Electrospinning a Dye-Sensitized Solar Cell
Abstract
:1. Introduction
2. Results
3. Discussion
4. Materials and Methods
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Kleidon, A. Sonne statt Flaute. Phys. Unserer Zeit 2019, 50, 120–127. [Google Scholar] [CrossRef]
- Hashmi, G.A.M.; Syed, W.A.A.; Hayat, M.; Shah, W.H.; Shah, N.A. SrCl2 an environment friendly alternate to CdCl2 treatment for CdTe thin films solar cell application. Mater. Res. Express 2019, 6, 106440. [Google Scholar] [CrossRef]
- Liu, Y.H.; Chen, C.; Zhou, Y.; Kondrotas, R.; Tang, J. Butyldithiocarbamate acid solution processing: Its fundamentals and applications in chalcogenide thin film solar cells. J. Mater. Chem. C 2019, 7, 11068–11084. [Google Scholar] [CrossRef]
- Siddiqui, H. Lead-free perovskite quantum structures towards the efficient solar cell. Mater. Lett. 2019, 249, 99–103. [Google Scholar] [CrossRef]
- Hong, L.; Yao, H.F.; Wu, Z.; Cui, Y.; Zhang, T.; Xu, Y.; Yu, R.N.; Liao, Q.; Gao, B.W.; Xian, K.H.; et al. Eco-Compatible Solvent-Processed Organic Photovoltaic Cells with Over 16% Efficiency. Adv. Mater. 2019, 31, 1903441. [Google Scholar] [CrossRef]
- Freitag, M.; Teuscher, J.; Saygili, Y.; Zhang, X.Y.; Giordano, F.; Liska, P.; Hua, J.L.; Zakeeruddin, S.M.; Moser, J.-E.; Grätzel, M.; et al. Dye-sensitized solar cells for efficient power generation under ambient lighting. Nat. Photonics 2017, 11, 372–378. [Google Scholar] [CrossRef]
- Zhou, H.Z.; Wu, L.Q.; Gao, Y.R.; Ma, T.L. Dye-sensitized solar cells using 20 natural dyes as sensitizers. J. Photochem. Photobiol. A 2011, 219, 188–194. [Google Scholar] [CrossRef]
