Mineralogical Characteristics and Color Genesis of Vesuvianite Jade from Hanzhong, Shaanxi Province, China
Abstract
1. Introduction
2. Materials and Methods
2.1. Samples
2.2. Methods
3. Results and Discussion
3.1. Conventional Gemological Testing
3.2. Petrography and Microstructure
3.3. X-Ray Powder Diffraction
3.4. X-Ray Fluorescence Spectroscopy
3.5. Infrared Spectroscopy
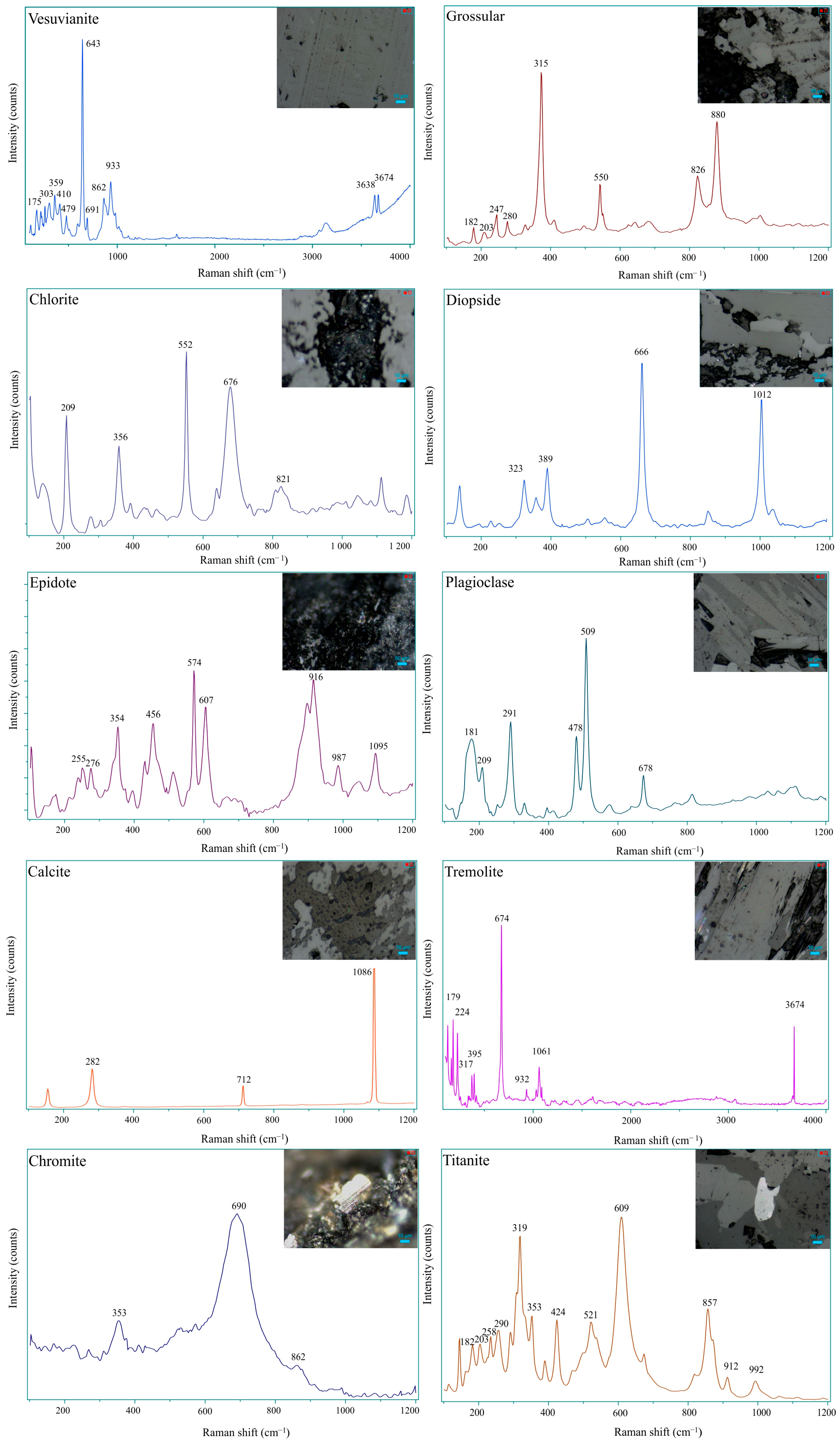
3.6. Raman Spectroscopy
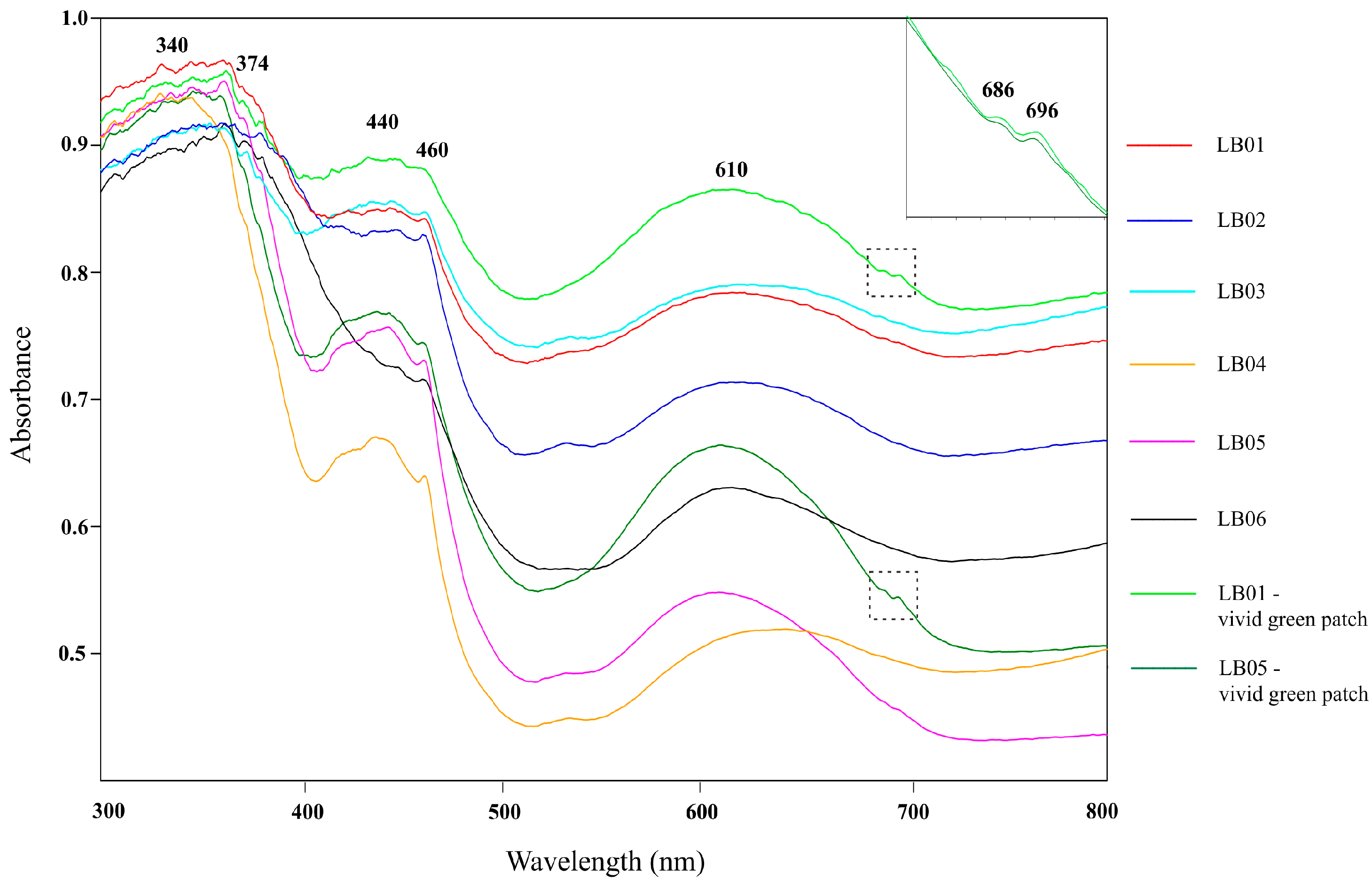
3.7. UV-Visible Spectroscopy
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Spencer, L.J. Vesuvianite. In Encyclopædia Britannica, 11th ed.; Chisholm, H., Ed.; Cambridge University Press: Cambridge, UK, 1911; Volume 27, p. 1063. [Google Scholar][Green Version]
- Caucia, F.P.; Marinoni, L.; Scacchetti, M.; Riccardi, M.P.; Bartoli, O. The Vesuvianite Gems of the Val d’Ala (Piedmont, Italy). Minerals 2020, 10, 535. [Google Scholar] [CrossRef]
- Lu, Z.; He, X.; Lin, C.; Liang, L.; Jin, X.; Guo, Q. Color and genesis of californite from Pakistan: Insights from μ-XRF mapping, optical spectra and X-ray photoelectron spectroscopy. Sci. Rep. 2020, 10, 285. [Google Scholar] [CrossRef]
- Pabst, A. Vesuvianite from Georgetown, California. Am. Mineral. 1936, 21, 1–10. [Google Scholar]
- Chamberlain, S.C. The Jeffrey mine: In Asbestos, Quebec, Canada. Rocks Miner. 1980, 55, 188–191. [Google Scholar] [CrossRef]
- Cao, Z.; Zhu, H. Mineralogical study on macrocrystals of vesuvianite. Acta Petrol. Mineral. 2020, 19, 69–77. [Google Scholar]
- Dyrek, K.; Platonov, A.N.; Sojka, Z.; Zabinski, W. Optical absorption and EPR study of Cu2+ ions in vesuvianite (“cyprine”) from Sauland, Telemark, Norway. Eur. J. Mineral. 1992, 4, 1285–1289. [Google Scholar] [CrossRef]
