Application of Response Surface Methodology for Ethanol Conversion into Hydrocarbons Using ZSM-5 Zeolites
Abstract
1. Introduction
2. Results and Discussion
2.1. Catalyst Characterization
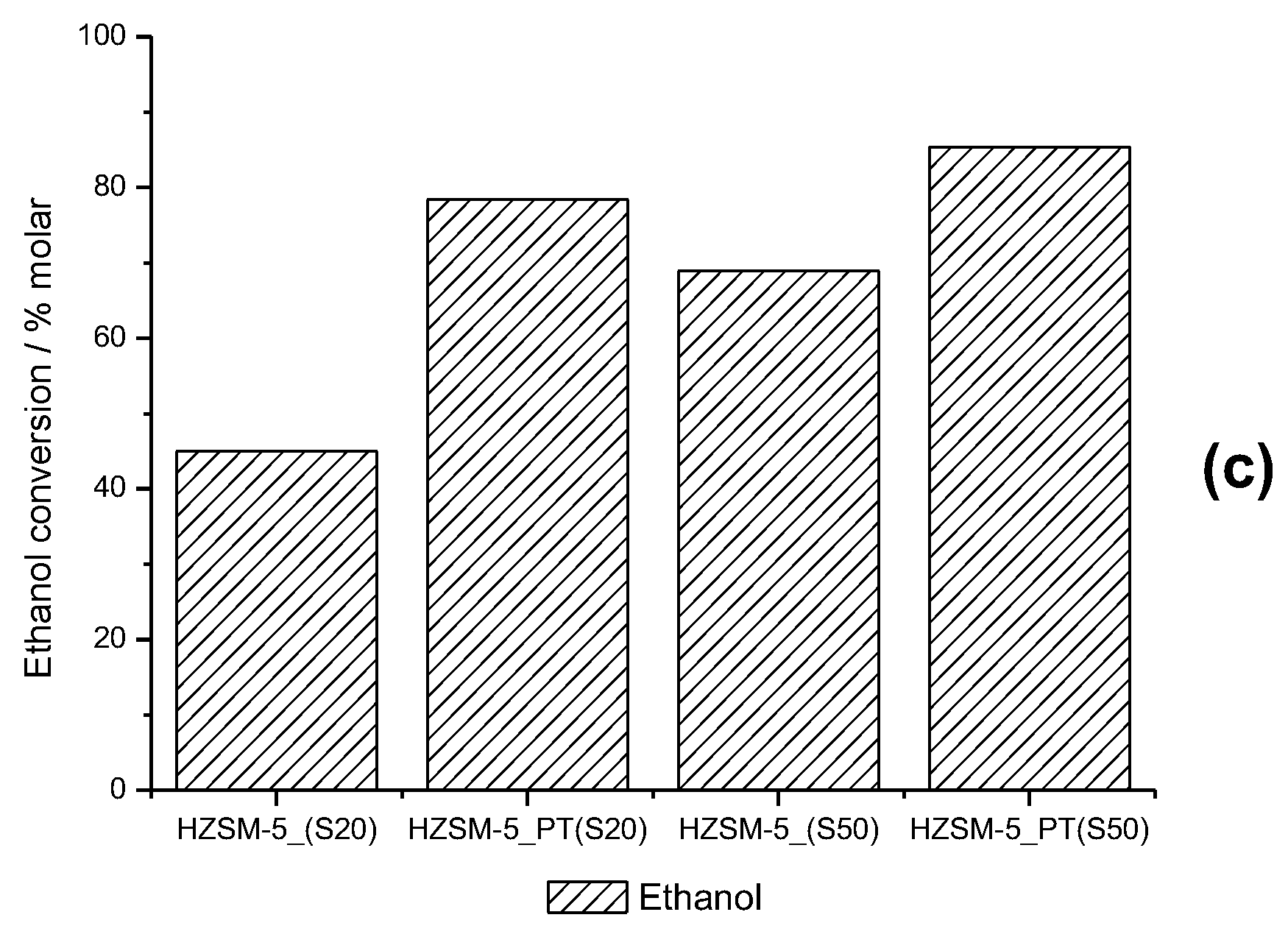
2.2. Screening of Catalyst and Reaction Parameters
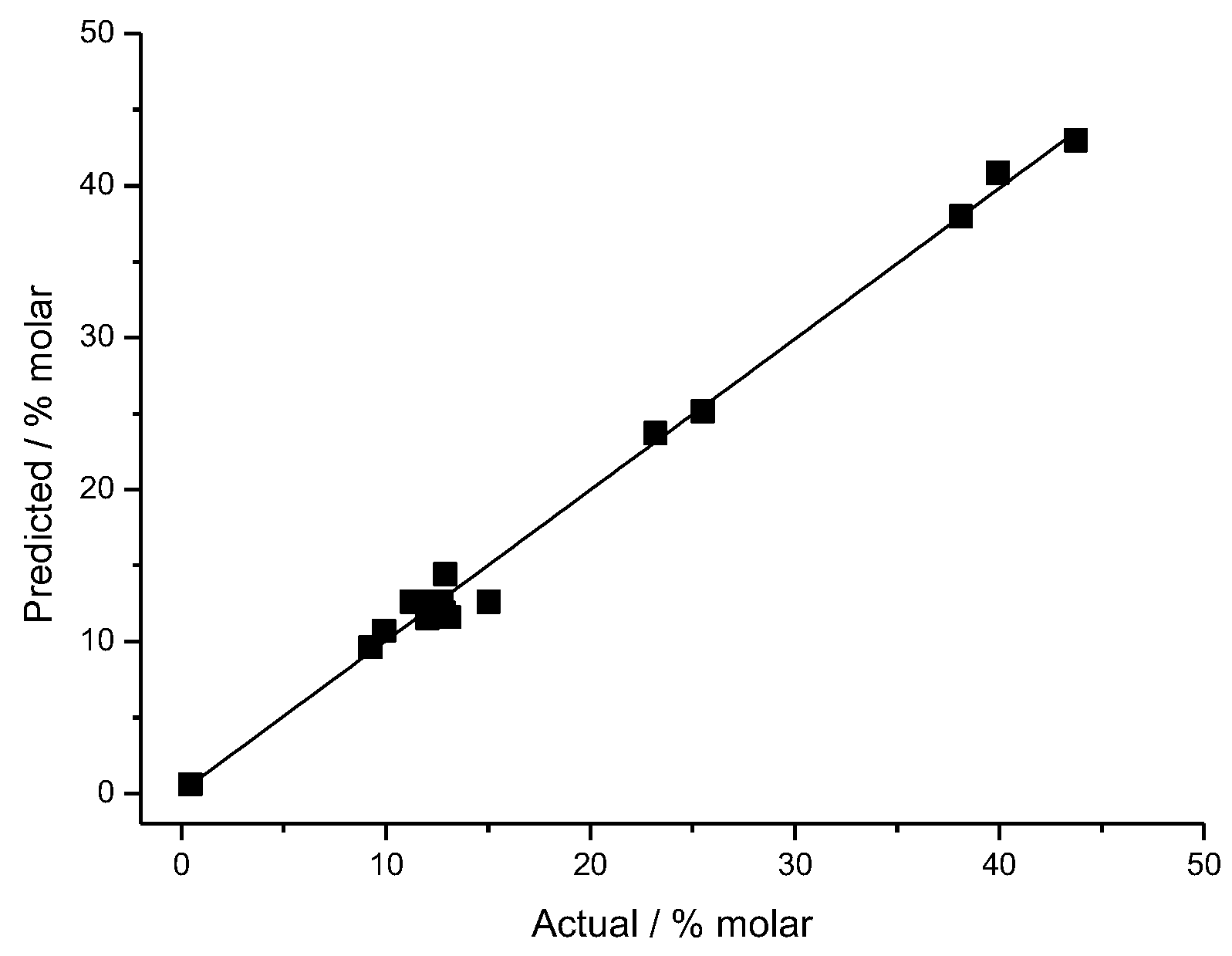
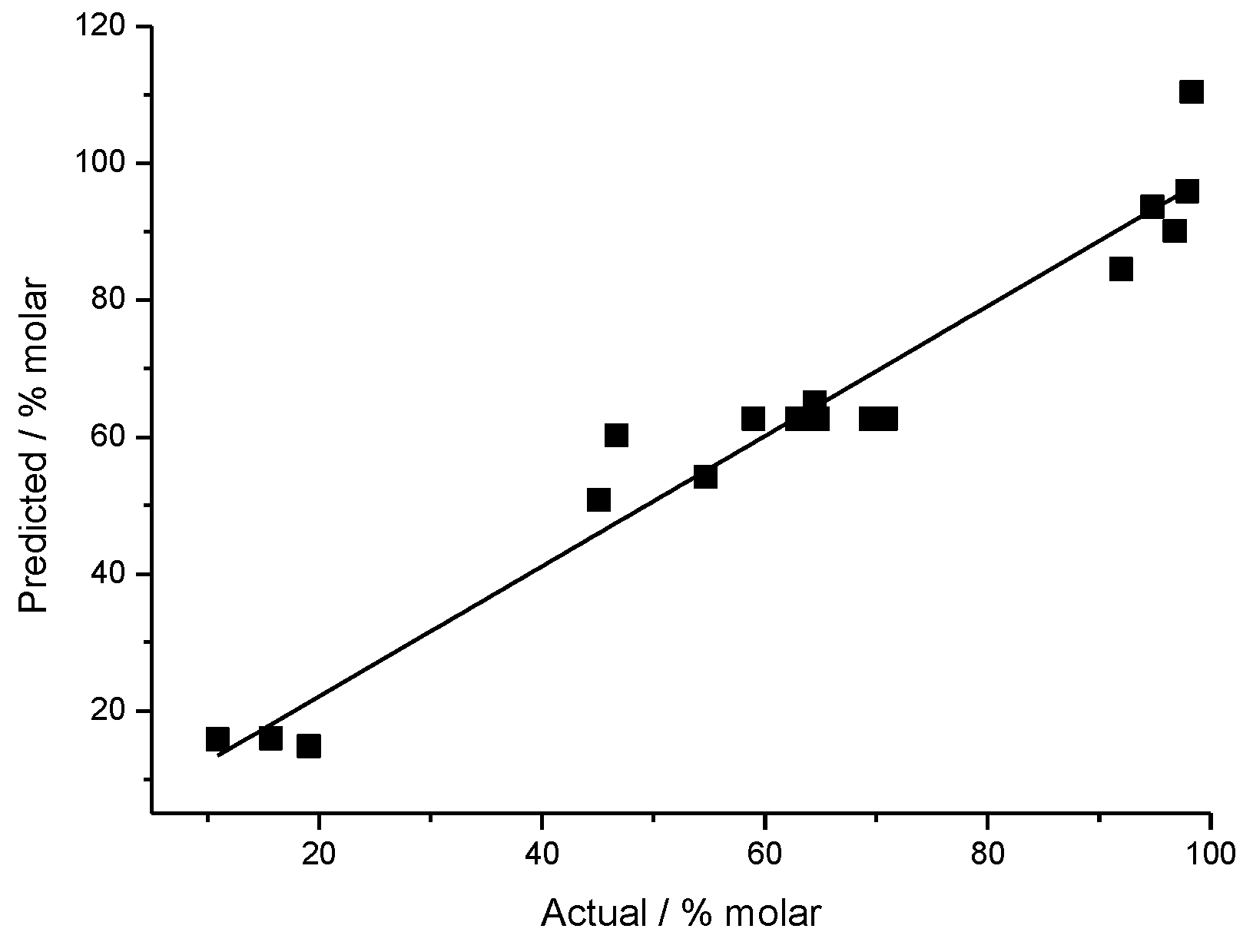
2.3. Development of Regression Equation
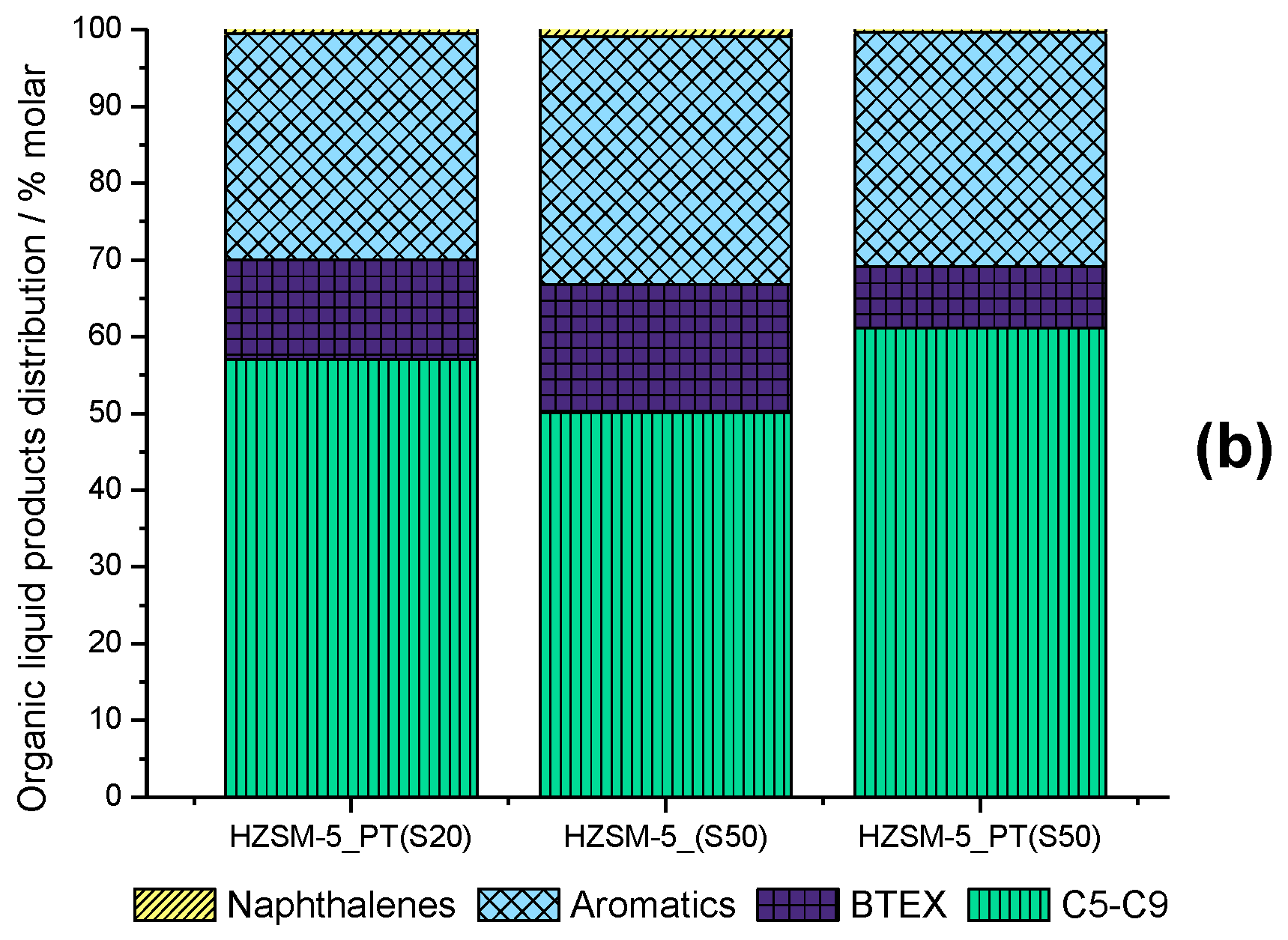
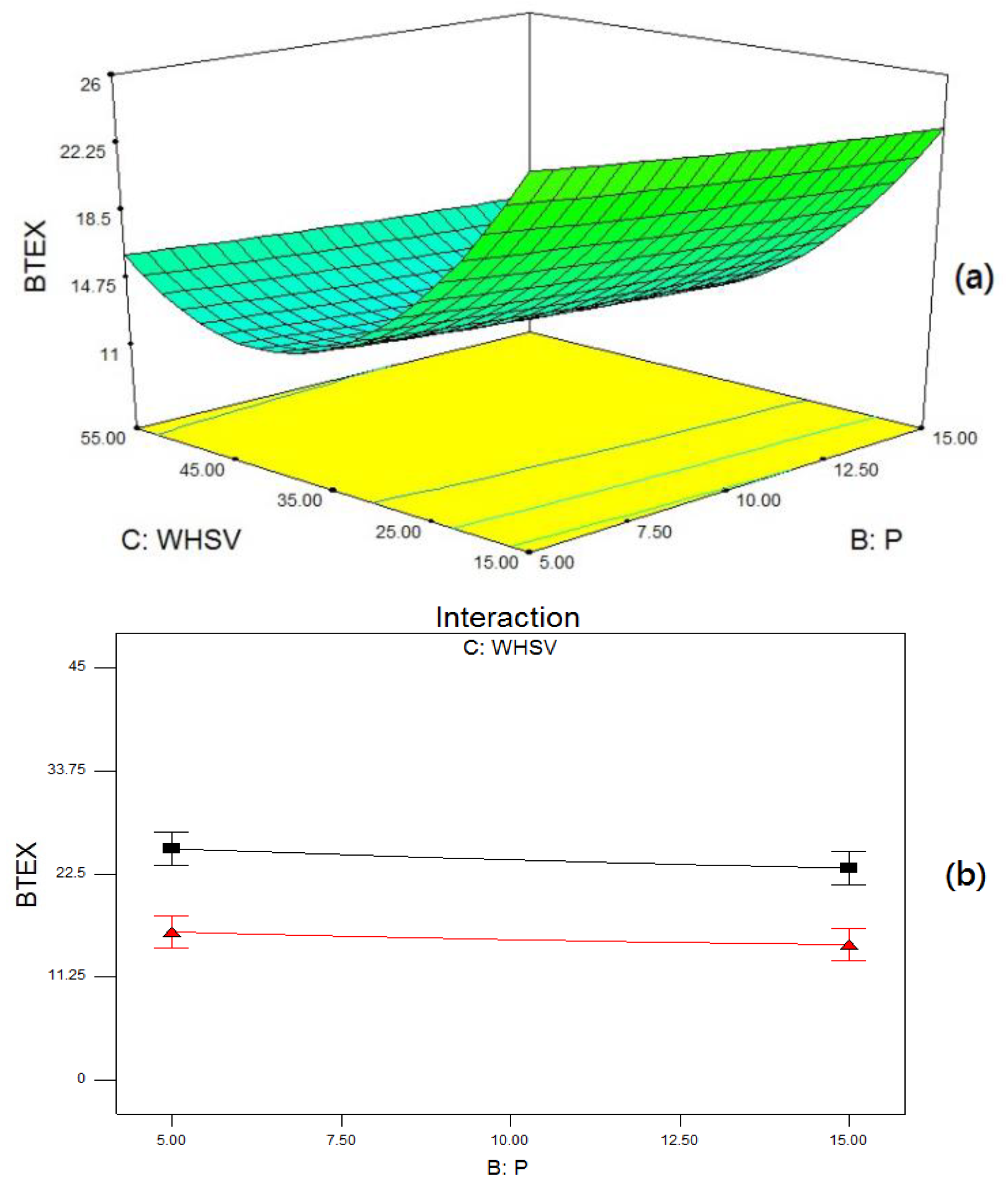
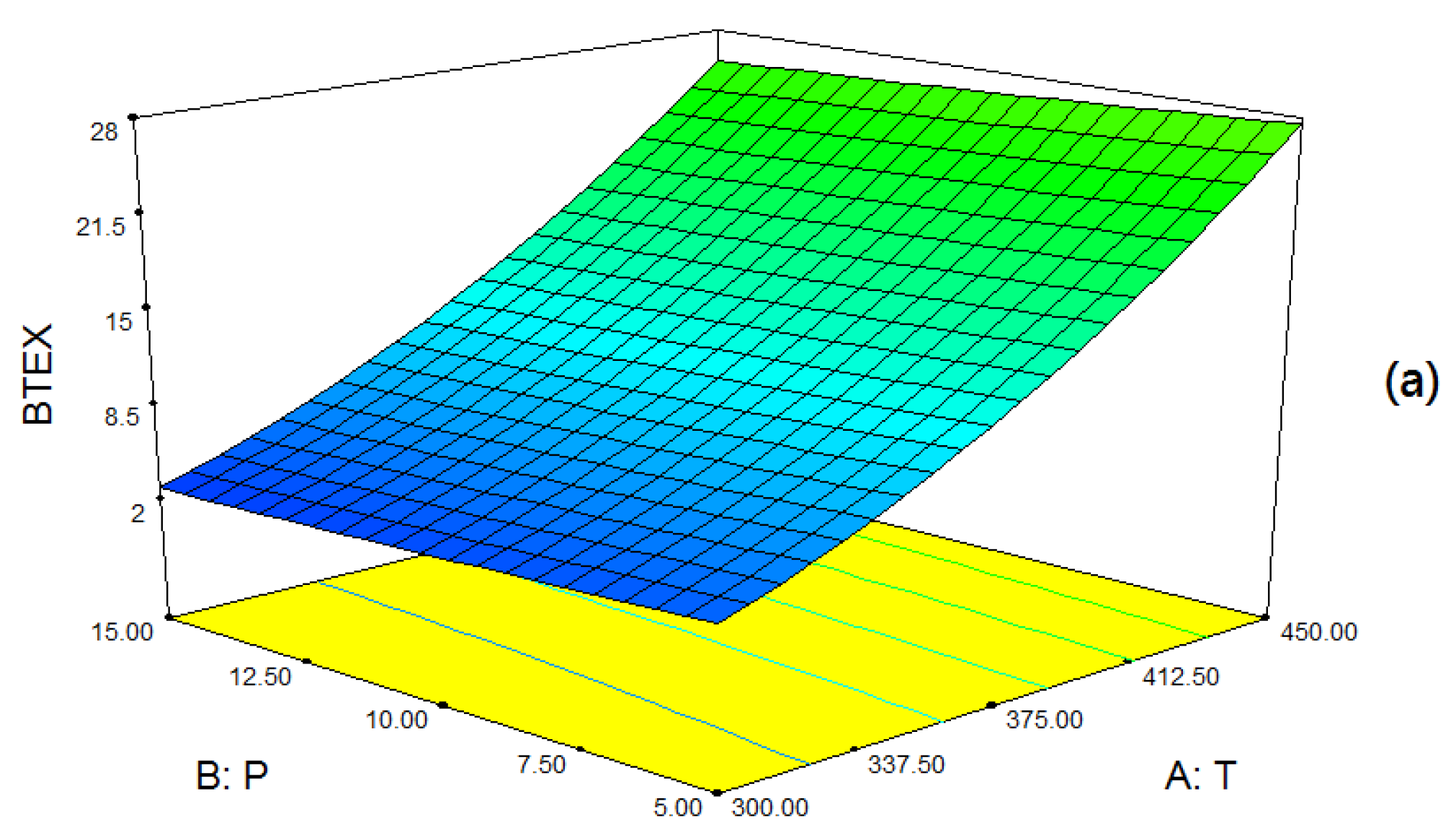
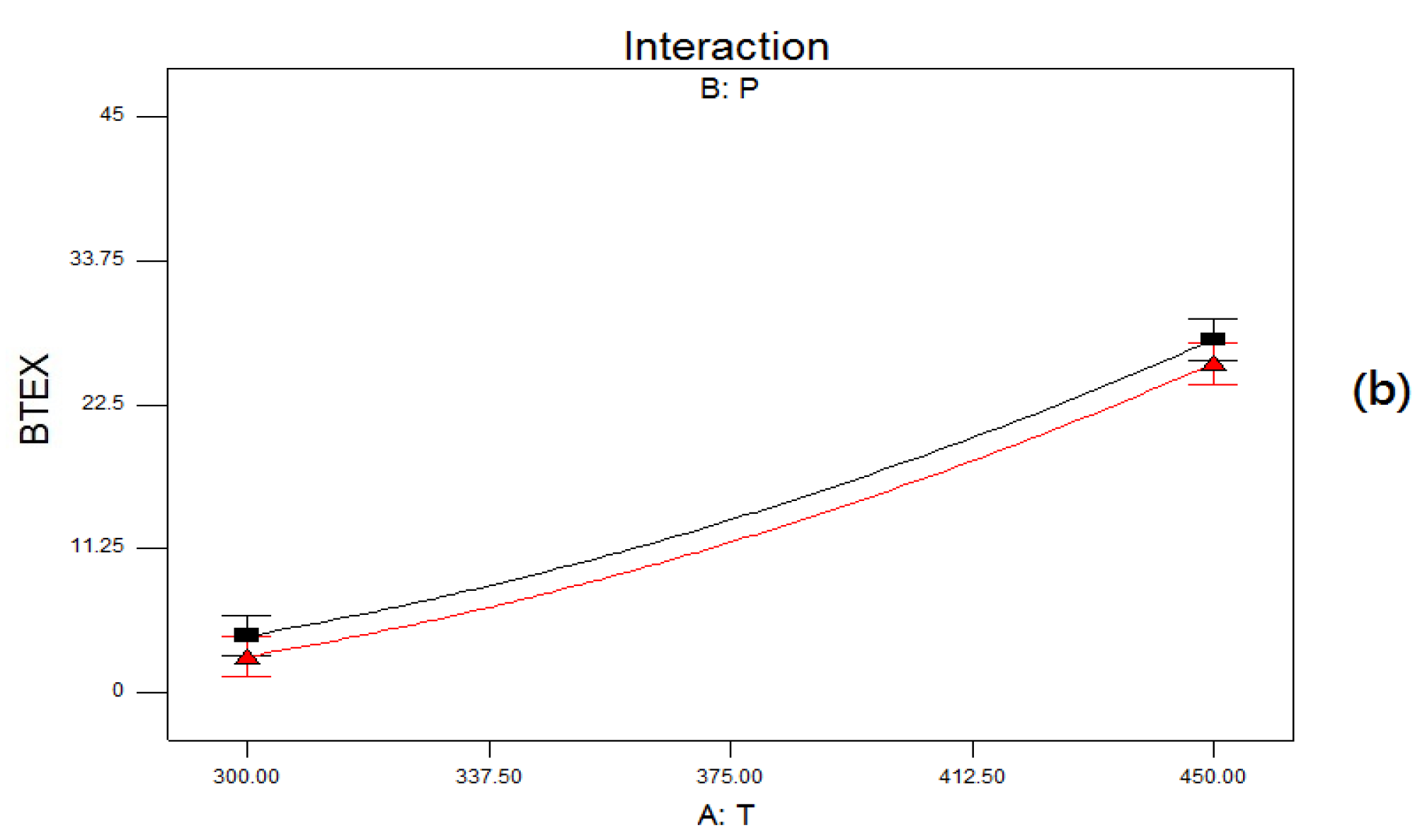
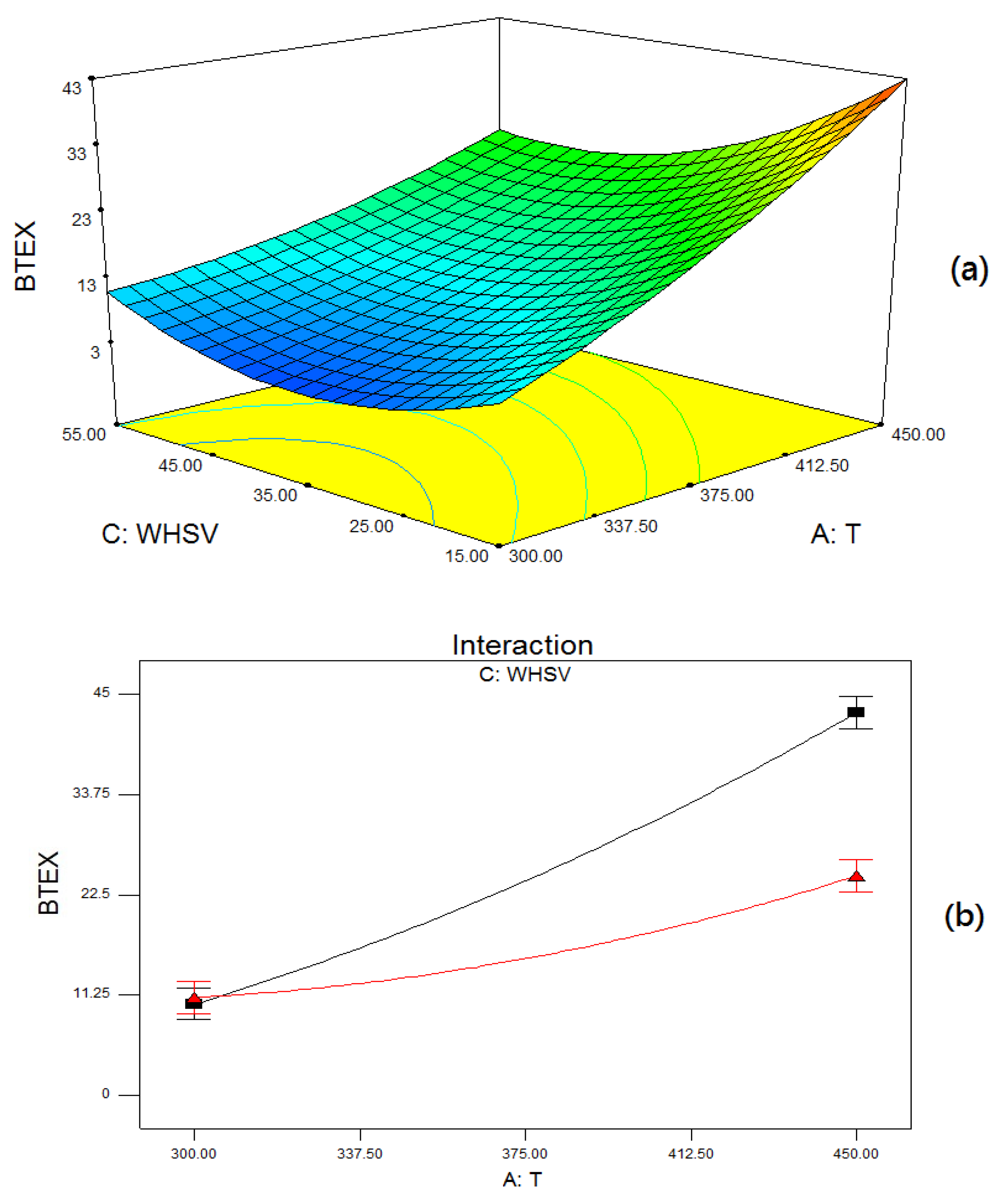
2.4. Reaction Parameter Study
2.5. Optimization of BTEX and Ethylene Production
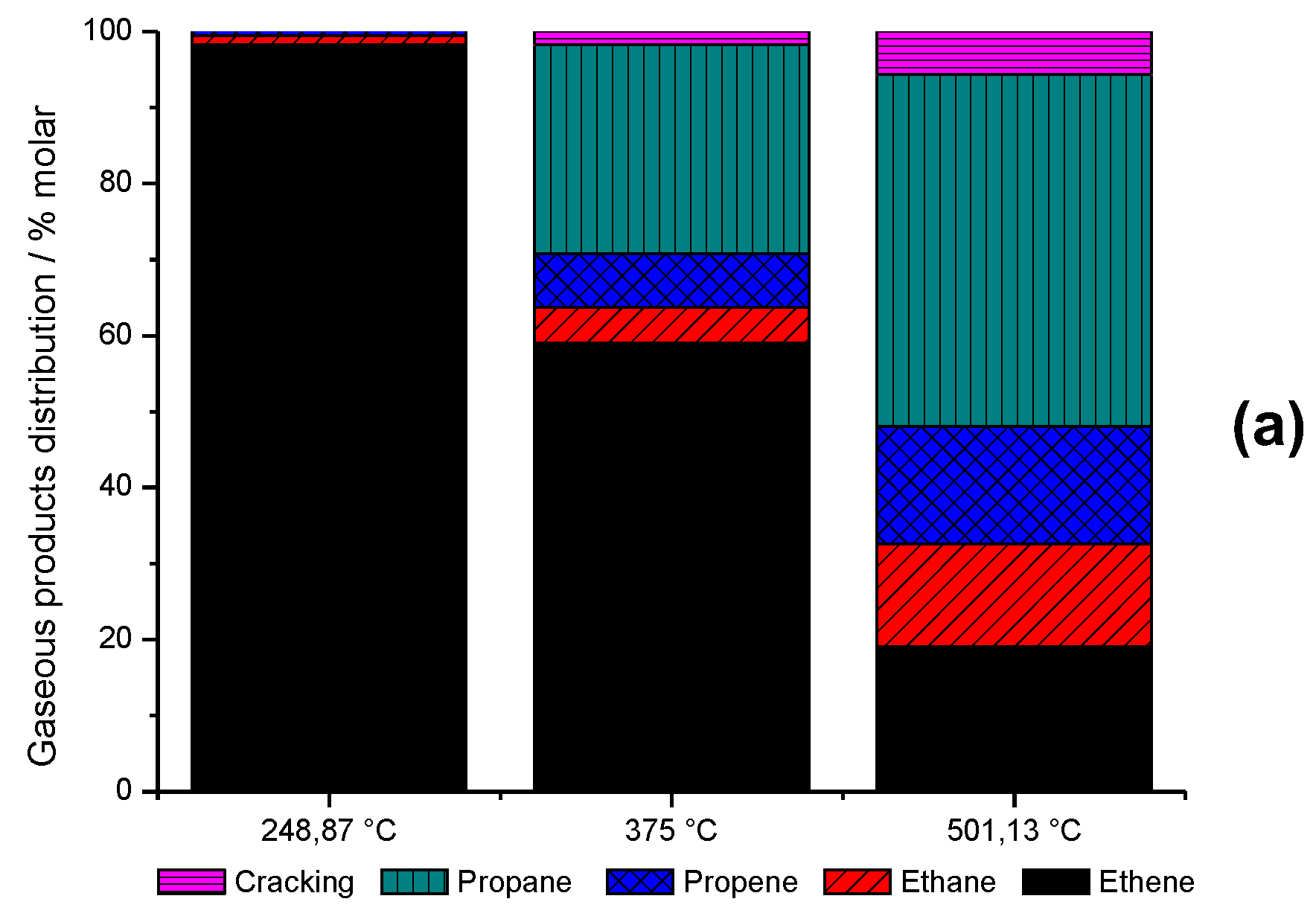
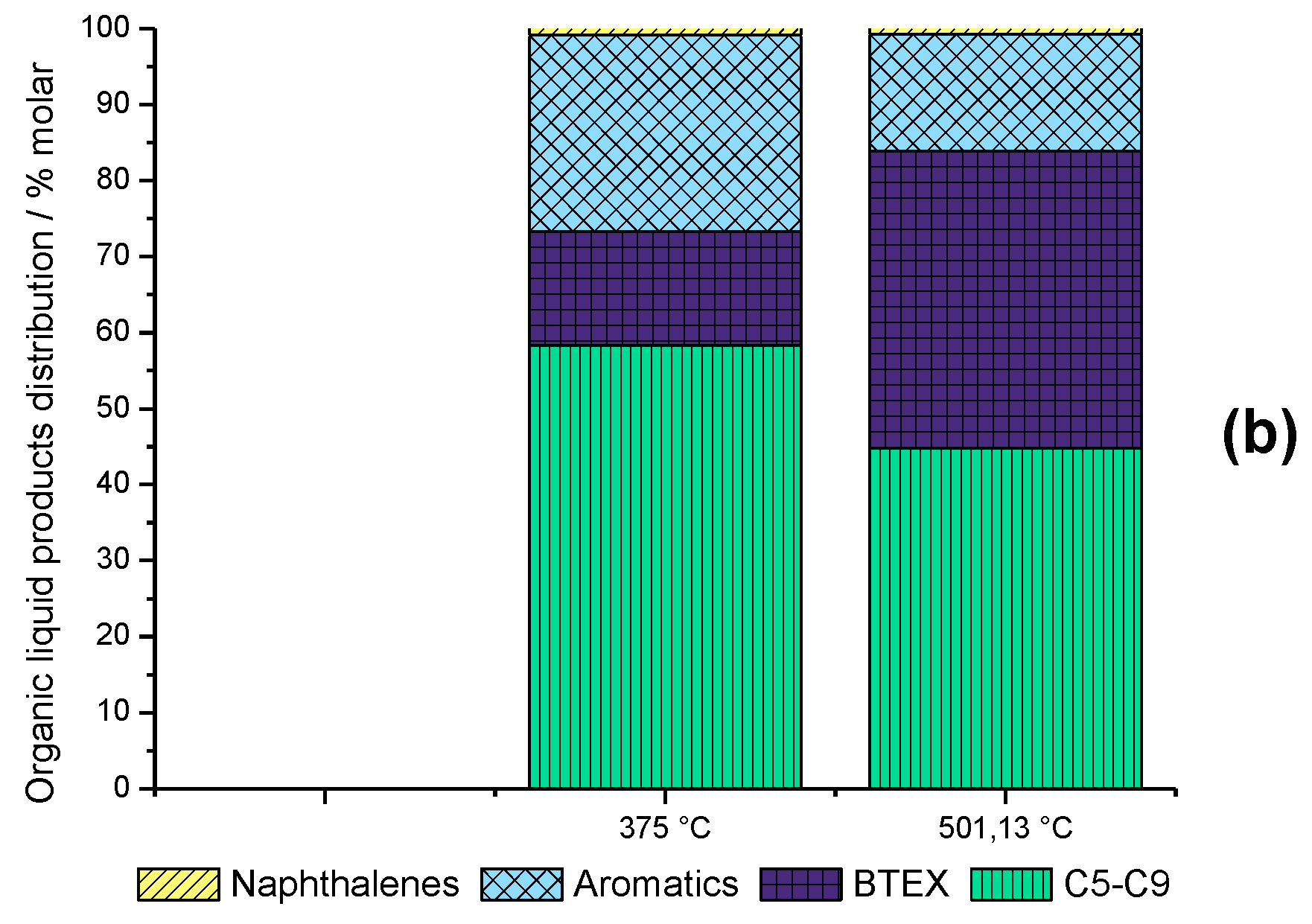
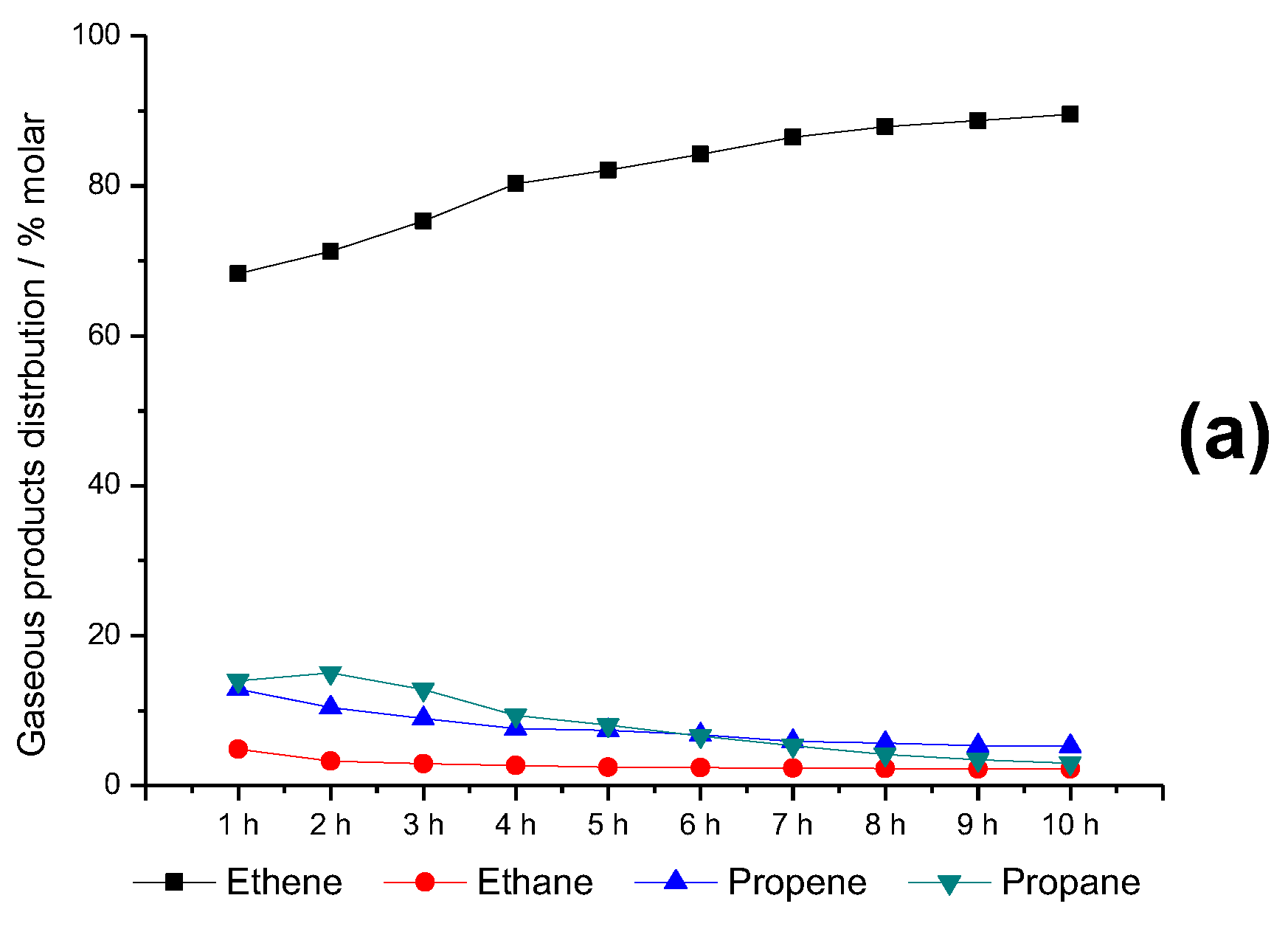
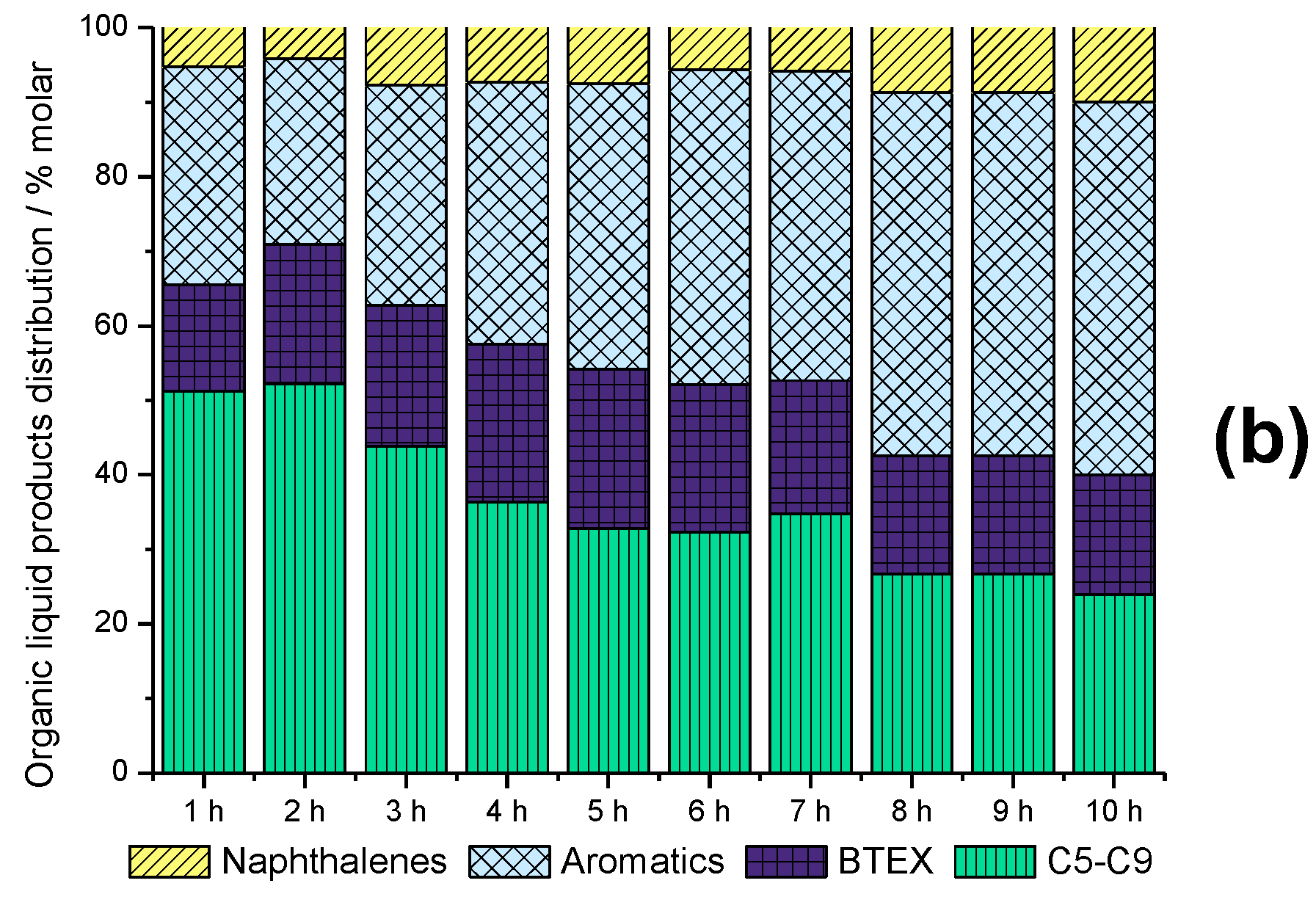
2.6. Time-on-Stream Evaluation of HZSM-5_(S50)
3. Materials and Methods
3.1. Catalyst Characterization
3.2. Catalytic Evaluation
3.3. Experiment Design and Statistical Study
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Lovón-Quintana, J.L.; Rodriguez-Guerrero, J.K.; Valença, P.G. Carbonate hydroxyapatiteas a catalyst for etanol conversion to hydrocarbons fuels. Appl. Catal. A 2017, 542, 136–145. [Google Scholar] [CrossRef]