- Narayan, M.R. Review: Dye sensitized solar cells based on natural photosensitizers. Renew. Sustain. Energy Rev. 2012, 16, 208–215. [Google Scholar] [CrossRef]
- Calogero, G.; Marco, G.D.; Cazzanti, S.; Caramori, S.; Argazzi, R.; Carlo, A.D.; Bignozzi, C.A. Efficient dye-sensitized solar cells using red turnip and purple wild Sicilian prickly pear fruits. Int. J. Mol. Sci. 2010, 11, 254–267. [Google Scholar] [CrossRef]
- Kabir, F.; Sakib, S.N.; Uddin, S.S.; Efaz, E.T.; Himel, M.T.F. Enhance cell performance of DSSC by dye mixture, carbon nanotube and post TiCl4 treatment along with degradation study. Sustain. Energy Technol. Assess. 2019, 35, 298–307. [Google Scholar] [CrossRef]
- Alhamed, M.; Issa, A.S.; Doubal, A.W. Studying of natural dyes properties as photo-sensitizers for dye sensitized solar cells (DSSC). J. Electron Dev. 2012, 16, 1370–1383. [Google Scholar]
- Al-Bath’hi, S.A.M.; Alaei, I.; Sopyan, I. Natural photosensitizers for dye sensitized solar cells. Int. J. Renew. Energy Res. 2013, 3, 138–143. [Google Scholar]
- Juhász Junger, I.; Homburg, S.V.; Meissner, H.; Grethe, T.; Schwarz-Pfeiffer, A.; Fiedler, J.; Herrmann, A.; Blachowicz, T.; Ehrmann, A. Influence of the pH value of anthocyanins on the electrical properties of dye-sensitized solar cells. AIMS Energy 2017, 5, 258–267. [Google Scholar] [CrossRef]
