Kilohertz Macromolecular Crystallography Using an EIGER Detector at Low X-ray Fluxes
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Setup
2.2. Protein Samples and Data Collection Procedures
2.3. Data Reduction and Treatment
3. Results
3.1. Investigation at a Frame Rate of 1 kHz
3.2. Frame Rates of 2 and 3 kHz
3.3. Frame Rates of 2.4 kHz
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Dinapoli, R.; Bergamaschi, A.; Henrich, B.; Horisberger, R.; Johnson, I.; Mozzanica, A.; Schmid, E.; Schmitt, B.; Schreiber, A.; Shi, X.; et al. EIGER: Next generation single photon counting detector for X-ray applications. Nucl. Instrum. Methods Phys. Res. Sect. A Accel. Spectometers Detect. Assoc. Equip. 2011, 650, 79–83. [Google Scholar] [CrossRef]
- Ciccone, L.; Fruchart-Gaillard, C.; Mourier, G.; Savko, M.; Nencetti, S.; Orlandini, E.; Servent, D.; Stura, E.A.; Shepard, W. Copper mediated amyloid-β binding to Transthyretin. Sci. Rep. 2018, 8, 13744. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.-Y.; Olieric, V.; Ma, P.; Howe, N.; Vogeley, L.; Liu, X.; Warshamanage, R.; Weinert, T.; Panepucci, E.; Kobilka, B.; et al. In meso in situ serial X-ray crystallography of soluble and membrane proteins at cryogenic temperatures. Acta Crystallogr. Sect. D Struct. Biol. 2016, 72, 93–112. [Google Scholar] [CrossRef] [PubMed]
- Leonarski, F.; Redford, S.; Mozzanica, A.; Lopez-Cuenca, C.; Panepucci, E.; Nass, K.; Ozerov, D.; Vera, L.; Olieric, V.; Buntschu, D.; et al. Fast and accurate data collection for macromolecular crystallography using the JUNGFRAU detector. Nat. Methods 2018, 15, 799–804. [Google Scholar] [CrossRef] [PubMed]
- Orville, A.M. Entering an era of dynamic structural biology. BMC Biol. 2018, 16, 55. [Google Scholar] [CrossRef] [PubMed]
