Synthesis and Characterization of Crown-Ether Appended Hexaazatrinaphthylene-Based Liquid-Crystalline Derivative
Abstract
1. Introduction
2. Materials and Methods
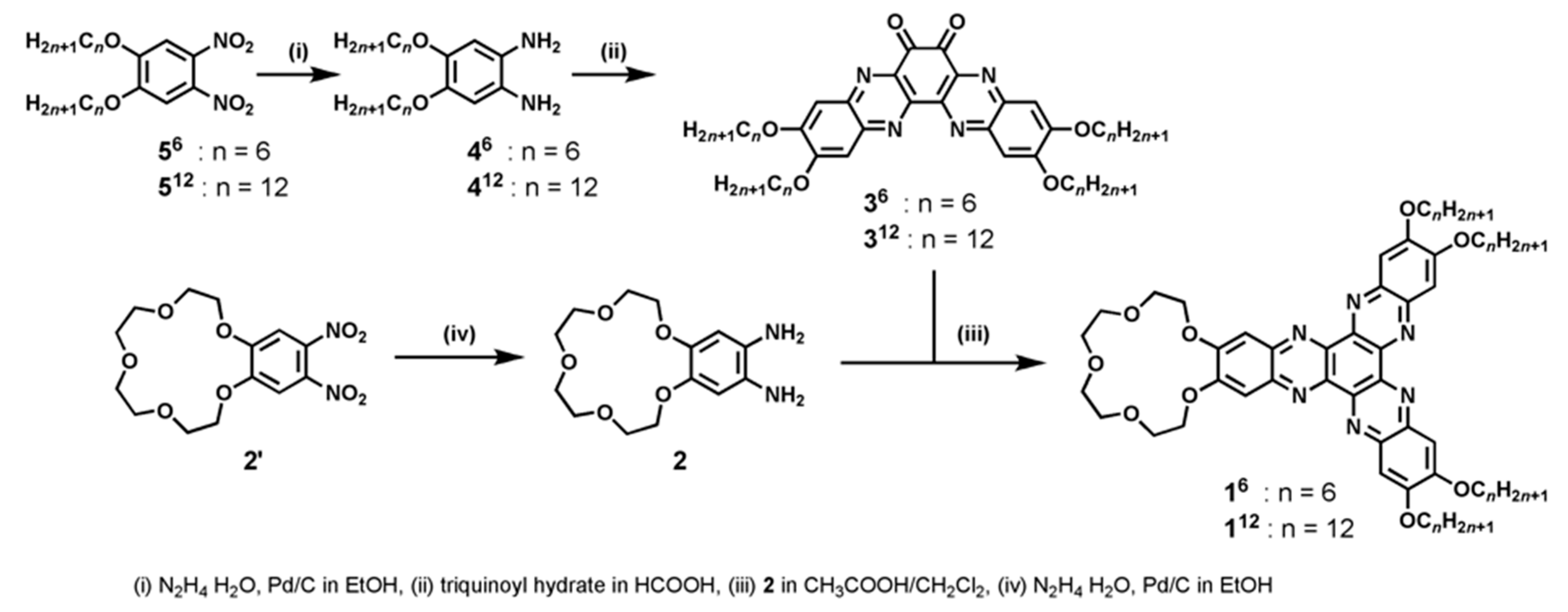
2.1. Synthesis of 112
2.2. Synthesis of 16
3. Results and Discussion
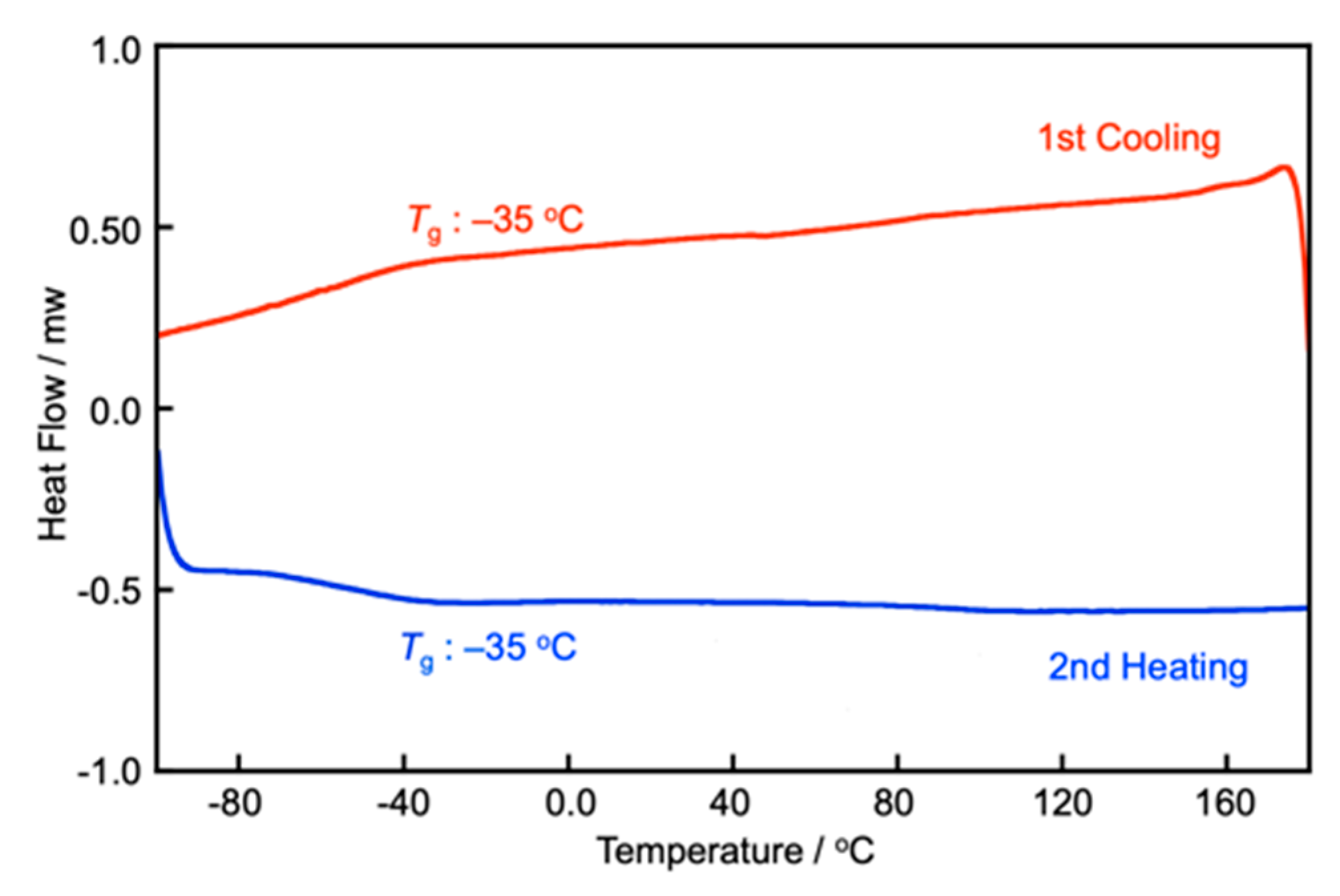
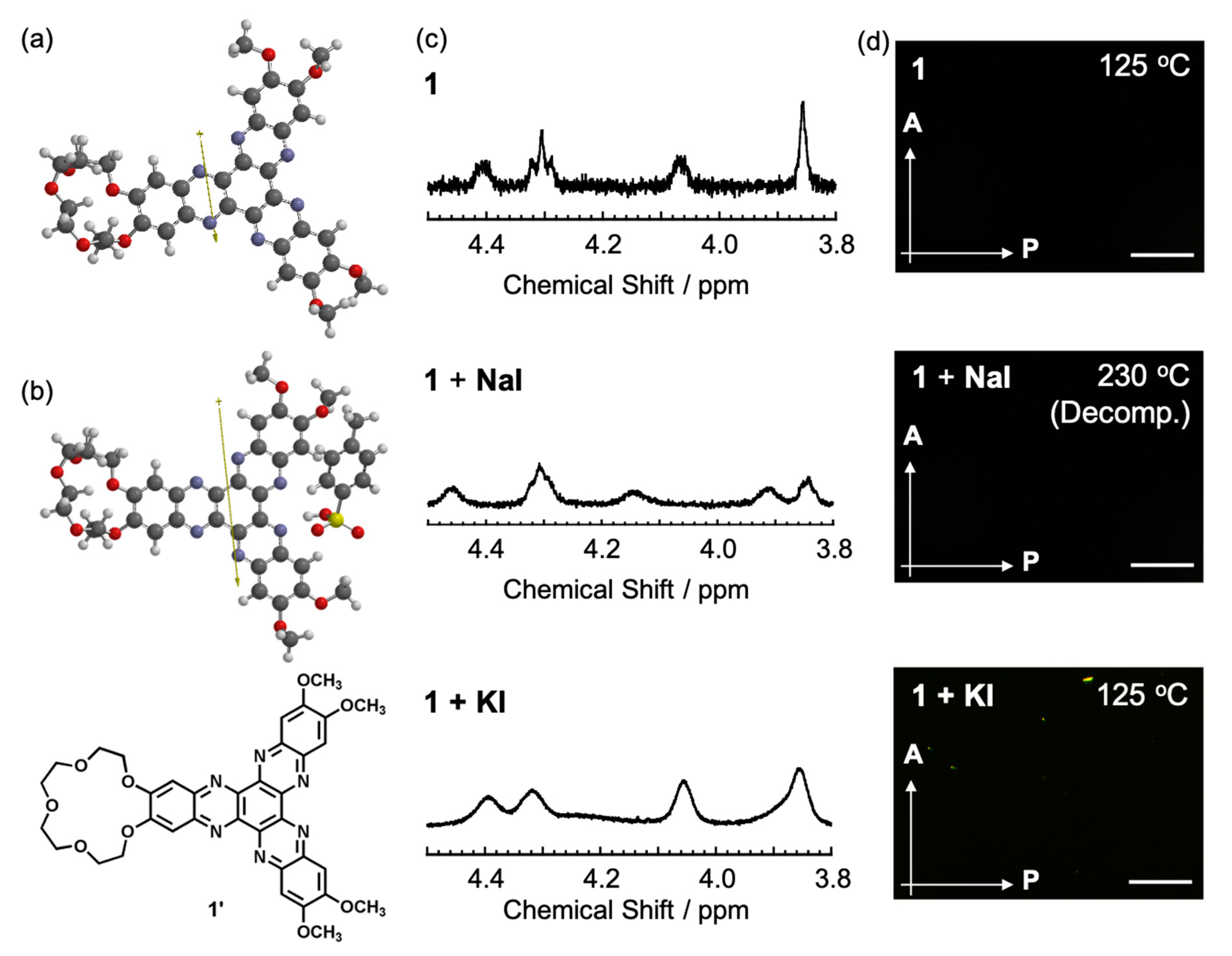
3.1. Synthesis and Characterization of Compounds 16 and 112
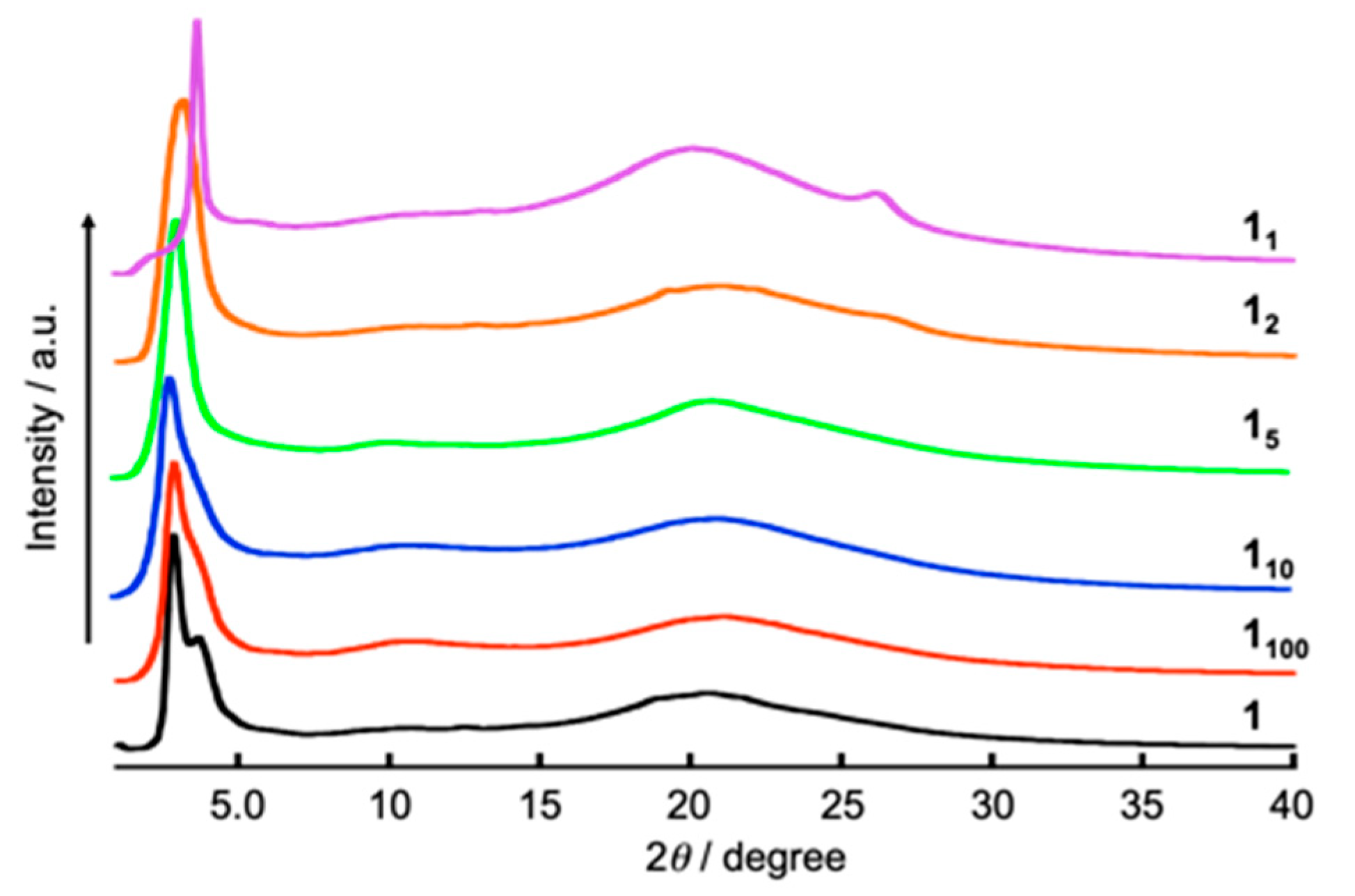
3.2. Influence on Self-Assembled Liquid-Crystalline Structures of 112 by Addition of BSA and Metal Ions
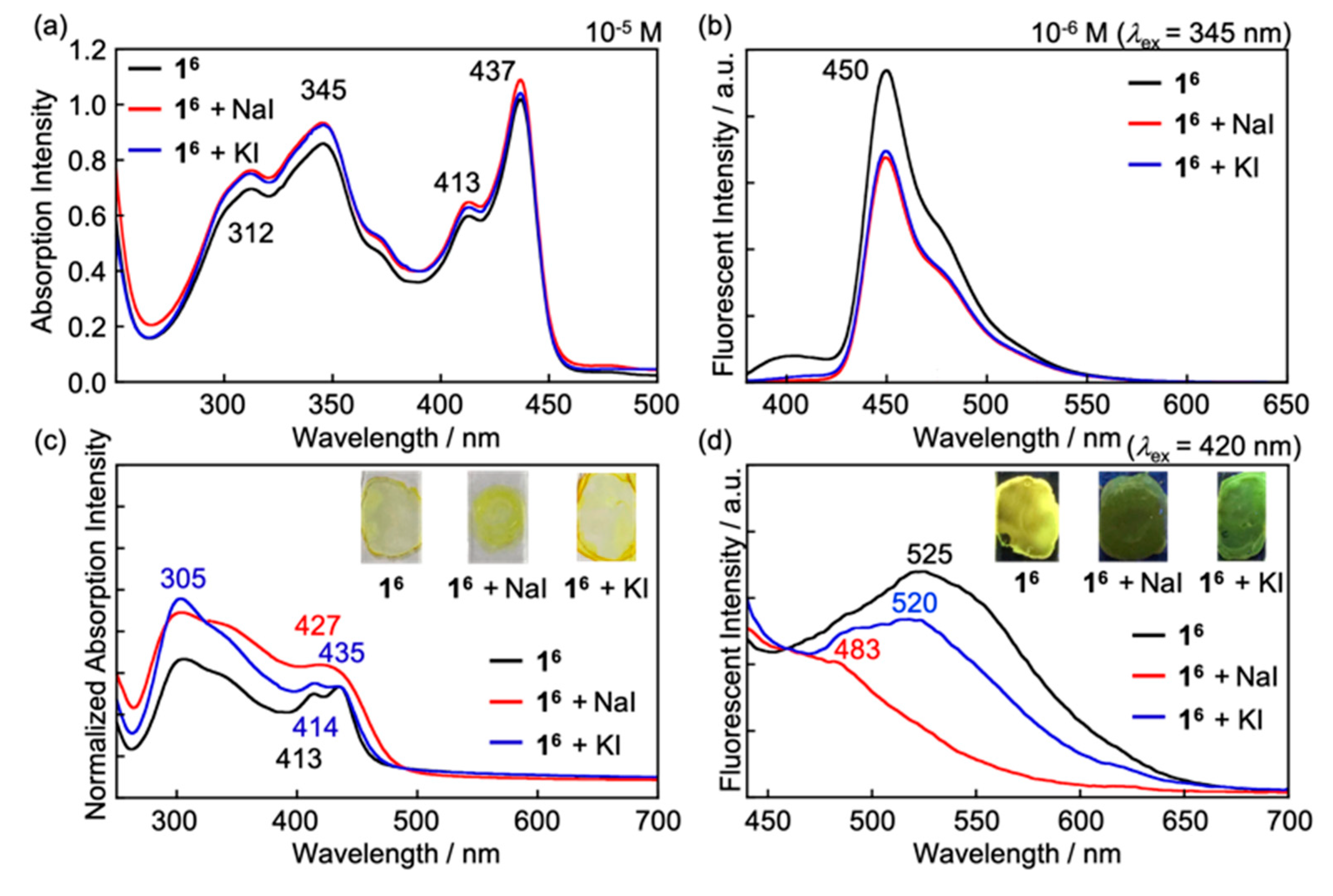
3.3. Influence on Electronic Properties of 16 by Addition of Metal Ions
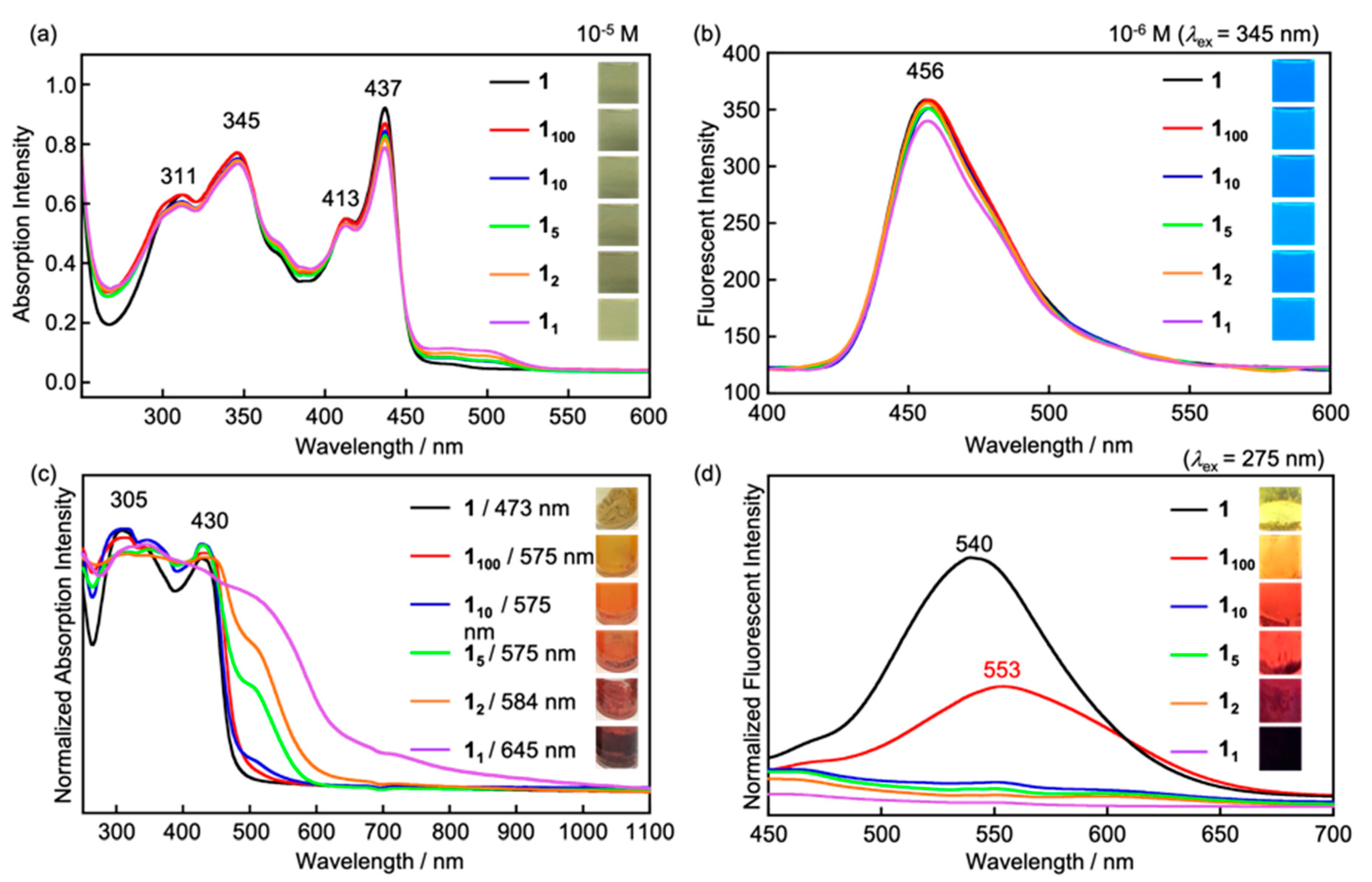
3.4. Influence on Electronic Properties of 112 by Addition of BSA
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Adam, D.; Schuhmacher, P.; Simmerer, J.; Häussling, L.; Siemensmeyer, K.; Etzbachi, K.H.; Ringsdorf, H.; Haarer, D. Fast photoconduction in the highly ordered columnar phase of a discotic liquid crystal. Nature 1994, 371, 141–143. [Google Scholar] [CrossRef]
