Heterotridentate Organomonophosphines in Pt(κ3–X1P1X2)(Y) (X1,2 = N1,2 or S1,2), Pt(κ3–P1N1X1)(Y) (X1 = O, C, S or Se) Pt(κ3–P1S1Cl1)(Cl) and Pt(κ3–P1Si1N1)(OL)—Structural Aspects
Abstract
1. Introduction
2. Results and Discussion
2.1. Pt(κ3–P1N1N2)(Y) Derivatives
2.2. Pt(κ3–P1N1O1)(Y) Derivatives
2.3. Pt(κ3–P1N1C1)(Y) Derivatives
2.4. Pt(κ3–P1N1S1)(Y) Derivatives
2.5. Pt(κ3–P1N1Se1)(Y) Derivatives
2.6. Pt(κ3–N1P1N2)(Cl) and Pt(κ3–S1P1S2)(Cl) Derivatives
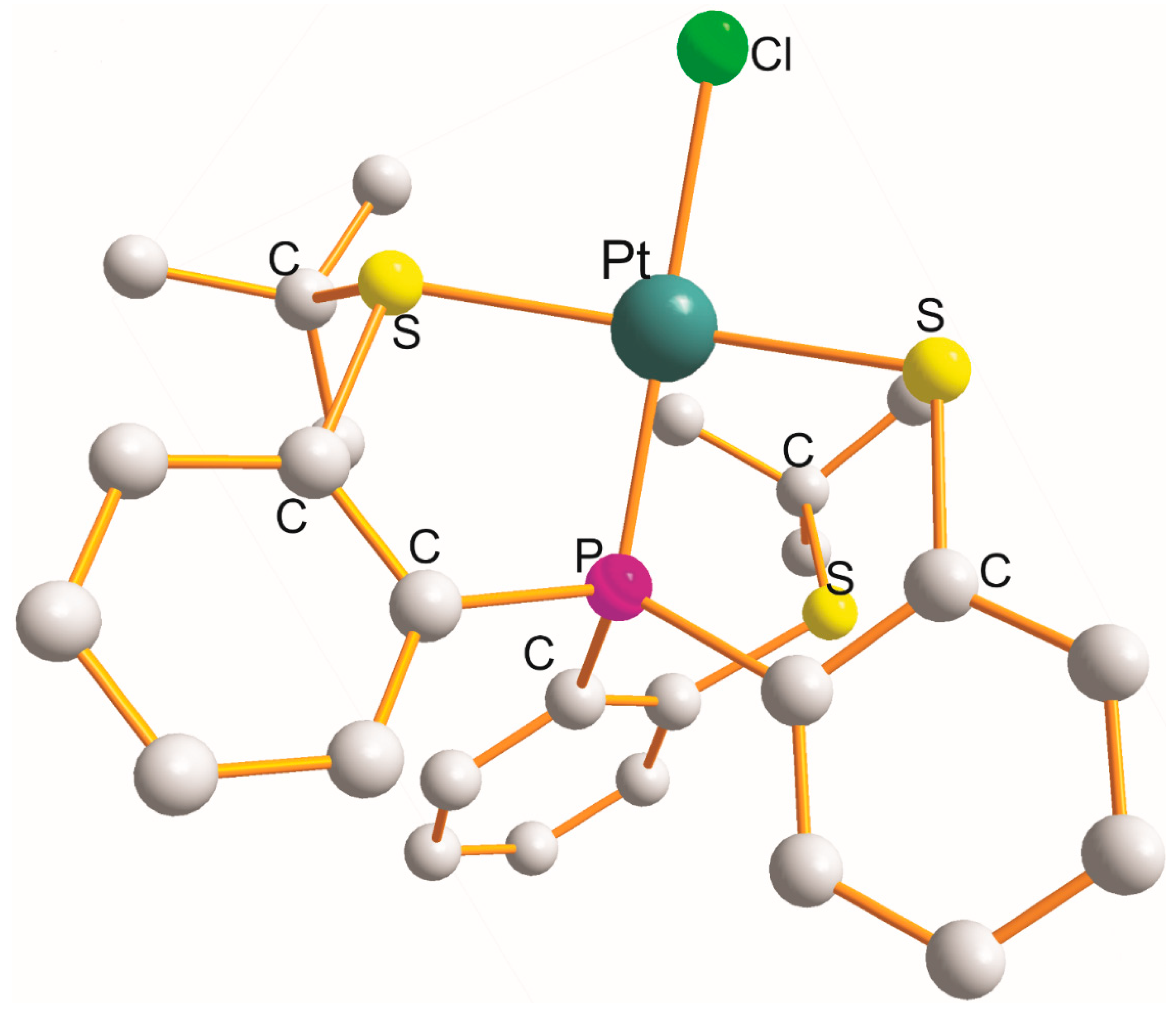
2.7. Pt(κ3–P1S1Cl1)(Cl) and Pt(κ3–P1Si1N1)(OL) Derivatives
- 5 + 5—membered: P1C2P1C2N2 (1 example), P1C2N1NCO1 (1 example), P1C2N1NCC1 (1 example) and S1C2N1C2S2 (2 examples)
- 6 + 5—membered: P1C3N1C2N2 (6 examples), P1C3N1NCN2, (1 example), P1C3N1C2C1, (1 example) and P1C3N1NCS1 (1 example)
- 5 + 6—membered: P1C2N1C3O1 (3 examples), P1C2S1C2BCl1, (1 example) and P1C2Si1C3N1 (1 example)
- 6 + 6—membered: P1C3N1C3N2, P1C3N1C3O1, P1C3N1C3S1, P1C3N1C3Se1 and N1C2NP1NC2N2 (each 1 example)
3. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| But2P1(CH2)(C5H3N1)(CH2)N2Et2 | (2-(di-t-butylphosphinomethyl)-6-diethyl-aminomethyl)pyridine) |
| ButS1(C6H4)P1(C6H4SBut)(C6H4)S2) | (2-((2-(t-butylsulfanyl)phenyl)(2-t-butyl-sulfanyl)phenyl)phosphino)benzenethiazato |
| cyh2P1(C6H4)Si1(CH3)(C7H6N1(Me2)) | ((2-(dicyclohexylphosphanyl)(2-(dimethyl-amino)methyl)phenyl)methylsilyl) |
| m | monoclinic |
| N1(C6H6)N(C6H10)NP1(Pri)(C6H6)N2 | (2-isopropyl-1,3-bis(2-pyridylmethyl)-octahydro-1H-1,3,2-benzodiazophosphore |
| or | orthorhombic |
| Ph2P1((C6H4CHN1NC(S1).NHMe) | (2-(diphenylphosphino)thiosemicarbazide |
| Ph2P1(C23H28S1)(B(Ph2)Cl1) | ((2,7-di-t-butyl-5-((chloro)(diphenyl)-25-boranyl)-9,9-dimethyl-9H-thioxantin-4-yl)(diphonylphosphine) |
| Ph2P1(C6H4N1)(C7H4ClO1)(C7H8N2) | (4-chloro-2-(((2-(diphenylphosphino)phenylimino)methylphenylate |
| Ph2P1(C6H4N1)(C8H7NOO1) | (2-(((2-(diphenylphosphino)phenylimino)methyl-4-methoxyphenylato) |
| Ph2P1(C7H5N1)(C2H2O)N2(C6H4OH) | (N-(2-(diphenylphosphinobenzylidene)-N-(2-hydroxyphenyl)glycinamidato) |
| Ph2P1(C7H5N1)(C2H2O)N2(C6H4OH) | (N2-(2-(diphenylphosphino)benzylidene)-N-(3-hydroxyphenyl)glycinamidato |
| Ph2P1(C7H5N1)(C2H2O)N2(C7H6OH) | (N-(2-(diphenylphosphino)benzylidene)-N-(2-hydroxymethylphenyl)glycine-amidato) |
| Ph2P1(C7H5N1)(C3H6)N2(C7H5O2) | (N-(2-((2-(diphenylphosphino)benzylidene)amino)propyl)-2-hydroxybenzamidato) |
