Multi-Modal Laser-Fabricated Nanocomposites with Non-Invasive Tracking Modality and Tuned Plasmonic Properties
Abstract
:1. Introduction
2. Materials and Methods
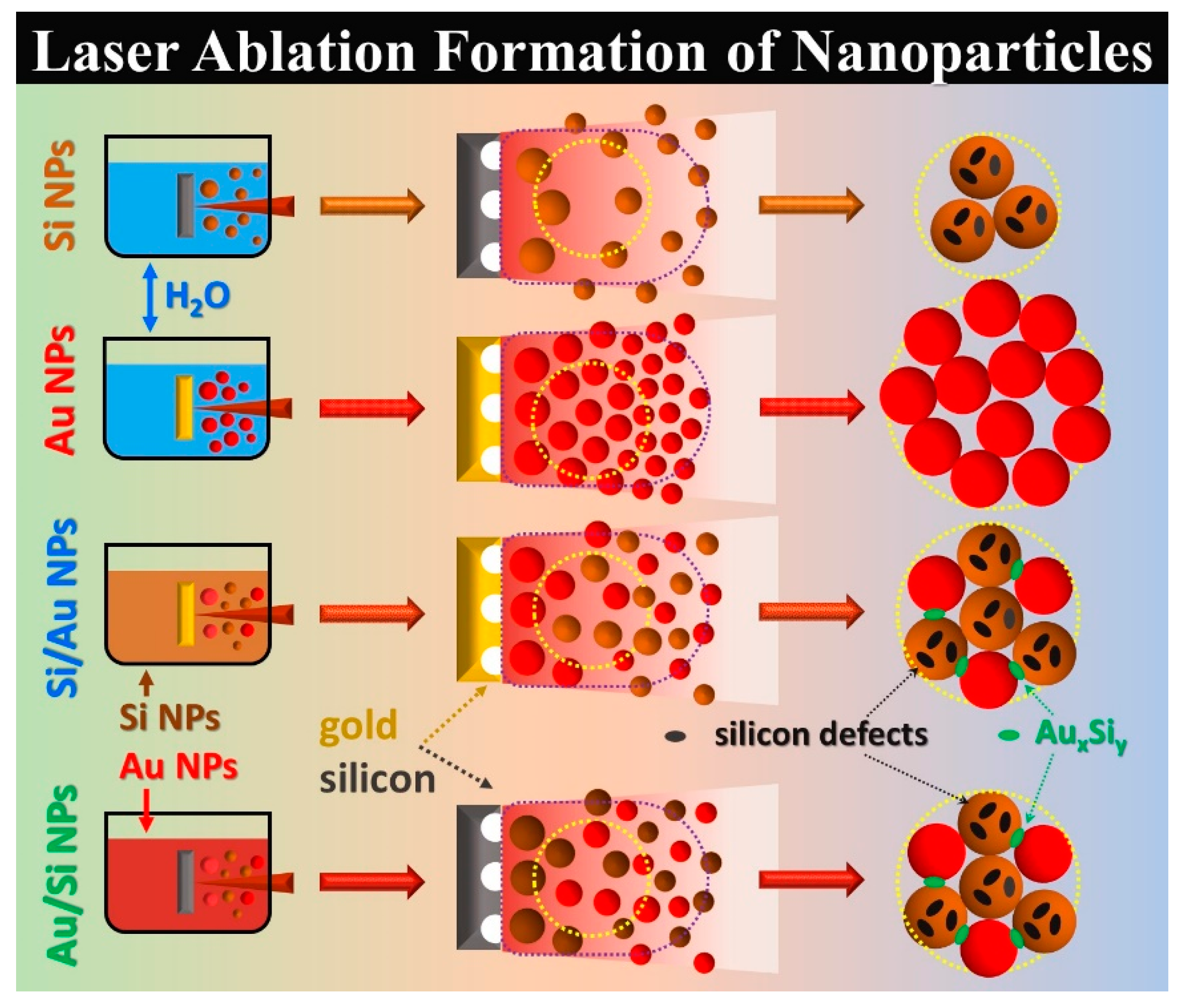
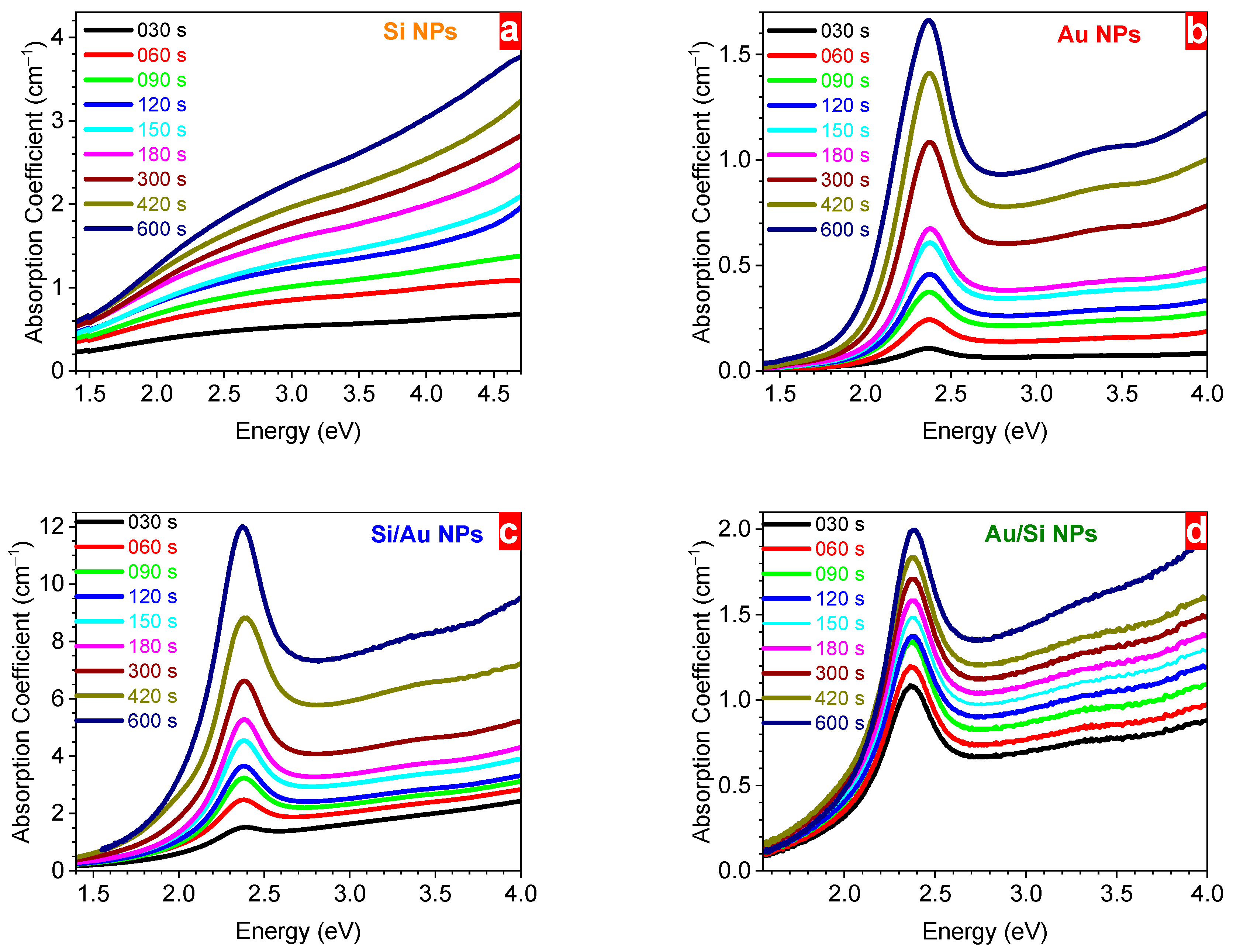
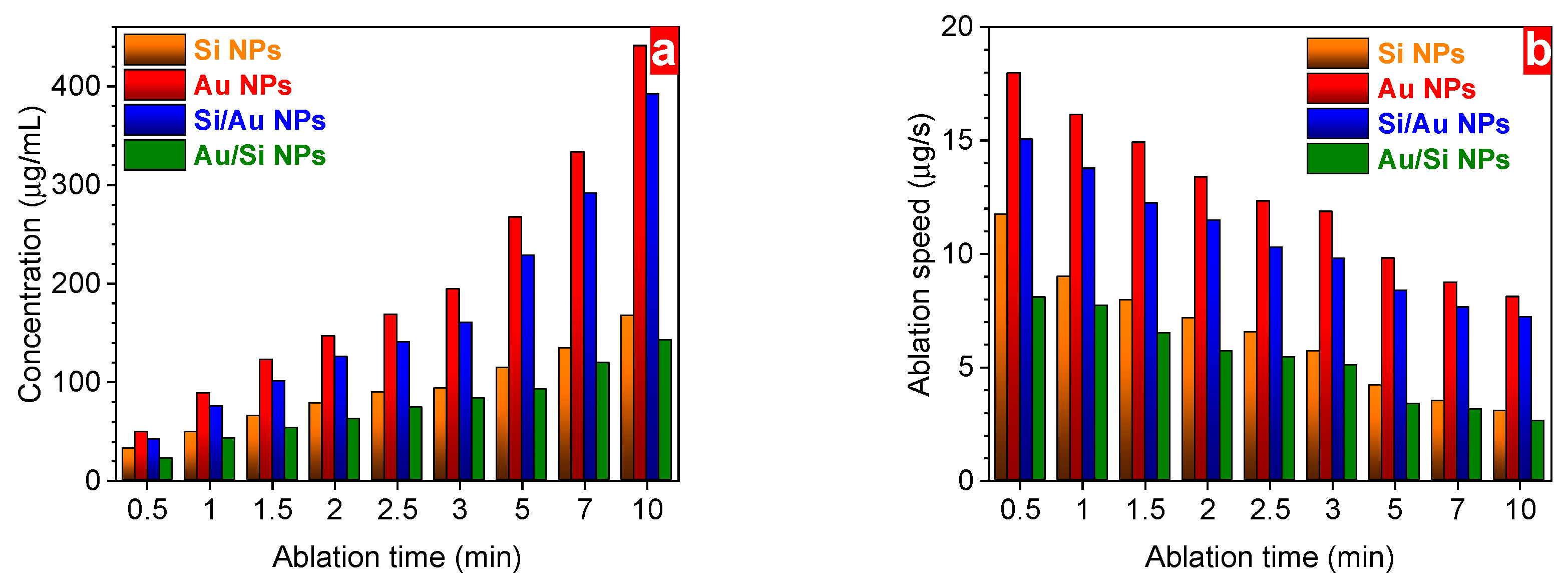
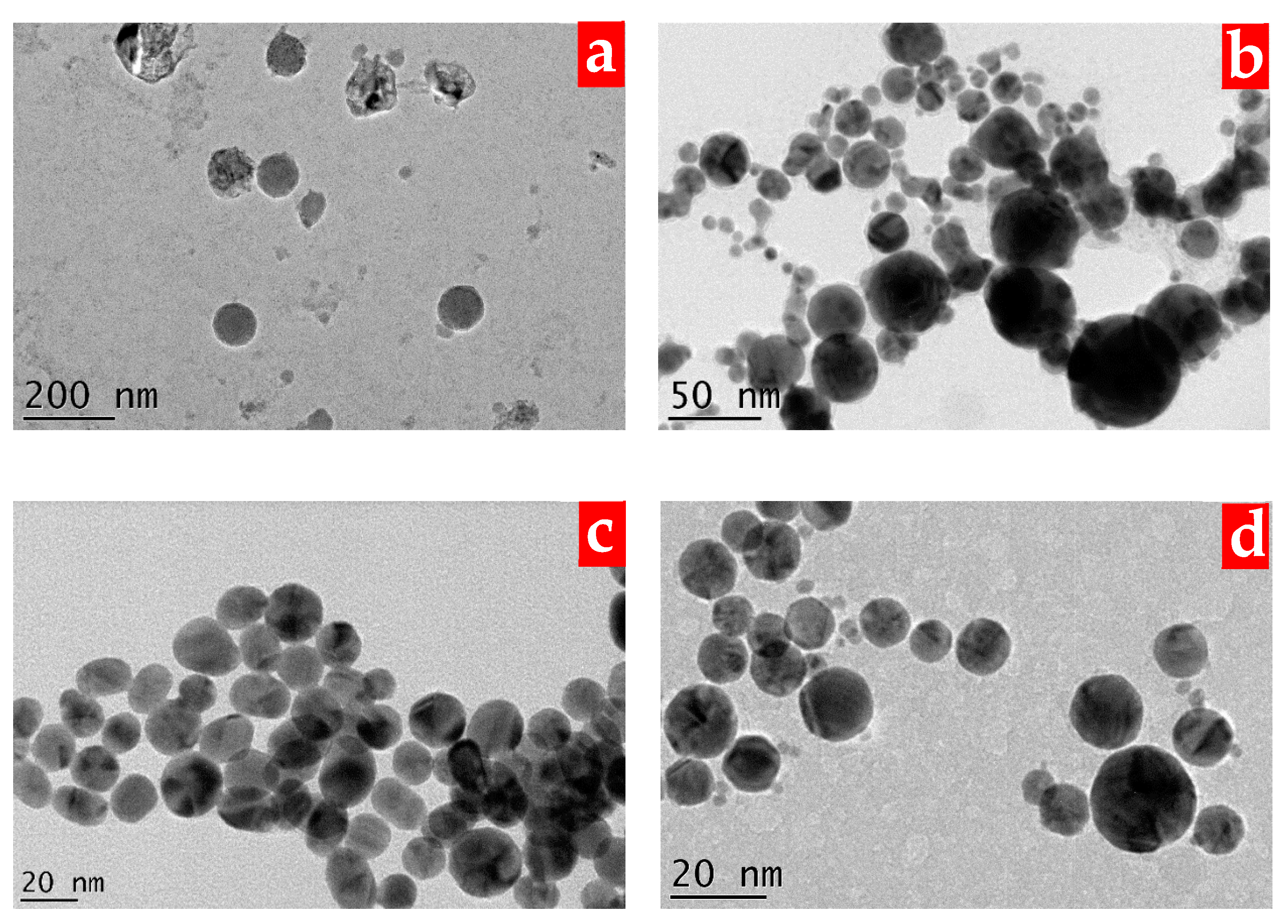
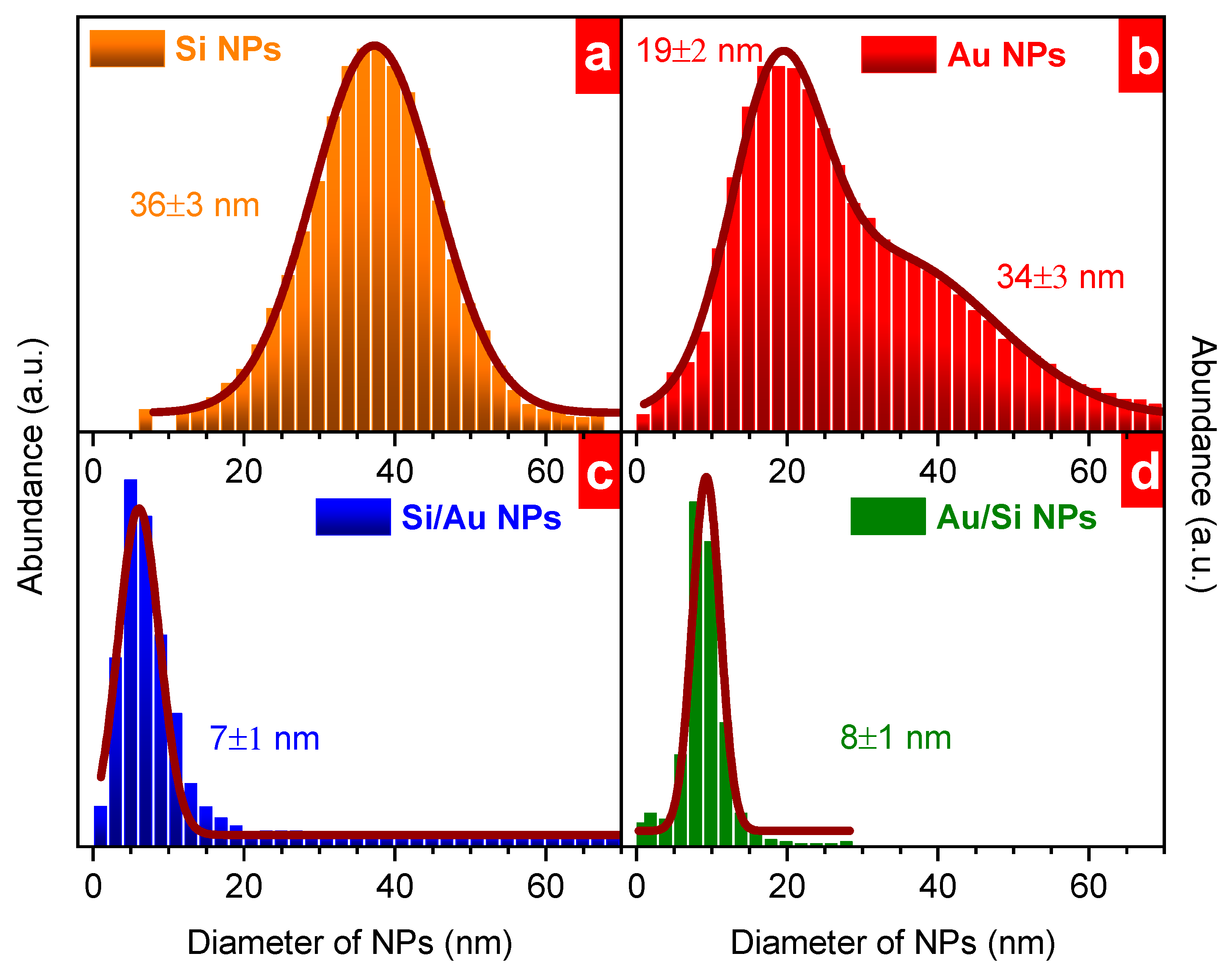
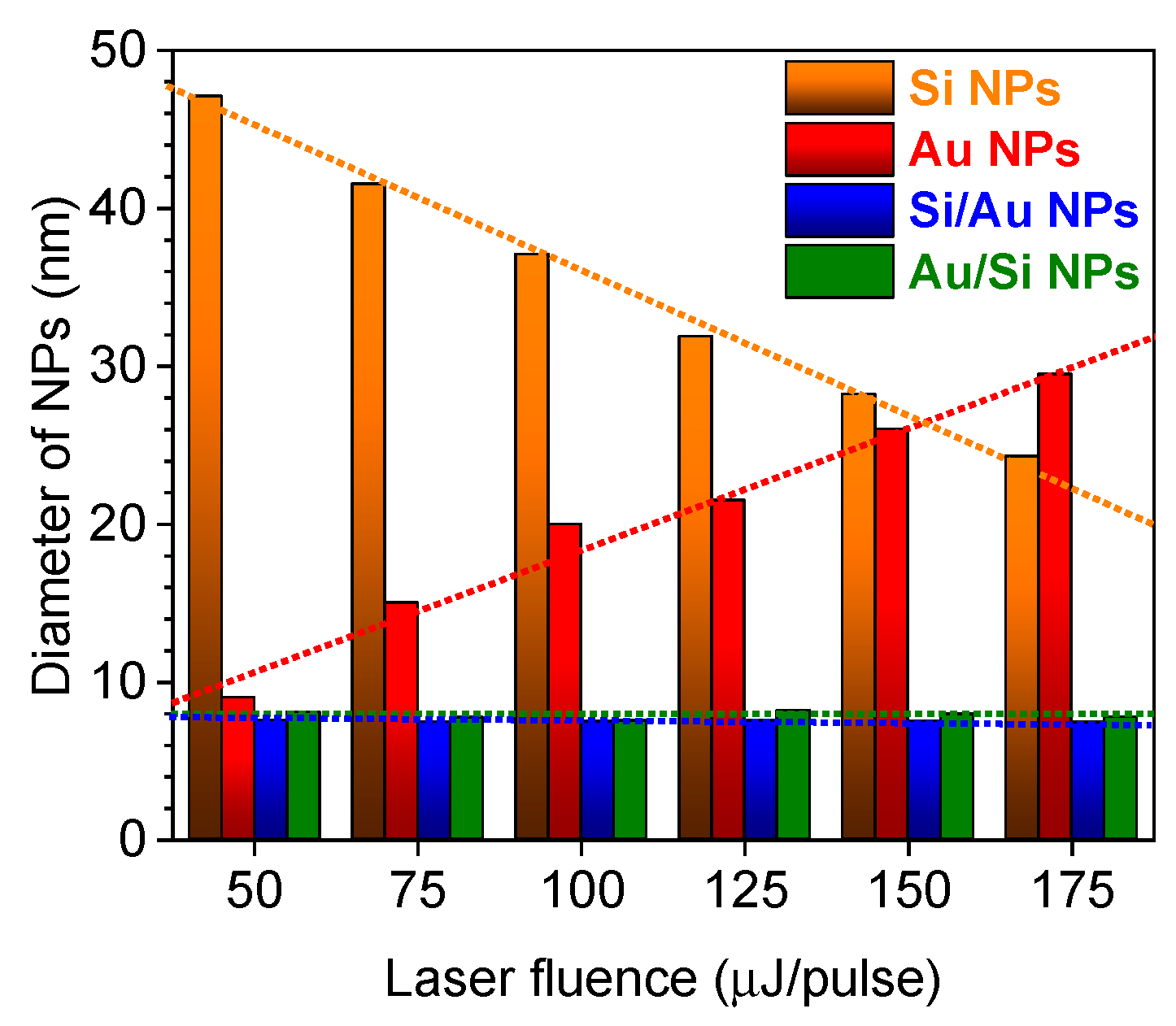
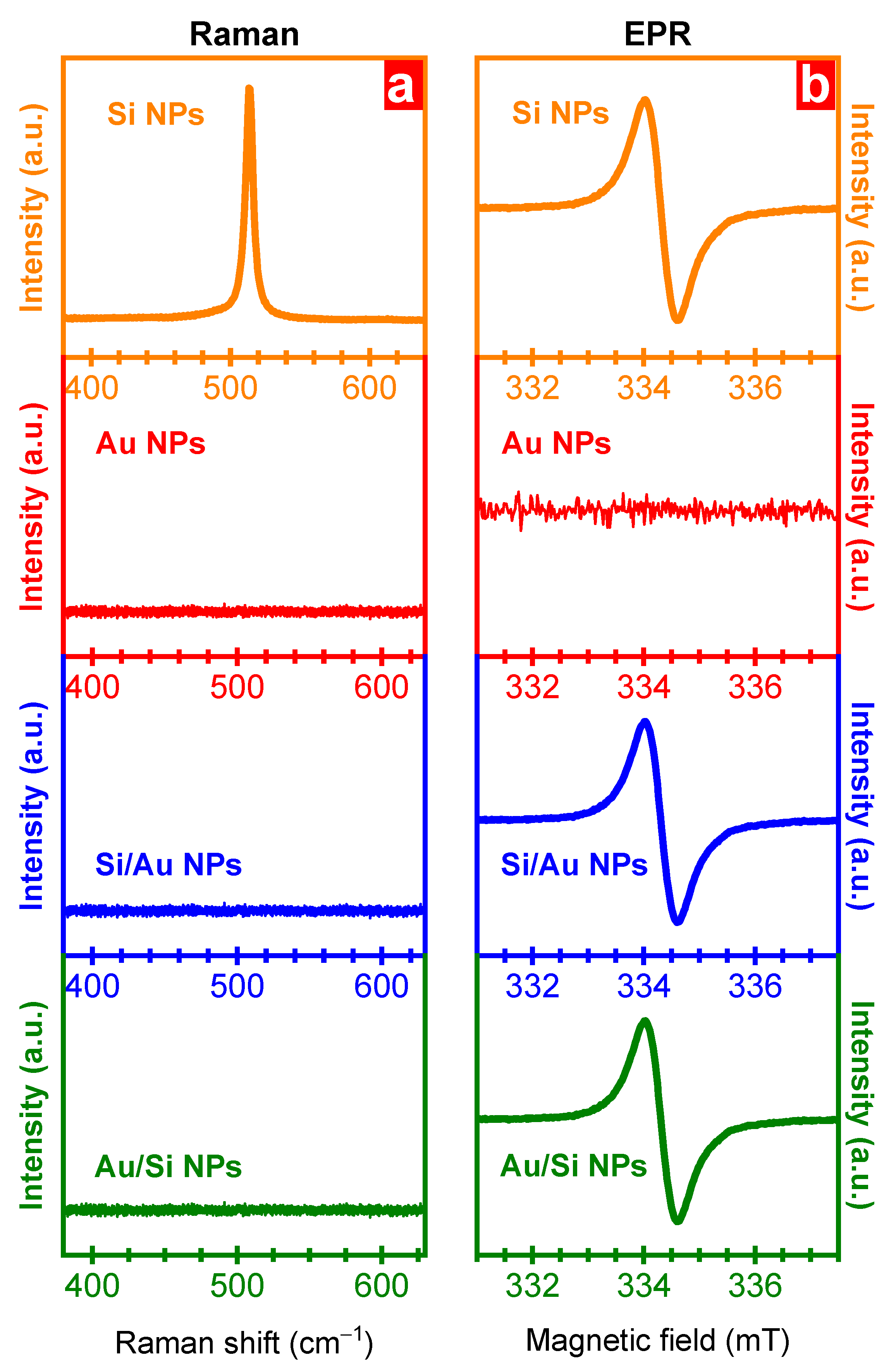
3. Results and Discussion
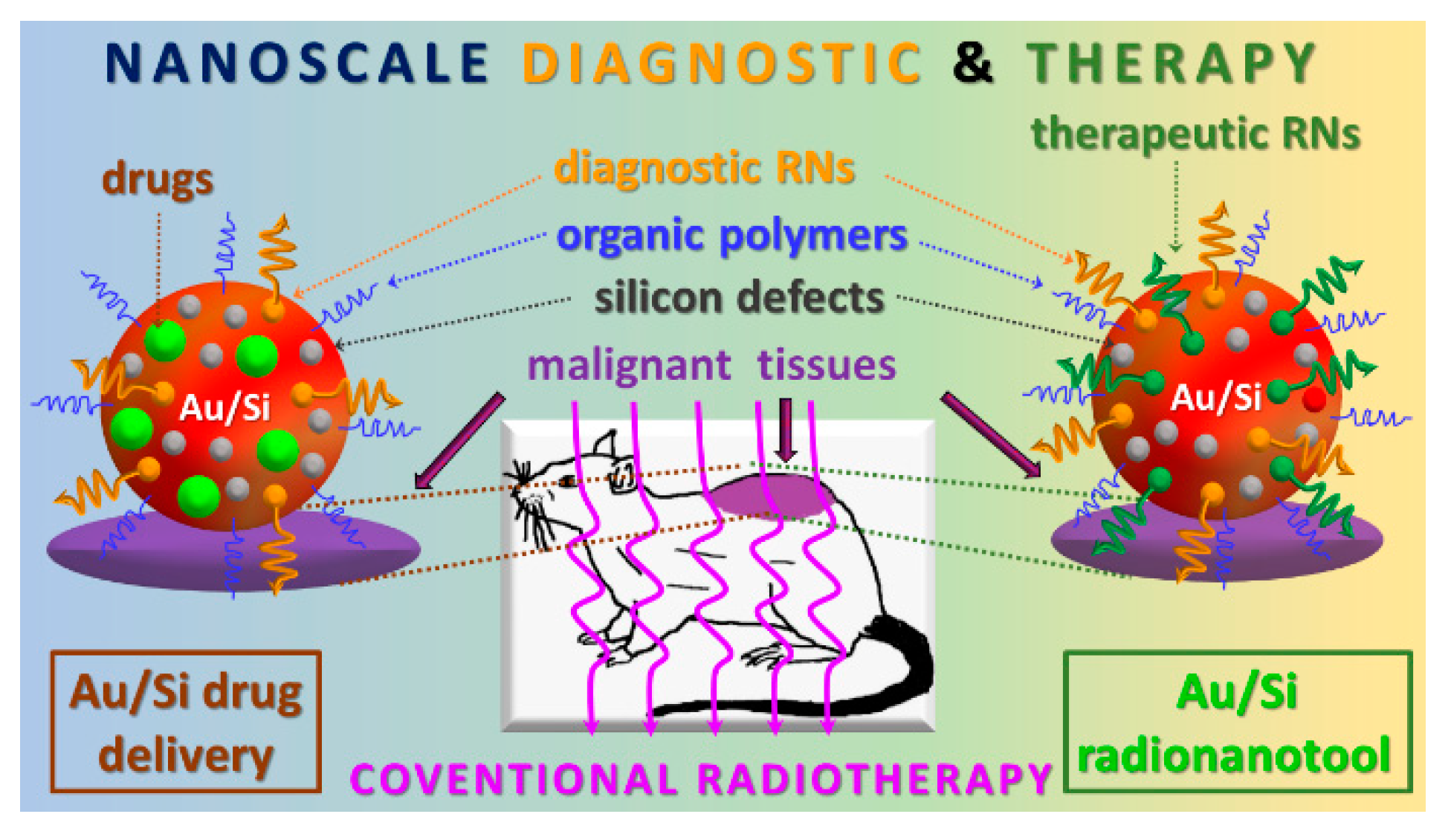
4. Conclusions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Peng, K.-Q.; Wang, X.; Wu, X.-L.; Lee, S.-T. Platinum Nanoparticle Decorated Silicon Nanowires for Efficient Solar Energy Conversion. Nano Lett. 2009, 9, 3704–3709. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.; Yu, Z.; Burkhard, G.F.; Hsu, C.-M.; Connor, S.T.; Xu, Y.; Wang, Q.; McGehee, M.; Fan, S.; Cui, Y. Optical Absorption Enhancement in Amorphous Silicon Nanowire and Nanocone Arrays. Nano Lett. 2009, 9, 279–282. [Google Scholar] [CrossRef] [PubMed]
- Pathak, N.K.; Chander, N.; Komarala, V.K.; Sharma, R.P. Plasmonic Perovskite Solar Cells Utilizing Au@SiO2 Core-Shell Nanoparticles. Plasmonics 2017, 12, 237–244. [Google Scholar] [CrossRef]
- Ryabchikov, Y.V.; Lysenko, V.; Nychyporuk, T. Enhanced Thermal Sensitivity of Silicon Nanoparticles Embedded in (nano-Ag)/SiNx for Luminescent Thermometry. J. Phys. Chem. C 2014, 118, 12515–12519. [Google Scholar] [CrossRef]
- Ryabchikov, Y.V.; Alekseev, S.A.; Lysenko, V.; Bremond, G.; Bluet, J.-M. Photoluminescence thermometry with alkyl-terminated silicon nanoparticles dispersed in low-polar liquids. Phys. Status Solidi-R 2013, 7, 414–417. [Google Scholar] [CrossRef]