- Veblen, D.R.; Wiechmann, M.J. Domain structure of low-symmetry vesuvianite from Crestmore, California. Am. Mineral. 1991, 76, 397–404. [Google Scholar]
- Xu, Y. Gemmological and Mineralogical Studies on Characteristics of Hydrogrossular Jade and Vesuvianite Jade from Tawmaw Area, Burma. Master’s Thesis, China University of Geosciences (Beijing), Beijing, China, 2014. [Google Scholar]
- Zhong, H. Skarn-type vesuvianite jade: Discovery of the Huilong jade deposit and characteristics of the jade. Min. Resour. Geol. 1996, 10, 256–258, (In Chinese with English abstract). [Google Scholar]
- Li, G.; Yu, Y. Gemmological study on vesuvianite jade from Manasi, Xinjiang. In Proceedings of the International Symposium on Jade Science, Beijing, China, 11 September 2011; pp. 201–206, (In Chinese with English abstract). [Google Scholar]
- Groat, L.A.; Hawthorne, F.C.; Ercit, T.S. The chemistry of vesuvianite. Can. Mineral. 1992, 30, 19–48. [Google Scholar]
- Fitzgerald, S.; Leavens, P.B.; Rossman, G.R.; Yap, G.P.A.; Rose, T. Vesuvianite from Pajsberg, Sweden, and the role of Be in the vesuvianite structure. Can. Mineral. 2016, 54, 1525–1537. [Google Scholar] [CrossRef]
- Czaja, M.; Lisiecki, R.; Chrobak, A.; Mazurak, Z.; Fronc, K.; Kądziołka-Gaweł, M. The absorption- and luminescence spectra of Mn3+ in beryl and vesuvianite. Phys. Chem. Miner. 2018, 45, 475–488. [Google Scholar] [CrossRef]
- Fitzgerald, S.; Rheingold, A.L.; Leavens, P.B. Crystal structure of a Cu-bearing vesuvianite. Am. Mineral. 1986, 71, 1011–1014. [Google Scholar]