- Yan, T.; Yang, L.; Dai, W.; Wang, C.; Wu, G.; Guan, N.; Hunger, M.; Li, L. On the deactivation mechanism of zeolite catalyst in etanol to butadiene conversion. J. Catal. 2018, 367, 7–15. [Google Scholar] [CrossRef]
- Boldog, I.; Čičmanec, P.; Ganjkhanlou, Y.; Bulánek, R. Surfactant template synthesis of porous VOx-ZrO2 catalysts for ethanol conversion to acetaldehyde. Catal. Today 2018, 304, 64–71. [Google Scholar] [CrossRef]
- Xia, W.; Wang, F.; Mu, X.; Chen, K. Remarkably enhanced selectivity for conversion of ethanol to propylene over ZrO2 catalysts. Fuel Process. Technol. 2017, 166, 140–145. [Google Scholar] [CrossRef]
- Wang, W.-C.; Tao, L. Bio-jet fuel conversion technologies. Renew. Sust. Energ. Rev. 2016, 53, 801–822. [Google Scholar] [CrossRef]
- Yang, J.; Xin, Z.; He, Q.S.; Corscadden, K.; Niu, H. An overview on performance characteristics of bio-jet fuels. Fuel 2019, 237, 916–936. [Google Scholar] [CrossRef]
- Zhan, N.; Hu, Y.; Yu, D.; Han, Y.; Huang, H. Lanthanum-phosphorous modified HZSM-5 catalysts in dehydration of ethanol to ethylene: A comparative analysis. Catal. Commun. 2010, 11, 633–637. [Google Scholar] [CrossRef]
- Ramasamy, K.K.; Wag, Y. Ethanol conversion to hydrocarbons on HZSM-5: Effect of reaction conditions and Si/Al ratio on the product distributions. Catal Today 2014, 273, 89–99. [Google Scholar] [CrossRef]
- Coqueblin, H.; Richard, A.; Uzio, D.; Pinard, L.; Pouilloux, Y.; Epron, F. Effect of the metal promoter on the performances of H-ZSM5 in ethylene aromatization. Catal. Today 2016, 289, 62–69. [Google Scholar] [CrossRef]
