Oxidized Palladium Supported on Ceria Nanorods for Catalytic Aerobic Oxidation of Benzyl Alcohol to Benzaldehyde in Protic Solvents
Abstract
1. Introduction
2. Results and Discussion
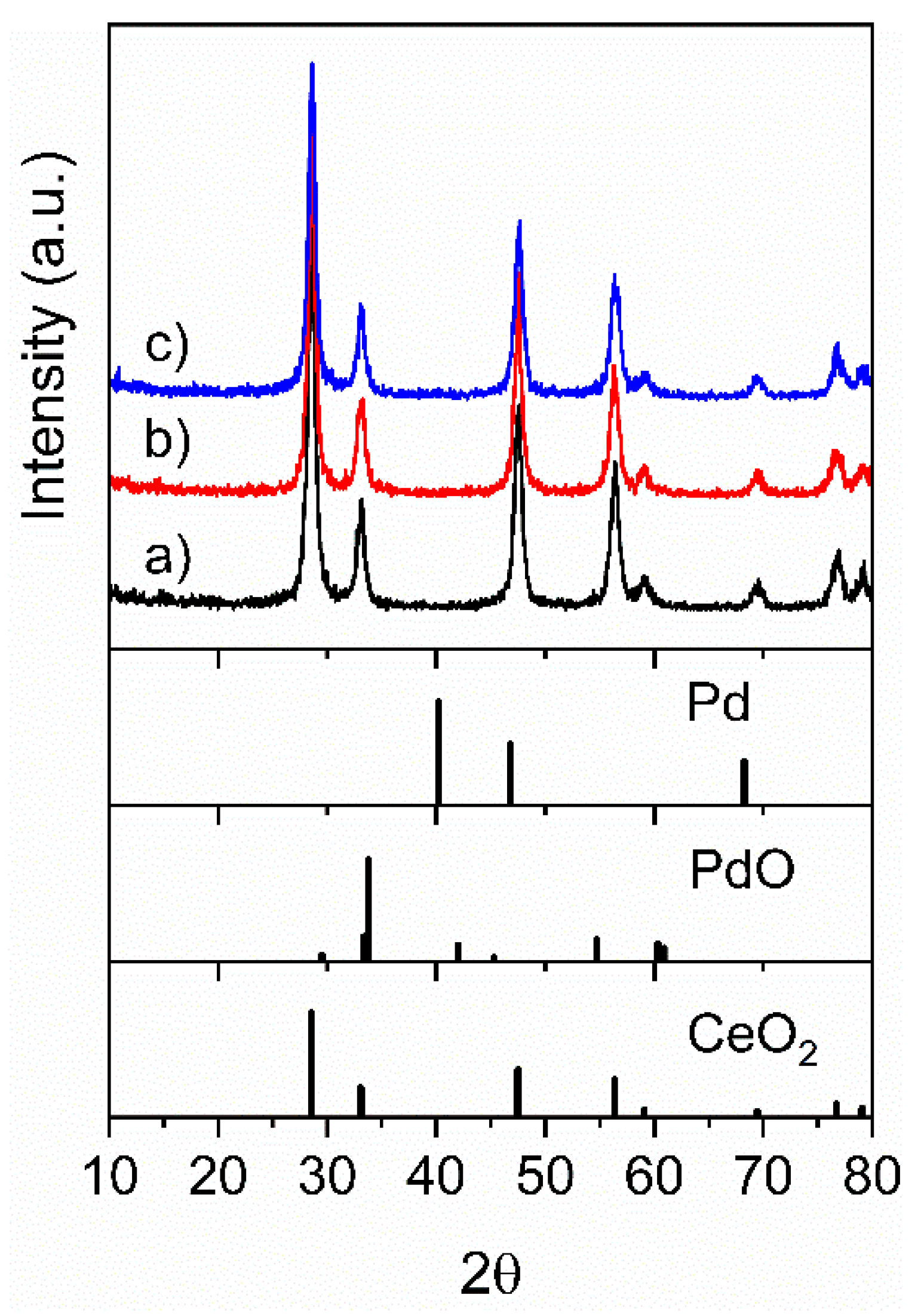
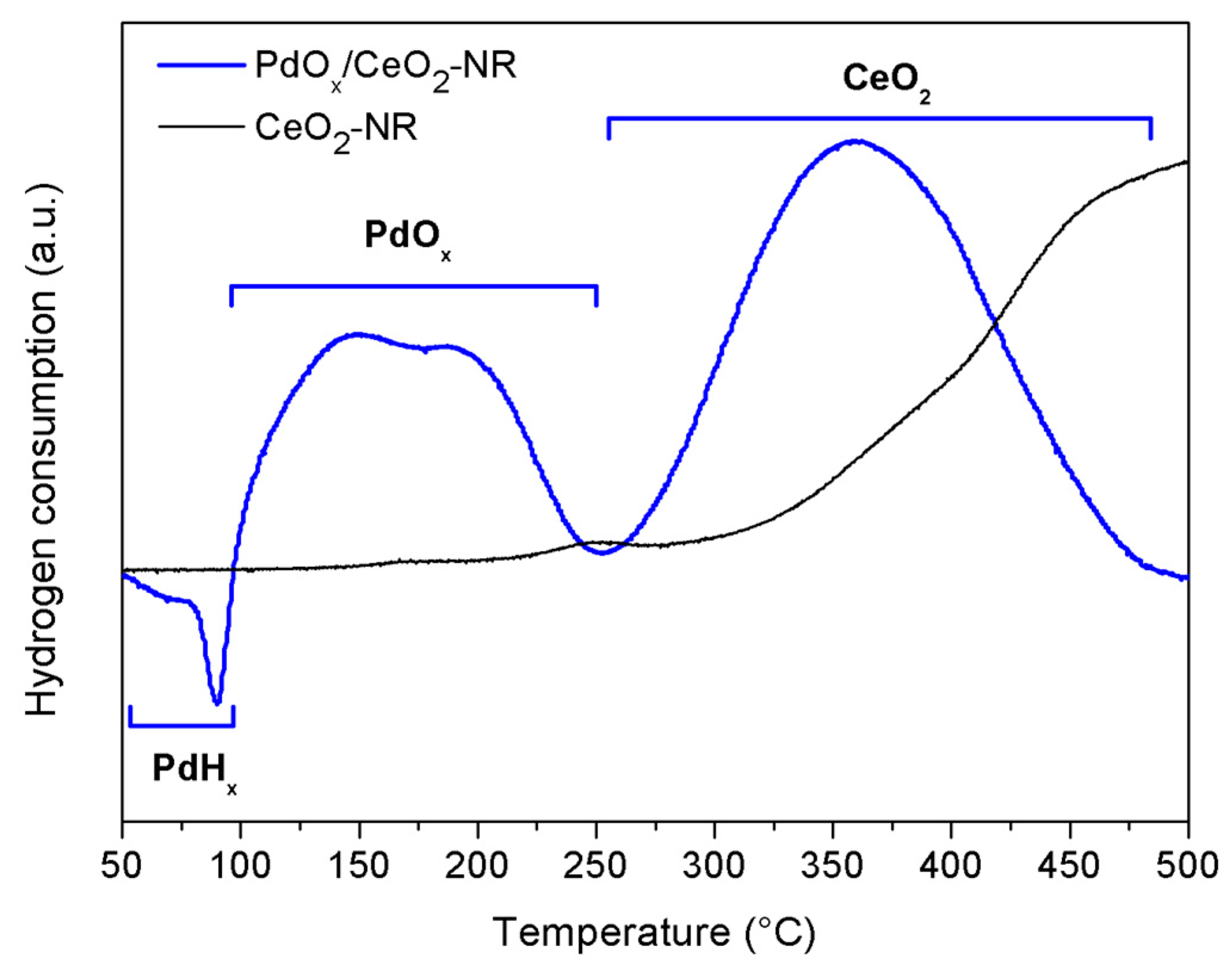
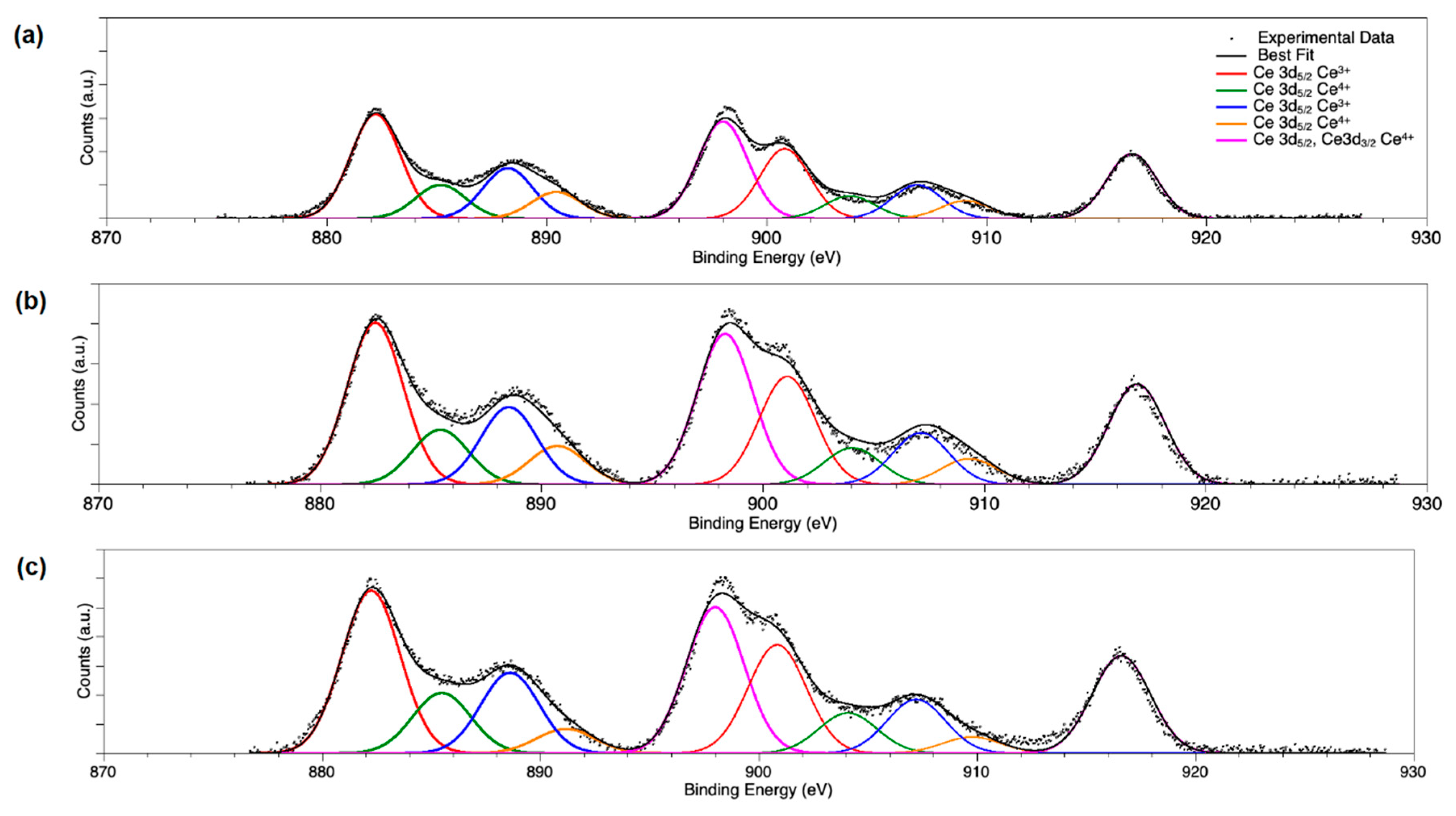
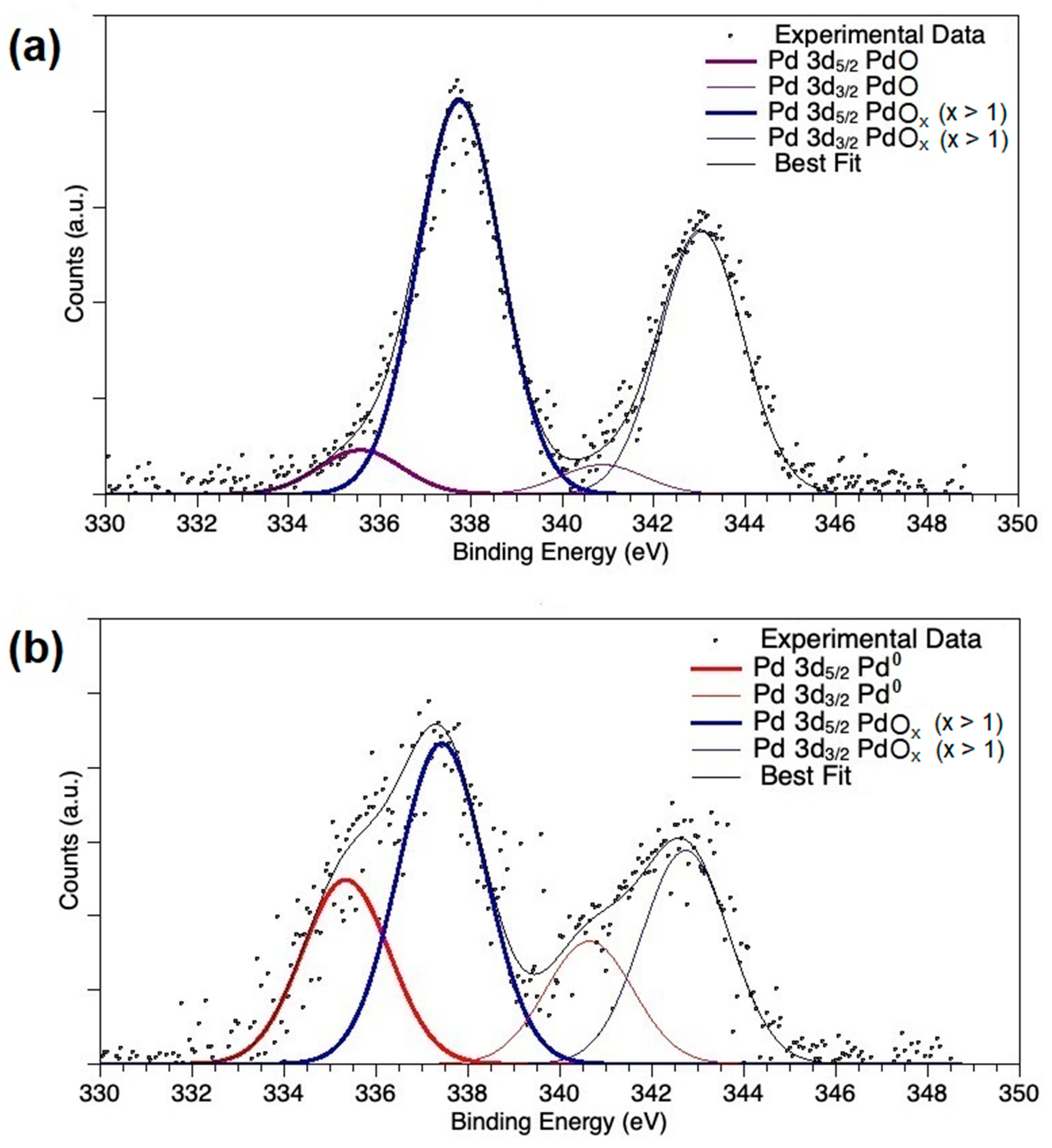
2.1. Catalyst Characterization
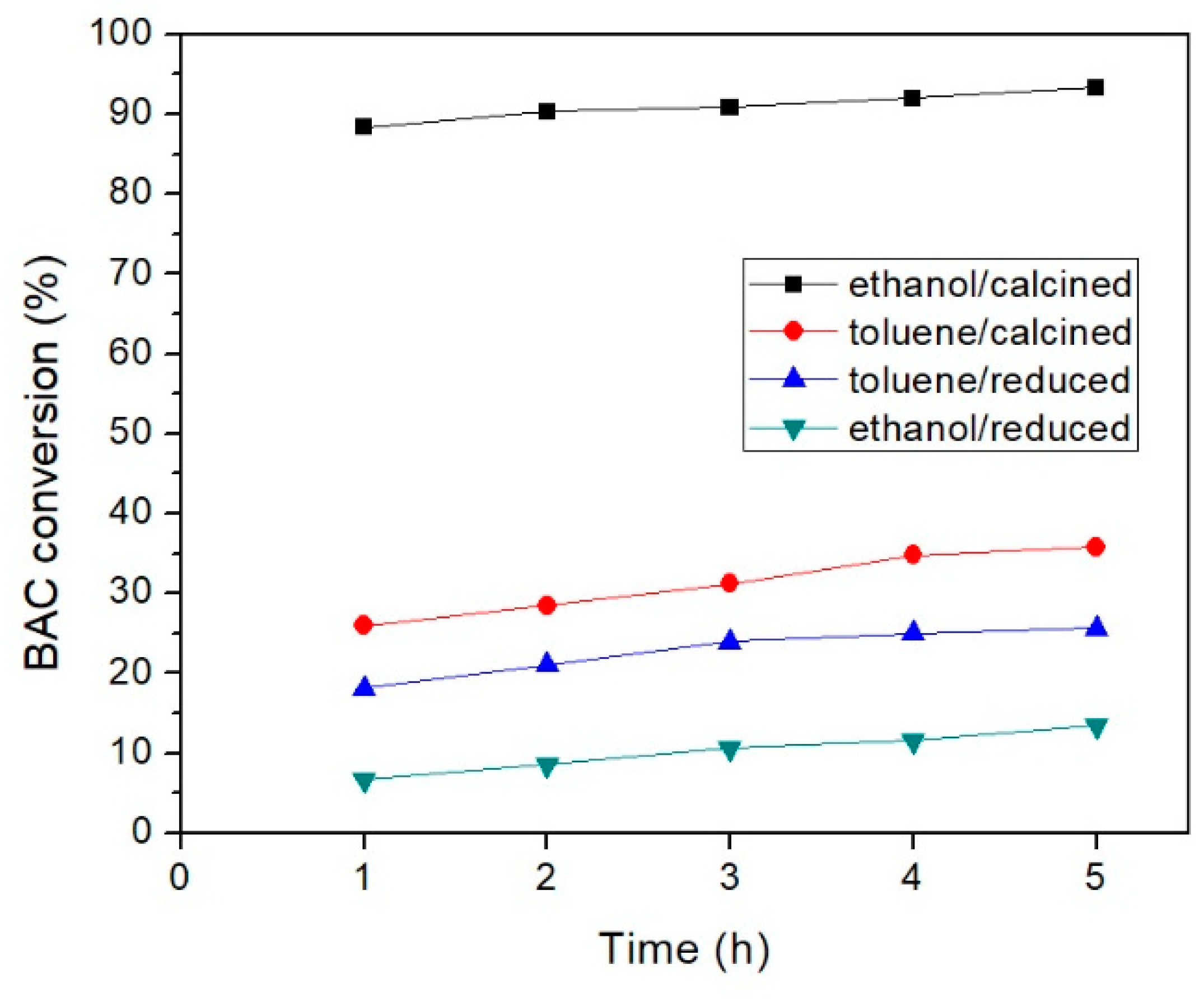
2.2. Catalytic Activity
2.2.1. Effect of Temperature and Solvent
2.2.2. Effect of Pd Oxidation State
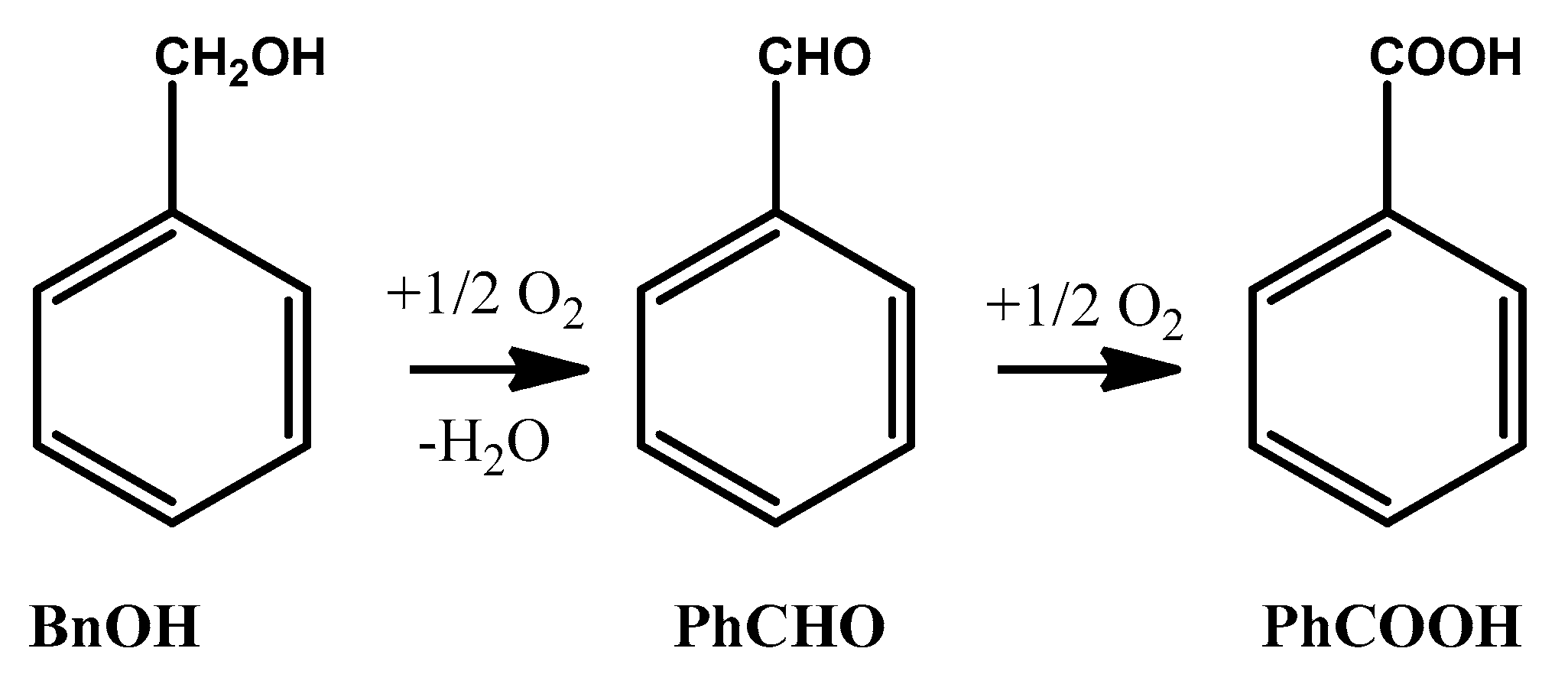
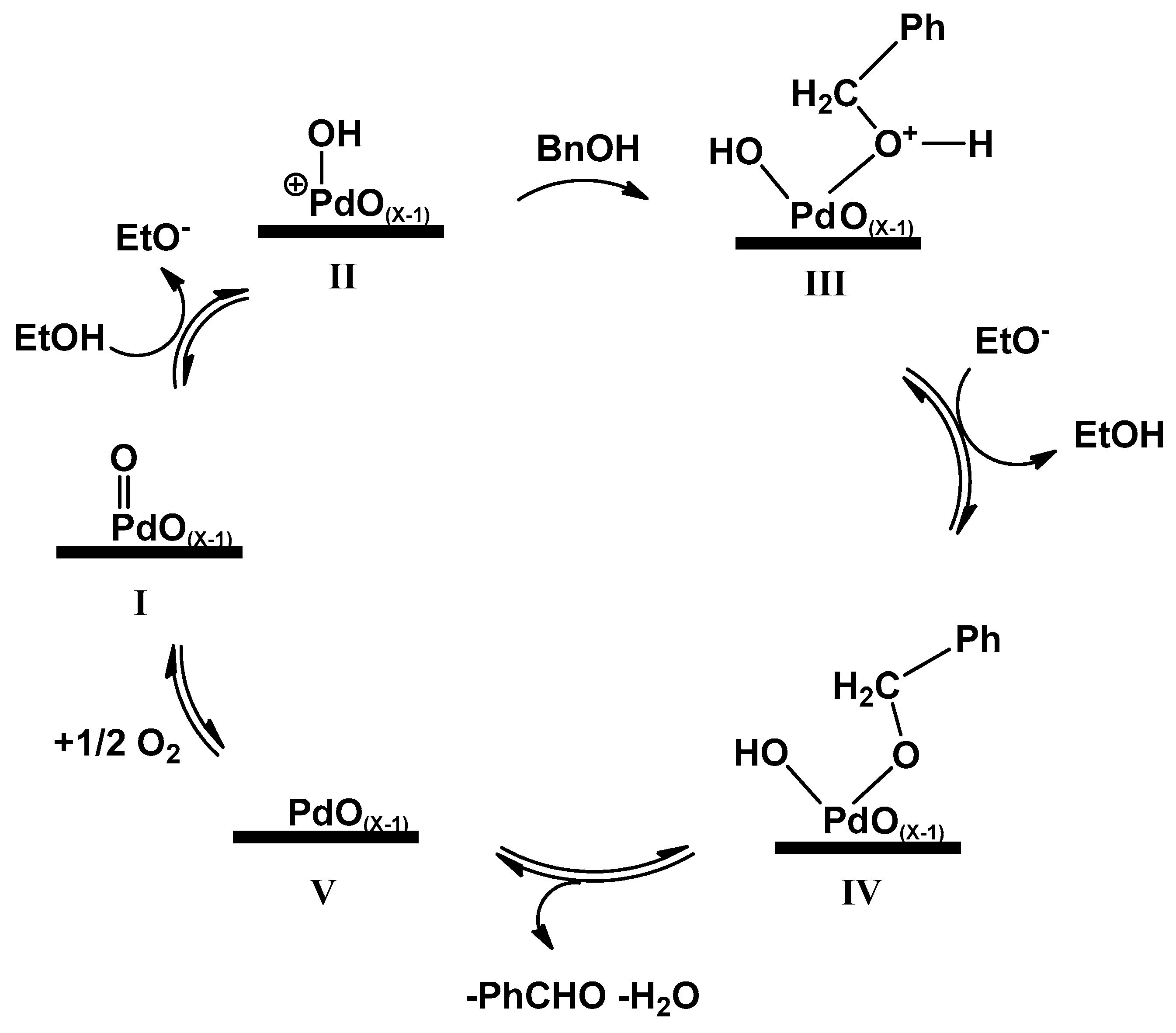
2.2.3. Mechanistic Aspects of BnOH Aerobic Oxidation, Catalyzed with PdOx/CeO2-NR
2.2.4. Leaching and Reusability Tests
3. Materials and Methods
3.1. Catalyst Preparation
3.2. Catalyst Characterization
3.3. Catalytic Tests
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Isaifan, R.J.; Ntais, S.; Baranova, E.A. Particle size effect on catalytic activity of carbon-supported Pt nanoparticles for complete ethylene oxidation. Appl. Catal. A Gen. 2013, 464–465, 87–94. [Google Scholar] [CrossRef]
- Kesavan, J.K.; Luisetto, I.; Tuti, S.; Meneghini, C.; Battocchio, C.; Iucci, G. Ni supported on YSZ: XAS and XPS characterization and catalytic activity for CO2 methanation. J. Mater. Sci. 2017, 52, 10331–10340. [Google Scholar] [CrossRef]
- Marconi, E.; Tuti, S.; Luisetto, I. Structure-Sensitivity of CO2 Methanation over Nanostructured Ni Supported on CeO2 Nanorods. Catalysts 2019, 9, 375. [Google Scholar] [CrossRef]
- Somorjai, G.A.; Tao, F.; Park, J.Y. The Nanoscience Revolution: Merging of Colloid Science, Catalysis and Nanoelectronics. Top. Catal. 2008, 47, 1–14. [Google Scholar] [CrossRef]
- Wyrwalski, F.; Giraudon, J.-M.; Lamonier, J.-F. Synergistic Coupling of the Redox Properties of Supports and Cobalt Oxide Co3O4 for the Complete Oxidation of Volatile Organic Compounds. Catal. Lett. 2010, 137, 141–149. [Google Scholar] [CrossRef]
- Rojluechaia, S.; Chavadeja, S.; Schwankb, J.W.; Meeyoo, V. Catalytic activity of ethylene oxidation over Au, Ag and Au–Ag catalysts: Support effect. Catal. Commun. 2007, 8, 57–64. [Google Scholar] [CrossRef]
- Comotti, M.; Li, W.C.; Spliethoff, B.; Schüth, F. Support Effect in High Activity Gold Catalysts for CO Oxidation. J. Am. Chem. Soc. 2006, 128, 917–924. [Google Scholar] [CrossRef]
- Jia, A.; Lou, L.-L.; Zhang, C.; Zhang, Y.; Liu, S. Selective oxidation of benzyl alcohol to benzaldehyde with hydrogen peroxide over alkali-treated ZSM-5 zeolite catalysts. J. Mol. Catal. A Chem. 2009, 306, 123–129. [Google Scholar] [CrossRef]
