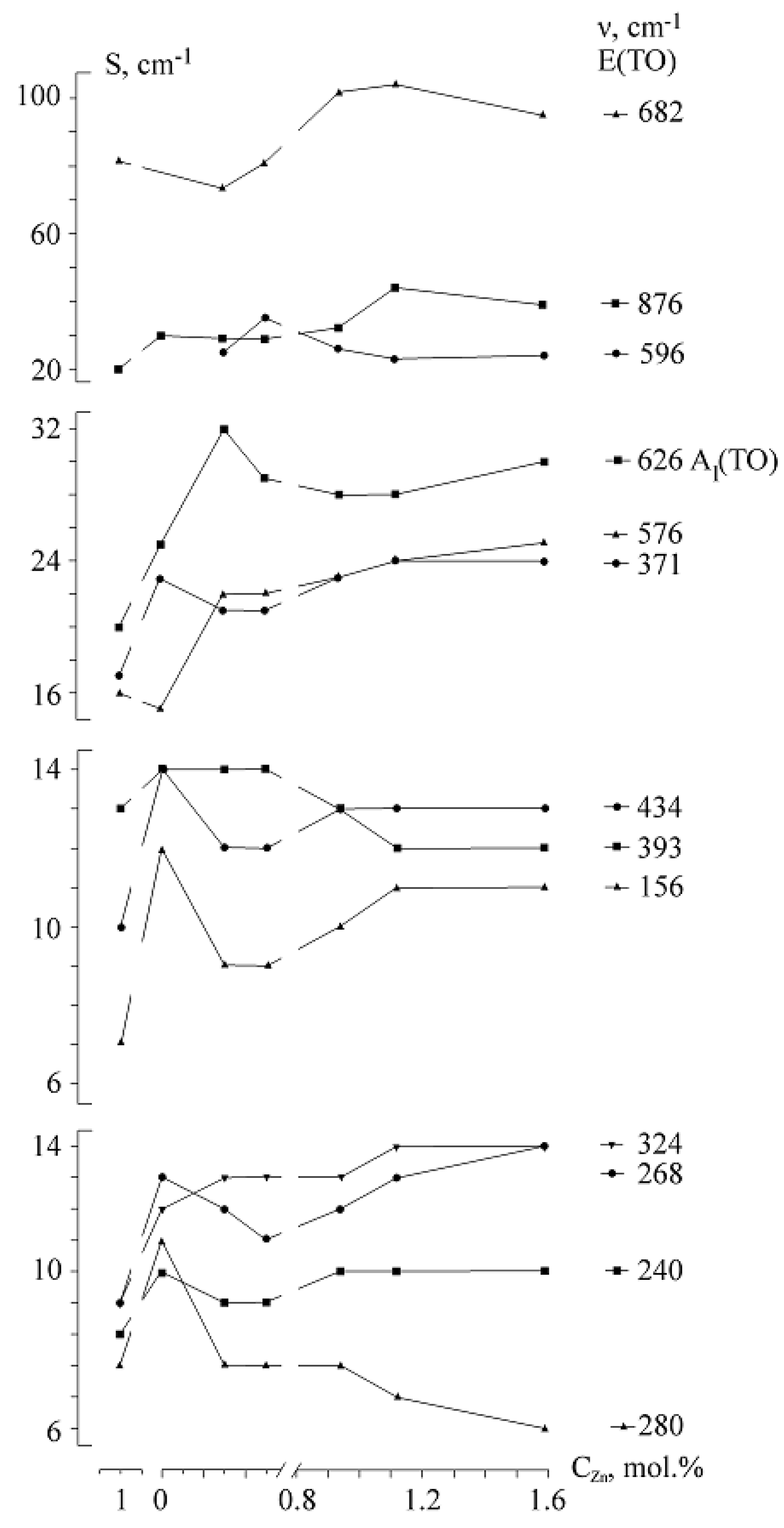
Concentration dependences of widths of many bands in the spectra of LiNbO
3: Zn crystals have a minimum in the concentration region of 0.05–1.12 mol%,
Figure 24.