- Lucioli, S.; Di Bari, C.; Forni, C.; Di Carlo, A.; Barrajón-Catalán, E.; Micol, V.; Nota, P.; Teoli, F.; Matteocci, F.; Frattarelli, A.; et al. Anthocyanic pigments from elicited in vitro grown shoot cultures of Vaccinium corymbosum L., cv. Brigitta Blue, as photosensitizer in natural dye-sensitized solar cells (NDSSC). J. Photochem. Photobiol. B 2018, 188, 69–76. [Google Scholar] [CrossRef]
- Li, Y.; Ku, S.-H.; Chen, S.-M.; Ali, M.A.; AlHemaid, F.M.A. Photoelectrochemistry for red cabbage extract as natural dye to develop a dye-sensitized solar cell. Int. J. Electrochem. Sci. 2013, 8, 1237–1245. [Google Scholar]
- Gokilamani, N.; Muthukumarasamy, N.; Thambidurai, M.; Ranjitha, A.; Velauthapillai, D. Utilization of natural anthocyanin pigments as photosensitizers for dye-sensitized solar cells. J. Sol-Gel Sci. Technol. 2013, 66, 212–219. [Google Scholar] [CrossRef]
- Wongcharee, K.; Meeyoo, V.; Chavadej, S. Dye-sensitized solar cell using natural dyes extracted from rosella and blue pea flowers. Sol. Energy Mater. Sol. Cells 2007, 91, 566–571. [Google Scholar] [CrossRef]
- Senthil, T.S.; Muthukumarasamy, N.; Velauthapillai, D.; Agilan, S.; Thambidurai, M.; Balasundaraprabhu, R. Natural dye (cyanidin 3-O-glucoside) sensitized nanocrystalline TiO2 solar cell fabricated using liquid electrolyte/quasi-solid-state polymer electrolyte. Renew. Energy 2011, 36, 2484–2488. [Google Scholar] [CrossRef]
- Hao, S.C.; Wu, J.H.; Huang, Y.F.; Lin, J.M. Natural dyes as photosensitizers for dye-sensitized solar cell. Sol. Energy 2006, 80, 209–214. [Google Scholar] [CrossRef]