- Choudhary, V.R.; Chaudhari, P.A.; Narkhede, V.S. Solvent-free liquid phase oxidation of benzyl alcohol to benzaldehyde by molecular oxygen using non-noble transition metal containing hydrotalcite-like solid catalysts. Catal. Commun. 2003, 4, 171–175. [Google Scholar] [CrossRef]
- Lingaiah, N.; Reddy, K.M.; Babu, N.S.; Rao, K.N.; Suryanarayana, I.; Prasad, P.S.S. Aerobic selective oxidation of benzyl alcohol over vanadium substituted ammonium salt of 12-molybdophosphoric acid. Catal. Commun. 2006, 7, 245–250. [Google Scholar] [CrossRef]
- Davis, S.E.; Ide, M.S.; Davis, R.J. Selective oxidation of alcohols and aldehydes over supported metal nanoparticles. Green Chem. 2013, 15, 17–45. [Google Scholar] [CrossRef]
- Marko, I.E.; Giles, P.R.; Tsukazaki, M.; Chellé-Regnaut, I.; Urch, C.J.; Brown, S.M. Efficient, Aerobic, Ruthenium-Catalyzed Oxidation of Alcohols into Aldehydes and Ketones. J. Am. Chem. Soc. 1997, 119, 12661–12662. [Google Scholar] [CrossRef]
- Dijksman, A.; Marino-Gonza′lez, A.; Mairata i Payeras, A.; Arends, I.W.C.E.; Sheldon, R.A. Efficient and Selective Aerobic Oxidation of Alcohols into Aldehydes and Ketones Using Ruthenium/TEMPO as the Catalytic System. J. Am. Chem. Soc. 2001, 123, 6826–6833. [Google Scholar] [CrossRef] [PubMed]
- Yamaguchi, K.; Mizuno, N. Supported Ruthenium Catalyst for the Heterogeneous Oxidation of Alcohols with Molecular Oxygen. Angew. Chem. Int. Ed. 2002, 41, 4538–4542. [Google Scholar] [CrossRef]
- Han, D.; Xu, T.; Su, J.; Xu, X.; Ding, Y. Gas-Phase Selective Oxidation of Benzyl Alcohol to Benzaldehyde with Molecular Oxygen over Unsupported Nanoporous Gold. ChemCatChem 2010, 2, 383–386. [Google Scholar] [CrossRef]
- Zhan, G.; Huang, J.; Du, M.; Sun, D.; Abdul-Rauf, I.; Lin, W.; Hong, Y.; Li, Q. Liquid phase oxidation of benzyl alcohol to benzaldehyde with novel uncalcined bioreduction Au catalysts: High activity and durability. Chem. Eng. J. 2012, 187, 232–238. [Google Scholar] [CrossRef]
- Liu, H.; Liu, Y.; Li, Y.; Tang, Z.; Jiang, H. Metal-Organic Framework Supported Gold Nanoparticles as a Highly Active Heterogeneous Catalyst for Aerobic Oxidation of Alcohols. J. Phys. Chem. C 2010, 114, 13362–13369. [Google Scholar] [CrossRef]
- Ma, C.Y.; Cheng, J.; Wang, H.L.; Hu, Q.; Tian, H.; He, C.; Hao, Z.P. Characteristics of Au/HMS catalysts for selective oxidation of benzyl alcohol to benzaldehyde. Catal. Today 2010, 158, 246–251. [Google Scholar] [CrossRef]