- Sousa, Z.S.B.; Veloso, C.O.; Henriques, C.A.; Silva, V.T. Ethanol Conversion into olefins and aromatics over HZSM-5 zeolite: Influence of reaction conditions and surface reactions studies. J. Mol. Catal. A 2016, 422, 266–274. [Google Scholar] [CrossRef]
- Oliveira, T.K.R.; Rosset, M.; Perez-Lopez, O.W. Ethanol dehydration to diethyl ether over Cu-Fe/ZSM-5 catalysts. Catal. Commun. 2018, 104, 32–36. [Google Scholar] [CrossRef]
- Santos, L.R.M.; Silva, M.A.P.; Menezes, S.C.; Chinelatto, L.S.; Lam, Y.L. Creation of mesopores and structural re-organization in Beta zeolite during alkaline treatment. Microporous Mesoporous Mater. 2016, 226, 260–266. [Google Scholar] [CrossRef]
- Purova, R.; Narasimharao, K.; Ahmed, N.S.I.; Al-Thabaiti, S.; Al-Shehri, A.; Mokhtar, M.; Schwieger, W. Pillared HMCM-36 zeolite catalyst for biodiesel production by esterification of palmitic acid. J. Mol. Catal. A 2015, 406, 159–167. [Google Scholar] [CrossRef]
- Sun, K.; Lu, J.; Ma, L.; Hang, Y.; Fu, Z.; Ding, J. A comparative study on the catalytic performance of different types of zeolites for biodiesel production. Fuel 2015, 158, 848–854. [Google Scholar] [CrossRef]
- Possato, L.G.; Diniz, R.N.; Garetto, T.; Pulcinelli, S.H.; Santilli, C.V.; Martins, L. A comparative study of glycerol dehydration catalyzed by micro/mesoporous MFI zeolites. J. Catal. 2013, 300, 102–112. [Google Scholar] [CrossRef]