- Kumar, M. Self-organization of disc-like molecules: Chemical aspects. Chem. Soc. Rev. 2006, 35, 83–109. [Google Scholar] [CrossRef] [PubMed]
- Laschat, S.; Baro, A.; Steinke, N.; Giesselmann, F.; Hägele, C.; Scalia, G.; Judele, R.; Kapatsina, E.; Sauer, S.; Schreivogel, A.; et al. Discotic liquid crystals: From tailor-made synthesis to plastic electronics. Angew. Chem. Int. Ed. 2007, 46, 4832–4887. [Google Scholar] [CrossRef]
- Pisula, W.; Kastler, M.; Wasserfallen, D.; Mondeshki, M.; Piris, J.; Schnell, I.; Müllen, K. Relation between supramolecular order and charge carrier mobility of branched alkyl hexa-peri-hexabenzocoronenes. Chem. Mater. 2006, 18, 3634–3640. [Google Scholar] [CrossRef]
- Sergeyev, S.; Pisula, W.; Geerts, Y.H. Discotic liquid crystals: A new generation of organic semiconductors. Chem. Soc. Rev. 2007, 36, 1902. [Google Scholar] [CrossRef]
- Lemaur, V.; Filho, D.A.D.S.; Coropceanu, V.; Lehmann, M.; Geerts, Y.; Piris, J.; Debije, M.G.; van de Craats, A.M.; Senthilkumar, K.; Siebbeles, L.D.A.; et al. Charge transport properties in discotic liquid crystals: A quantum-chemical insight into structure−property relationships. J. Am. Chem. Soc. 2004, 126, 3271–3279. [Google Scholar] [CrossRef]