| Ph2P1(C7H5N1)(C5H7O)N2(C10H10N2) | R7C-(N-(5,7-(dimethyl-1,8-naphtylridin-2-yl)-N2-(2-(diphenylphosphinyl)benzylidene) valinamidato) |
| Ph2P1(C7H5N1)(C6H4)N2(C10H9NO3) | (N2-benzyloxycarbinol)-N-(2-(((2-(diphenylphosphanyl)phenyl)methylidene) amino)phenyl)glycinamide) |
| Ph2P1(C7H5N1)(C7H8C1)(C7H8N2) | (2-(1b)-1-(((2-diphenylphosphinobenzylidene)amine) ethyl)phenyl) |
| Ph2P1(C7H5N1)(MeS1)(ButNH2) | (N-{N-[2-(diphenylphosphino)benzylidene)]-D/L-methionyl}-terc-butylamine |
| Ph2P1(C7H5N1)(NC5H4N2) | (2-(2-(diphenylphosphino)benzylidene)-1-(pyridine-2-yl)diazanido |
| Ph2P1(C7H5N1C3H6Se1(Ph) | (N-(2-(diphenylphosphino)benzylidene)-N-(3-(phenylseleno)propyl)amine |
| Ph2P1(C7H5N1O1) | (2-diphenylphosphino)-2-aminobenzaldehyde) |
| Ph2P1(C7H6N1)(C7H8N)(C7H8N2) | (N2-(2-(diphenylphosphino)benzyl)-N,N-bis(2-pyridyl-2-ethyl)amine) |
| Ph2P1(C7H6N1)(NC7H5O1)Ph2P2(C15H13N2O) | (N-(2-(diphenylphosphino)-1-phenylformyl)benzohydiazino)-N-(2-(diphenylphosphino)-1-phenylvinyl)benzohydeazone) |
| Ph2P1(C7H6N1 = NCC1C5H6) | (2-((2-(diphenylphosphino)-4-methylphenyl)diazinyl)-5-methylphenyl)pyridine |
| PriS1(C6H4)P1(C6H4SPri).(C6H4)S2) | (2-(((2-(isopropysulfanyl)phenyl)(2-isopropylsulfanyl)phenyl)phosphino)benzenethiazato) |
| py | pyridine |
| tr | triclinic |
References
- Holloway, C.E.; Melnik, M. Structural aspect of platinum coordination compounds: Part III—Monomeric square planar (PtA2XY and PtABXY) and trigonal bipyramidal PtII coordination compounds. Rev. Inorg. Chem. 2004, 24, 135–299. [Google Scholar] [CrossRef]
- Holloway, C.E.; Melnik, M. Structural aspect of platinum coordination compounds: Part I—Monomeric Pt0, PtI and PtIIA4 derivatives. Rev. Inorg. Chem. 2002, 22, 163–284. [Google Scholar] [CrossRef]
- Holloway, C.E.; Melnik, M. Structural Aspect of Platinum Coordination Compounds: Part II—Monomeric PtII Compounds with PtA3B and PtA2B2 Composition. Rev. Inorg. Chem. 2003, 23, 125–287. [Google Scholar] [CrossRef]
- Melnik, M.; Holloway, C.E. Stereochemistry of platinum coordination compounds. Coord. Chem. Rev. 2006, 250, 2261–2270. [Google Scholar] [CrossRef]
- Melník, M.; Mikuš, P. Distortion isomers of cis-PtP2X2 and cis-PtP2XY derivatives—Structural aspects. Rev. Inorg. Chem. 2020, 40, 153–165. [Google Scholar] [CrossRef]
- Melník, M.; Mikuš, P. Ligand isomerism in Pt(II) complexes—Structural aspects. Rev. Inorg. Chem. 2022, 42, 21–28. [Google Scholar] [CrossRef]
