Ag- and Cu-Promoted Mesoporous Ta-SiO2 Catalysts Prepared by Non-Hydrolytic Sol-Gel for the Conversion of Ethanol to Butadiene
Abstract
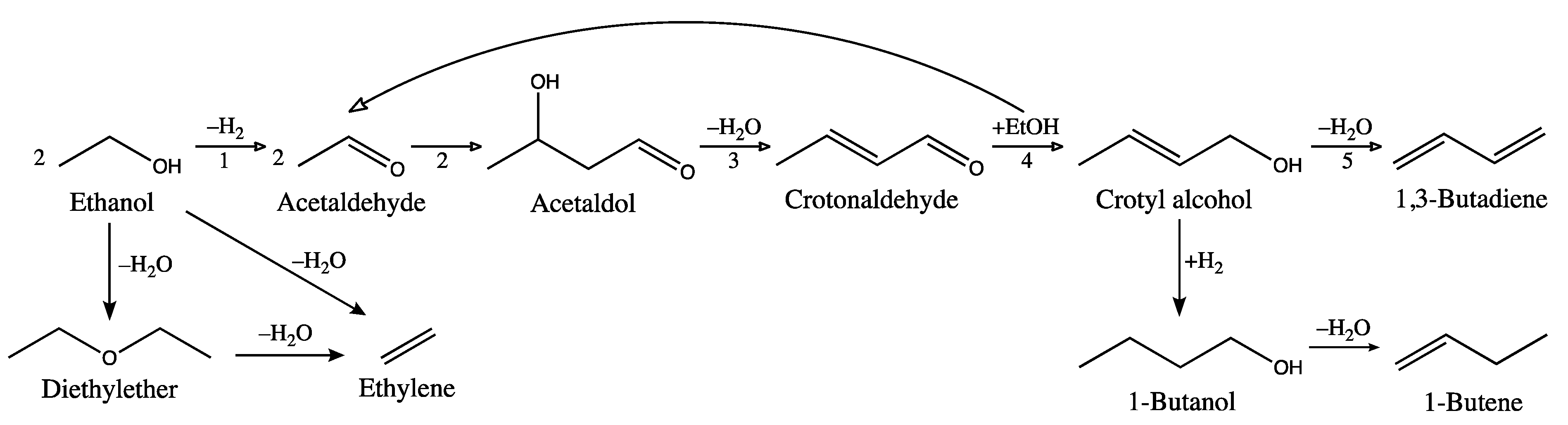
1. Introduction
2. Results
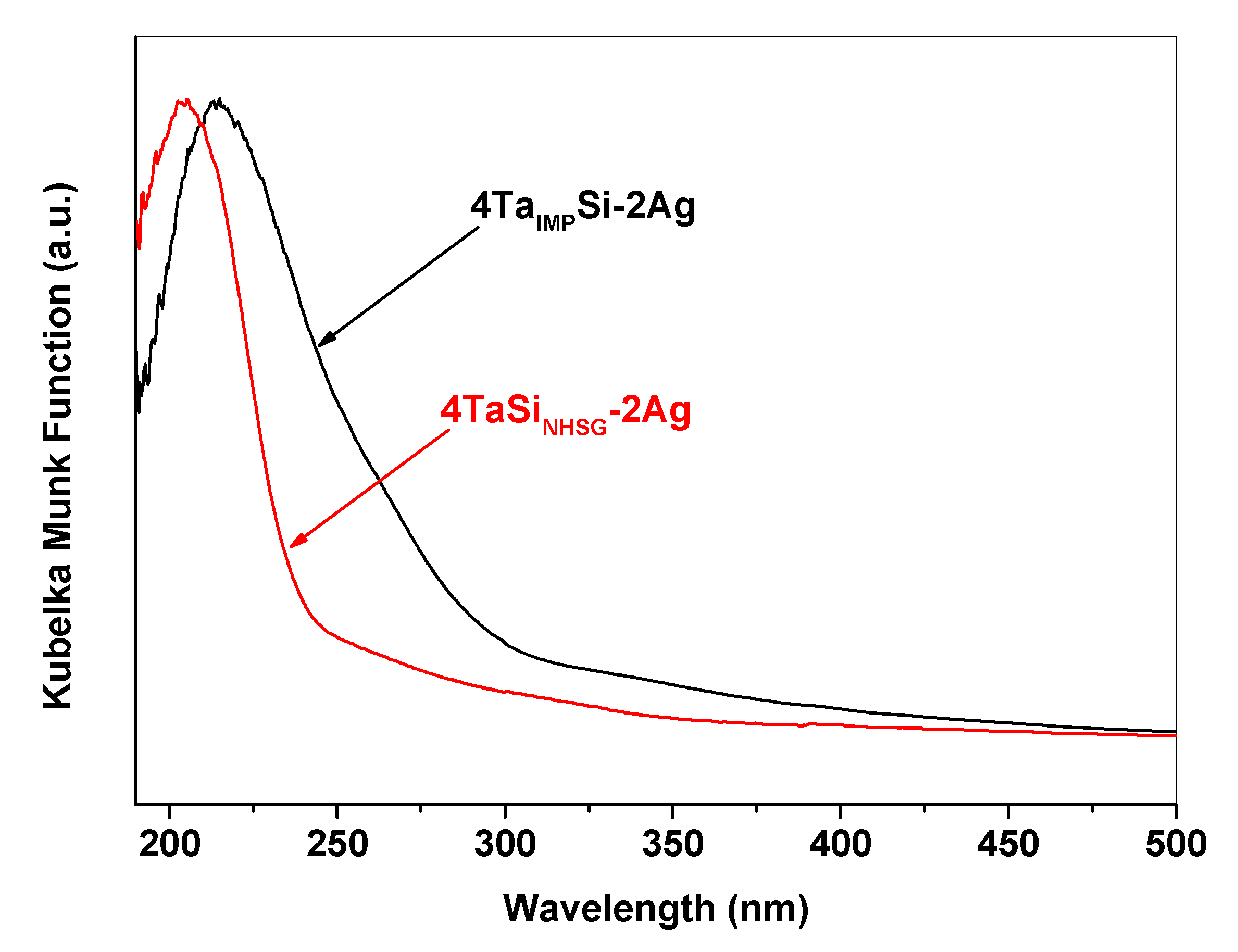
2.1. Incorporation of Ta in the Silica Matrix and Impact on Catalysis
2.2. Influence of Tantalum and Silver Loading on Butadiene Yield
2.3. One-pot NHSG Preparation of Ag-TaSiO2 Bifunctional Catalyst
2.4. Copper as an Effective Alternative to Silver
3. Conclusions
4. Experimental
4.1. Catalyst Preparation
4.2. Characterization
4.3. Ethanol to Butadiene Reaction
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Cespi, D.; Passarini, F.; Vassura, I.; Cavani, F. Butadiene from biomass, a life cycle perspective to address sustainability in the chemical industry. Green Chem. 2016, 18, 1625–1638. [Google Scholar] [CrossRef]
- Shylesh, S.; Gokhale, A.A.; Scown, C.D.; Kim, D.; Ho, C.R.; Bell, A.T. From Sugars to Wheels: The Conversion of Ethanol to 1,3-Butadiene over Metal-Promoted Magnesia-Silicate Catalysts. ChemSusChem 2016, 9, 1462–1472. [Google Scholar] [CrossRef] [PubMed]
- Pomalaza, G.; Capron, M.; Ordomsky, V.; Dumeignil, F.; Pomalaza, G.; Capron, M.; Ordomsky, V.; Dumeignil, F. Recent Breakthroughs in the Conversion of Ethanol to Butadiene. Catalysts 2016, 6, 203. [Google Scholar] [CrossRef]
- White, W.C. Butadiene production process overview. Chem. Biol. Interact. 2007, 166, 10–14. [Google Scholar] [CrossRef] [PubMed]
- Ren, T.; Patel, M.; Blok, K. Olefins from conventional and heavy feedstocks: Energy use in steam cracking and alternative processes. Energy 2006, 31, 425–451. [Google Scholar] [CrossRef]
- Chaudhary, L.; Pradhan, P.; Soni, N.; Singh, P.; Tiwari, A. Algae as a feedstock for bioethanol production: New entrance in biofuel world. Int. J. ChemTech Res. 2014, 6, 1381–1389. [Google Scholar]
