Development of Nickel-BTC-MOF-Derived Nanocomposites with rGO Towards Electrocatalytic Oxidation of Methanol and Its Product Analysis
Abstract
1. Introduction
2. Results and Discussion
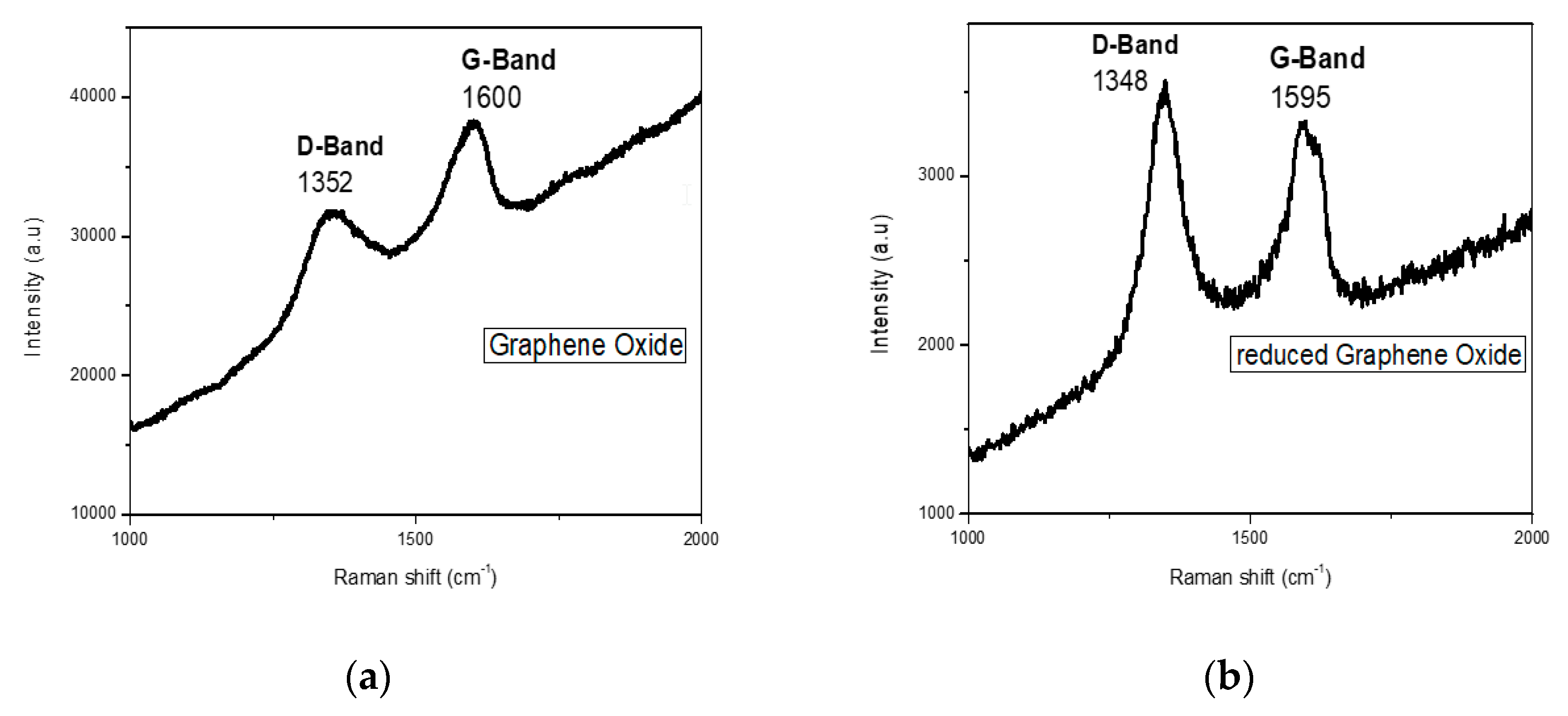
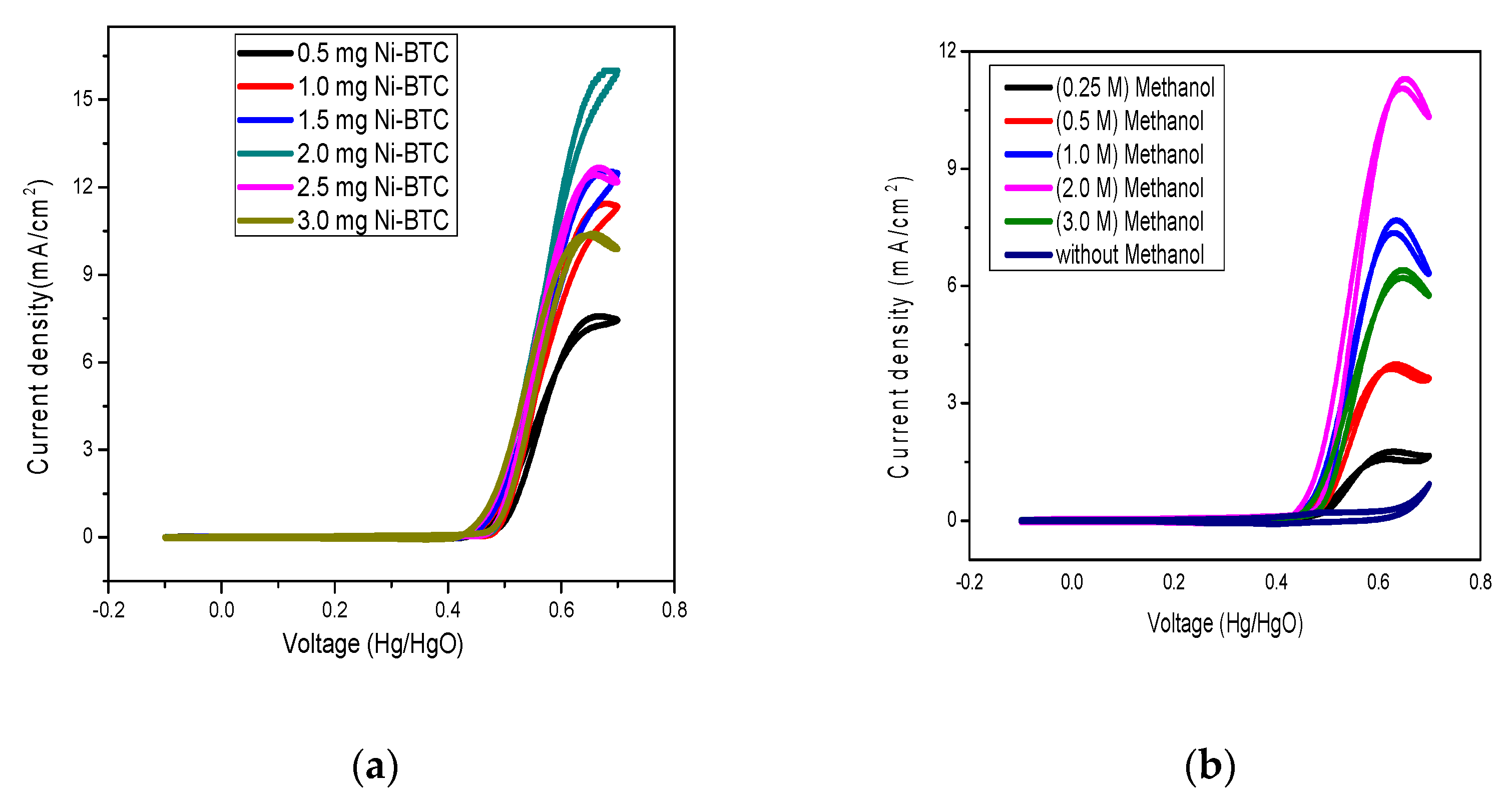
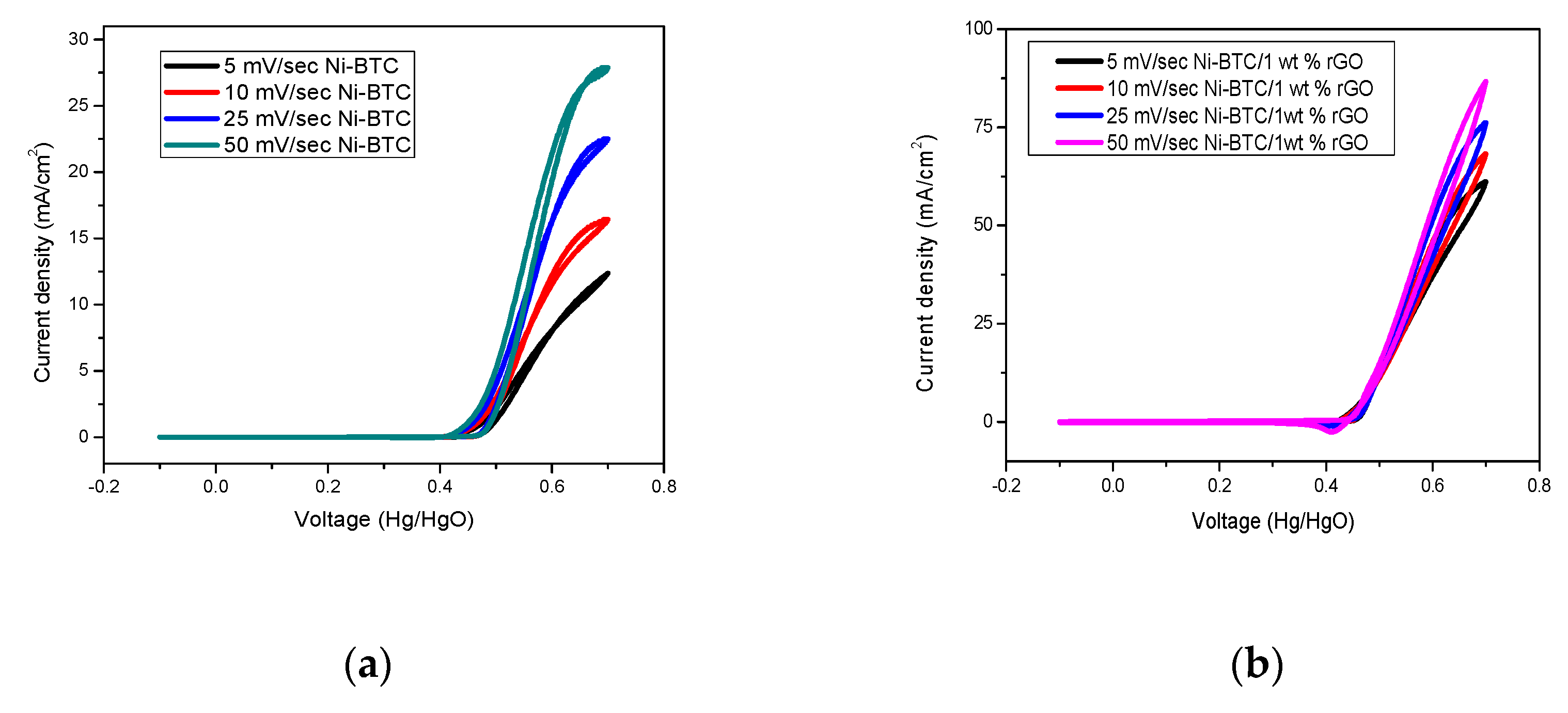
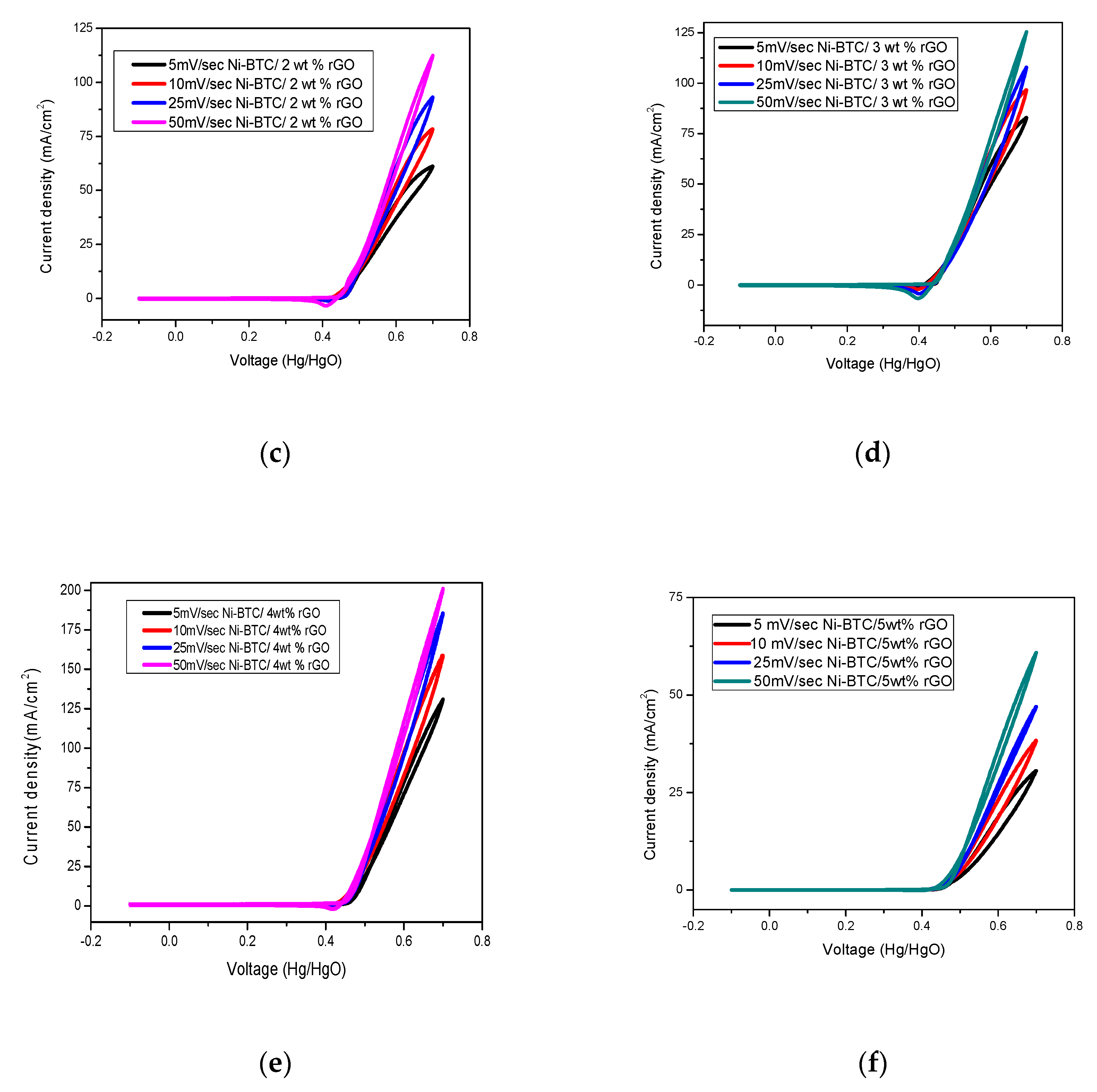
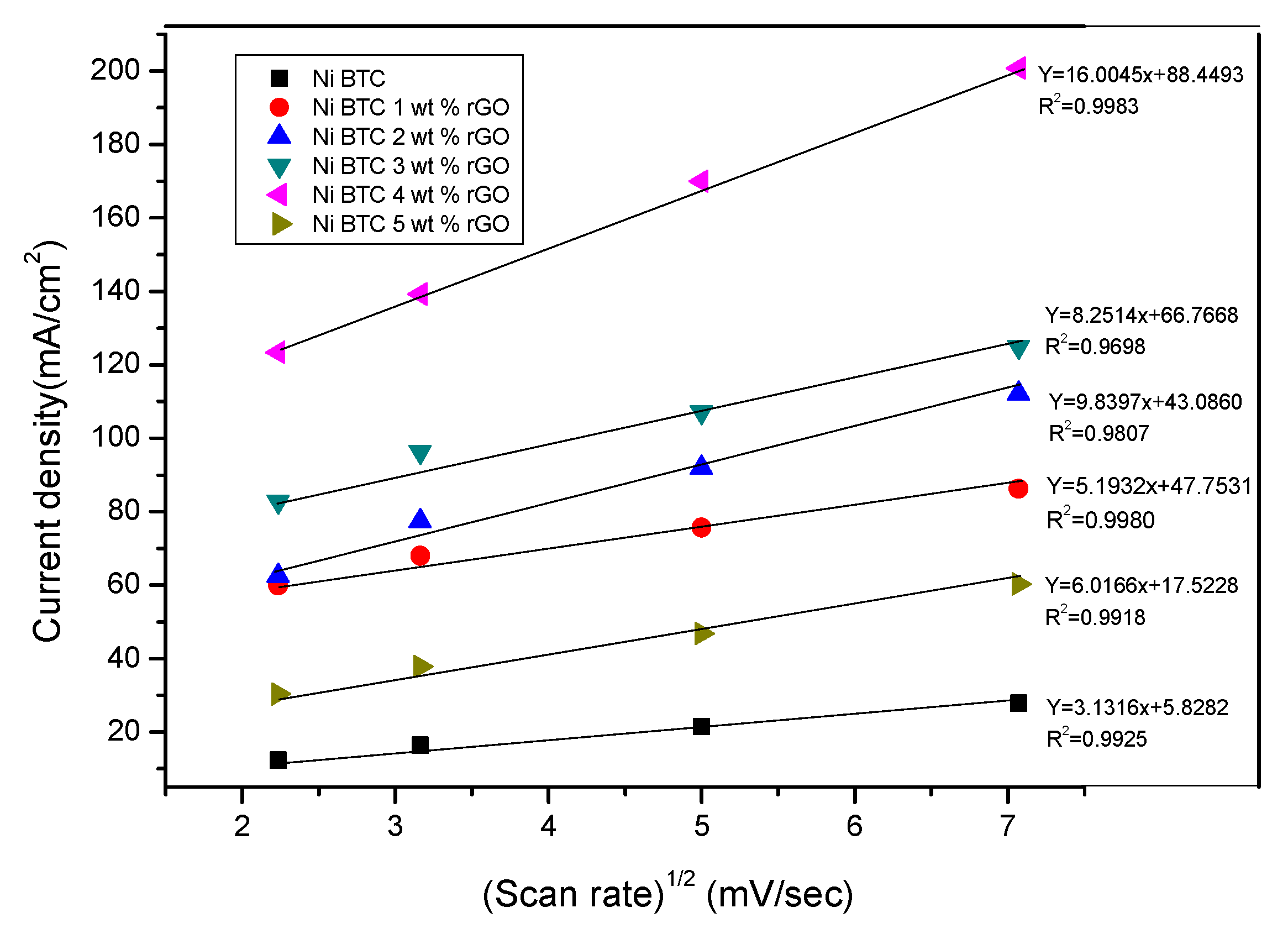
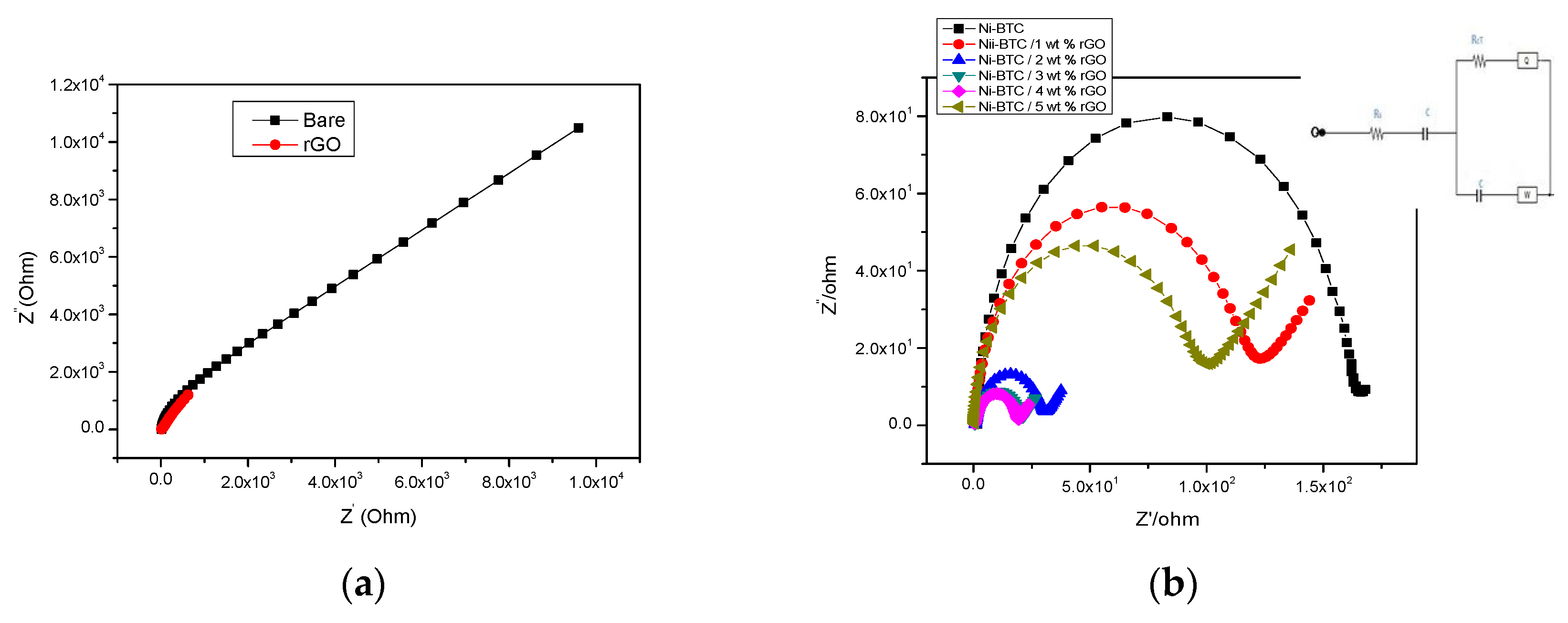
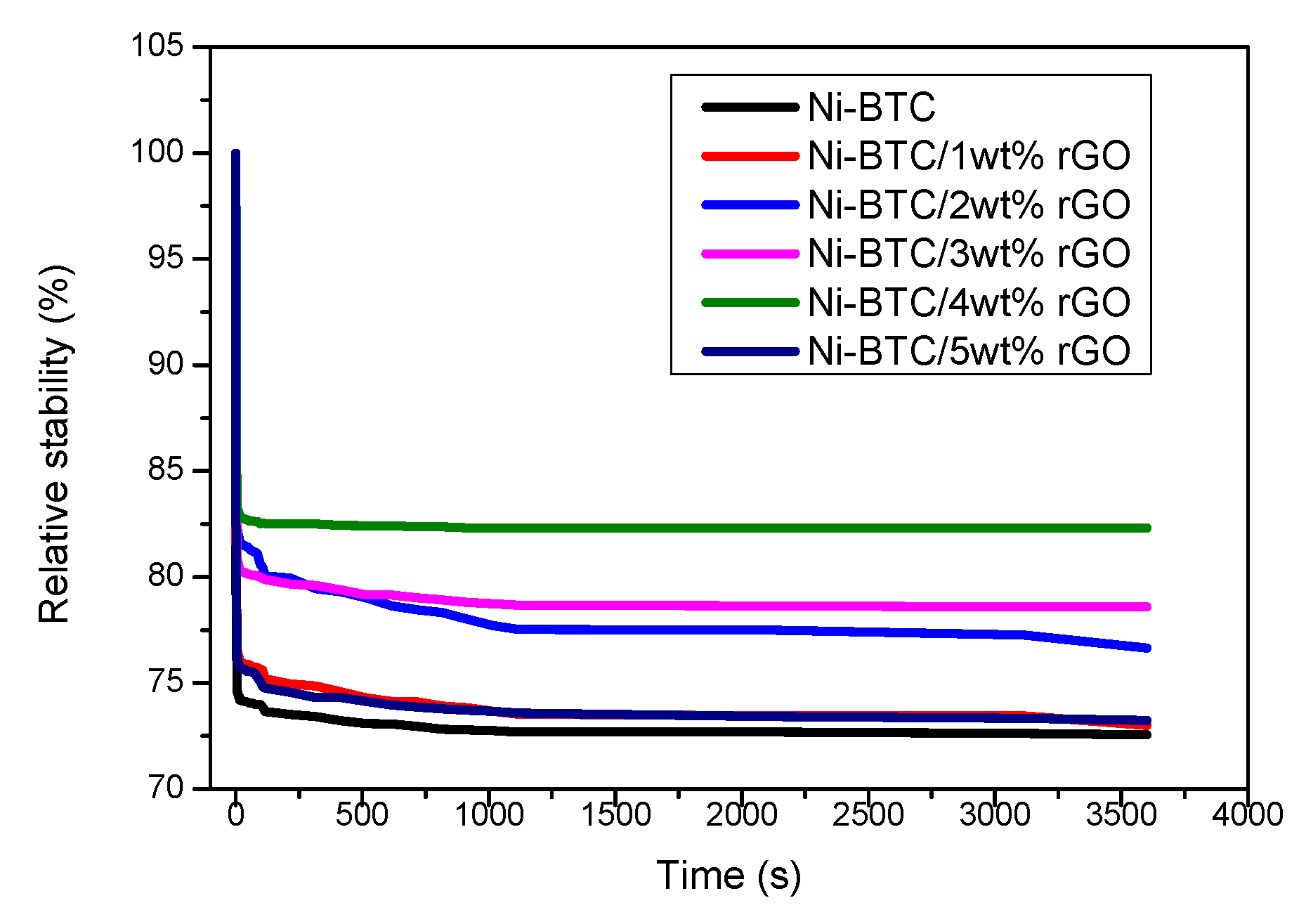
2.1. Electrochemical Measurements
2.2. Preparation of Working Electrodes
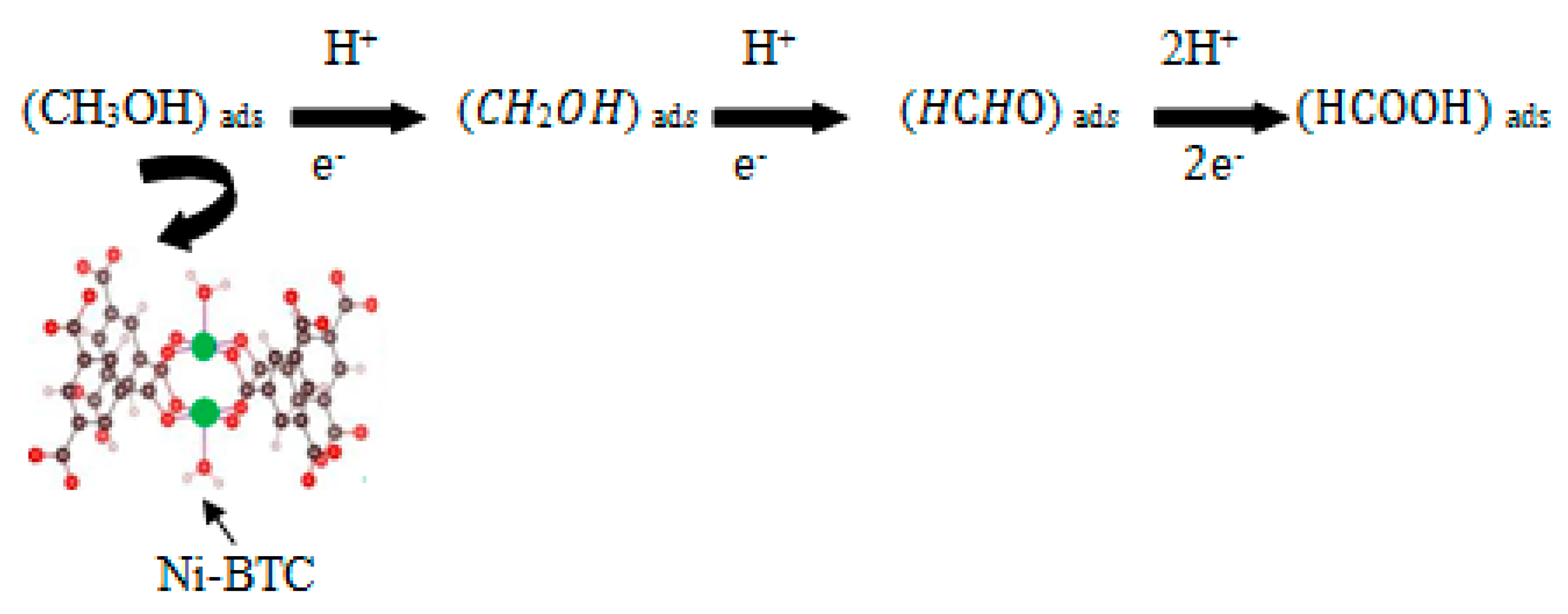
Bulk Electrolysis and Product Analysis
3. Material and Methods
3.1. Materials
3.2. Instrumentation
3.3. Synthesis of Ni-BTC-MOF
3.4. Synthesis of GO and rGO
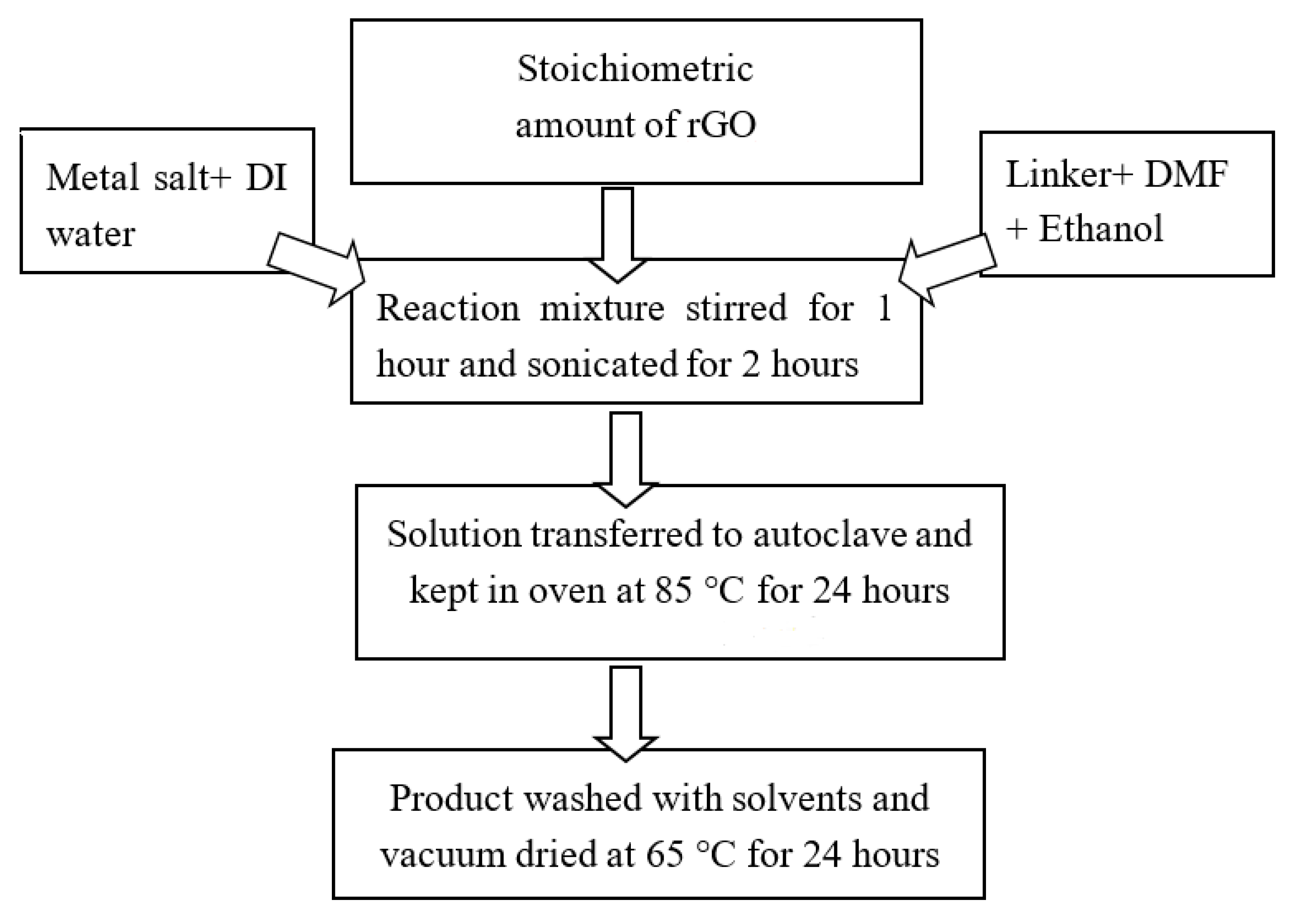
3.5. Synthesis of Ni-BTC-MOF/rGO Composites
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Dincer, I. Renewable energy and sustainable development: A crucial review. Renew. Sustain. Energy Rev. 2000, 4, 157–175. [Google Scholar] [CrossRef]
- Mintsouli, I.; Georgieva, J.; Armyanov, S.; Valova, E.; Avdeev, G.; Hubin, A.; Steenhaut, O.; Dille, J.; Tsiplakides, D.; Balomenou, S. Pt-Cu electrocatalysts for methanol oxidation prepared by partial galvanic replacement of Cu/carbon powder precursors. Appl. Catal. B Environ. 2013, 136, 160–167. [Google Scholar] [CrossRef]
- Appleby, A.J. Fuel Cell Handbook; Krieger Pub Co.: Malabar, FL, USA, 1989. [Google Scholar]
- Steele, B.; Heinzel, A. Materials for fuel-cell technologies. Nature 2001, 414, 345–352. [Google Scholar] [CrossRef]
- Franceschini, E.A.; Bruno, M.M.; Williams, F.J.; Viva, F.A.; Corti, H.R. High-activity mesoporous Pt/Ru catalysts for methanol oxidation. ACS Appl. Mater. Interfaces 2013, 5, 10437–10444. [Google Scholar] [CrossRef]
- Tian, G.L.; Zhang, Q.; Zhang, B.; Jin, Y.G.; Huang, J.Q.; Su, D.S.; Wei, F. Toward full exposure of “active sites”: Nanocarbon electrocatalyst with surface enriched nitrogen for superior oxygen reduction and evolution reactivity. Adv. Funct. Mater. 2014, 24, 5956–5961. [Google Scholar] [CrossRef]
- Kaichev, V.V.; Popova, G.Y.; Chesalov, Y.A.; Saraev, A.; Zemlyanov, D.; Beloshapkin, S.; Knop-Gericke, A.; Schlögl, R.; Andrushkevich, T.; Bukhtiyarov, V. Selective oxidation of methanol to form dimethoxymethane and methyl formate over a monolayer V2O5/TiO2 catalyst. J. Catal. 2014, 311, 59–70. [Google Scholar] [CrossRef]
- Kakati, N.; Maiti, J.; Lee, S.H.; Jee, S.H.; Viswanathan, B.; Yoon, Y.S. Anode catalysts for direct methanol fuel cells in acidic media: Do we have any alternative for Pt or Pt–Ru? Chem. Rev. 2014, 114, 12397–12429. [Google Scholar] [CrossRef]
