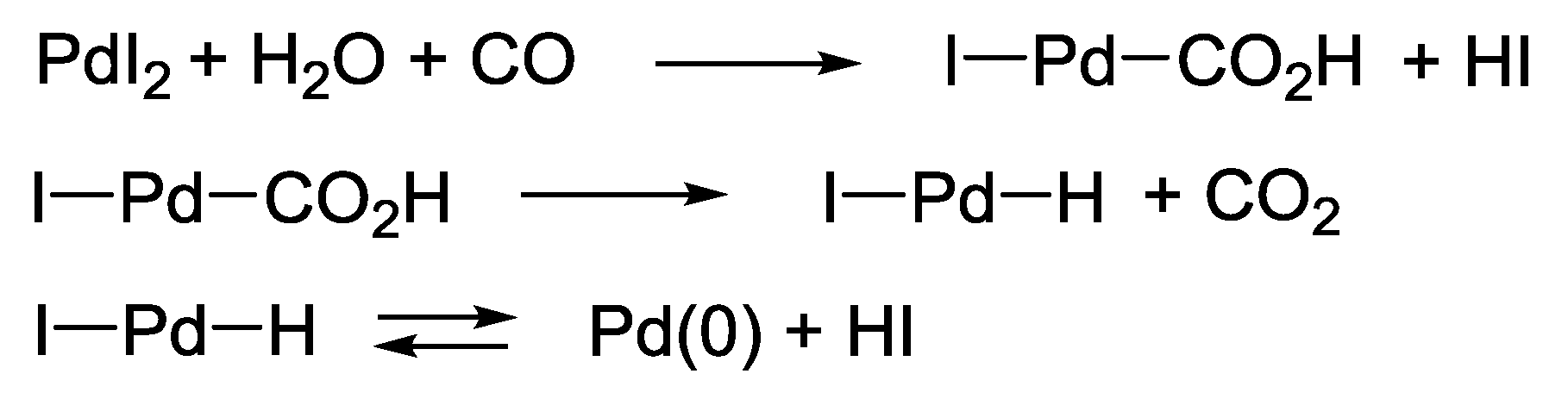PdI2-Based Catalysis for Carbonylation Reactions: A Personal Account
Abstract
1. Introduction
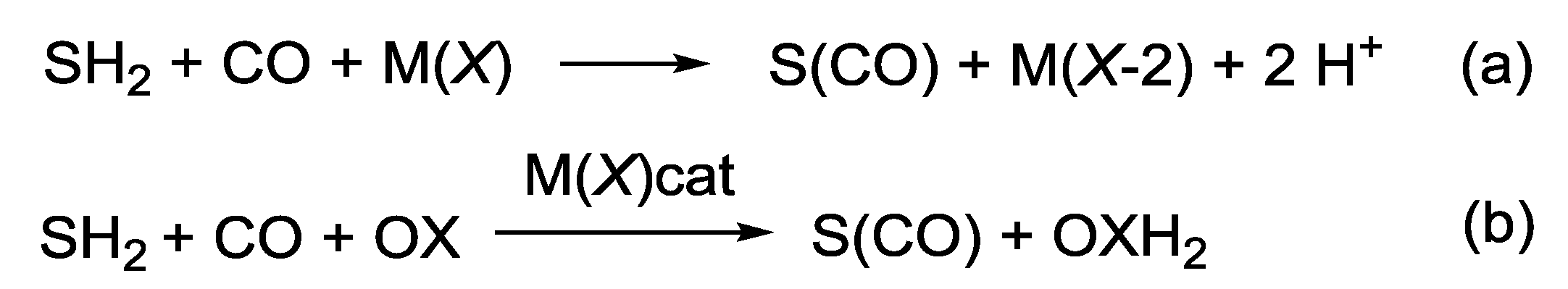
2. PdI2-Catalyzed Oxidative Carbonylations
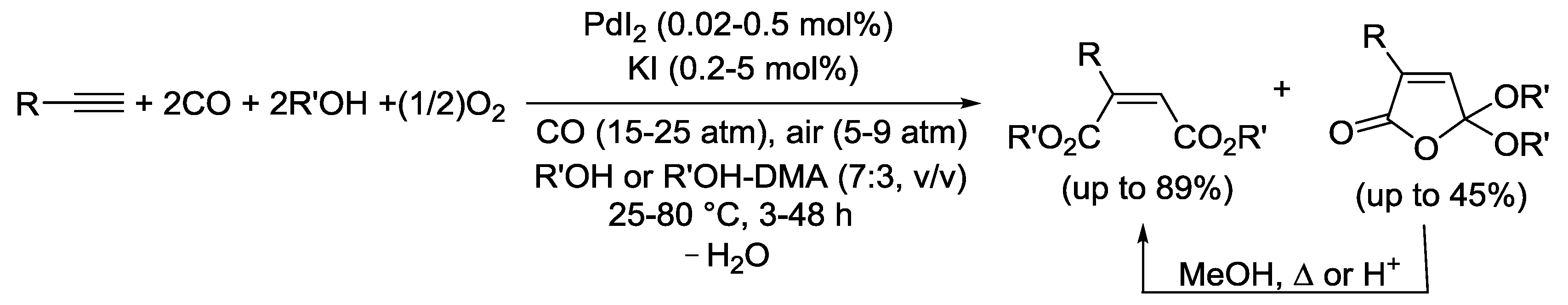
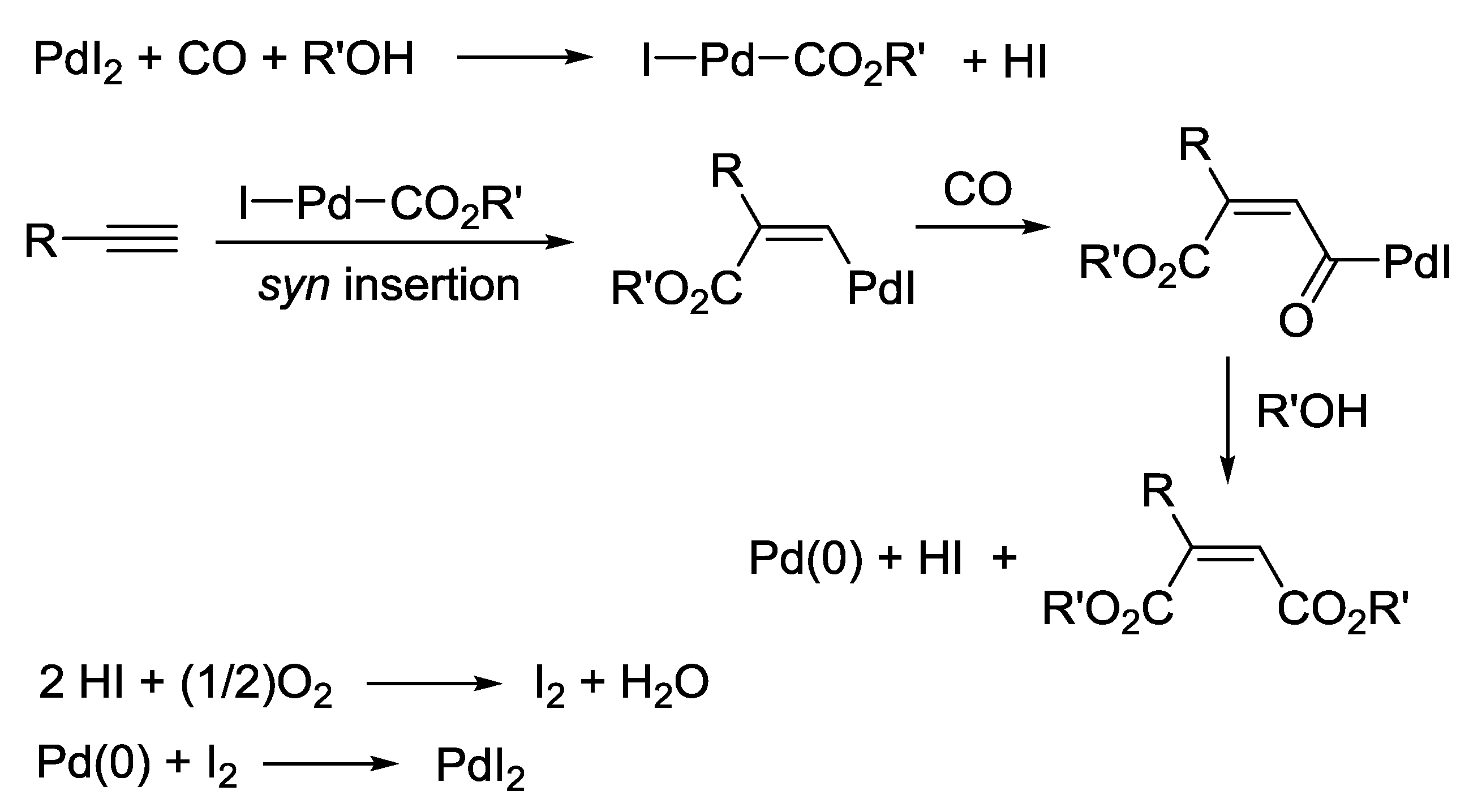
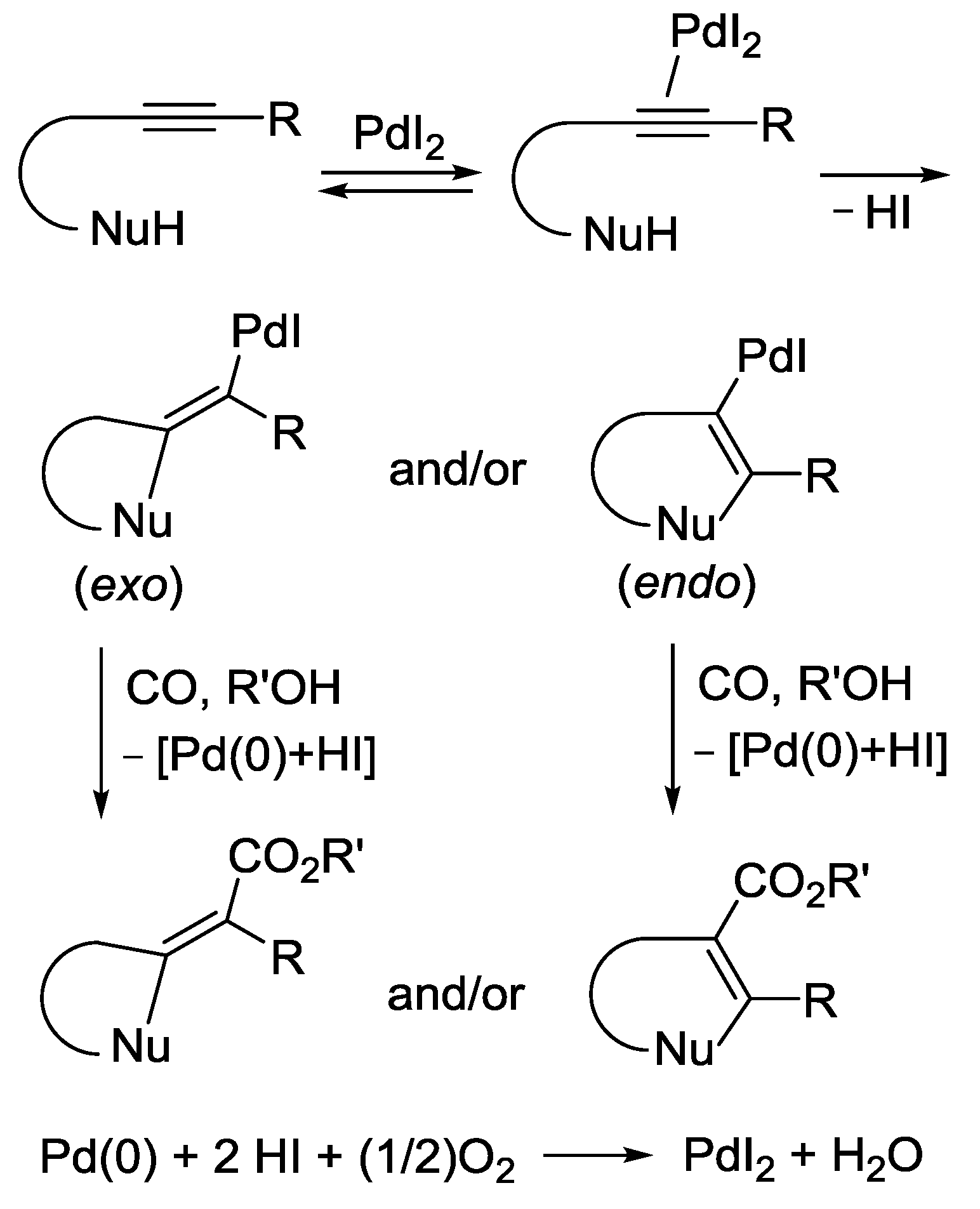
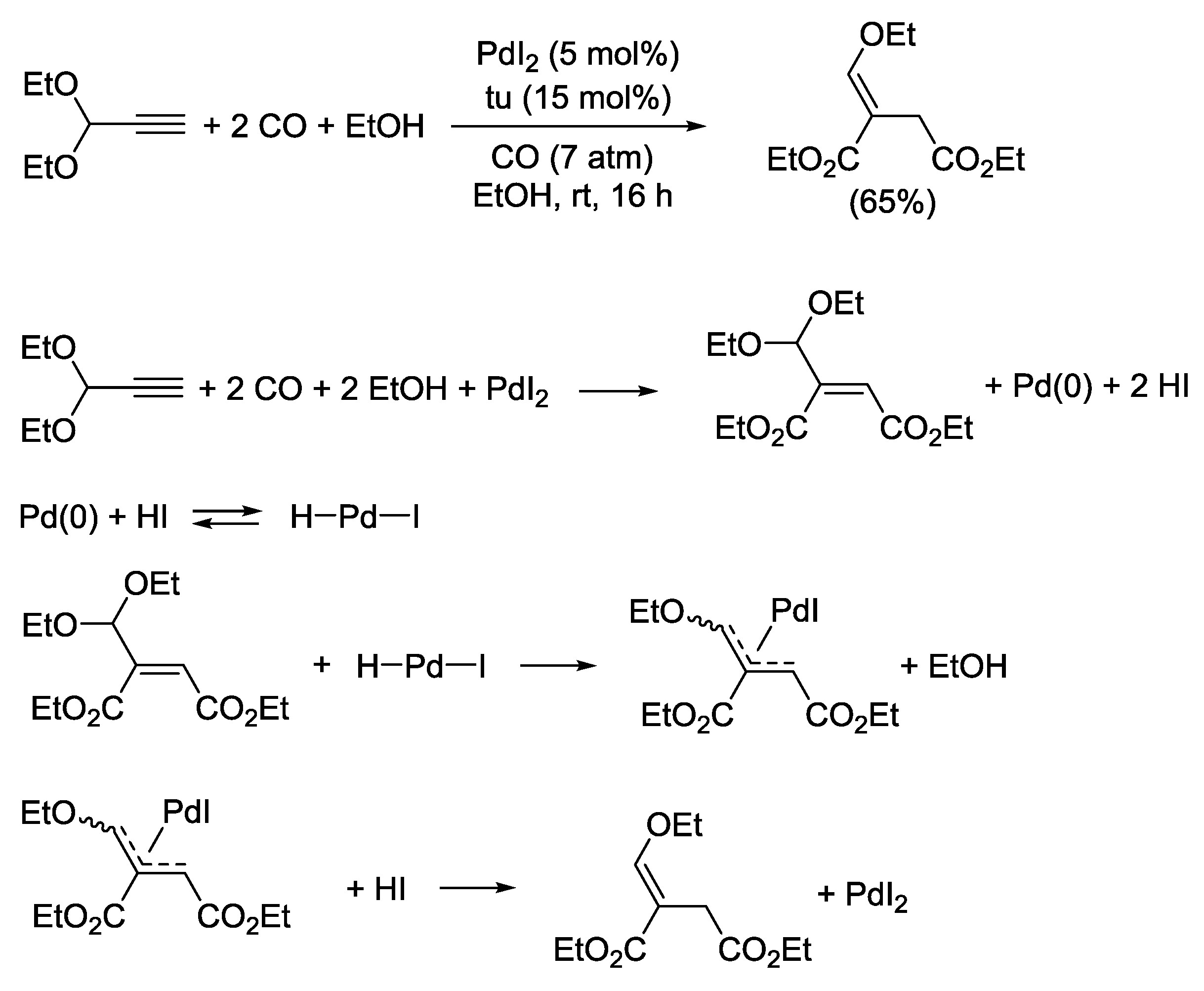
2.1. Background: PdI2-Catalyzed Oxidative Dialkoxycarbonylation of Alkynes
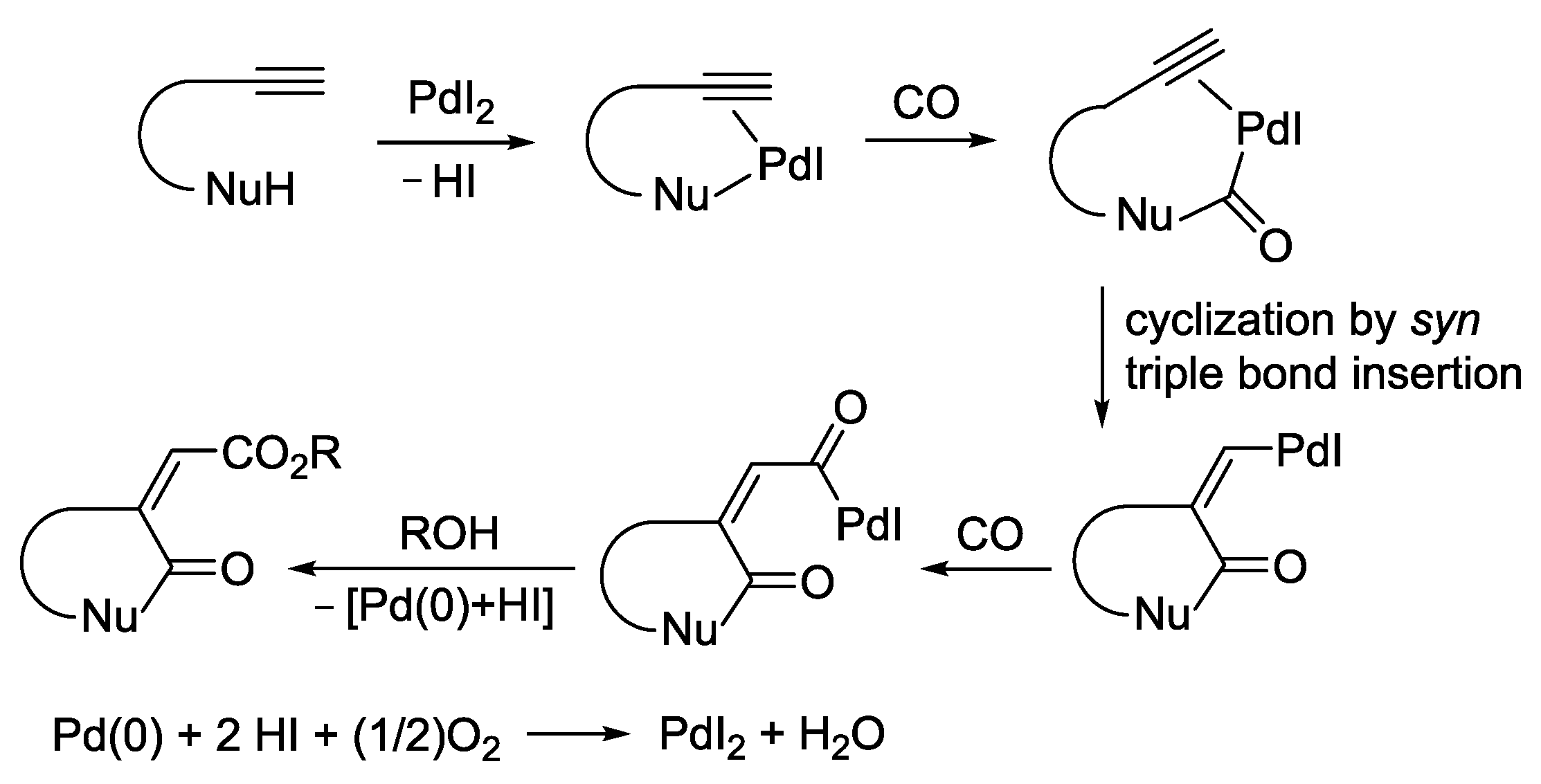
2.2. PdI2-Catalyzed Oxidative Cyclocarbonylation-Alkoxycarbonylation of Functionalized Alkynes
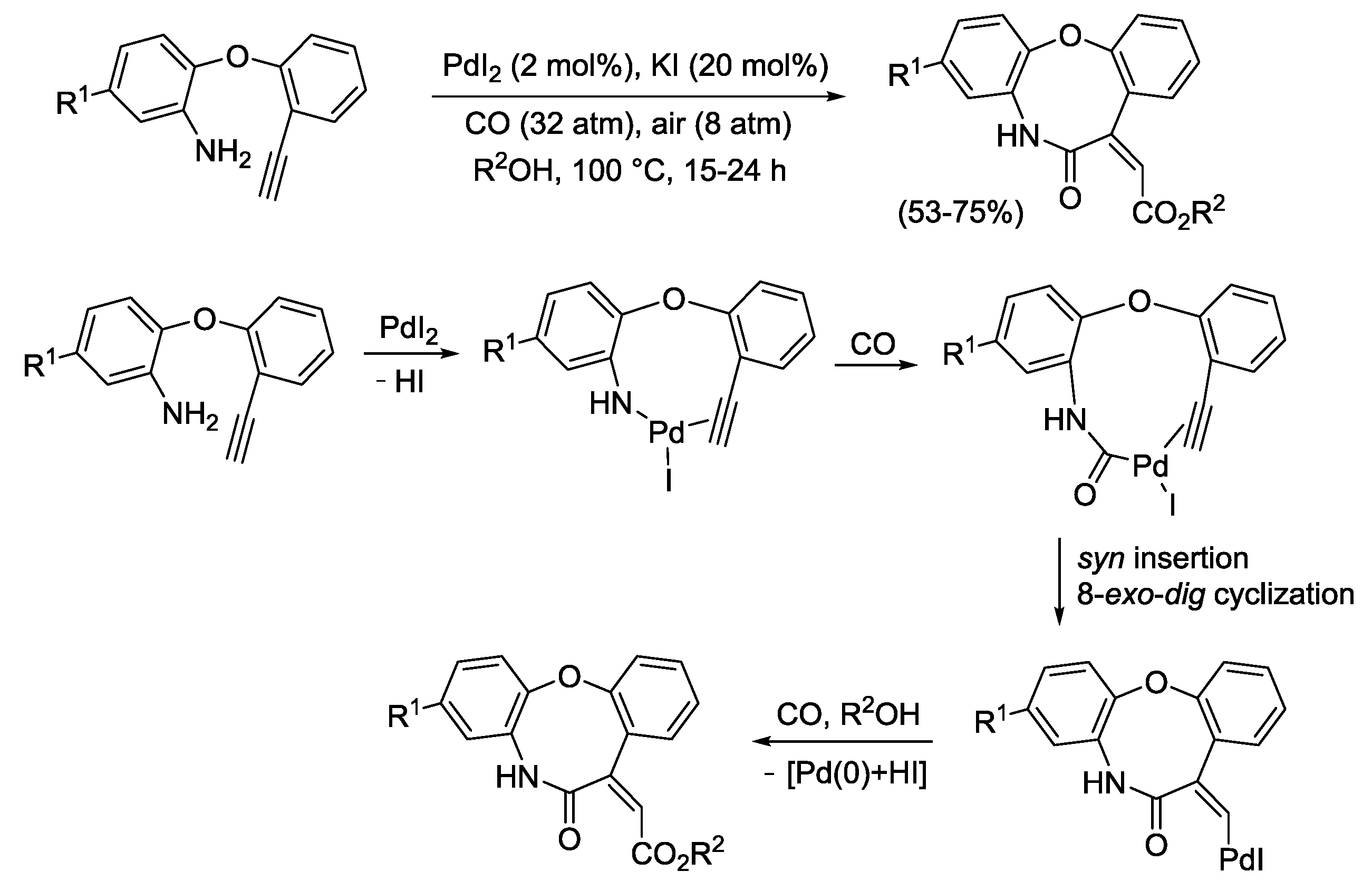
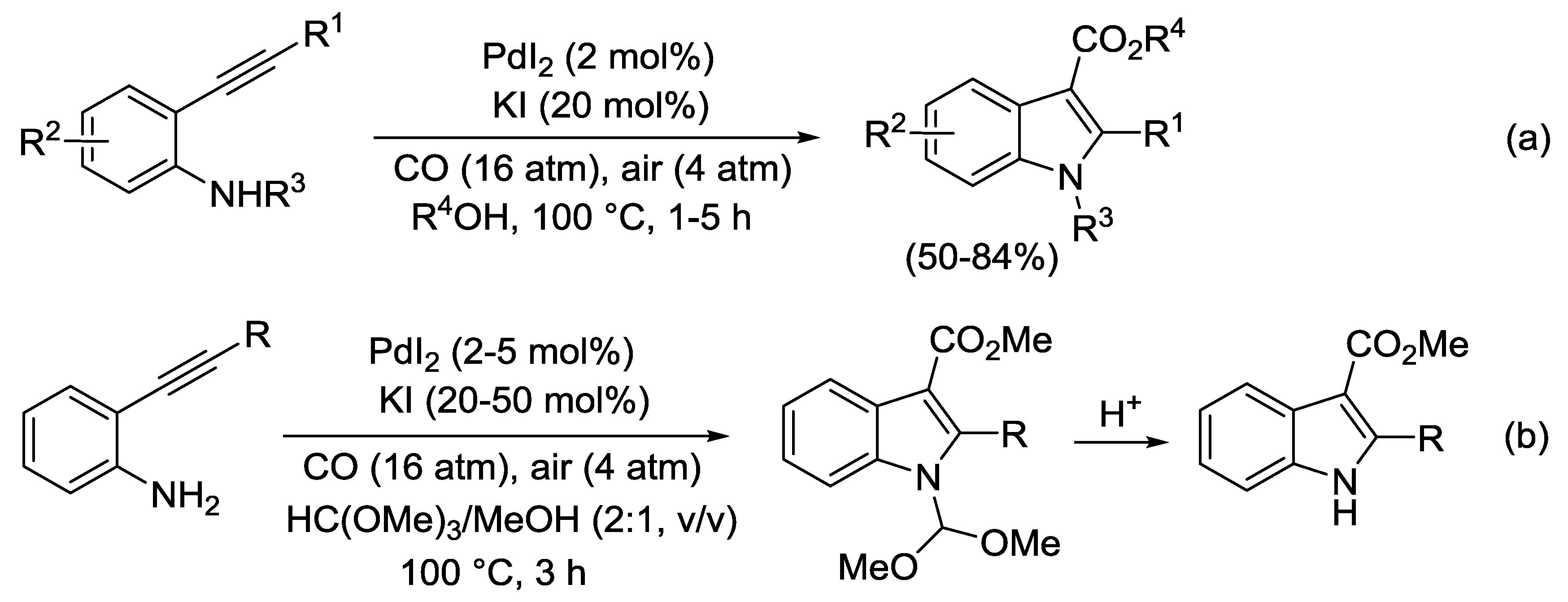
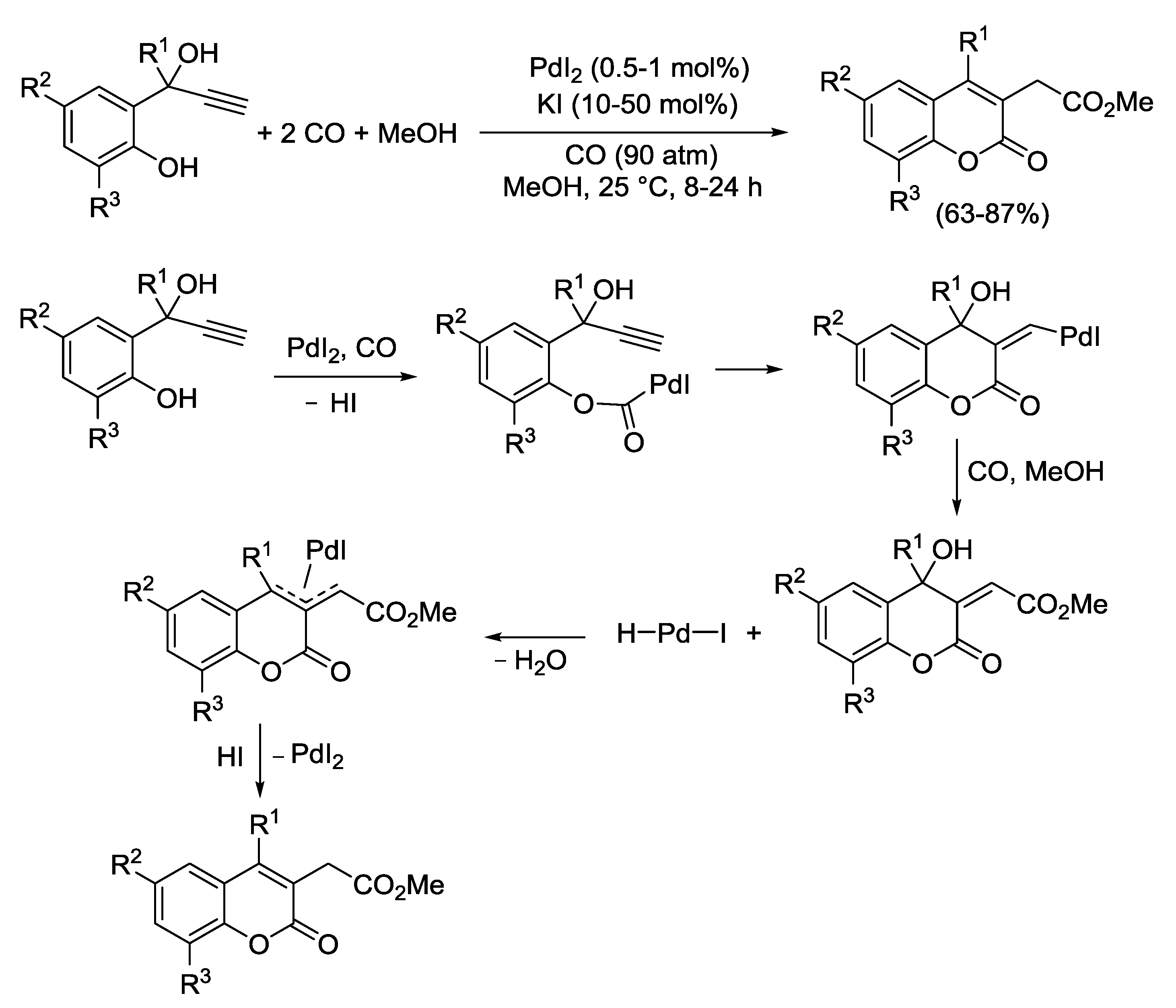
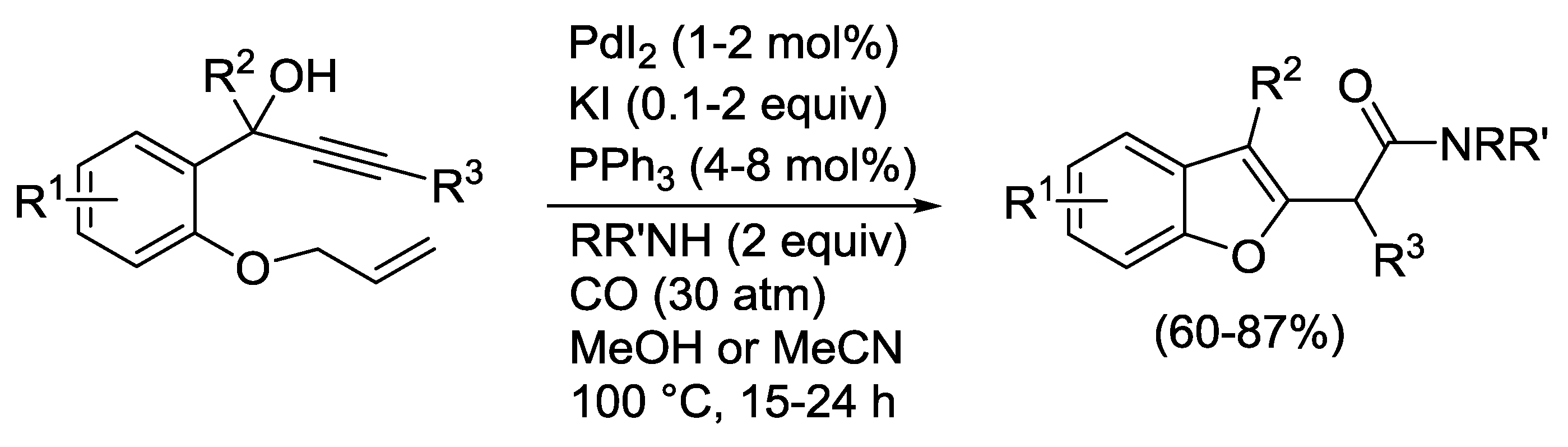
2.3. PdI2-Catalyzed Oxidative Heterocyclization-Alkoxycarbonylation of Functionalized Alkynes
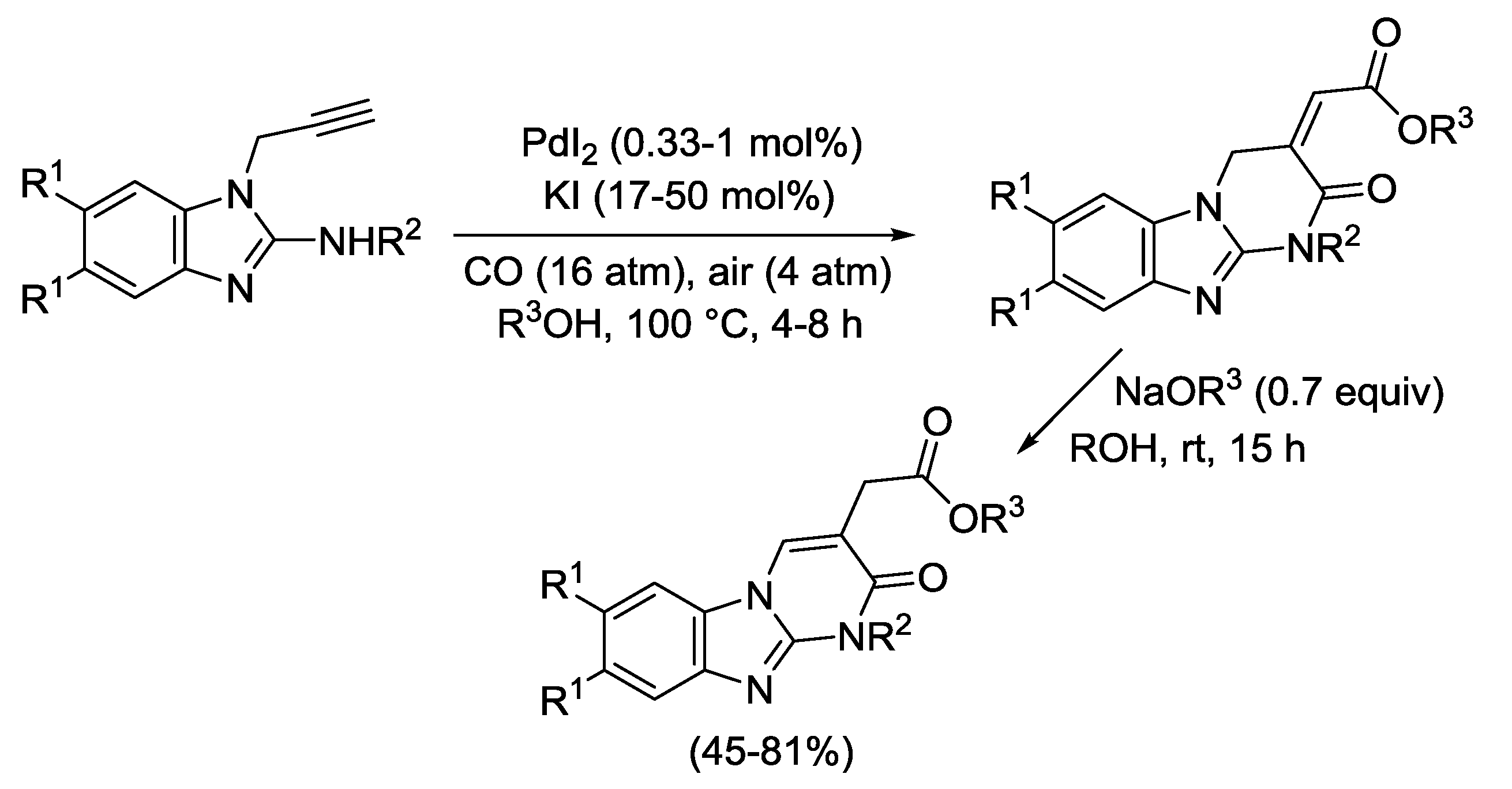
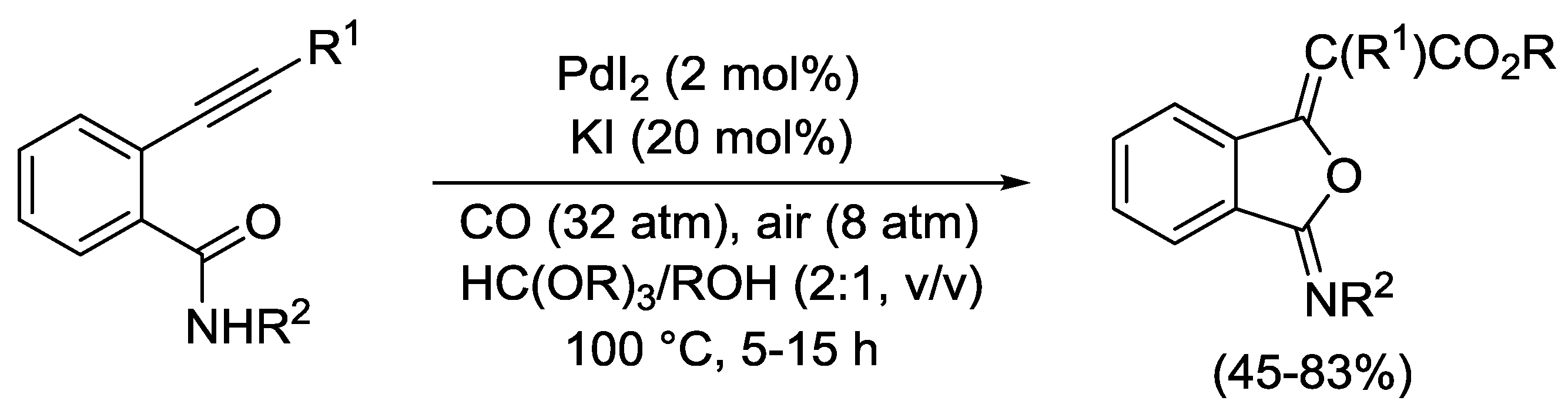
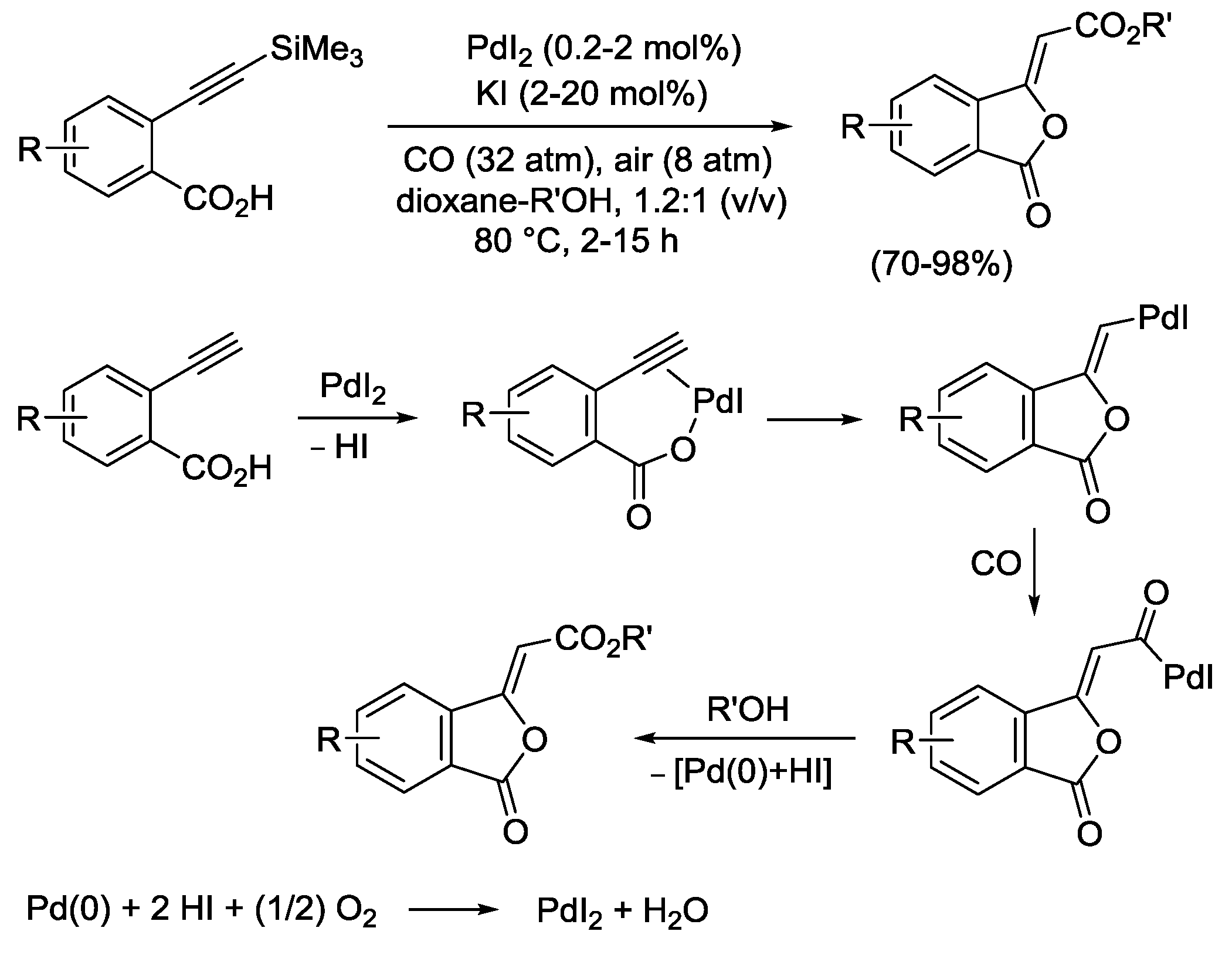
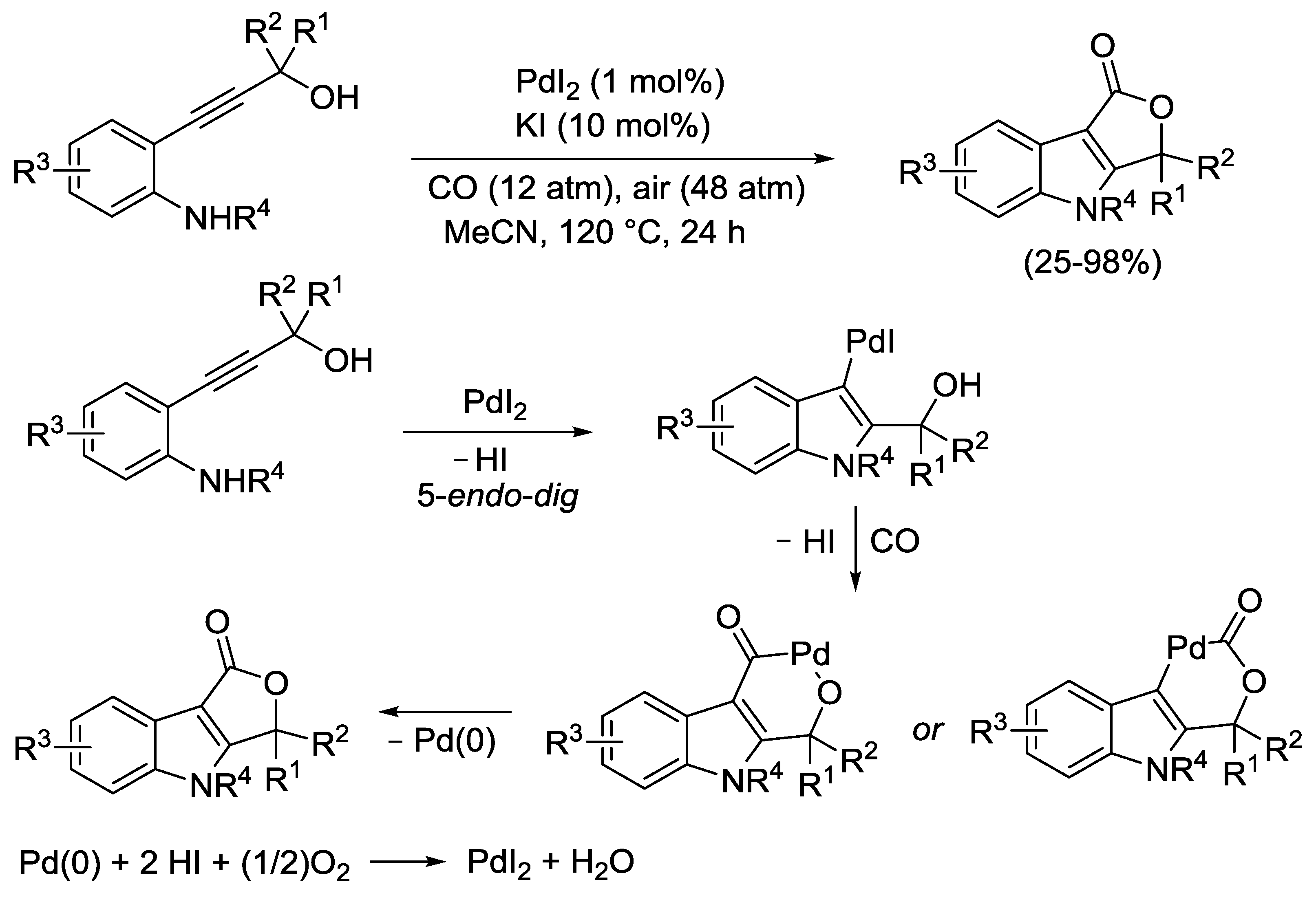
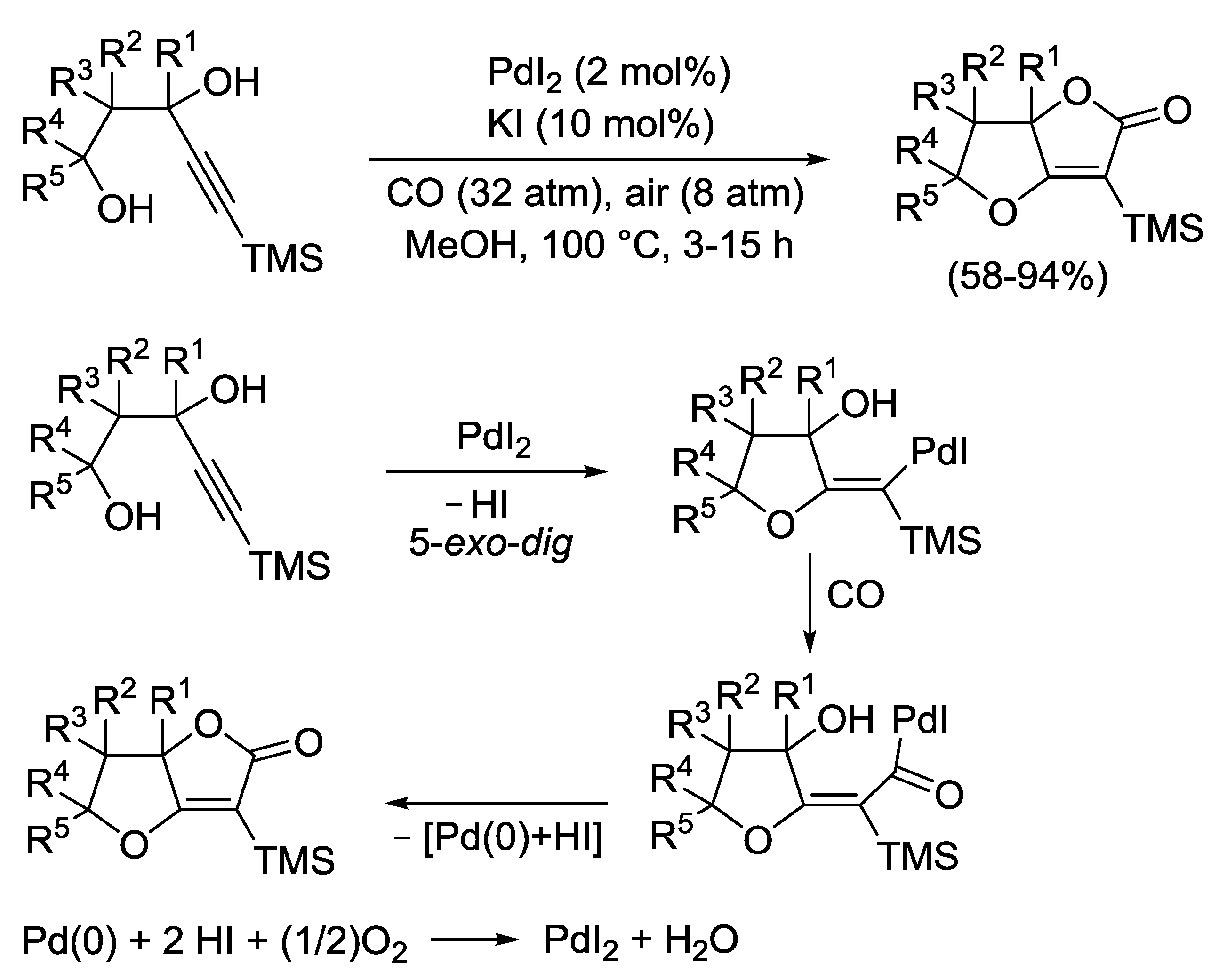
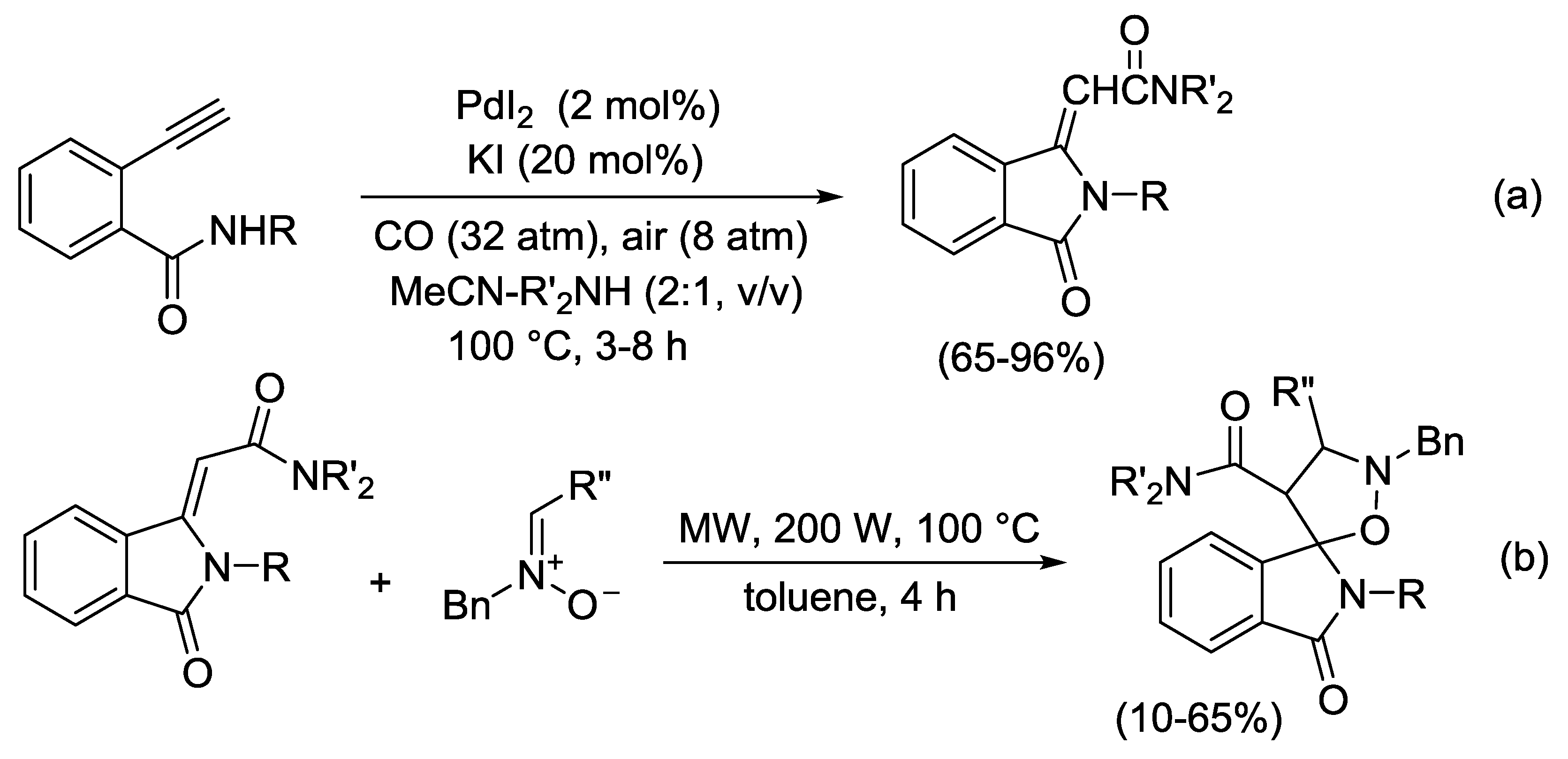
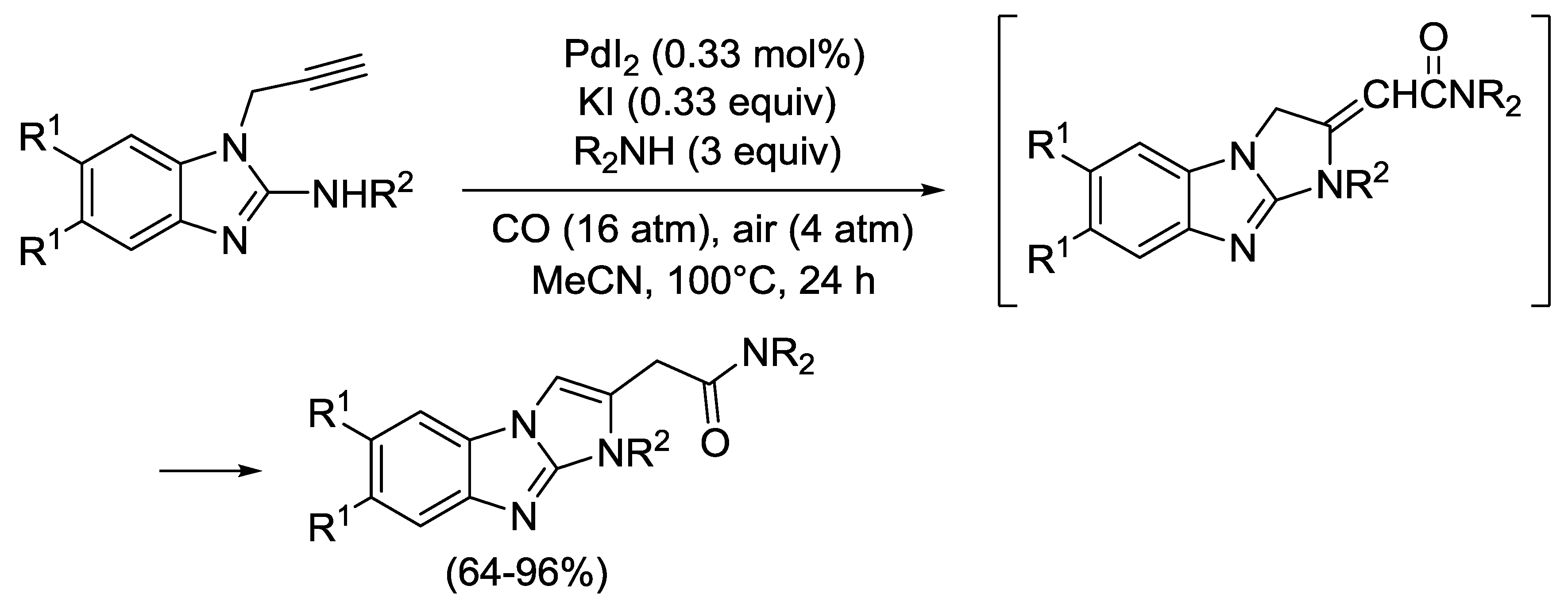
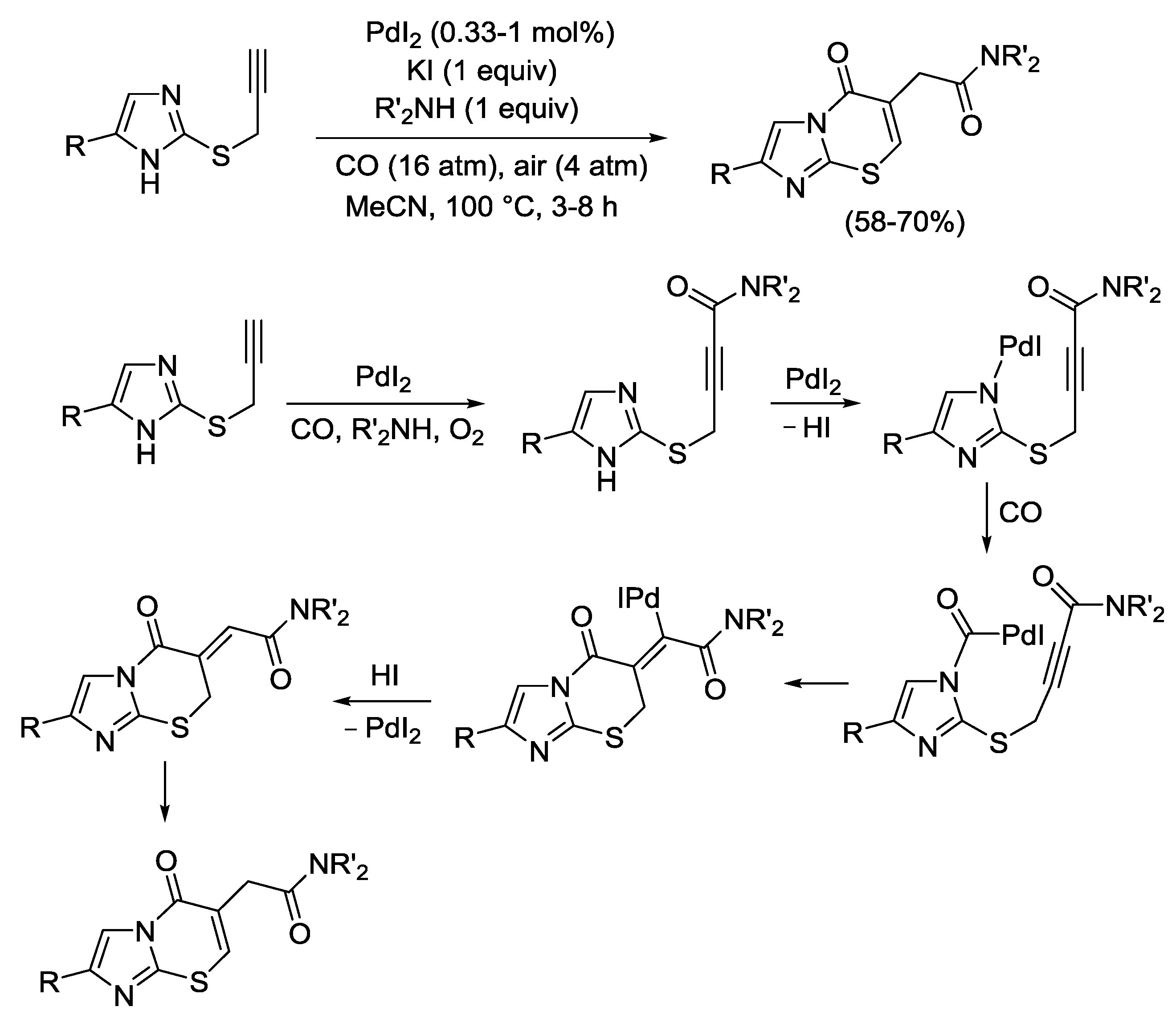
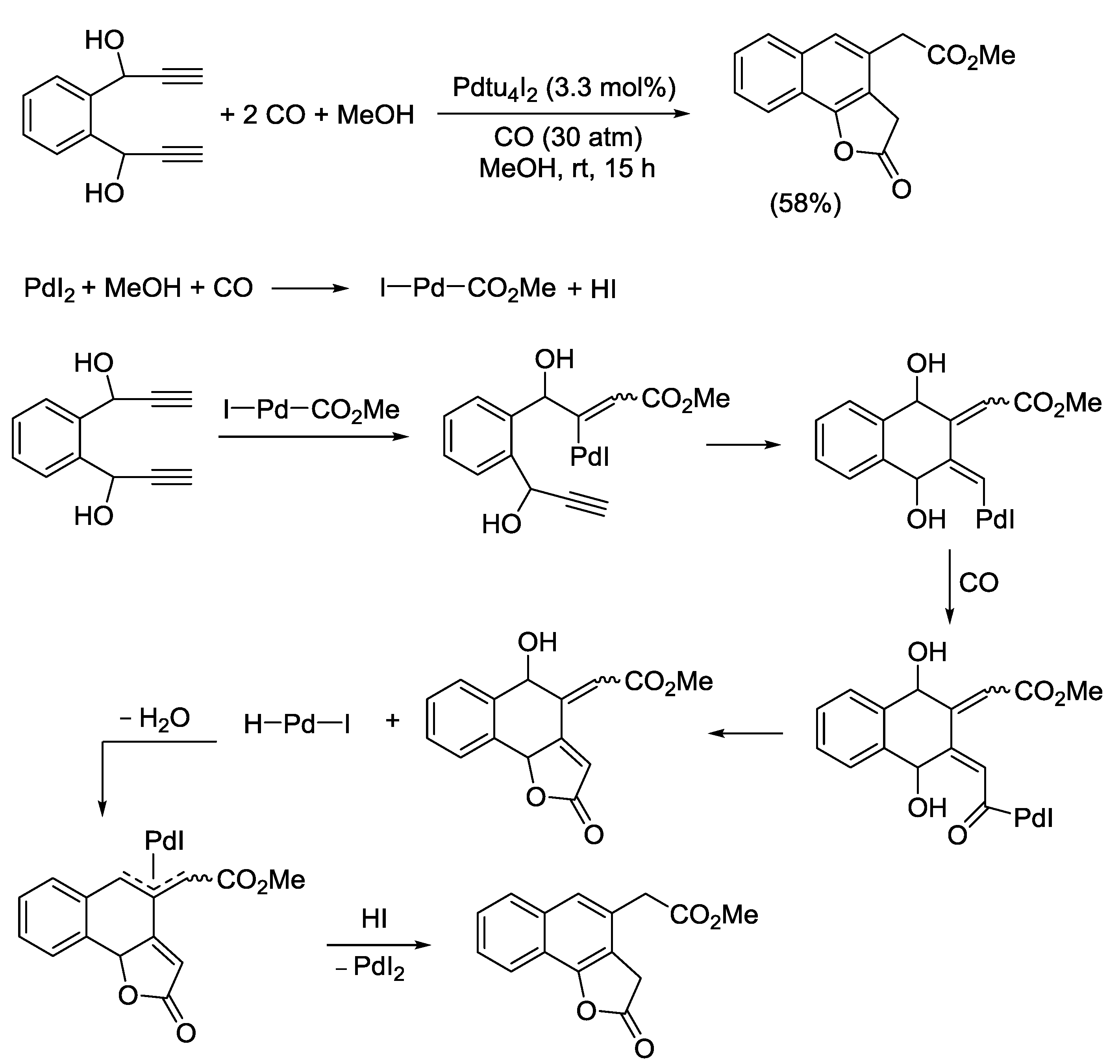
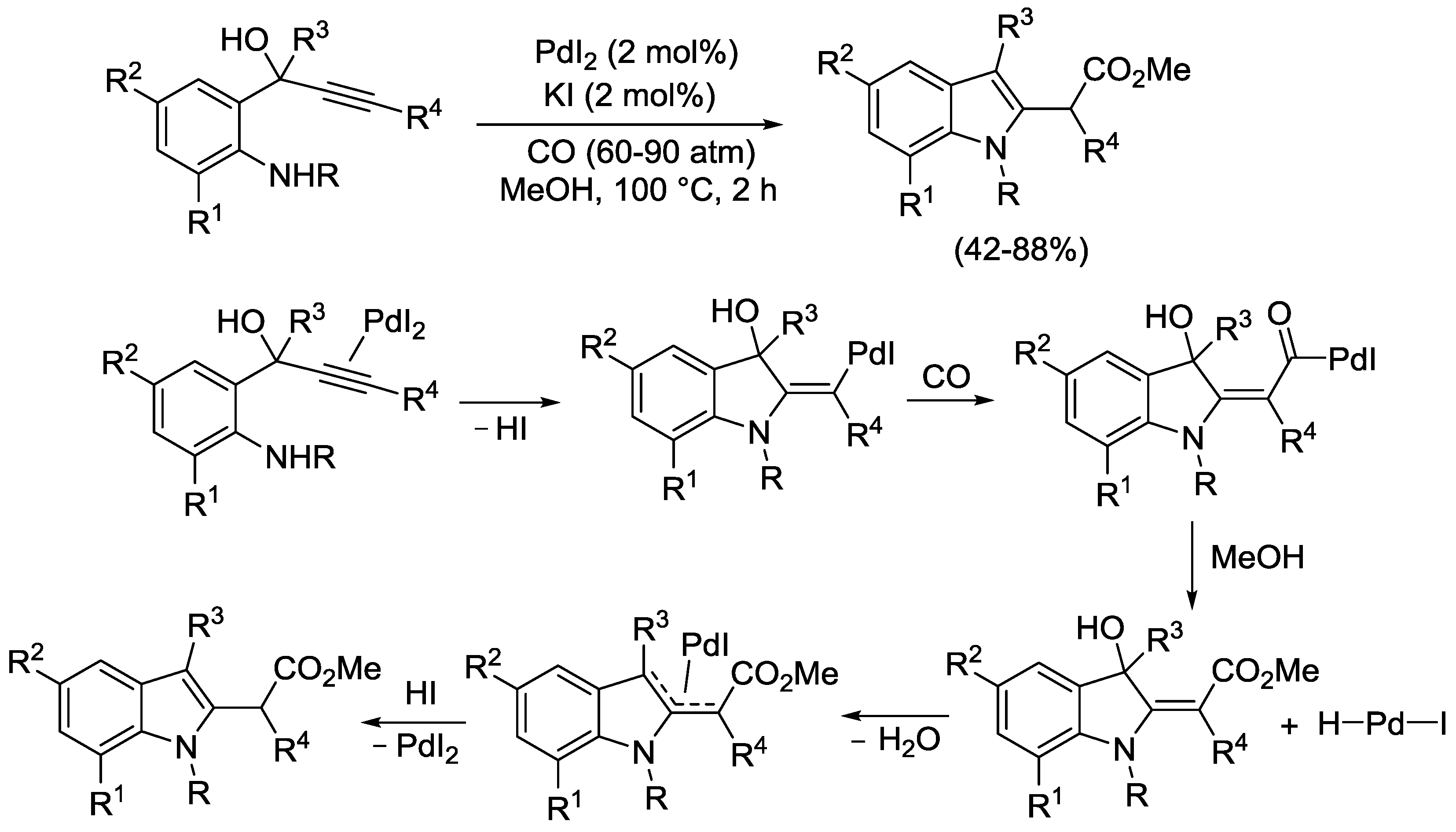
2.4. PdI2-Catalyzed Oxidative Carbonylative Double Cyclization of Functionalized Alkynes
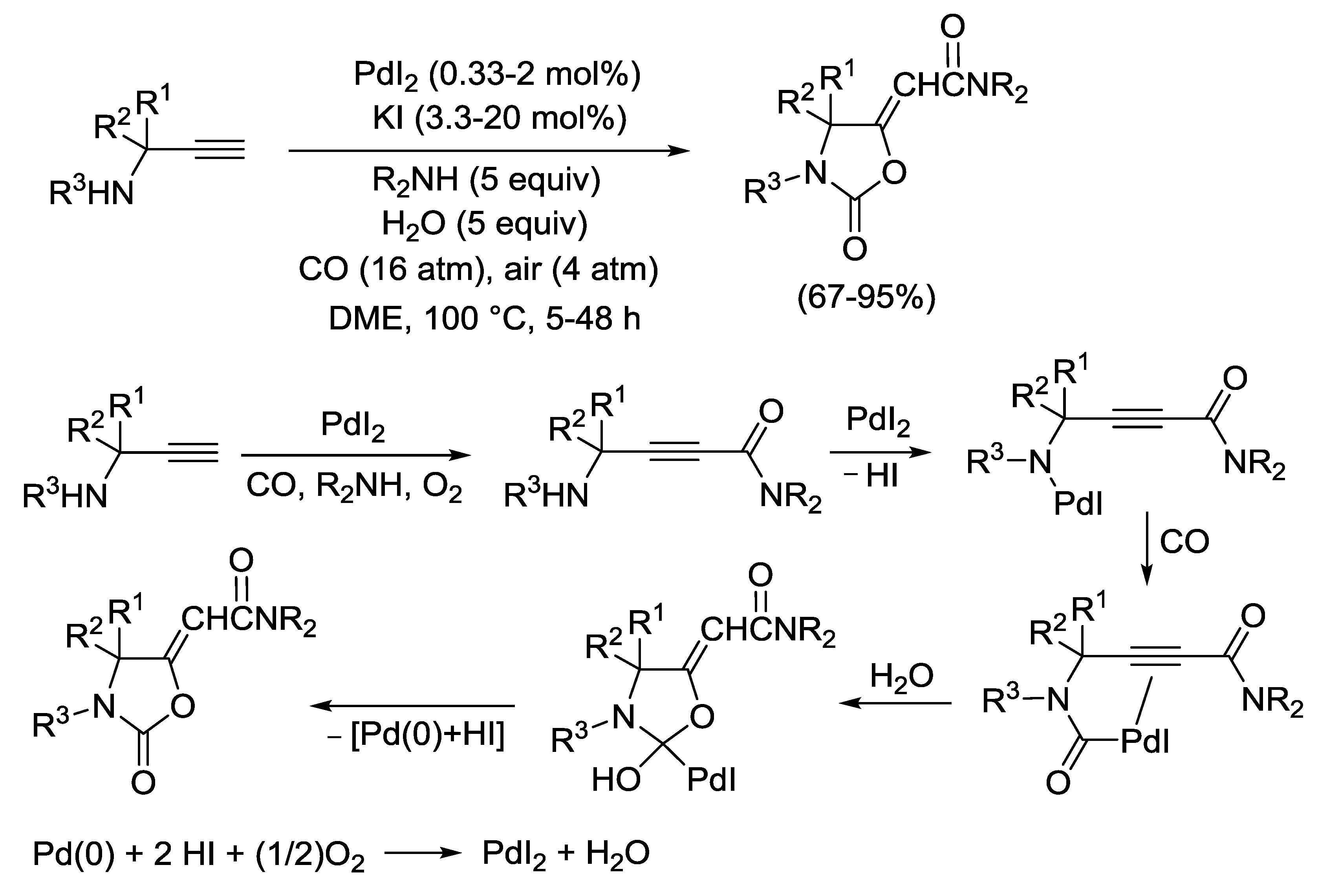
2.5. PdI2-Catalyzed Oxidative Monoaminocarbonylation of Terminal Alkyne Derivatives
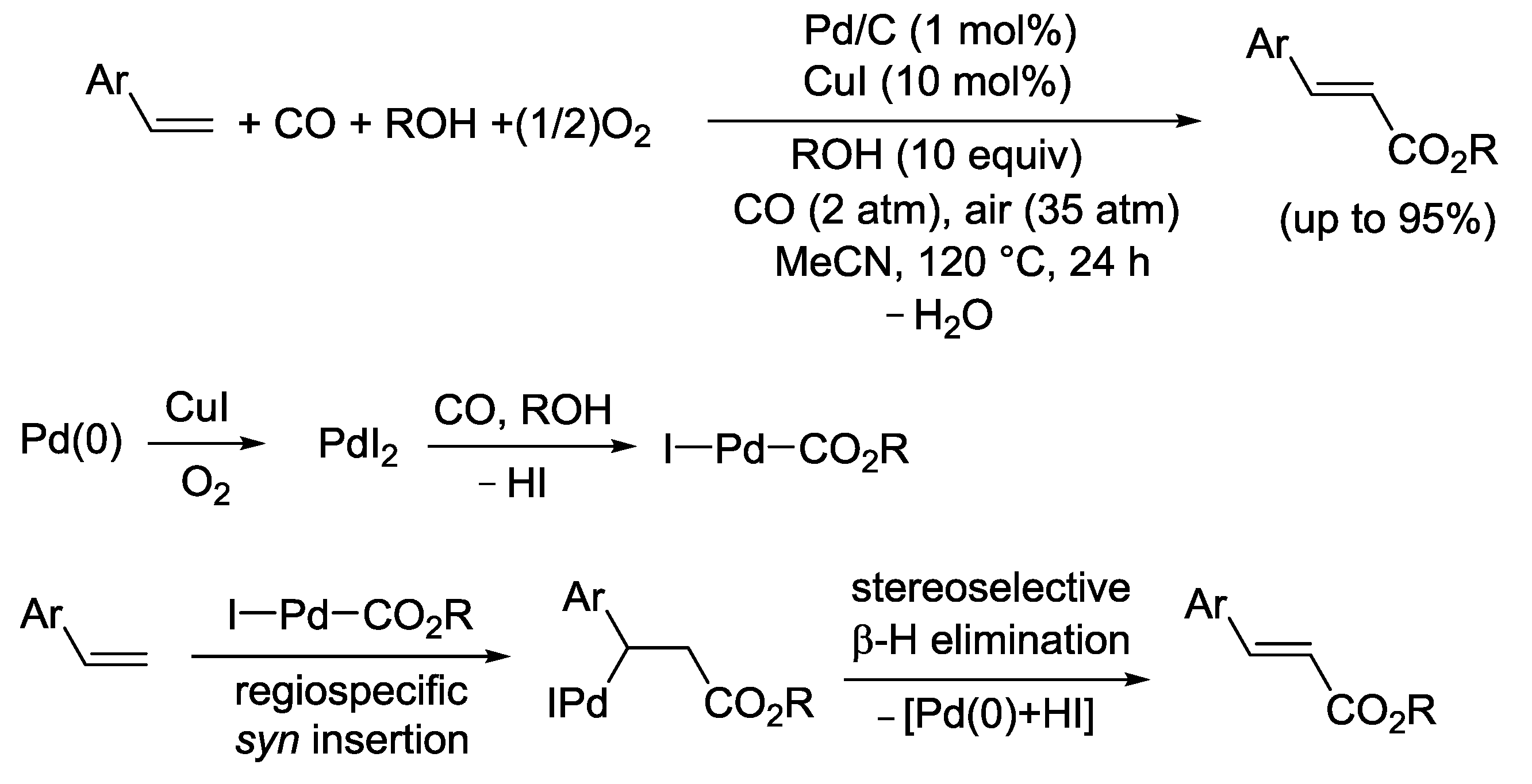
2.6. PdI2-Catalyzed Oxidative Monoalkoxycarbonylation of α-Olefins
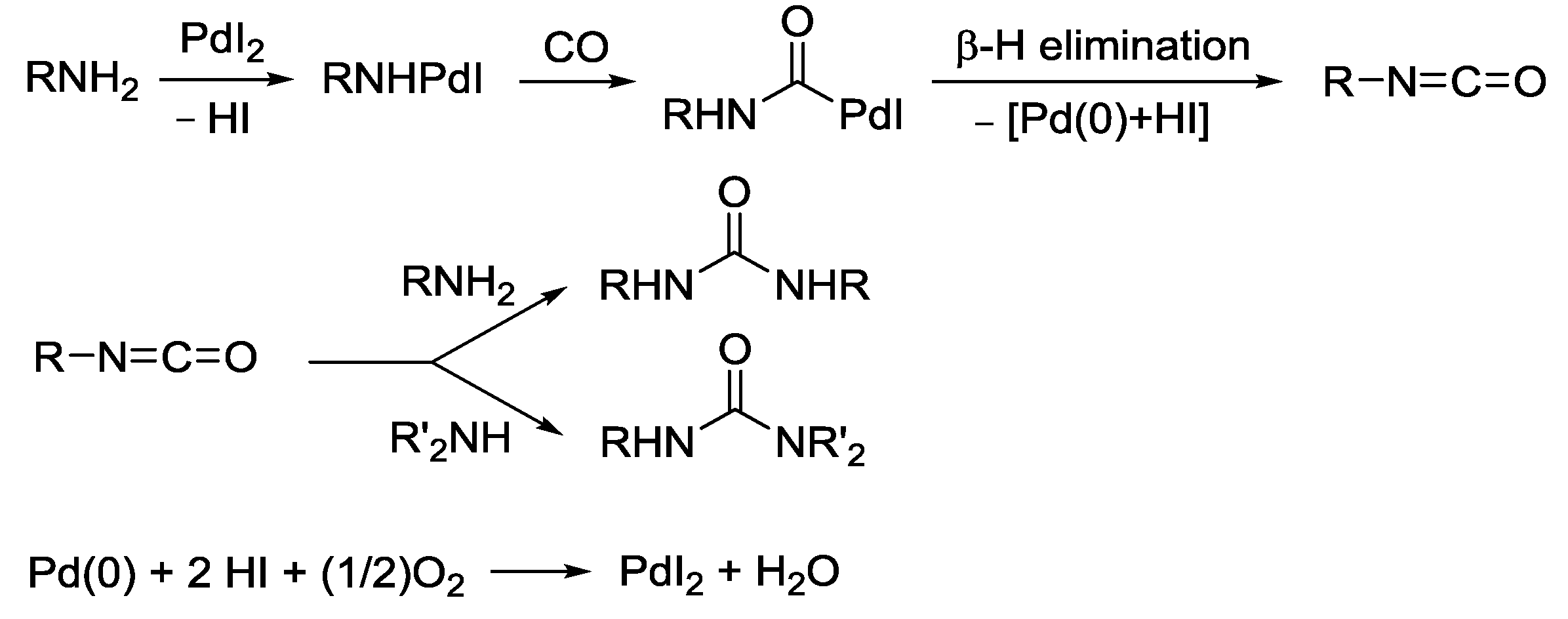
2.7. PdI2-Catalyzed Oxidative Carbonylation of Amines, β-Amino Alcohols, and 2-Aminophenols to Ureas, Oxamides, Oxazolidinones, and Benzoxazolones
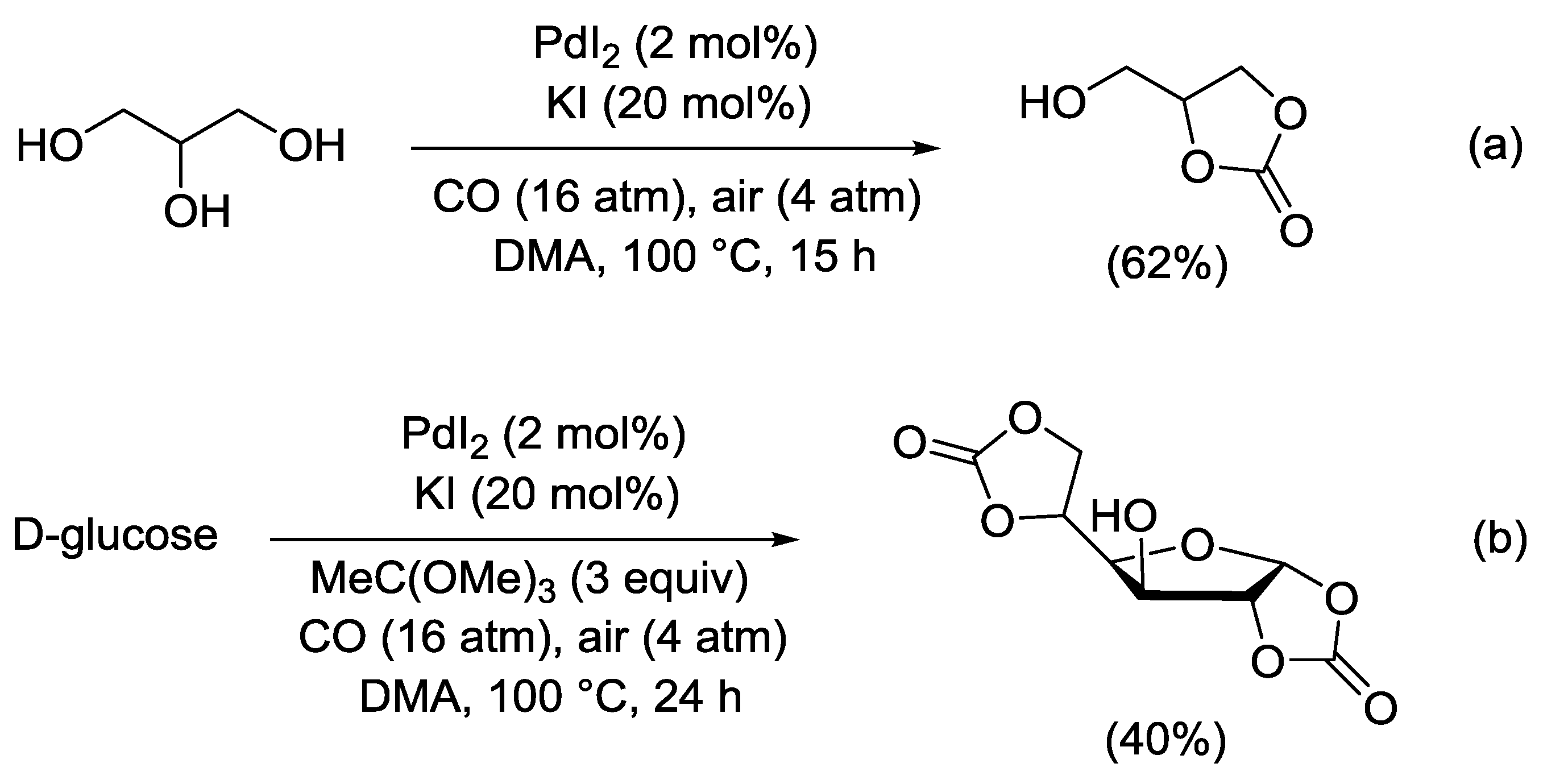
2.8. PdI2-Catalyzed Oxidative Carbonylation of 1,2- and 1,3-Diols to Cyclic Carbonates
3. PdI2-Catalyzed Non-Oxidative Carbonylations
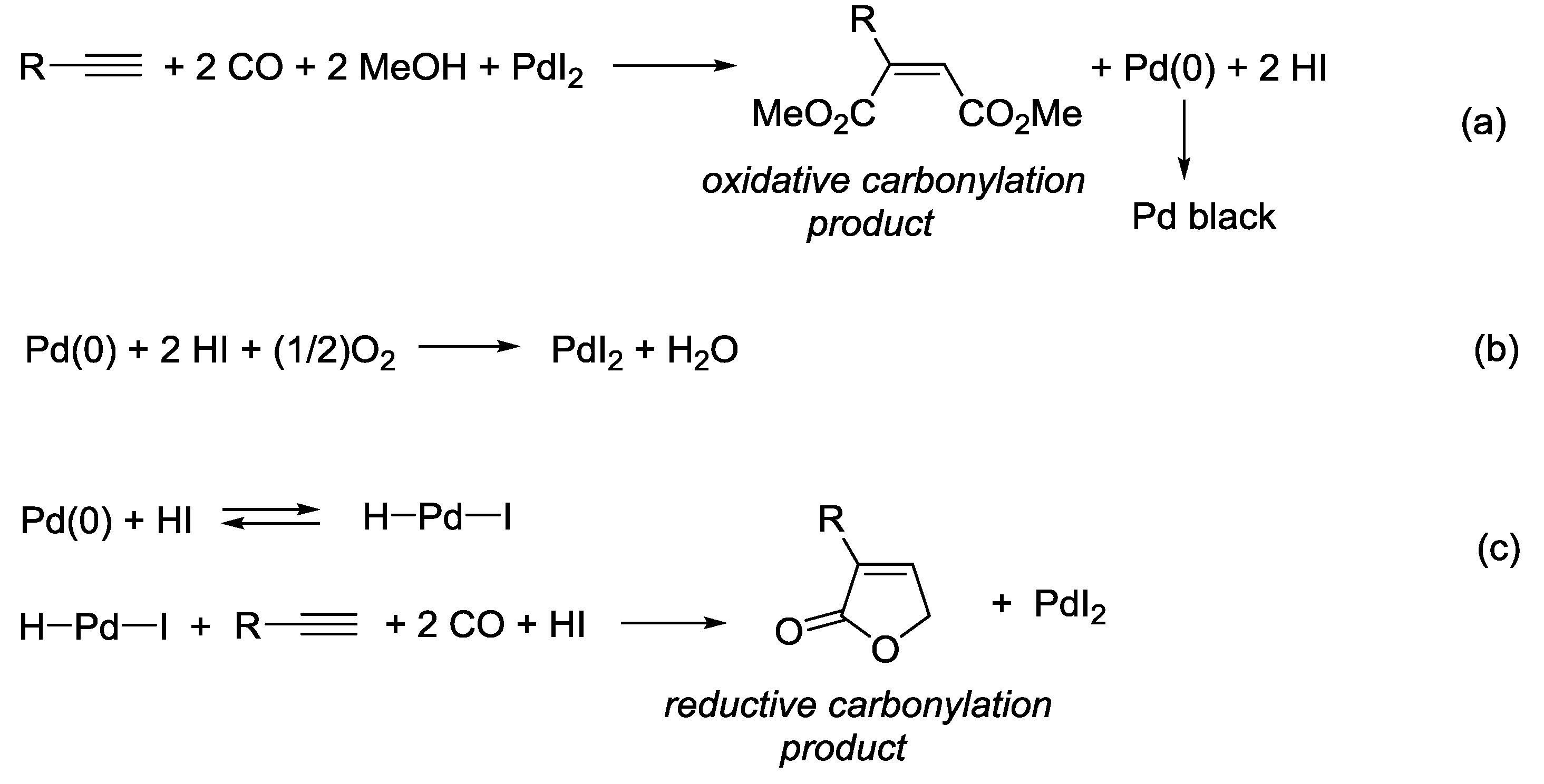
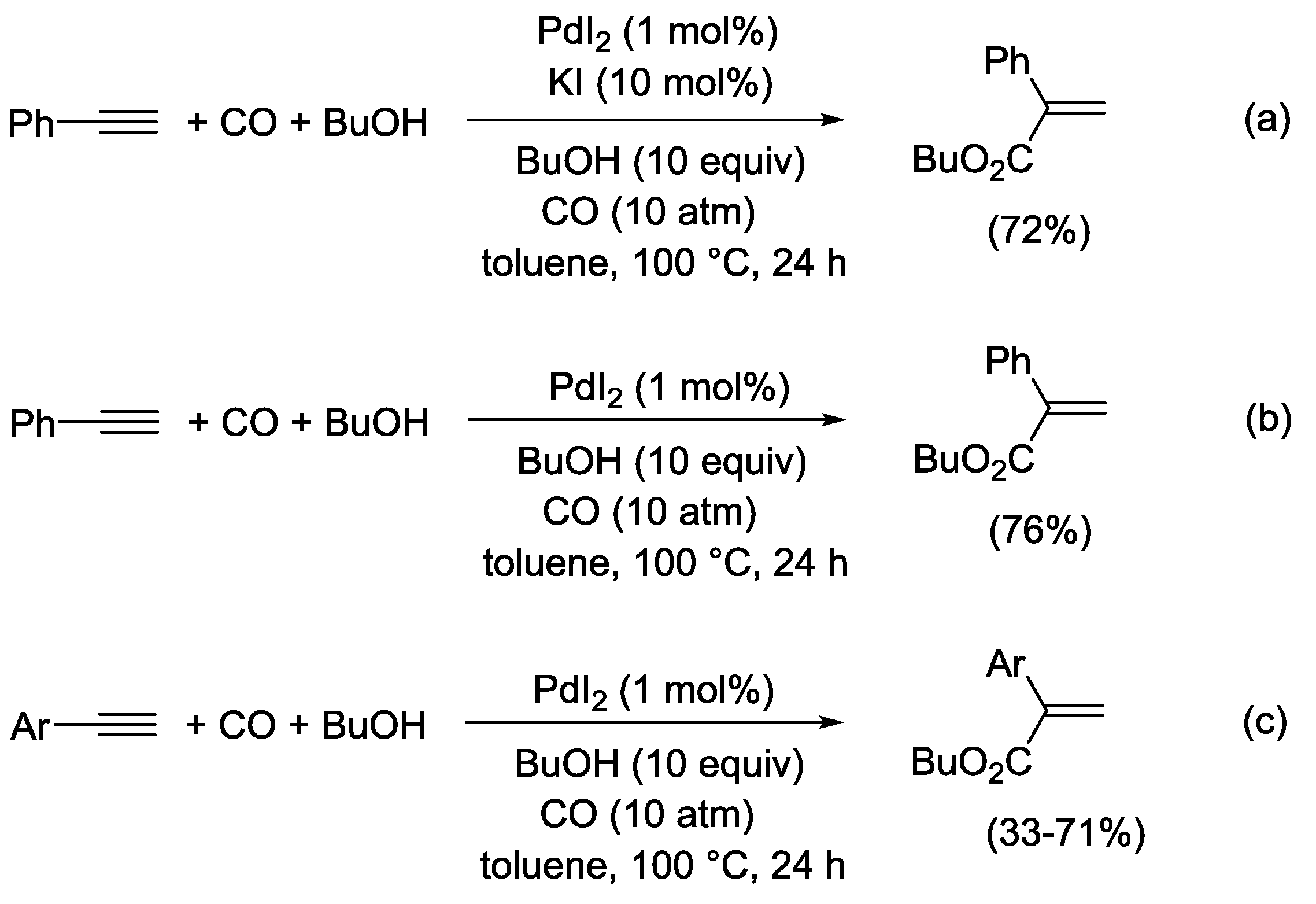
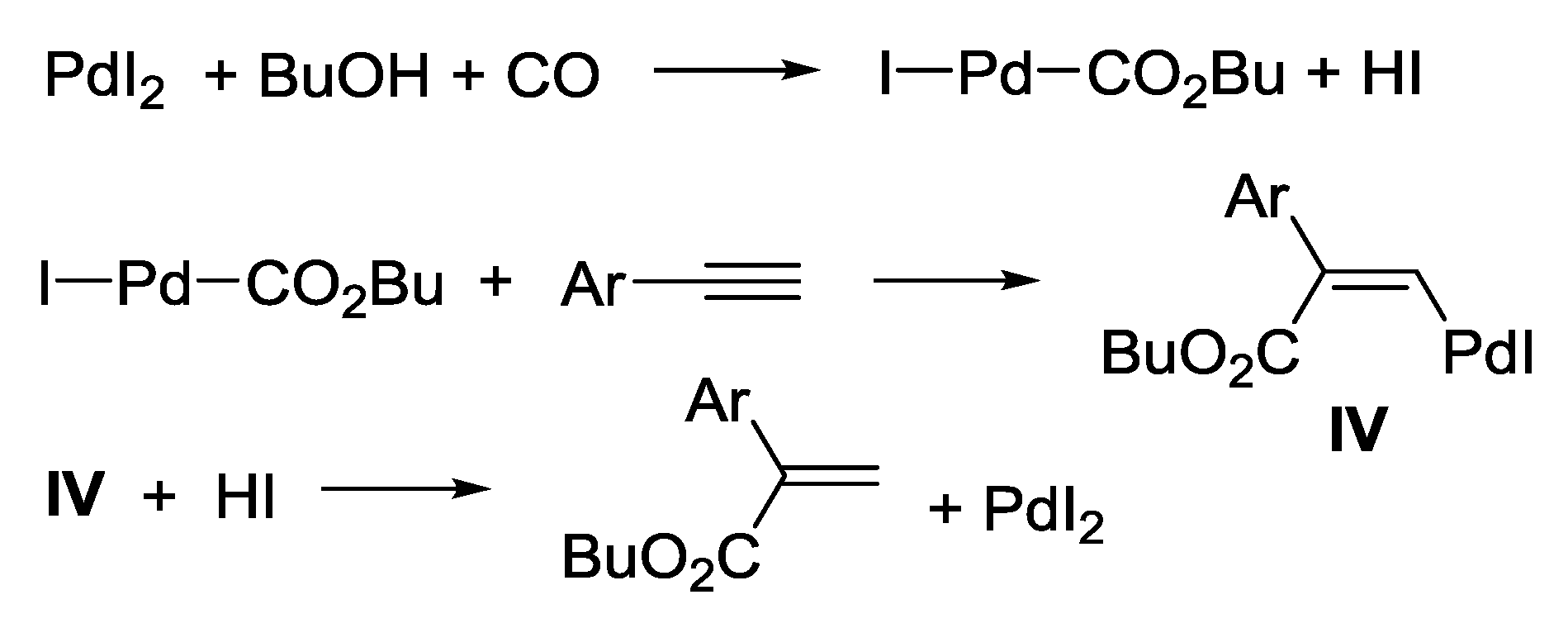
3.1. Combination Between Oxidative and Reductive Carbonylation and Additive Carbonylation of Terminal Alkynes
3.2. Combination Between Oxidative and Reductive Carbonylation within the Same Molecule and Sequential Homobimetallic Catalysis
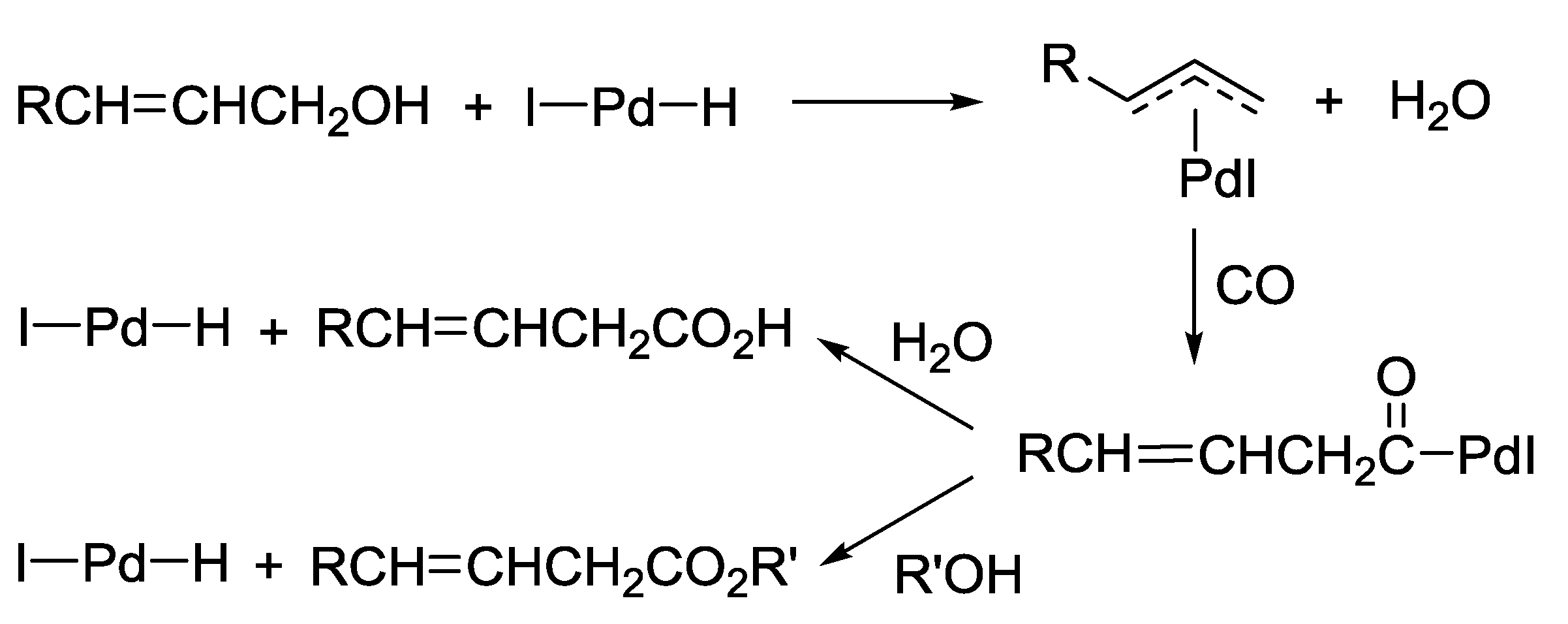
3.3. Substitutive Carbonylation of Allylic Alcohols to β,γ-Unsaturated Acids or Esters
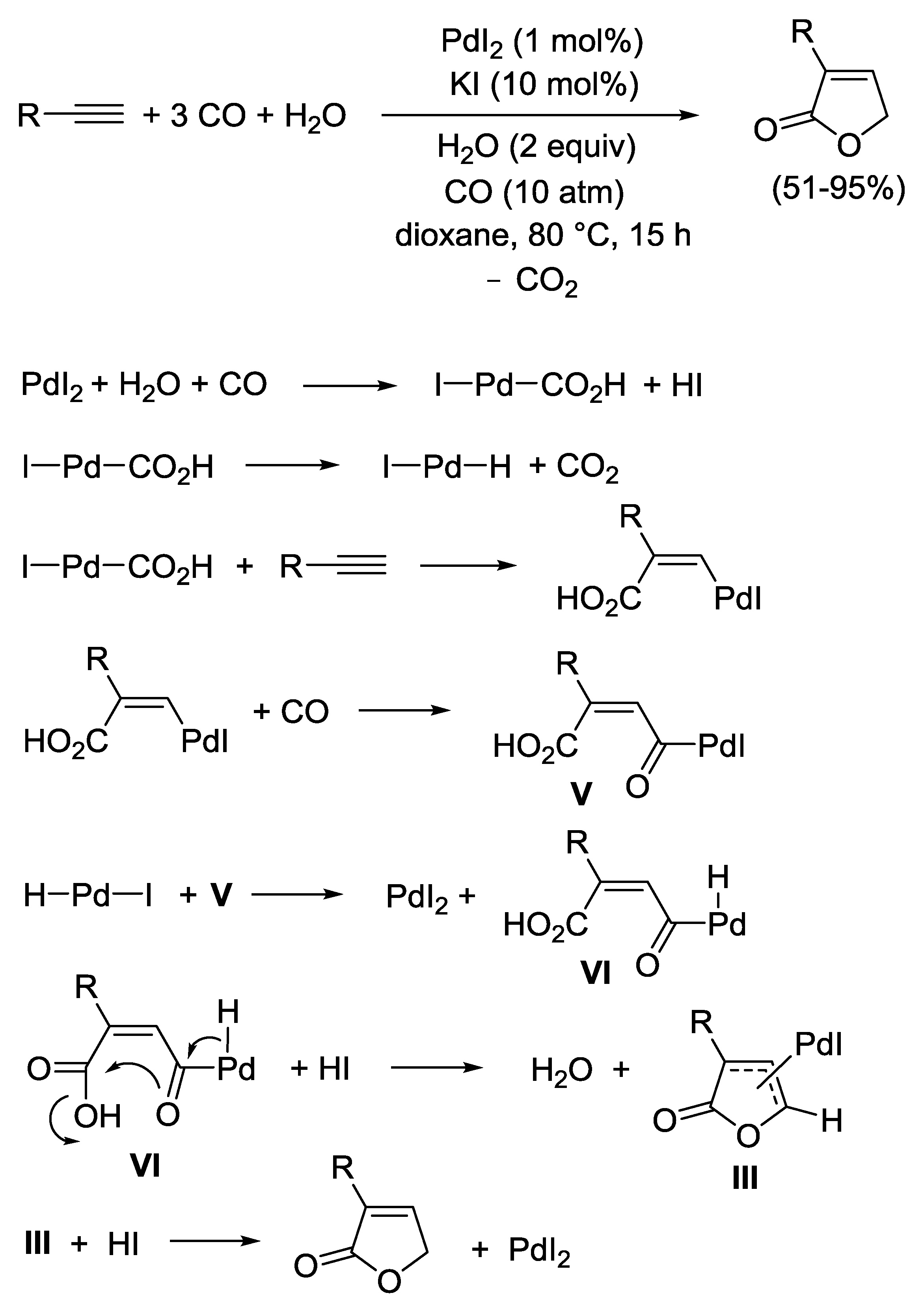
3.4. Reductive Carbonylation of Terminal Alkynes
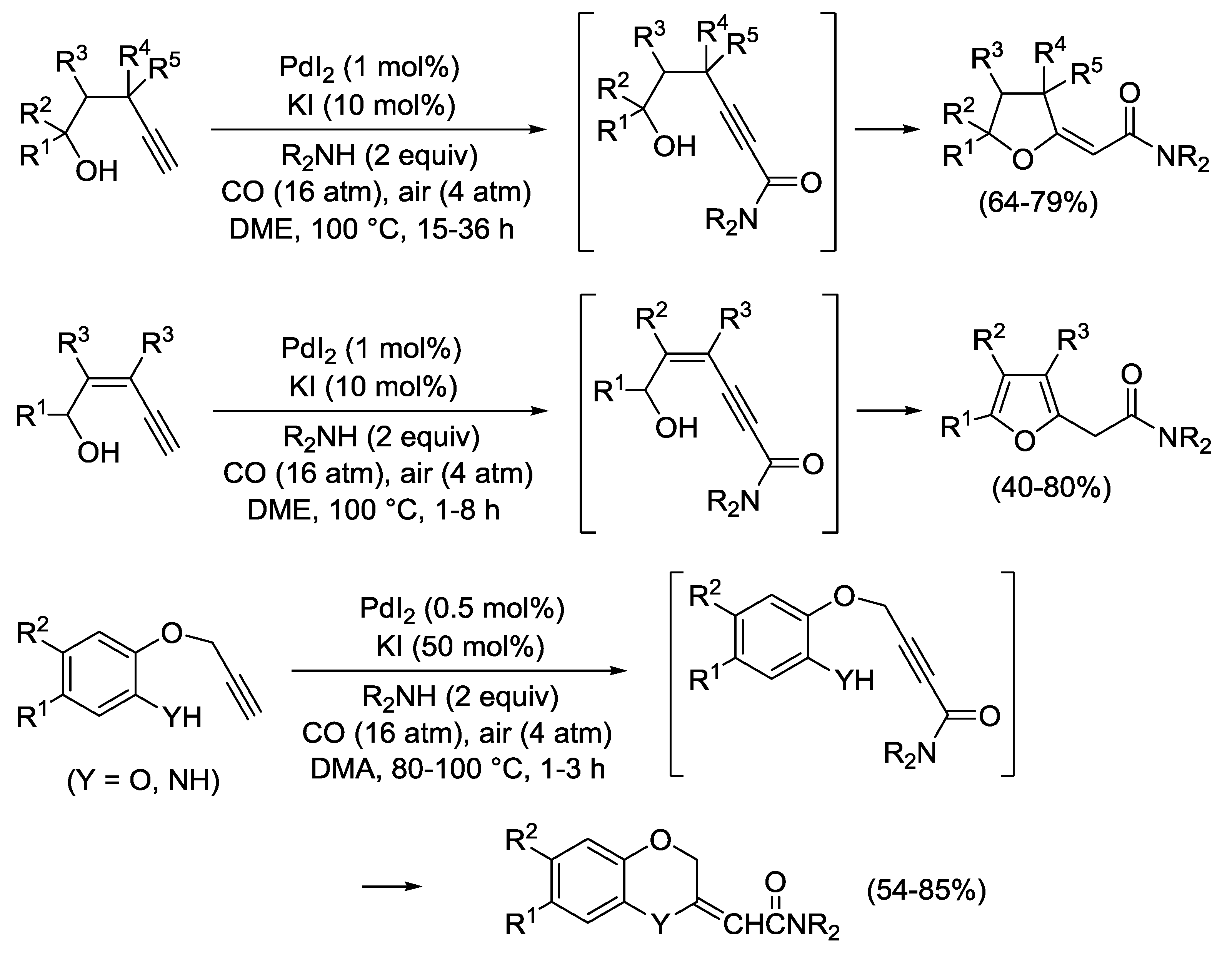
3.5. Propargylic Rearrangement Followed by Heterocyclization
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Peng, J.B.; Geng, H.Q.; Wu, X.F. The Chemistry of CO: Carbonylation. Chem 2019, 5, 526–552. [Google Scholar] [CrossRef]
- Ma, K.; Martin, B.S.; Yin, X.; Dai, M. Natural product syntheses via carbonylative cyclizations. Nat. Prod. Rep. 2019, 36, 174–219. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, D.U.; Neumann, K.T.; Lindhardt, A.T.; Skrydstrup, T. Recent developments in carbonylation chemistry using [13C]CO, [11C]CO, and [14C]CO. J. Label. Compd. Radiopharm. 2019, 61, 949–987. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Hu, Y.; Wu, X.F. Non-noble metal-catalysed carbonylative transformations. Chem. Soc. Rev. 2018, 47, 172–194. [Google Scholar] [CrossRef] [PubMed]
- Peng, J.B.; Qi, X.; Wu, X.F. Recent Achievements in Carbonylation Reactions: A Personal Account. Synlett 2017, 28, 175–194. [Google Scholar]
- Shen, C.; Wu, X.-F. Palladium-Catalyzed Carbonylative Multicomponent Reactions. Chem. Eur. J. 2017, 23, 2973–2987. [Google Scholar] [CrossRef]
- Pineiro, M.; Dias, L.D.; Damas, L.; Aquino, G.L.; Calvete, M.J.; Pereira, M.M. Microwave irradiation as a sustainable tool for catalytic carbonylation reactions. Inorganica Chim. Acta 2017, 455, 364–377. [Google Scholar] [CrossRef]
