Abstract
In this work, we synthesized two new s-triazine incorporates pyrazole/piperidine/aniline moieties. Molecular structure investigations in the light of X-ray crystallography combined with Hirshfeld and DFT calculations were presented. Intermolecular interactions controlling the molecular packing of 4-(3,5-dimethyl-1H-pyrazol-1-yl)-N-phenyl-6-(piperidin-1-yl)-1,3,5-triazin-2-amine; 5a and N-(4-bromophenyl)-4-(3,5-dimethyl-1H-pyrazol-1-yl)-6-(piperidin-1-yl)-1,3,5-triazin-2-amine; 5b were analyzed using Hirshfeld calculations. The most dominant interactions are the H...H, N...H and H...C contacts in both compounds. The N...H and H...C interactions in 5a and the N...H, Br...H and H...H interactions in 5b are the most important. In addition, DFT calculations were used to compute the molecular structures of 5a and 5b; then, their electronic properties, as well as the 1H- and 13C-NMR spectra, were predicted. Both compounds are polar where 5a (1.018 Debye) has lower dipole moment than 5b (4.249 Debye). The NMR chemical shifts were calculated and very good correlations between the calculated and experimental data were obtained (R2 = 0.938–0.997).
1. Introduction
s-Triazine scaffold has been extensively studied in the literature due to its wide applications in different fields, including coordination chemistry [1] and drug discovery development, such as Enasidenib (Idhifa), as a representative example, having the s-triazine privileged structure, which has been approved by FDA in 2017 for the IDH2-positive acute leukemia treatment [2,3]. Other pharmaceutical applications of the substituted s-triazine core structure were reported as fungicidal [4], antiprotozoal [5], antibacterial [6], sedative and anti-inflammatory [7], cytotoxic [8], antiviral [9], analgesic [10], and herbicide activity [11].
A combination of the s-triazine privileged structure with other N-heterocycles such as pyrazoles [12,13,14] and quinazoline have raised a great of attention in cancer chemotherapeutics targeting enzyme inhibitors such as TK and EGFR inhibitors [15,16].
In addition, s-triazine core structure has gained attention in the pharmaceutical industry but has also had a great impact in the polymer industrial applications due to ease of functionalization and mechanical and thermal stability [17]. Indeed, this fascinating structure is exploited in the metal–organic framework (MOFs) [18] as well as the storage and capture of carbon dioxide chemistry [19,20,21].
Several examples that have been reported in the literature have showed that replacement of the three chlorine atoms in the 2,4,6-trichlorotriazine (TCT) with different N-heterocycles or amines are an important task and exhibited several applications [22,23,24] (Figure 1). For example, El-Faham et al. have been designed and synthesized in a series of s-triazine hybrids comprising mon-/di-pyrazolyl along with piperidine moiety, which were subsequently assessed in vitro towards cancer activity and also in vivo toxicity in zebrafish embryos [25] (Compound I, Figure 1).
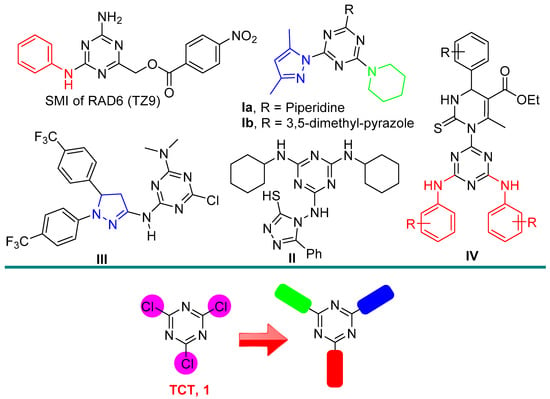
Figure 1.
Some reported hybrids-based s-triazine (I–IV) and our target product from 2,4,6-trichlorotriazine (TCT, 1) with different moieties.
Matthew A. Sanders et al., have reported a tri-substituted s-triazine as a novel and lead compound TZ9 (Figure 1) for selective RAD6B inhibitory activity and cellular anti-cancer activity [26]. El-Hamamsy and co-workers have been designed and synthesized a new scaffold-based, s-triazine-targeting hDHFR for lung cancer treatment. Compound II was found to have significant activity compared to methotrexate against the A549 cancer cell line [27]. Prashanth et al., reported novel lead compounds based on s-triazine tethered pyrazole moiety as a potential anti-cancer agent targeting EFGR kinase inhibitors with IC50 = 229.4 nm (Compound III, Figure 1) [28]. Additionally, the Singh research team have reported a new series of hybrids derived from monastrol-1,3,5-triazine and were studied in vitro and in vivo for their anti-cancer activity. The results indicated the potential efficacy of compound IV against cancer via inhibition of EGFR-TK (Figure 1) [29].
Therefore, the synthesis of new s-triazine derivatives connected to different amines such as aniline, piperidine, or heterocycles, such as pyrazole rings, could be useful for the drug discovery or as a new material for different applications.
In continuation of this research program towards the synthesis of s-triazine core structure with different moieties for biological activity applications. We reported here, the synthesis and characterization of two new compounds based on an s-triazine privileged structure having three different moieties such pyrazole, piperidine, and an aniline derivative. The chemical insights of the new compounds were explored via combined experimental and computational investigations.
2. Materials and Methods
Chemicals were purchased from Sigma-Aldrich Company (Chemie GmbH, Taufkirchen, Germany). 1H- and 13C-NMR spectra were recorded in CDCl3 and DMSO-d6 on a Jeol Spectrometer (Jeol, Tokyo, Japan) (400 MHz and 500 MHz). Infrared spectra were recorded on a Thermo Scientific Nicolet iS10 FT-IR spectrometer (Thermo Fisher Scientific, Waltham, MA, USA). X-ray diffraction data were collected on a Rigaku Oxford Diffraction Supernova diffractometer and processed with CrysAlisPro software v. 1.171.41.93a (Rigaku Oxford Diffraction, Yarnton, UK, 2020) using Cu Kα radiation.
Compounds 2a,b and 3a,b, which were required for this study, were prepared according to reported literature [25]. The synthetic pathway for the desired compounds are depicted in Scheme 1.
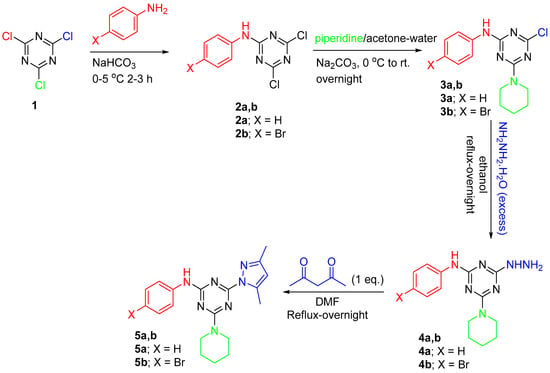
Scheme 1.
Synthesis of a new hits tethered pyrazole/piperidine/aniline-based s-triazine privileged structure.
General procedure (GP1) for the synthesis of compounds 4a,b
For the synthesis of monohydrazino s-triazine derivatives, 4a,b excess hydrazine hydrate-80% (5.0 equiv.) was added dropwise to a solution of disubstituted s-triazine derivative 3a,b (1.0 equiv.) in refluxing ethanol overnight, the evolution of the reaction was monitored by TLC (ethyl acetate/hexane 3:7). After completion of the reaction, the solution was cooled, and the precipitate was filtered and washed with diethyl ether to afford monohydrazino s-triazine derivatives as white solids in a very good yield and purity. Spectroscopic data fully agreed with the proposed structures. All monohydrazino s-triazine derivatives were used directly without further purification in the next step to synthesize the corresponding pyrazolo s-triazene derivatives.