- Mathe, N.R.; Scriba, M.R.; Rikhotso, R.S.; Coville, N.J. Microwave-irradiation polyol synthesis of PVP-protected Pt–Ni electrocatalysts for methanol oxidation reaction. Electrocatalysis 2018, 9, 388–399. [Google Scholar] [CrossRef]
- Li, W.; Liang, C.; Qiu, J.; Zhou, W.; Han, H.; Wei, Z.; Sun, G.; Xin, Q. Carbon nanotubes as support for cathode catalyst of a direct methanol fuel cell. Carbon 2002, 40, 787–790. [Google Scholar] [CrossRef]
- Mehek, R.; Iqbal, N.; Noor, T.; Nasir, H.; Mehmood, Y.; Ahmed, S. Novel Co-MOF/graphene oxide electrocatalyst for methanol oxidation. Electrochim. Acta 2017, 255, 195–204. [Google Scholar] [CrossRef]
- Heli, H.; Jafarian, M.; Mahjani, M.; Gobal, F. Electro-oxidation of methanol on copper in alkaline solution. Electrochim. Acta 2004, 49, 4999–5006. [Google Scholar] [CrossRef]
- Liu, F.; Lee, J.Y.; Zhou, W. Multi-Segment Pt–RuNi Nanorods for Methanol Electro-Oxidation at Room Temperature. J. Electrochem. Soc. 2006, 153, A2133–A2138. [Google Scholar] [CrossRef]
- He, Y.-B.; Li, G.-R.; Wang, Z.-L.; Ou, Y.-N.; Tong, Y.-X. Pt nanorods aggregates with enhanced electrocatalytic activity toward methanol oxidation. J. Phys. Chem. C 2010, 114, 19175–19181. [Google Scholar] [CrossRef]
- Alia, S.M.; Zhang, G.; Kisailus, D.; Li, D.; Gu, S.; Jensen, K.; Yan, Y. Porous platinum nanotubes for oxygen reduction and methanol oxidation reactions. Adv. Funct. Mater. 2010, 20, 3742–3746. [Google Scholar] [CrossRef]
- Varela, F.R.; Savadogo, O. The effect of anode catalysts on the behavior of low temperature direct propane polymer electrolyte fuel cells (DPFC). J. New Mater. Electrochem. Syst. 2006, 9, 127. [Google Scholar]
- Cai, Z.; Martin, C.R. Electronically conductive polymer fibers with mesoscopic diameters show enhanced electronic conductivities. J. Am. Chem. Soc. 1989, 111, 4138–4139. [Google Scholar] [CrossRef]
- Hsin, Y.L.; Hwang, K.C.; Yeh, C.-T. Poly (vinylpyrrolidone)-modified graphite carbon nanofibers as promising supports for PtRu catalysts in direct methanol fuel cells. J. Am. Chem. Soc. 2007, 129, 9999–10010. [Google Scholar] [CrossRef]
- Li, Y.; Gao, W.; Ci, L.; Wang, C.; Ajayan, P.M. Catalytic performance of Pt nanoparticles on reduced graphene oxide for methanol electro-oxidation. Carbon 2010, 48, 1124–1130. [Google Scholar] [CrossRef]
- Zhu, Q.-L.; Xu, Q. Metal–organic framework composites. Chem. Soc. Rev. 2014, 43, 5468–5512. [Google Scholar] [CrossRef]
- Huang, X.; Qi, X.; Boey, F.; Zhang, H. Graphene-based composites. Chem. Soc. Rev. 2012, 41, 666–686. [Google Scholar] [CrossRef]