- Melník, M.; Mikuš, P. Heterotridentate organodiphosphines in Pt(η3–P1X1P2)(Y) (X1 = B, S, or Si) and Pt(η3–P1P2Si1)(Y) derivatives-structural aspects. Rev. Inorg. Chem. 2021, 42, 21–28. [Google Scholar] [CrossRef]
- Melník, M.; Mikuš, P. Organodiphosphines in Pt{η2-P(X)nP}Cl2 (n = 9–15, 17, 18) derivatives—Structural aspects. Rev. Inorg. Chem. 2021, 41, 41–48. [Google Scholar] [CrossRef]
- Melník, M.; Mikuš, P. Heterotridentate organodiphosphines in Pt(η3–P1X1P2)(Y) derivatives-structural aspects. Rev. Inorg. Chem. 2021, 41, 41–48. [Google Scholar] [CrossRef]
- Vuzman, D.; Poverenov, E.; Shimon, L.J.W.; Diskin-Posner, Y.; Milstein, D. Cationic, Neutral and Anionic Platinum(II) Complexes Based on an Electron-Rich PNN Ligand. New Modes of Reactivity Based on Pincer Hemilability and Dearomatization. Organometallics 2008, 27, 2627–2634. [Google Scholar] [CrossRef]
- Durran, S.E.; Elsegood, M.R.J.; Hammond, S.R.; Smith, M.B. Flexible κ4-PNN′O-Tetradentate Ligands: Synthesis, Complexation and Structural Studies. Dalton Trans. 2010, 39, 7136–7146. [Google Scholar] [CrossRef] [PubMed]
- Durran, S.E.; Elsegood, M.R.J.; Hammond, S.R.; Smith, M.B. Coordination Studies of a New Unsymmetrical κ4-PNN‘N‘ ‘-Tetradentate Ligand: Stepwise Formation and Structural Characterization. Inorg. Chem. 2007, 46, 2755–2766. [Google Scholar] [CrossRef] [PubMed]
- Fuentes, J.A.; Clarke, M.L.; ZSlawin, A.M. A Supramolecular Approach to Chiral Ligand Modification: Coordination Chemistry of a Multifunctionalised Tridentate Amine-Phosphine Ligand. New J. Chem. 2008, 32, 689–693. [Google Scholar] [CrossRef]
- Elsegood, M.R.J.; Sanchez-Ballester, N.M.; Smith, M.B. New κ3-PNN′- and κ4-PNN′O-Polydentate Ligands: Synthesis, Coordination and Structural Studies. Inorg. Chim. Acta 2011, 379, 115–121. [Google Scholar] [CrossRef]
- Chang, M.; Horiki, H.; Nakajima, K.; Kobayashi, A.; Chang, H.-C.; Kato, M. Acid–Base Behavior of Substituted Hydrazone Complexes Controlled by the Coordination Geometry. Bull. Chem. Soc. Jpn. 2010, 83, 905–910. [Google Scholar] [CrossRef]
- Watkins, S.E.; Craig, D.C.; Colbran, S.B. Towards Co-Operative Reactivity in Conjoint Classical-Organometallic Heterometallic Complexes: The Co-Ordination Chemistry of Novel Ligands with Triphenylphosphine and Bis(Pyridylethyl)Amine or Triazacyclononane Domains. J. Chem. Soc. Dalton Trans. 2002, 12, 2423–2436. [Google Scholar] [CrossRef]
