Crystallographic Evidence of η1-Coordination of Bulky Aminopyridine in Halide-Containing Iron (II) Complexes
Abstract
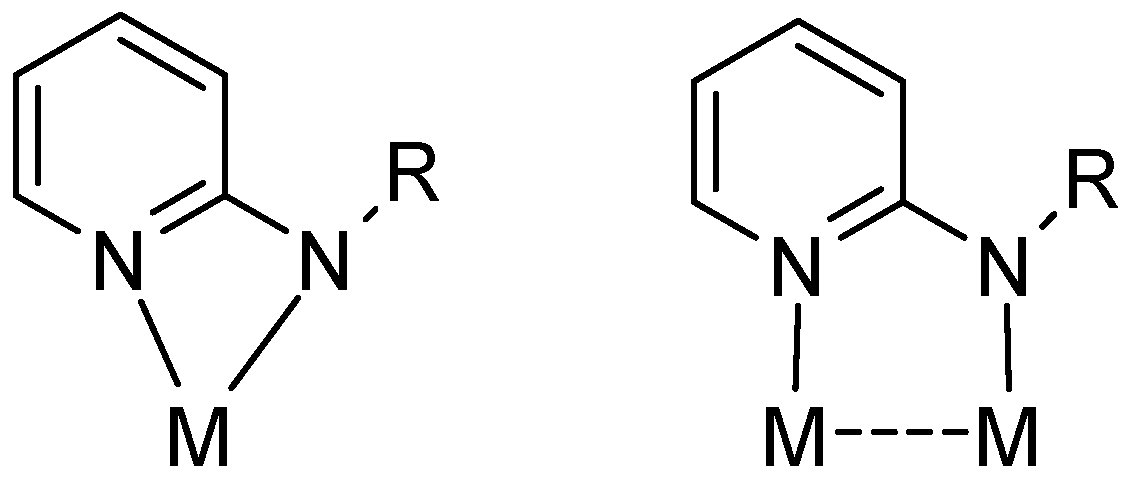
:1. Introduction
2. Materials and Methods
2.1. General Information
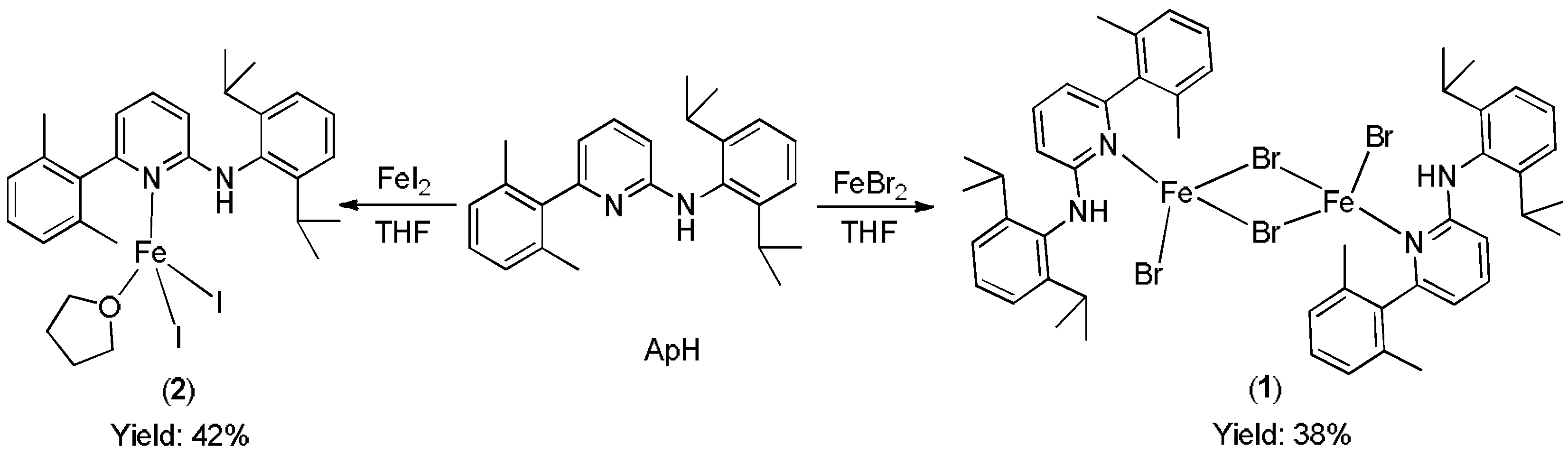
2.2. Syntheses
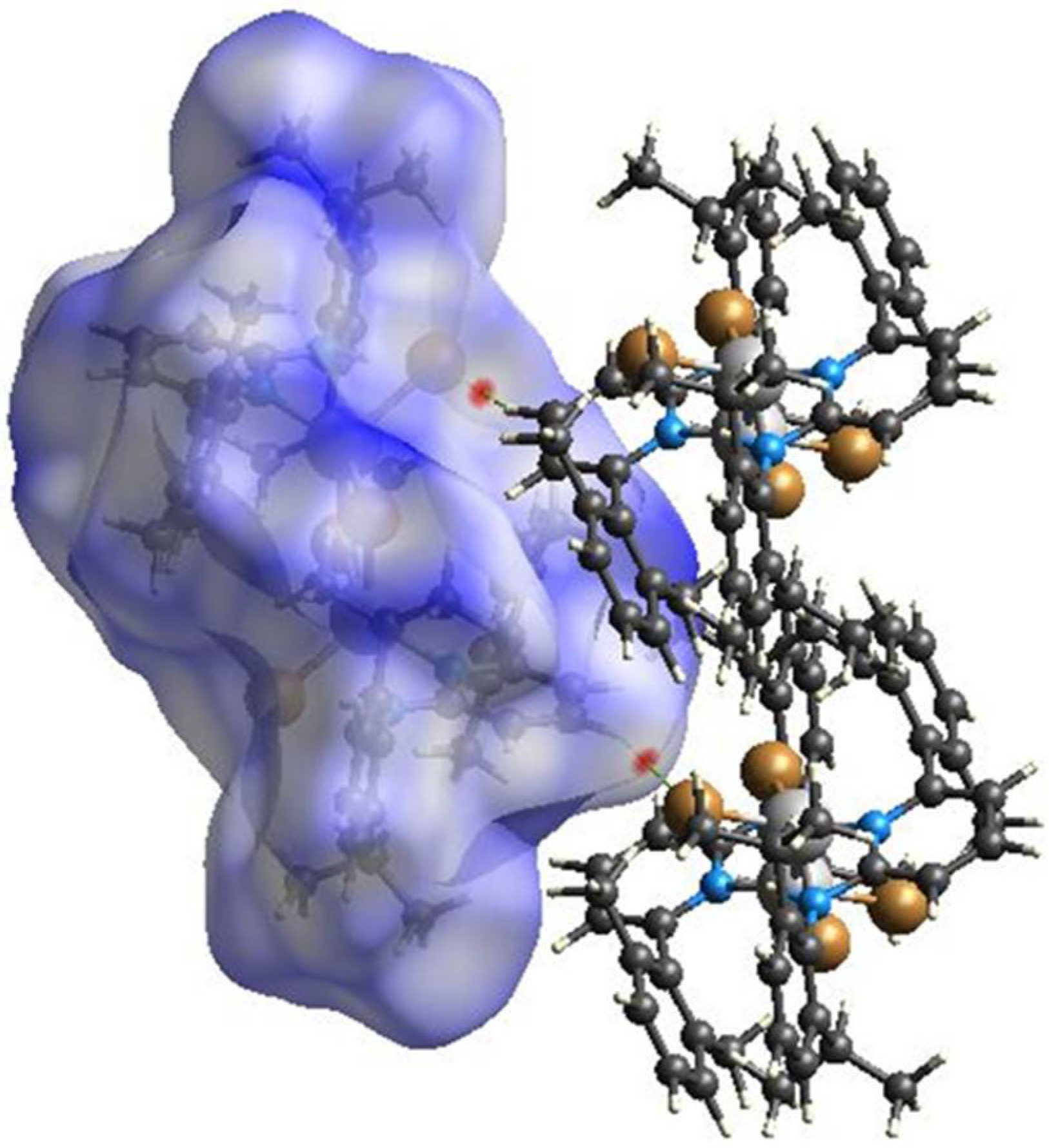
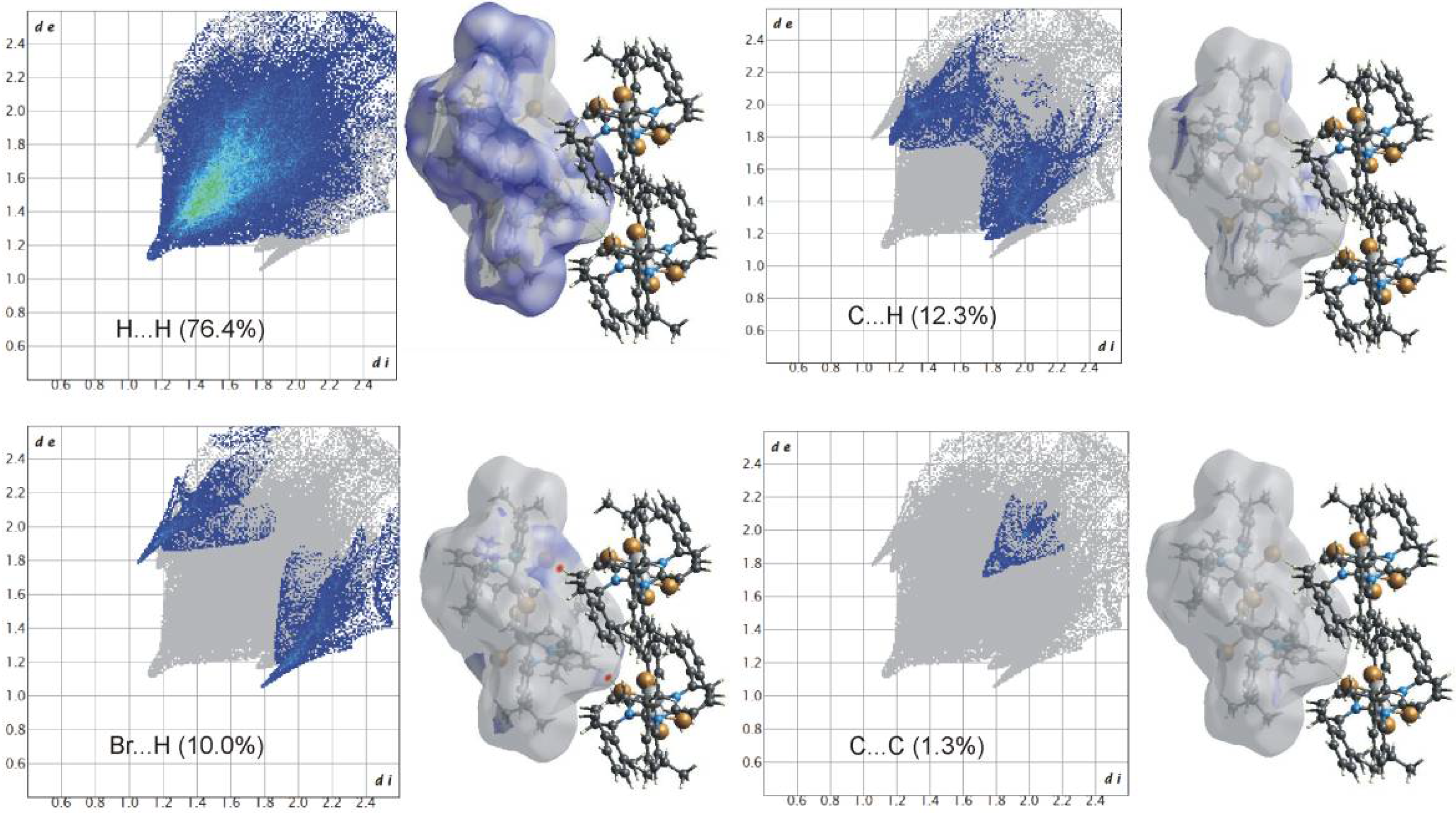
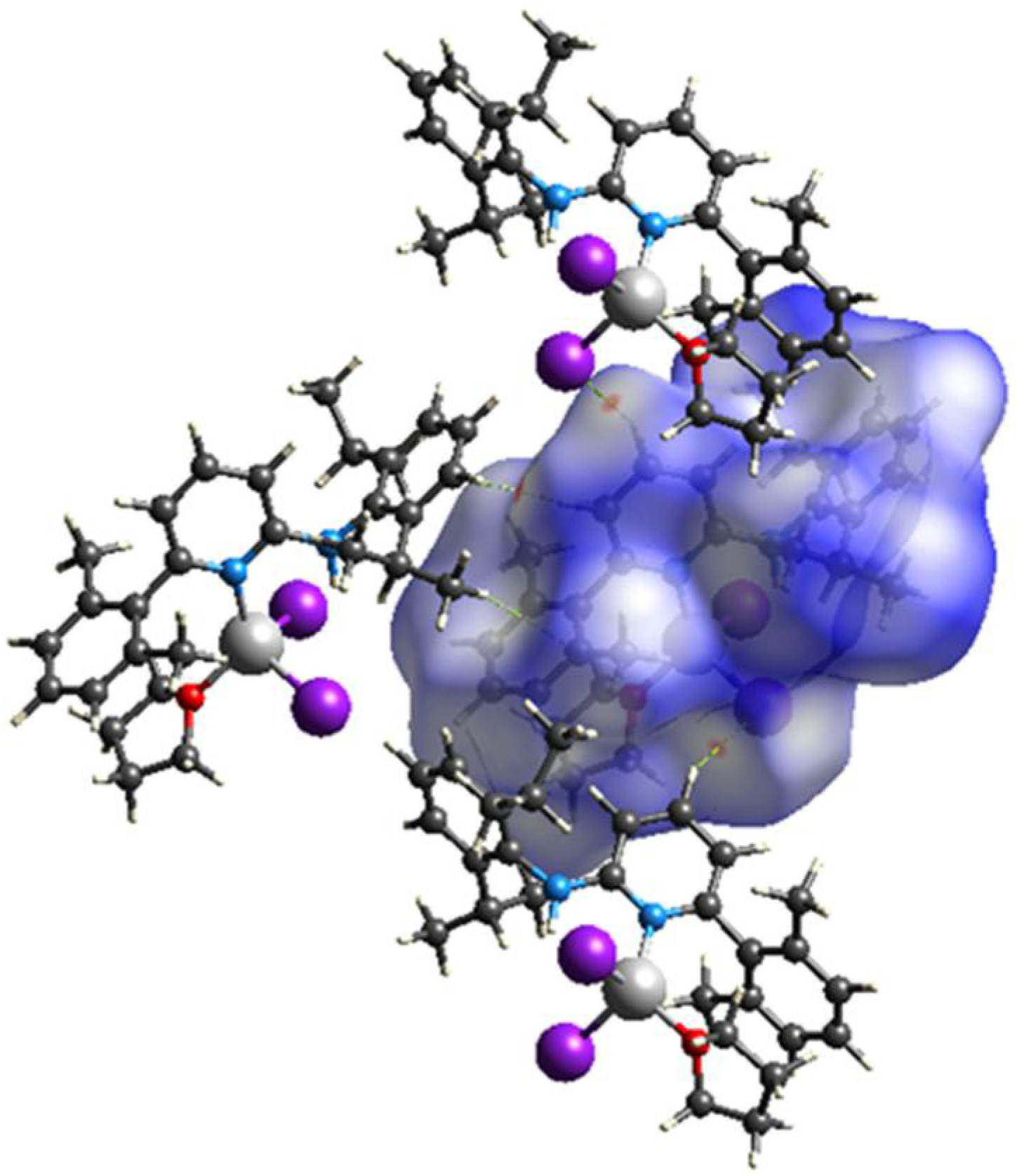
2.3. Hirshfeld Surface Analysis
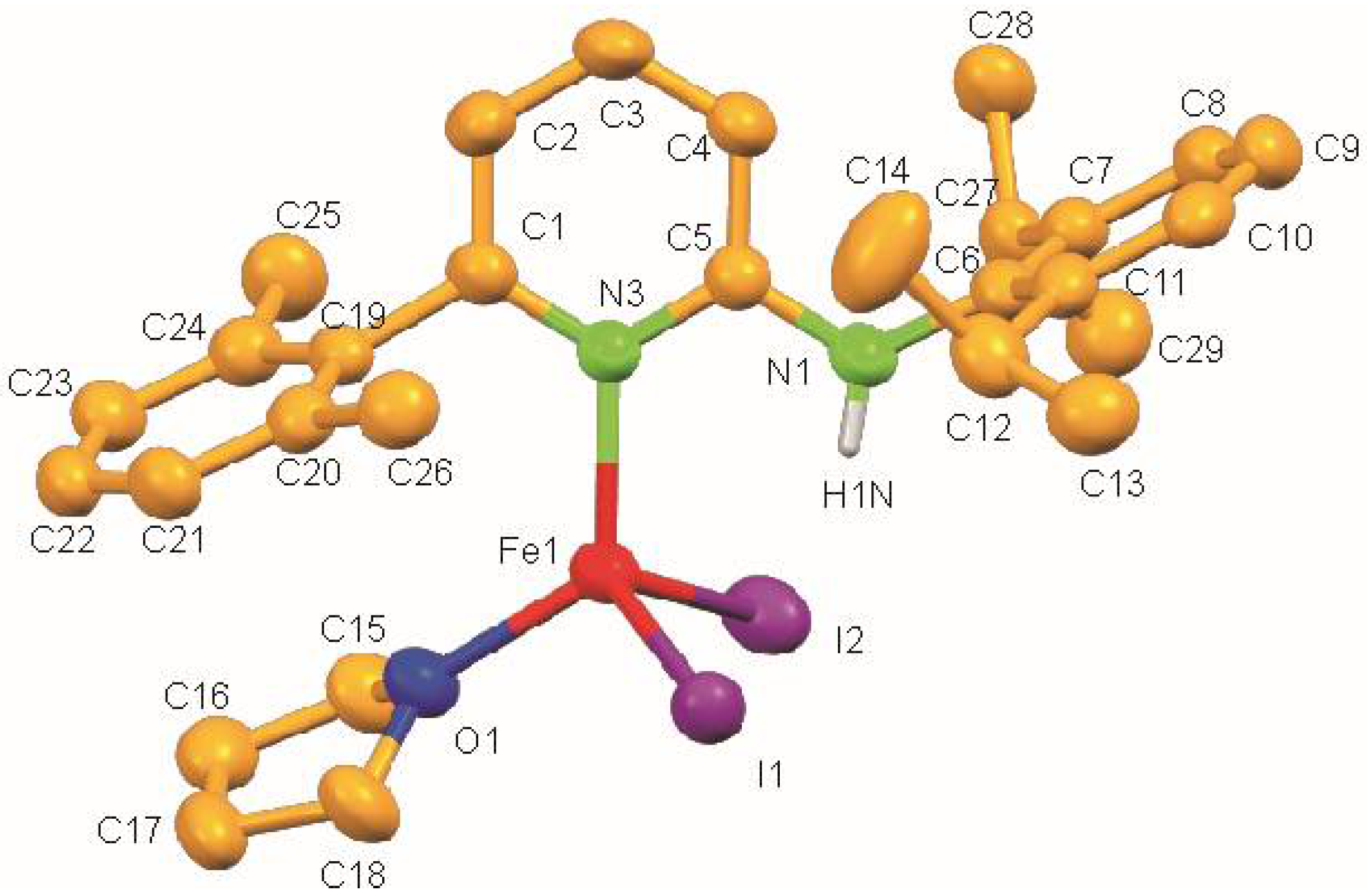
3. Results
4. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kempe, R. The strained η2-NAmido−NPyridine coordination of aminopyridinato ligands. Eur. J. Inorg. Chem. 2003, 2003, 791–803. [Google Scholar] [CrossRef]
- Noor, A. Coordination chemistry of bulky aminopryridinates with main group and transition metals. Top. Curr. Chem. 2021, 379, 6. [Google Scholar] [CrossRef] [PubMed]
- Scott, N.M.; Schareina, T.; Tok, O.; Kempe, R. Lithium and Potassium Amides of Sterically Demanding Aminopyridines. Eur. J. Inorg. Chem. 2004, 2004, 3297–3304. [Google Scholar] [CrossRef]