- Stock, N.; Biswas, S. Synthesis of metal-organic frameworks (MOFs): Routes to various MOF topologies, morphologies, and composites. Chem. Rev. 2011, 112, 933–969. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Yeo, S.; Jeon, J.-D.; Kwak, S.-Y. Enhancement of hydrogen storage capacity and hydrostability of metal–organic frameworks (MOFs) with surface-loaded platinum nanoparticles and carbon black. Microporous Mesoporous Mater. 2015, 202, 8–15. [Google Scholar] [CrossRef]
- Khan, I.A.; Badshah, A.; Nadeem, M.A.; Haider, N.; Nadeem, M.A. A copper based metal-organic framework as single source for the synthesis of electrode materials for high-performance supercapacitors and glucose sensing applications. Int. J. Hydrogen Energy 2014, 39, 19609–19620. [Google Scholar] [CrossRef]
- Jahan, M.; Liu, Z.; Loh, K.P. A Graphene oxide and copper-centered metal organic framework composite as a tri-functional catalyst for HER, OER, and ORR. Adv. Funct. Mater. 2013, 23, 5363–5372. [Google Scholar] [CrossRef]
- Tang, H.; Cai, S.; Xie, S.; Wang, Z.; Tong, Y.; Pan, M.; Lu, X. Metal–organic-framework-derived dual metal- and nitrogen-doped carbon as efficient and robust oxygen reduction reaction catalysts for microbial fuel cells. Adv. Sci. 2016, 3, 1500265. [Google Scholar] [CrossRef]
- Ma, T.Y.; Dai, S.; Jaroniec, M.; Qiao, S.Z. Metal–organic framework derived hybrid Co3O4-carbon porous nanowire arrays as reversible oxygen evolution electrodes. J. Am. Chem. Soc. 2014, 136, 13925–13931. [Google Scholar] [CrossRef]
- Fu, Y.; Sun, D.; Chen, Y.; Huang, R.; Ding, Z.; Fu, X.; Li, Z. An amine-functionalized titanium metal–organic framework photocatalyst with visible-light-induced activity for CO2 reduction. Angew. Chem. Int. Ed. 2012, 51, 3364–3367. [Google Scholar] [CrossRef]
- Dang, L.Q.; Nguyen, M.T.; Van Truong, N.; Le, P.H.; Long, N. Investigation of Carbon Supported Ru–Pt Nanoparticles for High–Performance Electrocatalytic Oxidation of Methanol. Int. J. Electrochem. Sci. 2017, 12, 10187–10198. [Google Scholar] [CrossRef]
- Behmenyar, G.; Akın, A.N. Investigation of carbon supported Pd–Cu nanoparticles as anode catalysts for direct borohydride fuel cell. J. Power Sources 2014, 249, 239–246. [Google Scholar] [CrossRef]
- Song, G.; Wang, Z.; Wang, L.; Li, G.; Huang, M.; Yin, F. Preparation of MOF (Fe) and its catalytic activity for oxygen reduction reaction in an alkaline electrolyte. Chin. J. Catal. 2014, 35, 185–195. [Google Scholar] [CrossRef]
- Hanif, S.; Shi, X.; Iqbal, N.; Noor, T.; Anwar, R.; Kannan, A. ZIF derived PtNiCo/NC cathode catalyst for proton exchange membrane fuel cell. Appl.Catal. B Environ. 2019, 258, 117947. [Google Scholar] [CrossRef]
- Noor, T.; Zaman, N.; Nasir, H.; Iqbal, N.; Hussain, Z. Electro catalytic study of NiO-MOF/rGO composites for methanol oxidation reaction. Electrochim. Acta 2019, 307, 1–12. [Google Scholar] [CrossRef]