- Song, H.; Kang, M.; Jin, R.; Jin, F.; Chen, J. Application of Ionic Liquids to the Carbonylation Reactions. Progr. Chem. 2016, 28, 1313–1327. [Google Scholar]
- Gehrtz, P.H.; Hirschbeck, V.; Ciszek, B.; Fleischer, I. Carbonylations of Alkenes in the Total Synthesis of Natural Compounds. Synthesis 2016, 48, 2679. [Google Scholar]
- Wu, X.F. Palladium-catalyzed carbonylative transformation of aryl chlorides and aryl tosylates. RSC Adv. 2016, 6, 83831–83837. [Google Scholar] [CrossRef]
- Wu, X.F.; Beller, M. Transition Metal Catalyzed Carbonylative Synthesis of Heterocycles. In Topics in Heterocyclic Chemistry; Heterocycles, H., Ed.; Springer: Berlin, Germany, 2016; Volume 42. [Google Scholar]
- Kalck, P.; Urrutigoïty, M. Recent improvements in the alkoxycarbonylation reaction catalyzed by transition metal complexes. Inorganica Chim. Acta 2015, 431, 110–121. [Google Scholar] [CrossRef]
- Wu, L.; Fang, X.; Liu, Q.; Jackstell, R.; Beller, M.; Wu, X.F. Palladium-Catalyzed Carbonylative Transformation of C(sp3)–X Bonds. ACS Catal. 2014, 4, 2977–2989. [Google Scholar] [CrossRef]
- Gadge, S.T.; Bhanage, B.M. Recent developments in palladium catalysed carbonylation reactions. RSC Adv. 2014, 4, 10367. [Google Scholar] [CrossRef]
- Wu, X.F.; Neumann, H.; Beller, M. Synthesis of Heterocycles via Palladium-Catalyzed Carbonylations. Chem. Rev. 2013, 113, 1–35. [Google Scholar] [CrossRef]
- Wu, X.F.; Neumann, H.; Beller, M. Palladium-Catalyzed Oxidative Carbonylation Reactions. ChemSusChem 2013, 6, 229–241. [Google Scholar] [CrossRef]
- Wu, X.F.; Neumann, H. Ruthenium and Rhodium-Catalyzed Carbonylation Reactions. ChemCatChem 2012, 4, 447–458. [Google Scholar] [CrossRef]
- Liu, Q.; Zhang, H.; Lei, A. Oxidative Carbonylation Reactions: Organometallic Compounds (R—M) or Hydrocarbons (R—H) as Nucleophiles. Angew. Chem. Int. Ed. 2011, 50, 10788–10799. [Google Scholar] [CrossRef]
- Omae, I. Transition metal-catalyzed cyclocarbonylation in organic synthesis. Coord. Chem. Rev. 2011, 255, 139–160. [Google Scholar] [CrossRef]
- Barnard, C.F.J. Palladium-Catalyzed Carbonylation—A Reaction Come of Age. Organometallics 2008, 27, 5402–5422. [Google Scholar] [CrossRef]
- Kollár, L. Modern Carbonylation Methods; Wiley-VCH: Weinheim, Germany, 2008. [Google Scholar]
- Beller, M. Catalytic Carbonylation Reaction; Springer: Berlin, Germany, 2006. [Google Scholar]
- Gabriele, B. Recent advences in the PdI2-catalyzed carbonylative synthesis of heterocycles from acetylenic substrates: A personal account. Targets Heterocycl Syst. 2018, 22, 41–55. [Google Scholar]
- Gabriele, B. Synthesis of Heterocycles by Palladium-Catalyzed Carbonylative Reactions. In Advances in Transition-Metal Mediated Heterocyclic Synthesis; Solé, D., Fernández, I., Eds.; Academic Press-Elsevier: London, UK, 2018; Volume 3, pp. 55–127. [Google Scholar]
- Gabriele, B.; Mancuso, R.; Salerno, G. Oxidative Carbonylation as a Powerful Tool for the Direct Synthesis of Carbonylated Heterocycles. Eur. J. Org. Chem. 2012, 2012, 6825–6839. [Google Scholar] [CrossRef]
- Gabriele, B.; Salerno, G.; Costa, M. Oxidative Carbonylations. Top. Organomet. Chem. 2006, 18, 239–272. [Google Scholar]
- Gabriele, B.; Salerno, G. PdI2. In e-EROS (Electronic Encyclopedia of Reagents for Organic Synthesis); Crich, D., Ed.; Wiley–Interscience: New York, NY, USA, 2006. [Google Scholar]
- Gabriele, B.; Salerno, G.; Costa, M.; Chiusoli, G.P. Recent Advances in the Synthesis of Carbonyl Compounds by Palladium-Catalyzed Oxidative Carbonylation Reactions of Unsaturated Substrates. Curr. Org. Chem. 2004, 35, 919–946. [Google Scholar] [CrossRef]
- Gabriele, B.; Salerno, G.; Costa, M. PdI2—Catalyzed Synthesis of Heterocycles. Synlett 2004, 2004, 2468–2483. [Google Scholar] [CrossRef]
- Gabriele, B.; Salerno, G.; Costa, M.; Chiusoli, G.P. Recent developments in the synthesis of heterocyclic derivatives by PdI2-catalyzed oxidative carbonylation reactions. J. Organomet. Chem. 2003, 687, 219–228. [Google Scholar] [CrossRef]