- Ahmed-Said, Y.; Leake, B.E. The conditions of metamorphism of a grossular-wollastonite vesuvianite skarn from the Omey Granite, Connemara, western Ireland, with special inference to the chemistry of vesuvianite. Mineral. Mag. 1996, 60, 541–550. [Google Scholar] [CrossRef]
- Dey, A.; Roy Choudhury, S.; Mukherjee, S.; Sanyal, S.; Sengupta, P. Origin of vesuvianite-garnet veins in calc-silicate rocks from part of the Chotanagpur Granite Gneiss Complex, East Indian Shield: The quantitative P-T-XCO2 topology in parts of the system CaO-MgO-Al2O3-SiO2-H2O-CO2 (+Fe2O3, F). Am. Mineral. 2019, 104, 744–760. [Google Scholar] [CrossRef]
- Ballivián Justiniano, C.A.; Rodríguez, M.G.; Panikorovskii, T.L.; Benítez, M.E.; Recio, C.; Ramos, C.P.; Lanfranchini, M.E.; Di Salvo, F. Vesuvianite as a key tool for the reconstruction of skarn formation conditions: An example from the Sauce Chico Complex, Argentina. Am. Mineral. 2025. [Google Scholar] [CrossRef]
- Zhao, Y.; Li, D. Study on vesuvianite in Qinjia copper-tin mine, Guangxi. Adv. Geosci. 2023, 13, 1296–1301. [Google Scholar] [CrossRef]
- Zhang, Q.; Zhao, Z.; Wang, Y. Gemological characteristics of ‘Baohecui’ from Hanzhong, Shaanxi Province, China. In Proceedings of the China International Jewelry Academic Exchange Conference, Beijing, China, 13 November 2019; pp. 330–333, (In Chinese with English abstract). [Google Scholar]
- Liu, H. Gemological and Mineralogical Characteristics and Genesis of the Idocrase Jade in Hanzhong, Shaanxi. Master’s Thesis, Guilin University of Technology, Guilin, China, 2022. [Google Scholar]
- Whitney, D.L.; Evans, B.W. Abbreviations for names of rock-forming minerals. Am. Mineral. 2010, 95, 185–187. [Google Scholar] [CrossRef]