- Ryabchikov, Y.V.; Alekseev, S.A.; Lysenko, V.; Bremond, G.; Bluet, J.-M. Photoluminescence of silicon nanoparticles chemically modified by alkyl groups and dispersed in low-polar liquids. J. Nanopart. Res. 2013, 15, 1535. [Google Scholar] [CrossRef]
- Cui, Y.; Lieber, C.M. Functional Nanoscale Electronic Devices Assembled Using Silicon Nanowire Building Blocks. Science 2001, 2915, 851–853. [Google Scholar] [CrossRef]
- Chen, L.J. Silicon nanowires: The key building block for future electronic devices. J. Mater. Chem. 2007, 17, 4639–4643. [Google Scholar] [CrossRef]
- Ryabchikov, Y.V.; Belogorokhov, I.A.; Vorontsov, A.S.; Osminkina, L.A.; Timoshenko, V.Y.; Kashkarov, P.K. Dependence of the Singlet Oxygen Photosensitization Efficiency on Morphology of Porous Silicon. Phys. Status Solidi A 2007, 204, 1271–1275. [Google Scholar] [CrossRef]
- Konstantinova, E.A.; Demin, V.A.; Vorontzov, A.S.; Ryabchikov, Y.V.; Belogorokhov, I.A.; Osminkina, L.A.; Forsh, P.A.; Kashkarov, P.K.; Timoshenko, V.Y. Electron Paramagnetic Resonance and Photoluminescence Study of Si Nanocrystals—Photosensitizers of Singlet Oxygen Molecules. J. Non-Cryst. Solids 2006, 352, 1156–1159. [Google Scholar] [CrossRef]
- Ryabchikov, Y.V.; Belogorokhov, I.A.; Gongalskiy, M.B.; Osminkina, L.A.; Timoshenko, V.Y. Photosensitized Generation of Singlet Oxygen in Powders and Aqueous Suspensions of Silicon Nanocrystals. Semiconductors 2011, 45, 1059–1063. [Google Scholar] [CrossRef]
- Tu, C.-C.; Awasthi, K.; Chen, K.-P.; Lin, C.-H.; Hamada, M.; Ohta, N.; Li, Y.-K. Time-gated Imaging on Live Cancer Cells Using Silicon Quantum Dot Nanoparticles with Long-lived Fluorescence. ACS Photonics 2017, 4, 1306–1315. [Google Scholar] [CrossRef]
- Kharin, A.Y.; Lysenko, V.V.; Rogov, A.; Ryabchikov, Y.V.; Geloen, A.; Tishchenko, I.; Marty, O.; Sennikov, P.G.; Kornev, R.A.; Zavestovskaya, I.N.; et al. Bi-modal nonlinear optical contrast from Si nanoparticles for cancer theranostics. Adv. Opt. Mater. 2019, 7, 1801728. [Google Scholar] [CrossRef]