- Xu, X.; James, S.L.; Mingos, D.M.P.; White, A.J.P.; Williams, D.J. Platinum(II) phosphine and orotate complexes with aminopyridine co-ligands, and their molecular recognition via hydrogen bonding. J. Chem. Soc. Dalton Trans. 2000, 29, 3783–3790. [Google Scholar] [CrossRef]
- Kempe, R. Rare Earth Polymerization Catalysts Supported by Bulky Aminopyridinato Ligands. Z. Anorg. Allg. Chem. 2010, 636, 2135–2147. [Google Scholar] [CrossRef] [Green Version]
- Glatz, G.; Demeshko, S.; Motz, G.; Kempe, R. First Row Transition Metal Aminopyridinates—The Missing Complexes. Eur. J. Inorg. Chem. 2009, 2009, 1385–1392. [Google Scholar] [CrossRef]
- Chen, M.S.; White, M.C. A Predictably Selective Aliphatic C–H Oxidation Reaction for Complex Molecule Synthesis. Science 2007, 318, 783–787. [Google Scholar] [CrossRef]
- Nielsen, A.; Larsen, F.B.; Bond, A.D.; McKenzie, C.J. Regiospecific Ligand Oxygenation in Iron Complexes of a Carboxylate-Containing Ligand Mediated by a Proposed FeV–Oxo Species. Angew. Chem. Int. Ed. 2006, 118, 1632–1636. [Google Scholar] [CrossRef]
- Costas, M.; Chen, K.; Que, L., Jr. Biomimetic nonheme iron catalysts for alkane hydroxylation. Coord. Chem. Rev. 2000, 200–202, 517–544. [Google Scholar] [CrossRef]