- Lafuente, B.; Downs, R.T.; Horkowitz, H.R.; Pufahl, P.K. The RRUFF™ project: An integrated study of the chemistry, crystallography, Raman and infrared spectroscopy of minerals. In Highlights in Mineralogical Sciences; Armbruster, T., Danisi, R.M., Eds.; De Gruyter: Berlin, Germany, 2015; pp. 1–30. [Google Scholar]
- Paluszkiewicz, C.; Żabiński, W. Vibrational spectroscopy as a tool for discrimination of high and low vesuvianite. Vib. Spectrosc. 2004, 35, 77–80. [Google Scholar] [CrossRef]
- Groat, L.A.; Hawthorne, F.C.; Rossman, G.R.; Ercit, T.S. The infrared spectroscopy of vesuvianite in the OH region. Can. Mineral. 1995, 33, 609–626. [Google Scholar]
- Borovikova, E.Y.; Kurazhkovskaya, V.S. Infrared spectra and factor group analysis of vesuvianites in OH region. Vib. Spectrosc. 2005, 39, 95–98. [Google Scholar] [CrossRef]
- Yang, M.; Ye, M.; Han, H.; Ren, G.; Han, L.; Zhang, Z. Near-Infrared spectroscopic study of chlorite minerals. J. Spectrosc. 2018, 2018, 6958260. [Google Scholar] [CrossRef]
- Shirozu, H. Infrared spectra of trioctahedral chlorites in relation to chemical composition. Clay Sci. 1985, 6, 167–176. [Google Scholar]
- Liu, J.; Li, R.; Xie, M.; Xu, Z. Gemmological characteristics of grossular jade as jadeite imitation. J. Gems Gemmol. 2014, 16, 47–50, (In Chinese with English abstract). [Google Scholar]
- Zhang, Z. Gemological Characteristics of Vesuvianite Jade and Hydrogrossular Jade in Liuba, Shaanxi Province. Master’s Thesis, China University of Geosciences, Beijing, China, 2022. [Google Scholar]
- Hainschwang, T.; Notari, F. Specular reflectance infrared spectroscopy—A review and update of a little exploited method for gem identification. J. Gemmol. 2008, 17, 23–29. [Google Scholar] [CrossRef]