- Li, K.; Valla, J.; Garcia-Martinez, J. Realizing the Commercial Potential of Hierarchical Zeolites: New Opportunities in Catalytic Cracking. ChemCatChem 2014, 6, 46–66. [Google Scholar] [CrossRef]
- Garcia-Martinez, J.; Johnson, M.; Valla, J.; Li, K.; Ying, J.Y. Mesostructured zeolite Y-high hydrothermal stability and superior FCC catalytic performance. Catal. Sci. Technol. 2012, 2, 987–994. [Google Scholar] [CrossRef]
- In, H.; Li, X.; Fang, Y.; Yi, X.; Hu, W.; Chu, Y.; Zhang, F.; Zheng, A.; Zhang, H.; Li, X. Catalytic dehydration of ethanol over post-treated ZSM-5 zeolites. J. Catal. 2014, 312, 204–215. [Google Scholar]
- Phung, T.K.; Radikapratama, R.; Garbarino, G.; Lagazzo, A.; Riani, P.; Busca, G. Tuning of product selectivity in the conversion of ethanol to hydrocarbons over H-ZSM-5 based zeolite catalysts. Fuel Process. Technol. 2015, 137, 290–297. [Google Scholar] [CrossRef]
- Sheng, Q.; Guo, S.; Ling, K.; Zhao, L. Catalytic Dehydration of Ethanol to Ethylene over Alkali-Treated HZSM-5 Zeolites. J. Braz. Soc. 2014, 25, 1365–1371. [Google Scholar] [CrossRef]
- Choopun, W.; Jitkarnka, S. Catalytic activity and stability of HZSM-5 zeolite and hierarchical uniform mesoporous MSU-ZSM-5 material during bio-ethanol dehydration. J. Clean. Prod. 2016, 135, 368–378. [Google Scholar] [CrossRef]