- Noor, T.; Ammad, M.; Zaman, N.; Iqbal, N.; Yaqoob, L.; Nasir, H. A Highly Efficient and Stable Copper BTC Metal Organic Framework Derived Electrocatalyst for Oxidation of Methanol in DMFC Application. Catal. Lett. 2019, 1–16. [Google Scholar] [CrossRef]
- Sarwar, E.; Noor, T.; Iqbal, N.; Mehmood, Y.; Ahmed, S.; Mehek, R. Effect of Co-Ni Ratio in Graphene Based Bimetallic Electro-catalyst for Methanol Oxidation. Fuel Cells 2018, 18, 189–194. [Google Scholar] [CrossRef]
- Rahim, M.A.; Hameed, R.A.; Khalil, M. Nickel as a catalyst for the electro-oxidation of methanol in alkaline medium. J. Power Sources 2004, 134, 160–169. [Google Scholar] [CrossRef]
- Sreeprasad, T.; Maliyekkal, S.M.; Lisha, K.; Pradeep, T. Reduced graphene oxide–metal/metal oxide composites: Facile synthesis and application in water purification. J. Hazard. Mater. 2011, 186, 921–931. [Google Scholar] [CrossRef]
- Krishnamoorthy, K.; Jeyasubramanian, K.; Premanathan, M.; Subbiah, G.; Shin, H.S.; Kim, S.J. Graphene oxide nanopaint. Carbon 2014, 72, 328–337. [Google Scholar] [CrossRef]
- Zhou, L.; Kong, X.; Gao, M.; Lian, F.; Li, B.; Zhou, Z.; Cao, H. Hydrothermal fabrication of MnCO3@ rGO composite as an anode material for high-performance lithium ion batteries. Inorg. Chem. 2014, 53, 9228–9234. [Google Scholar] [CrossRef]
- Petit, C.; Bandosz, T.J. MOF–graphite oxide composites: Combining the uniqueness of graphene layers and metal–organic frameworks. Adv. Mater. 2009, 21, 4753–4757. [Google Scholar] [CrossRef]
- Israr, F.; Kim, D.K.; Kim, Y.; Oh, S.J.; Ng, K.C.; Chun, W. Cost effective and low energy consuming hydrothermal synthesis of Ni based MOF. J. Energ. Eng. 2015, 24, 51–54. [Google Scholar] [CrossRef]
- Israr, F.; Chun, D.; Kim, Y.; Kim, D.K. High yield synthesis of Ni-BTC metal–organic framework with ultrasonic irradiation: Role of polar aprotic DMF solvent. Ultrason. Sonochem. 2016, 31, 93–101. [Google Scholar] [CrossRef] [PubMed]
- Yaghi, O.; Li, H.; Groy, T. Construction of porous solids from hydrogen-bonded metal complexes of 1, 3, 5-benzenetricarboxylic acid. J. Am. Chem. Soc. 1996, 118, 9096–9101. [Google Scholar] [CrossRef]
- Jabarian, S.; Ghaffarinejad, A. Electrochemical Synthesis of NiBTC Metal Organic Framework Thin Layer on Nickel Foam: An Efficient Electrocatalyst for the Hydrogen Evolution Reaction. J. Inorg. Organomet. Polym. Mater. 2019, 29, 1565–1574. [Google Scholar] [CrossRef]
- Jabbar, A.; Yasin, G.; Khan, W.Q.; Anwar, M.Y.; Korai, R.M.; Nizam, M.N.; Muhyodin, G. Electrochemical deposition of nickel graphene composite coatings: Effect of deposition temperature on its surface morphology and corrosion resistance. RSC Adv. 2017, 7, 31100–31109. [Google Scholar] [CrossRef]