- Izzo, F.; Germinario, C.; Grifa, C.; Langella, A.; Mercurio, M. External reflectance FTIR dataset (4000–400 cm−1) for the identification of relevant mineralogical phases forming Cultural Heritage materials. Infrared Phys. Technol. 2020, 106, 103266. [Google Scholar] [CrossRef]
- Galuskin, E.; Janeczek, J.; Kozanecki, M.; Sitarz, M.; Jastrzębski, W.; Wrzalik, R.; Stadnicka, K. Single-crystal Raman investigation of vesuvianite in the OH region. Vib. Spectrosc. 2007, 44, 36–41. [Google Scholar] [CrossRef]
- Peng, M.; Mao, H.K.; Dien, L.; Chao, E.C.T. Raman spectroscopy of garnet-group minerals. Chin. J. Geochem. 1994, 13, 176–183. [Google Scholar] [CrossRef]
- Gillet, P.; Fiquet, G.; Malezieux, J.M.; Geiger, C.A. High-pressure and high-temperature Raman spectroscopy of end-member garnets: Pyrope, grossular and andradite. Eur. J. Mineral. 1992, 4, 651–664. [Google Scholar] [CrossRef]
- Prieto, A.C.; Dubessy, J.; Cathelineau, M. Structure-composition relationships in trioctahedral chlorites: A vibrational spectroscopy study. Clays Clay Miner. 1991, 39, 531–539. [Google Scholar] [CrossRef]
- Wang, A.; Freeman, J.J.; Jolliff, B.L. Understanding the Raman spectral features of phyllosilicates. J. Raman Spectrosc. 2015, 46, 829–845. [Google Scholar] [CrossRef]
- Akbulut, M. Raman spectral features of a natural chromitite from NW Türkiye: Characterization of magnesiochromite spectra and implications. Period. Mineral. 2025, 94, 21–36. [Google Scholar] [CrossRef]
- Chopelas, A. Single crystal Raman spectrum of uvarovite, Ca3Cr2Si3O12. Phys. Chem. Miner. 2005, 32, 525–530. [Google Scholar] [CrossRef]