Figure 24 demonstrates that widths of some bands change nonlinearly with increasing Zn
2+ concentration in a LiNbO
3:Zn crystal. The bandwidths decrease in concentration ranges of Zn
2+ 0–0.94 mol.%, and then they increase in concentration range of Zn
2+ 0.94–1.59 mol.%. The minimum is the lowest for concentration dependences of band widths with frequencies of 156, 240, 268, 371, 434, 576 and 876 cm
−1 (E (TO)) and 254 and 274 cm
−1 (A
1 (TO)). These frequencies correspond to the vibrations of the Nb
5+ and Li
+ cations in oxygen octahedra and vibrations of oxygen octahedra. A decrease in widths of bands with frequencies of 254 and 274 cm
–1 corresponding to totally symmetric (A
1 (TO)) vibrations of Nb
5+ and Li
+ ions along the polar axis, clearly indicates the ordering of the cationic sublattice of LN crystal along the polar axis in the concentration range of Zn
2+ 0.05–1.12 mol%. A decrease in the width of a band with a frequency of 626 cm
–1 corresponding to totally symmetric (A
1 (TO)) vibrations of oxygen octahedra means that oxygen octahedra become more perfect,
Figure 24.
There is a region of increased ordering of the structure in LiNbO
3:Zn crystals (0.05–0.94 mol.% ZnO) when the order of alternation of the main, impurity cations and vacancies along the polar axis is increased in the cation sublattice, and the oxygen octahedra are close to perfect. The maximal ordering of the LiNbO
3:Zn crystal structure the is observed at Zn
2+ concentrations of ≈0.05–0.94 mol.% ZnO. At the same time, the band widths in the Raman spectrum of LiNbO
3:Zn crystals ([Zn] ≈ 0.05–0.94 mol.% ZnO) are smaller than in the spectrum of a CLN crystal and approach widths of bands in the spectrum of the SLN crystal,
Figure 24. This indicates a high degree of ordering of the structural units of the cationic sublattice of LiNbO
3:Zn (0.05–0.94 mol.% ZnO) crystals and its approach to the degree of ordering of a SLN crystal. We obtained similar results earlier on Raman spectra for congruent lithium niobate crystals doped with Mg
2+ and Gd
3+ [
5,
78,
79].
Our spectroscopic data for LiNbO
3: Zn crystals ([Zn] = 0–1.59 mol.%) correlate well with the concentration dependence of the unit cell parameters determined by X-ray diffraction [
5,
80]. The minimum is observed in the concentration range 1–2 mol.% ZnO on the concentration dependence of the parameter c the unit cell. While, according to Vegard’s law, there should be an increase in the parameter c with an increase in the ionic radius of the dopant cation in comparison with the replaced cation of the matrix. The ionic radii of Zn
2+, Li
+, and Nb
5+ are 0.74, 0.68, and 0.68 Å [
5].
A low-intensity 682 cm
−1 band is present in the spectra of CLN, LiNbO
3: Zn etc. crystals (
Figure 1,
Figure 14,
Figure 18 and
Figure 23). Its manifestation, according to the data of [
55], is due to the activity of biphonons in the Raman scattering. However, according to other studies [
57,
58,
63,
64], this band belongs to fundamental phonons of the A
1 (TO) symmetry type. We did fail to find a band with a frequency of 682 cm
−1 as well as a band with a frequency of 120 cm
−1 (
Figure 18) corresponding to two-particle states of acoustic phonons with a zero total wave vector equal in the Raman spectrum of a SLN single crystal.