- Pinard, L.; Tayeb, K.B.; Hamieh, S.; Vezin, H.; Canaff, C.; Maury, S.; Delpoux, O.; Pouilloux, Y. On the involvement of radical “coke” in the ethanol conversion to hydrocarbons over HZSM-5 zeolite. Catal. Today 2013, 218–219, 57–64. [Google Scholar] [CrossRef]
- Conceição, L.R.V.; Costa, C.E.F.; Flho, G.N.R.; Filho, E.R.; Zamian, J.R. Ethanolysis Optimization of Jupadi (Raphia taedigera Mart.) Oil to Biodiesel Using Response Surface Methodology. J. Braz. Soc. 2015, 26, 1321–1330. [Google Scholar]
- Tolosa, T.C.; Rogez, H.; Silva, E.M.; Souza, J.N. Optimization of Acid Hydrolysis of Myricetin-3-O-rhamnoside Using Response Surface Methodology. J. Braz. Soc. 2018, 29, 2475–2481. [Google Scholar] [CrossRef]
- Claus, T.; Palombini, S.V.; Carbonera, F.; Figueiredo, I.L.; Matsushita, M.; Visentainer, J.V. Response Surface Methodology Applied in the Study of Emulsion Formulations in the Presence of Leaves of Rosemary (Rosmarinus officinalis L.) as a Source of Natural Antioxidants. J. Braz. Soc. 2015, 26, 2097–2104. [Google Scholar]
- Groen, J.C.; Jansen, J.C.; Moulijn, J.A.; Pérez-Ramírez, J. Optimal Aluminum-Assisted Mesoporosity Development in MFI Zeolites by Desilication. J. Phys. Chem. 2004, 108, 13062–13065. [Google Scholar] [CrossRef]
- Groen, J.C.; Peffer, L.A.A.; Moulijn, J.A.; Pérez-Ramírez, J. Role of intrinsic zeolite properties on mesopore formation by desilication of MFI structures. Stud. Surf. Sci. Catal. 2005, 156, 401–408. [Google Scholar]