- Muktham, R.; Bhargava, S.K.; Bankupalli, S.; Ball, A.S. A Review on 1st and 2nd Generation Bioethanol Production-Recent Progress. J. Sustain. Bioenergy Syst. 2016, 6, 72–92. [Google Scholar] [CrossRef]
- Gérardy, R.; Morodo, R.; Estager, J.; Luis, P.; Debecker, D.P.; Monbaliu, J.C.M. Sustaining the Transition from a petrobased to a Biobased Chemical Industry with Flow Chemistry. Top. Curr. Chem. 2019, 377, 1. [Google Scholar] [CrossRef]
- Gallezot, P. Conversion of biomass to selected chemical products. Chem. Soc. Rev. 2012, 41, 1538–1558. [Google Scholar] [CrossRef]
- Dumeignil, F.; Capron, M.; Katryniok, B.; Wojcieszak, R.; Löfberg, A.; Girardon, J.S.; Desset, S.; Araque-Marin, M.; Jalowiecki-Duhamel, L.; Paul, S. Biomass-derived platform molecules upgrading through catalytic processes: Yielding chemicals and fuels. J. Jpn. Pet. Inst. 2015, 58, 257–273. [Google Scholar] [CrossRef]
- Kim, T.W.; Kim, J.W.; Kim, S.Y.; Chae, H.J.; Kim, J.R.; Jeong, S.Y.; Kim, C.U. Butadiene production from bioethanol and acetaldehyde over tantalum oxide-supported spherical silica catalysts for circulating fluidized bed. Chem. Eng. J. 2015, 278, 217–223. [Google Scholar] [CrossRef]
- Sun, J.; Wang, Y. Recent Advances in Catalytic Conversion of Ethanol to Chemicals. ACS Catal. 2014, 4, 1078–1090. [Google Scholar] [CrossRef]
- Makshina, E.V.; Dusselier, M.; Janssens, W.; Degrève, J.; Jacobs, P.A.; Sels, B.F. Review of old chemistry and new catalytic advances in the on-purpose synthesis of butadiene. Chem. Soc. Rev. 2014, 43, 7917–7953. [Google Scholar] [CrossRef] [PubMed]
- Talalay, A.; Talalay, L.S.K. The Russian Synthetic Rubber from Alcohol. A Survey of the Chemistry and Technology of the Lebedev Process for Producing Sodium-Butadiene Polymers. Rubber Chem. Technol. 1942, 15, 403–429. [Google Scholar] [CrossRef]
- Corson, B.B.; Jones, H.E.; Welling, C.E.; Hinckley, J.A.; Stahly, E.E. Butadiene from Ethyl Alcohol. Catalysis in the One-and Two-Stop Processes. Ind. Eng. Chem. 1950, 42, 359–373. [Google Scholar] [CrossRef]
