Heteropolyacid Salt Catalysts for Methanol Conversion to Hydrocarbons and Dimethyl Ether: Effect of Reaction Temperature
Abstract
1. Introduction
2. Results and Discussion
2.1. Characterization of the Catalysts
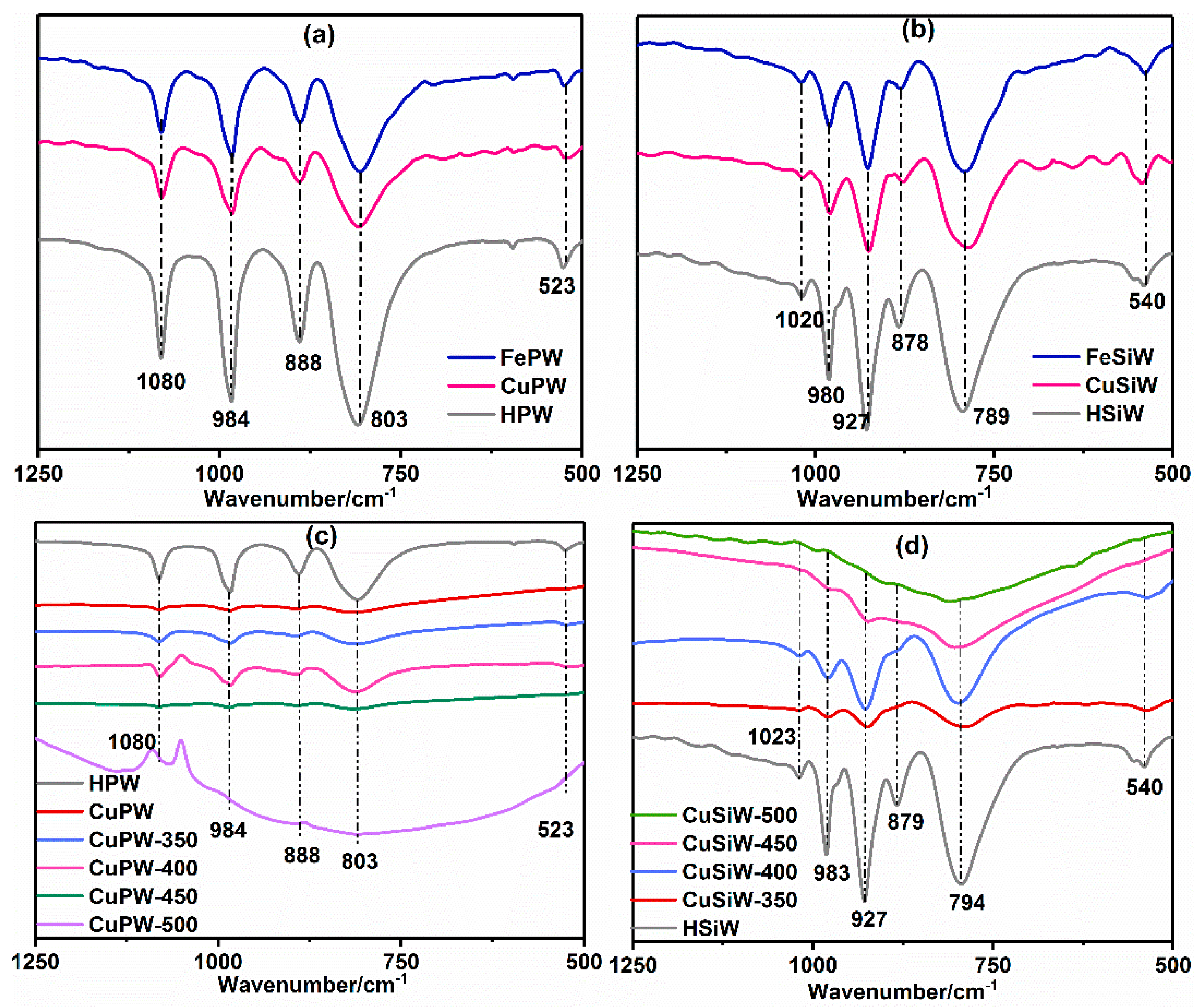
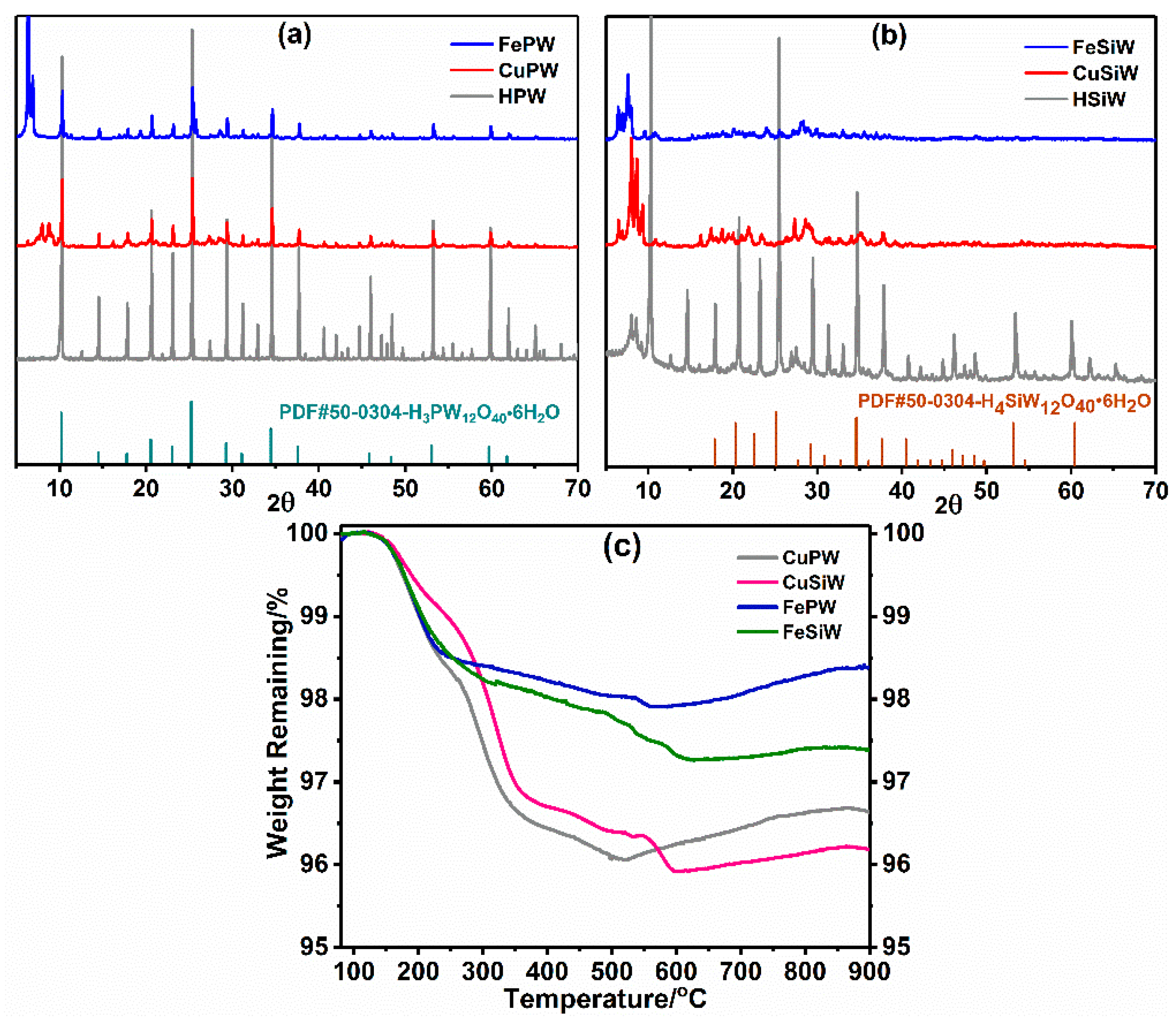
2.1.1. Structure and Thermal Stability of Heteropolyacid Salts
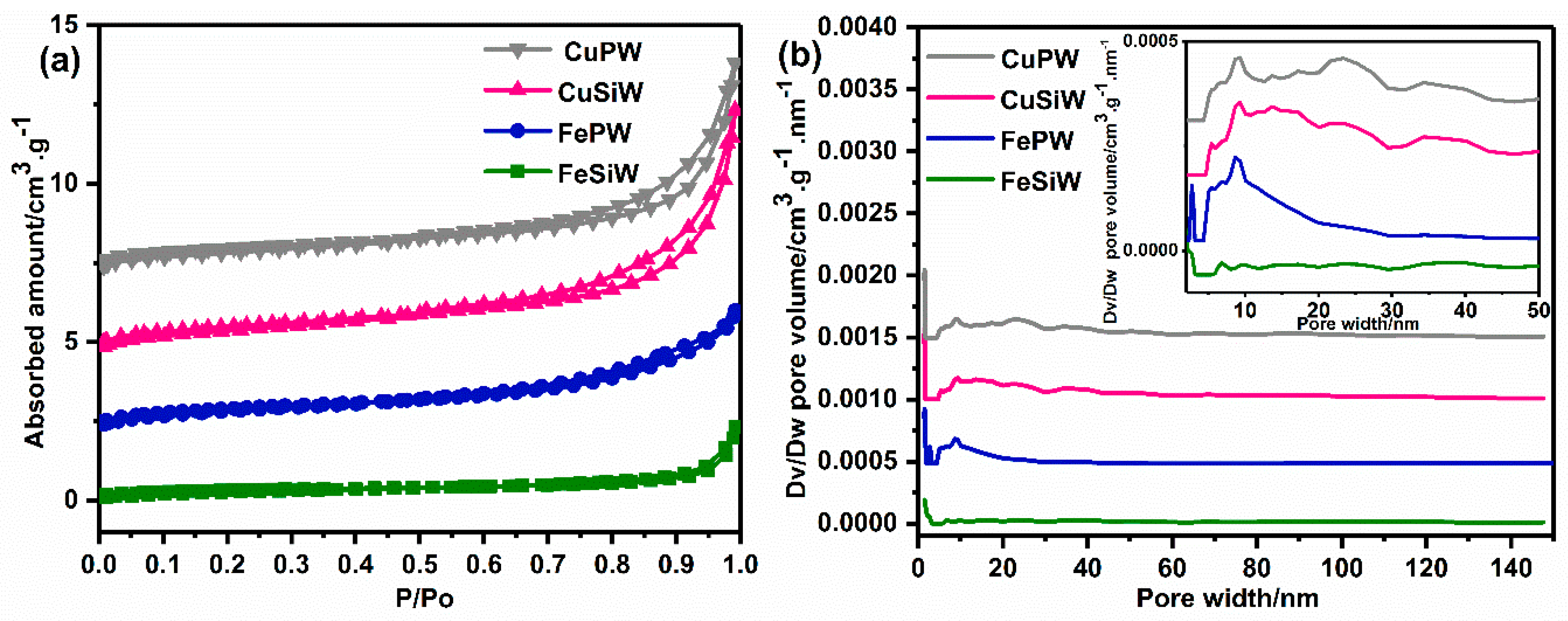
2.1.2. Composition and Textural Properties of Heteropolyacid Salt Catalysts
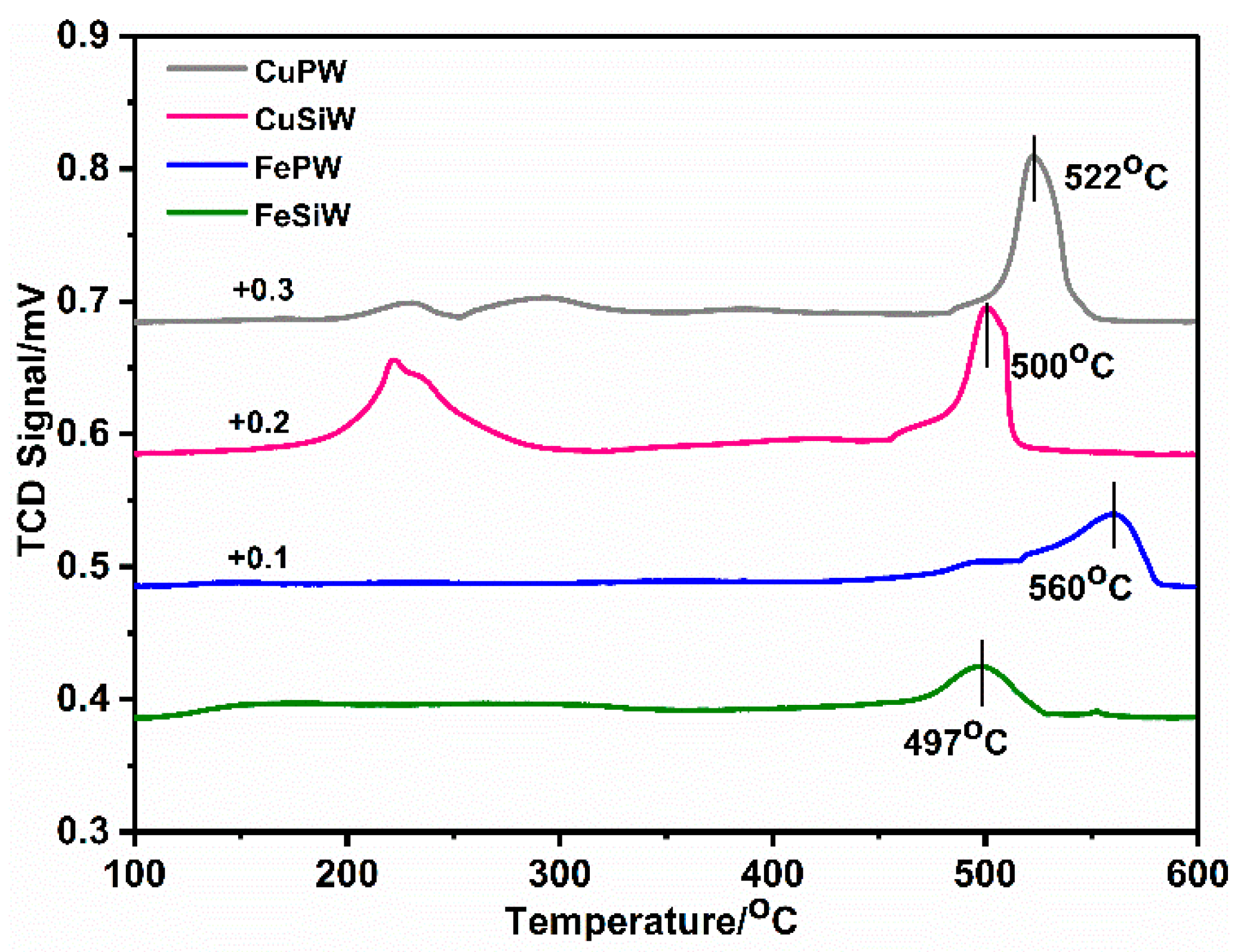
2.1.3. Acid Properties of Heteropolyacid Salt Catalysts
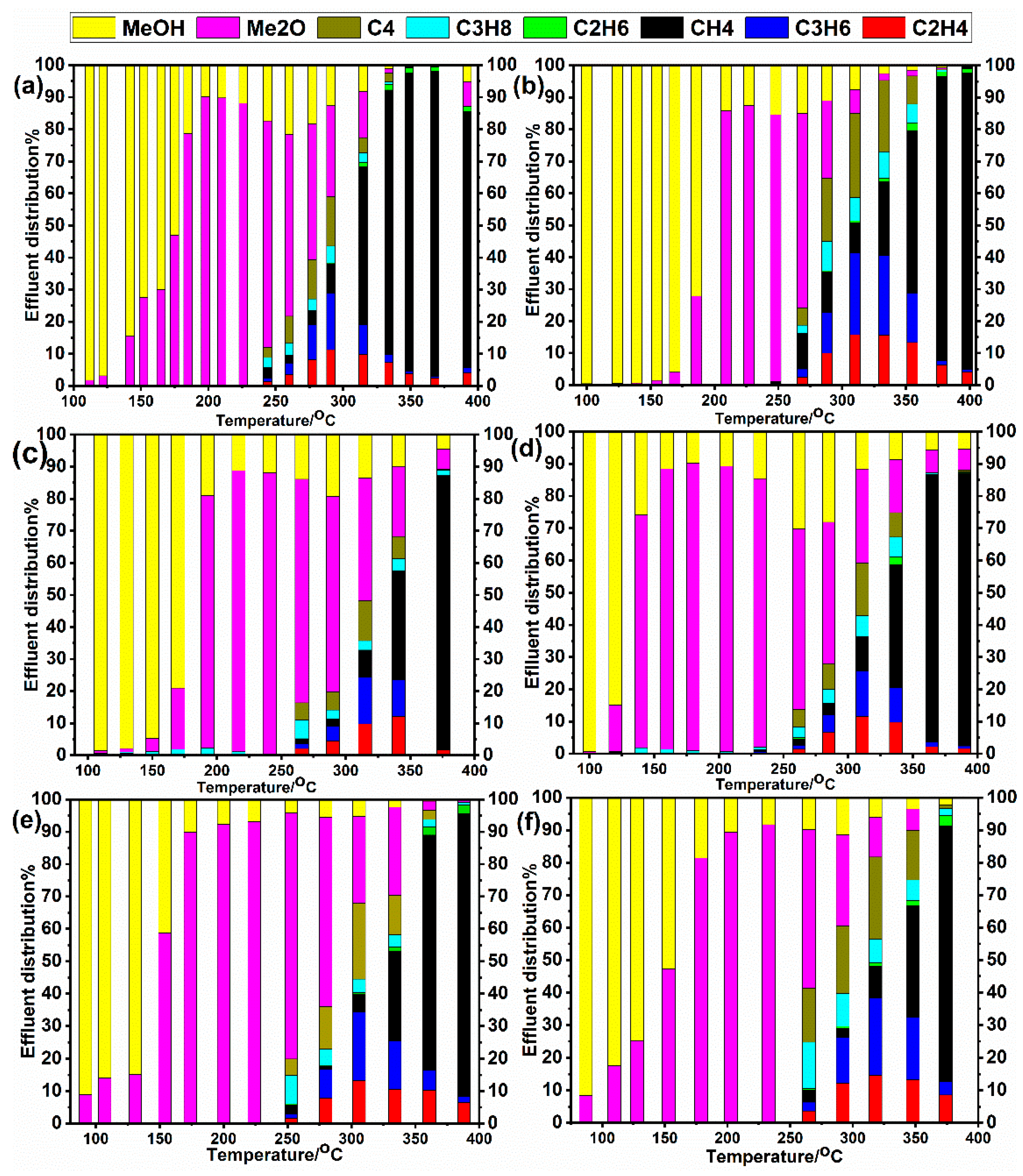
2.2. Temperature Programmed Reaction of Methanol Conversion
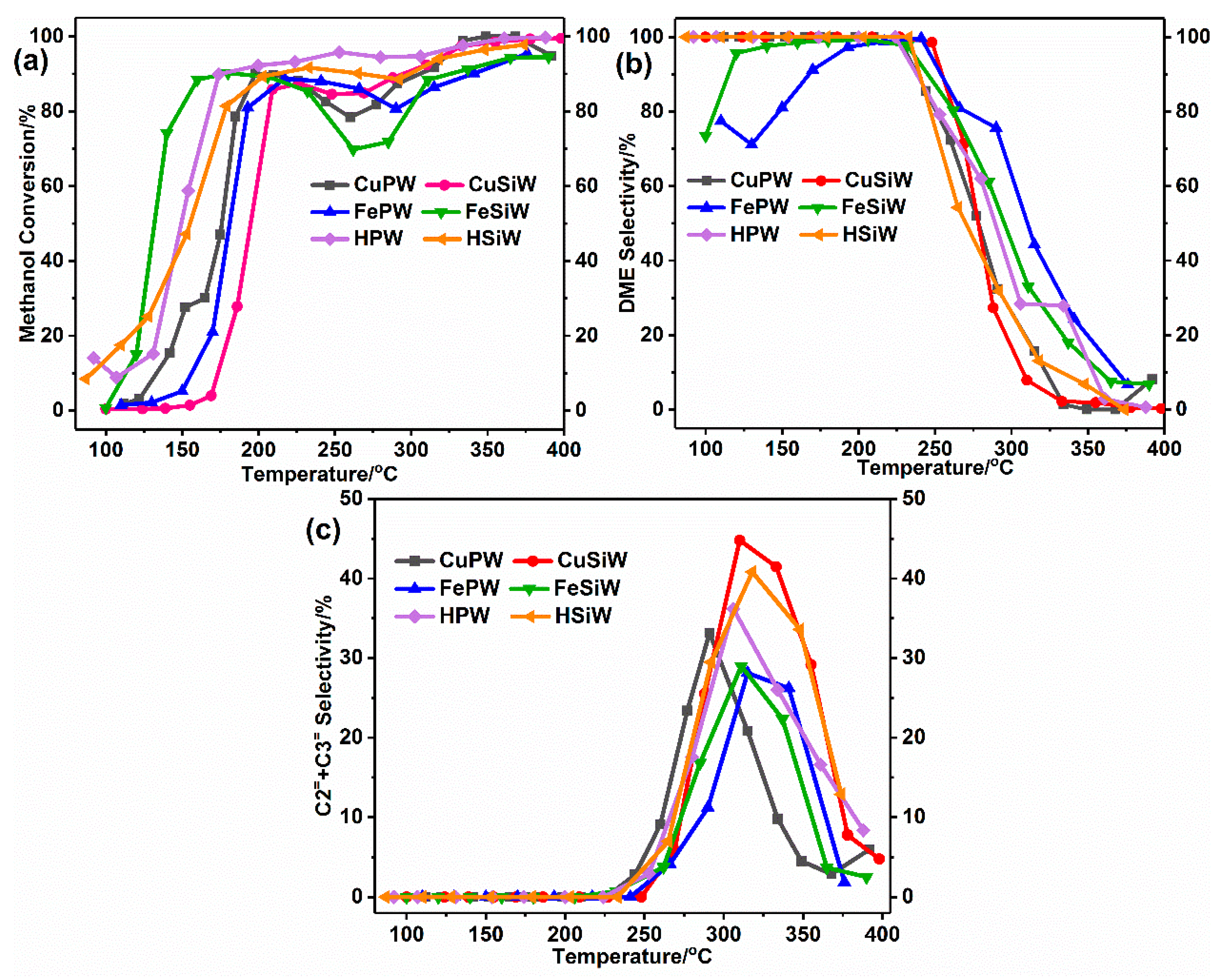
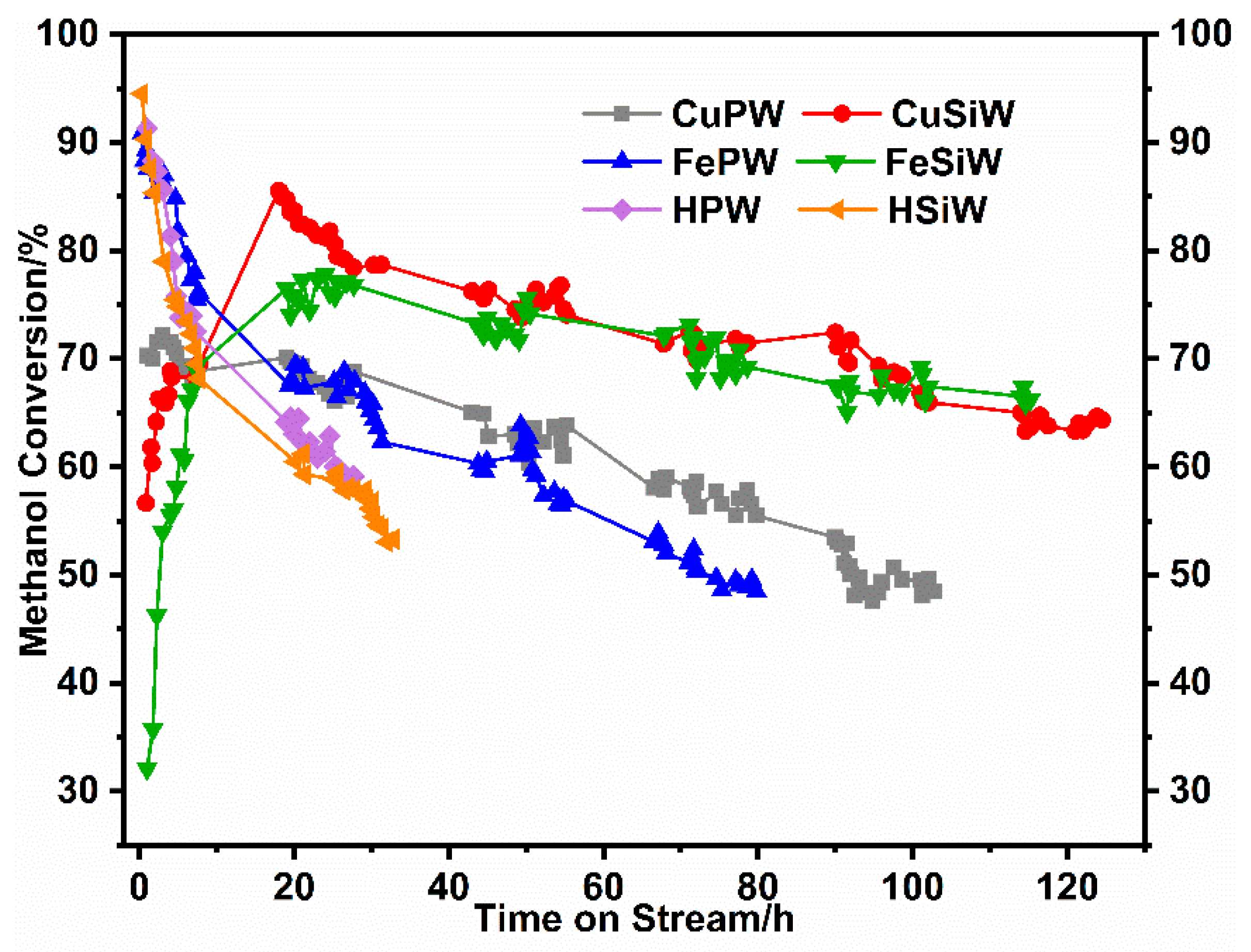
2.3. Catalytic Performance of Heteropolyacid Salts for Methanol to DME
3. Experimental
3.1. Chemical Reagents
3.2. Measurement
3.3. Synthesis of Heteropolyacid Salts Catalyst
3.4. Catalytic Evaluation
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Mokrani, T.; Scurell, M. Gas Conversion to Liquid Fuels and Chemicals: The Methanol Route-Catalysis and Processes Development. Catal. Rev. Sci. Eng. 2009, 51, 1–145. [Google Scholar] [CrossRef]
- Galvita, V.V.; Semin, G.L.; Belyaev, V.D.; Yurieva, T.M.; Sobyanin, V.A. Production of hydrogen from dimethyl ether. Appl. Catal. A Gen. 2001, 216, 85–90. [Google Scholar] [CrossRef]
- Takeishi, K.; Suzuki, H. Steam reforming of dimethyl ether. Appl. Catal. A Gen. 2004, 260, 111–117. [Google Scholar] [CrossRef]
- Ramos, F.S.; Duarte de Farias, A.M.; Borges, L.E.P.; Monteiro, J.L.; Fraga, M.A.; Sousa-Aguiar, E.F.; Appel, L.G. Role of dehydration catalyst acid properties on one-step DME synthesis over physical mixtures. Catal. Today 2005, 101, 39–44. [Google Scholar] [CrossRef]
- Dermeche, L.; Salhi, N.; Hocine, S.; Thouvenot, R.; Rabia, C.; Mol, J. Effective Dawson type polyoxometallate catalysts for methanol oxidation. Catal. A Chem. 2012, 356, 29–35. [Google Scholar] [CrossRef]