- Tollemeto, M.; Huang, Z.; Christensen, J.B.; Nielsen, H.M.; Rønholt, S. Mucoadhesive Dendrons Conjugated to Mesoporous Silica Nanoparticles as a Drug Delivery Approach for Orally Administered Biopharmaceuticals. ACS Appl. Mater. Interfaces 2023, 15, 8798–8810. [Google Scholar] [CrossRef] [PubMed]
- Fathy, M.M.; Yassin, F.M.; Elshemey, W.M.; Fahmy, H.M. Insight on the Dependence of the Drug Delivery Applications of Mesoporous Silica Nanoparticles on Their Physical Properties. Silicon 2023, 15, 61–70. [Google Scholar] [CrossRef]
- Ahmed, H.; Gomte, S.S.; Prathyusha, E.; Prabakaran, A.; Agrawal, M.; Alexander, A. Biomedical applications of mesoporous silica nanoparticles as a drug delivery carrier. J. Drug Deliv. Sci. Technol. 2022, 76, 103729. [Google Scholar] [CrossRef]
- Karthikeyan, L.; Sobhana, S.; Yasothamani, V.; Gowsalya, K.; Vivek, R. Multifunctional theranostic nanomedicines for cancer treatment: Recent progress and challenges. Biomed. Eng. Adv. 2023, 5, 100082. [Google Scholar] [CrossRef]
- Jiang, W.; Low, B.Q.L.; Long, R.; Low, J.; Loh, H.; Tang, K.Y.; Chai, C.H.T.; Zhu, H.; Zhu, H.; Li, Z.; et al. Active Site Engineering on Plasmonic Nanostructures for Efficient Photocatalysis. ACS Nano 2023, 17, 4193–4229. [Google Scholar] [CrossRef] [PubMed]
- Verma, P.; Kuwahara, Y.; Mori, K.; Raja, R.; Yamashita, H. New insights in establishing the structure-property relations of novel plasmonic nanostructures for clean energy applications. EnergyChem 2022, 4, 100070. [Google Scholar] [CrossRef]
- Mitra, S.; Mitali, M. Diverse bio-sensing and therapeutic applications of plasmon enhanced nanostructures. Mater. Today 2022, 57, 225–261. [Google Scholar] [CrossRef]
