Kinetic Study of Peroxidase-Catalyzed Oxidation of 2-Hydroxyanthracene and 9-Phenanthrol in Presence of Biosurfactant Escin
Abstract
1. Introduction
2. Results and Discussion
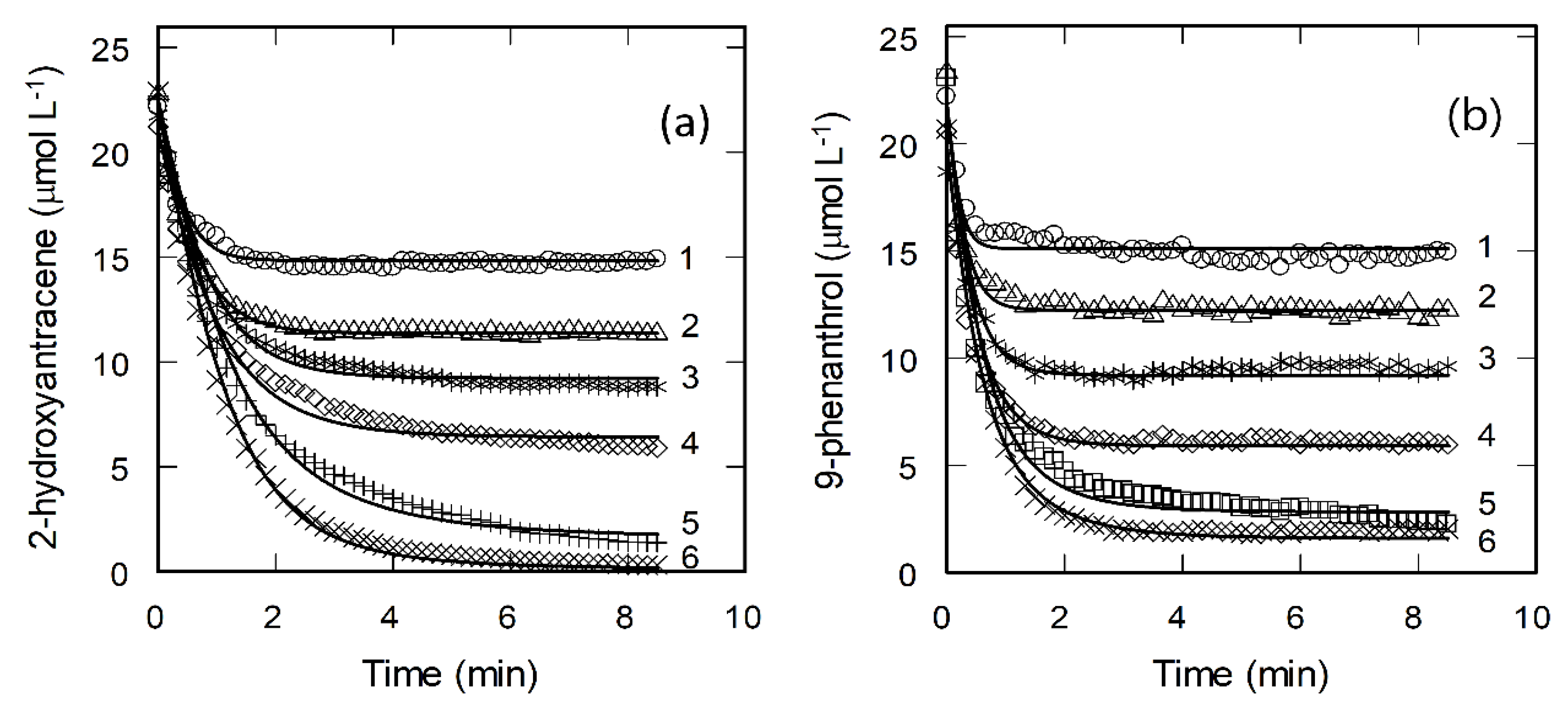
2.1. Kinetics of Peroxidase-Catalyzed 2-Hydroxyanthracene and 9-Phenanthrol Oxidation in the Absence and Presence of Biosurfactant Escin
2.2. Enhancing Effect of Escin Monomers on Biocatalytic Oxidation of 2-Hydroxyanthracene and 9-Phenanthrol
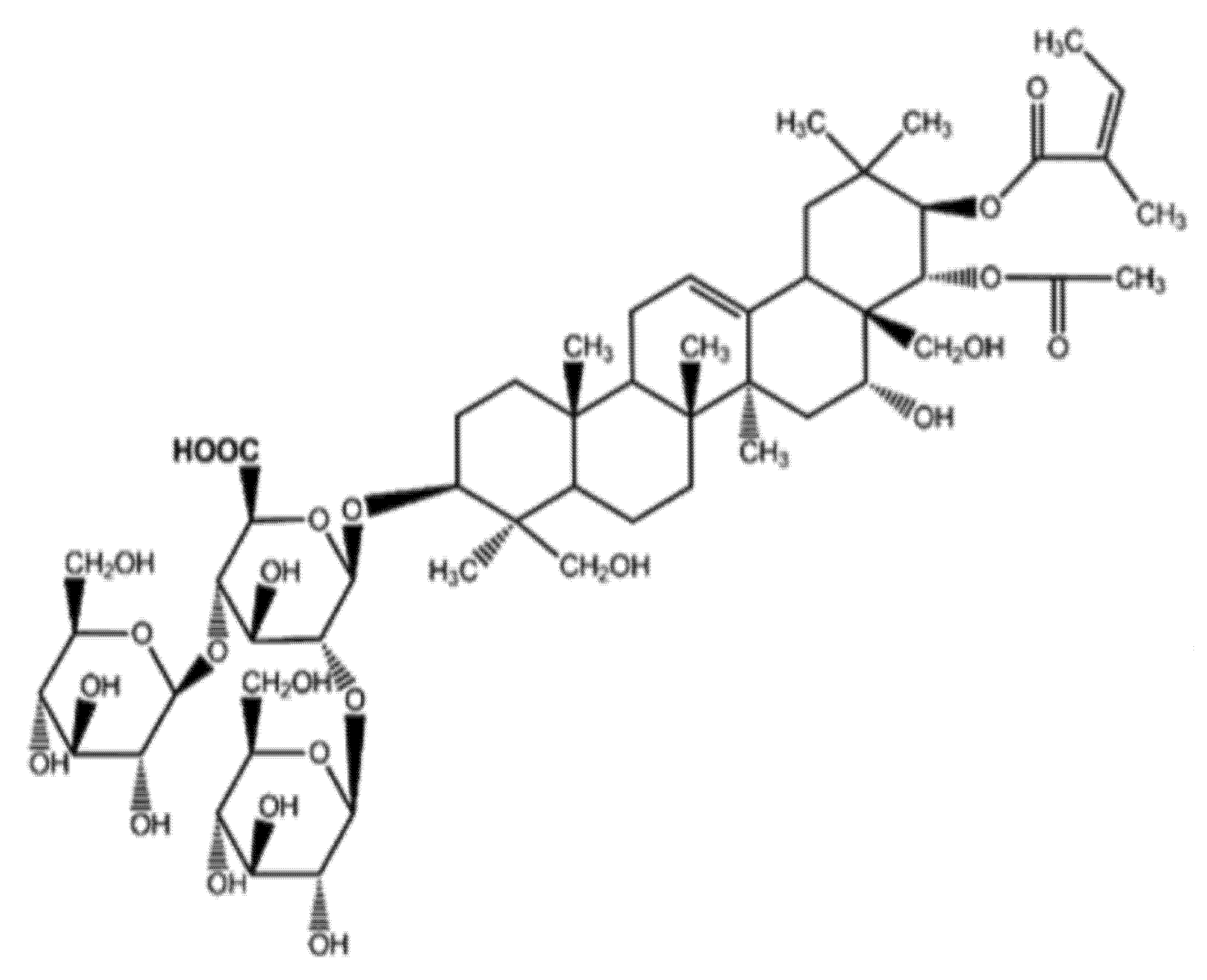
2.3. Mechanism of Escin Action
3. Materials and Methods
3.1. Materials
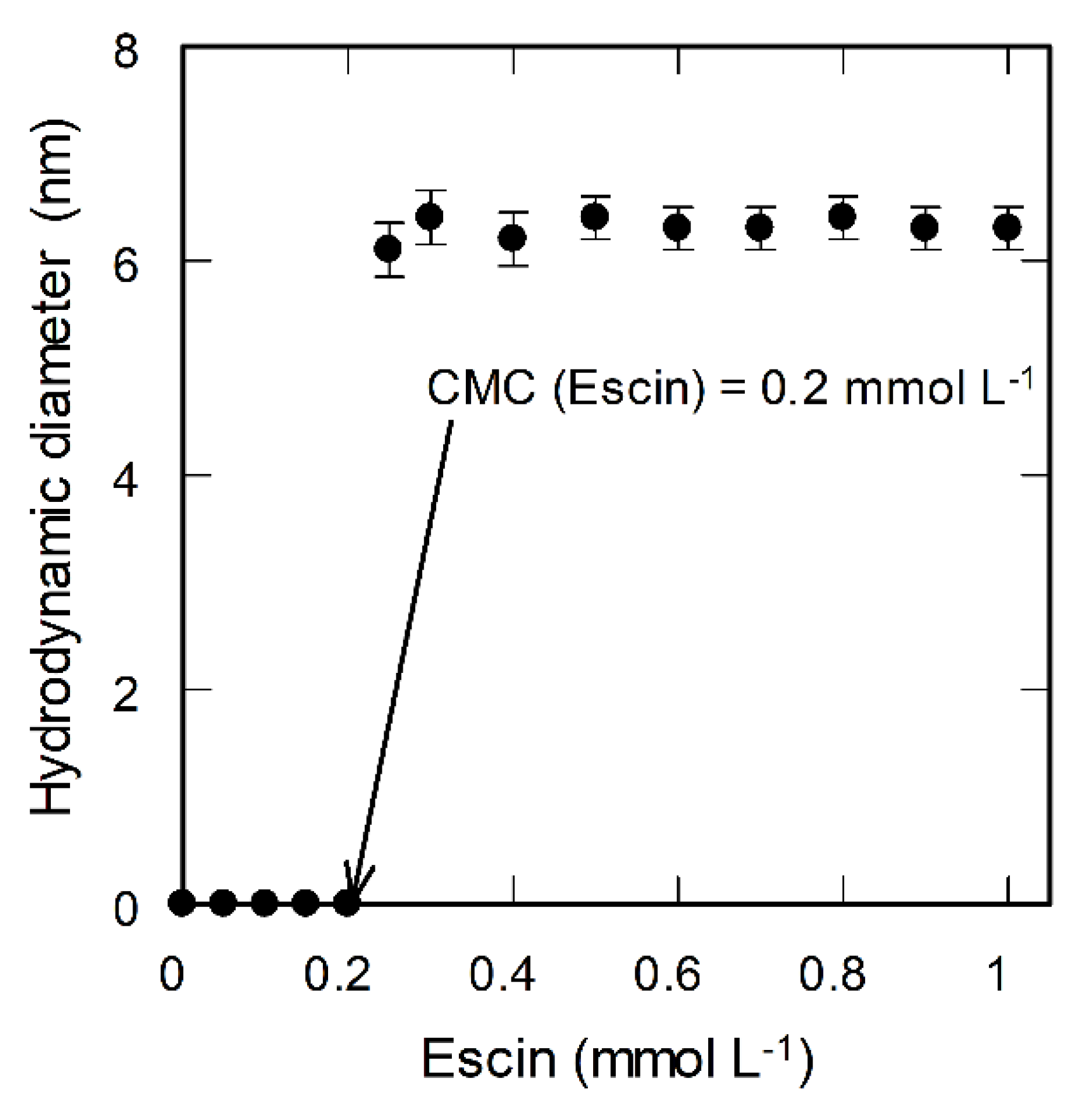
3.2. CMC Determination
3.3. Kinetic Analysis
3.4. Mathematical Calculations
4. Conclusions
Funding
Acknowledgments
Conflicts of Interest
References
- Anku, W.W.; Mamo, M.A.; Govender, P.P. Phenolic Compounds in Water: Sources, Reactivity, Toxicity and Treatment Methods. In Phenolic Compounds. Natural Sources, Importance and Applications; Soto-Hernández, M., Ed.; IntechOpen: London, UK, 2017; Volume 17, pp. 419–443. [Google Scholar]
- Glezer, V. Environmental effects of substituted phenols. In The Chemistry of Phenols Part 2; Rappoport, Z., Ed.; John Wiley & Sons: Chichester, UK, 2003; Volume 18, pp. 1347–1368. [Google Scholar]
- Kurnik, K.; Treder, K.; Skorupa-Kłaput, M.; Tretyn, A.; Tyburski, J. Removal of Phenol from Synthetic and Industrial Wastewater by Potato Pulp Peroxidases. Water Air Soil Pollut. 2015, 226, 254–273. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.; Guo, M.Y.; Gao, Y.H.; Bai, X.H.; Zhou, X.W. Expression and characteristics of manganese peroxidase from Ganoderma lucidum in Pichia pastoris and its application in the degradation of four dyes and phenol. BMC Biotechnol. 2017, 17, 19–31. [Google Scholar] [CrossRef] [PubMed]