- Youn, I.M.; Park, S.H.; Roh, H.G.; Lee, C.S. Investigation on the fuel spray and emission reduction characteristics for dimethyl ether (DME) fueled multi-cylinder diesel engine with common-rail injection system. Fuel Process. Technol. 2011, 92, 1280–1287. [Google Scholar] [CrossRef]
- Wood, D.A.; Nwaoha, C.; Towler, B.F. Gas-to-liquids (GTL): A review of an industry offering several routes for monetizing natural gas. J. Nat. Gas Sci. Eng. 2012, 9, 196–208. [Google Scholar] [CrossRef]
- Fleisch, T.H.; Basu, A.R.; Sills, A. Introduction and advancement of a new clean global fuel: The status of DME developments in China and beyond. J. Nat. Gas Sci. Eng. 2012, 9, 94–107. [Google Scholar] [CrossRef]
- Pontzen, F.; Liebner, W.; Gronemann, V.; Rothaemel, M.; Ahlers, B. CO2-based methanol and DME Efficient technologies for industrial scale production. Catal. Today 2011, 171, 242–250. [Google Scholar] [CrossRef]
- Hu, H.; Cao, F.H.; Ying, W.Y.; Sun, Q.W.; Fang, D.Y. Study of coke behavior of catalyst during methanol-to-olefins process based on a special TGA reactor. Chem. Eng. J. 2010, 160, 770–778. [Google Scholar] [CrossRef]
- Liu, Z.M.; Sun, C.L.; Wang, G.W.; Wang, Q.X.; Cai, G.Y. New progress in R&D of lower olefin synthesis. Fuel Process. Technol. 2000, 62, 161–172. [Google Scholar]
- Gertsberg, L.Y.; Surzhan, G.P.; Barvin, V.I. Efficient method of dimethyl sulfate production. Tr. Buryat. Inst. Estestv. Nauk Buryat. Filial Sib. Otd. Akad. Nauk SSSR 1971, 10, 228–229. [Google Scholar]
- Liu, D.H.; Huang, X.; Hu, L.; Fang, D.Y.; Ying, W.Y.; Chen, D.S. Synthesis of ethylidene diacetate from dimethyl ether, CO and H2. J. Nat. Gas Chem. 2010, 19, 165–168. [Google Scholar] [CrossRef]
- Bahmanpour, A.M.; Héroguel, F.; Baranowski, C.J.; Luterbacher, J.S.; Kröcher, O. Selective synthesis of dimethyl ether on eco-friendly K10 montmorillonite clay. Appl. Catal. A Gen. 2018, 560, 165–170. [Google Scholar] [CrossRef]