- Crispin, X.; Cornil, J.; Friedlein, R.; Okudaira, K.K.; Lemaur, V.; Crispin, A.; Kestemont, G.; Lehmann, M.; Fahlman, M.; Lazzaroni, R.; et al. Electronic Delocalization in discotic liquid crystals: A joint experimental and theoretical study. J. Am. Chem. Soc. 2004, 126, 11889–11899. [Google Scholar] [CrossRef]
- Lehmann, M.; Kestemont, G.; Aspe, R.G.; Buess-Herman, C.; Koch, M.H.J.; Debije, M.G.; Piris, J.; de Haas, M.P.; Warman, J.M.; Watson, M.D.; et al. High charge-carrier mobility in π-deficient discotic mesogens: Design and structure-property relationship. Chem. Eur. J. 2005, 11, 3349–3362. [Google Scholar] [CrossRef]
- Roussel, O.; Kestemont, G.; Tant, J.; De Halleux, V.; Aspe, R.G.; Levin, J.; Remacle, A.; Gearba, I.R.; Ivanov, D.A.; Lehmann, M.; et al. Discotic liquid crystals as electron carrier materials. Mol. Cryst. Liq. Cryst. 2003, 396, 35–39. [Google Scholar] [CrossRef]
- Ong, C.W.; Liao, S.-C.; Chang, T.H.; Hsu, H.-F. In situ synthesis of Hexakis(alkoxy)diquinoxalino[2,3-a:2’,3’-c]phenazines: Mesogenic phase transition of the electron-deficient discotic compounds. J. Org. Chem. 2004, 69, 3181–3185. [Google Scholar] [CrossRef]