4-Hydrazinyl-N-phenyl-6-(piperidin-1-yl)-1,3,5-triazin-2-amine 4a
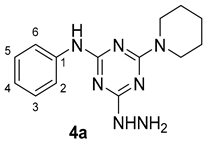
The reaction between 4-chloro-N-phenyl-6-(piperidin-1-yl)-1,3,5-triazin-2-amine 3a (1.45 g, 5 mmol) and excess hydrazine hydrate (1.26 mL, 25.0 mmol) was carried out in refluxing ethanol (30 mL) for 12 h according to the general procedure (GP1). TLC analysis (ethyl acetate/hexane 3:7) showed a single spot which indicated completion of the reaction and formation of the product 4a as a pure compound. Thereafter, the resulting white solid was filtered and used in the next step without further purification (1.10 g, 3.86 mmol, 77%), m.p. 136–140 °C; IR (KBr, cm−1): 3288, 3160 (2NH), 1586 (C=N), 1506–1440 (aromatic C-C); 1H-NMR (400 MHz, DMSO-d6) δ 9.00 (s, 1H, -NHNH2), 7.95 (s, 1H, -NH-), 7.74 (d, J = 8.0 Hz, 2H, H-2, H-6), 7.23 (t, J = 7.7 Hz, 2H, H-3, H-5), 6.90 (t, J = 7.3 Hz, 1H, H-4), 3.72 (s, 4H, 2NCH2-), 1.61 (q, J = 6.2 Hz, 2H, -CH2-), 1.52–1.45 (m, 4H, 2-CH2-); 13C-NMR (126 MHz, DMSO-d6) δ 168.19, 164.62, 164.40 (3C=N), 141.13 (C1), 128.85 (C3,5), 121.71 (C4), 119.91 (C2,6), 44.11 (2CH2N), 25.99 (2-CH2-), 24.92 (-CH2); Chemical Formula: C14H19N7.
N-(4-Bromophenyl)-4-hydrazinyl-6-(piperidin-1-yl)-1,3,5-triazin-2-amine
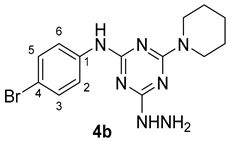
The reaction of N-(4-bromophenyl)-4-chloro-6-(piperidin-1-yl)-1,3,5-triazin-2-amine 3b (1.843 g, 5.00 mmol) and excess hydrazine hydrate (1.26 mL, 25.0 mmol) in refluxing ethanol (35 mL) was carried out according to the general procedure (GP1). After 12 h, the TLC analysis (ethyl acetate/hexane 3:7) provided evidence that the desired hydrazine derivative 4b was produced in its pure form. Ethanol and excess hydrazine were evaporated by vacuo as a part of work up procedure followed by an addition of 15 mL of n-hexane to the product as a white precipitate, which was collected by filtration (1.55 g, 4.75 mmol, 95%); 198–201 °C; IR (KBr, cm−1): 3224, 3230 (2NH), 1581 (C=N), 1506–1440 (aromatic C-C); 1H-NMR (400 MHz, DMSO-d6) δ 9.16 (s, 1H, -NHNH2), 7.97 (s, 1H, -NH-), 7.74 (d, J = 8.5 Hz, 2H, H-2, H-6), 7.38 (d, J = 8.6 Hz, 2H, H-3, H-5), 4.19 (s, 1H, -NH), 3.71 (s, 4H, 2NCH2-), 1.60 (q, J = 5.8 Hz, 2H, -CH2-), 1.51–1.44 (m, 4H, 4H, 2-CH2-); 13C-NMR (101 MHz, DMSO-d6) δ 168.35, 164.64 (3C=N), 140.71 (C1), 131.57 (C4), 121.80 (C3,5), 113.12 (C2,6), 44.18 (2CH2N-), 26.04 (2-CH2-), 24.96 (-CH2-); Chemical Formula: C14H18BrN7.
General procedure (GP2) for the synthesis of compounds 5a,b
For the synthesis of mono-pyrazole s-triazine derivatives 5a,b, a solution of acetyl acetone (1.2 equiv.) in DMF was added to a stirring solution of monohydrazino s-triazene derivative 4a,b (1 equivalent) in DMF at rt. The reaction mixture was treated with triethylamine (0.8 equiv.) and was allowed to reflux while stirring overnight. It was noticed that the reactivity of bromo-hydrazino s-triazine derivative 5b was relatively lower than that of the other hydrazino s-triazine derivatives, which required starting with excess amounts of acetylacetone (1.8 equiv.) and triethylamine (1.2 equiv.) for each equivalent of 4b. After completion of the reaction as identified by TLC analysis (ethyl acetate/hexane 5:5), it was allowed to cool down to rt., and then a small amount of ice-cold water was added with continuous stirring for 30 min. Thereafter, the reaction mixture was placed into an ice bath for 1 h and the precipitate of the product was separated by filtration and washed with several portions of ice-cold water and then dried overnight under vacuum to afford the product a very good yield. Some of the final products needed further purification by column chromatography on silica gel (gradient 20–60% EtOAc in hexane).
4-(3,5-Dimethyl-1H-pyrazol-1-yl)-N-phenyl-6-(piperidin-1-yl)-1,3,5-triazin-2-amine
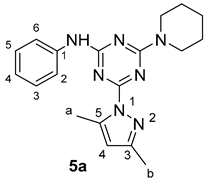
According to the general procedure (GP2), 4-hydrazinyl-N-phenyl-6-(piperidin-1-yl)-1,3,5-triazin-2-amine 4a (428 mg, 1.50 mmol) was reacted with acetylacetone (180 mg, 1.80 mmol) in the presence of triethylamine (121 mg, 1.20 mmol) in refluxing DMF (10 mL). The reaction progress was monitored by TLC (ethyl acetate/hexane 5:5), which indicated the completion of the reaction after 8 h. Subsequently, the reaction mixture was stirred with ice water and placed in an ice bath for about 30 min to create the white precipitate of compound 5a, which was filtered, washed with water, and dried under vacuum (503 mg, 1.44 mmol, 96%); m.p. 195–197 °C; IR (KBr, cm−1): 3440 (NH), 1588 (C=N), 1512–1440 (aromatic C-C); 1H-NMR (400 MHz, CDCl3) δ 7.57 (s, 2H, H-2, H-6), 7.33 (s, 2H, H-3, H-5), 7.05 (s, 1H, H-4), 6.00 (s, 1H, H-4; Pyrazole), 3.87–3.78 (m, 4H, 2NCH2-), 2.66 (s, 3H, H-b), 2.32 (s, 3H, H-a), 1.71 (s, 2H, -CH2-), 1.66 (s, 4H, 2-CH2-); 13C-NMR (101 MHz, CDCl3) δ 164.75, 164.19, 162.63 (3C=N), 151.58 (C5; Pyraz.), 143.37 (C3; Pyraz.), 138.82 (C1, Ph), 128.94 (C3,5; Ph), 123.13 (C4, Ph), 119.99 (C2,6; Ph), 110.84 (C4, Pyraz.), 45.09 (2CH2N-), 25.83 (2-CH2-), 24.75 (-CH2-), 16.21, 14.10 (2CH3; Pyraz.); Chemical Formula: C19H23N7.
Compound 5a was recrystallized from ethanol:
About 15 mL of absolute ethanol was added to compound 5a and the mixture was heated gently until the entire amount dissolved completely. Thereafter, the solution was allowed to cool at rt. overnight to afford the product its crystalline form.