- Carr, R.T.; Neurock, M.; Iglesia, E. Catalytic consequences of acid strength in the conversion of methanol to dimethyl ether. J. Catal. 2011, 278, 78–93. [Google Scholar] [CrossRef]
- Moses, P.G.; Nørskov, J.K. Methanol to Dimethyl Ether over ZSM-22: A Periodic Density Functional Theory Study. ACS Catal. 2013, 3, 735–745. [Google Scholar] [CrossRef]
- Ladera, R.; Finocchio, E.; Rojas, S.; Busca, G.; Fierro, J.L.G.; Ojeda, M. Supported WOx-based catalysts for methanol dehydration to dimethyl ether. Fuel 2013, 113, 1–9. [Google Scholar] [CrossRef]
- Arena, F.; Spadaro, L.; Di Blasi, L.; Bonura, G.; Frusteri, F. Integrated synthesis of dimethylether via CO2 hydrogenation. Stud. Surf. Sci. Catal. 2004, 147, 385–390. [Google Scholar]
- Bonura, G.; Cordaro, M.; Spadaro, L.; Cannilla, C.; Arena, F.; Frusteri, F. Hybrid Cu-ZnO-ZrO2/H-ZSM-5 system for the direct synthesis of DME by CO2 hydrogenation. Appl. Catal. B-Environ. 2013, 140–141, 16–24. [Google Scholar] [CrossRef]
- Bonura, G.; Cordaro, M.; Cannilla, C.; Mezzapica, A.; Spadaro, L.; Arena, F.; Frusteri, F. Catalytic behavior of a bifunctional system for the one step synthesis of DME by CO2 hydrogenation. Catal. Today 2014, 228, 51–57. [Google Scholar] [CrossRef]
- Raoof, F.; Taghizadeh, M.; Eliassi, A.; Yaripour, F. Effects of temperature and feed composition on catalytic dehydration of methanol to dimethyl ether over γ-alumina. Fuel 2008, 87, 2967–2971. [Google Scholar] [CrossRef]
- Tavan, Y.; Khosravi Nikou, M.R.; Shariati, A. Effect of the P/Al ratio on the performance of modified HZSM-5 for methanol dehydration reaction. J. Ind. Eng. Chem. 2014, 20, 668–673. [Google Scholar] [CrossRef]
- Akarmazyan, S.S.; Panagiotopoulou, P.; Kambolis, A.; Papadopoulou, C.; Kondarides, D.I. Methanol dehydration to dimethyl ether over Al2O3 catalysts. Appl. Catal. B-Environ. 2014, 145, 136–148. [Google Scholar] [CrossRef]