- Yeh, M.-C.; Liao, S.-C.; Chao, S.-H.; Ong, C.W. Synthesis of polyphilic hexaazatrinaphthylenes and mesomorphic properties. Tetrahedron 2010, 66, 8888–8892. [Google Scholar] [CrossRef]
- Kaafarani, B.R.; Kondo, T.; Yu, J.; Zhang, Q.; Dattilo, D.; Risko, C.; Jones, S.C.; Barlow, S.; Domercq, B.; Amy, F.; et al. High charge-carrier mobility in an amorphous hexaazatrinaphthylene derivative. J. Am. Chem. Soc. 2005, 127, 16358–16359. [Google Scholar] [CrossRef] [PubMed]
- Budd, P.M.; Ghanem, B.; Msayib, K.; McKeown, N.; Tattershall, C. A nanoporous network polymer derived from hexaazatrinaphthylene with potential as an adsorbent and catalyst support. J. Mater. Chem. 2003, 13, 2721–2726. [Google Scholar] [CrossRef]
- McKeown, N.; Gahnem, B.; Msayib, K.J.; Budd, P.M.; Tattershall, C.E.; Mahmood, K.; Tan, S.; Book, D.; Langmi, H.; Walton, A. Towards polymer-based hydrogen storage materials: Engineering ultramicroporous cavities within polymers of intrinsic microporosity. Angew. Chem. Int. Ed. 2006, 45, 1804–1807. [Google Scholar] [CrossRef] [PubMed]