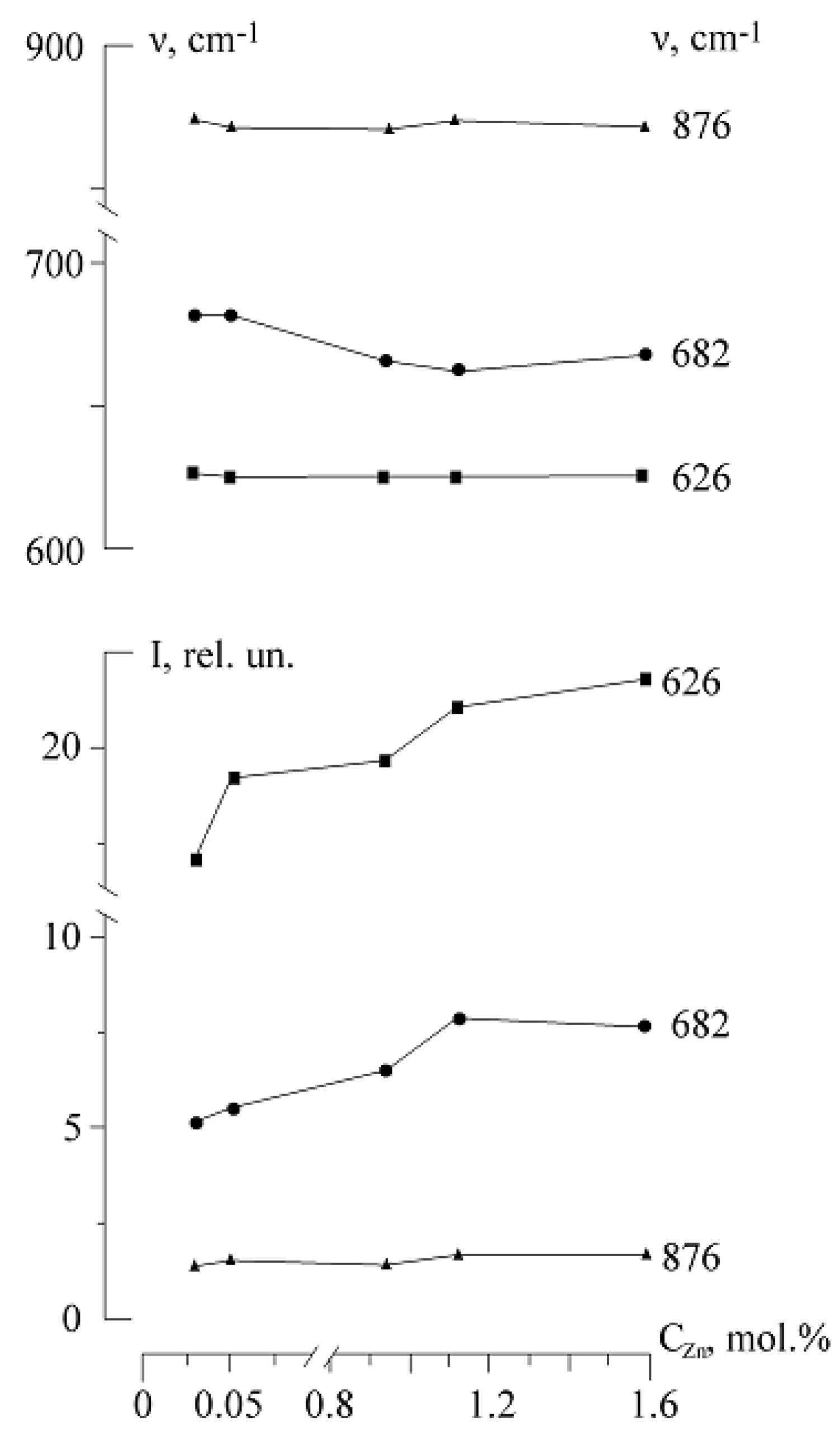
Figure 24 and
Figure 25 show that in the region of 0–1.12 mol.% ZnO width and intensity of the 682 cm
−1 band monotonously increase with increasing Zn
2+ concentration, and frequency, on the contrary, decreases. However, with a further increase in the Zn
2+ concentration to 1.59 mol.%, there is a significant decrease in the band width (at 9 cm
−1,
Figure 24,
Table 7). This indicates the manifestation of ordering effects. In this case, the frequency and the intensity of the band, on the contrary, increase. According to [
55], an increase in the intensity of the 682 cm
−1 band corresponds to an increase in the concentration of Nb
Li defects. However, this contradicts the results of [
5,
14,
15,
80,
81]. The mentioned studies unambiguously show that the number of Nb
Li defects decreases in LiNbO
3:Zn crystals with an increase in the concentration of Zn
2+. At Zn
2+ concentrations from 0 to 5 mol.% ZnO, Nb
Li point defects are mostly substituted with Zn
2+ cations. The 682 cm
−1 band is manifested in spectra of LiNbO
3:Zn crystals and does not appear in the spectrum of the SLN. It also has a high sensitivity to the concentration of Zn
2+ cations in the crystal structure, and the behavior of its basic parameters (
Figure 24 and
Figure 25) may indicate a threshold incorporation of Zn
2+ in the crystal structure at concentrations ≈0.94–1.12 mol.% ZnO.