- Bansal, N.; Kanwar, S.S. Peroxidase(s) in Environment Protection. Sci. World J. 2013, 2013, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Krainer, F.W.; Glieder, A. An updated view on horseradish peroxidases: Recombinant production and biotechnological applications. Appl. Microbiol. Biotechnol. 2015, 99, 1611–1625. [Google Scholar] [CrossRef] [PubMed]
- Dunford, H.B. Horseradish peroxidase: Structure and kinetic properties. In Peroxidases in Chemistry and Biology; Everse, J., Everse, K.E., Grisham, M.B., Eds.; CRC Press: Boca Raton, FL, USA, 1991; Volume 2, pp. 1–25, 225–227. [Google Scholar]
- Ji, G.L.; Zhang, H.B.; Huang, F.; Huang, X. Effects of nonionic surfactant Triton X-100 on the laccase-catalyzed conversion of bisphenol A. J. Environ. Sci. 2009, 21, 1486–1490. [Google Scholar] [CrossRef]
- Robinson, P.K. Enzymes: Principles and biotechnological applications. Essays Biochem. 2015, 59, 1–41. [Google Scholar] [CrossRef]
- Ziemys, A.; Kulys, J. Heme peroxidase clothing and inhibition with polyphenolic substances revealed by molecular modeling. Comput. Biol. Chem. 2005, 29, 83–90. [Google Scholar] [CrossRef]
- Mao, L.; Luo, S.; Huang, Q.; Lu, J. Horseradish Peroxidase Inactivation: Heme Destruction and Influence of Polyethylene Glycol. Sci. Rep. 2013, 3, 3126–3135. [Google Scholar] [CrossRef]
- Kulys, J.; Ivanec-Goranina, R. Peroxidase catalyzed phenolic compounds oxidation in presence of surfactant Dynol 604: A kinetic investigation. Enzyme Microb. Technol. 2009, 44, 368–372. [Google Scholar] [CrossRef]