- Sundaresan, S.; Eppelsheimer, J.; Carrella, L.M.; Rentschler, E. Three Novel Thiazole-Arm Containing 1,3,4-Oxadiazole-Based [HS-HS] Fe(II) Dinuclear Complexes. Crystals 2022, 12, 404. [Google Scholar] [CrossRef]
- Sundaresan, S.; Kühne, I.A.; Kelly, C.T.; Barker, A.; Salley, D.; Müller-Bunz, H.; Powell, A.K.; Morgan, G.G. Anion Influence on Spin State in Two Novel Fe(III) Compounds: [Fe(5F-sal2333)]X. Crystals 2019, 9, 19. [Google Scholar] [CrossRef] [Green Version]
- Prakash, O.; Chábera, P.; Rosemann, N.W.; Huang, P.; Häggström, L.; Ericsson, T.; Strand, D.; Persson, P.; Bendix, J.; Lomoth, R.; et al. A Stable Homoleptic Organometallic Iron(IV) Complex. Chem. Eur. J. 2020, 26, 12728–12732. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, M.; Singh, K.K.; Panda, C.; Weitz, A.; Hendrich, M.P.; Collins, T.J.; Dhar, B.B.; Sen Gupta, S. Formation of a Room Temperature Stable FeV(O) Complex: Reactivity Toward Unactivated C–H Bonds. J. Am. Chem. Soc. 2014, 136, 9524–9527. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Noor, A.; Wagner, F.R.; Kempe, R. Metal–metal distances at the limit: A coordination compound with an ultrashort chromium–chromium bond. Angew. Chem. Int. Ed. 2008, 47, 7246–7249. [Google Scholar] [CrossRef] [PubMed]