- Nowak-Jary, J.; Machnicka, B. Pharmacokinetics of magnetic iron oxide nanoparticles for medical applications. J. Nanobiotechnol. 2022, 20, 305. [Google Scholar] [CrossRef]
- Naghdi, M.; Ghovvati, M.; Rabiee, N.; Ahmadi, S.; Abbariki, N.; Sojdeh, S.; Ojaghi, A.; Bagherzadeh, M.; Akhavan, O.; Sharifi, E.; et al. Magnetic nanocomposites for biomedical applications. Adv. Colloid Interface Sci. 2022, 308, 102771. [Google Scholar] [CrossRef]
- Laha, S.S.; Thorat, N.D.; Singh, G.; Sathish, C.I.; Yi, J.; Dixit, A.; Vinu, A. Rare-Earth Doped Iron Oxide Nanostructures for Cancer Theranostics: Magnetic Hyperthermia and Magnetic Resonance Imaging. Small 2022, 18, 2104855. [Google Scholar] [CrossRef]
- Falzarano, M.S.; Flesia, C.; Cavalli, R.; Guiot, C.; Ferlini, A. Nanodiagnostics and Nanodelivery Applications in Genetic Alterations. Curr. Pharm. Des. 2018, 24, 1717–1726. [Google Scholar] [CrossRef]
- Li, J.; Liu, J.; Chen, C. Remote Control and Modulation of Cellular Events by Plasmonic Gold Nanoparticles: Implications and Opportunities for Biomedical Applications. ACS Nano 2017, 11, 2403–2409. [Google Scholar] [CrossRef]
- Iqbal, M.Z.; Ren, W.; Saeed, M.; Chen, T.; Ma, X.; Yu, X.; Zhang, J.; Zhang, L.; Li, A.; Wu, A. A facile fabrication route for binary transition metal oxide-based Janus nanoparticles for cancer theranostic applications. Nano Res. 2018, 11, 5735–5750. [Google Scholar] [CrossRef]
- Ma, J.; Lee, S.M.-Y.; Yi, C.; Li, C.-W. Controllable synthesis of functional nanoparticles by microfluidic platforms for biomedical applications—A review. Lab. Chip 2017, 17, 209–226. [Google Scholar] [CrossRef] [PubMed]
- Ryabchikov, Y.V. Size Modification of Optically Active Contamination-Free Silicon Nanoparticles with Paramagnetic Defects by Their Fast Synthesis and Dissolution. Phys. Status Solidi A 2019, 216, A1800685. [Google Scholar] [CrossRef]