- Gabriele, B.; Costa, M.; Salerno, G.; Chiusoli, G.P. A new synthesis of trimethyl aconitate by palladium-catalysed triple carbonylation of propynyl alcohol. J. Chem. Soc. Chem. Commun. 1992, 1992, 1007–1008. [Google Scholar] [CrossRef]
- Gabriele, B.; Costa, M.; Salerno, G.; Chiusoli, G.P. An efficient and selective palladium-catalysed oxidative dicarbonylation of alkynes to alkyl- or aryl-maleic esters. J. Chem. Soc. Perkin Trans. 1 1994, 1994, 83–87. [Google Scholar] [CrossRef]
- Gabriele, B.; Veltri, L.; Salerno, G.; Costa, M.; Chiusoli, G.P. Synthesis of Maleic Anhydrides and Maleic Acids by Palladium Catalyzed Oxidative Dicarbonylation of Alk-1-ynes. Eur. J. Org. Chem. 2003, 2003, 1722–1728. [Google Scholar] [CrossRef]
- Zhu, C.; Yang, B.; Bäckvall, J.-E. Highly Selective Cascade C–C Bond Formation via Palladium- Catalyzed Oxidative Carbonylation–Carbocyclization–Carbonylation–Alkynylation of Enallenes. J. Am. Chem. Soc. 2015, 137, 11868–11871. [Google Scholar] [CrossRef]
- Yang, B.; Qiu, Y.; Jiang, T.; Wulff, W.D.; Yin, X.; Zhu, C.; Bäckvall, J.E.; Bäckvall, J.E.; Bäckvall, J. Enantioselective Palladium-Catalyzed Carbonylative Carbocyclization of Enallenes via Cross-Dehydrogenative Coupling with Terminal Alkynes: Efficient Construction of α-Chirality of Ketones. Angew. Chem. 2017, 129, 4606–4610. [Google Scholar] [CrossRef]
- Zhu, C.; Yang, B.; Qiu, Y.; Bäckvall, J.E. Highly Selective Construction of Seven-Membered Carbocycles by Olefin-Assisted Palladium-Catalyzed Oxidative Carbocyclization-Alkoxycarbonylation of Bisallenes. Angew. Chem. 2016, 128, 14617–14620. [Google Scholar] [CrossRef]
- Gabriele, B.; Costa, M.; Salerno, G.; Chiusoli, G.P. Stereoselective synthesis of β-lactones containing α-Z-alkoxycarbonylmethylene chains by palladium-catalysed oxidative carbonylation of tertiary α-hydroxyalkynes. J. Chem. Soc. Chem. Commun. 1994, 199, 1429–1430. [Google Scholar] [CrossRef]
- Gabriele, B.; Salerno, G.; De Pascali, F.; Costa, M.; Chiusoli, G.P. Stereoselective synthesis of (Z)-α-(alkoxycarbonyl)methylene β- and γ-lactones by palladium-catalysed oxidative carbonylation of alkynols. J. Chem. Soc. Perkin Trans. 1 1997, 1997, 147–154. [Google Scholar] [CrossRef]
- Bonardi, A.; Costa, M.; Gabriele, B.; Salerno, G.; Chiusoli, G.P. Versatile Synthesis of beta-Lactams, gamma-Lactams or Oxazolines by Palladium-catalysed Oxidative Carbonylation of 1-Substituted Prop-2-ynylamines. Tetrahedron Lett. 1995, 36, 7495–7498. [Google Scholar] [CrossRef]
- Gabriele, B.; Salerno, G.; Veltri, L.; Costa, M.; Massera, C. Stereoselective Synthesis of (E)-3-(Methoxycarbonyl) methylene-1,3-dihydroindol-2-ones by Palladium-Catalyzed Oxidative Carbonylation of 2-Ethynylanilines. Eur. J. Org. Chem. 2001, 2001, 4607–4613. [Google Scholar] [CrossRef]
- Gabriele, B.; Salerno, G.; Fazio, A.; Veltri, L. Versatile Synthesis of Pyrrole-2-acetic Esters and (Pyridine-2-one)-3-acetic amides by Palladium-Catalyzed, Carbon Dioxide-Promoted Oxidative Carbonylation of (Z)-(2-En-4-ynyl)amines. Adv. Synth. Catal. 2006, 348, 2212–2222. [Google Scholar] [CrossRef]
- Mancuso, R.; Raut, D.S.; Marino, N.; De Luca, G.; Giordano, C.; Catalano, S.; Barone, I.; Andò, S.; Gabriele, B. A Palladium-Catalyzed Carbonylation Approach to Eight-Membered Lactam Derivatives with Antitumor Activity. Chem. A Eur. J. 2016, 22, 3053–3064. [Google Scholar] [CrossRef]
- Mancuso, R.; Veltri, L.; Russo, P.; Grasso, G.; Cuocci, C.; Romeo, R.; Gabriele, B. Palladium-Catalyzed Carbonylative Synthesis of Functionalized Benzimidazopyrimidinones. Synthesis 2018, 50, 267–277. [Google Scholar]
- Gabriele, B.; Salerno, G.; De Pascali, F.; Tomasi Scianò, G.; Costa, M.; Chiusoli, G.P. Novel synthesis of furan-2-acetic esters by palladium-catalysed oxidative cyclization-alkoxycarbonylation of (Z)-2-En-4-yn-1-ols. Tetrahedron Lett. 1997, 38, 6877–6880. [Google Scholar] [CrossRef]
- Gabriele, B.; Salerno, G.; De Pascali, F.; Costa, M.; Chiusoli, G.P. An Efficient and General Synthesis of Furan-2-acetic Esters by Palladium-Catalyzed Oxidative Carbonylation of (Z)-2-En-4-yn-1-ols. J. Org. Chem. 1999, 64, 7693–7699. [Google Scholar] [CrossRef]