N-(4-Bromophenyl)-4-(3,5-dimethyl-1H-pyrazol-1-yl)-6-(piperidin-1-yl)-1,3,5-triazin-2-amine
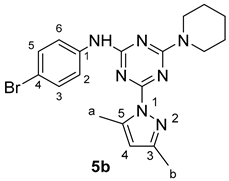
The reaction of N-(4-bromophenyl)-4-hydrazinyl-6-(piperidin-1-yl)-1,3,5-triazin-2-amine 4b (728 mg, 2.00 mmol) and acetylacetone (360 mg, 3.6 mmol) in the presence of triethylamine (243 mg, 2.40 mmol) was carried out in refluxing DMF (15 mL) while stirring overnight. Once the reaction was complete based on TLC analysis (ethyl acetate/hexane 5:5), it was cooled in an ice bath while stirring with 15 mL of ice water to afford the product 5b as a white precipitate, which was separated by filtration, washed with water, and dried under vacuum (668 mg, 1.57 mmol, 79%); m.p. 198–201 °C; IR (KBr, cm−1): 3444 (NH), 1585 (C=N), 1530–1480 (aromatic C-C); 1H-NMR (400 MHz, CDCl3) δ 7.44–7.46 (d, J = 13.3 Hz, 4H, H-2, H-3, H-5, H-6), 6.00 (s, 1H, H-4; Pyrazole), 3.81 (s, 4H, 2NCH2-), 2.65 (s, 3H, H-b), 2.31 (s, 3H, H-a), 1.71 (s, 2H, -CH2-), 1.64 (s, 4H, 2-CH2-); 13C-NMR (126 MHz, CDCl3) δ 160.70–168.00 (3C=N), 151.02 (C5; Pyraz.), 142.37 (C3; Pyraz.), 136.56 (C1, Ph), 130.78 (C3,5; Ph), 120.65 (C2,6; Ph), 114.82 (C4; Ph), 110.10 (C4; Pyraz.), 44.22 (2CH2N-), 24.73 (2-CH2-), 23.58 (-CH2-), 15.06, 13.02 (2CH3; Pyraz.); Chemical Formula: C19H22BrN7.
Compound 5b was recrystallized from DCM/Diethyl ether:
Compound 5b was dissolved in about 10 mL of DCM with gentle heating until the dissolution was complete, then a layer of diethyl ether (50 mL) was allowed to slide through the flask. The crystals of compound 5b were collected after 48 h.
Crystal structure determination
The technical experimental method for the mounting data and solved the chemical structures [30,31,32,33] are amended in the Supplementary Materials (SI). The crystallographic details are summarized in Table 1.

Table 1.
Crystal Data of compounds 5a and 5b.
Computational Study
Computational investigation software [34,35,36,37,38,39,40] employed in this study are provided in the Supplementary Materials (SI).
3. Results and Discussion
3.1. Chemistry
The target s-triazine hybridized structures with different moieties have been synthesized, as shown in Scheme 1. Insertion of the aniline derivative was carried out initially via reaction of 2,4,6-trichlorotriazine (TCT) with the substituted aniline at 0–5 °C for 2–3 h. Subsequently, the second reactive chlorine atom was replaced by the piperidine moiety in acetone-water as a solvent at 0 °C to rt. overnight. Next, the least reactive chlorine atom was replaced with hydrazine. Finally, the hydrazine derivative was cyclized with acetyl acetone to afford the last pyrazole moiety in DMF under reflux condition. The chemical features of the synthesized s-triazine having different three moieties were assigned and confirmed via NMR, IR, and a single crystal X-ray diffraction analysis technique.
3.2. Crystal Structure Description
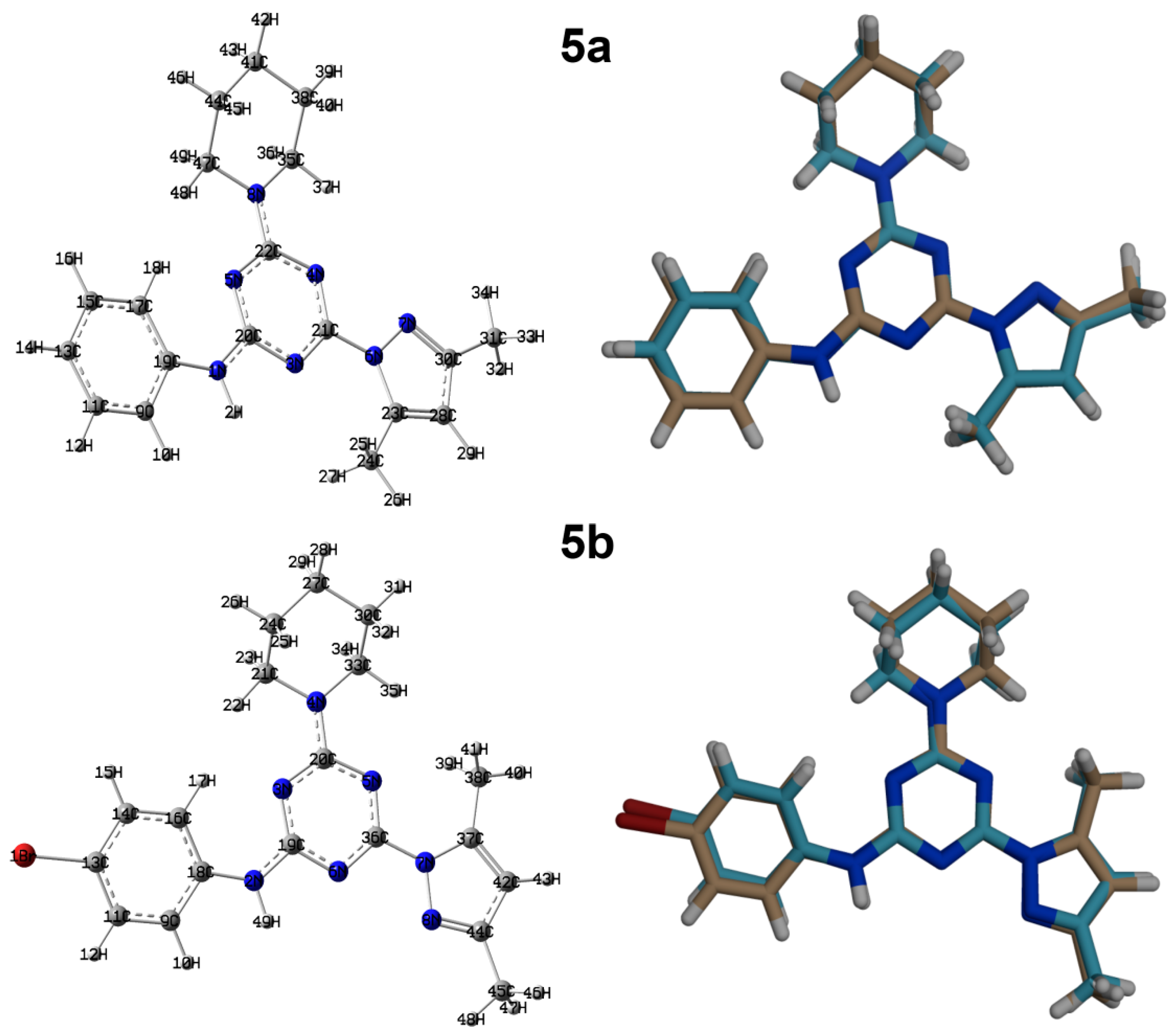
The X-ray structure of 5a showing atom numbering and thermal ellipsoids drawn at 30% probability level is shown in Figure 2A. The compound crystallized in the orthorhombic system and Pbca space group with unit cell parameters of a = 11.49940(10) Å, b = 14.91530(10) Å, and c = 21.10870(10) Å (Table 1). The unit cell volume is 3620.50(4) Å3 with Z = 8, and one molecule as asymmetric unit. The structure comprised three rings connected with the s-triazine core, which are the aminophenyl, pyrazolyl, and morpholine moieties. The morpholine moiety occurred in the chair form and the mean plane passing through the s-triazine core makes angles of 17.27 and 14.71° with the phenyl and pyrazolyl moieties, respectively. Selected bond lengths and angles are listed in Table 2.