- Kirillin, M.Y.; Sergeeva, E.A.; Agrba, P.D.; Krainov, A.D.; Ezhov, A.A.; Shuleiko, D.V.; Kashkarov, P.K.; Zabotnov, S.V. Laser-ablated silicon nanoparticles: Optical properties and perspectives in optical coherence tomography. Laser Phys. 2015, 25, 075604. [Google Scholar] [CrossRef]
- Radičić, R.; Maletić, D.; Blažeka, D.; Car, J.; Krstulović, N. Synthesis of Silver, Gold, and Platinum Doped Zinc Oxide Nanoparticles by Pulsed Laser Ablation in Water. Nanomaterials 2022, 12, 3484. [Google Scholar] [CrossRef]
- Pini, F.; Pilot, R.; Ischia, G.; Agnoli, S.; Amendola, V. Au–Ag Alloy Nanocorals with Optimal Broadband Absorption for Sunlight-Driven Thermoplasmonic Applications. ACS Appl. Mater. Interfaces 2022, 14, 28924–28935. [Google Scholar] [CrossRef]
- Doñate-Buendia, C.; Spellauge, M.; Streubel, R.; Riahi, F.; Barcikowski, S.; Huber, H.P.; Gökce, B. Double-pulse laser ablation in liquids: Nanoparticle bimodality reduction by sub-nanosecond interpulse delay optimization. J. Phys. D Appl. Phys. 2023, 56, 104001. [Google Scholar] [CrossRef]
- Verma, A.K.; Soni, R.K. Laser ablation synthesis of bimetallic gold-palladium core@shell nanoparticles for trace detection of explosives. Opt. Laser Technol. 2023, 163, 109429. [Google Scholar] [CrossRef]
- Ryabchikov, Y.V. Facile Laser Synthesis of Multimodal Composite Silicon/Gold Nanoparticles with Variable Chemical Composition. J. Nanopart. Res. 2019, 21, 85. [Google Scholar] [CrossRef]
- Ryabchikov, Y.V.; Behrends, J. Expedient Paramagnetic Properties of Surfactant-Free Plasmonic Silicon-Based Nanoparticles. Opt. Quant. Electron. 2020, 52, 177. [Google Scholar] [CrossRef]
- Nasiri, P.; Doranian, D.; Sari, A.H. Synthesis of Au/Si nanocomposite using laser ablation method. Opt. Laser Technol. 2019, 113, 217–224. [Google Scholar] [CrossRef]