- Dayeh, V.R.; Chow, S.L.; Schirmer, K.; Lynn, D.H.; Bols, N.C. Evaluating the toxicity of Triton X-100 to protozoan, fish, and mammalian cells using fluorescent dyes as indicators of cell viability. Ecotoxicol. Environ. Saf. 2004, 57, 375–382. [Google Scholar] [CrossRef]
- Li, M.H. Effects of nonionic and ionic surfactants on survival, oxidative stress, and cholinesterase activity of planarian. Chemosphere 2008, 70, 1796–1803. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.F.; Zeng, G.M.; Zhong, H.; Yuan, X.Z.; Fu, H.Y.; Zhou, M.F.; Ma, X.L.; Li, H.; Li, J.B. Effect of dirhamnolipid on the removal of phenol catalyzed by laccase in aqueous solution. World J Microbiol. Biotechnol. 2012, 28, 175–181. [Google Scholar] [CrossRef] [PubMed]
- Tsibranska, S.; Ivanova, A.; Tcholakova, S.; Denkov, N. Self-Assembly of Escin Molecules at the Air-Water Interface as Studied by Molecular Dynamics. Langmuir 2017, 33, 8330–8341. [Google Scholar] [CrossRef] [PubMed]
- Schrlau, J.E.; Kramer, A.L.; Chlebowski, A.; Truong, L.; Tanguay, R.L.; Massey Simonich, S.L.; Semprini, L. Formation of Developmentally Toxic Phenanthrene Metabolite Mixtures by Mycobacterium sp. ELW1. Environ. Sci. Technol. 2017, 51, 8569–8578. [Google Scholar] [CrossRef] [PubMed]
- Lampi, M.A.; Gurska, J.; McDonald, K.I.; Xie, F.; Huang, X.D.; Dixon, D.G.; Greenberg, B.M. Photoinduced toxicity of polycyclic aromatic hydrocarbons to Daphnia magna: Ultraviolet-mediated effects and the toxicity of polycyclic aromatic hydrocarbon photoproducts. Environ. Toxicol. Chem. 2006, 25, 1079–1087. [Google Scholar] [CrossRef] [PubMed]
- Dudek-Makuch, M.; Studzińska-Sroka, E. Horse chestnut—Efficacy and safety in chronic venous insufficiency: An overview. Rev. Bras. Farmacogn. 2015, 25, 533–541. [Google Scholar] [CrossRef]