- Noor, A.; Glatz, G.; Müller, R.; Kaupp, M.; Demeshko, S.; Kempe, R. Carboalumination of a chromium–chromium quintuple bond. Nat. Chem. 2009, 1, 322–325. [Google Scholar] [CrossRef] [PubMed]
- Noor, A.; Qayyum, S.; Schwarz, S.; Dietel, T.; Kempe, R. Formation of a dimeric tungsten(I) complex via C-H activation. Dalton Trans. 2020, 49, 1992–1996. [Google Scholar] [CrossRef] [PubMed]
- Noor, A.; Bauer, T.; Todorova, T.K.; Weber, B.; Gagliardi, L.; Kempe, R. The ligand-based quintuple bond-shortening concept and some of its limitations. Chem. Eur. J. 2013, 19, 9825–9832. [Google Scholar] [CrossRef]
- Noor, A.; Glatz, G.; Müller, R.; Kaupp, M.; Demeshko, S.; Kempe, R. Metal–metal distances at the limit: Cr–Cr 1.73 Å—The importance of the ligand and its fine tuning. Z. Anorg. Allg. Chem. 2009, 635, 119–1152. [Google Scholar] [CrossRef]
- Wagner, F.R.; Noor, A.; Kempe, R. Ultrashort metal–metal distances and extreme bond orders. Nat. Chem. 2009, 1, 529–536. [Google Scholar] [CrossRef]
- Noor, A.; Kempe, R. The shortest metal-metal bond. Chem. Rec. 2010, 10, 413–416. [Google Scholar] [CrossRef]