- Pellegrini, C. X-ray free-electron lasers: From dreams to reality. Phys. Scr. 2016, 111, 014004. [Google Scholar] [CrossRef]
- Zhavoronkov, N.; Gritsai, Y.; Bargheer, M.; Woerner, M.; Elsaesser, T.; Zamponi, F.; Uschmann, I.; Förster, E. Microfocus Cu Kα source for femtosecond X-ray science. Opt. Lett. 2005, 30, 1737–1739. [Google Scholar] [CrossRef]
- Korn, G.; Thoss, A.; Stiel, H.; Vogt, U.; Richardson, M.; Elsaesser, T.; Faubel, M. Ultrashort 1-kHz laser plasma hard X-ray source. Opt. Lett. 2002, 27, 866–868. [Google Scholar] [CrossRef]
- Zamponi, F.; Ansari, Z.; Schmising, C.V.K.; Rothhardt, P.; Zhavoronkov, N.; Woerner, M.; Elsaesser, T.; Bargheer, M.; Trobitzsch-Ryll, T.; Haschke, M. Femtosecond hard X-ray plasma sources with a kilohertz repetition rate. Appl. Phys. A 2009, 96, 51–58. [Google Scholar] [CrossRef]
- Weisshaupt, J.; Juvé, V.; Holtz, M.; Ku, S.; Woerner, M.; Elsaesser, T.; Ališauskas, S.; Pugžlys, A.; Baltuška, A. High-brightness table-top hard X-ray source driven by sub-100-femtosecond mid-infrared pulses. Nat. Photon. 2014, 8, 927–930. [Google Scholar] [CrossRef]
- Caffrey, M.; Li, D.; Howe, N.; Shah, S.T. ‘Hit and run’ serial femtosecond crystallography of a membrane kinase in the lipid cubic phase. Philos. Trans. R. Soc. B Biol. Sci. 2014, 369, 20130621. [Google Scholar] [CrossRef] [PubMed]
- Botha, S.; Baitan, D.; Jungnickel, K.E.J.; Oberthür, D.; Schmidt, C.; Stern, S.; Wiedorn, M.O.; Perbandt, M.; Chapman, H.N.; Betzel, C. De novo protein structure determination by heavy-atom soaking in lipidic cubic phase and SIRAS phasing using serial synchrotron crystallography. IUCrJ 2018, 5, 524–530. [Google Scholar] [CrossRef] [PubMed]
- Rousse, A.; Rischel, C.; Gauthier, J.C. Femtosecond X-ray crystallography. Rev. Mod. Phys. 2001, 73, 17–31. [Google Scholar] [CrossRef]