- Sabour, B.; Peyrovi, M.H.; Hamoule, T.; Rashidzadeh, M. Catalytic dehydration of methanol to dimethyl ether (DME) over Al-HMS catalysts. J. Ind. Eng. Chem. 2014, 20, 222–227. [Google Scholar] [CrossRef]
- Sang, Y.; Li, H.; Zhu, M.; Ma, K.; Jiao, Q.; Wu, Q. Catalytic performance of metal ion doped MCM-41 for methanol dehydration to dimethyl ether. J. Porous Mater. 2013, 20, 1509–1518. [Google Scholar] [CrossRef]
- Ladera, R.; Finocchio, E.; Rojas, S.; Fierro, J.L.G.; Ojeda, M. Supported niobium catalysts for methanol dehydration to dimethyl ether: FTIR studies of acid properties. Catal. Today 2012, 192, 136–143. [Google Scholar] [CrossRef]
- Lertjiamratn, K.; Praserthdam, P.; Arai, M.; Panpranot, J. Modification of acid properties and catalytic properties of AlPO4 by hydrothermal pretreatment for methanol dehydration to dimethyl ether. Appl. Catal. A 2010, 378, 119–123. [Google Scholar] [CrossRef]
- Vishwanathan, V.; Roh, H.S.; Kim, J.W.; Jun, K.W. Surface Properties and Catalytic Activity of TiO2–ZrO2 Mixed Oxides in Dehydration of Methanol to Dimethyl Ether. Catal. Lett. 2004, 96, 23–28. [Google Scholar] [CrossRef]
- Tokay, K.C.; Dogu, T.; Dogu, G. Dimethyl ether synthesis over alumina based catalysts. Chem. Eng. J. 2012, 184, 278–285. [Google Scholar] [CrossRef]
- Ladera, R.M.; Ojeda, M.; Fierro, J.L.G.; Rojas, S. TiO2-supported heteropoly acid catalysts for dehydration of methanol to dimethyl ether: Relevance of dispersion and support interaction. Catal. Sci. Technol. 2015, 5, 484–491. [Google Scholar] [CrossRef]
- Xu, M.; Lunsford, J.H.; Goodman, D.W.; Bhattacharayya, A. Synthesis of dimethyl ether (DME) from methanol over solid-acid catalysts. Appl. Catal. A 1997, 149, 289–301. [Google Scholar] [CrossRef]
- Takeguchi, T.; Yanagisawa, K.; Inui, T.; Inoue, M. Effect of the property of solid acid upon syngas-to-dimethyl ether conversion on the hybrid catalysts composed of Cu-Zn-Ga and solid acids. Appl. Catal. A 2000, 192, 201–209. [Google Scholar] [CrossRef]