- Bersani, D.; Andò, S.; Vignola, P.; Moltifiori, G.; Marino, I.-G.; Lottici, P.P.; Diella, V. Micro-Raman spectroscopy as a routine tool for garnet analysis. Spectrochim. Acta A Mol. Biomol. Spectrosc. 2009, 73, 484–491. [Google Scholar] [CrossRef] [PubMed]
- Chukanov, N.V.; Vigasina, M.F. Vibrational (Infrared and Raman) Spectra of Minerals and Related Compounds. In Springer Mineralogy; Springer: Cham, Switzerland, 2020; Chapter 4; pp. 741–1255. [Google Scholar] [CrossRef]
- Mattson, S.; Rossman, G. Identifying characteristics of charge transfer transitions in minerals. Phys. Chem. Miner. 1987, 14, 94–99. [Google Scholar] [CrossRef]
- Burns, R.G. Mineralogical Applications of Crystal Field Theory; Cambridge University Press: Cambridge, UK, 1993; pp. 222–227. [Google Scholar] [CrossRef]
- Evans, H.; Rossman, G. Fe2+/Fe3+ intervalence charge transfer and enhanced d-d absorption in mixed valence iron minerals at elevated temperatures. Am. Mineral. 2025. [Google Scholar] [CrossRef]
- Hålenius, U. Optical absorption spectra of Cr-bearing vesuvianite: Detection and measurement of Cr3+ at the octahedral Y3-site. Period. Mineral. 2000, 69, 35–48. [Google Scholar]
- Fritsch, E.; Rossman, G. An update on color in gems. Part I: Introduction and colors caused by dispersed metal ions. Gems Gemol. 1986, 23, 126–139. [Google Scholar] [CrossRef]
- Matsyuk, S.; Platonov, A.; Sobolev, N. Influence of crystallochemical factors on the optical spectra and color of chromium-containing garnets. J. Appl. Spectrosc. 1994, 60, 114–123. [Google Scholar] [CrossRef]