- Marangos, J.P. Introduction to the new science with X-ray free electron lasers. Contemp. Phys. 2011, 6, 551–569. [Google Scholar] [CrossRef]
- Hauf, C.; Salvador, A.-A.H.; Holtz, M.; Woerner, M.; Elsaesser, T. Phonon driven charge dynamics in polycrystalline acetylsalicylic acid mapped by ultrafast x-ray diffraction. Struct. Dyn. 2019, 6, 014503. [Google Scholar] [CrossRef]
- Chapman, H.N. X-Ray Free-Electron Lasers for the Structure and Dynamics of Macromolecules. Annu. Rev. Biochem. 2019, 88, 35–38. [Google Scholar] [CrossRef]
- Grünbein, M.L.; Bielecki, J.; Gorel, A.; Stricker, M.; Bean, R.; Cammarata, M.; Dörner, K.; Fröhlich, L.; Hartmann, E.; Hauf, S.; et al. Megahertz data collection from protein microcrystals at an X-ray free-electron laser. Nat. Commun. 2018, 9, 3487. [Google Scholar] [CrossRef]
- Schmidt, M. Time-Resolved Macromolecular Crystallography at Pulsed X-ray Sources. Int. J. Mol. Sci. 2019, 20, 1401. [Google Scholar] [CrossRef]
- Wiedorn, M.O.; Oberthür, D.; Bean, R.; Schubert, R.; Werner, N.; Abbey, B.; Aepfelbacher, M.; Adriano, L.; Allahgholi, A.; Al-Qudami, N.; et al. Megahertz serial crystallography. Nat. Commun. 2018, 9, 4025. [Google Scholar] [CrossRef]
- Chapman, H.N.; Fromme, N.; Barty, A.; White, T.A.; Kirian, R.A.; Aquila, A.; Hunter, M.S.; Schulz, J.; DePonte, D.P.; Weierstall, U.; et al. Femtosecond X-ray protein nanocrystallography. Nature 2011, 470, 73–77. [Google Scholar] [CrossRef]
- Schlichting, I. Serial femtosecond crystallography: The first five years. IUCrJ 2015, 2, 246–255. [Google Scholar] [CrossRef] [PubMed]
- Stagno, J.R.; Liu, Y.; Bhandari, Y.R.; Conrad, C.E.; Panja, S.; Swain, M.; Fan, L.; Nelson, G.; Li, C.; Wendel, D.R.; et al. Structures of riboswitch RNA reaction states by mix-and-inject XFEL serial crystallography. Nature 2017, 541, 242–246. [Google Scholar] [CrossRef] [PubMed]