- Andrijanto, E.; Shoelarta, S.; Subiyanto, G.; Rifki, S. Facile synthesis of graphene from graphite using ascorbic acid as reducing agent. In Proceedings of the AIP ICAMST 2015 Conference, Semarang, Indonesia, 6–7 October 2015. [Google Scholar]
- Zhang, Y.; Liu, H.; Zhu, Z.; Wong, K.-W.; Mi, R.; Mei, J.; Lau, W.-M. A green hydrothermal approach for the preparation of graphene/α-MnO2 3D network as anode for lithium ion battery. Electrochim. Acta 2013, 108, 465–471. [Google Scholar] [CrossRef]
- Zeng, G.; Chen, Y.; Chen, L.; Xiong, P.; Wei, M. Hierarchical cerium oxide derived from metal-organic frameworks for high performance supercapacitor electrodes. Electrochim. Acta 2016, 222, 773–780. [Google Scholar] [CrossRef]
- Sun, K.; Li, L.; Yu, X.; Liu, L.; Meng, Q.; Wang, F.; Zhang, R. Functionalization of mixed ligand metal-organic frameworks as the transport vehicles for drugs. J. Colloid Interface Sci. 2017, 486, 128–135. [Google Scholar] [CrossRef]
- Wu, Y.; Song, X.; Li, S.; Zhang, J.; Yang, X.; Shen, P.; Gao, L.; Wei, R.; Zhang, J.; Xiao, G. 3D-monoclinic M–BTC MOF (M= Mn, Co, Ni) as highly efficient catalysts for chemical fixation of CO2 into cyclic carbonates. J. Ind. Eng. Chem. 2018, 58, 296–303. [Google Scholar] [CrossRef]
- Hamidipour, L.; Farzaneh, F. Cobalt metal organic framework as an efficient heterogeneous catalyst for the oxidation of alkanes and alkenes. React. Kinet. Mech. Catal. 2013, 109, 67–75. [Google Scholar] [CrossRef]
- Machado, B.F.; Serp, P. Graphene-based materials for catalysis. Catal. Sci. Technol. 2012, 2, 54–75. [Google Scholar] [CrossRef]
- Hidayah, N.; Liu, W.-W.; Lai, C.-W.; Noriman, N.; Khe, C.-S.; Hashim, U.; Lee, H.C. Comparison on graphite, graphene oxide and reduced graphene oxide: Synthesis and characterization. AIP Conf. Proc. 2017, 1892, 150002. [Google Scholar]
- Feng, H.; Cheng, R.; Zhao, X.; Duan, X.; Li, J. A low-temperature method to produce highly reduced graphene oxide. Nat. Commun. 2013, 4, 1539. [Google Scholar] [CrossRef] [PubMed]
- Antolini, E.; Salgado, J.R.; Gonzalez, E.R. The methanol oxidation reaction on platinum alloys with the first row transition metals: The case of Pt–Co and–Ni alloy electrocatalysts for DMFCs: A short review. Appl. Catal. B Environ. 2006, 149, 137–149. [Google Scholar] [CrossRef]
- Cordeiro, C.; De Vries, M.; Cremers, T.; Westerink, B. The role of surface availability in membrane-induced selectivity for amperometric enzyme-based biosensors. Sens. Actuators B Chem. 2016, 223, 679–688. [Google Scholar] [CrossRef]
- Das, A.K.; Layek, R.K.; Kim, N.H.; Jung, D.; Lee, J.H. Reduced graphene oxide (RGO)-supported NiCo2O4 nanoparticles: An electrocatalyst for methanol oxidation. Nanoscale 2014, 6, 10657–10665. [Google Scholar] [CrossRef]