- Xu, F.; Chen, X.; Tang, Z.; Wu, D.; Fu, R.; Jiang, D. Redox-active conjugated microporous polymers: A new organic platform for highly efficient energy storage. Chem. Commun. 2014, 50, 4788–4790. [Google Scholar] [CrossRef]
- Xiao, R.; Tobin, J.M.; Zha, M.; Hou, Y.-L.; He, J.; Vilela, F.; Xu, Z. A nanoporous graphene analog for superfast heavy metal removal and continuous-flow visible-light photoredox catalysis. J. Mater. Chem. A 2017, 5, 20180–20187. [Google Scholar] [CrossRef]
- Hisaki, I.; Suzuki, Y.; Gomez, E.; Ji, Q.; Tohnai, N.; Nakamura, T.; Douhal, A. Acid responsive hydrogen-bonded organic frameworks. J. Am. Chem. Soc. 2019, 141, 2111–2121. [Google Scholar] [CrossRef]
- Bradshaw, J.S.; Izatt, R. Crown ethers: The search for selective ion ligating agents. Acc. Chem. Res. 1997, 30, 338–345. [Google Scholar] [CrossRef]
- Isoda, K. Acid-responsive N-heteroacene-based material showing multi-emission colors. ChemistryOpen 2017, 6, 242–246. [Google Scholar] [CrossRef]
- Isoda, K.; Sato, Y.; Matsukuma, D. Fluorescent N-heteroacene-based π-conjugated liquid responsive to HCl vapor. ChemistrySelect 2017, 2, 7222–7226. [Google Scholar] [CrossRef]