- Kyriienko, P.I.; Larina, O.V.; Soloviev, S.O.; Orlyk, S.M.; Calers, C.; Dzwigaj, S. Ethanol Conversion into 1,3-Butadiene by the Lebedev Method over MTaSiBEA Zeolites (M = Ag, Cu, Zn). ACS Sustain. Chem. Eng. 2017, 5, 2075–2083. [Google Scholar] [CrossRef]
- Angelici, C.; Weckhuysen, B.M.; Bruijnincx, P.C.A. Chemocatalytic Conversion of Ethanol into Butadiene and Other Bulk Chemicals. ChemSusChem 2013, 6, 1595–1614. [Google Scholar] [CrossRef]
- Müller, P.; Burt, S.P.; Love, A.M.; McDermott, W.P.; Wolf, P.; Hermans, I. Mechanistic Study on the Lewis Acid Catalyzed Synthesis of 1,3-Butadiene over Ta-BEA Using Modulated Operando DRIFTS-MS. ACS Catal. 2016, 6, 6823–6832. [Google Scholar] [CrossRef]
- Sushkevich, V.L.; Ivanova, I.I.; Ordomsky, V.V.; Taarning, E. Design of a Metal-Promoted Oxide Catalyst for the Selective Synthesis of Butadiene from Ethanol. ChemSusChem 2014, 7, 2527–2536. [Google Scholar] [CrossRef]
- Sushkevich, V.L.; Ivanova, I.I.; Taarning, E. Mechanistic Study of Ethanol Dehydrogenation over Silica-Supported Silver. ChemCatChem 2013, 5, 2367–2373. [Google Scholar] [CrossRef]
- Sushkevich, V.L.; Ivanova, I.I.; Taarning, E. Ethanol conversion into butadiene over Zr-containing molecular sieves doped with silver. Green Chem. 2015, 17, 2552–2559. [Google Scholar] [CrossRef]
- Kyriienko, P.I.; Larina, O.V.; Soloviev, S.O.; Orlyk, S.M.; Dzwigaj, S. High selectivity of TaSiBEA zeolite catalysts in 1,3-butadiene production from ethanol and acetaldehyde mixture. Catal. Commun. 2016, 77, 123–126. [Google Scholar] [CrossRef]
- Debecker, D.P. Innovative Sol-Gel Routes for the Bottom-Up Preparation of Heterogeneous Catalysts. Chem. Rec. 2018, 18, 662–675. [Google Scholar] [CrossRef] [PubMed]
- Livage, J. Sol–gel synthesis of heterogeneous catalysts from aqueous solutions. Catal. Today 1998, 41, 3–19. [Google Scholar] [CrossRef]