- Weinert, T.; Olieric, N.; Cheng, R.; Brunle, S.; James, D.; Ozerov, D.; Gashi, D.; Vera, L.; Marsh, M.; Jaeger, K.; et al. Serial millisecond crystallography for routine room-temperature structure determination at synchrotrons. Nat. Commun. 2017, 8, 542. [Google Scholar] [CrossRef] [PubMed]
- Neutze, R.; Wouts, R.; van der Spoel, D.; Weckert, E.; Hajdu, J. Potential for biomolecular imaging with femtosecond X-ray pulses. Nature 2000, 406, 752–757. [Google Scholar] [CrossRef] [PubMed]
- Mancuso, A.P.; Aquila, A.; Batchelor, L.; Bean, R.J.; Bielecki, J.; Borchers, G.; Doerner, K.; Giewekemeyer, K.; Graceffa, R.; Kelsey, O.D.; et al. The Single Particles, Clusters and Biomolecules and Serial Femtosecond Crystallography instrument of the European XFEL: Initial installation. J. Synchrotron Radiat. 2019, 26, 660–676. [Google Scholar] [CrossRef]
- Martin-Garcia, J.M.; Conrad, C.E.; Nelson, G.; Stander, N.; Zatsepin, N.A.; Zook, J.; Zhu, L.; Geiger, J.; Chun, E.; Kissick, D.; et al. Serial millisecond crystallography of membrane and soluble protein microcrystals using synchrotron radiation. IUCrJ 2017, 4, 439–454. [Google Scholar] [CrossRef]
- Gati, C.; Bourenkov, G.; Klinge, M.; Rehders, D.; Stellato, F.; Oberthür, D.; Yefanov, O.; Sommer, B.P.; Mogk, S.; Duszenko, M.; et al. Serial crystallography on in vivo grown microcrystals using synchrotron radiation. IUCrJ 2014, 1, 87–94. [Google Scholar] [CrossRef]
- Rossmann, M.G. Serial crystallography using synchrotron radiation. IUCrJ 2014, 1, 84–86. [Google Scholar] [CrossRef]
- Bonvalet, A.; Darmon, A.; Lambry, J.C.; Martin, J.L.; Audebert, P. 1 kHz tabletop ultrashort hard X-ray source for time-resolved X-ray protein crystallography. Opt. Lett. 2006, 31, 2753–2755. [Google Scholar] [CrossRef]