- Gabriele, B.; Salerno, G.; De Pascali, F.; Costa, M.; Chiusoli, G.P. Palladium-catalysed synthesis of 2E-[(methoxycarbonyl)methylene]tetrahydrofurans: Oxidative cyclization-methoxycarbonylation of 4-yn-1-ols versus cycloisomerization-hydromethoxylation. J. Organomet. Chem. 2000, 593, 409–415. [Google Scholar] [CrossRef]
- Bacchi, A.; Costa, M.; Della Ca’, N.; Gabriele, B.; Salerno, G.; Cassoni, S. Heterocyclic Derivative Synthesis by Palladium-Catalyzed Oxidative Cyclization-Alkoxycarbonylation of Substituted γ-Oxoalkynes. J. Org. Chem. 2005, 70, 4971–4979. [Google Scholar] [CrossRef]
- Della Ca’, N.; Campanini, F.; Gabriele, B.; Salerno, G.; Massera, C.; Costa, M. Cascade Reactions: Catalytic Synthesis of Functionalized 1,3-Dihydroisobenzofuran and Tetrahydrofuran Derivatives by Sequential Nucleophilic Ring Opening-Heterocyclization- Oxidative Carbonylation of Alkynyloxiranes. Adv. Synth. Catal. 2009, 351, 2423–2432. [Google Scholar] [CrossRef]
- Bacchi, A.; Chiusoli, G.P.; Costa, M.; Gabriele, B.; Righi, C.; Salerno, G. Palladium-catalysed sequential carboxylation-alkoxycarbonylation of acetylenic amines. Chem. Commun. 1997, 1997, 1209–1210. [Google Scholar] [CrossRef]
- Chiusoli, G.; Costa, M.; Gabriele, B.; Salerno, G. Sequential reaction of carbon dioxide and carbon monoxide with acetylenic amines in the presence of a palladium catalyst. J. Mol. Catal. A Chem. 1999, 143, 297–310. [Google Scholar] [CrossRef]
- Bacchi, A.; Chiusoli, G.P.; Costa, M.; Sani, C.; Gabriele, B.; Salerno, G. Nitrogen heterocycles by palladium-catalysed oxidative cyclization-alkoxycarbonylation of acetylenic ureas. J. Organomet. Chem. 1998, 562, 35–43. [Google Scholar] [CrossRef]
- Bacchi, A.; Costa, M.; Gabriele, B.; Pelizzi, G.; Salerno, G. Efficient and General Synthesis of 5-(Alkoxycarbonyl)methylene-3-oxazolines by Palladium-Catalyzed Oxidative Carbonylation of Prop-2-ynylamides. J. Org. Chem. 2002, 67, 4450–4457. [Google Scholar] [CrossRef]
- Gabriele, B.; Salerno, G.; Fazio, A.; Campana, F.B. Unprecedented carbon dioxide effect on a Pd-catalysed oxidative carbonylation reaction: A new synthesis of pyrrole-2-acetic esters. Chem. Commun. 2002, 2002, 1408–1409. [Google Scholar] [CrossRef]
- Bacchi, A.; Costa, M.; Della Ca’, N.; Fabbricatore, M.; Fazio, A.; Gabriele, B.; Nasi, C.; Salerno, G. Synthesis of 1-(Alkoxycarbonyl)methylene-1,3-dihydroisobenzofurans and 4-(Alkoxycarbonyl)benzo[c]pyrans by Palladium-Catalysed Oxidative Carbonylation of 2-Alkynylbenzyl Alcohols, 2-Alkynylbenzaldehydes and 2-Alkynylphenyl Ketones. Eur. J. Org. Chem. 2004, 2004, 574–585. [Google Scholar] [CrossRef]
- Gabriele, B.; Veltri, L.; Maltese, V.; Spina, R.; Mancuso, R.; Salerno, G. Versatile Synthesis of Isoquinolines and Isochromenes by Pd-Catalyzed Oxidative Carbonylation of (2-Alkynyl)benzylideneamine Derivatives. Eur. J. Org. Chem. 2011, 2011, 5626–5635. [Google Scholar] [CrossRef]
- Costa, M.; Della Ca’, N.; Gabriele, B.; Massera, C.; Salerno, G.; Soliani, M. Synthesis of 4H-3,1-Benzoxazines, Quinazolin-2-ones, and Quinoline-4-ones by Palladium-Catalyzed Oxidative Carbonylation of 2-Ethynylaniline Derivatives. J. Org. Chem. 2004, 69, 2469–2477. [Google Scholar] [CrossRef]
- Pancrazzi, F.; Motti, E.; Costa, M.; Mancuso, R.; Gabriele, B.; Della Ca’, N. (Z)-4-(Carbomethoxymethylene)-2-(4-fluorophenyl)-4H-benzo[d][1,3] oxazine. Molbank 2017, 2017, M927. [Google Scholar] [CrossRef]
- Gabriele, B.; Mancuso, R.; Salerno, G.; Lupinacci, E.; Ruffolo, G.; Costa, M. Versatile Synthesis of Quinoline-3-carboxylic Esters and Indol-2-Acetic Esters by Palladium-Catalyzed Carbonylation of 1-(2-Aminoaryl)-2-yn-1-ols. J. Org. Chem. 2008, 73, 4971–4977. [Google Scholar] [CrossRef]