- Debecker, D.P.; Mutin, P.H. Non-hydrolytic sol-gel routes to heterogeneous catalysts. Chem. Soc. Rev. 2012, 41, 3624–3650. [Google Scholar] [CrossRef] [PubMed]
- Skoda, D.; Styskalik, A.; Moravec, Z.; Bezdicka, P.; Babiak, M.; Klementova, M.; Barnes, C.E.; Pinkas, J. Novel non-hydrolytic templated sol–gel synthesis of mesoporous aluminosilicates and their use as aminolysis catalysts. RSC Adv. 2016, 6, 24273–24284. [Google Scholar] [CrossRef]
- Skoda, D.; Styskalik, A.; Moravec, Z.; Bezdicka, P.; Pinkas, J. Templated non-hydrolytic synthesis of mesoporous zirconium silicates and their catalytic properties. J. Mater. Sci. 2015, 50, 3371–3382. [Google Scholar] [CrossRef]
- Styskalik, A.; Skoda, D.; Pinkas, J.; Mathur, S. Non-hydrolytic synthesis of titanosilicate xerogels by acetamide elimination and their use as epoxidation catalysts. J. Sol-Gel Sci. Technol. 2012, 63, 463–472. [Google Scholar] [CrossRef]
- Debecker, D.P.; Hulea, V.; Mutin, P.H. Mesoporous mixed oxide catalysts via non-hydrolytic sol–gel: A review. Appl. Catal. A 2013, 451, 192–206. [Google Scholar] [CrossRef]
- Styskalik, A.; Skoda, D.; Barnes, C.; Pinkas, J. The Power of Non-Hydrolytic Sol-Gel Chemistry: A Review. Catalysts 2017, 7, 168. [Google Scholar] [CrossRef]
- Bouchmella, K.; Stoyanova, M.; Rodemerck, U.; Debecker, D.P.; Hubert Mutin, P. Avoiding rhenium loss in non-hydrolytic synthesis of highly active Re–Si–Al olefin metathesis catalysts. Catal. Commun. 2015, 58, 183–186. [Google Scholar] [CrossRef]
- Maksasithorn, S.; Praserthdam, P.; Suriye, K.; Devillers, M.; Debecker, D.P. WO3-based catalysts prepared by non-hydrolytic sol-gel for the production of propene by cross-metathesis of ethene and 2-butene. Appl. Catal. A 2014, 488, 200–207. [Google Scholar] [CrossRef]