- Zhu, D.; Guo, C.; Liu, J.; Wang, L.; Du, Y.; Qiao, S.-Z. Two-dimensional metal–organic frameworks with high oxidation states for efficient electrocatalytic urea oxidation. Chem. Commun. 2017, 53, 10906–10909. [Google Scholar] [CrossRef]
- Zhang, X.; Jiang, Z.H.; Yao, Z.P.; Song, Y.; Wu, Z.D. Effects of scan rate on the potentiodynamic polarization curve obtained to determine the Tafel slopes and corrosion current density. Corros. Sci. 2009, 51, 581–587. [Google Scholar] [CrossRef]
- Parwaiz, S.; Bhunia, K.; Das, A.K.; Khan, M.M.; Pradhan, D. Cobalt-doped ceria/reduced graphene oxide nanocomposite as an efficient oxygen reduction reaction catalyst and supercapacitor material. J. Phys. Chem. C 2017, 121, 20165–20176. [Google Scholar] [CrossRef]
- Huang, T.; Mao, S.; Zhou, G.; Zhang, Z.; Wen, Z.; Huang, X.; Ci, S.; Chen, J. A high-performance catalyst support for methanol oxidation with graphene and vanadium carbonitride. Nanoscale 2015, 7, 1301–1307. [Google Scholar] [CrossRef]
- Yu, E.H.; Scott, K.; Reeve, R.W. A study of the anodic oxidation of methanol on Pt in alkaline solutions. J. Electroanal. Chem. 2003, 547, 17–24. [Google Scholar] [CrossRef]
- Niu, L.; Li, Q.; Wei, F.; Chen, X.; Wang, H. Electrochemical impedance and morphological characterization of platinum-modified polyaniline film electrodes and their electrocatalytic activity for methanol oxidation. J. Electroanal. Chem. 2003, 544, 121–128. [Google Scholar] [CrossRef]
- Zhu, X.; Zhang, P.; Xu, S.; Yan, X.; Xue, Q. Free-standing three-dimensional graphene/manganese oxide hybrids as binder-free electrode materials for energy storage applications. ACS Appl. Mater. Interfaces 2014, 6, 11665–11674. [Google Scholar] [CrossRef] [PubMed]
- Wu, Q.; Jiang, M.; Zhang, X.; Cai, J.; Lin, S. A novel octahedral MnO/RGO composite prepared by thermal decomposition as a noble-metal free electrocatalyst for ORR. J. Mater. Sci. 2017, 52, 6656–6669. [Google Scholar] [CrossRef]
- Wang, W.; Li, Y.; Wang, H. Tin oxide nanoparticle-modified commercial PtRu catalyst for methanol oxidation. Micro Nano Lett. 2013, 8, 23–26. [Google Scholar] [CrossRef]
- Huang, Y.; Cai, J.; Liu, M.; Guo, Y. Fabrication of a novel PtPbBi/C catalyst for ethanol electro-oxidation in alkaline medium. Electrochim. Acta 2012, 83, 1–6. [Google Scholar] [CrossRef]
- Wang, H.; Da, H.; Wang, R.; Ji, S. Beef-derived mesoporous carbon as highly efficient support for PtRuIr electrocatalysts and their high activity for CO and methanol oxidation. S. Afr. J. Chem. 2014, 67, 33–39. [Google Scholar]
- Ye, W.; Zhang, X.; Chen, Y.; Du, Y.; Zhou, F.; Wang, C. Pulsed electrodeposition of reduced graphene oxide on glass carbon electrode as an effective support of electrodeposited Pt microspherical particles: Nucleation studies and the application for methanol electro-oxidation. Int. J. Electrochem. Sci. 2013, 8, e2139. [Google Scholar]