- Gabriele, B.; Veltri, L.; Salerno, G.; Mancuso, R.; Costa, M. Multicomponent Cascade Reactions: A Novel and Expedient Approach to Functionalized Indoles by an Unprecedented Nucleophilic Addition-Heterocyclization-Oxidative Alkoxycarbonylation Sequence. Adv. Synth. Catal. 2010, 352, 3355–3363. [Google Scholar] [CrossRef]
- Gabriele, B.; Veltri, L.; Mancuso, R.; Salerno, G.; Costa, M. A General Synthesis of Indole-3-carboxylic Esters by Palladium-Catalyzed Direct Oxidative Carbonylation of 2-Alkynylaniline Derivatives. Eur. J. Org. Chem. 2012, 2012, 2549–2559. [Google Scholar] [CrossRef]
- Mancuso, R.; Ziccarelli, I.; Armentano, D.; Marino, N.; Giofrè, S.V.; Gabriele, B. Divergent Palladium Iodide Catalyzed Multicomponent Carbonylative Approaches to Functionalized Isoindolinone and Isobenzofuranimine Derivatives. J. Org. Chem. 2014, 79, 3506–3518. [Google Scholar] [CrossRef]
- Araniti, F.; Mancuso, R.; Ziccarelli, I.; Sunseri, F.; Abenavoli, M.R.; Gabriele, B. 3-[(Methoxycarbonyl)methylene]isobenzofuran-1-imines as a New Class of Potential Herbicides. Molecules 2014, 19, 8261–8275. [Google Scholar] [CrossRef]
- Gabriele, B.; Veltri, L.; Mancuso, R.; Salerno, G.; Maggi, S.; Aresta, B.M. A Palladium Iodide-Catalyzed Carbonylative Approach to Functionalized Pyrrole Derivatives. J. Org. Chem. 2012, 77, 4005–4016. [Google Scholar] [CrossRef]
- Gabriele, B.; Veltri, L.; Mancuso, R.; Plastina, P.; Salerno, G.; Costa, M. Palladium-Catalyzed Oxidative Heterocyclodehydration-Alkoxycarbonylation of 3-Yne-1,2-diols: A Novel and Expedient Approach to Furan-3-carboxylic Esters. Tetrahedron Lett. 2010, 51, 1663–1665. [Google Scholar] [CrossRef]
- Gabriele, B.; Mancuso, R.; Maltese, V.; Veltri, L.; Salerno, G. Synthesis of Furan-3-carboxylic and 4-Methylene-4,5-dihydrofuran-3-carboxylic Esters by Direct Palladium Iodide Catalyzed Oxidative Carbonylation of 3-Yne-1, 2-diol Derivatives. J. Org. Chem. 2012, 77, 8657–8668. [Google Scholar] [CrossRef]
- Mancuso, R.; Ziccarelli, I.; Fini, F.; Della Ca’, N.; Marino, N.; Carfagna, C.; Gabriele, B. A Regio- and Stereoselective Carbonylative Approach to Alkyl (Z)-2-[3-Oxoisobenzofuran-1-(3H)-ylidene] acetates. Adv. Synth. Catal. 2019, 361, 690–695. [Google Scholar] [CrossRef]
- Acerbi, A.; Carfagna, C.; Costa, M.; Mancuso, R.; Gabriele, B.; Della Ca’, N. An Unprecedented Pd-Catalyzed Carbonylative Route to Fused Furo[3,4-b]indol-1-ones. Chem. A Eur. J. 2018, 24, 4835–4840. [Google Scholar] [CrossRef]
- Mancuso, R.; Miliè, R.; Palumbo Piccionello, A.; Olivieri, D.; Della Ca’, N.; Carfagna, C.; Gabriele, B. Catalytic Carbonylative Double Cyclization of 2-(3-Hydroxy-1-yn-1-yl) phenols in Ionic Liquids Leading to Furobenzofuranone Derivatives. J. Org. Chem. 2019, 84, 7303–7311. [Google Scholar] [CrossRef]
- Mancuso, R.; Ziccarelli, I.; Chimento, A.; Marino, N.; Della Ca’, N.; Sirianni, R.; Pezzi, V.; Gabriele, B. Catalytic Double Cyclization Process for Antitumor Agents against Breast Cancer Cell Lines. iScience 2018, 3, 279–288. [Google Scholar] [CrossRef]
- Gabriele, B.; Salerno, G.; Veltri, L.; Costa, M. Synthesis of 2-ynamides by direct palladium-catalyzed oxidative aminocarbonylation of alk-1-ynes. J. Organomet. Chem. 2001, 622, 84–88. [Google Scholar] [CrossRef]
- Gabriele, B.; Salerno, G.; Plastina, P.; Costa, M.; Crispini, A. Expedient Synthesis of 4-Dialkylamino-5H-furan-2-ones by One-Pot Sequential Pd-Catalyzed Oxidative Carbonylation of 2-Yn-1-ols—Conjugate Addition-Lactonization. Adv. Synth. Catal. 2004, 35, 351–358. [Google Scholar] [CrossRef]
- Plastina, P.; Gabriele, B.; Salerno, G. Palladium-Catalyzed Oxidative Aminocarbonylation of Alkynols. Synthesis 2007, 2007, 3083–3087. [Google Scholar] [CrossRef]
- Gabriele, B.; Plastina, P.; Salerno, G.; Costa, M. A New Synthesis of 4-Dialkylamino-1, 5-dihydropyrrol-2-ones by Pd-Catalyzed Oxidative Aminocarbonylation of 2-Ynylamines. Synlett 2005, 2005, 935–938. [Google Scholar] [CrossRef]
- Gabriele, B.; Salerno, G.; Plastina, P. A New Synthesis of 2-[(Dialkylcarbamoyl)Methylene]Tetrahydrofuran Derivatives by Pd-Catalyzed Oxidative Aminocarbonylation of 4-yn-1-ols. Lett. Org. Chem. 2004, 1, 134–136. [Google Scholar] [CrossRef]