- Li, H.; Qin, F.; Yang, Z.; Cui, X.; Wang, J.; Zhang, L. New Reaction Pathway Induced by Plasmon for Selective Benzyl Alcohol Oxidation on BiOCl Possessing Oxygen Vacancies. J. Am. Chem. Soc. 2017, 139, 3513–3521. [Google Scholar] [CrossRef]
- Blackburn, T.F.; Schwartz, J. Homogeneous catalytic oxidation of secondary alcohols to ketones by molecular oxygen under mild conditions. J. Chem. Soc. Chem. Commun. 1977, 157–158. [Google Scholar] [CrossRef]
- Karimi, B.; Abedi, S.; Clark, J.H.; Budarin, V. Highly Efficient Aerobic Oxidation of Alcohols Using a Recoverable Catalyst: The Role of Mesoporous Channels of SBA-15 in Stabilizing Palladium Nanoparticles. Angew. Chem. 2006, 118, 4894–4897. [Google Scholar] [CrossRef]
- Hackett, S.F.J.; Brydson, R.M.; Gass, M.H.; Harvey, I.; Newman, A.D.; Wilson, K.; Lee, A.F. High-Activity, Single-Site Mesoporous Pd/Al2O3 Catalysts for Selective Aerobic Oxidation of Allylic Alcohols. Angew. Chem. 2007, 119, 8747–8750. [Google Scholar] [CrossRef]
- Zhou, Y.; Xiang, Z.; Cao, D.; Liu, C. Preparation and Characterization of Covalent Organic Polymer Supported Palladium Catalysts for Oxidation of CO and Benzyl Alcohol. Ind. Eng. Chem. Res. 2014, 53, 1359–1367. [Google Scholar] [CrossRef]
- Xu, J.; Shang, J.-K.; Chen, Y.; Wang, Y.; Li, Y.-X. Palladium nanoparticles supported on mesoporous carbon nitride for efficiently selective oxidation of benzyl alcohol with molecular oxygen. Appl. Catal. A Gen. 2017, 542, 380–388. [Google Scholar] [CrossRef]
- Sharma, M.; Das, B.; Deka, B.K.; Park, Y.B.; Bhargava, S.K.; Bania, K.K. Pd/Cu-Oxide Nanoconjugate at Zeolite-Y Crystallite Crafting the Mesoporous Channels for Selective Oxidation of Benzyl-Alcohols. ACS Appl. Mater. Interfaces 2017, 9, 35453–35462. [Google Scholar] [CrossRef] [PubMed]
- Akbari, S.; Mokhtari, J.; Mirjafari, Z. Solvent-free and melt aerobic oxidation of benzyl alcohols using Pd/Cu2(BDC)2DABCO–MOF prepared by one-step and through reduction by dimethylformamide. R. Soc. Chem. 2017, 7, 40881–40886. [Google Scholar] [CrossRef]
- Enache, D.I.; Barker, D.; Edwards, J.K.; Taylor, S.H.; Knight, D.W.; Carley, A.F.; Hutchings, G.J. Solvent-free oxidation of benzyl alcohol using titania-supported gold–palladium catalysts: Effect of Au–Pd ratio on catalytic performance. Catal. Today 2007, 122, 407–411. [Google Scholar] [CrossRef]