- Kraft, P.; Bergamaschi, A.; Broennimann, C.; Dinapoli, R.; Eikenberry, E.F.; Henrich, B.; Johnson, I.; Mozzanica, A.; Schleputz, C.M.; Willmotta, P.R.; et al. Performance of single-proton-counting PILATUS detector modules. J. Synchrotron Radiat. 2009, 16, 368–375. [Google Scholar] [CrossRef]
- Johnson, I.; Bergamaschi, A.; Billich, H.; Cartier, S.; Dinapoli, R.; Greiffenberg, D.; Guizar-Sicairos, M.; Henrich, B.; Jungmann, J.; Mezza, D.; et al. Eiger: A single-photon counting X-ray detector. J. Instrum. 2014, 9, C05032. [Google Scholar] [CrossRef]
- Radicci, V.; Bergamaschi, A.; Dinapoli, R.; Greiffenberg, D.; Henrich, B.; Johnson, I.; Mozzanica, A.; Schmitt, B.; Shi, X. EIGER a new single photon counting detector for X-ray applications: Performance of the chip. J. Instrum. 2012, 7, C02019. [Google Scholar] [CrossRef]
- Casanas, A.; Warshamanage, R.; Finke, A.D.; Panepucci, E.; Olieric, V.; Nöll, A.; Tampé, R.; Brandstetter, S.; Förster, A.; Mueller, M.; et al. EIGER detector: Application in macromolecular crystallography. Acta Crystallogr. Sect. D Struct. Biol. 2016, 72, 1036–1048. [Google Scholar] [CrossRef]
- Duran, D.; Couster, S.L.; Desjardins, K.; Delmotte, A.; Fox, G.; Meijers, R.; Moreno, T.; Savko, M.; Shepard, W. PROXIMA 2A—A new fully tunable micro-focus beamline for macromolecular crystallography. J. Phys. Conf. Ser. 2013, 425, 012005. [Google Scholar] [CrossRef]
- McPherson, A.; Gavira, J.A. Introduction to protein crystallization. Acta Crystallogr. Sect. F Struct. Biol. Commun. 2014, 70, 2–20. [Google Scholar] [CrossRef] [PubMed]
- Kabsch, W. Integration, scaling, space-group assignments and post-refinement. Acta Crystallogr. Sect. D Struct. Biol. 2010, 66, 125–132. [Google Scholar] [CrossRef] [PubMed]