- Smeets, V.; Ben Mustapha, L.; Schnee, J.; Gaigneaux, E.M.; Debecker, D.P. Mesoporous SiO2-TiO2 epoxidation catalysts: Tuning surface polarity to improve performance in the presence of water. Mol. Catal. 2018, 452, 123–128. [Google Scholar] [CrossRef]
- Lafond, V.; Mutin, P.; Vioux, A. Non-hydrolytic sol–gel routes based on alkyl halide elimination: Toward better mixed oxide catalysts and new supports: Application to the preparation of a SiO2-TiO2 epoxidation catalyst. J. Mol. Catal. A 2002, 182, 81–88. [Google Scholar] [CrossRef]
- Cojocariu, A.M.; Mutin, P.H.; Dumitriu, E.; Fajula, F.; Vioux, A.; Hulea, V. Non-hydrolytic synthesis of mesoporous silica–titania catalysts for the mild oxidation of sulfur compounds with hydrogen peroxide. Chem. Commun. 2008, 42, 5357–5359. [Google Scholar] [CrossRef]
- Styskalik, A.; Abbott, J.G.; Orick, M.C.; Debecker, D.P.; Barnes, C.E. Synthesis, characterization and catalytic activity of single site, Lewis acidic aluminosilicates. Catal. Today 2019, 334, 131–139. [Google Scholar] [CrossRef]
- Hamden, Z.; Bouattour, S.; Ferraria, A.; Ferreira, D.; Ferreira, L.V.; Do Rego, A.B.; Boufi, S. In situ generation of TiO2 nanoparticles using chitosan as a template and their photocatalytic activity. J. Photochem. Photobiol. A 2016, 321, 211–222. [Google Scholar] [CrossRef]
- Zatsepin, A.; Zatsepin, D.; Boukhvalov, D.; Kuznetsova, Y.A.; Gavrilov, N.; Shur, V.Y.; Esin, A. Local atomic configurations, energy structure, and optical properties of implantation defects in Gd-doped silica glass: An XPS, PL, and DFT study. J. Alloys Compd. 2019, 796, 77–85. [Google Scholar] [CrossRef]
- Jacquemin, M.; Genet, M.J.; Gaigneaux, E.M.; Debecker, D.P. Calibration of the X-ray Photoelectron Spectroscopy Binding Energy Scale for the Characterization of Heterogeneous Catalysts: Is Everything Really under Control? ChemPhysChem 2013, 14, 3618–3626. [Google Scholar] [CrossRef]
