Biosynthesis of Nylon 12 Monomer, ω-Aminododecanoic Acid Using Artificial Self-Sufficient P450, AlkJ and ω-TA
Abstract
1. Introduction
2. Results and Discussion
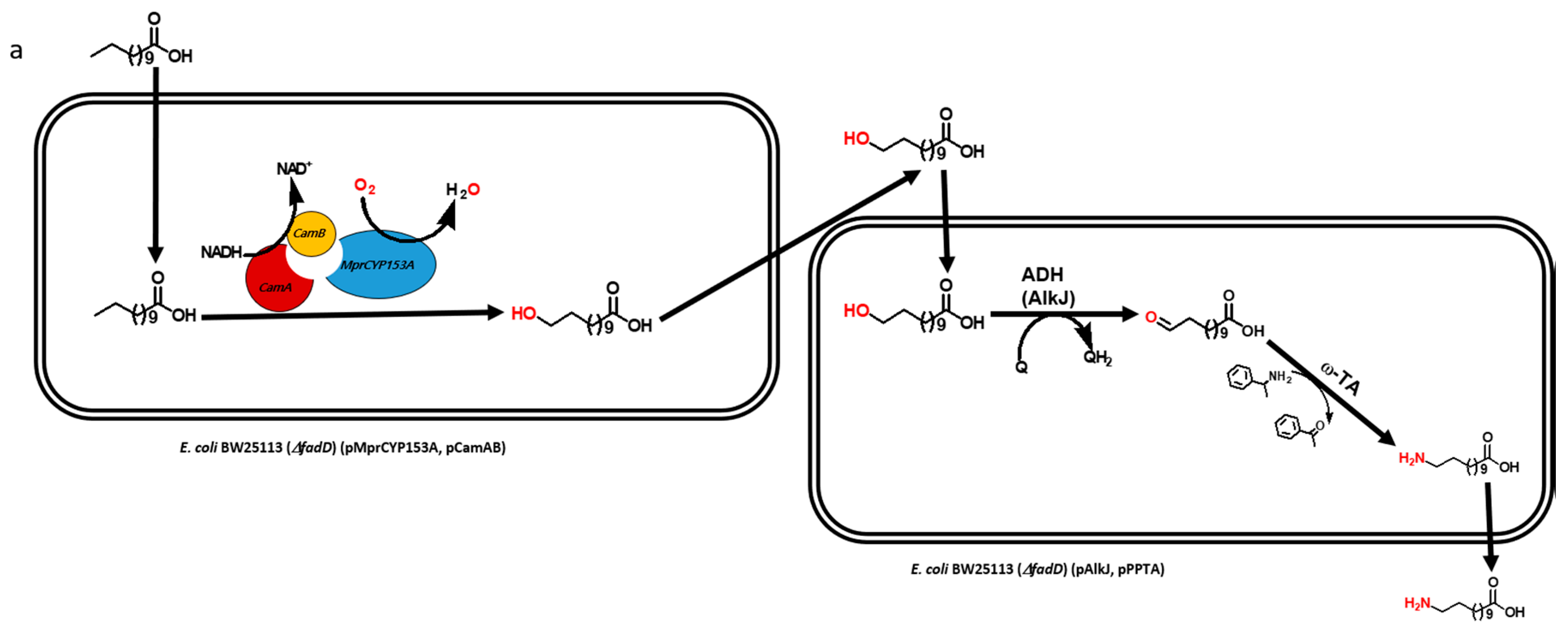
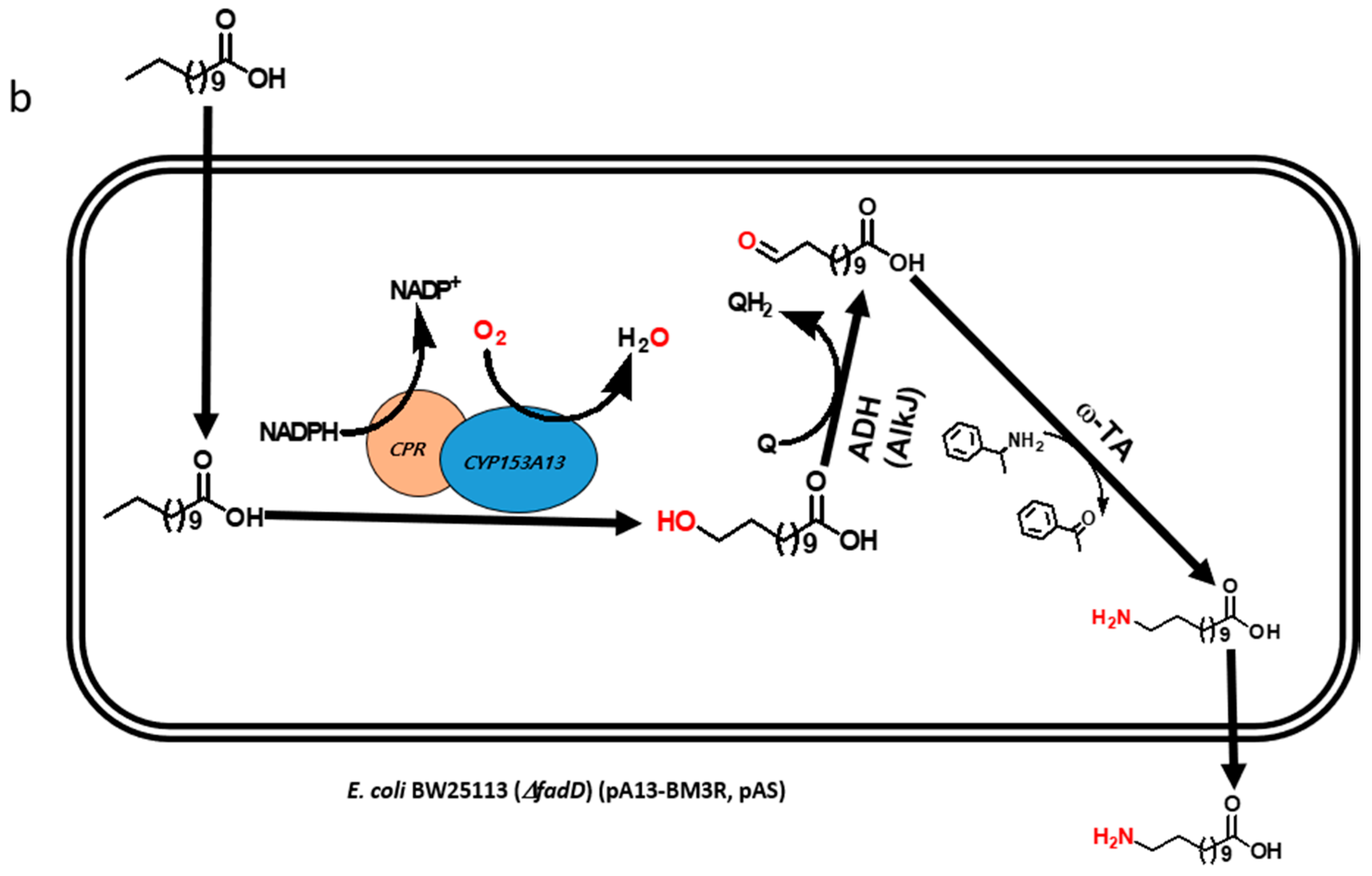