- Battye, T.G.; Kontogiannis, L.; Johnson, O.; Powell, H.R.; Leslie, A.G. iMOSFLM: A new graphical interface for diffraction-image processing with MOSFLM. Acta Crystallogr. Sect. D Biol Crystallogr. 2011, 67, 271–281. [Google Scholar] [CrossRef]
- Evans, P.R.; Murshudov, G.N. How good are my data and what is the resolution? Acta Crystallogr. Sect. D Struct. Biol. 2013, 69, 1204–1214. [Google Scholar] [CrossRef]
- Vagin, A.A.; Teplyakov, A. MOLREP: An automated program for molecular replacement. J. Appl. Crystallogr. 1997, 30, 1022–1025. [Google Scholar] [CrossRef]
- Murshudov, G.N.; Skubak, P.; Lebedev, A.A.; Pannu, N.S.; Steiner, R.A.; Nicholls, R.A.; Winn, M.D.; Long, F.; Vagin, A.A. REFMAC5 for the refinement of macromolecular crystal structures. Acta Crystallogr. Sect. D Struct. Biol. 2011, 67, 355–367. [Google Scholar] [CrossRef]
- Collaborative Computational Project. The CCP4 suite: Programs for protein crystallography. Acta Crystallogr. Sect. D Biol. Crystallogr. 1994, 50, 760–763. [Google Scholar] [CrossRef] [PubMed]
- Weiss, M.S.; Palm, G.J.; Hilgenfeld, R. Crystallization, structure solution and refinement of hen egg-white lysozyme at pH 8.0 in the presence of MPD. Acta Crystallogr. Sect. D Struct. Biol. 2000, 56, 952–958. [Google Scholar] [CrossRef] [PubMed]
- Kieffer, J.; Karkoulis, D. PyFAI, a versatile library for azimuthal regrouping. J. Phys. Conf. Ser. 2013, 425, 202012. [Google Scholar] [CrossRef]
- Kabsch, W. Processing of X-ray snapshots from crystals in random orientations. Acta Crystallogr. Sect. D Struct. Biol. 2014, 70, 2204–2216. [Google Scholar] [CrossRef]


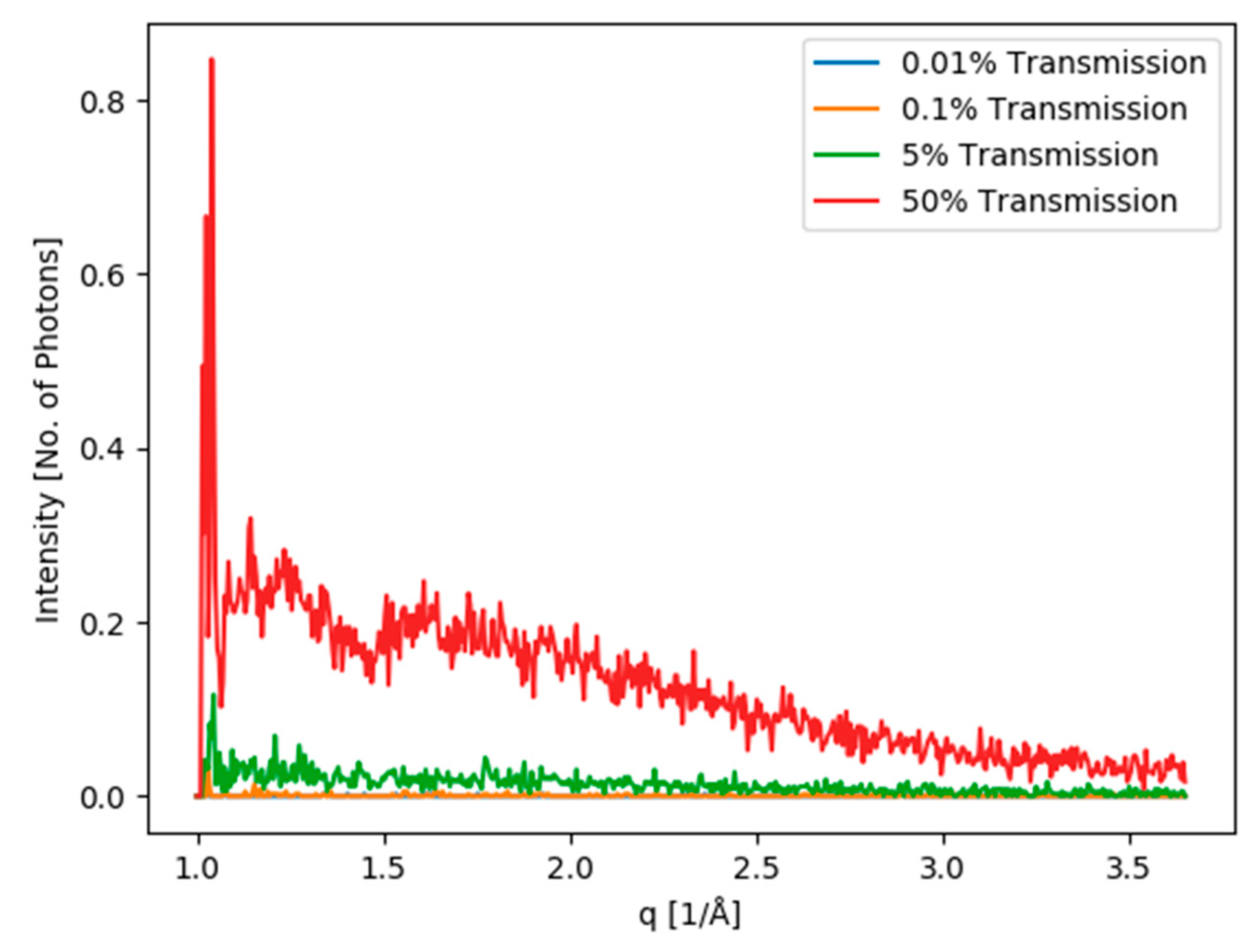

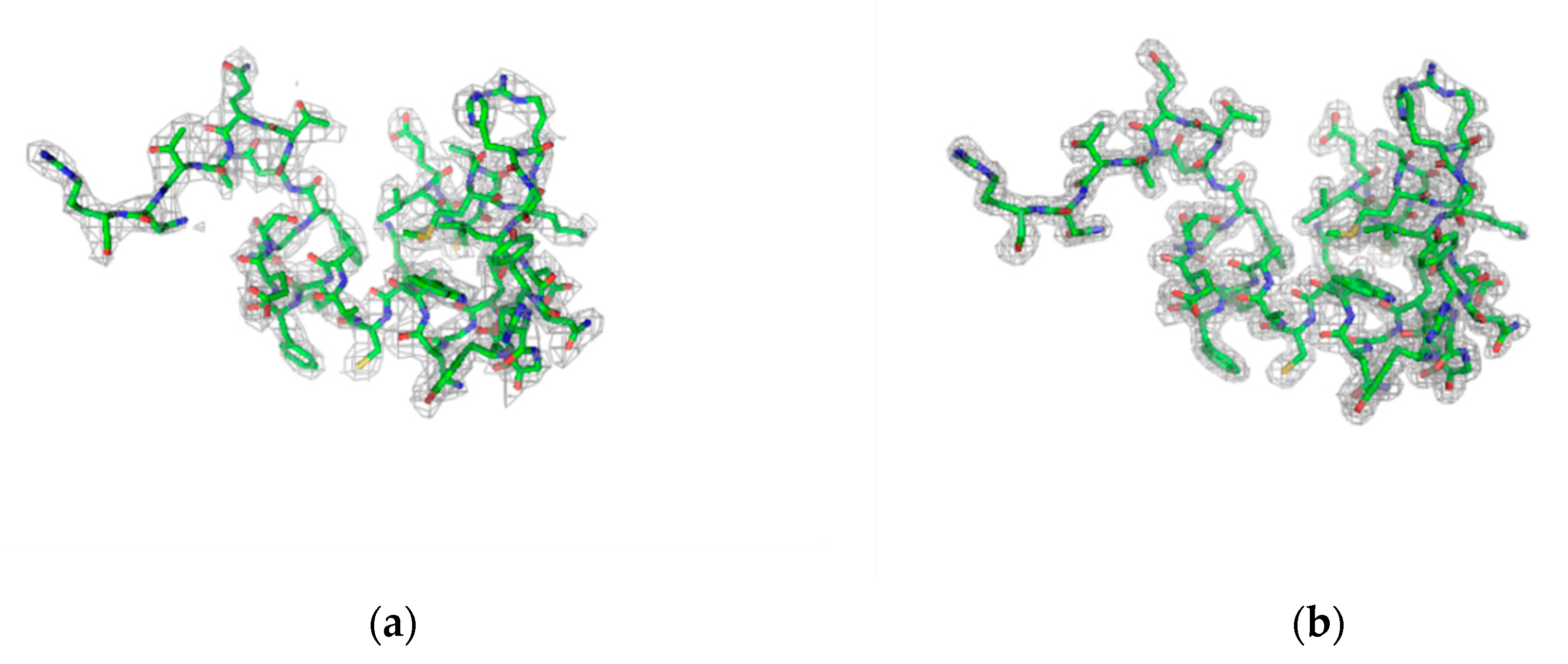
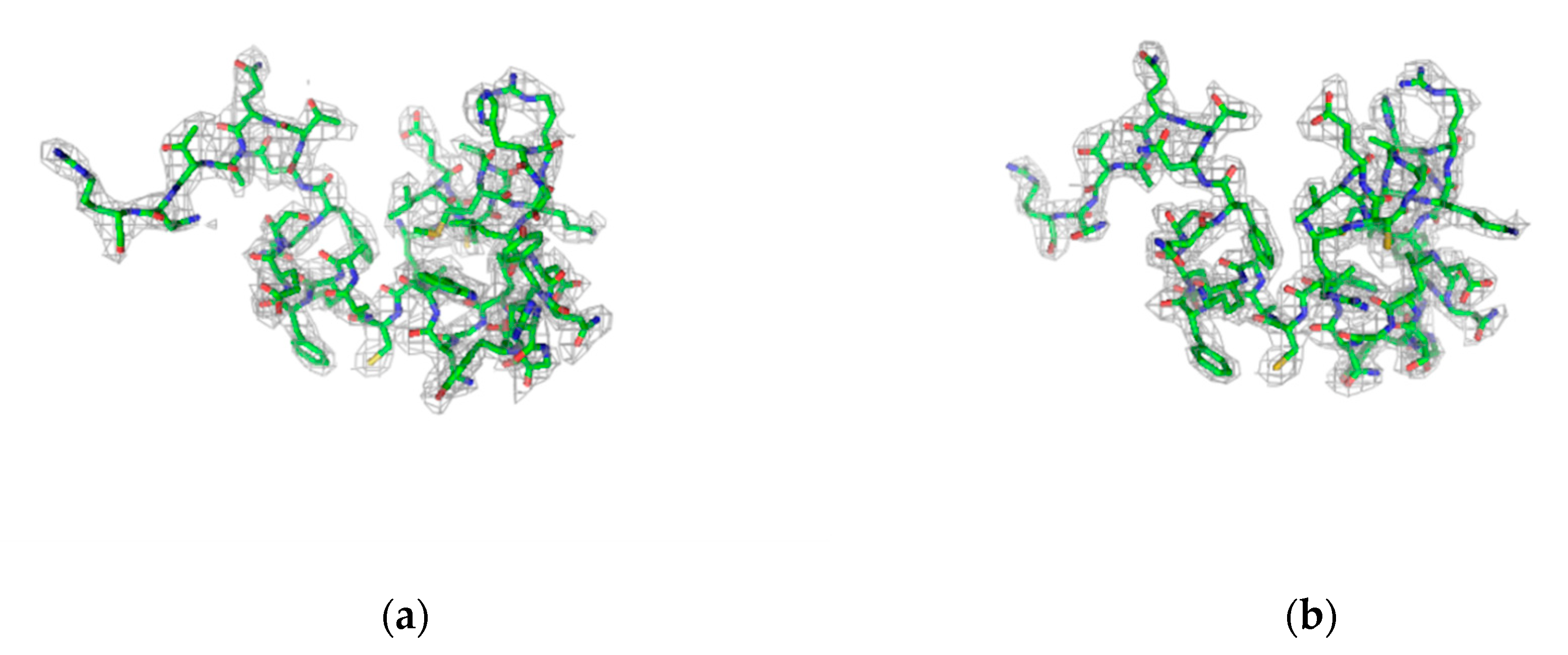
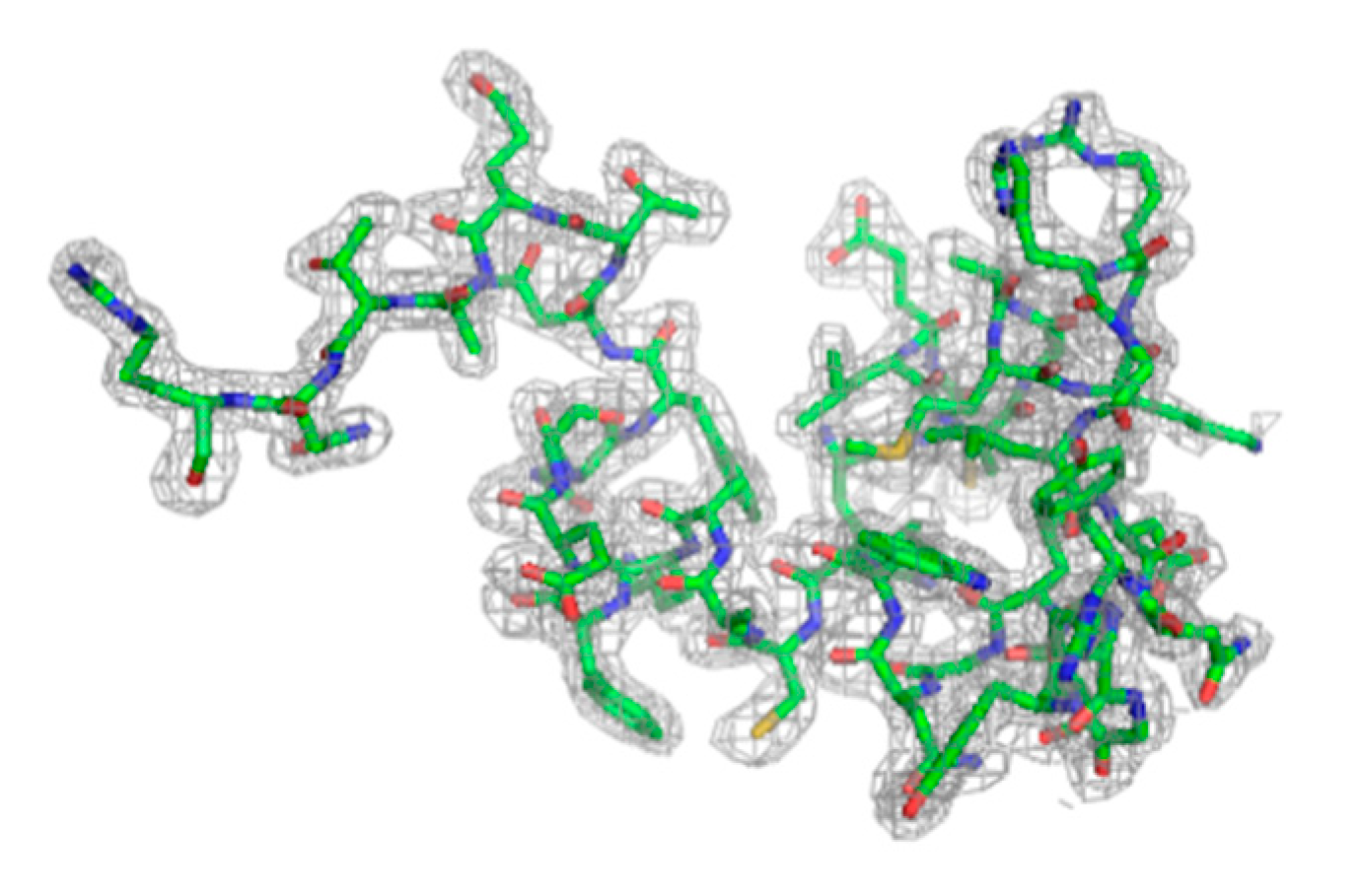
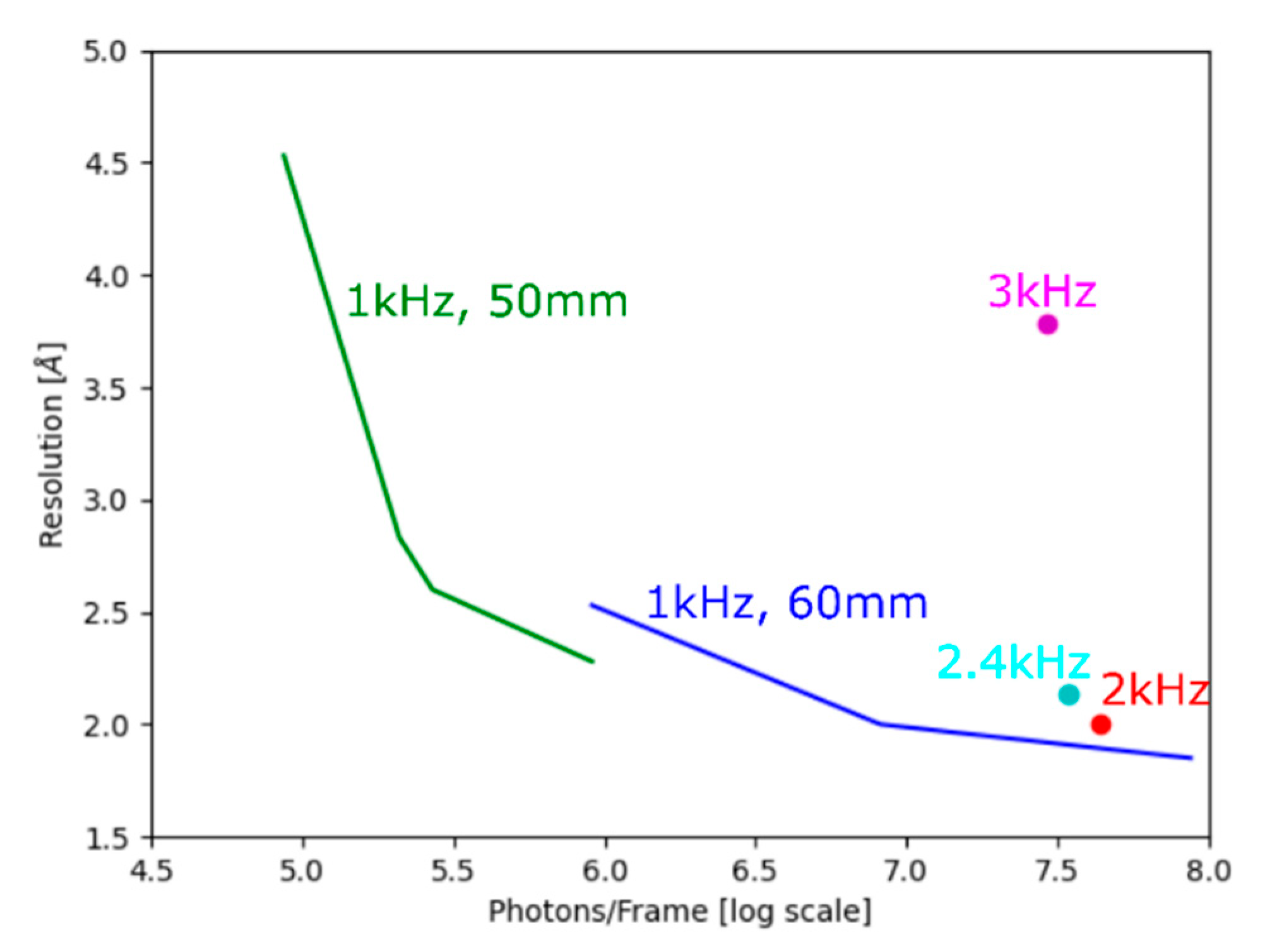