- Huang, W.; Wang, H.; Zhou, J.; Wang, J.; Duchesne, P.N.; Muir, D.; Zhang, P.; Han, N.; Zhao, F.; Zeng, M. Highly active and durable methanol oxidation electrocatalyst based on the synergy of platinum–nickel hydroxide–graphene. Nat. Commun. 2015, 6, 10035. [Google Scholar] [CrossRef]
- Zheng, Y.; Chen, H.; Dai, Y.; Zhang, N.; Zhao, W.; Wang, S.; Lou, Y.; Li, Y.; Sun, Y. Preparation and characterization of Pt/TiO2 nanofibers catalysts for methanol electro-oxidation. Electrochim. Acta 2015, 178, 74–79. [Google Scholar] [CrossRef]
- Vigier, F.; Rousseau, S.; Coutanceau, C.; Leger, J.-M.; Lamy, C. Electrocatalysis for the direct alcohol fuel cell. Top. Catal. 2006, 40, 111–121. [Google Scholar] [CrossRef]
- Asiri, H.A.; Anderson, A.B. Mechanisms for ethanol electrooxidation on Pt (111) and adsorption bond strengths defining an ideal catalyst. J. Electrochem. Soc. 2015, 162, F115–F122. [Google Scholar] [CrossRef]
- Yang, Y.; McElwee-White, L. Electrochemical oxidation of methanol using dppm-bridged Ru/Pd, Ru/Pt and Ru/Au catalysts. Dalton Trans. 2004, 15, 2352–2356. [Google Scholar] [CrossRef]
- Liu, Y.; Zhao, S.-F.; Guo, S.-X.; Bond, A.M.; Zhang, J.; Zhu, G.; Hill, C.L.; Geletii, Y.V. Electrooxidation of Ethanol and Methanol Using the Molecular Catalyst [{Ru4O4 (OH) 2 (H2O) 4}(γ-SiW10O36) 2]10−. J. Am. Chem. Soc. 2016, 138, 2617–2628. [Google Scholar] [CrossRef] [PubMed]
- Nahm, C.; Kim, C.; Park, Y.; Park, B. Nanoporous Pt thin films with superior catalytic activities by the electrochemical dissolution of Al. Met. Mater. Int. 2009, 15, 989–992. [Google Scholar] [CrossRef]
- Nguyen, T.; Thuy, V.; Luu, C.L.; Hoang, T.C.; Nguyen, T.; Bui, T.H.; Duy, N.; Phuc, H.; Pham, T.; Thuy, P. Synthesis of MOF-199 and application to CO2 adsorption. Adv. Nat. Sci. Nanosci. Nanotechnol. (Online) 2013, 4. [Google Scholar] [CrossRef]











| Sample Element | Ni-BTC | Ni-BTC/1 wt % rGO | Ni-BTC/2 wt % rGO | Ni-BTC/3 wt % rGO | Ni-BTC/4 wt % rGO | Ni-BTC/5 wt % rGO |
|---|---|---|---|---|---|---|
| C wt % | 35.66 | 39.66 | 42.13 | 45.65 | 53.11 | 53.69 |
| O wt % | 26.89 | 29.05 | 36.99 | 41.88 | 39.67 | 41.06 |
| Ni wt % | 37.45 | 31.29 | 20.92 | 12.48 | 7.22 | 5.25 |
| Catalyst | R2 | Diffusion Co-Efficient (cm2/s) |
|---|---|---|
| Ni-BTC | 0.99256 | 1.643 × 10−5 |
| Ni-BTC/1 wt % rGO | 0.98716 | 15.35 × 10−5 |
| Ni-BTC/2 wt % rGO | 0.99269 | 23.60 × 10−5 |
| Ni-BTC/3 wt % rGO | 0.98708 | 32.74 × 10−5 |
| Ni-BTC/4 wt % rGO | 0.99833 | 84.57 × 10−5 |
| Ni-BTC/5 wt % rGO | 0.99184 | 7.494 × 10−5 |
| Sr.No | Sample | Resistance (Rs) Ohm | Resistance (Rct) Ohm | Capacitance (C) Farad |