- Sato, Y.; Mutoh, Y.; Matsukuma, D.; Nakagawa, M.; Kawai, T.; Isoda, K. Tuning the electronic properties and acid-response behavior of N-heteroacene-based π-conjugated liquids by changing the number of π-conjugated substituents. Chem. Asian J. 2018, 13, 2619–2625. [Google Scholar] [CrossRef] [PubMed]
- Isoda, K.; Ikenaga, A. Synthesis of furan-substituted N-heteroacene-based liquid material and its acid-recognizing behavior. Crystals 2019, 9, 51. [Google Scholar] [CrossRef]
- Isoda, K.; Ishiyama, T.; Mutoh, Y.; Matsukuma, D. Stimuli-responsive room-temperature N-heteroacene liquid: In situ observation of the self-assembling process and its multiple properties. ACS Appl. Mater. Interfaces 2019, 11, 12053–12062. [Google Scholar] [CrossRef] [PubMed]
- Isoda, K.; Matsubara, M.; Ikenaga, A.; Akiyama, Y.; Mutoh, Y. Reversibly/irreversibly stimuli-responsive inks based on N-heteroacene liquids. J. Mater. Chem. C 2019, 7, 14075–14079. [Google Scholar] [CrossRef]
- Hariharan, P.; Pople, J. The effect of d-functions on molecular orbital energies for hydrocarbons. Chem. Phys. Lett. 1972, 16, 217–219. [Google Scholar] [CrossRef]