- Ladera, R.M.; Fierro, J.L.G.; Ojeda, M.; Rojas, S. TiO2-supported heteropoly acids for low-temperature synthesis of dimethyl ether from methanol. J. Catal. 2014, 312, 195–203. [Google Scholar] [CrossRef]
- Schnee, J.; Gaigneaux, E.M. Lifetime of the H3PW12O40 heteropolyacid in the methanol-to-DME process: A question of pre-treatment. Appl. Catal. A Gen. 2017, 538, 174–180. [Google Scholar] [CrossRef]
- Alharbi, W.; Kozhevnikova, E.F.; Kozhevnikov, I.V. Dehydration of Methanol to Dimethyl Ether over Heteropoly Acid Catalysts: The Relationship between Reaction Rate and Catalyst Acid Strength. ACS Catal. 2015, 5, 7186–7193. [Google Scholar] [CrossRef]
- Jones, A.L.; Carr, R.T.; Zones, S.I.; Iglesia, E. Acid strength and solvation in catalysis by MFI zeolites and effects of the identity, concentration and location of framework heteroatoms. J. Catal. 2014, 312, 58–68. [Google Scholar] [CrossRef]
- Hibi, T.; Takahashi, K.; Okuhara, T.; Misono, M.; Yoneda, Y. Catalysis by heteropoly compounds. X. Synthesis of lower olefins by conversion of dimethyl ether over 12-tungstophosphates. Appl. Catal. 1986, 24, 69–83. [Google Scholar] [CrossRef]
- Highfield, J.C.; Moffat, J.B. Characterization of sorbed intermediates and implications for the mechanism of chain growth in the conversion of methanol and ethanol to hydrocarbons over 12-tungstophosphoric acid using infrared photoacoustic spectroscopy. J. Catal. 1986, 98, 245–258. [Google Scholar] [CrossRef]
- Seo, C.W.; Jung, K.D.; Lee, K.Y.; Yoo, K.S. Dehydration of methanol over Nordstrandite based catalysts for dimethyl ether synthesis. J. Ind. Eng. Chem. 2009, 15, 649–652. [Google Scholar] [CrossRef]
- Çiftci, A.; Varisli, D.; Cem Tokay, K.; Asli Sezgi, N.; Dogu, T. Dimethyl ether, diethyl ether & ethylene from alcohols over tungstophosphoric acid based mesoporous catalysts. Chem. Eng. J. 2012, 207, 85–93. [Google Scholar]