- Gabriele, B.; Salerno, G.; Veltri, L.; Mancuso, R.; Li, Z.; Crispini, A.; Bellusci, A. A New Synthesis of 2,3-Dihydrobenzo[1,4]dioxine and 3,4-Dihydro-2H-benzo[1,4]oxazine Derivatives by Tandem Palladium-Catalyzed Oxidative Aminocarbonylation−Cyclization of 2-Prop-2-ynyloxyphenols and 2-Prop-2-ynyloxyanilines. J. Org. Chem. 2006, 71, 7895–7898. [Google Scholar] [CrossRef]
- Gabriele, B.; Plastina, P.; Salerno, G.; Mancuso, R. An Easy and Convenient Synthesis of 2-Furan-2-ylacetamides by Sequential Palladium-Catalyzed Oxidative Aminocarbonylation of (Z)-2-En-4-yn-1-ols/Conjugate Addition/Aromatization. Synthesis 2006, 2006, 4247–4251. [Google Scholar] [CrossRef]
- Gabriele, B.; Mancuso, R.; Ziccarelli, I.; Salerno, G. A new approach to isoindolinone derivatives by sequential palladium iodide-catalyzed oxidative aminocarbonylation–heterocyclization of 2-ethynylbenzamides. Tetrahedron Lett. 2012, 53, 6694–6696. [Google Scholar] [CrossRef]
- Giofrè, S.V.; Cirmi, S.; Mancuso, R.; Nicolò, F.; Lanza, G.; Legnani, L.; Campisi, A.; Chiacchio, M.A.; Navarra, M.; Gabriele, B.; et al. Synthesis of spiro[isoindole-1,5’-isoxazolidin]-3(2H)-ones as potential inhibitors of the MDM2-p53 interaction. Beilstein J. Org. Chem. 2016, 12, 2793–2807. [Google Scholar] [CrossRef]
- Veltri, L.; Giofrè, S.V.; Devo, P.; Romeo, R.; Dobbs, A.P.; Gabriele, B. A Palladium Iodide-Catalyzed Oxidative Aminocarbonylation–Heterocyclization Approach to Functionalized Benzimidazoimidazoles. J. Org. Chem. 2018, 83, 1680–1685. [Google Scholar] [CrossRef]
- Veltri, L.; Grasso, G.; Rizzi, R.; Mancuso, R.; Gabriele, B. Palladium-Catalyzed Carbonylative Multicomponent Synthesis of Functionalized Benzimidazothiazoles. Asian J. Org. Chem. 2016, 5, 560–567. [Google Scholar] [CrossRef]
- Gabriele, B.; Veltri, L.; Mancuso, R.; Carfagna, C. Cascade Reactions: A Multicomponent Approach to Functionalized Indane Derivatives by a Tandem Palladium-Catalyzed Carbamoylation/Carbocyclization Process. Adv. Synth. Catal. 2014, 356, 2547–2558. [Google Scholar] [CrossRef]
- Gabriele, B.; Plastina, P.; Salerno, G.; Mancuso, R.; Costa, M. An Unprecedented Pd-Catalyzed, Water-Promoted Sequential Oxidative Aminocarbonylation-Cyclocarbonylation Process Leading to 2-Oxazolidinones. Org. Lett. 2007, 9, 3319–3322. [Google Scholar] [CrossRef]
- Mancuso, R.; Maner, A.; Ziccarelli, I.; Pomelli, C.; Chiappe, C.; Della Ca’, N.; Veltri, L.; Gabriele, B. Auto-Tandem Catalysis in Ionic Liquids: Synthesis of 2-Oxazolidinones by Palladium-Catalyzed Oxidative Carbonylation of Propargylic Amines in EmimEtSO4. Molecules 2016, 21, 897. [Google Scholar] [CrossRef]
- Veltri, L.; Mancuso, R.; Altomare, A.; Gabriele, B. Divergent Multicomponent Tandem Palladium-Catalyzed Aminocarbonylation-Cyclization Approaches to Functionalized Imidazothiazinones and Imidazothiazoles. ChemCatChem 2015, 7, 2206–2213. [Google Scholar] [CrossRef]
- Maffei, M.; Giacoia, G.; Mancuso, R.; Gabriele, B.; Motti, E.; Costa, M.; Della Ca’, N. A highly efficient Pd/CuI-catalyzed oxidative alkoxycarbonylation of α-olefins to unsaturated esters. J. Mol. Catal. A Chem. 2017, 426, 435–443. [Google Scholar] [CrossRef]
- Gabriele, B.; Mancuso, R.; Salerno, G.; Costa, M. A novel and efficient method for the Pd-catalysed oxidative carbonylation of amines to symmetrically and unsymmetrically substituted ureas. Chem. Commun. 2003, 2003, 486–487. [Google Scholar] [CrossRef]
- Gabriele, B.; Salerno, G.; Mancuso, R.; Costa, M. Efficient Synthesis of Ureas by Direct Palladium-Catalyzed Oxidative Carbonylation of Amines. J. Org. Chem. 2004, 69, 4741–4750. [Google Scholar] [CrossRef]
- Della Ca’, N.; Bottarelli, P.; Dibenedetto, A.; Aresta, M.; Gabriele, B.; Salerno, G.; Costa, M. Palladium-catalyzed synthesis of symmetrical urea derivatives by oxidative carbonylation of primary amines in carbon dioxide medium. J. Catal. 2011, 282, 120–127. [Google Scholar]