- Groen, J.C.; Peffer, L.A.A.; Moulijn, J.A.; Pérez-Ramírez, J. Mesoporosity development in ZSM-5 zeolite upon optimized desilication conditions in alkaline medium. Colloids Surf. A 2004, 241, 53–58. [Google Scholar] [CrossRef]
- Sedighi, M.; Towfighi, J. Methanol conversion over SAPO-34 catalysts; Systematic study of temperature, space-time, and initial gel composition on product distribution and stability. Fuel 2015, 153, 382–392. [Google Scholar] [CrossRef]
- Silva, G.F.; Camargo, F.L.; Ferreira, A.L.O. Application of response surface methodology for optimization of biodiesel production by transesterification of soybean oil with ethanol. Fuel Process Technol. 2011, 92, 407–413. [Google Scholar] [CrossRef]
- Jan, O.; Resende, F.L.P. Liquid hydrocarbon production via ethylene oligomerization over Ni-Hβ. Fuel Process Technol. 2018, 179, 269–276. [Google Scholar] [CrossRef]
- Palla, V.C.S.; Shee, D.; Maity, S.K. Conversion of n-butanol to gasoline range hydrocarbons, butylenes and aromatics. Appl. Catal. A 2016, 526, 28–36. [Google Scholar] [CrossRef]
- PROCAT. Available online: http://www.procat-ufrj.com/index.php/en (accessed on 5 April 2019).
- PID Eng&Tech. Available online: http://www.pidengtech.com/products-and-services/microactivity-effi (accessed on 5 April 2019).