- Ehrmann, A.; Blachowicz, T. Comment on ‘Dye-sensitized solar cells using Aloe Vera and Cladode of Cactus extracts as natural sensitizers’. Chem. Phys. Lett. 2019, 714, 227–229. [Google Scholar] [CrossRef]
- Juhász Junger, I.; Udomrungkhajornchai, S.; Grimmelsmann, N.; Blachowicz, T.; Ehrmann, A. Effect of Caffeine Copigmentation of Anthocyanin Dyes on DSSC Efficiency. Materials 2019, 12, 2692. [Google Scholar] [CrossRef] [PubMed]
- Ehrmann, A.; Blachowicz, T. Recent coating materials for textile-based solar cells. AIMS Mater. 2019, 6, 234–251. [Google Scholar] [CrossRef]
- Herrmann, A.; Fiedler, J.; Ehrmann, A.; Grethe, T.; Schwarz-Pfeiffer, A.; Blachowicz, T. Examination of the sintering process dependent micro- and nanostructure of TiO2 on textile substrates. Proc. SPIE 2016, 9898, 98980S. [Google Scholar]
- Opwis, K.; Gutmann, J.S.; Lagunas Alonso, A.R.; Rodriguez Henche, M.J.; Mayo, M.E.; Breuil, F.; Leonardi, E.; Sorbello, L. Preparation of a textile-based dye-sensitized solar cell. Int. J. Photoenergy 2016, 2016, 3796074. [Google Scholar] [CrossRef]
- Jun, M.J.; Cha, S.I.; Seo, S.H.; Kim, H.S.; Lee, D.Y. Float printing deposition to control the morphology of TiO2 photoanodes on woven textile metal substrates for TCO-free flexible dye-sensitized solar cells. RSC Adv. 2016, 6, 67331–67339. [Google Scholar]
- Pu, X.; Song, W.X.; Liu, M.M.; Sun, C.W.; Du, C.H.; Jiang, C.Y.; Huang, X.; Zou, D.; Hu, W.G.; Wang, Z.L. Wearable power-textiles by integrating fabric triboelectric nanogenerators and fiber-shaped dye-sensitized solar cells. Adv. Energy Mater. 2016, 6, 1601048. [Google Scholar] [CrossRef]