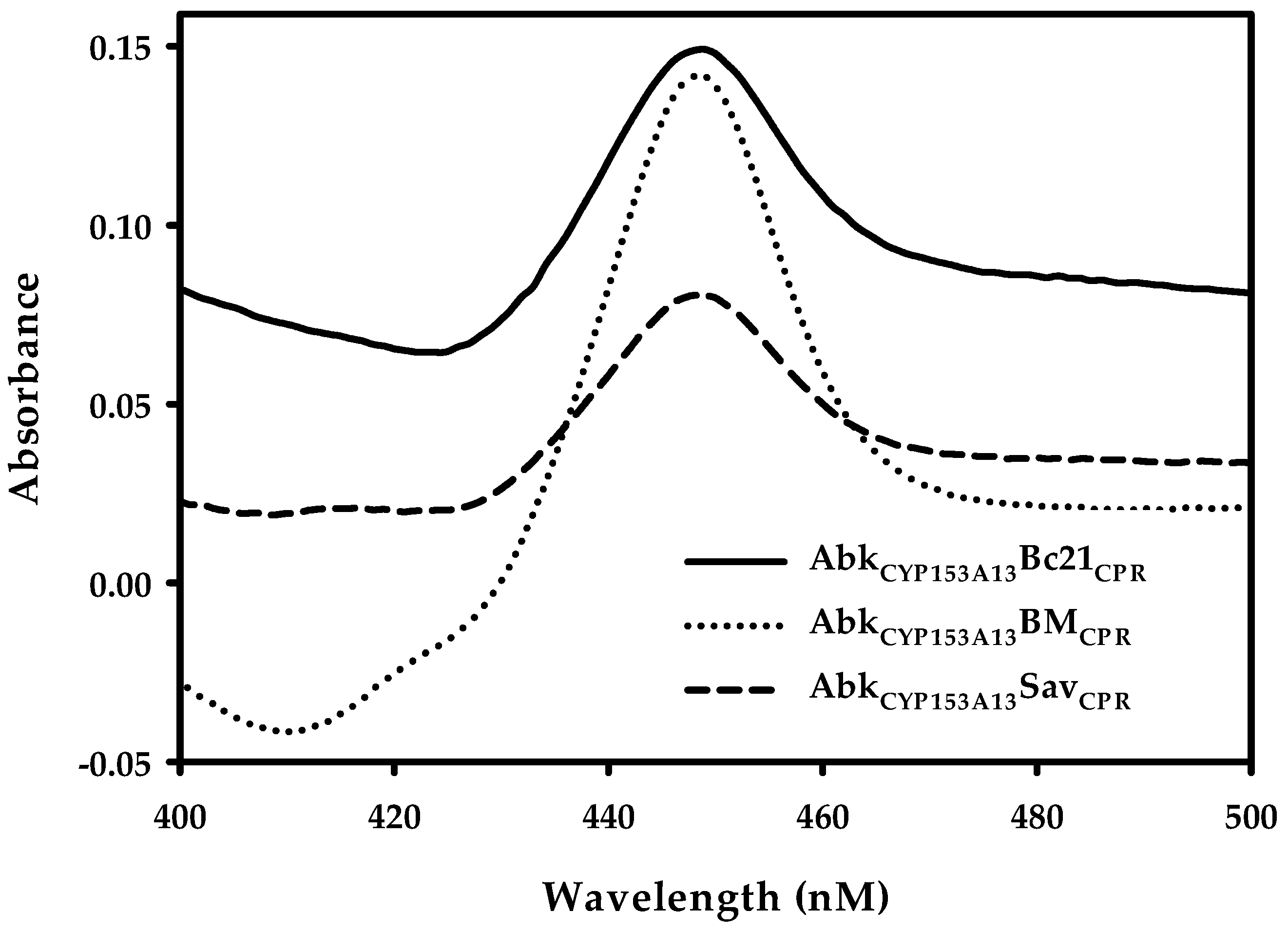
2.1. Construction and Expression of Artificial Self-Sufficient P450s (ArtssP450s)