The width of 876 cm
−1 band (E (TO), corresponds to the stretching bridge oscillations of oxygen atoms along the polar axis), behaves similarly to the 682 cm
−1 band width. The 876 cm
−1 band is forbidden by the selection rules in the spectrum of the centrosymmetric paraelectric phase of a LN crystal [
63,
64]. The change in the behavior of the 876 cm
−1 band width is observed at a concentration of 1.12 mol.% ZnO,
Figure 25. In the literature, the intensity of the 876 cm
−1 band is used to assess the quality of ferroelectrics properties of crystals with an oxygen-octahedral structure [
5,
82]. An increase in the intensity of this band means more ordered cation sublattice along the polar axis, which in its turn indicates an increase in the spontaneous polarization of the crystal.
The presence of a region of increased ordering of the structural units of the cation sublattice in LiNbO
3:Zn crystals can be explained as follows. The main point defects of CLN (Li/Nb = 0.946) are Nb
Li—Nb
5+ cations in lithium sites. Niobium occupies the position of Li
+ cations of a perfect stoichiometric structure [
3,
5,
22]. For electroneutrality reasons, the formation of Nb
Li defect leads to the appearance of four-point defects in the form of vacant oxygen octahedra. Zn
2+ cations шncorporating into the congruent crystal structure displace Nb
Li defects, which is energetically efficient [
5,
45]. Small amounts of Zn
2+ cations occupying lithium oxygen octahedra (in which Nb
Li defects were located), order the alternation of cations and vacancies along the polar axis and reduce the crystal deficiency with respect to Li
+ vacancies [
5,
45]. The addition of Zn
2+ cation to the vacant oxygen octahedron of a perfect structure along with a decrease in Li
+ vacancies leads to an additional increase in the structure’s defects due to the violation of the existing order of cations and vacancies along the polar axis of the crystal.
Thus, when small amounts of Zn2+ cations are introduced into the cation sublattice of a congruent lithium niobate crystal, two mechanisms compete with each other. One leads to the ordering of cations along the polar axis and a decrease in cation vacancies. The other leads to disruption of the order of the cations along the polar axis by the impurity cations Zn2+. At low concentrations of Zn2+ cations, the ordering mechanism prevails. It leads to a decrease in widths of bands in the Raman spectrum and the c parameter of the unit cell. With an increase in the concentration of Zn2+ cations, the disordering mechanism begins to predominate, and widths of bands and parameter c increase.
The data obtained allow us to state that there is a smooth change in the ordering of the structural units of the cation sublattice and crystal properties when the Zn
2+ cations incorporate into the structure of the congruent lithium niobate crystal. This is explained by the fact that with an increase in Zn
2+ concentration, two interrelated processes take place simultaneously—the displacement of Nb
Li defects and the entry of Zn
2+ cations into vacant octahedra of a perfect structure. According to the data of [
6,
14,
15], even at 3 mol.% ZnO, Nb
Li defects are present in the crystal. And their final displacement occurs only when the concentration of Zn
2+ ≥ 8 mol.% ZnO. Thus, Zn
2+ cations can be quite fine and effective regulators of the number of Nb
Li defects in the structure of the LN crystal. This is important for the purposeful creation of optical materials with specified characteristics.