- Simpson, R.; White, R.G.; Watts, J.F.; Baker, M.A. XPS investigation of monatomic and cluster argon ion sputtering of tantalum pentoxide. Appl. Surf. Sci. 2017, 405, 79–87. [Google Scholar] [CrossRef]
- Arnould, C.; Volcke, C.; Lamarque, C.; Thiry, P.A.; Delhalle, J.; Mekhalif, Z. Titanium modified with layer-by-layer sol–gel tantalum oxide and an organodiphosphonic acid: A coating for hydroxyapatite growth. J. Colloid Interface Sci. 2009, 336, 497–503. [Google Scholar] [CrossRef]
- Greczynski, G.; Hultman, L. X-ray photoelectron spectroscopy: Towards reliable binding energy referencing. Prog. Mater. Sci. 2019. [Google Scholar] [CrossRef]
- Hasegawa, Y.; Ayame, A. Investigation of oxidation states of titanium in titanium silicalite-1 by X-ray photoelectron spectroscopy. Catal. Today 2001, 71, 177–187. [Google Scholar] [CrossRef]
- Debecker, D.P.; Bouchmella, K.; Delaigle, R.; Eloy, P.; Poleunis, C.; Bertrand, P.; Gaigneaux, E.M.; Mutin, P.H. One-step non-hydrolytic sol–gel preparation of efficient V2O5-TiO2 catalysts for VOC total oxidation. Appl. Catal. B 2010, 94, 38–45. [Google Scholar] [CrossRef]
- Blanc, D.; Zhang, W.; Massard, C.; Mugnier, J. Synthesis and characterisation of tantalum-incorporating silica hybrid sol–gel thin films for optical applications. Opt. Mater. 2006, 28, 331–335. [Google Scholar] [CrossRef]
- Ko, Y.S.; Ahn, W.S. Synthesis and characterization of tantalum silicalite molecular sieves with MFI structure. Microporous Mesoporous Mater. 1999, 30, 283–291. [Google Scholar] [CrossRef]
- Ruddy, D.A.; Tilley, T.D. Kinetics and mechanism of olefin epoxidation with aqueous H2O2 and a highly selective surface-modified TaSBA15 heterogeneous catalyst. J. Am. Chem. Soc. 2008, 130, 11088–11096. [Google Scholar] [CrossRef] [PubMed]
- Dzwigaj, S.; Millot, Y.; Che, M. Ta (V)-single site BEA zeolite by two-step postsynthesis method: Preparation and characterization. Catal. Lett. 2010, 135, 169–174. [Google Scholar] [CrossRef]