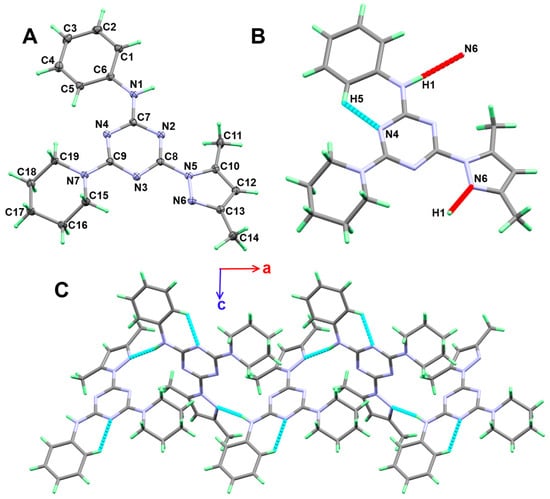
Figure 2.
(A) Atom numbering and thermal ellipsoids at 30% probability level (B) hydrogen bond contacts (C) and molecular packing along ac plane for 5a.

Table 2.
Selected bond lengths [Å] and angles [°] for 5a and 5b.
The molecular structure of 5a is stabilized by C-H…N intramolecular hydrogen bond between the one C-H bond from the phenyl moiety as hydrogen bond donor and the neighbouring nitrogen atom from the s-triazine core. The donor–acceptor distance of the C-H…N intramolecular hydrogen bond is 2.9366(13) Å. The intramolecular C-H…N and the intermolecular N-H…N hydrogen bond contacts are shown in Figure 2B as turquoise and red dotted lines, respectively. Viewing of the supramolecular structure of 5a is shown in Figure 2C, while the hydrogen bond parameters are depicted in Table 3.

Table 3.
Hydrogen bonds for 5a and 5b [Å and °].
On the other hand, compound 5b in the orthorhombic system and Pbcn space group with unit cell parameters of a = 37.4570(3) Å, b = 8.62930(10) Å, and c = 24.8053(2) Å (Table 1). The unit cell volume is 8017.76(13) Å3 with Z = 16 and two molecules as an asymmetric unit. The structure of the two molecular units of 5b is shown in Figure 3A,B. Moreover, the structure comprised three rings connected with the s-triazine core, which are the p-bromoaminophenyl, pyrazolyl, and morpholine moieties. The morpholine moiety also occurred in the chair form. In this case, the twists of the phenyl ring moiety from the mean plane passing through the s-triazine core are found more than in 5a in unit A (29.44°) and less in unit B (10.46°). The corresponding values for the pyrazolyl moiety are 8.22 and 7.17°, respectively.
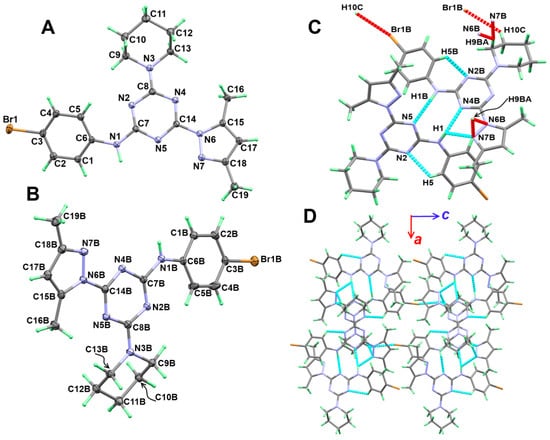
Figure 3.
(A,B) Atom numbering and thermal ellipsoids at 30% probability level, (C) hydrogen bond contacts (D) and molecular packing along ac plane for 5b.
Moreover, the molecular structure of the two units in 5b is stabilized by slightly shorter C-H…N intramolecular interactions, with a donor–acceptor distance of 2.887(2) and 2.870(2) Å for C5-H5…N2 and C5b-H5b…N2B interactions, respectively. The packing of this compound is controlled by a number of C-H…N, N-H…N and C-H…Br hydrogen bonds (Table 3 and Figure 3C). Viewing of the packing scheme of 5b is shown in Figure 3D.
3.3. Analysis of Molecular Packing
The intermolecular interactions play important role in the crystal stability. In this regard, the different contacts in the crystal structures of 5a and 5b were analysed with the aid of Hirshfeld calculations. These contacts and their percentages are summarized in Figure 4. For 5a, the structure contains three elements which are C, H, and N. Hence, the C…C, C…H, C…N, H…H, N…H and N…N are the only possible contacts. Of these interactions, only five contacts occurred, which are H…C, H…H and N…H. Their percentages are 22.2, 58.4, and 19.0%, respectively. Hence, the H…H, N…H and H…C contacts are the most dominant. Among these contacts, the N…H and H…C are the most important, based on the dnorm map shown in Figure 5. These are the only contacts which appeared as red regions, indicating shorter distances than the van der Waals (vdWs) radii sum of the interacting atoms.
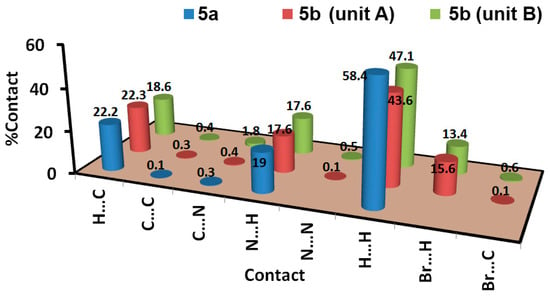
Figure 4.
The percentages of all possible intermolecular interactions in the studied systems.
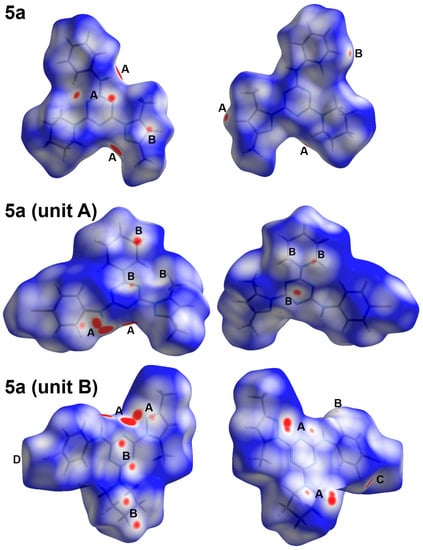
Figure 5.
The dnorm maps of the studied systems showing the most important interactions: (A) N…H, (B) H…C, (C) H…H and (D) Br…H.
Another piece of evidence which confirms the importance of these contacts is the sharp spikes that appeared in the decomposed fingerprint plots of the N…H and H…C contacts. The sharp spikes indicate significantly short interactions (Figure 6). Summary of the short N…H and H…C interactions having distances smaller than the vdWs radii sum of the interacting atoms are listed in Table 4.
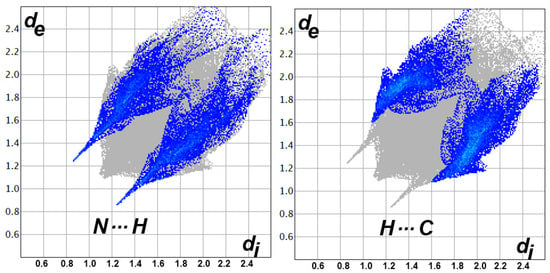
Figure 6.
Decomposed fingerprint plots for the short intermolecular interactions in 5a.