- Miedziak, P.J.; He, Q.; Edwards, J.K.; Taylor, S.H.; Knight, D.W.; Tarbit, B.; Kiely, C.J.; Hutchings, G.J. Oxidation of benzyl alcohol using supported gold–palladium nanoparticles. Catal. Today 2011, 163, 47–54. [Google Scholar] [CrossRef]
- Marx, S.; Baiker, A. Beneficial Interaction of Gold and Palladium in Bimetallic Catalysts for the Selective Oxidation of Benzyl Alcohol. J. Phys. Chem. C 2009, 113, 6191–6201. [Google Scholar] [CrossRef]
- Ma, C.Y.; Dou, B.J.; Li, J.J.; Cheng, J.; Hu, Q.; Hao, Z.P.; Qiao, S.Z. Catalytic oxidation of benzyl alcohol on Au or Au–Pd nanoparticles confined in mesoporous silica. Appl. Catal. B Environ. 2009, 92, 202–208. [Google Scholar] [CrossRef]
- Chen, Y.; Lim, H.; Tang, Q.; Gao, Y.; Sun, T.; Yan, Q.; Yang, Y. Solvent-free aerobic oxidation of benzyl alcohol over Pd monometallic and Au–Pd bimetallic catalysts supported on SBA-16 mesoporous molecular sieves. Appl. Catal. A Gen. 2010, 380, 55–65. [Google Scholar] [CrossRef]
- Khawaji, M.; Chadwick, D. Au-Pd Bimetallic Nanoparticles Immobilised on Titanate Nanotubes: A Highly Active Catalyst for Selective Oxidation. ChemCatChem 2017, 9, 4353–4363. [Google Scholar] [CrossRef]
- Yan, Z.; Tomer, A.; Perrussel, G.; Ousmane, M.; Katryniok, B.; Dumeignil, F.; Ponchel, A.; Liebens, A.; Pera-Titus, M. A Pd/CeO2 “H2Pump” for the Direct Amination of Alcohols. ChemCatChem 2016, 8, 3347–3352. [Google Scholar] [CrossRef]
- Meng, L.; Jia, A.-P.; Lu, J.-Q.; Luo, L.-F.; Huang, W.-X.; Luo, M.-F. Synergetic Effects of PdO Species on CO Oxidation over PdO–CeO2 Catalysts. J. Phys. Chem. C 2011, 115, 19789–19796. [Google Scholar] [CrossRef]
- Liu, J.; Wang, F.; Dewil, R. CeO2 Nanocrystalline-Supported Palladium Chloride: An Effective Catalyst for Selective Oxidation of Alcohols by Oxygen. Catal. Lett. 2009, 130, 448–454. [Google Scholar] [CrossRef]
- Alhumaimess, M.; Lin, Z.; Weng, W.; Dimitratos, N.; Dummer, N.F.; Taylor, S.H.; Bartley, J.K.; Kiely, C.J.; Hutchings, G.J. Oxidation of benzyl alcohol by using gold nanoparticles supported on ceria foam. ChemSusChem 2012, 5, 125–131. [Google Scholar] [CrossRef]
- Zhang, H.; Xie, Y.; Sun, Z.; Tao, R.; Huang, C.; Zhao, Y.; Liu, Z. In-situ loading ultrafine AuPd particles on ceria: Highly active catalyst for solvent-free selective oxidation of benzyl alcohol. Langmuir 2011, 27, 1152–1157. [Google Scholar] [CrossRef]
- Lei, Y.; Li, W.; Liu, Q.; Lin, Q.; Zheng, X.; Huang, Q.; Guan, S.; Wang, X.; Wang, C.; Li, F. Typical crystal face effects of different morphology ceria on the activity of Pd/CeO2 catalysts for lean methane combustion. Fuel 2018, 233, 10–20. [Google Scholar] [CrossRef]