- Styskalik, A.; Kordoghli, I.; Poleunis, C.; Delcorte, A.; Aprile, C.; Fusaro, L.; Debecker, D.P. Highly Porous Hybrid Metallosilicate Materials Prepared by Non-Hydrolytic Sol-Gel: Hydrothermal Stability and Catalytic Properties in Ethanol Dehydration. ChemRxiv 2019. [Google Scholar] [CrossRef]
- Debecker, D.P.; Boissiere, C.; Laurent, G.; Huet, S.; Eliaers, P.; Sanchez, C.; Backov, R. First acidic macro-mesocellular aluminosilicate monolithic foams “SiAl(HIPE)” and their catalytic properties. Chem. Commun. 2015, 51, 14018–14021. [Google Scholar] [CrossRef]
- Phung, T.K.; Busca, G. Ethanol dehydration on silica-aluminas: Active sites and ethylene/diethyl ether selectivities. Catal. Commun. 2015, 68, 110–115. [Google Scholar] [CrossRef]
- Balthis, J.; Rochow, E.; White, D. Silicon Tetraacetate. Inorg. Synth. 1953, 4, 45–47. [Google Scholar]
- Bradley, D.C.; Thomas, I.M. Metallo-Organic Compounds Containing Metal–Nitrogen Bonds: Part III. Dialkylamino Compounds of Tantalum. Can. J. Chem. 1962, 40, 1355–1360. [Google Scholar] [CrossRef]
- Wakuda, D.; Hatamura, M.; Suganuma, K. Novel method for room temperature sintering of Ag nanoparticle paste in air. Chem. Phys. Lett. 2007, 441, 305–308. [Google Scholar] [CrossRef]
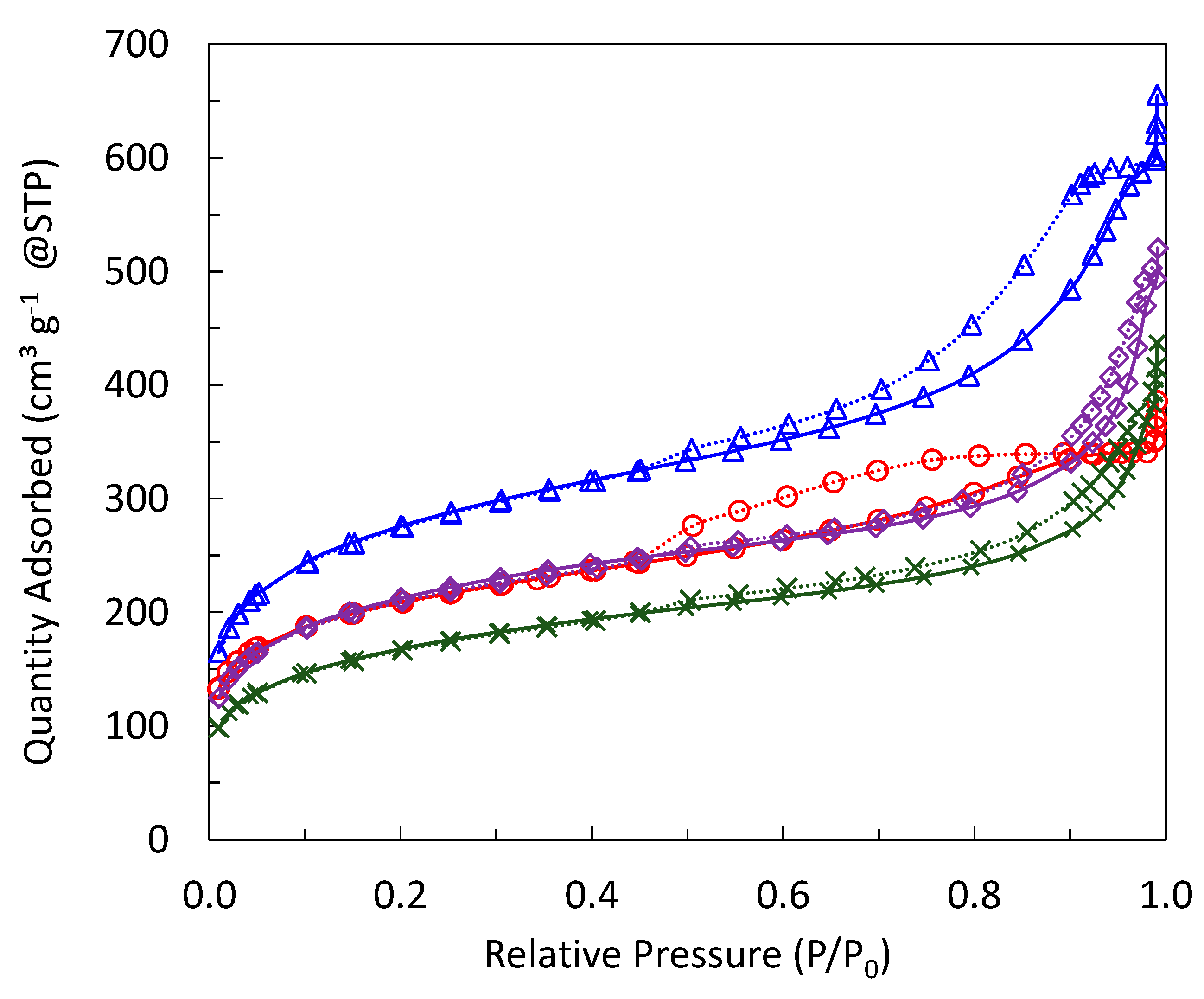
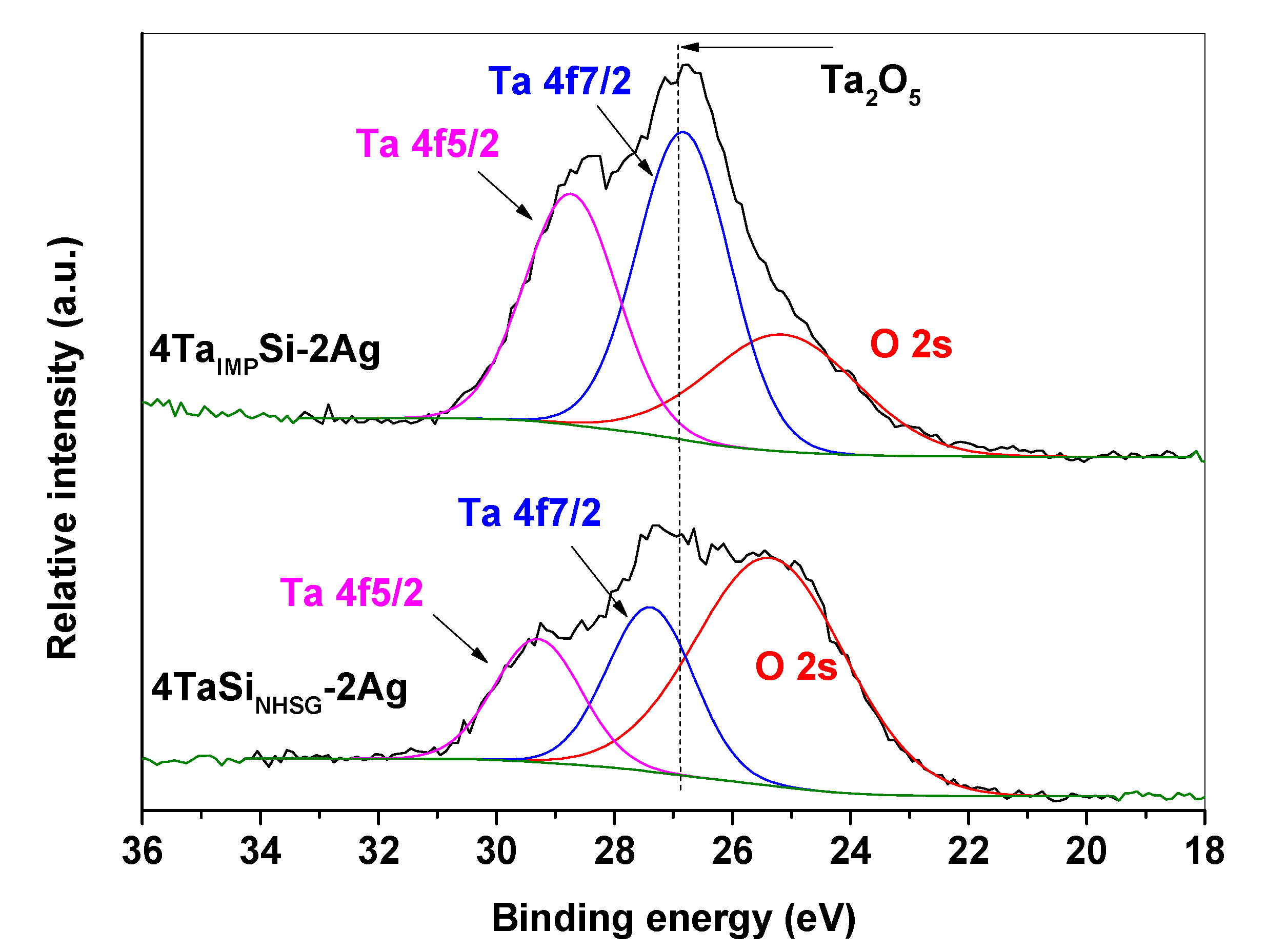
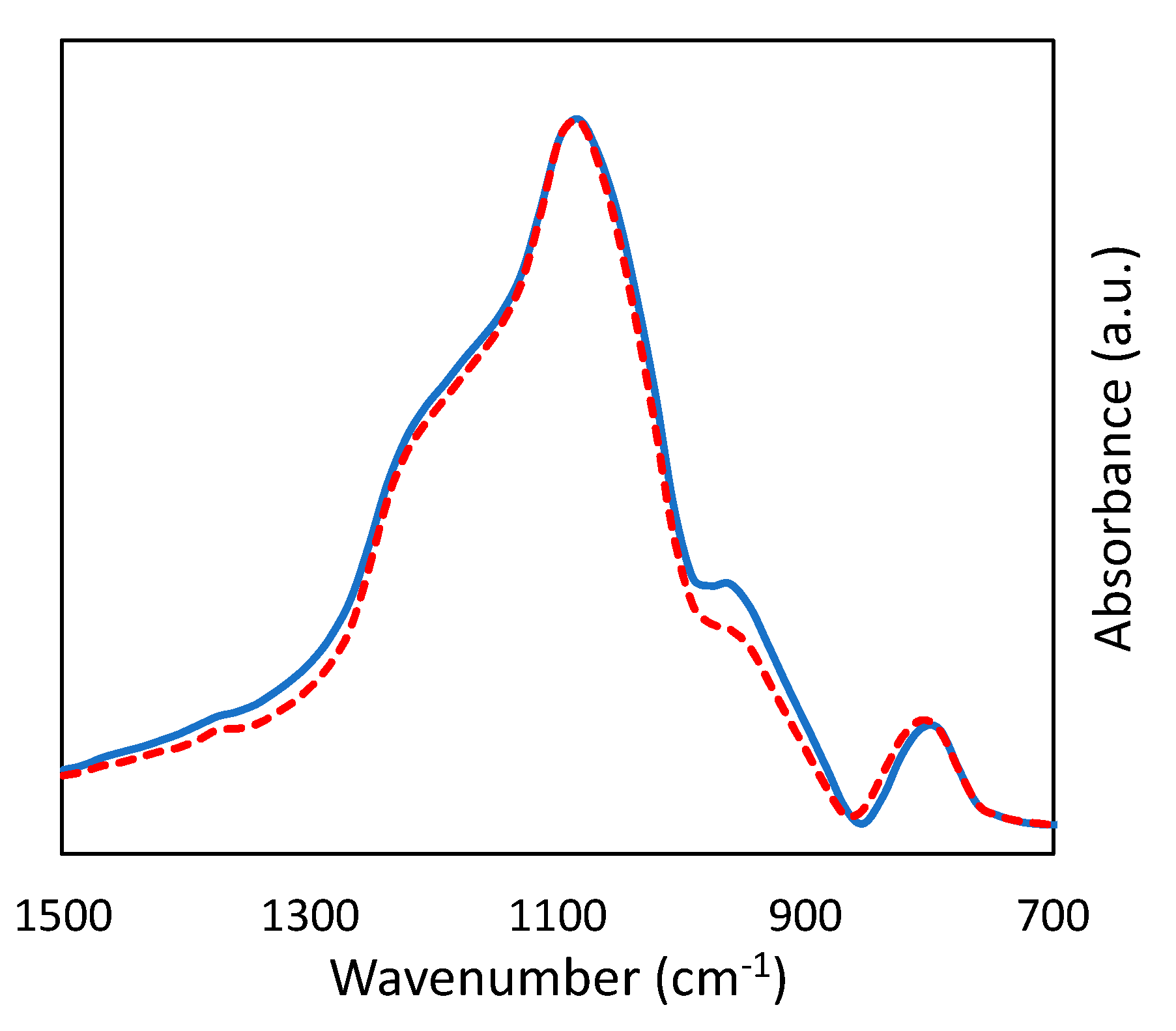


| Sample | SBET (m2 g−1) | Vp (mL g−1) a | Vµ (mL g−1) b | Dp (nm) c | % Ta (w/w) d | % Ag (w/w) d | Surface Si/Ta e | Acid Sites (mmol g−1) f |
|---|---|---|---|---|---|---|---|---|
| 4TaSiNHSGg | 990 | 0.96 | 0.17 | 3.9 | 4.3 | - | 65 | n.m. |
| 4TaSiNHSG-2Ag | 690 | 0.65 | 0.11 | 3.8 | 4.8 | 1.7 | 61 | 3.7 10−2 |
| SiNHSG | 760 | 0.76 | 0.13 | 4.0 | - | - | - | n.m. |
| 4TaIMPSi-2Ag | 600 | 0.59 | 0.09 | 4.0 | 4.1 | 1.6 | 34 | 1.5 10−2 |
| Catalyst | Ethanol Conversion (%) | Butadiene (%) | Acetaldehyde (%) | Ethylene (%) | Diethylether (%) | Others (%) | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Yield | Sel | Yield | Sel | Yield | Sel | Yield | Sel | Yield | Sel | ||