- Ahmad, M.; Perera, S.D.; Shaw, B.L.; Thornton-Pett, M. Uni-, Bi- and Ter-Dentate Complexes Formed from PPh2CH2C(R)NNHC(O)Ph (R = But or Ph) and Pd or Pt: Crystal Structures of [PdCl{PPh2CH2C(But)NNC(Ph)O}], [Pt{PPh2CHC(Ph)NNC(Ph)O}{PPh2CH2C(Ph)NNHC(O)Ph}] and [Pd{PPh2CHC(But)NHNC(O)Ph}2]. J. Chem. Soc. Dalton Trans. 2002, 9, 1954–1962. [Google Scholar] [CrossRef]
- Ní Dhubhghaill, O.M.; Lennon, J.; Drew, M.G.B. Palladium(Ii) and Platinum(Ii) Complexes with Tridentate Iminophosphine Ligands; Synthesis and Structural Studies. Dalton Trans. 2005, 19, 3213–3220. [Google Scholar] [CrossRef]
- Jircitano, A.J.; Mertes, K.B. Ligands with Dual Denticity: Crystal and Molecular Structure of Dichloro-Bis(o-Diphenylphosphino)-Benzaldehyde)Platinum(II). Inorg. Chim. Acta 1985, 103, L11–L13. [Google Scholar] [CrossRef]
- Kano, N.; Yamamura, M.; Meng, X.; Yasuzuka, T.; Kawashima, T. Different Coordination Modes of 2-(Diphenylphosphino)Azobenzenes in Complexation with Hard and Soft Metals. Dalton Trans. 2012, 41, 11491–11496. [Google Scholar] [CrossRef]
- Ramírez, P.; Contreras, R.; Valderrama, M.; Carmona, D.; Lahoz, F.J.; Balana, A.I. Cyclometallated Platinum(II) Complexes Containing the Chiral Ligand [2-(Diphenyl-Phosphanyl)-Benzylidene]-(1-Phenyl-Ethyl)-Amine: Synthesis and Molecular Structures of the Compounds [PtCl(Me){κ2-(R)-Ph2P(C6H4)CHNCH(Ph)Me-P,N}] and [Pt{κ3-(S)-Ph2P(C6H4)CHN. J. Organomet. Chem. 2008, 693, 349–356. [Google Scholar] [CrossRef]
- You, D.; Kang, S.O.; Ko, J.J.; Choi, M. Cycloplatinated complexes of thiosemicarbazones. Synthesis and crystal structure of [Ph2PC6H4CHNNC(S)NHCH3PtCl]. Bull. Korean Chem. Soc. 1997, 18, 305–310. [Google Scholar]
- Ankersmit, H.A.; Veldman, N.; Spek, A.L.; Vrieze, K.; van Koten, G. Methyl-, Acetyl- and Allyl-Palladium and -Platinum Complexes Containing Novel Terdentate PNS and NN’S Ligands. Inorg. Chim. Acta 1996, 252, 339–354. [Google Scholar] [CrossRef]
- Durran, S.E.; Elsegood, M.R.J.; Smith, M.B. New Complexes of Functionalised Ligands Bearing P/N/Se or P2Se Donor Sets. New J. Chem. 2002, 26, 1402–1408. [Google Scholar] [CrossRef]
- Edwards, P.G.; Kariuki, B.; Newman, P.D. Coordination Behaviour in Transition Metal Complexes of Asymmetric NPN Ligands. Polyhedron 2011, 30, 935–941. [Google Scholar] [CrossRef]
- Takeda, N.; Tanaka, Y.; Oma, R.; Sakakibara, F.; Unno, M. Activation of C-S Bond by Group 10 Metal Complexes: Reaction of Phosphine Ligand Tethered with Three tert-Butylthiophenyl Groups with Group 10 Metal Compounds. Bull. Chem. Soc. Jpn. 2016, 89, 922–930. [Google Scholar] [CrossRef]