| Dataset | lyz1 1 kHz 0.5Io | lyz1 1 kHz 5Io | lyz1 1 kHz 50Io | lyz1 2 kHz 50Io | lyz1 2.4 kHz 5Io | lyz1 3 kHz 50Io | lyz2 1 kHz 0.01Io | lyz2 1 kHz 0.05Io | lyz2 1 kHz 0.1Io | lyz2 1 kHz 0.5Io |
|---|---|---|---|---|---|---|---|---|---|---|
| Dataset abbreviation | L11_05 | L11_5 | L11_50 | L12_50 | L12.4_5 | L13_50 | L21_001 | L21_005 | L21_01 | L21_05 |
| Rotation speed (°/s) | 100 | 100 | 100 | 200 | 120 | 300 | 100 | 100 | 100 | 100 |
| Exposure time (s) | 0.001 | 0.001 | 0.001 | 0.0005 | 0.00041 | 0.0003 | 0.001 | 0.001 | 0.001 | 0.001 |
| Set transmission (%) | 0.5 | 5 | 50 | 50 | 5 | 50 | 0.01 | 0.05 | 0.1 | 0.5 |
| Flux 1 (photons/s) on crystal | 9.1 × 108 | 8.2 × 109 | 8.7 × 1010 | 8.7 × 1010 | 8.2 × 109 | 8.7 × 1010 | 8.7 × 107 | 2.1 × 108 | 2.7 × 108 | 9.1 × 108 |
| Unit cell | ||||||||||
| a = b (Å) | 75.97 | 76.14 | 76.3 | 79.16 | 76.12 | 78.97 | 75.8 | 75.9 | 75.99 | 76.08 |
| c (Å) | 35.47 | 35.54 | 35.61 | 37.01 | 35.54 | 35.01 | 35.16 | 35.36 | 35.4 | 35.44 |
| Mosaicity (°) * MOSFLM | 0.11 | 0.11 | 0.15 | 0.68 * | 0.14 | 0.74 * | 0.07 | 0.07 | 0.07 | 0.07 |
| Resolution (Å) | 2.53 (2.64–2.53) | 2.0 (2.07–2.0) | 1.85 (1.93–1.85) | 2.0 (2.09–2.0) | 2.14 (2.18–2.14) | 3.78 (3.86–3.78) | 4.53 (4.64–4.53) | 2.83 (2.96–2.83) | 2.6 (2.71–2.6) | 2.28 (2.40–2.28) |
| No. of reflection | 92,496 | 179,722 | 209,230 | 199,428 | 156,432 | 151,616 | 15,923 | 61,382 | 86,921 | 179,654 |
| No. of unique reflection | 3762 | 7453 | 9408 | 8388 | 6308 | 6160 | 1009 | 2965 | 3922 | 7444 |
| Completeness (%) | 99.5 (97.3) | 99.6 (96.3) | 99.7 (96.9) | 99.9 (95.6) | 99.7 (97.0) | 98.1 (92.0) | 99.1 (95.1) | 99.4 (93.4) | 99.5 (94.0) | 99.5 (94.9) |
| Multiplicity | 24.6 (24.2) | 24.1 (16.1) | 22.2 (12.5) | 23.8 (17.1) | 24.8 (19.1) | 24.6 (16.3) | 15.8 (17.8) | 20.7 (19.0) | 22.2 (19.8) | 24.1 (17.9) |