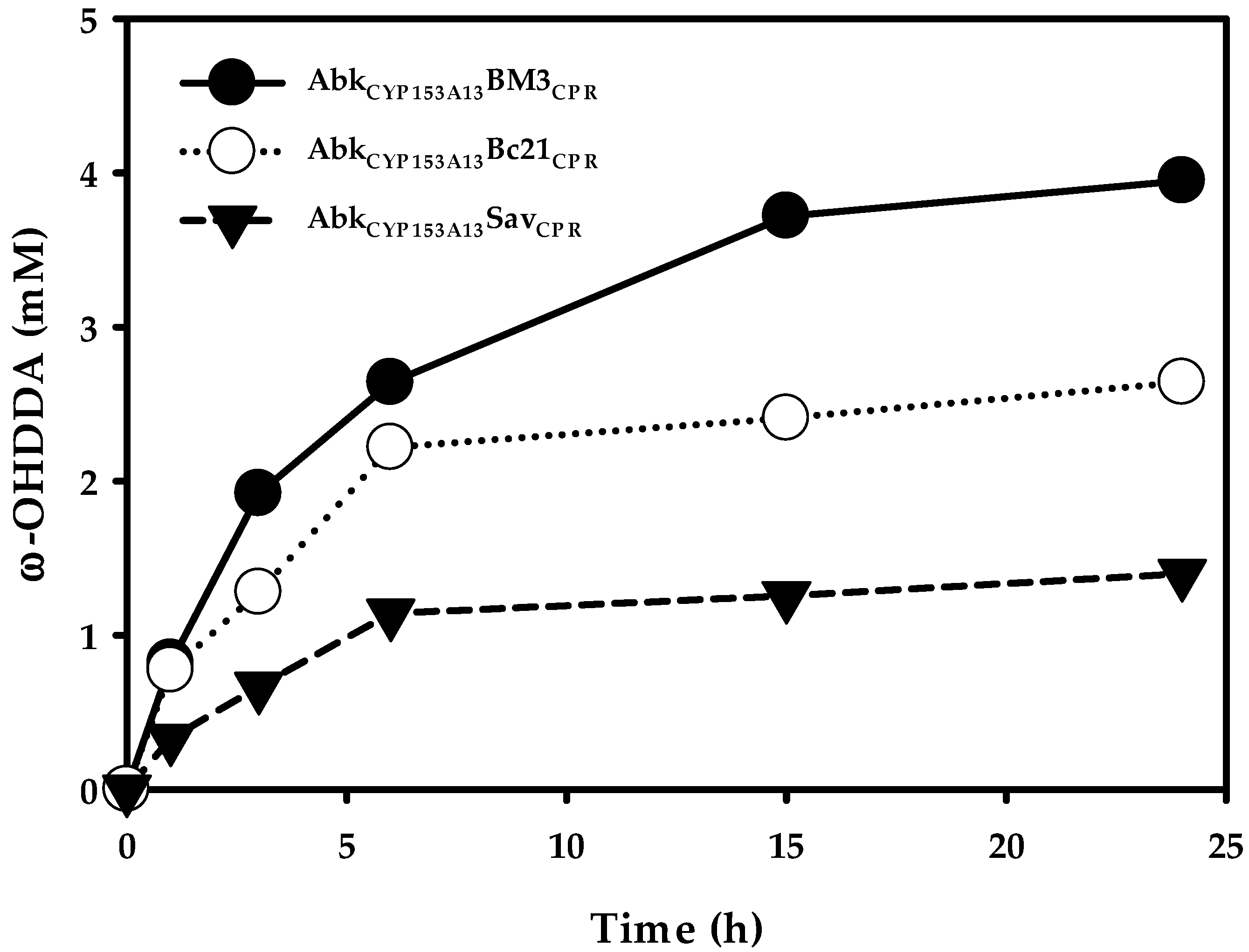
2.2. Production of ω-OHDDA Using ArtssP450s
2.3. Biosysnthesis