- Gabriele, B.; Mancuso, R.; Veltri, L.; Della Ca’, N. Polemic against conclusions drawn in “Palladium/iodide catalyzed oxidative carbonylation of aniline to diphenylurea: Effect of ppm amounts of iron salts” (J. Catal. 369 (2019) 257–266). J. Catal. 2019, in press. [Google Scholar] [CrossRef]
- Mancuso, R.; Raut, D.S.; Della Ca’, N.; Fini, F.; Carfagna, C.; Gabriele, B. Catalytic Oxidative Carbonylation of Amino Moieties to Ureas, Oxamides, 2-Oxazolidinones, and Benzoxazolones. ChemSusChem 2015, 8, 2204–2211. [Google Scholar] [CrossRef]
- Gabriele, B.; Salerno, G.; Brindisi, D.; Costa, M.; Chiusoli, G.P. Synthesis of 2-Oxazolidinones by Direct Palladium-Catalyzed Oxidative Carbonylation of 2-Amino-1-alkanols. Org. Lett. 2000, 2, 625–627. [Google Scholar] [CrossRef]
- Gabriele, B.; Mancuso, R.; Salerno, G.; Costa, M. An Improved Procedure for the Palladium-Catalyzed Oxidative Carbonylation of β-Amino Alcohols to Ozaxolidin-2-ones. J. Org. Chem. 2003, 68, 601–604. [Google Scholar] [CrossRef]
- Mancuso, R.; Miliè, R.; Ziccarelli, I.; Novello, M.; Della Ca’, N.; Gabriele, B. (S)-4-Isopropyl-5,5-diphenyloxazolidin-2-one. Molbank 2018, 2018, M1017. [Google Scholar] [CrossRef]
- Gabriele, B.; Mancuso, R.; Salerno, G.; Ruffolo, G.; Costa, M.; Dibenedetto, A. A novel and efficient method for the catalytic direct oxidative carbonylation of 1,2- and 1,3-diols to 5-membered and 6-membered cyclic carbonates. Tetrahedron Lett. 2009, 50, 7330–7332. [Google Scholar] [CrossRef]
- Gabriele, B.; Mancuso, R.; Salerno, G.; Veltri, L.; Costa, M.; Dibenedetto, A. A General and Expedient Synthesis of 5- and 6-Membered Cyclic Carbonates by Palladium-Catalyzed Oxidative Carbonylation of 1,2- and 1,3-Diols. ChemSusChem 2011, 4, 1778–1786. [Google Scholar] [CrossRef]
- Gabriele, B.; Salerno, G.; Costa, M.; Chiusoli, G.P. Combined oxidative and reductive carbonylation of terminal alkynes with palladium iodide-thiourea catalysts. J. Organomet. Chem. 1995, 503, 21–28. [Google Scholar] [CrossRef]
- Queirolo, M.; Vezzani, A.; Mancuso, R.; Gabriele, B.; Costa, M.; Della Ca’, N. Neutral vs anionic palladium iodide-catalyzed carbonylation of terminal arylacetylenes. J. Mol. Catal. A Chem. 2015, 398, 115–126. [Google Scholar] [CrossRef]
- Bonardi, A.; Costa, M.; Gabriele, B.; Salerno, G.; Chiusoli, G.P. Coupling of oxidative and reductive processes: Catalytic carbonylation of acetals of prop-2-ynal. J. Chem. Soc. Chem. Commun. 1994, 1994, 2429–2430. [Google Scholar] [CrossRef]
- Costa, M.; De Souza Santos, L.; Chiusoli, G.P.; Gabriele, B.; Salerno, G. Palladium-catalyzed ring closure to a naphthofuranoneacetic ester by selective carbonylation of diacetylenic precursors. J. Mol. Catal. 1993, 78, 151–158. [Google Scholar] [CrossRef]
- De Munno, G.; Gabriele, B.; Salerno, G. X-ray structure of palladium (II) tetrakis-thiourea iodide, a catalyst for carbonylation reactions. Inorg. Chim. Acta 1995, 234, 181–183. [Google Scholar] [CrossRef]
- Gabriele, B.; Mancuso, R.; Salerno, G.; Plastina, P. A Novel Palladium-Catalyzed Dicarbonylation Process Leading to Coumarins. J. Org. Chem. 2008, 73, 756–759. [Google Scholar] [CrossRef]
- Araniti, F.; Mancuso, R.; Lupini, A.; Giofrè, S.V.; Sunseri, F.; Gabriele, B.; Abenavoli, M.R. Phytotoxic Potential and Biological Activity of Three Synthetic Coumarin Derivatives as New Natural-Like Herbicides. Molecules 2015, 20, 17883–17902. [Google Scholar] [CrossRef]
- Gabriele, B.; Mancuso, R.; Salerno, G.; Veltri, L. Sequential homobimetallic catalysis: An unprecedented tandem Pd(0)-catalysed deprotection—Pd(II)-catalysed heterocyclization reaction leading to benzofurans. Chem. Commun. 2005, 2005, 271–273. [Google Scholar] [CrossRef]
- Gabriele, B.; Mancuso, R.; Salerno, G.; Costa, M. Cascade Reactions: Sequential Homobimetallic Catalysis Leading to Benzofurans and β, γ-Unsaturated Esters. Adv. Synth. Catal. 2006, 37, 1101–1109. [Google Scholar] [CrossRef]