| ISa | 7.32 | 19.35 | 18.16 | 27.23 | 16.83 | 23.92 | 4.09 | 4.72 | 4.18 | 7.11 |
| I/σ(I) | 10.8 (3.9) | 26.2 (5.8) | 37.7 (3.1) | 45.0 (6.1) | 22.1 (4.2) | 21.0 (4.8) | 5.7 (3.4) | 7.1 (4.1) | 7.7 (3.1) | 8.3 (3.0) |
| Rmeas (%) | 28.2 (58.2) | 10.1 (54.5) | 6.2 (48.0) | 5.9 (13.8) | 11.2 (55.1) | 6.0 (22.3) | 47.7 (49.8) | 42 (76.0) | 37.7 (61.8) | 29.1 (48.2) |
| CC1/2 | 99.2 (95.9) | 99.9 (96.0) | 99.9 (95.1) | 99.9 (88.2) | 99.9 (90.2) | 91.7 (76.3) | 96.1 (95.8) | 97.8 (92.4) | 98.5 (89.5) | 99.3 (82.2) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Khakurel, K.P.; Espinoza, S.; Savko, M.; Polovinkin, V.; Dohnalek, J.; Shepard, W.; Angelova, A.; Hajdu, J.; Andreasson, J.; Angelov, B. Kilohertz Macromolecular Crystallography Using an EIGER Detector at Low X-ray Fluxes. Crystals 2020, 10, 1146. https://doi.org/10.3390/cryst10121146
Khakurel KP, Espinoza S, Savko M, Polovinkin V, Dohnalek J, Shepard W, Angelova A, Hajdu J, Andreasson J, Angelov B. Kilohertz Macromolecular Crystallography Using an EIGER Detector at Low X-ray Fluxes. Crystals. 2020; 10(12):1146. https://doi.org/10.3390/cryst10121146
Chicago/Turabian StyleKhakurel, Krishna P., Shirly Espinoza, Martin Savko, Vitaly Polovinkin, Jan Dohnalek, William Shepard, Angelina Angelova, Janos Hajdu, Jakob Andreasson, and Borislav Angelov. 2020. "Kilohertz Macromolecular Crystallography Using an EIGER Detector at Low X-ray Fluxes" Crystals 10, no. 12: 1146. https://doi.org/10.3390/cryst10121146
APA StyleKhakurel, K. P., Espinoza, S., Savko, M., Polovinkin, V., Dohnalek, J., Shepard, W., Angelova, A., Hajdu, J., Andreasson, J., & Angelov, B. (2020). Kilohertz Macromolecular Crystallography Using an EIGER Detector at Low X-ray Fluxes. Crystals, 10(12), 1146. https://doi.org/10.3390/cryst10121146