- Micek-Ilnicka, A.; Bielanska, E.; Litynska-Dobrzynska, L.; Bielanski, A. Carbon nanotubes, silica and titania supported heteropolyacid H3PW12O40 as the catalyst for ethanol conversion. Appl. Catal. A 2012, 421, 91–98. [Google Scholar] [CrossRef]
- Varisli, D.; Tokay, C.K.; Ciftci, A.; Dogu, T.; Dogu, G. Methanol dehydration reaction to produce clean diesel alternative dimethylether over mesoporous aluminosilicate-based catalysts. Turk. J. Chem. 2009, 33, 355–366. [Google Scholar]
- Yoshio, O.; Toshihide, B.; Junya, S.; Tominaga, K. Conversion of methanol into hydrocarbons catalysed by metal salts of heteropolyacids. J. Chem. Soc. Chem. Commun. 1981, 9, 400–401. [Google Scholar]
- Toshihide, B.; Junya, S.; Yoshio, O. The Conversion of Methanol into Hydrocarbons over Metal Salts of Heteropolyacids. Bull. Chem. Soc. Jpn. 1982, 55, 2657–2658. [Google Scholar]
- Nakka, L.; Molinari, J.E.; Wachs, I.E. Surface and Bulk Aspects of Mixed Oxide Catalytic Nanoparticles: Oxidation and Dehydration of CH3OH by Polyoxometallates. J. Am. Chem. Soc. 2009, 131, 15544–15554. [Google Scholar] [CrossRef]
- Rocchiccioli-Deltcheff, C.; Fournier, M.; Franck, R.; Thouvenot, R. Vibrational investigations of polyoxometalates. 2. Evidence for anion-anion interactions in molybdenum (VI) and tungsten (VI) compounds related to the Keggin structure. Inorg. Chem. 1983, 22, 207–226. [Google Scholar] [CrossRef]
- Bielańnski, A.; Datka, J.; Gil, B.; Małecka-Lubańska, A.; Micek-Ilnicka, A. FTIR study of hydration of dodecatungstosilicic acid. Catal. Lett. 1999, 57, 61–64. [Google Scholar] [CrossRef]
- Marcì, G.; García-López, E.; Vaiano, V.; Sarno, G.; Sannino, D.; Palmisano, L. Keggin heteropolyacids supported on TiO2 used in gas-solid (photo)catalytic propene hydration and in liquid-solid photocatalytic glycerol dehydration. Catal. Today 2017, 281, 60–70. [Google Scholar] [CrossRef]