- Emslie, D.J.H.; Cowie, B.E.; Oakley, S.R.; Huk, N.L.; Jenkins, H.A.; Harrington, L.E.; Britten, J.F. A Study of M–X–BR3 (M = Pt, Pd or Rh; X = Cl or I) Interactions in Square Planar Ambiphilic Ligand Complexes: Structural, Spectroscopic, Electrochemical and Computational Comparisons with Borane-Free Analogues. Dalton Trans. 2012, 41, 3523–3535. [Google Scholar] [CrossRef] [PubMed]
- Takaya, J.; Ito, S.; Nomoto, H.; Saito, N.; Kirai, N.; Iwasawa, N. Fluorine-Controlled C–H Borylation of Arenes Catalyzed by a PSiN-Pincer Platinum Complex. Chem. Commun. 2015, 51, 17662–17665. [Google Scholar] [CrossRef]
- Yang, L.; Powell, D.R.; Houser, R.P. Structural Variation in Copper(i) Complexes with Pyridylmethylamide Ligands: Structural Analysis with a New Four-Coordinate Geometry Index, τ4. Dalton Trans. 2007, 9, 955–964. [Google Scholar] [CrossRef]




| Complex | Space gr. Cryst. cl. Z | a [Å] b [Å] c [Å] | α[°] β[°] γ[°] | Chromophore (Chelate Rings) Ʈ4 b | Pt-L c [Å] | L-Pt-L c [°] | Ref. REFCODE |
|---|---|---|---|---|---|---|---|
| A: Pt(κ3-P1N1N2)(Y) | |||||||
| [Pt{κ3-But2P(CH2)(C5H3N1). (CH2)N2Et2}(Cl)].C6H6 (at 120 K) | tr Pī 2 | 9.158(0) 10.963(0) 16.018(0) | 77.29(0) 76.97(0) 69.11(0) | PtP1N1N2Cl P1C2N1C2N2 0.044 | P1 2.236(1) N1 1.997(2) N2 2.149(2) Cl 2.296(2) | P1,N1 85.5 d N1,N2 83.4 d P1,N2 168.0 P1,Cl 98.5 N2,Cl 92.6 N1,Cl 176.0 | [10] WOGDAY |
| [Pt{κ3-Ph2P1(C7H5N1). (C2H2O)N2C6H4OH)}(CH3)]. (CHCl3) (at 150 K) | tr Pī 2 | 9.917(1) 11.944(2) 14.872(2) | 99.17(0) 103.82(0) 112.53(0) | PtP1N1N2C P1C3N1C2N2 0.032 | P1 2.179(1) N1 2.050(2) N2 2.089(2) H3C 2.045(2) | P1,N1 95.3 e N1,N2 80.5 d P1,N2 173.7 P1,C 89.0 N2,C 95.5 N1,C 174.7 | [11] GAJMOV |
| [Pt{κ3-Ph2P1(C7H5N1). (C2H2O)N2(C6H4OH)}(CH3)]. 1.5 toluene (at 150 K) | m P21/c 4 | 11.882(1) 14.184(1) 21.892(1) | 103.75(0) | PtP1N1N2C P1C3N1C2N2 0.027 | P1 2.184(2) N1 2.061(3) N2 2.075(3) H3C 2.051(2) | P1,N1 95.4 e N1,N2 81.0 d P1,N2 176.4 P1,C 90.5 N2,C 93.0 N1,C 174.0 | [11] GAJMUB |
| [Pt{κ3-Ph2P1(C7H5N1). (C3H6)N2(C7H5O2)}(CH3)]. 2 toluene (at 150 K) | m P21/c 4 | 14.859(0) 15.607(0) 16.287(0) | 95.88(0) | PtP1N1N2C P1C3N1C2N2 0.033 | P1 2.189(1) N1 2.077(2) N2 2.070(2) H3C 2.062(2) | P1,N1 95.2 e N1,N2 80.3 d P1,N2 175.4 P1,C 89.4 N2,C 94.9 N1,C 172.6 | [11] GAJNAI |