- Mancuso, R.; Gabriele, B. The Sequential Homobimetallic Catalysis Concept Applied to the Synthesis of Benzofuran Derivatives (Highlight). Chem. Heterocycl. Compd. 2014, 50, 160–170. [Google Scholar] [CrossRef]
- Giordano, C.; Rovito, D.; Barone, I.; Mancuso, R.; Bonofiglio, D.; Giordano, F.; Catalano, S.; Gabriele, B.; Andò, S. Benzofuran-2-acetic ester derivatives induce apoptosis in breast cancer cells by upregulating p21 Cip/WAF1 gene expression in p53-independent manner. DNA Repair 2017, 51, 20–30. [Google Scholar] [CrossRef]
- Iannazzo, D.; Pistone, A.; Celesti, C.; Triolo, C.; Patanè, S.; Giofrè, S.V.; Romeo, R.; Ziccarelli, I.; Mancuso, R.; Gabriele, B.; et al. A Smart Nanovector for Cancer Targeted Drug Delivery Based on Graphene Quantum Dots. Nanomaterials 2019, 9, 282. [Google Scholar] [CrossRef]
- Gabriele, B.; Salerno, G.; Costa, M.; Chiusoli, G.P. Palladium-catalysed formation of maleic anhydrides from CO, CO2 and alk-1-ynes. Chem. Commun. 1999, 1999, 1381–1382. [Google Scholar] [CrossRef]
- Chiusoli, G.P.; Costa, M.; Cucchia, L.; Gabriele, B.; Salerno, G.; Veltri, L. Carbon dioxide effect on palladium-catalyzed sequential reactions with carbon monoxide, acetylenic compounds and water. J. Mol. Catal. A Chem. 2003, 204, 133–142. [Google Scholar] [CrossRef]
- Gabriele, B.; Mancuso, R.; Salerno, G.; Costa, M. Cascade Reactions: A New Synthesis of 2-Benzofuran-2-ylacetamides by Sequential Pd(0)-Catalyzed Deallylation−Pd(II)-Catalyzed Aminocarbonylative Heterocyclization of 1-(2-Allyloxyaryl)-2-yn-1-ols. J. Org. Chem. 2007, 72, 9278–9282. [Google Scholar] [CrossRef]
- Gabriele, B.; Mancuso, R.; Lupinacci, E.; Salerno, G.; Veltri, L. Tandem catalysis in ionic liquids: A recyclable catalytic synthesis of benzofuran derivatives. Tetrahedron 2010, 66, 6156–6161. [Google Scholar] [CrossRef]
- Gabriele, B.; Salerno, G.; Costa, M.; Chiusoli, G.P. A simple catalytic system for the substitutive carbonylation of allyl alcohols to β, γ-unsaturated acids or esters. J. Mol. Catal. A Chem. 1996, 111, 43–48. [Google Scholar] [CrossRef]
- Gabriele, B.; Salerno, G.; Costa, M.; Chiusoli, G.P. A new regioselective synthesis of 3-substituted furan-2(5H)-ones by palladium-catalysed reductive carbonylation of alk-1-ynes. Tetrahedron Lett. 1999, 40, 989–990. [Google Scholar] [CrossRef]
- Veltri, L.; Paladino, V.; Plastina, P.; Gabriele, B. A Palladium Iodide-Catalyzed Cyclocarbonylation Approach to Thiadiazafluorenones. J. Org. Chem. 2016, 81, 6106–6111. [Google Scholar] [CrossRef]
- Veltri, L.; Amuso, R.; Cuocci, C.; Vitale, P.; Gabriele, B. Palladium-Catalyzed Cyclocarbonylation Approach to Thiadiazafluorenones: A Correction. J. Org. Chem. 2019, 84, 8743–8749. [Google Scholar] [CrossRef]

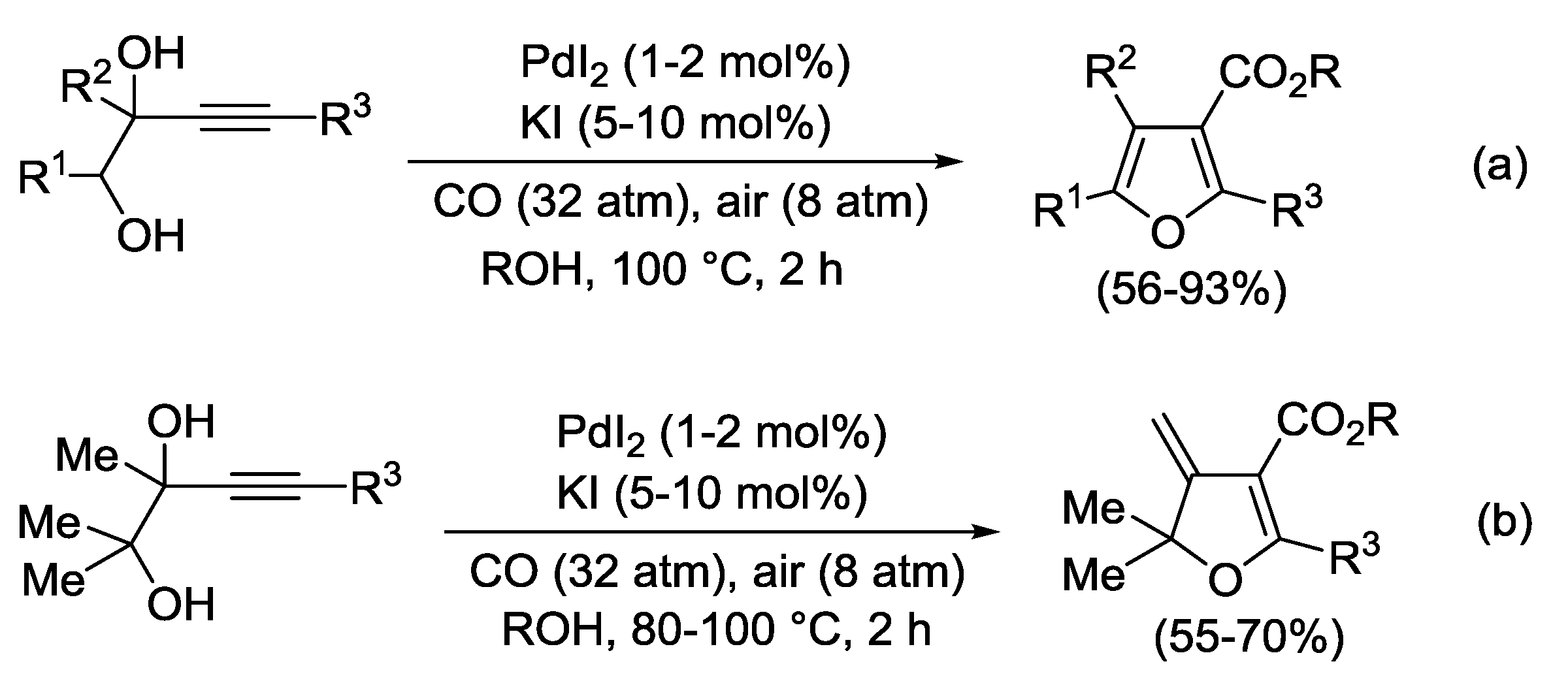

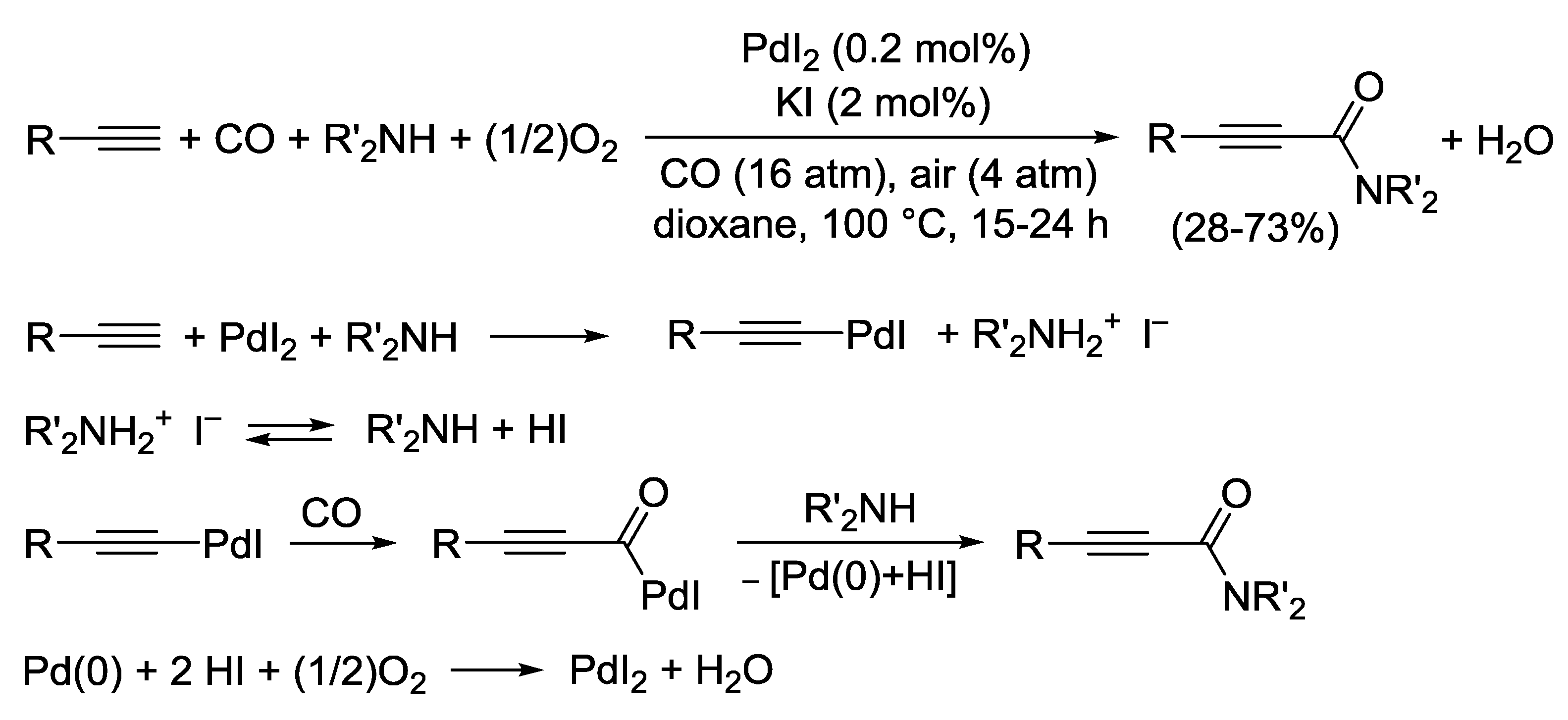

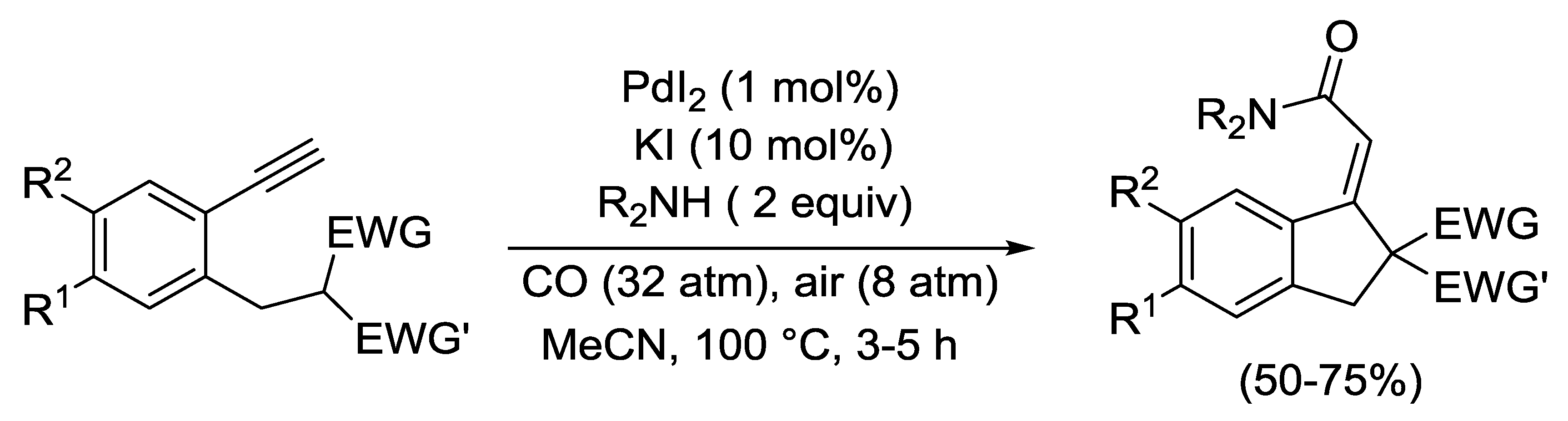
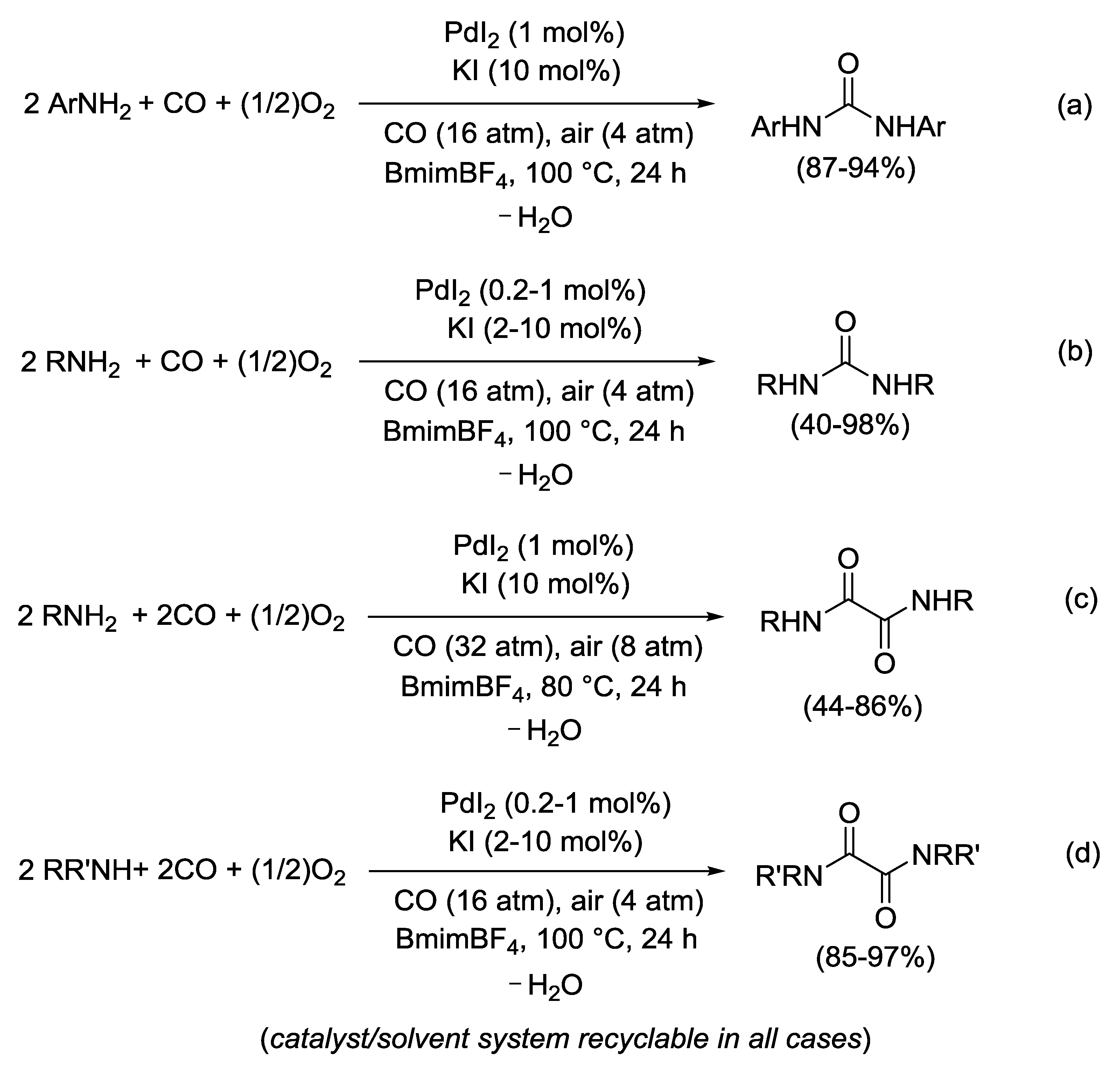


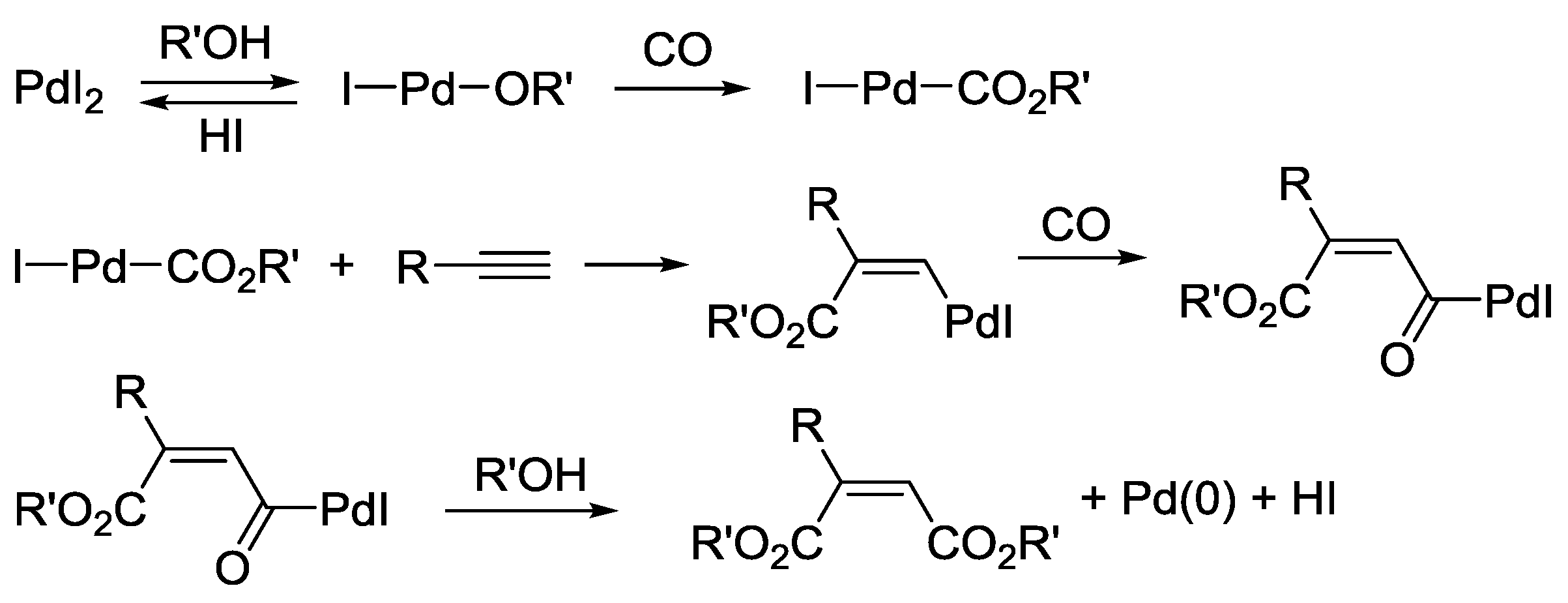
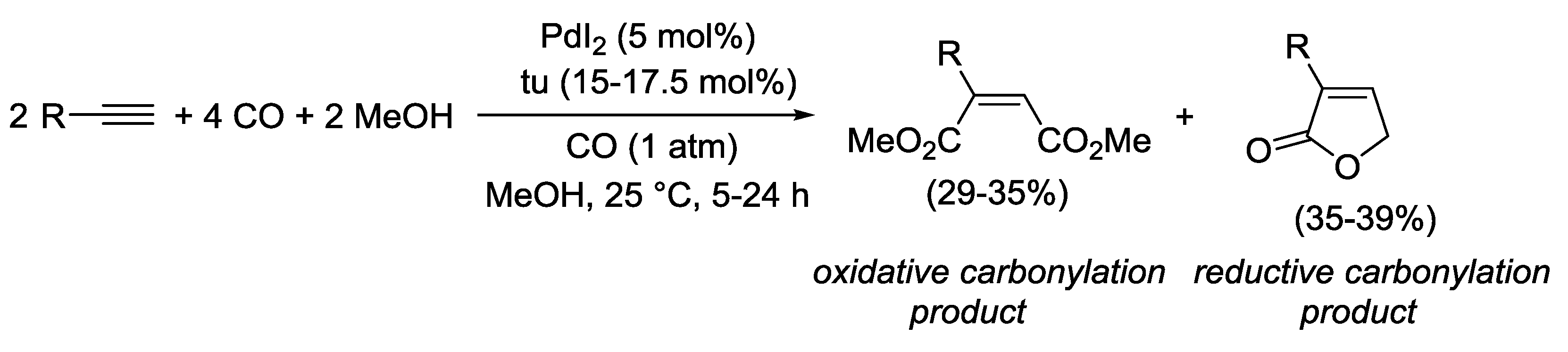
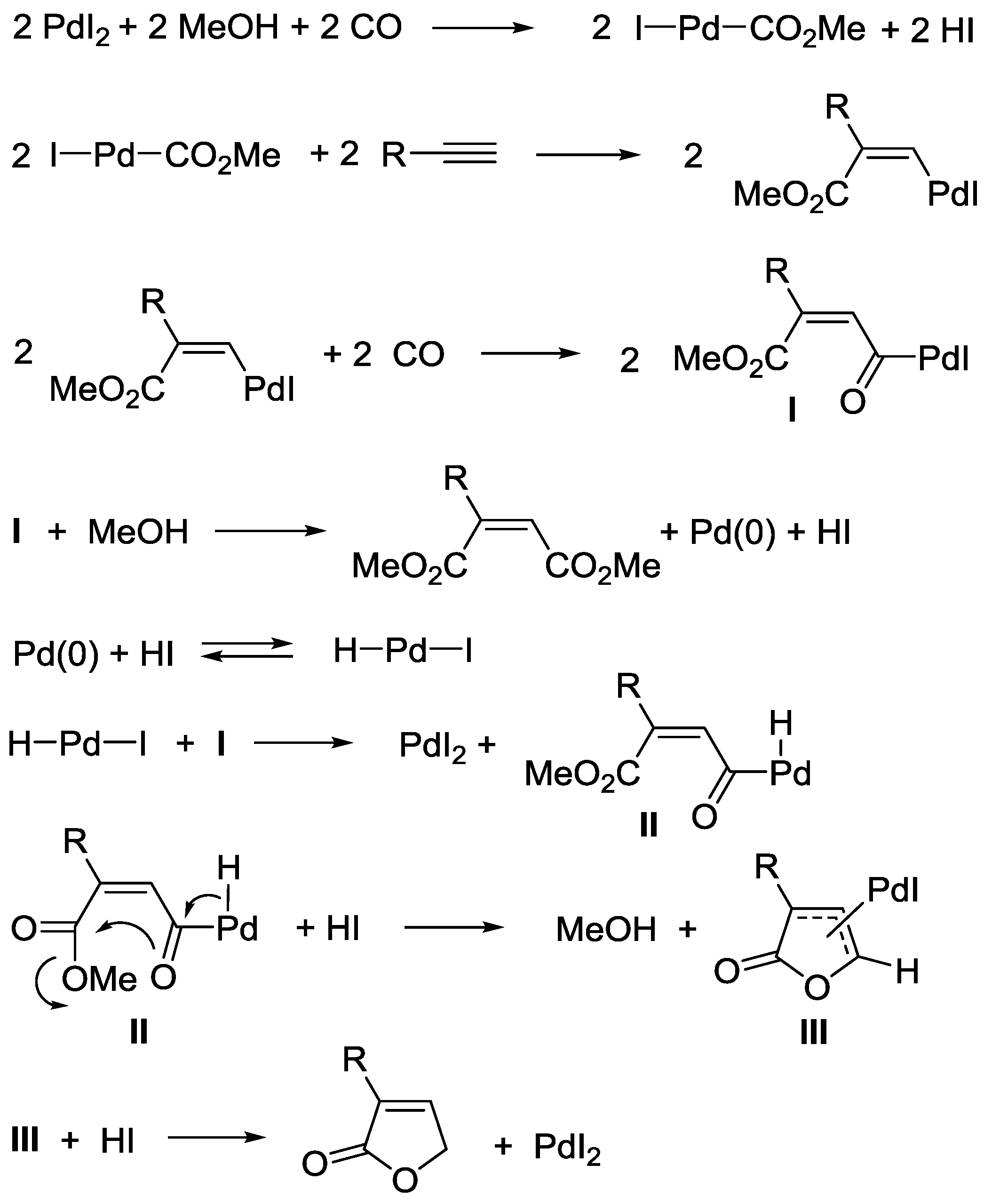
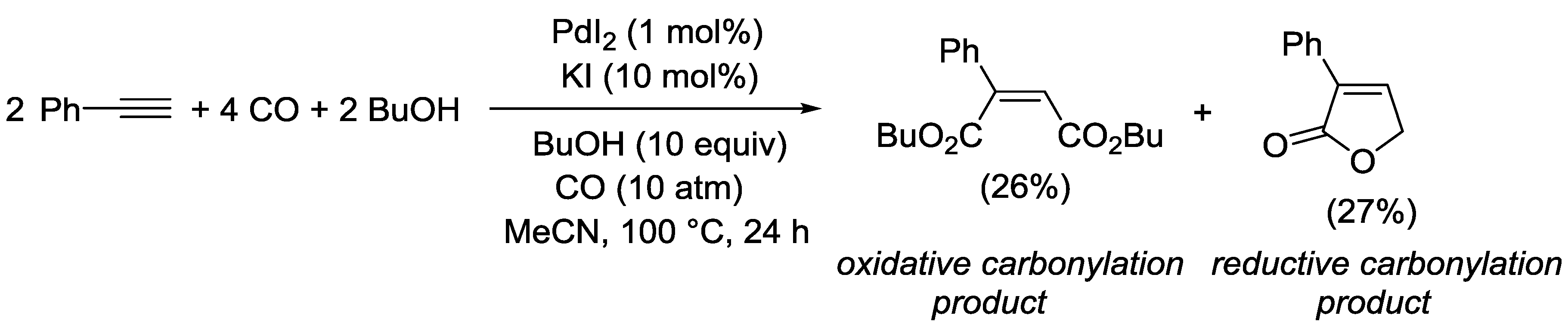

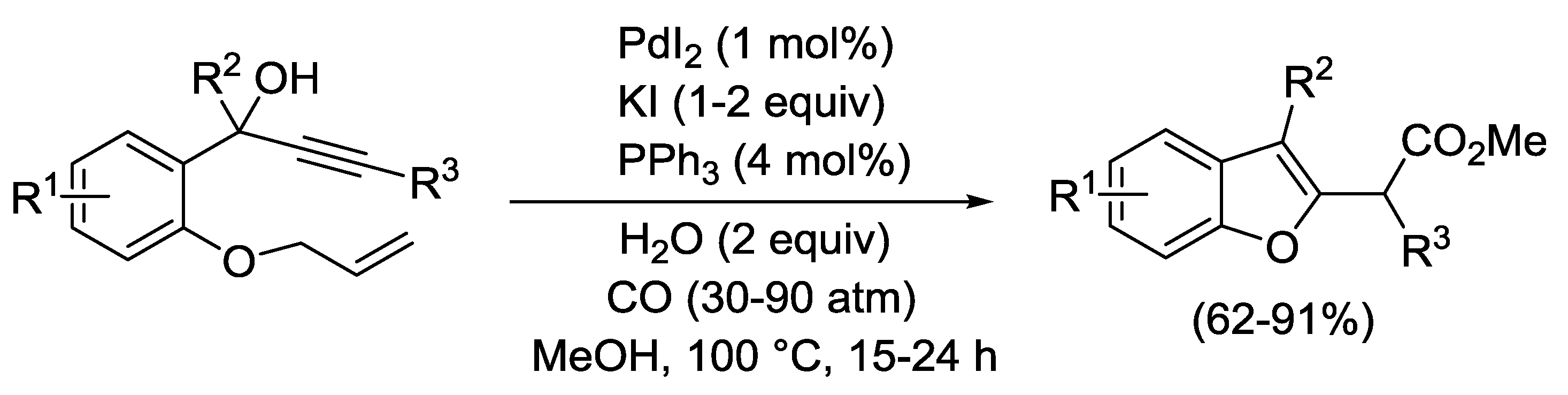

© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mancuso, R.; Della Ca’, N.; Veltri, L.; Ziccarelli, I.; Gabriele, B. PdI2-Based Catalysis for Carbonylation Reactions: A Personal Account. Catalysts 2019, 9, 610. https://doi.org/10.3390/catal9070610
Mancuso R, Della Ca’ N, Veltri L, Ziccarelli I, Gabriele B. PdI2-Based Catalysis for Carbonylation Reactions: A Personal Account. Catalysts. 2019; 9(7):610. https://doi.org/10.3390/catal9070610
Chicago/Turabian StyleMancuso, Raffaella, Nicola Della Ca’, Lucia Veltri, Ida Ziccarelli, and Bartolo Gabriele. 2019. "PdI2-Based Catalysis for Carbonylation Reactions: A Personal Account" Catalysts 9, no. 7: 610. https://doi.org/10.3390/catal9070610
APA StyleMancuso, R., Della Ca’, N., Veltri, L., Ziccarelli, I., & Gabriele, B. (2019). PdI2-Based Catalysis for Carbonylation Reactions: A Personal Account. Catalysts, 9(7), 610. https://doi.org/10.3390/catal9070610