|---|---|---|---|---|
| 1 | Ni-BTC | 1.340 | 158.00 | 6.209e−6 |
| 2 | Ni-BTC/1 wt % rGO | 0.624 | 112.90 | 8.316e−6 |
| 3 | Ni-BTC/2 wt % rGO | 0.663 | 28.23 | 1.444e−5 |
| 4 | Ni-BTC/3 wt % rGO | 0.464 | 19.12 | 1.229e−5 |
| 5 | Ni-BTC/4 wt % rGO | 0.328 | 18.12 | 9.156e−6 |
| 6 | Ni-BTC/5 wt % rGO | 0.496 | 102.00 | 4.728e−6 |
| Catalyst | Rct (Ohm) | Tafel Slope (mV/dec) |
|---|---|---|
| Ni-BTC | 158 | 59.65 |
| Ni-BTC/1 wt % rGO | 112 | 33.69 |
| Ni-BTC/2 wt % rGO | 28.23 | 29.58 |
| Ni-BTC/3 wt % rGO | 19.12 | 28.10 |
| Ni-BTC/4 wt % rGO | 18.12 | 27.89 |
| Ni-BTC/5 wt % rGO | 102 | 75.47 |
| Catalytic Materials | Methanol Concentration (M) | Catalyst Amount (mg/cm2) | Scan Rate (mV/s) | Anodic Potential (V) | Peak Current Density (mA/cm2) | Resistance (Ohm) | Ref. |
|---|---|---|---|---|---|---|---|
| Pt-Ni | 1 | 20 | 50 | 1.44 vs. RHE | 265.6 | - | [9] |
| Ru@Pt/MWCNT | 2 | 4 | 50 | 1.81 vs. RHE | 182.4 | 606 | [29] |
| NiCo2O4-rGO | 0.5 | - | 50 | 1.26 vs. RHE | 48.0 | 1000 | [52] |
| Nanoporous Pt/Al thin films | 2 | - | 100 | 0.8 vs. RHE | 6.0 | - | [76] |
| Ni-BTC | 2 | 1.07 | 50 | 1.66 vs. RHE | 27.155 | 158 | This work |
| Ni-BTC/1 wt % rGO | 2 | 1.07 | 50 | 1.66 vs. RHE | 86.346 | 112 | This work |
| Ni-BTC/2 wt % rGO | 2 | 1.07 | 50 | 1.66 vs. RHE | 112.04 | 28.23 | This work |
| Ni-BTC/3 wt % rGO | 2 | 1.07 | 50 | 1.66 vs. RHE | 125.83 | 19.12 | This work |
| Ni-BTC/4 wt % rGO | 2 | 1.07 | 50 | 1.66 vs. RHE | 200.02 | 18.12 | This work |
| Ni-BTC/5 wt % rGO | 2 | 1.07 | 50 | 1.66 vs. RHE | 60.72 | 102 | This work |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yaqoob, L.; Noor, T.; Iqbal, N.; Nasir, H.; Zaman, N. Development of Nickel-BTC-MOF-Derived Nanocomposites with rGO Towards Electrocatalytic Oxidation of Methanol and Its Product Analysis. Catalysts 2019, 9, 856. https://doi.org/10.3390/catal9100856
Yaqoob L, Noor T, Iqbal N, Nasir H, Zaman N. Development of Nickel-BTC-MOF-Derived Nanocomposites with rGO Towards Electrocatalytic Oxidation of Methanol and Its Product Analysis. Catalysts. 2019; 9(10):856. https://doi.org/10.3390/catal9100856
Chicago/Turabian StyleYaqoob, Lubna, Tayyaba Noor, Naseem Iqbal, Habib Nasir, and Neelam Zaman. 2019. "Development of Nickel-BTC-MOF-Derived Nanocomposites with rGO Towards Electrocatalytic Oxidation of Methanol and Its Product Analysis" Catalysts 9, no. 10: 856. https://doi.org/10.3390/catal9100856
APA StyleYaqoob, L., Noor, T., Iqbal, N., Nasir, H., & Zaman, N. (2019). Development of Nickel-BTC-MOF-Derived Nanocomposites with rGO Towards Electrocatalytic Oxidation of Methanol and Its Product Analysis. Catalysts, 9(10), 856. https://doi.org/10.3390/catal9100856