| [Pt{κ3-Ph2P1(C7H5N1)(C6H4)N2 (C10H9NO3)}(CH3)]Et2O (at 150 K) | m P21/c 4 | 10.992(0) 20.133(0) 16.933(0) | 101.72(0) | PtP1N1N2C P1C3N1C2N2 0.040 | P1 2.184(1) N1 2.087(2) N2 2.086(2) H3C 2.062(2) | P1,N1 92.6 e N1,N2 79.4 d P1,N2 171.8 P1,C 91.5 N2,C 96.6 N1,C 173.7 | [12] QICYAD |
| [Pt{κ3-Ph2P1(C7H5N1)(C5H7O) N2(C10H10N2)}(CH3)]H2O (at 93 K) | m P21/c 4 | 8.739 (1) 14.988(2) 25.469(2) | 94.23(0) | PtP1N1N2C P1C3N1C2N2 0.052 | P1 2.190(1) N1 2.061(2) N2 2.094(2) H3C 2.083(2) | P1,N1 89.4 e N1,N2 79.6 d P1,N2 169.0 P1,C 92.2 N2,C 93.7 N1,C 172.1 | [13] DIYYIU |
| [Pt{κ3-Ph2P1(C7H5N1)(C2H2O) N2(C6H4OH)}(CH3)]CHCl3 (at 150 K) | m P21/c 4 | 10.191(0) 16.863(1) 17.525(1) | 97.30(0) | PtP1N1N2C P1C3N1C2N2 0.030 | P1 2.183(1) N1 2.059(1) N2 2.061(1) H3C 2.055(1) | P1,N1 95.1 e N1,N2 81.0 d P1,N2 175.8 P1,C 91.2 N2,C 92.6 N1,C 173.4 | [14] CAJLAC |
| [Pt{κ3–Ph2P1(C7H6N1 = NCC1. C5H6)}(Cl)] (at 150 K) | tr Pī 2 | 7.431(2) 10.031(3) 14.797(5) | 101.10(0) 95.70(0) 98.76(0) | PtP1N1N2C P1C3N1NCN2 0.038 | P1 2.219(1) N1 2.164(2) N2 2.050(1) Cl 2.297(2) | P1,N1 95.8 e N1,N2 79.3 d P1,N2 173.0 P1,Cl 90.8 N2,Cl 94.2 N1,Cl 173.4 | [15] XUYWEU |
| [Pt{κ3-Ph2P1(C7H5N1)(C7H8N) (C7H8N2)}(Cl)]PF6 | m P21/c 4 | 18.910(3) 10.098(1) 19.429(3) | 118.93(1) | PtP1N1N2Cl P1C3N1CN2 0.020 | P1 2.234(1) N1 2.120(1) N2 2.104(1) Cl 2.284(1) | P1,N1 93.3 e N1,N2 85.6 d P1,N2 178.7 P1,Cl 91.8 N2,Cl 89.2 N1,Cl 174.0 | [16] IFUQEF |
| B: Pt(κ3–P1N1O1)(Y) | |||||||
| [Pt{κ3-Ph2P1(C8H6N1)(N. C7H5O1)}{κ1-Ph2P. (C15H13N2O)}].CH2Cl2 (at 200 K) | tr Pī 2 | 12.614(2) 13.671(2) 15.754(3) | 100.26(0) 99.33(0) 110.68(0) | PtP1N1O1P P1C2N1NCO1 0.067 | P1 2.233(2) N1 1.985(2) O1 2.050(2) LP 2.261(1) | P1,N1 83.6 d N1,O1 78.8 d P1,O1 162.4 P1,P 102.9 O1,P 94.7 N1,Cl 173.3 | [17] EFODAE |
| [Pt{κ3-Ph2P1(C6H4N1). (C7H4ClO1)}(P(p-tolyl3)]ClO4 (at 200 K) | m P21/c 4 | 12.614(14) 20.280(20) 16.972(17) | 98.96(1) | PtP1N1O1P P1C2N1C3O1 0.028 | P1 2.21(1) N1 2.05(2) O1 2.03(2) LP 2.269(1) | P1,N1 82.7 N1,O1 91.2 P1,O1 172.1 P1,P 99.6 O1,P 86.5 N1,P 177.7 | [18] KAVZOX |
| [Pt{κ3-Ph2P1(C6H4N1). (C8H7OO1)} (Cl)] | m P21/n 4 | 12.350(12) 12.138(14) 15.550(17) | 97.70(1) | PtP1N1O1Cl P1C2N1C3O1 0.007 | P1 2.195(1) N1 2.005(2) O1 2.080(2) Cl 2.303(1) | P1,N1 83.6 d N1,O1 92.3 e P1,O1 178.5 P1,Cl 93.5 O1,Cl 87.9 N1,Cl 178.9 | [18] KAVZAJ |
| [Pt{κ3-Ph2P1(C6H4N1). (C8H7OO1)}(I)](CH2Cl2) | m P21/c 4 | 10.446(11) 16.389(17) 16.507(0) | 100.241(1) | PtP1N1O1I P1C2N1C3O1 0.014 | P1 2.207(1) N1 2.011(2) O1 2.045(2) I 2.620(1) | P1,N1 84.8 d N1,O1 91.9 e P1,O1 176.6 P1,I 92.6 O1,I 89.2 N1,I 178.2 | [18] KAVZEN |