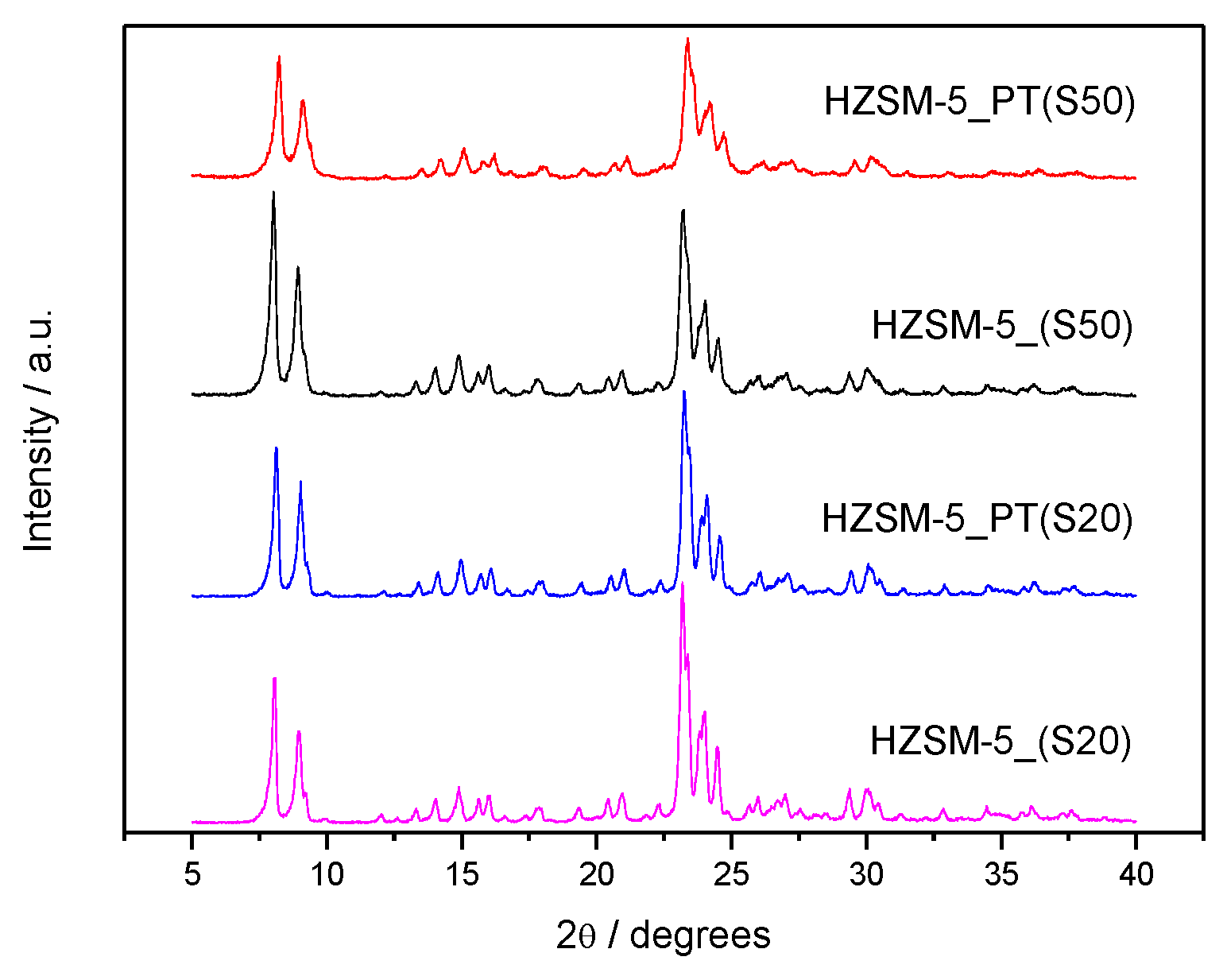
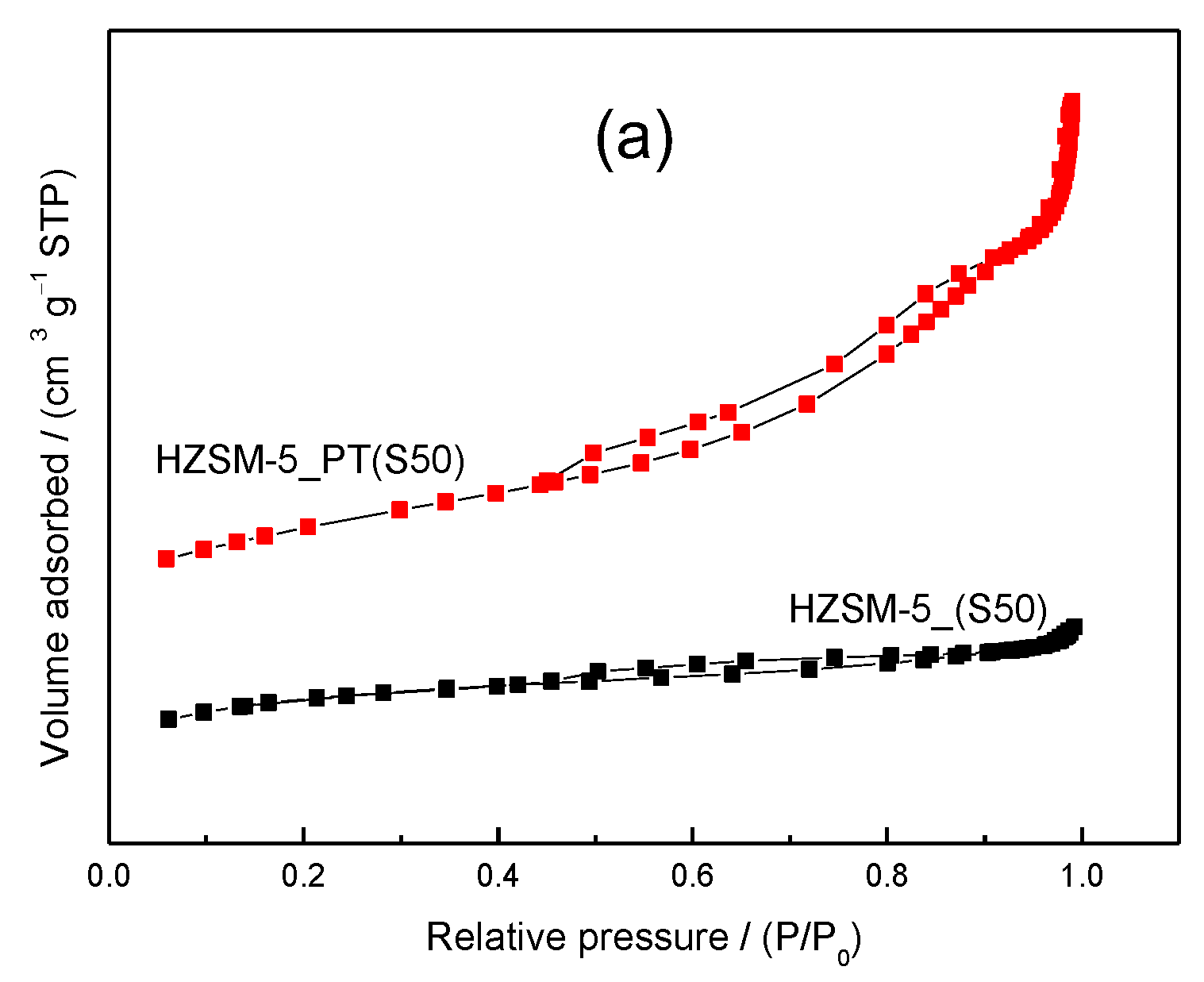
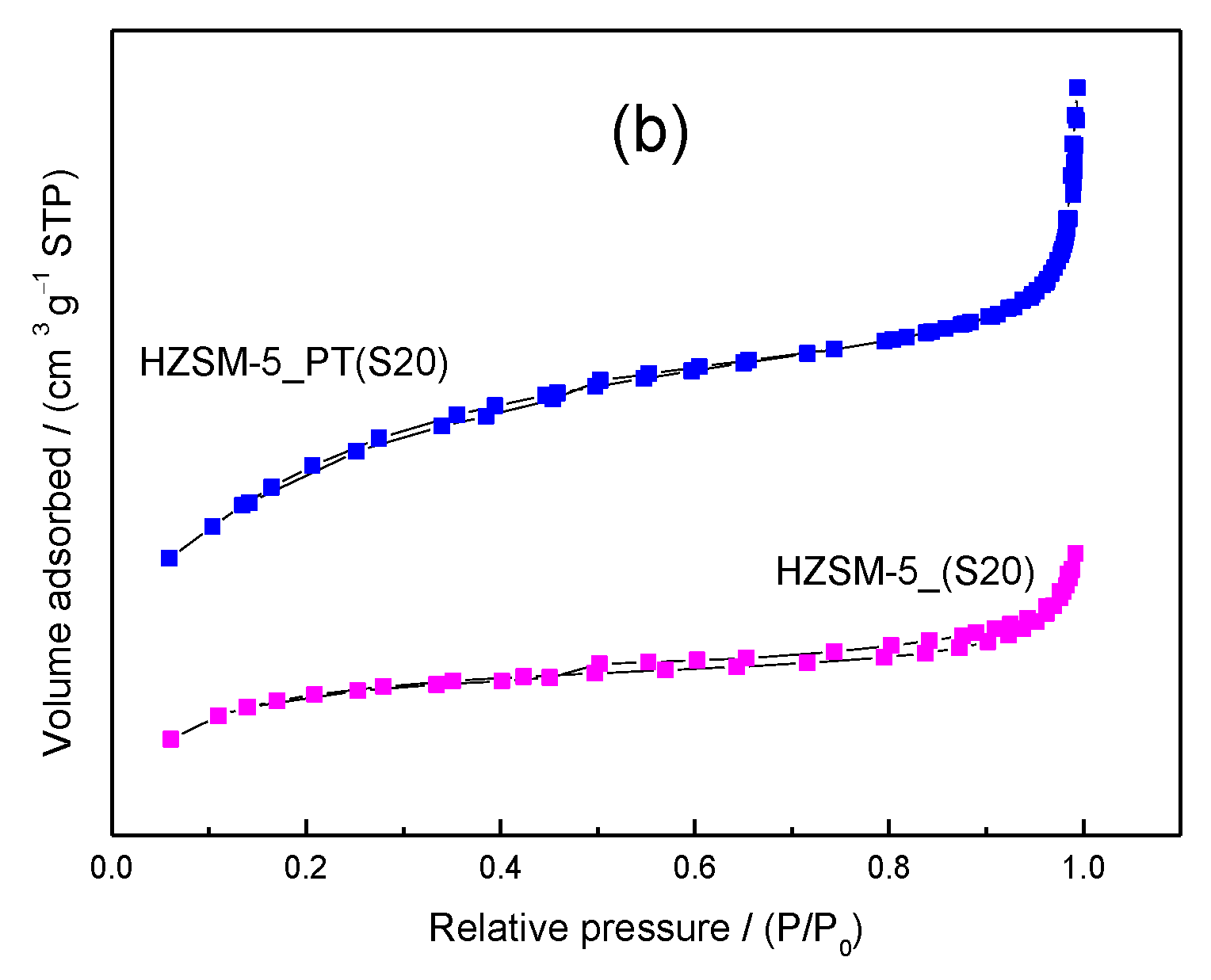

| Catalyst | SAR a (molar) | Cristallinity b (%) | SBET c (m2/g) | Smeso d (m2/g) | Vmicro d (cm3/g) | Vmeso e (cm3/g) |
|---|---|---|---|---|---|---|
| HZSM-5_PT(S50) | 38 | 93 | 491 | 212 | 0.10 | 0.37 |
| HZSM-5_(S50) | 53 | 100 | 419 | 61 | 0.18 | 0.07 |
| HZSM-5_PT(S20) | 23 | 99 | 352 | 42 | 0.11 | 0.04 |
| HZSM-5_(S20) | 23 | 100 | 337 | 12 | 0.16 | 0.02 |
| Source | Sum of Squares | Df c | Mean Square | F Value | p-Value Prob > F | Significance |
|---|---|---|---|---|---|---|
| Model | 2358.78 | 9 | 262.09 | 124.60 | <0.0001 | Highly significant |
| A-T | 1685.52 | 1 | 1685.52 | 801.30 | <0.0001 | Highly significant |
| B-P | 9.67 | 1 | 9.67 | 4.60 | 0.0644 | Not significant |
| C-WHSV | 140.53 | 1 | 140.53 | 66.81 | <0.0001 | Highly significant |
| AB | 0.02 | 1 | 0.02 | 0.01 | 0.9223 | Not significant |
| AC | 165.88 | 1 | 165.88 | 78.86 | <0.0001 | Highly significant |
| BC | 0.25 | 1 | 0.25 | 0.12 | 0.7385 | Not significant |
| A2 | 67.29 | 1 | 67.29 | 31.99 | 0.0005 | Significant |
| B2 | 0.26 | 1 | 0.26 | 0.12 | 0.7328 | Not significant |
| C2 | 182.46 | 1 | 182.46 | 86.74 | <0.0001 | Highly significant |
| Residual | 16.83 | 8 | 2.10 | |||
| Lack of Fit | 8.20 | 3 | 2.73 | 1.58 | 0.3041 | Not significant |
| Pure error | 8.63 | 5 | 1.73 | |||
| Std. Dev. a | 1.45 | R-squared | 0.9929 | |||
| Mean | 17.59 | Adj R-squared | 0.9849 | |||
| C.V.% b | 8.25 | Pred R-squared | 0.9432 | |||
| PRESS | 134.87 | Adeq Precision d | 39.1954 | |||
| (p < 0.0001) Highly significant; (0.0001 < p < 0.05) Significant; (p > 0.05) Not significant | ||||||
| Source | Sum of Squares | Df c | Mean Square | F Value | p-Value Prob > F | Significance |
|---|---|---|---|---|---|---|
| Model | 12470.23 | 6 | 2078.37 | 35.33 | <0.0001 | Highly significant |
| A-T | 11040.27 | 1 | 11040.27 | 187.68 | <0.0001 | Highly significant |
| B-P | 27.66 | 1 | 27.66 | 0.47 | 0.5071 | Not significant |
| C-WHSV | 822.15 | 1 | 822.15 | 13.98 | 0.0033 | Significant |
| AB | 41.86 | 1 | 41.86 | 0.71 | 0.4169 | Not significant |
| AC | 533.01 | 1 | 533.01 | 9.06 | 0.0119 | Significant |
| BC | 5.28 | 1 | 5.28 | 0.09 | 0.7700 | Not significant |
| Residual | 647.07 | 11 | 58.82 | |||
| Lack of Fit | 550.52 | 6 | 91.75 | 4.75 | 0.0541 | Not significant |
| Pure Error | 96.55 | 5 | 19.31 | |||
| Std. Dev. a | 7.67 | R-squared | 0.9507 | |||
| Mean | 62.64 | Adj R-squared | 0.9238 | |||
| C.V.% b | 12.24 | Pred R-squared | 0.8001 | |||
| PRESS | 2622.72 | Adeq Precision d | 19.9952 | |||
| (p < 0.0001) Highly significant; (0.0001 < p < 0.05) Significant; (p > 0.05) Not significant | ||||||
| Factor | Coding | Units | Levels | ||||
|---|---|---|---|---|---|---|---|
| −1.682 | −1 | 0 | 1 | 1.682 | |||
| Temperature | A | °C | 248.9 | 300.0 | 375.0 | 450.0 | 501.1 |