- Wang, X.; Chen, J.; Jianxin, Z.; Wang, Q.; Li, Z.; Qin, R.; Wu, C.; Xie, Z.; Zheng, L. The synergy between atomically dispersed Pd and cerium oxide for enhanced catalytic properties. Nanoscale 2017, 9, 6643–6648. [Google Scholar] [CrossRef]
- Lykaki, M.; Pachatouridou, E.; Carabineiro, S.A.C.; Iliopoulou, E.; Andriopoulou, C.; Kallithrakas-Kontos, N.; Boghosian, S.; Konsolakis, M. Ceria nanoparticles shape effects on the structural defects and surface chemistry: Implications in CO oxidation by Cu/CeO2 catalysts. Appl. Catal. B Environ. 2018, 230, 18–28. [Google Scholar] [CrossRef]
- Du, X.; Zhang, D.; Shi, L.; Gao, R.; Zhang, J. Morphology Dependence of Catalytic Properties of Ni/CeO2 Nanostructures for Carbon Dioxide Reforming of Methane. J. Phys. Chem. C 2012, 116, 10009–10016. [Google Scholar] [CrossRef]
- Wang, S.; Zhao, L.; Wang, W.; Zhao, Y.; Zhang, G.; Ma, X.; Gong, J. Morphology control of ceria nanocrystals for catalytic conversion of CO2 with methanol. Nanoscale 2013, 5, 5582–5588. [Google Scholar] [CrossRef] [PubMed]
- Xin, P.; Li, J.; Xiong, Y.; Wu, X.; Dong, J.; Chen, W.; Wang, Y.; Gu, L.; Luo, J.; Rong, H.; et al. Revealing the Active Species for Aerobic Alcohol Oxidation by Using Uniform Supported Palladium Catalysts. Angew. Chem. Int. Ed. Engl. 2018, 57, 4642–4646. [Google Scholar] [CrossRef] [PubMed]
- Tan, H.; Wang, J.; Yu, S.; Zhou, K. Support Morphology-Dependent Catalytic Activity of Pd/CeO2 for Formaldehyde Oxidation. Environ. Sci. Technol. 2015, 49, 8675–8682. [Google Scholar] [CrossRef] [PubMed]
- Liu, B.; Liu, R.; Li, Q.-J.; Yao, M.-G.; Zou, B.; Cui, T.; Liu, B.-B.; Liu, J. Study of high pressure structural stability of CeO2 nanoparticles. Chin. Phys. C 2013, 37. [Google Scholar] [CrossRef]
- Wang, W.-W.; Yu, W.-Z.; Du, P.-P.; Xu, H.; Jin, Z.; Si, R.; Ma, C.; Shi, S.; Jia, C.-J.; Yan, C.-H. Crystal Plane Effect of Ceria on Supported Copper Oxide Cluster Catalyst for CO Oxidation: Importance of Metal–Support Interaction. ACS Catal. 2017, 7, 1313–1329. [Google Scholar] [CrossRef]
- Feng, T.; Tang, R.; Shang, N.; Feng, C.; Gao, S.; Wang, C. Pd nanoparticles supported on CeO2 as efficient catalyst for hydrogen generation from formaldehyde solution at room temperature. Appl. Organomet. Chem. 2017, 31, e3889. [Google Scholar] [CrossRef]