- Sirtori, C.R. Aescin: Pharmacology, pharmacokinetics and therapeutic profile. Pharmacol. Res. 2001, 44, 183–193. [Google Scholar] [CrossRef]
- Wei, F.; Ma, L.Y.; Cheng, X.L.; Lin, R.C.; Jin, W.T.; Khan, I.A.; Lu, J.Q. Preparative HPLC for purification of four isomeric bioactive saponins from the seeds of Aesculus chinensis. J. Liq. Chromatogr. Relat. Technol. 2005, 28, 763–773. [Google Scholar] [CrossRef]
- Andersen, M.B.; Hsuanyu, Y.; Welinder, K.G.; Schneider, P.; Dunford, H.B. Spectral and kinetic properties of oxidized intermediantes of Coprinus cinereus peroxidase. Acta Chem. Scand. 1991, 45, 1080–1086. [Google Scholar] [CrossRef]
- Nelson, D.P.; Kiesow, L. Enthalpy of decomposition of hydrogen peroxide by catalase at 25 degrees C (with molar extinction coefficients of H2O2 solutions in the UV). Anal. Biochem. 1972, 49, 474–478. [Google Scholar] [CrossRef]
- Svir, I.; Klymenko, O.V.; Oleinick, A.I.; Platz, M.S. KinFitSim (Version 2.1)—A powerful tool for kinetic simulation of any reaction mechanism and fitting of any number of pairs of theoretical and experimental data sets. Radioelektron. Inform. 2004, 4, 21–24. [Google Scholar]
| AHDs | Escin, μmol L−1 | k2, mol−1 L s−1 | k3, mol−1 L s−1 | kin, mol−1 L s−1 |
|---|---|---|---|---|
| 2-hydroxyanthracene | 0 | (1.0 ± 0.3) × 108 | (1.0 ± 0.2) × 108 | (2.2 ± 0.1) × 104 |
| 2-hydroxyanthracene | 0.2 | (1.0 ± 0.2) × 108 | (1.0 ± 0.3) × 108 | (9.5 ± 0.3) × 103 |
| 2-hydroxyanthracene | 0.5 | (1.0 ± 0.3) × 108 | (1.0 ± 0.2) × 108 | (5.3 ± 0.2) × 103 |
| 2-hydroxyanthracene | 0.7 | (9.2 ± 0.5) × 107 | (9.0 ± 0.5) × 107 | (4.2 ± 0.2) × 103 |