- Gurbatov, S.O.; Puzikov, V.; Storozhenko, D.; Modin, E.; Mitsai, E.; Cherepakhin, A.; Shevlyagin, A.; Gerasimenko, A.V.; Kulinich, S.A.; Kuchmizhak, A.A. Multigram-Scale Production of Hybrid Au-Si Nanomaterial by Laser Ablation in Liquid (LAL) for Temperature-Feedback Optical Nanosensing, Light-to-Heat Conversion, and Anticounterfeit Labeling. ACS Appl. Mater. Interfaces 2023, 15, 3336–3347. [Google Scholar] [CrossRef]
- Abbas, Z.A.; Khashan, K.S.; Hussain, Z.T. Silver (Ag) doped cadmium oxide (CdO) nanoparticles were synthesized through laser ablation in liquid for the sensing applications. Opt. Quant. Electron. 2023, 55, 587. [Google Scholar] [CrossRef]
- Kulikov, I.; Panjwani, N.A.; Vereshchagin, A.A.; Spallek, D.; Lukianov, D.A.; Alekseeva, E.V.; Levin, O.V.; Behrends, J. Spins at work: Probing charging and discharging of organic radical batteries by electron paramagnetic resonance spectroscopy. Energy Environ. Sci. 2022, 15, 3275–3290. [Google Scholar] [CrossRef]
- Maximova, K.; Aristov, A.; Sentis, M.; Kabashin, A.V. Size-controllable synthesis of bare gold nanoparticles by femtosecond laser fragmentation in water. Nanotechnology 2015, 26, 065601. [Google Scholar] [CrossRef] [PubMed]
- Flimelova, M.; Ryabchikov, Y.V.; Behrends, J.; Bulgakova, N.M. Environmentally Friendly Improvement of Plasmonic Nanostructure Functionality towards Magnetic Resonance Applications. Nanomaterials 2023, 13, 764. [Google Scholar] [CrossRef] [PubMed]
- Lin, J.-S.; Chen, C.-C.; Diau, W.W.-G.; Liu, T.-F. Fabrication and characterization of eutectic gold–silicon (Au–Si) nanowires. J. Mater. Process. Technol. 2008, 206, 425–430. [Google Scholar] [CrossRef]