- Luo, J.-Y.; Meng, M.; Xian, H.; Tu, Y.-B.; Li, X.-G.; Ding, T. The Nanomorphology-Controlled Palladium-Support Interaction and the Catalytic Performance of Pd/CeO2 Catalysts. Catal. Lett. 2009, 133, 328–333. [Google Scholar] [CrossRef]
- Luisetto, I.; Tuti, S.; Romano, C.; Boaro, M.; Di Bartolomeo, E. Dry reforming of methane over Ni supported on doped CeO2: New insight on the role of dopants for CO2 activation. J. CO2 Util. 2019, 30, 63–78. [Google Scholar] [CrossRef]
- Kurnatowska, M.; Kepinski, L.; Mista, W. Structure evolution of nanocrystalline Ce1−xPdxO2−y mixed oxide in oxidizing and reducing atmosphere: Reduction-induced activity in low-temperature CO oxidation. Appl. Catal. B Environ. 2012, 117–118, 135–147. [Google Scholar] [CrossRef]
- Shen, J.; Hayes, R.E.; Wu, X.; Semagina, N. 100° Temperature Reduction of Wet Methane Combustion: Highly Active Pd–Ni/Al2O3 Catalyst versus Pd/NiAl2O4. ACS Catal. 2015, 5, 2916–2920. [Google Scholar] [CrossRef]
- Agostini, G.; Groppo, E.; Piovano, A.; Pellegrini, R.; Leofanti, G.; Lamberti, C. Preparation of supported Pd catalysts: From the Pd precursor solution to the deposited Pd2+ phase. Langmuir 2010, 26, 11204–11211. [Google Scholar] [CrossRef] [PubMed]
- Pinna, F.; Menegazzo, F.; Signoretto, M.; Canton, P.; Fagherazzi, G.; Pernicone, N. Consecutive Hydrogenation of Benzaldehyde Over Pd Catalysts Influence of Supports and Sulfur Poisoning. Appl. Catal. A Gen. 2001, 219, 195–200. [Google Scholar] [CrossRef]
- Luo, M.F.; Hou, Z.Y.; Yaun, X.X.; Zheng, X.M. Characterization study of CeO2 supported Pd catalyst for low-temperature carbon monoxide oxidation. Catal. Lett. 1998, 50, 205–209. [Google Scholar] [CrossRef]
- Gopinath, R.; Lingaiah, N.; Sreedhar, B.; Suryanarayana, I.; Sai Prasad, P.S.; Obuchi, A. Highly stable Pd/CeO2 catalyst for hydrodechlorination of chlorobenzene. Appl. Catal. B Environ. 2003, 46, 587–594. [Google Scholar] [CrossRef]
- Jiang, D.; Wang, W.; Zhang, L.; Zheng, Y.; Wang, Z. Insights into the Surface-Defect Dependence of Photoreactivity over CeO2 Nanocrystals with Well-Defined Crystal Facets. ACS Catal. 2015, 5, 4851–4858. [Google Scholar] [CrossRef]
- Shyu, J.Z.; Weber, W.H.; Gandhi, H.S. Surface Characterization of Alumina-Supported Ceria. J. Phys. Chem. 1988, 92, 4964–4970. [Google Scholar] [CrossRef]
- NIST X-ray Photoelectron Spectroscopy Database. Version 4.1. Available online: https://srdata.nist.gov/xps/ (accessed on 15 October 2018).