- Noor, A.; Kempe, R. M5M—Key compounds of the research field metal–metal quintuple bonding. Inorg. Chim. Acta 2014, 424, 75–82. [Google Scholar] [CrossRef]
- Xia, J.; Matyjaszewski, K. Controlled/“Living” Radical Polymerization. Atom Transfer Radical Polymerization Using Multidentate Amine Ligands. Macromolecules 1997, 30, 7697–7700. [Google Scholar] [CrossRef]
- Thierer, L.M.; Jenny, S.E.; Shastri, V.; Donley, M.R.; Round, L.M.; Piro, N.A.; Kassel, W.S.; Brown, C.L.; Dudley, T.J.; Zubris, D.L. Amino pyridine iron(II) complexes: Characterization and catalytic application for atom transfer radical polymerization and catalytic chain transfer. J. Organomet. Chem. 2020, 924, 121456. [Google Scholar] [CrossRef]
- Jing, C.; Wang, L.; Mahmood, Q.; Zhao, M.; Zhu, G.; Zhang, X.; Wang, X.; Wang, Q. Synthesis and characterization of aminopyridine iron(II) chloride catalysts for isoprene polymerization: Sterically controlled monomer enchainment. Dalton Trans. 2019, 48, 7862–7874. [Google Scholar] [CrossRef] [PubMed]
- Gibson, V.C.; O’Reilly, R.K.; Wass, D.F.; White, A.J.P.; Williams, D.J. Iron complexes bearing iminopyridine and aminopyridineligands as catalysts for atom transfer radical polymerization. Dalton Trans. 2003, 32, 2824–2830. [Google Scholar] [CrossRef]