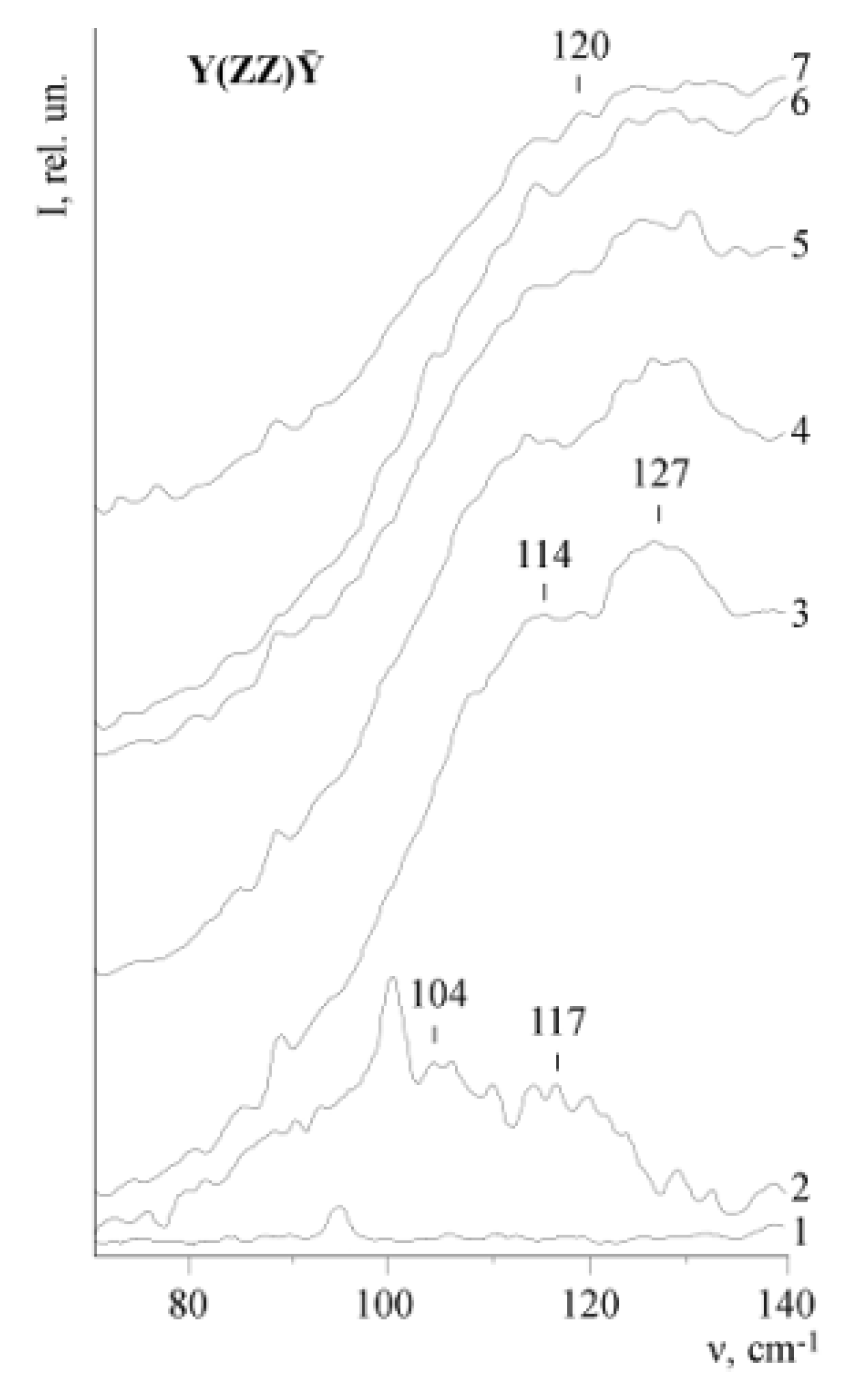
Figure 26 shows a fragment of the Raman spectrum of SLN, CLN, LiNbO
3:Zn single crystals in the low-frequency region (50–140 cm
−1). This region contains no bands corresponding to the fundamental phonons [
5,
36,
55]. A low-intensity wide 120 cm
−1 band is observed in the spectrum of the CLN crystal in scattering geometry Y(ZZ)Ȳ (phonons of A
1 (TO) symmetry type are active). The band corresponds to quasi-particle oscillations—two-particle states of acoustic phonons with a zero total wave vector. At the same time, this bands is absent from the Raman spectrum of a highly ordered SLN single crystal. It is important that the intensity of this band is sensitive to changes in the acoustic Q-factor of the LN crystal [
83]: the higher the intensity of the 120 cm
−1 band is, the lower the acoustic Q-factor quality is. The SLN single crystal has the highest acoustic Q-factor quality, thus the 120 cm
−1 band is absent from its spectrum.
The processing of the spectra by the programs for the separation of the contours of the spectral bands shows that the band with a frequency of ≈120 cm
−1 has a structure and is a superposition of two bands with frequencies ≈104 and 117 cm
−1 in the spectra of LN crystals,
Figure 26. The presence of components is also confirmed by calculations of the density of phonon states, performed in [
83,
84].