- Abderrafi, K.; Calzada, R.G.; Gongalsky, M.B.; Suarez, I.; Abarques, R.; Chirvony, V.S.; Timoshenko, V.Y.; Ibanez, R.; Martínez-Pastor, J.P. Silicon Nanocrystals Produced by Nanosecond Laser Ablation in an Organic Liquid. J. Phys. Chem. C 2011, 115, 5147–5151. [Google Scholar] [CrossRef]
- Shimasaki, M.; Show, Y.; Iwase, M.; Izumi, T.; Ichinohe, T.; Nozaki, S.; Morisaki, H. Correlation between light emission and dangling bonds in porous silicon. Appl. Surf. Sci. 1996, 92, 617–620. [Google Scholar] [CrossRef]
- Pan, S.; Wang, T.; Jin, K.; Cai, X. Understanding and designing metal matrix nanocomposites with high electrical conductivity: A review. J. Mater. Sci. 2022, 57, 6487–6523. [Google Scholar] [CrossRef]
- Yuan, J.; Yao, G.; Pan, S.; Murali, N.; Li, X. Size Control of In Situ Synthesized TiB2 Particles in Molten Aluminum. Metall. Mater. Trans. A 2021, 52, 2657–2666. [Google Scholar] [CrossRef]
- Havelka, O.; Yalcinkaya, F.; Wacławek, S.; Padil, V.V.T.; Amendola, V.; Černíka, M.; Torres-Mendieta, R. Sustainable and scalable development of PVDF-OH Ag/TiOx nanocomposites for simultaneous oil/water separation and pollutant degradation. Environ. Sci. Nano 2023. [Google Scholar] [CrossRef]
- Scaramuzza, S.; Faria, C.M.G.; Coviello, V.; Forrer, D.; Artiglia, L.; Badocco, D.; Pastore, P.; Ghigna, P.; Postuma, I.; Cansolino, L.; et al. A Laser Synthesis Route to Boron-Doped Gold Nanoparticles Designed for X-Ray Radiotherapy and Boron Neutron Capture Therapy Assisted by CT Imaging. Adv. Funct. Mater. 2023, 2303366. [Google Scholar] [CrossRef]
- Chen, F.; Wang, M.; Du, Z.; Pu, Z.; Zhu, B. 31I labeled pH-responsive gold nanoparticles for bimodal tumor diagnosis. Mater. Lett. 2023, 330, 133202. [Google Scholar] [CrossRef]
- Ünak, P.; Yasakçı, V.; Tutun, E.; Karatay, K.B.; Walczak, R.; Wawrowicz, K.; Żelechowska-Matysiak, K.; Majkowska-Pilip, A.; Bilewicz, A. Multimodal Radiobioconjugates of Magnetic Nanoparticles Labeled with 44Sc and 47Sc for Theranostic Application. Pharmaceutics 2023, 15, 850. [Google Scholar] [CrossRef] [PubMed]