Table 4.
All short intermolecular interactions in the studied systems.
The structure of 5b comprised an extra Br atom attached at the 4-position of the phenyl ring. Hence, more intermolecular interactions were detected in this compound (Figure 4). Since there are two different molecular units per asymmetric formula, there are two sets of results for this molecular system. Based on Hirshfeld calculations, and similar to 5a, the H…H, N…H and H…C are the most dominant contacts. In addition, there is significant amount of Br…H contacts. The H12B…C8 (2.632 Å), N5…H1B (2.055 Å), H4B…H4B (1.795 Å), and Br1B…H10C (2.902 Å) are the shortest contacts, respectively. All four of these contacts appeared as a red region in the dnorm map (Figure 5). An important difference between the two molecular units of 5b is that the Br…H and H…H interactions are more important in the molecular unit with the letter B in the atom numbering than the other unit. Moreover, the percentages of the H…H, Br…H, and H…C contacts are also different in both units (Figure 7).
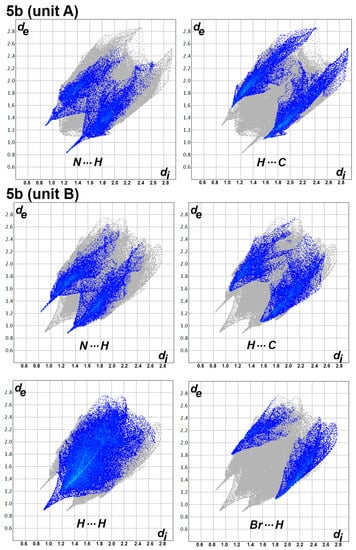
Figure 7.
Decomposed fingerprint plots for the short intermolecular interactions in 5b.
DFT studies
Optimized Geometry
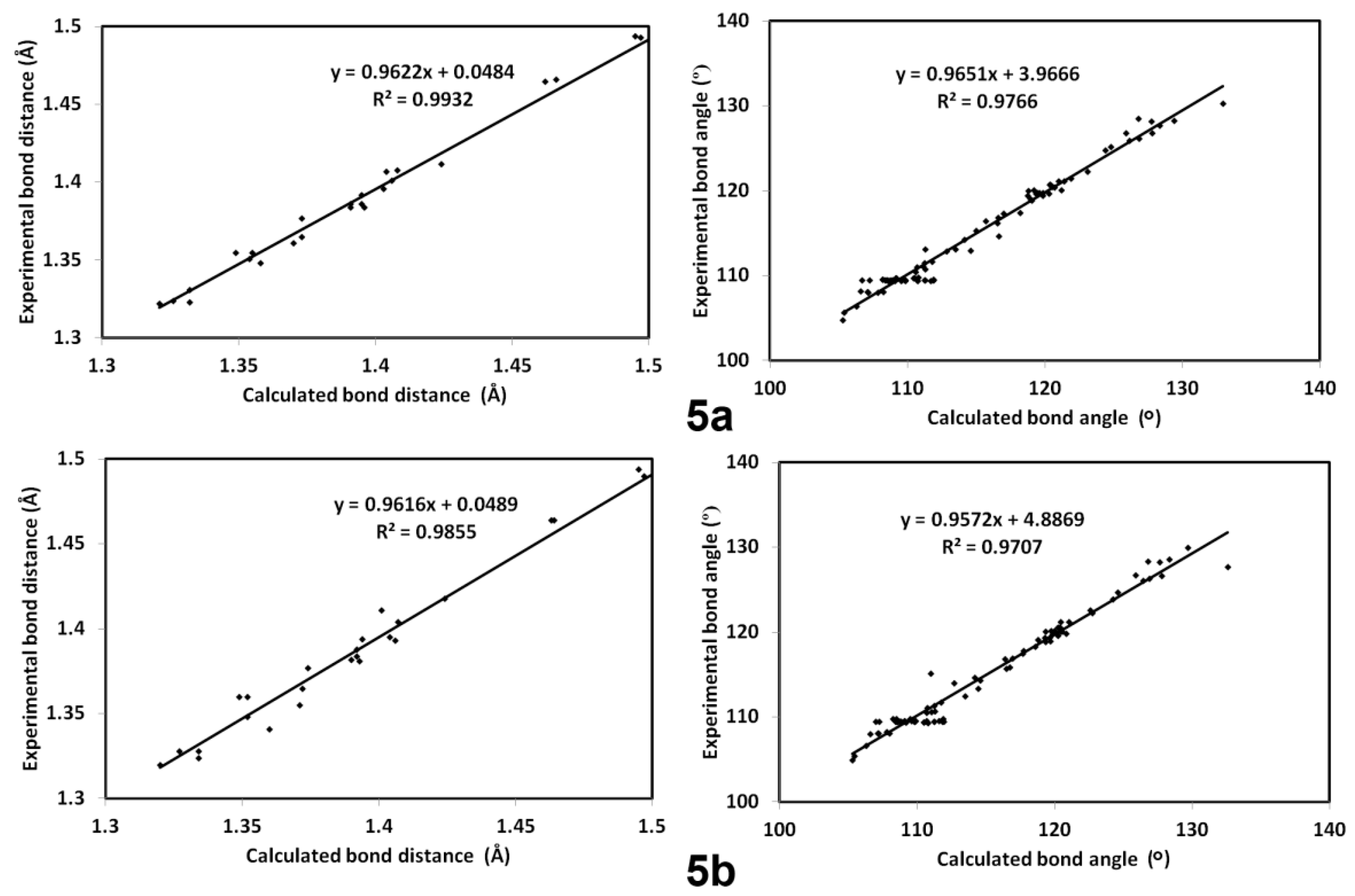
The calculated optimized structures of 5a and 5b agreed very well with the reported X-ray structures, as indicated from the excellent matching between them (Figure 8) as well as the excellent correlations between the calculated and experimental bond distances and angles (Figure 9). In addition, the differences between the calculated and experimental bond distances are found to not exceed 0.01 Å (Table S1; Supplementary data). Moreover, the correlation coefficients (R2) are calculated to be very close to unity (Figure 9). Some higher deviations occurred in the bond angles, which could be attributed to the relaxation of the molecular structure as a consequence of the geometry optimization in the absence of the crystal packing effect. Generally, the differences between the calculated and experimental bond angles did not exceed 2.8˚ (Table S1; Supplementary data).
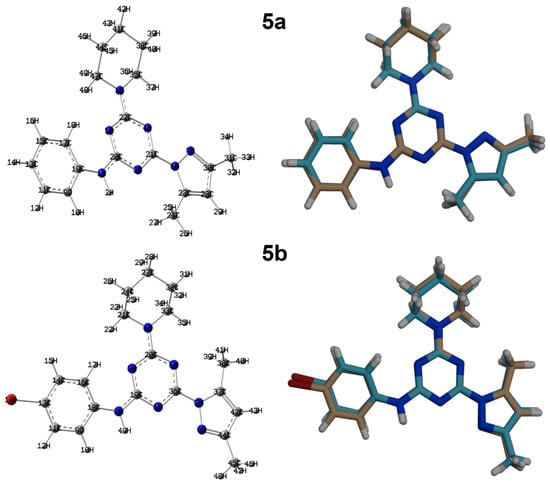
Figure 8.
Optimized structures (left) and overlay of the optimized with experimental structures (right) for the studied compounds.
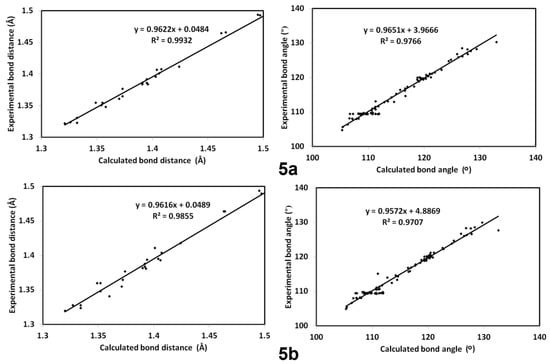
Figure 9.