of ω-AmDDA in One-Pot Reaction
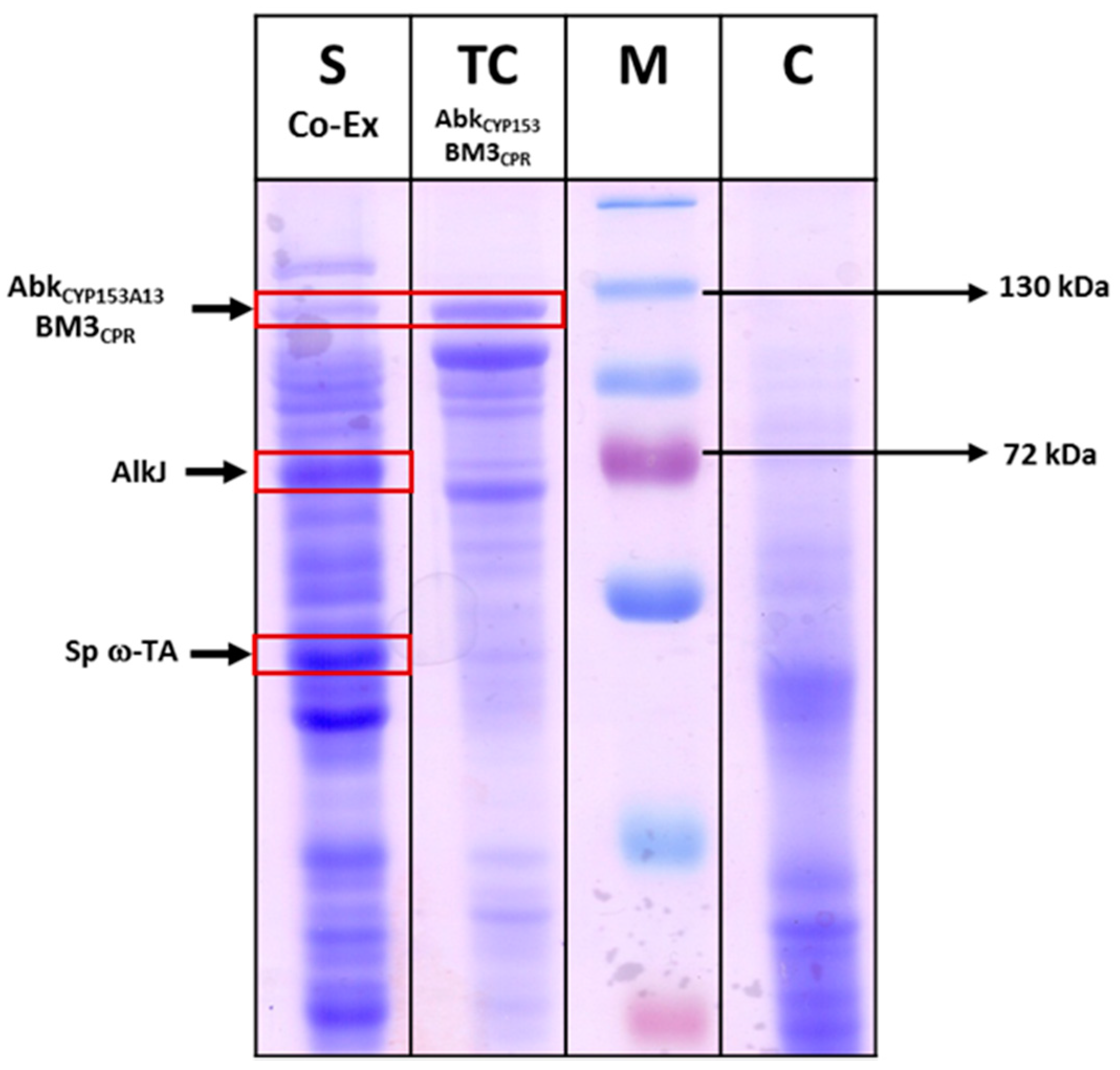
2.3.1. Co-expression of AbkCYP153A13BM3CPR with