- Altomare, A.; Burla, M.C.; Camalli, M.; Cascarano, G.L.; Giacovazzo, C.; Guagliardi, A.; Moliterni, A.G.G.; Polidori, G.; Spagna, R. SIR 97: A new tool for crystal determination and refinement. J. Appl. Crystallogr. 1999, 32, 115–119. [Google Scholar] [CrossRef]
- Sheldrick, G.M. A short history of SHELX. Acta Crystallogr. Sect. A 2008, 64, 112–122. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Farrugia, L.J. WinGX Suite for small-molecule single-crystal crystallography. J. Appl. Crystallogr. 1999, 32, 837–838. [Google Scholar] [CrossRef]
- Turner, M.J.; McKinnon, J.J.; Wolff, S.K.; Grimwood, D.J.; Spackman, P.R.; Jayatilaka, D.; Spackman, M.A. Crystal Explorer 17; University of Western Australia: Pert, WA, Australia, 2017. [Google Scholar]
- Ayuso, A.E.; Noor, A.; Irrgang, T.; Glatz, G.; Kempe, R. Crystal structure of (2,6-diisopropyl-phenyl)-(6-(2,6-dimethyl-phenyl)-pyridin-2-yl)-amine-(2,6-diisopropyl-phenyl)-(6-(2,6-dimethyl-phenyl)-pyridin-2-yl)-amido-lithium, Li(C25H29N2)(C25H30N2). Z. Kristallogr. NCS 2007, 222, 281–283. [Google Scholar] [CrossRef]


| Compound | 1 | 2 |
|---|---|---|