- Hariharan, P.C.; Pople, J.A. The influence of polarization functions on molecular orbital hydrogenation energies. Theor. Chim. Acta 1973, 28, 213–222. [Google Scholar] [CrossRef]
- Lee, C.; Yang, W.; Parr, R.G. Development of the Colle-Salvetti correlation-energy formula into a functional of the electron density. Phys. Rev. B 1988, 37, 785–789. [Google Scholar] [CrossRef]
- Isoda, K.; Takahashi, H.; Mutoh, Y.; Hoshino, N.; Akutagawa, T.; Hoshino, T. One-dimensional single-helix coordination polymer self-assembled by a crown-ether appended-N-heteroacene radical anion. Dalton Trans. 2019, 48, 13125–13129. [Google Scholar] [CrossRef]
- Isoda, K.; Abe, T.; Tadokoro, M. Room-temperature redox-active liquid crystals composed of tetraazanaphthacene derivatives. Chem. Asian J. 2013, 8, 2951–2954. [Google Scholar] [CrossRef]


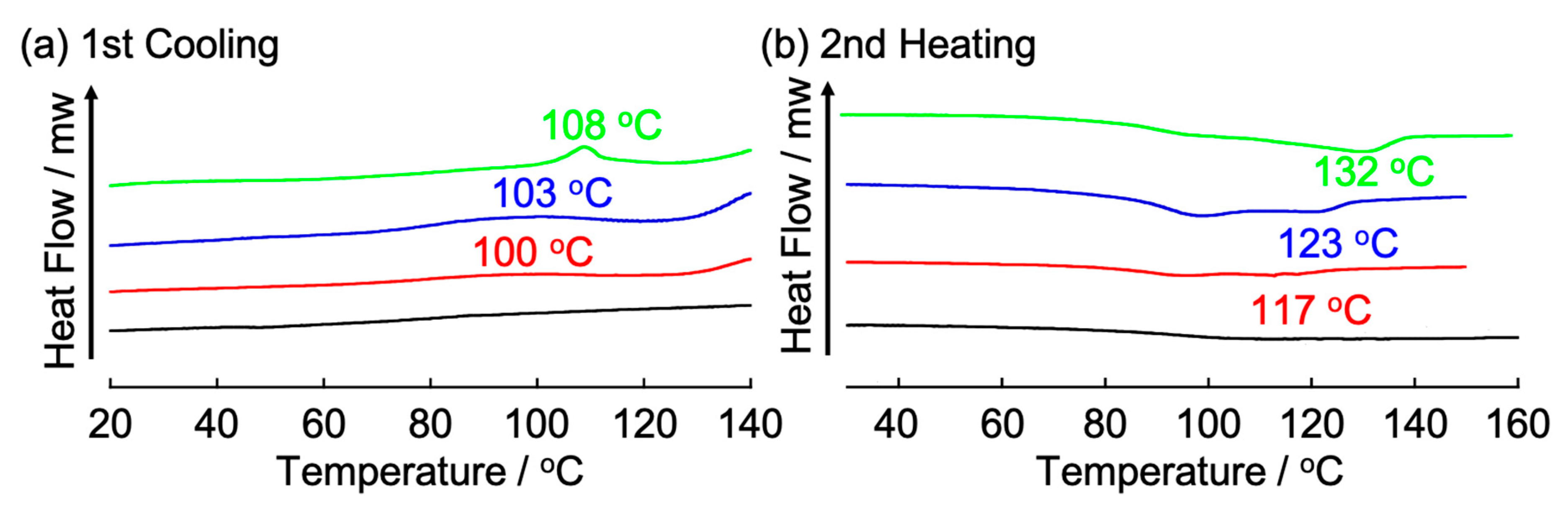
| Complex | m.p./°C a | Peak Top/°C b | ΔH/KJ/mol c | Angle (d Value)/° (Å) d |
|---|---|---|---|---|
| 112 | 114 | – | – | 2.8 (d110 31.4), 3.7 (d200 24.0) |
| 112100 | 121 | 100 | 3.56 | 2.8 (d100 31.4) |