- You, R.; Li, Z.; Cao, T.; Nan, B.; Si, R.; Huang, W. Synthesis in Glovebox: Utilizing Surface Oxygen Vacancies to Enhance Atomic Dispersion of Palladium on Ceria for CO Oxidation and Propane Combustion. ACS Appl. Nano Mater. 2018, 1, 4988–4997. [Google Scholar] [CrossRef]
- Tang, K.; Ren, Y.; Liu, W.; Wei, J.; Guo, J.; Wang, S.; Yang, Y. Insight Investigation of Active Palladium Surface Sites in Palladium-Ceria Catalysts for NO + CO Reaction. ACS Appl. Mater. Interfaces 2018, 10, 13614–13624. [Google Scholar] [CrossRef]
- Al-Saeedi, S.; Abdel-Rahman, L.; Abu-Dief, A.; Abdel-Fatah, S.; Alotaibi, T.; Alsalme, A.; Nafady, A. Catalytic Oxidation of Benzyl Alcohol Using Nanosized Cu/Ni Schiff-Base Complexes and Their Metal Oxide Nanoparticles. Catalysts 2018, 8, 452. [Google Scholar] [CrossRef]
- Savara, A.; Chan-Thaw, C.E.; Rossetti, I.; Villa, A.; Prati, L. Benzyl Alcohol Oxidation on Carbon-Supported Pd Nanoparticles: Elucidating the Reaction Mechanism. ChemCatChem 2014, 6, 3464–3473. [Google Scholar] [CrossRef]
- Zope, B.N.; Hibbitts, D.D.; Neurock, M.; Davis, R.J. Reactivity of the Gold/Water Interface During Selective Oxidation Catalysis. Science 2010, 330, 74–78. [Google Scholar] [CrossRef] [PubMed]
- Chan-Thaw, C.; Savara, A.; Villa, A. Selective Benzyl Alcohol Oxidation over Pd Catalysts. Catalysts 2018, 8, 431. [Google Scholar] [CrossRef]
- Vohs, J.M. Site requirements for the adsorption and reaction of oxygenates on metal oxide surfaces. Chem. Rev. 2013, 113, 4136–4163. [Google Scholar] [CrossRef] [PubMed]
- Moulder, J.F.; Stickle, W.F.; Sobol, P.E.; Bomben, K.D. Handbook of X-ray Photoelectron Spectroscopy; Physical Electronics Inc.: Eden Prairie, MN, USA, 1996. [Google Scholar]
- Shirley, D.A. High-Resolution X-ray Photoemission Spectrum of the Valence Bands of Gold. Phys. Rev. B 1972, 5, 4709–4714. [Google Scholar] [CrossRef]

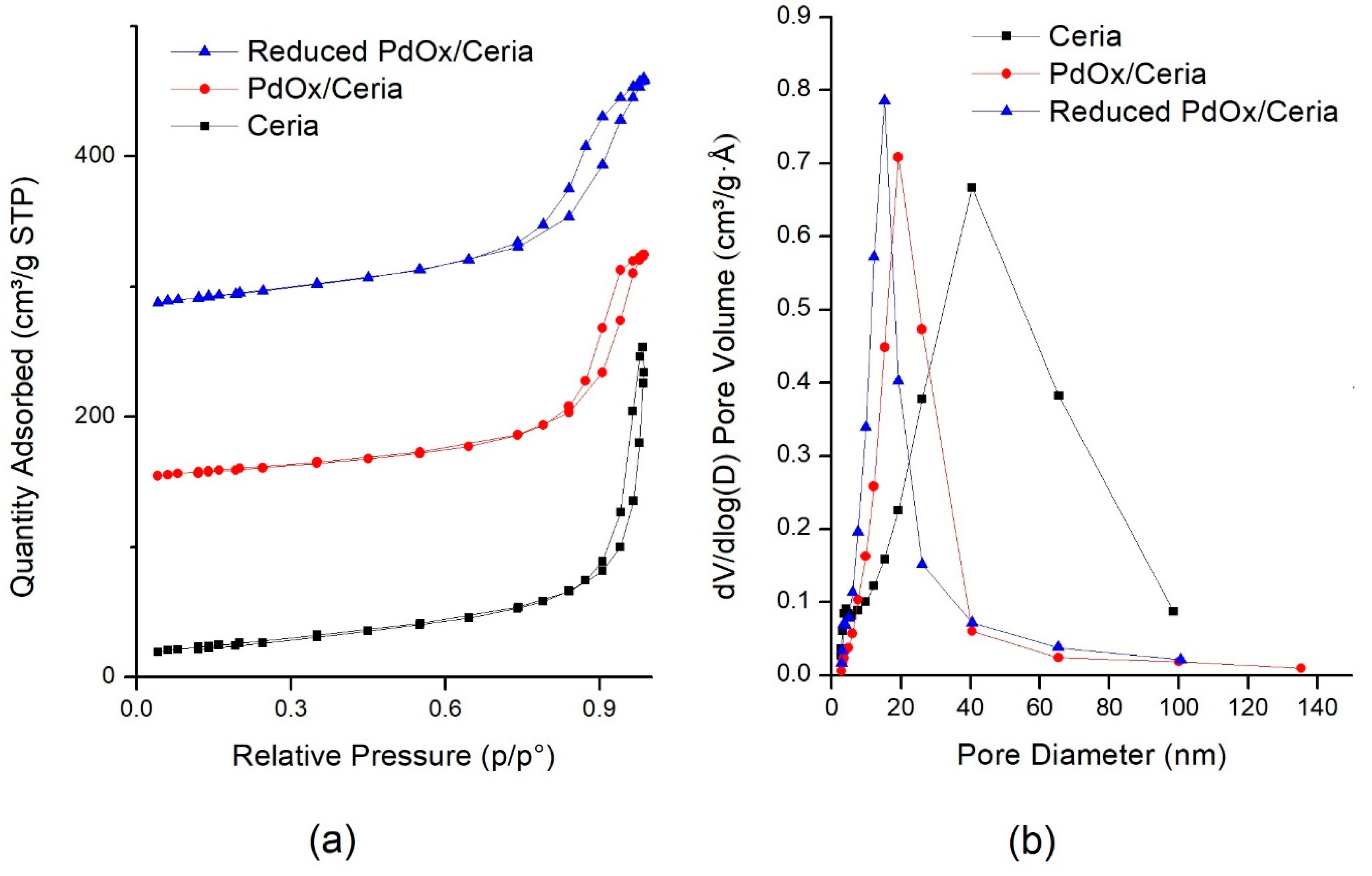
| Sample | Surface Area (m2 g−1) | Total Pore Volume (cm3 g−1) | Average Pore Diameter (nm) | CeO2 Crystallite Size 1 (nm) | CeO2 Cell Size, a 1 (Å) |
|---|---|---|---|---|---|
| CeO2-NR | 116 | 0.40 | 19 | 23.3 | 5.419 |
| PdOx/CeO2-NR | 88 | 0.30 | 16 | 21.8 | 5.419 |
| PdOx/CeO2-NR-Red | 93 | 0.32 | 12 | 22.4 | 5.414 |
| Catalyst | Pd0 % | PdO % | PdOx (x > 1) % | Pd %/(Pd + Ce) | Ce3+/Ce4+ |
|---|---|---|---|---|---|
| CeO2-NR | - | - | - | - | 0.24 |
| PdOx/CeO2-NR | 0 | 10 | 90 | 0.13 | 0.24 |
| PdOx/CeO2-NR-Red | 36.5 | 0 | 63.5 | 0.06 | 0.22 |
| Exp. | Catalyst | Catalyst Mass (mg) | Solvent | T (°C) | BnOH Conversion (%) 1 | PhCHO Selectivity (%) 1 |