| 2-hydroxyanthracene | 1 | (9.0 ± 0.6) × 107 | (9.5 ± 0.4) × 107 | (1.6 ± 0.3) × 103 |
| 2-hydroxyanthracene | 2 μmol L−1–0.2 mmol L−1 | (1.0 ± 0.2) × 108 | (1.0 ± 0.2) × 108 | (9.0 ± 0.2) × 102 |
| 9-phenanthrol | 0 | (3.0 ± 0.1) × 107 | (3.0 ± 0.1) × 107 | (5.0 ± 0.2) × 104 |
| 9-phenanthrol | 0.2 | (3.0 ± 0.1) × 107 | (3.1 ± 0.1) × 107 | (3.3 ± 0.4) × 104 |
| 9-phenanthrol | 0.5 | (3.0 ± 0.1) × 107 | (3.0 ± 0.1) × 107 | (1.5 ± 0.3) × 104 |
| 9-phenanthrol | 0.7 | (3.2 ± 0.2) × 107 | (3.3 ± 0.2) × 107 | (7.4 ± 0.2) × 103 |
| 9-phenanthrol | 1 | (3.0 ± 0.1) × 107 | (3.0 ± 0.2) × 107 | (3.3 ± 0.1) × 103 |
| 9-phenanthrol | 2 μmol L−1–0.2 mmol L−1 | (3.1 ± 0.2) × 107 | (3.0 ± 0.1) × 107 | (1.5 ± 0.1) × 103 |
© 2019 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ivanec-Goranina, R. Kinetic Study of Peroxidase-Catalyzed Oxidation of 2-Hydroxyanthracene and 9-Phenanthrol in Presence of Biosurfactant Escin. Catalysts 2019, 9, 591. https://doi.org/10.3390/catal9070591
Ivanec-Goranina R. Kinetic Study of Peroxidase-Catalyzed Oxidation of 2-Hydroxyanthracene and 9-Phenanthrol in Presence of Biosurfactant Escin. Catalysts. 2019; 9(7):591. https://doi.org/10.3390/catal9070591
Chicago/Turabian StyleIvanec-Goranina, Rūta. 2019. "Kinetic Study of Peroxidase-Catalyzed Oxidation of 2-Hydroxyanthracene and 9-Phenanthrol in Presence of Biosurfactant Escin" Catalysts 9, no. 7: 591. https://doi.org/10.3390/catal9070591
APA StyleIvanec-Goranina, R. (2019). Kinetic Study of Peroxidase-Catalyzed Oxidation of 2-Hydroxyanthracene and 9-Phenanthrol in Presence of Biosurfactant Escin. Catalysts, 9(7), 591. https://doi.org/10.3390/catal9070591