- Chai, Z.S.; Zhang, N.N.; Sun, P.; Huang, Y.; Zhao, C.X.; Fan, H.J.; Fan, X.; Mai, W.J. Tailorable and wearable textile devices for solar energy harvesting and simultaneous storage. ACS Nano 2016, 10, 9201–9207. [Google Scholar] [CrossRef]
- Chen, J.; Huang, Y.; Zhang, N.N.; Zou, H.Y.; Liu, R.Y.; Tao, C.Y.; Fan, X.; Wang, Z.L. Micro-cable structured textile for simultaneously harvesting solar and mechanical energy. Nat. Energy 2016, 1, 16138. [Google Scholar] [CrossRef]
- Kohn, S.; Großerhode, C.; Storck, J.L.; Grötsch, G.; Cornelißen, C.; Streitenberger, A.; Grassmann, C.; Schwarz-Pfeiffer, A.; Ehrmann, A. Commercially available teas as possible dyes for dye-sensitized solar cells. Optik 2019, 185, 178–182. [Google Scholar] [CrossRef]
- Mamun, A.; Trabelsi, M.; Klöcker, M.; Sabantina, L.; Großerhode, C.; Blachowicz, T.; Grötsch, G.; Cornelißen, C.; Streitenberger, A.; Ehrmann, A. Electrospun Nanofiber Mats with Embedded Non-Sintered TiO2 for Dye-Sensitized Solar Cells (DSSCs). Fibers 2019, 7, 60. [Google Scholar] [CrossRef]
- Juhász Junger, I.; Wehlage, D.; Böttjer, R.; Grothe, T.; Juhász, L.; Grassmann, C.; Blachowicz, T.; Ehrmann, A. Dye-Sensitized Solar Cells with Electrospun Nanofiber Mat-Based Counter Electrodes. Materials 2018, 11, 1604. [Google Scholar] [CrossRef] [PubMed]
- Kerker, E.; Steinhäußer, D.; Mamun, A.; Trabelsi, M.; Fiedler, J.; Sabantina, L.; Juhász Junger, I.; Schiek, M.; Ehrmann, A.; Kaschuba, R. Spectroscopic Investigation of Highly-Scattering Nanofiber Mats during Drying and Film Formation. Optik 2020. submitted. [Google Scholar]