| 4TaIMPSi-2Ag | 53.7 | 12.1 | 22.5 | 13.3 | 24.8 | 7.7 | 14.3 | 10.3 | 19.2 | 10.3 | 19.2 |
| 4TaSiNHSG-2Ag | 78.1 | 10.4 | 13.3 | 17.0 | 21.8 | 28.9 | 37.0 | 8.1 | 10.4 | 13.8 | 17.7 |
| 4TaSiNHSG-5Ag | 90.4 | 15.4 | 17.0 | 17.1 | 18.9 | 31.5 | 34.8 | 5.3 | 5.9 | 21.1 | 23.3 |
| 2TaSiNHSG-2Ag | 64.8 | 4.6 | 7.1 | 17.3 | 26.7 | 23.6 | 36.4 | 7.2 | 11.1 | 12.1 | 18.7 |
| 2TaSiNHSG-5Ag | 84.5 | 16.6 | 19.6 | 28.5 | 33.8 | 13.0 | 15.4 | 3.5 | 4.1 | 22.9 | 27.1 |
| 2TaIMPSi-5Ag | 64.5 | 14.6 | 22.6 | 34.5 | 53.5 | 6.1 | 9.4 | 3.5 | 5.4 | 5.9 | 9.1 |
| 2TaSi5AgNHSG | 65.2 | 8.9 | 13.7 | 19.9 | 30.5 | 22.2 | 34.0 | 4.9 | 7.5 | 9.5 | 14.6 |
| 2TaSi5CuNHSG | 75.3 | 23.3 | 30.9 | 29.8 | 39.6 | 1.4 | 1.9 | 0.8 | 1.1 | 20.0 | 26.6 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dochain, D.D.; Stýskalík, A.; Debecker, D.P. Ag- and Cu-Promoted Mesoporous Ta-SiO2 Catalysts Prepared by Non-Hydrolytic Sol-Gel for the Conversion of Ethanol to Butadiene. Catalysts 2019, 9, 920. https://doi.org/10.3390/catal9110920
Dochain DD, Stýskalík A, Debecker DP. Ag- and Cu-Promoted Mesoporous Ta-SiO2 Catalysts Prepared by Non-Hydrolytic Sol-Gel for the Conversion of Ethanol to Butadiene. Catalysts. 2019; 9(11):920. https://doi.org/10.3390/catal9110920
Chicago/Turabian StyleDochain, Denis D., Aleš Stýskalík, and Damien P. Debecker. 2019. "Ag- and Cu-Promoted Mesoporous Ta-SiO2 Catalysts Prepared by Non-Hydrolytic Sol-Gel for the Conversion of Ethanol to Butadiene" Catalysts 9, no. 11: 920. https://doi.org/10.3390/catal9110920
APA StyleDochain, D. D., Stýskalík, A., & Debecker, D. P. (2019). Ag- and Cu-Promoted Mesoporous Ta-SiO2 Catalysts Prepared by Non-Hydrolytic Sol-Gel for the Conversion of Ethanol to Butadiene. Catalysts, 9(11), 920. https://doi.org/10.3390/catal9110920