- Petriev, V.M.; Tischenko, V.K.; Mikhailovskaya, A.A.; Popov, A.A.; Tselikov, G.; Zelepukin, I.; Deyev, S.M.; Kaprin, A.D.; Ivanov, S.; Timoshenko, V.Y.; et al. Nuclear nanomedicine using Si nanoparticles as safe and effective carriers of 188Re radionuclide for cancer therapy. Sci. Rep.-UK 2019, 9, 2017. [Google Scholar] [CrossRef] [PubMed]
- Türkkanı, G.; Güngör, B.; Cetin, O.; İçhedef, C.; Parlak, Y.; Gümüşer, F.G.; Bilgin, B.E.S.; Teksöz, S. Synthesis, radiolabeling and in vitro evaluation of azathioprine loaded magnetic solid lipid nanoparticles. J. Radioanal. Nucl. Chem. 2023. [Google Scholar] [CrossRef]
- Goel, M.; Mackeyev, Y.; Krishnan, S. Radiolabeled nanomaterial for cancer diagnostics and therapeutics: Principles and concepts. Cancer Nano. 2023, 14, 15. [Google Scholar] [CrossRef] [PubMed]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ryabchikov, Y.V. Multi-Modal Laser-Fabricated Nanocomposites with Non-Invasive Tracking Modality and Tuned Plasmonic Properties. Crystals 2023, 13, 1381. https://doi.org/10.3390/cryst13091381
Ryabchikov YV. Multi-Modal Laser-Fabricated Nanocomposites with Non-Invasive Tracking Modality and Tuned Plasmonic Properties. Crystals. 2023; 13(9):1381. https://doi.org/10.3390/cryst13091381
Chicago/Turabian StyleRyabchikov, Yury V. 2023. "Multi-Modal Laser-Fabricated Nanocomposites with Non-Invasive Tracking Modality and Tuned Plasmonic Properties" Crystals 13, no. 9: 1381. https://doi.org/10.3390/cryst13091381
APA StyleRyabchikov, Y. V. (2023). Multi-Modal Laser-Fabricated Nanocomposites with Non-Invasive Tracking Modality and Tuned Plasmonic Properties. Crystals, 13(9), 1381. https://doi.org/10.3390/cryst13091381