- Sabantina, L.; Hes, L.; Rodríguez-Mirasol, J.; Cordero, T.; Ehrmann, A. Water Vapor Permeability through PAN Nanofiber Mat with Varying Membrane-Like Areas. Fibres Text. East. Eur. 2019, 27, 12–15. [Google Scholar] [CrossRef]

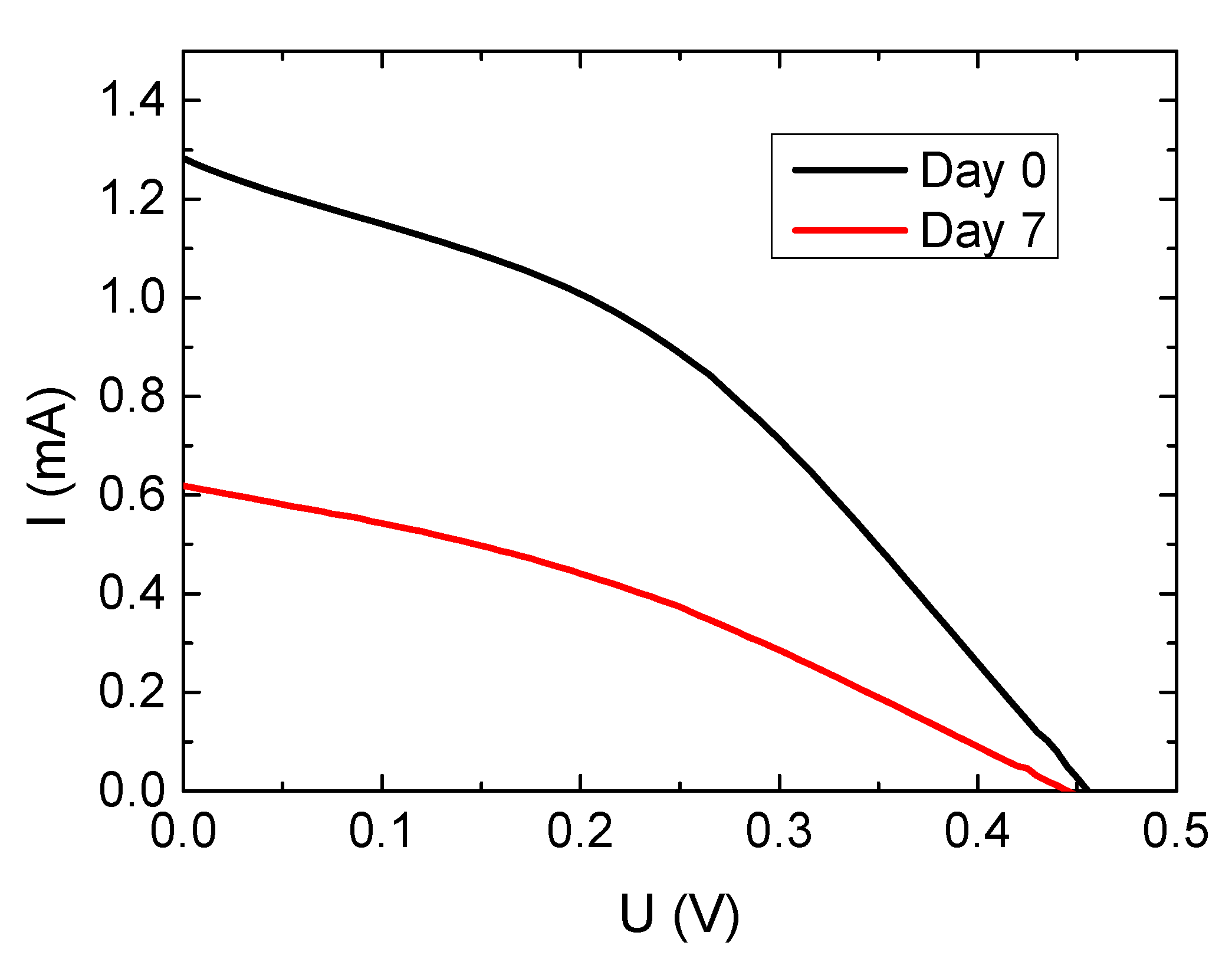
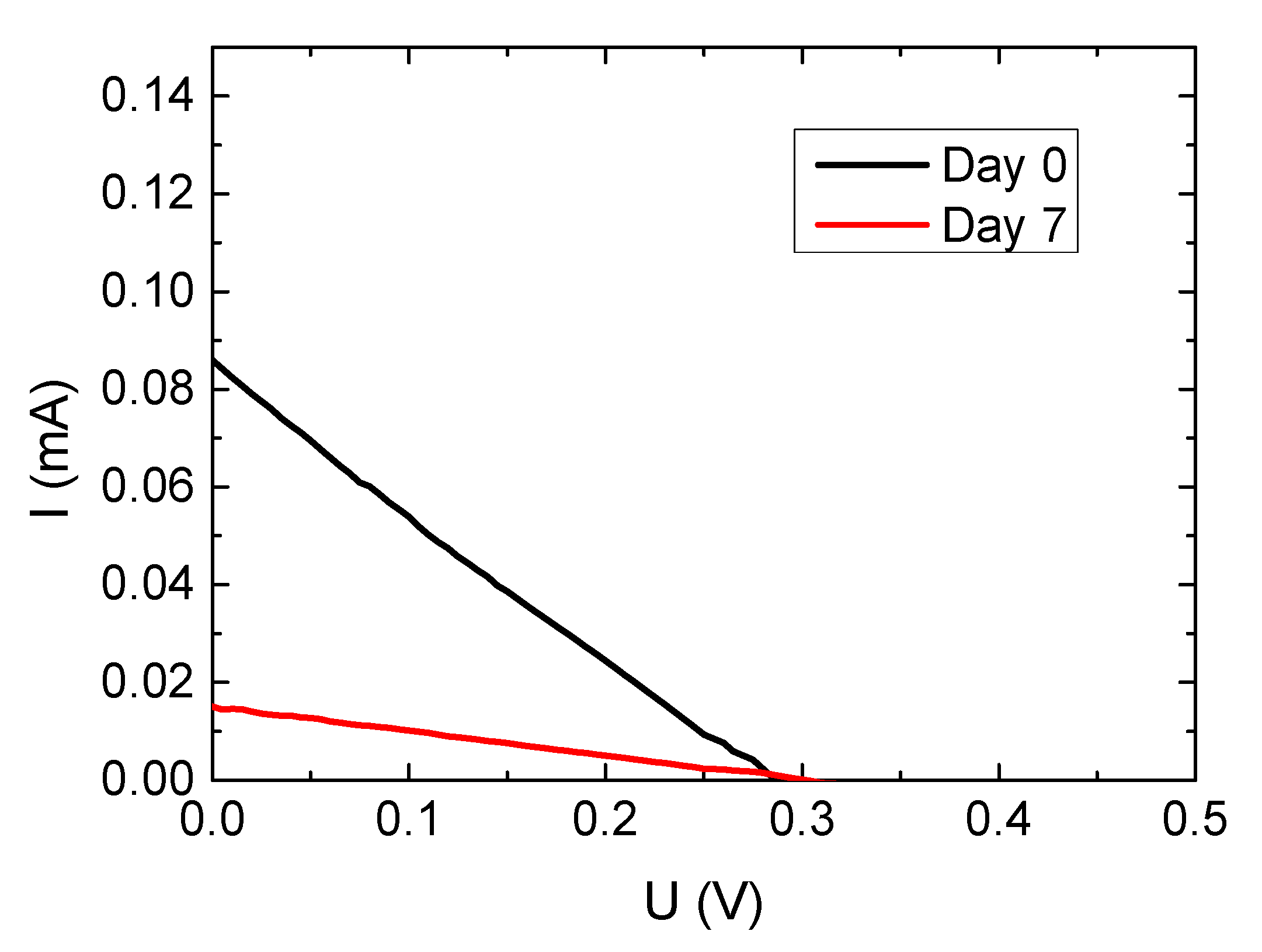
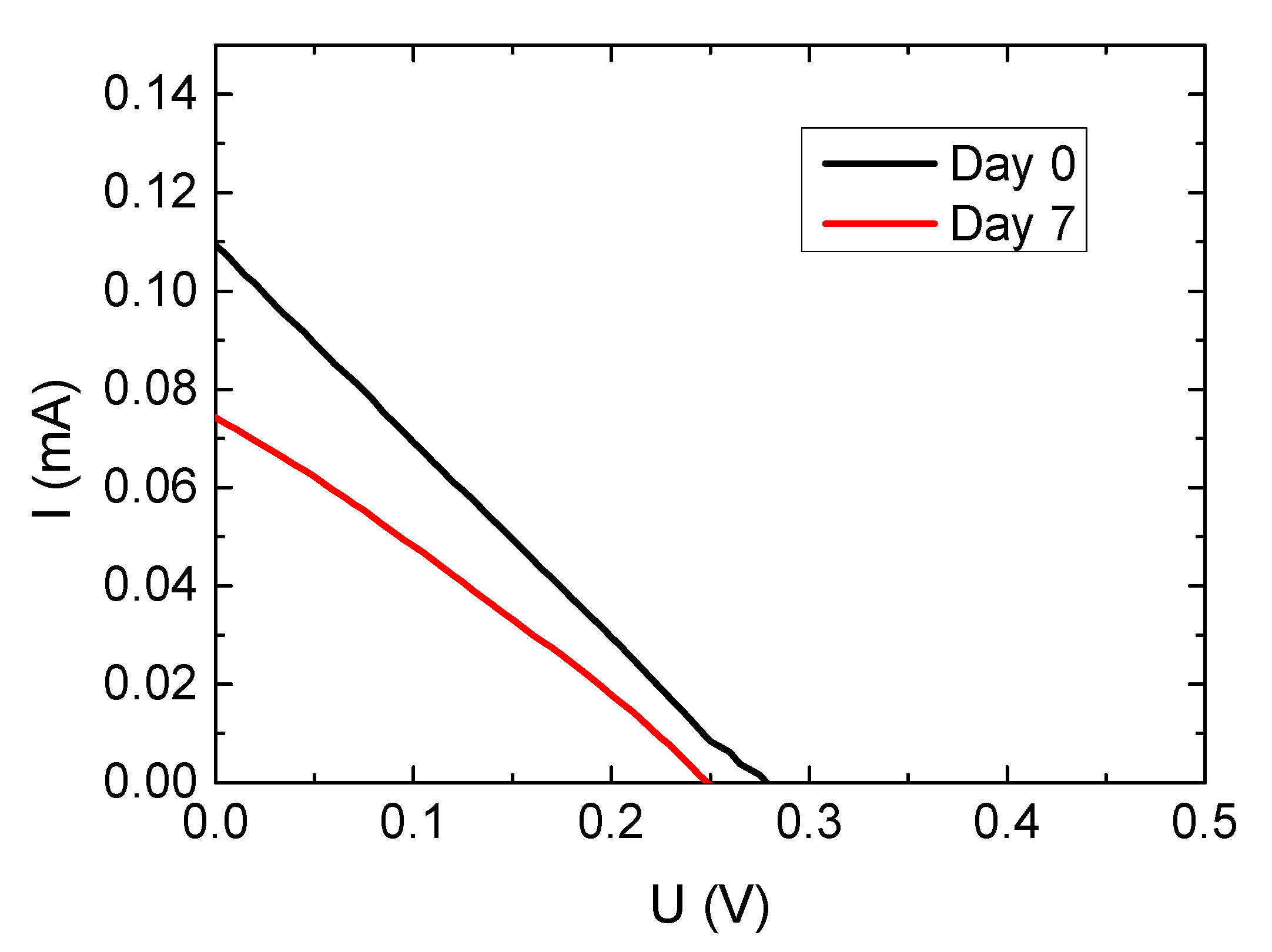
| Sample | JSC/mA | UOC/V | FF | Efficiency/% |
|---|---|---|---|---|
| Ref. glass–day 0 | 0.214 | 0.455 | 0.383 | 0.037 |
| Ref. glass–day 7 | 0.103 | 0.445 | 0.339 | 0.016 |
| Glass + nanofiber mat–day 0 | 0.015 | 0.290 | 0.246 | 0.0011 |
| Glass + nanofiber mat–day 7 | 0.003 | 0.305 | 0.265 | 0.0002 |
| Full nanofiber DSSC–day 0 | 0.019 | 0.280 | 0.259 | 0.0014 |
| Full nanofiber DSSC–day 7 | 0.013 | 0.260 | 0.283 | 0.0010 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kohn, S.; Wehlage, D.; Juhász Junger, I.; Ehrmann, A. Electrospinning a Dye-Sensitized Solar Cell. Catalysts 2019, 9, 975. https://doi.org/10.3390/catal9120975
Kohn S, Wehlage D, Juhász Junger I, Ehrmann A. Electrospinning a Dye-Sensitized Solar Cell. Catalysts. 2019; 9(12):975. https://doi.org/10.3390/catal9120975
Chicago/Turabian StyleKohn, Sophia, Daria Wehlage, Irén Juhász Junger, and Andrea Ehrmann. 2019. "Electrospinning a Dye-Sensitized Solar Cell" Catalysts 9, no. 12: 975. https://doi.org/10.3390/catal9120975
APA StyleKohn, S., Wehlage, D., Juhász Junger, I., & Ehrmann, A. (2019). Electrospinning a Dye-Sensitized Solar Cell. Catalysts, 9(12), 975. https://doi.org/10.3390/catal9120975