The straight line correlations between the calculated and experimental geometric parameters (Table S1; Supplementary data).
Both of the studied systems comprised CHN backbone and differ only in the substituent attached to the phenyl moiety. Graphical presentations of the natural charges at the different atomic sites are shown in Figure 10. The N atoms of the triazine core and the aniline moiety are more electronegative than the N-atoms of pyrazolyl and piperidinyl moieties in both compounds (Table S2; Supplementary data). The C-atoms of the CH3 groups are the most electronegative C-sites. In contrast, all C-atoms bonded to nitrogen have high positive charge. Of C-atoms, the s-triazine C-atoms are the most electropositive, as these carbons found located between two nitrogen sites in the s-triazine moiety. Moreover, all hydrogen sites have a positive charge where the NH protons are the most positively charged. The natural charge values are calculated to be 0.434 and 0.443 e for 5a and 5b, respectively. In the right part of Figure 11, the molecular electrostatic potential (MEP) maps of both compounds are shown. The red regions are related to the atomic sites with high electron density, while the blue regions are related to the atomic sites with lowest electron density, which represents the most possible sites as hydrogen bond acceptor and donors, respectively.
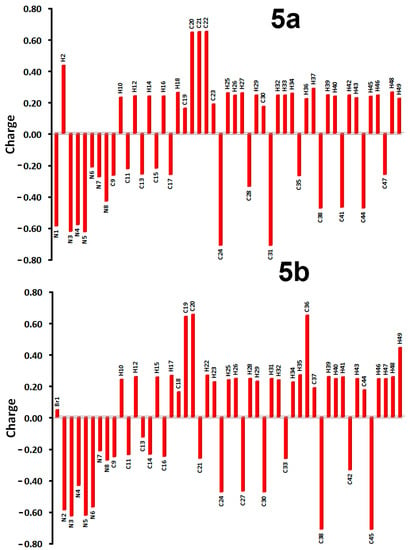
Figure 10.
Natural charge populations at the different atomic sites for 5a and 5b.
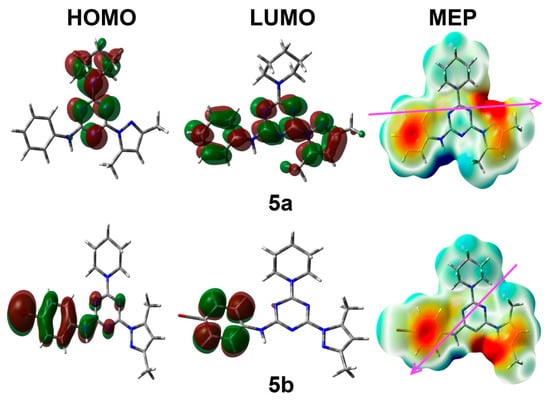
Figure 11.
The MEP, HOMO, and LUMO for the studied compounds. In MEP, the red and blue colors indicate the most negative and most positive regions, respectively.
Both compounds are polar molecules with net dipole moment of 1.018 and 4.249 Debye for 5a and 5b, respectively. The compound with bromo substituent at the phenyl ring (5b) has a higher polarity than the one with an unsubstituted phenyl ring (5a). The substituent has also a great effect on the direction of the dipole moment vector, as can be seen from Figure 11 (left part).
Moreover, the substituent at the phenyl moiety has a direct impact on the distribution of the HOMO and LUMO levels on the studied molecular systems. In case of 5a, the HOMO level is mainly located over the s-triazine core and the piperidinyl fragment, while the LUMO is distributed over the π-system of the s-triazine core, phenyl, and pyrazolyl moieties. In this case, the HOMO-LUMO energy gap is calculated to be 4.917 eV. On the other hand, the HOMO and LUMO are mainly located over the phenyl moiety in case of 5b. The HOMO has some distribution over the s-triazine core, where the HOMO-LUMO energy gap is slightly less in 5b (4.804 eV) compared to 5a.
3.4. Electronic Reactivity Parameters
The HOMO and LUMO energies are presented in Table 5. The molecular reactivity parameters such as electrophilicity index (ω = μ2/2η), electron affinity (A = −ELUMO), ionization potential (I = −EHOMO), hardness (η = (I−A)/2), as well as chemical potential (μ = − (I + A)/2) reactivity indices, were calculated based on the HOMO and LUMO energies [41,42,43,44], and the results are also listed in Table 5. The ionization potential and electron affinity values of 5b are lower than 5a. In contrast, the chemical hardness, chemical potential, and electrophilicity index have higher values for 5b than 5a.

Table 5.
Reactivity parameters in eV of the studied compounds.
3.5. NBO Analysis
The electron conjugation process has a great impact on the stability of molecular system [45,46]. In this regard, the different electron delocalization processes and their stabilization energies (E(2)) are summarized in Table 6. The σ→σ*, π→π*, n→π* and n→σ* intramolecular charge transfer (IMCT) processes stabilized the system up to 6.37, 47.40, 72.58, and 13.20 kcal/mol for the BD(1)C28-C30→ BD*(1)C23-C24, BD(2)N5-C22→BD*(2)N3-C20, LP(1)N8→BD*(2)N5-C22 and LP(1)N5→ BD*(1)N3-C20 in 5a, respectively. The corresponding values for 5b are 6.39, 44.78, 70.72, and 12.99 kcal/mol for the BD(1)C42-C44→BD*(1)C37-C38, BD(2)N5-C20→BD*(2)N6-C36, LP(1)N4→BD*(2)N5-C20, and LP(1)N6→ BD*(1)N3-C19, respectively.

Table 6.
The E(2) (kcal/mol) values for the charge transfer interactions in 5a and 5b a.
3.6. NMR Spectra
The calculated HNMR and CNMR chemical shifts by empolying GIAO method and final data are listed in Supplementary Materials in Tables S3 and S4. Figure 12 presented the correlations between computed NMR chemical shifts with experimental data. As we observed from the presented graphs, the correlation coefficients (R2) are high and close to 1. The R2 values range from 0.93 to 0.94, indicating the close agreement between the computed and experimental results.
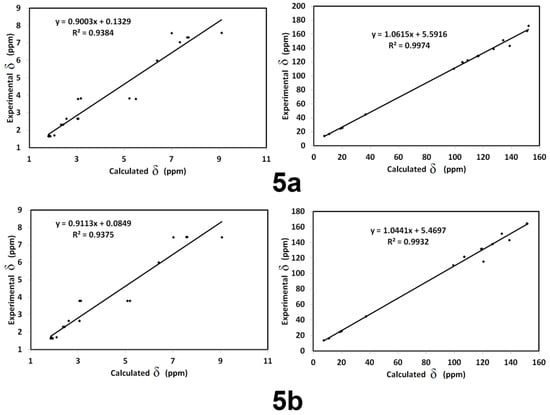
Figure 12.
1H and 13C NMR correlations between the calculated and experimental data.
4. Conclusions
Two new hybrids tethered pyrazole/piperidine/aniline moieties-based s-triazine privileged structure were synthesized in a high chemical yield using a very simple and efficient method. The supramolecular structures of 5a and 5b were quantitatively determined using Hirshfeld analysis. The calculated structures using DFT B3LYP/6–31G(d,p) agreed very well with the experimental X-ray geometries. Moreover, calculated NMR spectra are found in very good correlations with the experimental data. These compounds well be further explored by our groups with other derivatives in the near future for their biological applications.