| CCDC number | 2168545 | 2168544 |
| Empirical formula | C50H60Br4Fe2N4 | C29H38FeI2N2O |
| Formula weight | 1148.36 | 740.26 |
| crystal system | monoclinic | monoclinic |
| space group | C 2/c | P 21/c |
| a [Å] | 25.5750(5) | 10.3180(7) |
| b [Å] | 10.5150(5) | 16.1080(10) |
| c [Å] | 18.9610(8) | 18.6580(11) |
| α [deg] | ||
| β [deg] | 97.892(5) | 102.038(5) |
| γ [deg] | ||
| V, [Å3] | 5050.7(3) | 3032.8(3) |
| crystal size, [mm3] | 0.25 × 0.21 × 0.16 | 0.37 × 0.30 × 0.24 |
| ρcalcd, [g cm−3] | 1.510 | 1.621 |
| µ, [mm−1] (Mo Kα) | 3.775 | 2.556 |
| T, [K] | 133(2) | 133(2) |
| 2θ range, [deg] | 3.22–53.18 | 3.37–53.13 |
| No. of reflections unique | 5052 | 6095 |
| No. of reflections obs. [I > 2σ (I)] | 3014 | 4453 |
| No. of parameters | 281 | 326 |
| wR2 (all data) | 0.0816 | 0.1004 |
| R value [I > 2σ (I)] | 0.0352 | 0.0375 |
| Compound | Atoms | Bond Length | Atoms | Angles |
|---|---|---|---|---|
| 1 | Br1–Fe1 | 2.5260(8) | Fe1–Br1–Fe1 | 86.55(3) |
| Br2–Fe1 | 2.3985(8) | N2–Fe1–Br2 | 104.84(10) | |
| Fe1–N2 | 2.093(3) | N2–Fe1–Br1 | 138.02(10) | |
| Br2–Fe1–Br1 | 103.62(3) | |||
| N2–Fe1–Br1 | 98.07(10) | |||
| Br2–Fe1–Br1 | 120.32(3) | |||