- Lachheb, R.; Herrmann, A.; Damak, K.; Rüssel, C.; Maâlej, R. Optical absorption and photoluminescence properties of chromium in different host glasses. J. Lumin. 2017, 186, 152–157. [Google Scholar] [CrossRef]



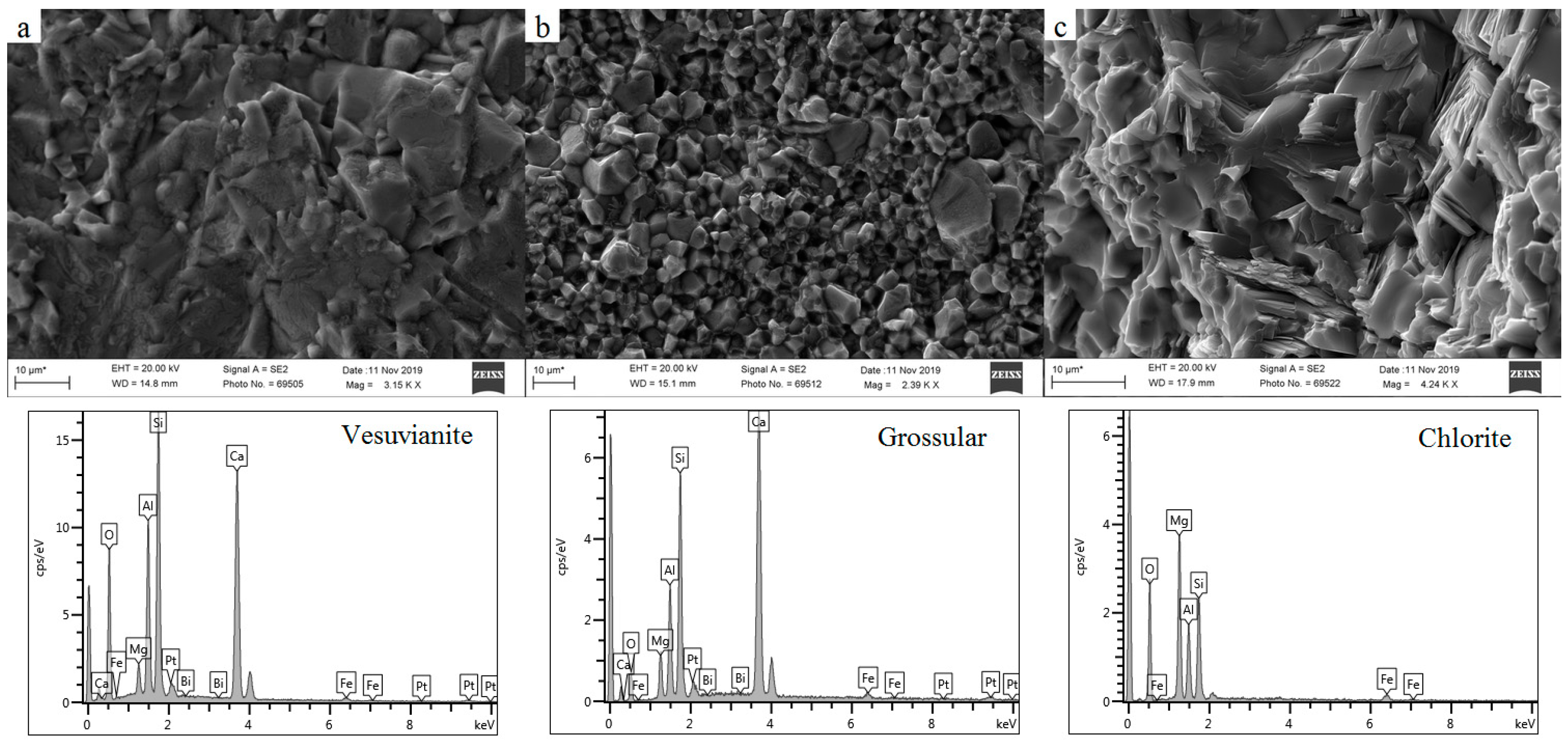
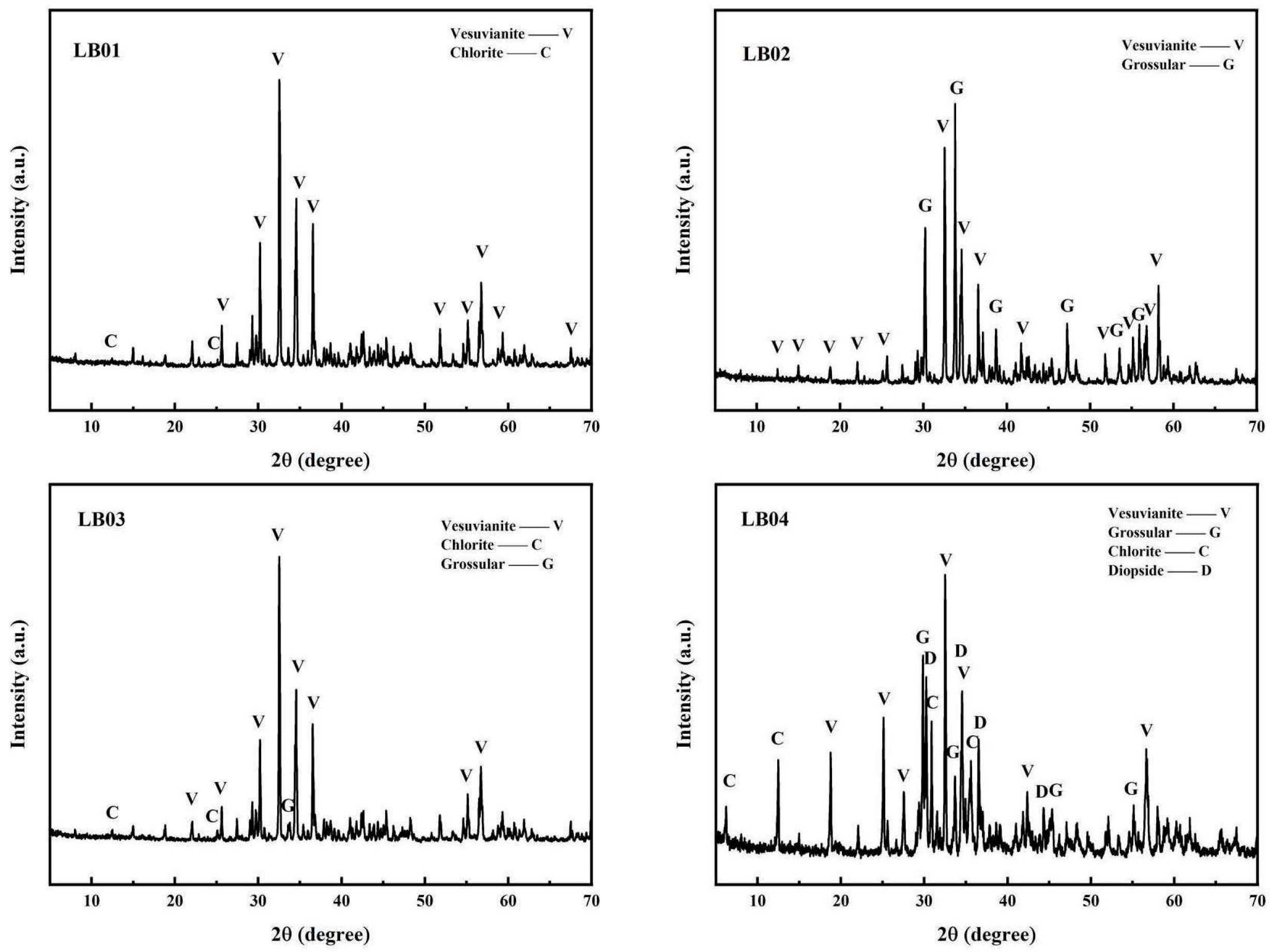
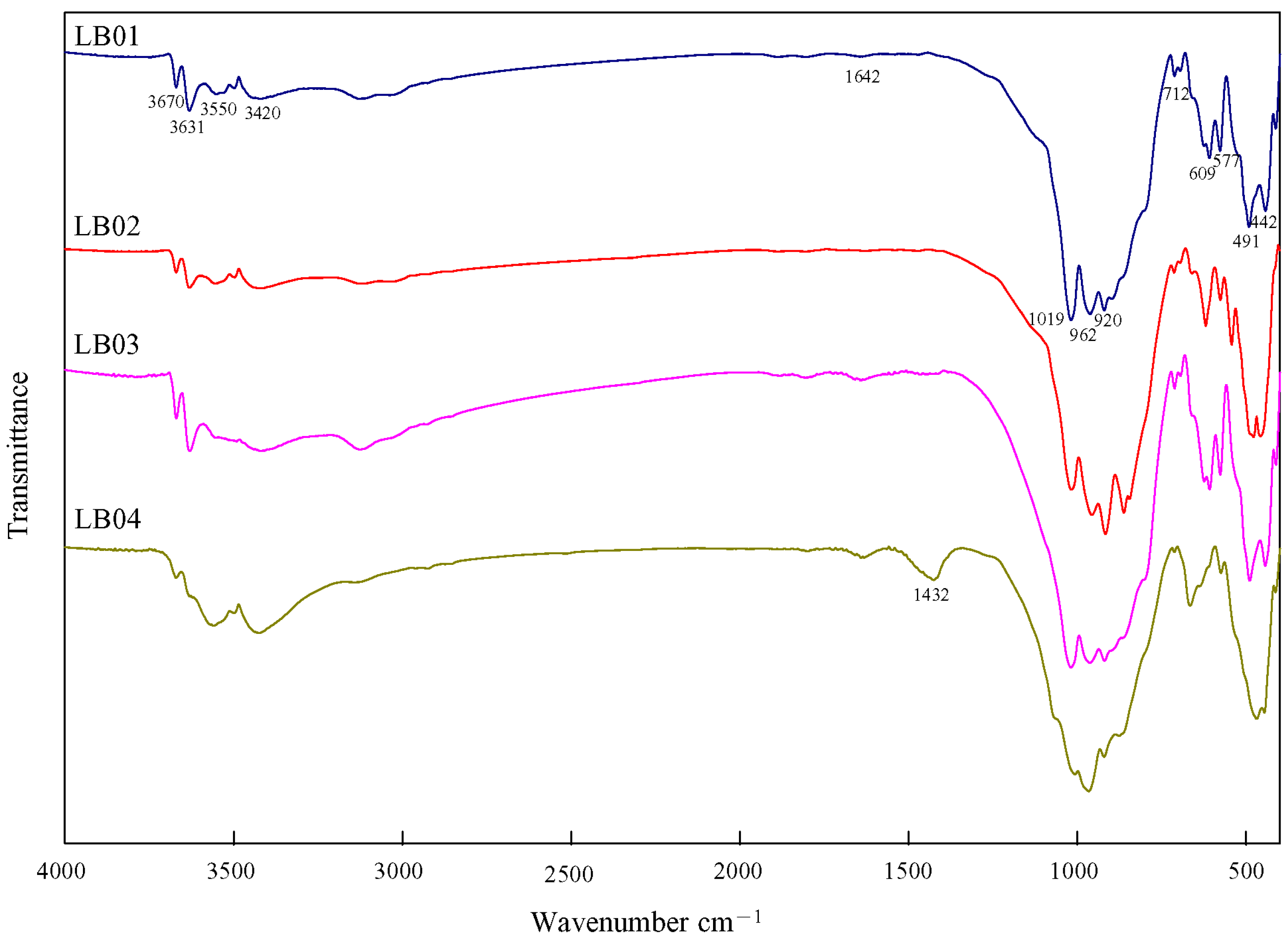
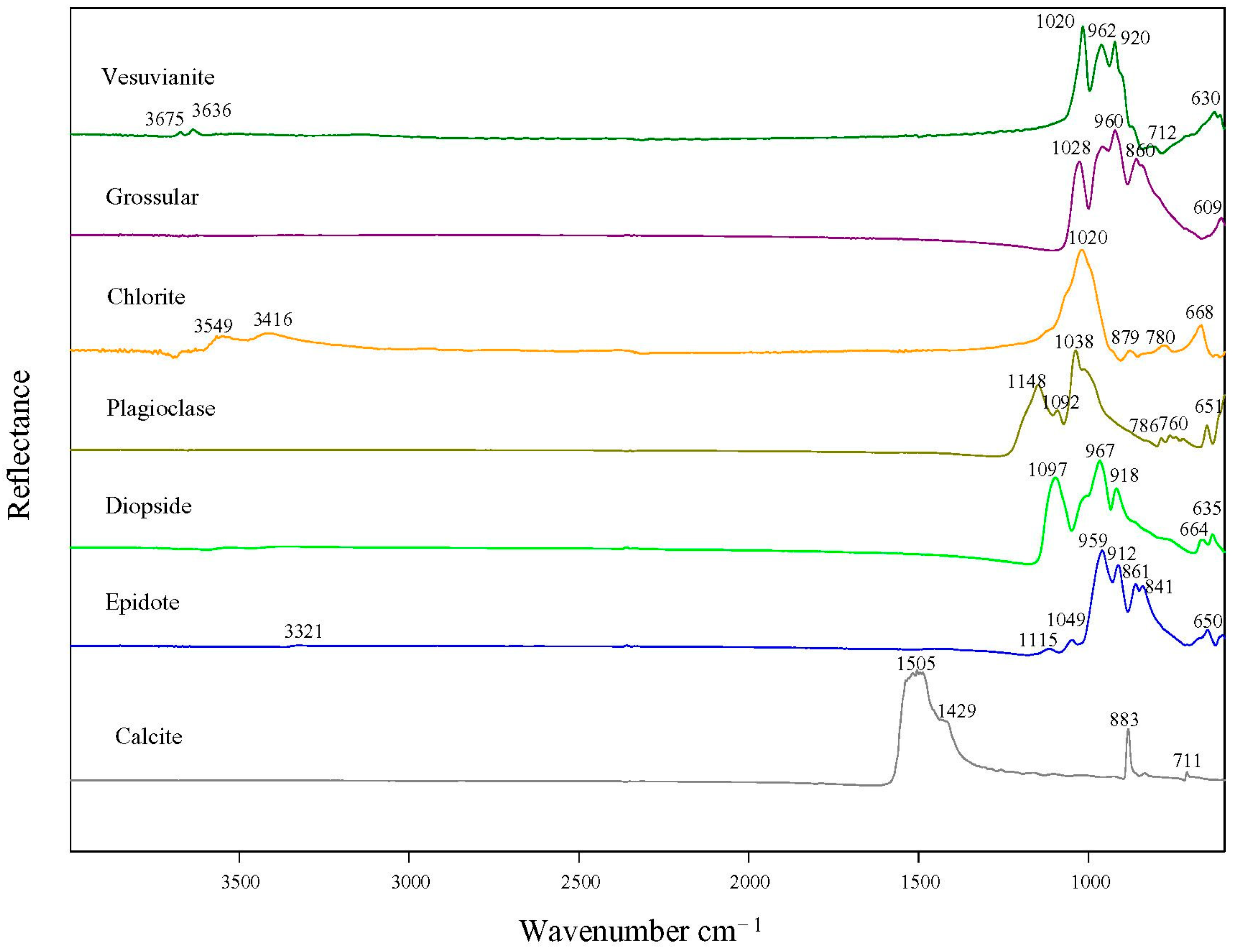
| Sample Number | Microscopic Characteristics | Refractive Index | Specific Gravity |
|---|---|---|---|
| LB01 | Predominantly yellowish-green with localized vivid green patches | 1.71 | 3.33 |
| LB02 | Predominantly yellowish-green with localized pale green patches | 1.71 | 3.31 |
| LB03 | Predominantly yellowish-green with localized pale gray patches | 1.71 | 3.30 |
| LB04 | Predominantly light green exhibiting distinct granular texture | 1.70 | 3.29 |
| LB05 | Predominantly yellowish-green with localized vivid green patches | 1.71 | 3.35 |
| LB06 | Intergrowth of white and yellowish-green zones; dark brown patches and dark green bands present | 1.72 | 3.47 |
| Sample Number | SiO2 | Al2O3 | CaO | MgO | TFeO | Cr2O3 | Analysis Region |
|---|---|---|---|---|---|---|---|
| LB01-1 | 37.89 | 20.59 | 37.72 | 2.87 | 0.72 | / | yellowish-green matrix |
| LB01-2 | 37.10 | 19.25 | 39.34 | 2.74 | 0.90 | 0.14 | vivid green patch |
| LB02-1 | 38.54 | 22.84 | 37.16 | 0.76 | 0.56 | / | yellowish-green matrix |
| LB02-2 | 37.06 | 21.82 | 38.25 | 1.79 | 0.98 | / | green matrix |
| LB02-3 | 37.53 | 20.02 | 35.44 | 5.08 | 1.30 | 0.13 | vivid green patch |
| LB02-4 | 34.93 | 21.48 | 22.54 | 18.96 | 1.06 | 0.32 | pale green patch |
| LB03-1 | 37.89 | 19.61 | 37.88 | 3.29 | 1.23 | / | yellowish-green matrix |
| LB03-2 | 37.98 | 19.09 | 37.92 | 3.16 | 1.76 | / | green matrix |