- Vita, A.; Italiano, G.; Ashraf, M.A.; Pino, L.; Specchia, S. Syngas production by steam and oxy-steam reforming of biogas on monolith-supported GeO2-based catalysts. Int. J. Hydrogen Energy 2018, 43, 11731–11744. [Google Scholar] [CrossRef]
- Borodina, E.; Meirer, F.; Lezcano-González, I.; Mokhtar, M.; Asiri, A.M.; Al-Thabaiti, S.A.; Basahel, S.N.; Ruiz-Martinez, J.; Wechhuysen, B.M. Influence of the Reaction Temperature on the Nature of the Active and Deactivating Species during Methanol to Olefins Conversion over H-SSZ-13. ACS Catal. 2015, 5, 992–1003. [Google Scholar] [CrossRef]
- Zhao, W.P.; Zhang, B.Z.; Wang, G.R.; Guo, H.C. Methane formation route in the conversion of methanol to hydrocarbons. J. Energy Chem. 2014, 23, 201–206. [Google Scholar] [CrossRef]
- Arena, F.; Di Chio, R.; Fazio, B.; Espro, C.; Spiccia, L.; Palella, A.; Spadaro, L. Probing the functionality of nanostructured MnCeOx catalysts in the carbon monoxide oxidation Part I. Influence of cerium addition on structure and CO oxidation activity. Appl. Catal. B-Environ. 2017, 210, 14–22. [Google Scholar] [CrossRef]
- Ortega, C.; Rezaei, M.; Hessel, V.; Kolb, G. Methanol to dimethyl ether conversion over a ZSM-5 catalyst: Intrinsic kinetic study on an external recycle reactor. Chem. Eng. J. 2018, 347, 741–753. [Google Scholar] [CrossRef]
- Weisz, P.B.; Prater, C.D. Interpretation of measurements in experimental catalysis. In Advances in Catalysis; Frankenburg, W.G., Komarewsky, V.I., Rideal, E.K., Eds.; Academic Press Inc.: Salt Lake City, UT, USA, 1954; Volume 6, pp. 143–196. [Google Scholar]
- Gupta, A.S.; Thodos, G. Mass and heat in the flow of fluids through fixed and fluidized beds of spherical particles. AIChE J. 1962, 8, 608–610. [Google Scholar] [CrossRef]
- Anderson, J.B. A criterion for isothermal behaviour of a catalyst pellet. Chem. Eng. Sci. 1963, 18, 147–148. [Google Scholar]