| Br1–Fe1–Br1A | 93.45(2) | |||
| N2–C5–N1 | 117.3(3) | |||
| 2 | I1–Fe1 | 2.6245(7) | O1–Fe1–N3 | 123.45(13) |
| I2–Fe1 | 2.6024(7) | O1–Fe1–I2 | 100.82(9) | |
| Fe1–O1 | 2.032(3) | N3–Fe1–I2 | 108.21(9) | |
| Fe1–N3 | 2.080(3) | O1–Fe1–I1 | 105.87(9) | |
| N3–Fe1–I1 | 106.25(0) | |||
| I2–Fe1–I1 | 112.22(3) | |||
| C5–N1–C6 | 123.4(4) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Noor, A. Crystallographic Evidence of η1-Coordination of Bulky Aminopyridine in Halide-Containing Iron (II) Complexes. Crystals 2022, 12, 697. https://doi.org/10.3390/cryst12050697
Noor A. Crystallographic Evidence of η1-Coordination of Bulky Aminopyridine in Halide-Containing Iron (II) Complexes. Crystals. 2022; 12(5):697. https://doi.org/10.3390/cryst12050697
Chicago/Turabian StyleNoor, Awal. 2022. "Crystallographic Evidence of η1-Coordination of Bulky Aminopyridine in Halide-Containing Iron (II) Complexes" Crystals 12, no. 5: 697. https://doi.org/10.3390/cryst12050697
APA StyleNoor, A. (2022). Crystallographic Evidence of η1-Coordination of Bulky Aminopyridine in Halide-Containing Iron (II) Complexes. Crystals, 12(5), 697. https://doi.org/10.3390/cryst12050697