| LB04-1 | 38.04 | 18.48 | 37.45 | 3.86 | 2.09 | / | yellowish-green matrix |
| LB04-2 | 37.83 | 18.76 | 36.81 | 4.39 | 2.13 | / | green matrix |
| LB05-1 | 37.14 | 18.93 | 40.04 | 2.93 | 0.91 | / | yellowish-green matrix |
| LB05-2 | 37.01 | 19.68 | 39.31 | 2.79 | 1.07 | / | green matrix |
| LB05-3 | 37.04 | 18.64 | 40.11 | 2.79 | 0.96 | 0.03 | vivid green patch |
| LB06-1 | 37.94 | 21.97 | 36.20 | 2.55 | 1.06 | / | yellowish-green matrix |
| LB06-2 | 37.27 | 19.63 | 38.91 | 2.39 | 1.63 | / | green matrix |
| LB06-3 | 30.23 | 31.15 | 0.09 | 35.04 | 3.13 | 0.03 | dark green band |
| LB06-4 | 27.45 | 21.54 | 12.80 | 14.11 | 6.52 | 16.67 | deep brown patch |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yuan, Y.; Shi, M.; Jia, R.; Huang, X.; Zhang, Y. Mineralogical Characteristics and Color Genesis of Vesuvianite Jade from Hanzhong, Shaanxi Province, China. Crystals 2025, 15, 765. https://doi.org/10.3390/cryst15090765
Yuan Y, Shi M, Jia R, Huang X, Zhang Y. Mineralogical Characteristics and Color Genesis of Vesuvianite Jade from Hanzhong, Shaanxi Province, China. Crystals. 2025; 15(9):765. https://doi.org/10.3390/cryst15090765
Chicago/Turabian StyleYuan, Ye, Miao Shi, Ru Jia, Xuren Huang, and Yi Zhang. 2025. "Mineralogical Characteristics and Color Genesis of Vesuvianite Jade from Hanzhong, Shaanxi Province, China" Crystals 15, no. 9: 765. https://doi.org/10.3390/cryst15090765
APA StyleYuan, Y., Shi, M., Jia, R., Huang, X., & Zhang, Y. (2025). Mineralogical Characteristics and Color Genesis of Vesuvianite Jade from Hanzhong, Shaanxi Province, China. Crystals, 15(9), 765. https://doi.org/10.3390/cryst15090765