| [Pt{κ3-Ph2P1(C8H7N1O1)}(Cl)] | or Pna21 4 | 18.88(2) 13.10(1) 9.66(1) | PtP1N1O1Cl P1C3N1C3O1 0.027 | P1 2.206(1) N1 1.88(1) O1 2.14(1) Cl 2.386(4) | P1,N1 94.8(4) e N1,O1 93.3(4) e P1,O1 175.5 P1,Cl 89.1(2) O1,Cl 84.0(2) N1,Cl174.8 | [19] DERNIX | |
| C: Pt(κ3–P1N1C1)(Y) | |||||||
| [Pt{κ3-Ph2P1(C7H6N1 = NC. C1C5H6)}(Cl)] (at 120 K) | m P21/n 4 | 8.632(4) 17.191(8) 15.216(7) | 96.3(0) | PtP1N1C1Cl P1C2N1NCC1 0.060 | P1 2.291(2) N1 1.972(2) C1 2.023(2) Cl 2.309(1) | P1,N1 85.2 d N1,C1 78.7 d P1,C1 163.9 P1,Cl 99.9 C1,Cl 84.0 N1,Cl174.6 | [20] YEHMOP |
| [Pt{κ3-Ph2P1(C7H5N1). (C7H8C1)}(py)]BF4 (at 100 K) | m P21 4 | 9.356(0) 19.892(1) 15.084(1) | 90.76(0) | PtP1N1C1N P1C3N1C2C1 0.042 | P1 2.292(1) N1 2.000(2) C1 2.035(2) pyN 2.026(1) | P1,N1 92.1 e N1,C1 82.3 d P1,C1 174.3 P1,N 92.4 C1,N 93.6 N1,Cl170.7 | [21] NIVCAX |
| D: Pt(κ3–P1N1S1)(Y) | |||||||
| [Pt{κ3-Ph2P1(C6H4CHN1NC. (S1)NHMe}(Cl)] | m P21/c 4 | 14.695(6) 16.683(7) 19.297(9) | 102.83(6) | PtP1N1S1Cl P1C3N1NCS1 0.022 | P1 2.239(5) N1 2.03(2) S1 2.298(5) Cl 2.304(5) | P1,N1 95.8(4) e N1,S1 84.9(4) d P1,S1 177.8(2) P1,Cl 89.5(2) S1,Cl 89.8(2) N1,Cl174.4 (4) | [22] HAFMOQ |
| [Pt{κ3-Ph2P1(C7H5N1)(MeS1). (But.NH2)}(I)] | tr Pī 2 | 10.529(1) 11.558(1) 14.550(1) | 77.37(1) 84.45(1) 79.72(1) | PtP1N1S1I P1C3N1C3S1 0.023 | P1 2.240(2) N1 2.056(6) S1 2.363(2) I 2.580(1) | P1,N1 89.1(1) e N1,S1 93.2(1) e P1,S1 176.2(2) P1,I 93.6(2) S1,I 84.2(2) N1,I 175.4(2) | [23] ROBHOP |
| E: Pt(κ3–P1N1Se1)(Cl) | |||||||
| [Pt{κ3-Ph2P1(C7H5N1). (C3H6Se1)(Ph)}(Cl)]BF4 (at 150K) | m P21/c 4 | 9.869(0) 23.847(0) 11.740(0) | 99.65(0) | PtP1N1Se1Cl P1C3N1C3Se1 0.012 | P1 2.407(14) N1 2.028(4) Se1 2.489(1) Cl 2.308(1) | P1,N1 87.5(1) e N1,Se1 95.7(1) e P1,Se1 176.7(1) P1,Cl 93.0(1) Se1,Cl 83.7(1) N1,Cl 178.8(1) | [24] MULZIC |
| Complex | Space gr. Cryst. cl. Z | a [Å] b [Å] c [Å] | α [°] β [°] γ [°] | Chromophore (Chelate Rings) Ʈ4 b | Pt-L c [Å] | L-Pt-L c [°] | Ref. REFCODE |
|---|---|---|---|---|---|---|---|
| [Pt{κ3-N1(C6H6)N(C6H10)N.. P1(Pri) (C6H6)N2}(Cl)].H2O (at 150 K) | or P212121 6 | 14.373(0) 9.906(0) 17.590(0) | PtN1P1N2Cl N1C2NP1NC2N2 0.032 | N1 2.035 P1 2.187 N2 2.039 Cl 2.375 | N1,P1 91.1 e P1,N2 91.0 e N1,N2 175.4 N1,Cl 90.4 N2,Cl 91.0 P1,Cl 173.0 | [25] IRAWOO | |