| 11210 | 128 | 103 | 4.34 | 2.8 (d100 31.4) |
| 1125 | 140 | 108 | 4.54 | 3.1 (d100 28.7) |
| 1122 | 240 (Decomp.) | – | – | 3.2 (d100 27.5) |
| 1121 | 250 (Decomp.) | – | – | 3.7 (d100 23.6) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Isoda, K.; Shimooka, K. Synthesis and Characterization of Crown-Ether Appended Hexaazatrinaphthylene-Based Liquid-Crystalline Derivative. Crystals 2020, 10, 377. https://doi.org/10.3390/cryst10050377
Isoda K, Shimooka K. Synthesis and Characterization of Crown-Ether Appended Hexaazatrinaphthylene-Based Liquid-Crystalline Derivative. Crystals. 2020; 10(5):377. https://doi.org/10.3390/cryst10050377
Chicago/Turabian StyleIsoda, Kyosuke, and Keigo Shimooka. 2020. "Synthesis and Characterization of Crown-Ether Appended Hexaazatrinaphthylene-Based Liquid-Crystalline Derivative" Crystals 10, no. 5: 377. https://doi.org/10.3390/cryst10050377
APA StyleIsoda, K., & Shimooka, K. (2020). Synthesis and Characterization of Crown-Ether Appended Hexaazatrinaphthylene-Based Liquid-Crystalline Derivative. Crystals, 10(5), 377. https://doi.org/10.3390/cryst10050377