| Pressure | B | Bar | 1.6 | 5.0 | 10.0 | 15.0 | 18.4 |
| WHSV | C | h−1 | 1.4 | 15.0 | 35.0 | 55.0 | 68.6 |
| Point | Std a | T (°C) | P (bar) | WHSV (h−1) | BTEX (mol%) | C2 = (mol%) |
|---|---|---|---|---|---|---|
| Factorial | 1 | 300.0(−1) | 5.0(−1) | 15.0(−1) | 11.9 | 92.0 |
| Factorial | 2 | 450.0(+1) | 5.0(−1) | 15.0(−1) | 45.0 | 15.7 |
| Factorial | 3 | 300.0(−1) | 15.0(+1) | 15.0(−1) | 9.0 | 94.8 |
| Factorial | 4 | 450.0(+1) | 15.0(+1) | 15.0(−1) | 41.0 | 10.9 |
| Factorial | 5 | 300.0(−1) | 5.0(−1) | 55.0(+1) | 12.7 | 96.8 |
| Factorial | 6 | 450.0(+1) | 5.0(−1) | 55.0(+1) | 25.9 | 54.7 |
| Factorial | 7 | 300.0(−1) | 15.0(+1) | 55.0(+1) | 9.7 | 97.9 |
| Factorial | 8 | 450.0(+1) | 15.0(+1) | 55.0(+1) | 23.5 | 45.1 |
| Axial | 9 | 248.9(−1.682) | 10.0(0) | 35.0(0) | 0.0 | 98.3 |
| Axial | 10 | 501.1(+1.682) | 10.0(0) | 35.0(0) | 39.1 | 19.1 |
| Axial | 11 | 375.0(0) | 1.6(−1.682) | 35.0(0) | 12.8 | 46.7 |
| Axial | 12 | 375.0(0) | 18.4(1.682) | 35.0(0) | 13.0 | 64.5 |
| Axial | 13 | 375.0(0) | 10.0(0) | 1.4(−1.682) | 40.0 | 1.6 |
| Axial | 14 | 375.0(0) | 10.0(0) | 68.6(1.682) | 20.0 | 57.0 |
| Center | 15 | 375.0(0) | 10.0(0) | 35.0(0) | 15.0 | 59.0 |
| Center | 16 | 375.0(0) | 10.0(0) | 35.0(0) | 12.6 | 64.8 |
| Center | 17 | 375.0(0) | 10.0(0) | 35.0(0) | 12.5 | 63.9 |
| Center | 18 | 375.0(0) | 10.0(0) | 35.0(0) | 11.6 | 62.9 |
| Center | 19 | 375.0(0) | 10.0(0) | 35.0(0) | 12.0 | 70.9 |
| Center | 20 | 375.0(0) | 10.0(0) | 35.0(0) | 11.1 | 69.5 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Souza de Carvalho Filho, J.F.; Maciel Pereira, M.; Gomes Aranda, D.A.; Monnerat Araujo Ribeiro de Almeida, J.; Falabella Sousa-Aguiar, E.; Nothaft Romano, P. Application of Response Surface Methodology for Ethanol Conversion into Hydrocarbons Using ZSM-5 Zeolites. Catalysts 2019, 9, 617. https://doi.org/10.3390/catal9070617
Souza de Carvalho Filho JF, Maciel Pereira M, Gomes Aranda DA, Monnerat Araujo Ribeiro de Almeida J, Falabella Sousa-Aguiar E, Nothaft Romano P. Application of Response Surface Methodology for Ethanol Conversion into Hydrocarbons Using ZSM-5 Zeolites. Catalysts. 2019; 9(7):617. https://doi.org/10.3390/catal9070617
Chicago/Turabian StyleSouza de Carvalho Filho, José Faustino, Marcelo Maciel Pereira, Donato Alexandre Gomes Aranda, João Monnerat Araujo Ribeiro de Almeida, Eduardo Falabella Sousa-Aguiar, and Pedro Nothaft Romano. 2019. "Application of Response Surface Methodology for Ethanol Conversion into Hydrocarbons Using ZSM-5 Zeolites" Catalysts 9, no. 7: 617. https://doi.org/10.3390/catal9070617
APA StyleSouza de Carvalho Filho, J. F., Maciel Pereira, M., Gomes Aranda, D. A., Monnerat Araujo Ribeiro de Almeida, J., Falabella Sousa-Aguiar, E., & Nothaft Romano, P. (2019). Application of Response Surface Methodology for Ethanol Conversion into Hydrocarbons Using ZSM-5 Zeolites. Catalysts, 9(7), 617. https://doi.org/10.3390/catal9070617