| [Pt{κ3-PriS1(C6H4)P1. (C6H4SPri) (C6H4)S2}(Cl)] (at 123 K) | m P21/n 4 | 8.790(0) 18.706(1) 15.508(1) | 95.89(0) | PtS1P1S2Cl S1C2P1C2S2 0.055 | S1 2.289 P1 2.189 S2 2.292 Cl 2.374 | S1,P1 88.2 d P1,S2 87.7 d S1,S2 162.1 S1,Cl 93.0 S2,Cl 90.8 P1,Cl 178.3 | [26] EZORAO |
| [Pt{κ3-ButS1(C6H4)P1. (C6H4SBut)(C6H4)S2}(Cl)].0.5CHCl3 (at 123 K) | m P21/n 4 | 10.250(1) 18.715(2) 15.320(1) | 96.65(0) | PtS1P1S2Cl S1C2P1C2S2 0.063 | S1 2.287 P1 2.198 S2 2.297 Cl 2.360 | S1,P1 88.7 d P1,S2 88.8 d S1,S2 158.7 N1,Cl 92.8 N2,Cl 90.8 P1,Cl 178.6 | [26] EZOQIV |
| [Pt{κ3-Ph2P1(C23H28S1). (B)(Ph2)Cl1)}(Cl)].2CH2Cl2 (at 123 K) | or Pna21 4 | 21.373(0) 8.959(0) 25.330(3) | PtP1S1Cl1Cl P1C2S1C2BCl1 0.033 | P1 2.212 S1 2.243 Cl1 2.391 Cl2 2.321 | P1,S1 87.9 d S1,Cl1 87.1 e P1,Cl1 174.7 P1,Cl 93.4 Cl1,Cl 91.7 S1,Cl 173.3 | [27] DASMER | |
| [Pt{κ3-cyh2P1(C6H4)Si1. (CH3)(C7H6)N1(CH3)2)}. (OSO2CF3)] (at 123 K) | m P21/c 4 | 19.851(1) 20.837(3) 15.443(2) | 99.52(0) | PtP1Si1N1O P1C2Si1C3N1 0.033 | P1 2.228 Si1 2.260 N1 2.177 LO 2.353 | P1,Si1 85.8 d Si1,N1 82.7 e P1,N1 169.2 P1,O 95.0 N1,O 86.4 Si1,O 179.0 | [28] WUXFAI |
| Metallocyclic Rings | α- L-Pt-L [°] | β- L’-Pt-Y [°] | Ʈ4 |
|---|---|---|---|
| 5 + 5—membered | 163.0 | 176.2 | 0.058 |
| 6 + 5—membered | 172.9 | 173.6 | 0.037 |
| 5 + 6—membered | 174.2 | 177.5 | 0.023 |
| 6 + 6—membered | 176.5 | 175.6 | 0.022 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Melník, M.; Mikuš, P. Heterotridentate Organomonophosphines in Pt(κ3–X1P1X2)(Y) (X1,2 = N1,2 or S1,2), Pt(κ3–P1N1X1)(Y) (X1 = O, C, S or Se) Pt(κ3–P1S1Cl1)(Cl) and Pt(κ3–P1Si1N1)(OL)—Structural Aspects. Crystals 2022, 12, 1772. https://doi.org/10.3390/cryst12121772
Melník M, Mikuš P. Heterotridentate Organomonophosphines in Pt(κ3–X1P1X2)(Y) (X1,2 = N1,2 or S1,2), Pt(κ3–P1N1X1)(Y) (X1 = O, C, S or Se) Pt(κ3–P1S1Cl1)(Cl) and Pt(κ3–P1Si1N1)(OL)—Structural Aspects. Crystals. 2022; 12(12):1772. https://doi.org/10.3390/cryst12121772
Chicago/Turabian StyleMelník, Milan, and Peter Mikuš. 2022. "Heterotridentate Organomonophosphines in Pt(κ3–X1P1X2)(Y) (X1,2 = N1,2 or S1,2), Pt(κ3–P1N1X1)(Y) (X1 = O, C, S or Se) Pt(κ3–P1S1Cl1)(Cl) and Pt(κ3–P1Si1N1)(OL)—Structural Aspects" Crystals 12, no. 12: 1772. https://doi.org/10.3390/cryst12121772
APA StyleMelník, M., & Mikuš, P. (2022). Heterotridentate Organomonophosphines in Pt(κ3–X1P1X2)(Y) (X1,2 = N1,2 or S1,2), Pt(κ3–P1N1X1)(Y) (X1 = O, C, S or Se) Pt(κ3–P1S1Cl1)(Cl) and Pt(κ3–P1Si1N1)(OL)—Structural Aspects. Crystals, 12(12), 1772. https://doi.org/10.3390/cryst12121772