Figure 26 shows that the intensity of the 120 cm
–1 band changes with a change in the concentration of Zn
2+ in LiNbO
3:Zn crystals. With an increase in Zn
2+ concentration, the structure of the 120 cm
−1 band gradually disappears,
Figure 26. At a concentration of 1.59 mol.%, only one maximum appears at a frequency of 120 cm
−1. For crystals of LiNbO
3:Mg and LiNbO
3:Gd, the incorporation of an impurity cation into the lattice occurs stepwise and is more sharp than for LiNbO
3:Zn crystals [
5,
6,
16,
44]. The broadening of the 254 and 274 cm
−1 bands (A
1 (TO)), the disappearance of the structure and an increase in the intensity of the 120 cm
−1 band occur in these crystals a narrower range of impurity concentrations [
5,
78,
79].
The observed effects for the 120 cm
−1 band, in our opinion, can be explained by the resonant interaction of fundamental oscillations with frequencies of 254 and 274 cm
−1 A
1 (TO) between themselves and with two-particle states of acoustic phonons in the region of 120 cm
−1 with a zero total wave vector. The manifestation of such an interaction in the vibrational spectrum of an anharmonic crystal was considered in [
83,
84]. For a highly ordered LN cation sublattice (in particular, for crystals close to the stoichiometric composition), the interaction covers a relatively narrow part of the spectrum. For such highly ordered structures, fundamental vibrations with frequencies of 254 and 274 cm
−1 (A
1 (TO)) interact with two-particle states of acoustic phonons (A
1 (TO)) almost independently of each other. This is expressed in the appearance of two maxima 104 and 117 cm
−1 in the spectrum in the region of two-particle excitations of acoustic phonons. With increasing disorder in the cation sublattice (as well as increasing anharmonicity with increasing crystal temperature), fundamental oscillations with frequencies of 254 and 274 cm
−1 begin to interact with each other. At the same time, the magnitude of the part of the spectrum where the resonant interaction of fundamental oscillations and two-particle states of acoustic phonons occurs is expanding. This is expressed in an increase in the intensity and width of the bands corresponding to the two-particle states of acoustic phonons with a zero total wave vector. With a sufficiently strong interaction, the maxima of 104 and 117 cm
−1 merge into a wide maximum with a frequency of 120 cm
−1,
Figure 15,
Figure 18,
Figure 19 and
Figure 26.
Thus, the appearance of a 120 cm
−1 band in the Raman spectrum may be caused by the deviation of the order of cations (Li
+, Nb
5+, vacant octahedra) along the polar axis in non-stoichiometric crystals from the order of cations in a perfect structure of stoichiometric composition. At this an increase in the number of Nb
Li defects is observed. Probably, the intensity of the 120 cm
−1 band increases with an increase in the number of defects (including Nb
Li defects) associated with the disruption of the order of alternation of Li
+, Nb
5+ cations and vacant octahedra along the polar axis. The splitting of the band into two components can then be due to an improvement in the selection rules for the wave vector of two-particle states of acoustic phonons of the A
1 (TO) symmetry type. This occurs due to a decrease in the uncertainty of the quasiparticle wave vector when ordering the alternation of cations and voids along the polar axis [
83,
84]. An increase in disorder in the cation sublattice, on the contrary, should lead to an increase in the uncertainty in the wave vector of two-particle states of acoustic phonons of the A
1 (TO) symmetry type and the probability of two-phonon transitions [
83,
84]. Oscillations will appear in an ever-increasing region of the Brillouin zone in the Raman spectrum. All this should lead to a blurring of the structure of the 120 cm
–1 band, an increase in its intensity and a broadening of the bands corresponding to totally symmetric fundamental vibrations of cations. The experimental data obtained also indicate a decrease in the number of Nb
Li defects with an increase in the concentration of Zn
2+ in LiNbO
3:Zn crystals. However, to reliably establish the dependence of the band intensity with a frequency of 120 cm
−1 on the number of Nb
Li defects, further studies of the Raman spectra of LiNbO
3:Zn crystals at high Zn
2+ concentrations, up to 8 mol.%, are necessary. At such high Zn
2+ concentrations, Nb
Li defects are practically absent [
5,
6], and the band intensity with a frequency of 120 cm
−1 can be close to zero.