|---|---|---|---|---|---|---|
| 1 | CeO2-NR | 32 | toluene | 111 | 0 | - |
| 2 | CeO2-NR | 32 | ethanol | 78 | 0 | - |
| 3 | PdOx/CeO2-NR | 32 | toluene | 50 | 9 | 100 |
| 4 | PdOx/CeO2-NR | 32 | toluene | 65 | 15 | 100 |
| 5 | PdOx/CeO2-NR | 32 | toluene | 100 | 29 | 100 |
| 6 | PdOx/CeO2-NR | 32 | toluene | 111 | 35 | 100 |
| 7 | PdOx/CeO2-NR | 32 | AcCN | 65 | 6 | 100 |
| 8 | PdOx/CeO2-NR | 32 | AcCN | 82 | 14 | 100 |
| 9 | PdOx/CeO2-NR | 32 | ethanol | 65 | 50 | >97 |
| 10 | PdOx/CeO2-NR | 32 | ethanol | 78 | 93 | 96 |
| 10(II) 2 | PdOx/CeO2-NR | 32 | ethanol | 78 | 73 | 96 |
| 10(III) 3 | PdOx/CeO2-NR | 32 | ethanol | 78 | 54 | 97 |
| 11 | PdOx/CeO2-NR | 32 | methanol | 65 | 41 | >97 |
| 12 | PdOx/CeO2-NR | 32 | THF | 66 | <3 | - |
| 13 | PdOx/CeO2-NR-Red | 32 | toluene | 111 | 25 | 100 |
| 14 | PdOx/CeO2-NR-Red | 32 | ethanol | 78 | 13 | 100 |
| 15 4 | PdOx/CeO2-NR | 16 | ethanol | 78 | 28 | >97 |
| Solvent | Proticity | Dielectric Constant | Boiling Point (°C) |
|---|---|---|---|
| toluene | aprotic | 2.4 | 111 |
| THF | aprotic | 7.52 | 66 |
| AcCN | aprotic | 37.5 | 82 |
| ethanol | protic | 24.5 | 78 |
| methanol | protic | 32.7 | 65 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Moeini, S.S.; Battocchio, C.; Casciardi, S.; Luisetto, I.; Lupattelli, P.; Tofani, D.; Tuti, S. Oxidized Palladium Supported on Ceria Nanorods for Catalytic Aerobic Oxidation of Benzyl Alcohol to Benzaldehyde in Protic Solvents. Catalysts 2019, 9, 847. https://doi.org/10.3390/catal9100847
Moeini SS, Battocchio C, Casciardi S, Luisetto I, Lupattelli P, Tofani D, Tuti S. Oxidized Palladium Supported on Ceria Nanorods for Catalytic Aerobic Oxidation of Benzyl Alcohol to Benzaldehyde in Protic Solvents. Catalysts. 2019; 9(10):847. https://doi.org/10.3390/catal9100847
Chicago/Turabian StyleMoeini, Seyed Sepehr, Chiara Battocchio, Stefano Casciardi, Igor Luisetto, Paolo Lupattelli, Daniela Tofani, and Simonetta Tuti. 2019. "Oxidized Palladium Supported on Ceria Nanorods for Catalytic Aerobic Oxidation of Benzyl Alcohol to Benzaldehyde in Protic Solvents" Catalysts 9, no. 10: 847. https://doi.org/10.3390/catal9100847
APA StyleMoeini, S. S., Battocchio, C., Casciardi, S., Luisetto, I., Lupattelli, P., Tofani, D., & Tuti, S. (2019). Oxidized Palladium Supported on Ceria Nanorods for Catalytic Aerobic Oxidation of Benzyl Alcohol to Benzaldehyde in Protic Solvents. Catalysts, 9(10), 847. https://doi.org/10.3390/catal9100847