| Sample Name | W/M a | W/Mtheo | SBET b (m2/g) | Smicro c (m2/g) | Sext c (m2/g) | Vmicro c (cm3/g) | Vmeso d (cm3/g) | RMeso e % | RMacro e % | Average Pore Size (nm) | |
|---|---|---|---|---|---|---|---|---|---|---|---|
| CuPW | 6.75 | 8 | 3.05 | 0.56 | 2.49 | 2.34·10−4 | 1.03·10−2 | 62.5 | 37.5 | 13.48 | |
| CuSiW | 5.56 | 6 | 3.48 | 0.22 | 3.26 | 5.10·10−5 | 1.20·10−2 | 62.1 | 37.9 | 13.73 | |
| FePW | 9.44 | 12 | 3.41 | 0.91 | 2.50 | 3.69·10−4 | 6.42·10−3 | 76.0 | 24.0 | 7.28 | |
| FeSiW | 8.35 | 9 | 1.21 | 0.31 | 0.90 | 1.05·10−4 | 3.51·10−3 | 38.8 | 61.2 | 11.42 | |
| Sample Name | Weak Acid Sites (mmol/g) (100–250 °C) | Medium Acid Sites (mmol/g) (250–450 °C) | Strong Acid Sites (mmol/g) (Above 450 °C) | Total (mmol/g) |
|---|---|---|---|---|
| CuPW | 0.28 | 0.20 | 1.19 | 1.67 |
| CuSiW | 1.02 | 0.07 | 1.03 | 2.12 |
| FePW | 0.15 | 0.16 | 1.29 | 1.60 |
| FeSiW | 0.39 | 0.47 | 0.82 | 1.68 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yu, Y.; Sun, D.; Wang, S.; Xiao, M.; Sun, L.; Meng, Y. Heteropolyacid Salt Catalysts for Methanol Conversion to Hydrocarbons and Dimethyl Ether: Effect of Reaction Temperature. Catalysts 2019, 9, 320. https://doi.org/10.3390/catal9040320
Yu Y, Sun D, Wang S, Xiao M, Sun L, Meng Y. Heteropolyacid Salt Catalysts for Methanol Conversion to Hydrocarbons and Dimethyl Ether: Effect of Reaction Temperature. Catalysts. 2019; 9(4):320. https://doi.org/10.3390/catal9040320
Chicago/Turabian StyleYu, Yuehong, Daoming Sun, Shuanjin Wang, Min Xiao, Luyi Sun, and Yuezhong Meng. 2019. "Heteropolyacid Salt Catalysts for Methanol Conversion to Hydrocarbons and Dimethyl Ether: Effect of Reaction Temperature" Catalysts 9, no. 4: 320. https://doi.org/10.3390/catal9040320
APA StyleYu, Y., Sun, D., Wang, S., Xiao, M., Sun, L., & Meng, Y. (2019). Heteropolyacid Salt Catalysts for Methanol Conversion to Hydrocarbons and Dimethyl Ether: Effect of Reaction Temperature. Catalysts, 9(4), 320. https://doi.org/10.3390/catal9040320