Supplementary Materials
The following are available online at https://www.mdpi.com/article/10.3390/cryst11121500/s1, Crystal structure determination; protocol and sofwares utilized in this study for computational investigation; Figures S1–S12: 1HNMR, 13CNMR, and IR spectrum for the synthezied compounds; Table S1–S4: Computational study investigation.
Author Contributions
Conceptualization, A.E.-F. and A.B.; methodology, I.S.; software, S.M.S. and M.H.; formal analysis, I.S. and M.H.; investigation, I.S.; resources, A.E.-F. and A.B.; data curation, I.S., S.M.S., M.H. and A.A.; writing—original draft preparation, S.M.S., M.H. and A.B.; writing—review and editing, S.M.S., A.B. and A.E.-F.; visualization, A.B., and A.E.-F.; supervision, A.A., A.B. and A.E.-F.; project administration, A.E.-F. and A.B.; funding acquisition, A.B. All authors have read and agreed to the published version of the manuscript.
Funding
Researchers Supporting Project number (RSP-2021/64), King Saud University, Riyadh, Saudi Arabia.
Acknowledgments
The authors would like to extend their sincere appreciation to the Researchers Supporting Project (RSP-2021/64), King Saud University, Riyadh, Saudi Arabia.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Barakat, A.; El-Faham, A.; Haukka, M.; Al-Majid, A.M.; Soliman, S.M. s-Triazine pincer ligands: Synthesis of their metal complexes, coordination behavior, and applications. Appl. Organomet. Chem. 2021, 35, e6317. [Google Scholar] [CrossRef]
- Dugan, J.; Pollyea, D. Enasidenib for the treatment of acute myeloid leukemia. Expert Rev. Clin. Pharmacol. 2018, 11, 755–760. [Google Scholar] [CrossRef]
- Kim, E.S. Enasidenib: First global approval. Drugs 2017, 77, 1705–1711. [Google Scholar] [CrossRef] [PubMed]
- Singh, U.; Bhat, H.; Gahtori, P. Antifungal activity, SAR and physicochemical correlation of some thiazole-1, 3, 5-triazine derivatives. J. Mycol. Med. 2012, 22, 134–141. [Google Scholar] [CrossRef]
- Shanmugam, M.; Narayanan, K.; Chidambaranathan, V.; Kabilan, S. Synthesis, spectral characterization and antimicrobial studies of novel s-triazine derivatives. Spectrochim. Acta Mol. Biomol. Spectrosc. 2013, 105, 383–390. [Google Scholar] [CrossRef] [PubMed]
- Shah, D.R.; Modh, R.P.; Chikhalia, K.H. Privileged s-triazines: Structure and pharmacological applications. Future Med. Chem. 2014, 6, 463–477. [Google Scholar]
- Shanmugakala, R.; Tharmaraj, P.; Sheela, C.; Chidambaranathan, N. Transition metal complexes of s-triazine derivative: New class of anticonvulsant, antiinflammatory, and neuroprotective agents. Med. Chem. Res. 2014, 23, 329–342. [Google Scholar] [CrossRef]
- Menicagli, R.; Samaritani, S.; Signore, G.; Vaglini, F.; Dalla Via, L. In vitro cytotoxic activities of 2-Alkyl-4,6-diheteroalkyl-1,3,5-triazines: New molecules in anticancer research. J. Med. Chem. 2004, 47, 4649–4652. [Google Scholar] [CrossRef]
- Viira, B.; Selyutina, A.; García-Sosa, A.T.; Karonen, M.; Sinkkonen, J.; Merits, A.; Maran, U. Design, discovery, modelling, synthesis, and biological evaluation of novel and small, low toxicity s-triazine derivatives as HIV-1 non-nucleoside reverse transcriptase inhibitors. Biorg. Med. Chem. 2016, 24, 2519–2529. [Google Scholar] [CrossRef] [PubMed]
- El-Gazzar, A.-R.B.; Hafez, H.N. Synthesis of 4-substituted pyrido [2, 3-d] pyrimidin-4 (1H)-one as analgesic and anti-inflammatory agents. Bioorg. Med. Chem. Lett. 2009, 19, 3392–3397. [Google Scholar] [CrossRef]
- Behki, R.; Topp, E.; Dick, W.; Germon, P. Metabolism of the herbicide atrazine by Rhodococcus strains. Appl. Environ. Microbiol. 1993, 59, 1955–1959. [Google Scholar] [CrossRef] [PubMed]
- Sharma, A.; Ghabbour, H.; Khan, S.T.; Beatriz, G.; Albericio, F.; El-Faham, A. Novel pyrazolyl-s-triazine derivatives, molecular structure and antimicrobial activity. J. Mol. Struct. 2017, 1145, 244–253. [Google Scholar] [CrossRef]
- Mikhaylichenko, S.N.; Patel, S.M.; Dalili, S.; Chesnyuk, A.A.; Zaplishny, V.N. Synthesis and structure of new 1, 3, 5-triazine-pyrazole derivatives. Tetrahedron Lett. 2009, 50, 2505–2508. [Google Scholar] [CrossRef]
- Lasri, J.; Haukka, M.; Al-Rasheed, H.H.; Abutaha, N.; El-Faham, A.; Soliman, S.M. Synthesis, Structure and In Vitro Anticancer Activity of Pd(II) Complex of Pyrazolyl-s-Triazine Ligand; A New Example of Metal-Mediated Hydrolysis of s-Triazine Pincer Ligand. Crystals 2021, 11, 119. [Google Scholar] [CrossRef]
- Pathak, P.; Shukla, P.K.; Kumar, V.; Kumar, A.; Verma, A. Quinazoline clubbed 1,3,5-triazine derivatives as VEGFR2 kinase inhibitors: Design, synthesis, docking, in vitro cytotoxicity and in ovo antiangiogenic activity. Inflammopharmacology 2018, 26, 1441–1453. [Google Scholar] [CrossRef]
- Pathak, P.; Rimac, H.; Grishina, M.; Verma, A.; Potemkin, V. Hybrid Quinazoline 1, 3, 5-Triazines as Epidermal Growth Factor Receptor (EGFR) Inhibitors with Anticancer Activity: Design, Synthesis, and Computational Study. ChemMedChem 2021, 16, 822–838. [Google Scholar] [CrossRef]
- Osman, S.M.; Alasmary, F.A.; Kenawy, E.R.; Aly, E.S.A.; Khattab, S.N.; El-Faham, A. Synthesis, characterization and comparative thermal degradation kinetics of s-Triazine based polymers. J. Polym. Res. 2021, 28, 1–12. [Google Scholar] [CrossRef]
- Liao, J.H.; Lai, C.Y.; Yang, C.L. Synthesis, Characterization and Photoluminescence of Lanthanide Metal-organic Frameworks, Constructed from Triangular 4, 4′, 4 ″-s-triazine-1, 3, 5-triyl-p-aminobenzoate Ligands. J. Chin. Chem. Soc. 2014, 61, 1115–1120. [Google Scholar] [CrossRef]
- Gomes, R.; Bhaumik, A. A new triazine functionalized luminescent covalent organic framework for nitroaromatic sensing and CO2 storage. RSC Adv. 2016, 6, 28047–28054. [Google Scholar] [CrossRef]
- Liu, A.; Zhang, J.; Lv, X. Novel hydrazine-bridged covalent triazine polymer for CO2 capture and catalytic conversion. Chinese J. Catal. 2018, 39, 1320–1328. [Google Scholar] [CrossRef]
- Wu, M.-X.; Yang, Y.-W. Applications of covalent organic frameworks (COFs): From gas storage and separation to drug delivery. Chin Chem Lett. 2017, 28, 1135–1143. [Google Scholar] [CrossRef]
- Barakat, A.; El-Senduny, F.F.; Almarhoon, Z.; Al-Rasheed, H.H.; Badria, F.A.; Al-Majid, A.M.; Ghabbour, H.A.; El-Faham, A. Synthesis, X-ray crystal structures, and preliminary antiproliferative activities of new s-triazine-hydroxybenzylidene hydrazone derivatives. J. Chem. 2019, 2019. [Google Scholar] [CrossRef]
- Barakat, A.; Soliman, S.M.; El-Faham, A.; Ali, M.; Al-Majid, A.M.; Yousuf, S.; Choudhary, M.I. Three multi-components reaction: Synthesis and X-ray single-crystal of hydroacridinone-based hydrazino-s-triazine derivative as a new class of urease inhibitor. Crystals. 2020, 10, 14. [Google Scholar] [CrossRef]
- Soliman, S.M.; Lasri, J.; Haukka, M.; Elmarghany, A.; Al-Majid, A.M.; El-Faham, A.; Barakat, A. Synthesis, X-ray structure, Hirshfeld analysis, and DFT studies of a new Pd (II) complex with an anionic s-triazine NNO donor ligand. J. Mol. Struct. 2020, 1217, 128463. [Google Scholar] [CrossRef]
- Farooq, M.; Sharma, A.; Almarhoon, Z.; Al-Dhfyan, A.; El-Faham, A.; Abu Taha, N.; Wadaan, M.A.M.; de la Torre, B.G.; Albericio, F. Design and synthesis of mono-and di-pyrazolyl-s-triazine derivatives, their anticancer profile in human cancer cell lines, and in vivo toxicity in zebrafish embryos. Bioorg. Chem. 2019, 87, 457–464. [Google Scholar] [CrossRef]
- Sanders, M.A.; Brahemi, G.; Nangia-Makker, P.; Balan, V.; Morelli, M.; Kothayer, H.; Westwell, A.D.; Shekhar, M.P. Novel inhibitors of Rad6 ubiquitin conjugating enzyme: Design, synthesis, identification, and functional characterization. Mol. Cancer Ther. 2013, 12, 373–383. [Google Scholar] [CrossRef] [PubMed]
- Balaha, M.F.; El-Hamamsy, M.H.; El-Din, N.A.; El-Mahdy, N.A. Synthesis, evaluation and docking study of 1, 3, 5-triazine derivatives as cytotoxic agents against lung cancer. J. Appl. Pharm. Sci. 2016, 6, 28–45. [Google Scholar] [CrossRef]
- Raghu, M.S.; Kumar, C.P.; Prashanth, M.K.; Kumar, K.Y.; Prathibha, B.S.; Kanthimathi, G.; Alissa, S.A.; Alghulikah, H.A.; Osman, S.M. Novel 1, 3, 5-triazine-based pyrazole derivatives as potential antitumor agents and EFGR kinase inhibitors: Synthesis, cytotoxicity, DNA binding, molecular docking and DFT studies. New. J. Chem. 2021, 45, 13909–13924. [Google Scholar] [CrossRef]
- Srivastava, J.K.; Pillai, G.G.; Bhat, H.R.; Verma, A.; Singh, U.P. Design and discovery of novel monastrol-1, 3, 5-triazines as potent anti-breast cancer agent via attenuating epidermal growth factor receptor tyrosine kinase. Sci. Rep. 2017, 7, 1–18. [Google Scholar] [CrossRef]
- Rikagu Oxford Diffraction. CrysAlisPro; Agilent Technologies Inc.: Yarnton, UK, 2020. [Google Scholar]
- Sheldrick, G.M. SHELXT—Integrated space-group and crystal-structure determination. Acta Cryst. 2015, A71, 3–8. [Google Scholar] [CrossRef]
- Sheldrick, G.M. Crystal structure refinement with SHELXL. Acta Cryst. 2015, C71, 3–8. [Google Scholar]
- Hübschle, C.B.; Sheldrick, G.M.; Dittrich, B. ShelXle: A Qt graphical user interface for SHELXL. B. J. Appl. Cryst. 2011, 44, 1281–1284. [Google Scholar] [CrossRef] [PubMed]
- Turner, M.J.; McKinnon, J.J.; Wolff, S.K.; Grimwood, D.J.; Spackman, P.R.; Jayatilaka, D.; Spackman, M.A. Crystal Explorer17; University of Western Australia: 2017. Available online: http://hirshfeldsurface.net (accessed on 17 July 2019).
- Frisch, M.J.; Trucks, G.W.; Schlegel, H.B.; Scuseria, G.E.; Robb, M.A.; Cheeseman, J.R.; Scalmani, G.; Barone, V.; Mennucci, B.; Petersson, G.A.; et al. GAUSSIAN 09; Revision A02; Gaussian Inc.: Wallingford, CT, USA, 2009. [Google Scholar]
- GaussView; Version 4.1; Denningto, R., II, Keith, T., Millam, J., Eds.; Semichem Inc.: Shawnee Mission, KS, USA, 2007. [Google Scholar]
- Reed, A.E.; Curtiss, L.A.; Weinhold, F. Intermolecular interactions from a natural bond orbital, donor-acceptor viewpoint. Chem. Rev. 1988, 88, 899–926. [Google Scholar] [CrossRef]
- Cheeseman, J.R.; Trucks, G.W.; Keith, T.A.; Frisch, M.J. A Comparison of Models for Calculating Nuclear Magnetic Resonance Shielding Tensors. J. Chem. Phys. 1996, 104, 5497–5509. [Google Scholar] [CrossRef]
- Marten, B.; Kim, K.; Cortis, C.; Friesner, R.A.; Murphy, R.B.; Ringnalda, M.N.; Sitkoff, D.; Honig, B. New Model for Calculation of Solvation Free Energies: Correction of Self-Consistent Reaction Field Continuum Dielectric Theory for Short-Range Hydrogen-Bonding Effects. J. Phys. Chem. 1996, 100, 11775–11788. [Google Scholar] [CrossRef]
- Tannor, D.J.; Marten, B.; Murphy, R.; Friesner, R.A.; Sitkoff, D.; Nicholls, A.; Ringnalda, M.; Goddard, W.A.; Honig, B. Accurate first principles calculation of molecular charge distributions and solvation energies from ab initio quantum mechanics and continuum dielectric theory. J. Am. Chem. Soc. 1994, 116, 11875–11882. [Google Scholar] [CrossRef]
- Foresman, J.B.; Frisch, Æ. Exploring Chemistry with Electronic Structure Methods, 2nd ed.; Gaussian: Pittsburgh, PA, USA, 1996. [Google Scholar]
- Chang, R. Chemistry, 7th ed.; McGraw-Hill: New York, NY, USA, 2001. [Google Scholar]
- Parr, R.G.; Yang, W. Density-Functional Theory of Atoms and Molecules; Oxford University Press: New York, NY, USA, 1989. [Google Scholar]
- Parr, R.G.; Szentpaly, L.V.; Liu, S. Electrophilicity index. J. Am. Chem. Soc. 1999, 121, 1922–1924. [Google Scholar] [CrossRef]
- Hubert Joe, I.; Kostova, I.; Ravikumar, C.; Amalanathan, M.; Pinzaru, S.C. Theoretical and vibrational spectral investigation of sodium salt of acenocoumarol. J. Raman Spectrosc. 2009, 40, 1033–1038. [Google Scholar]
- Sebastian, S.; Sundaraganesan, N.; Spectrochim. The spectroscopic (FT-IR, FT-IR gas phase, FT-Raman and UV) and NBO analysis of 4-Hydroxypiperidine by density functional method. Acta Part A Mol. Biomol. Spectrosc. 2010, 75, 941–952. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).