AlkJ and Sp ω-TA
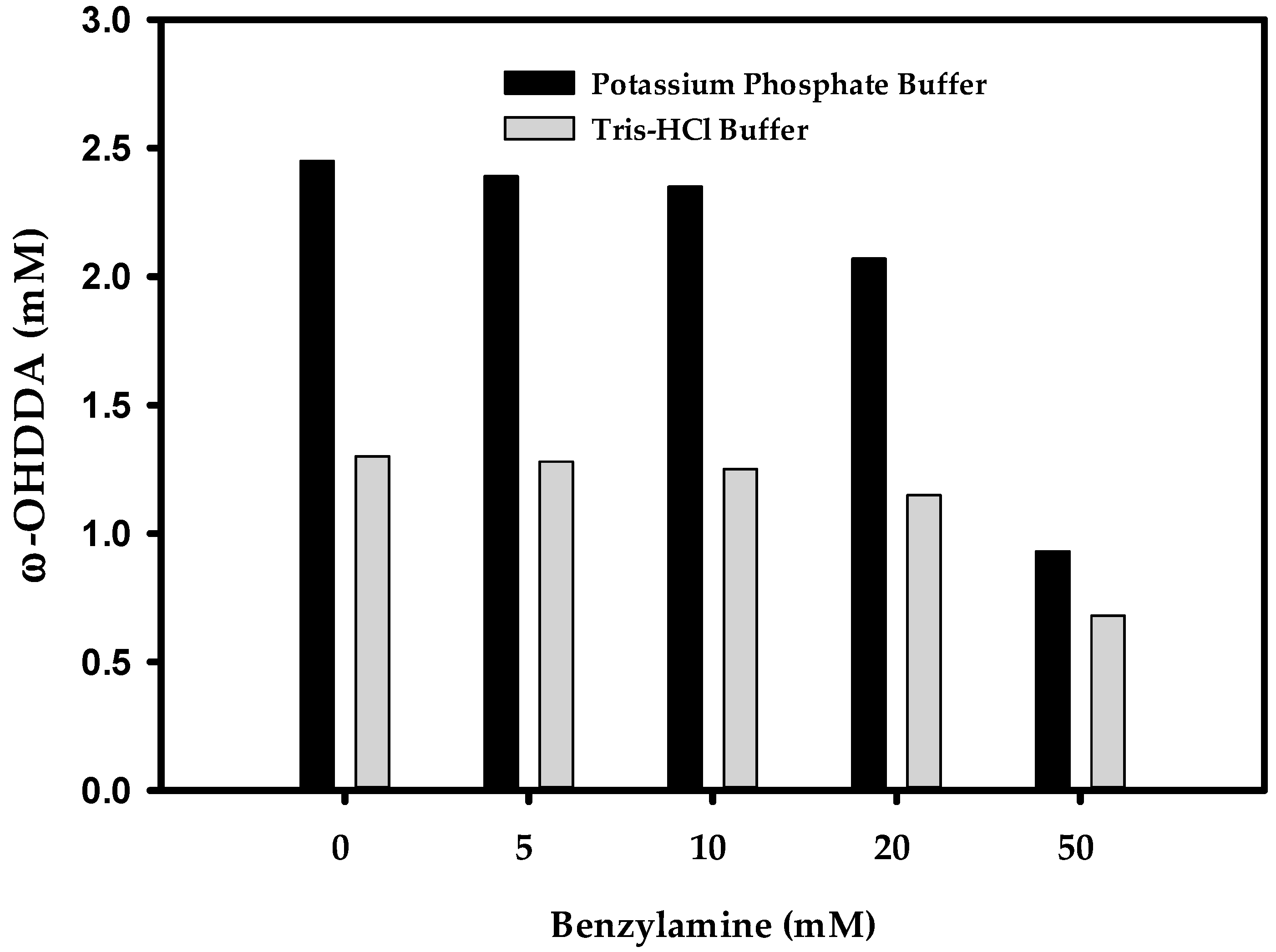
2.3.2. Effect of Benzylamine (BA) on ω-OHDDA Productivity
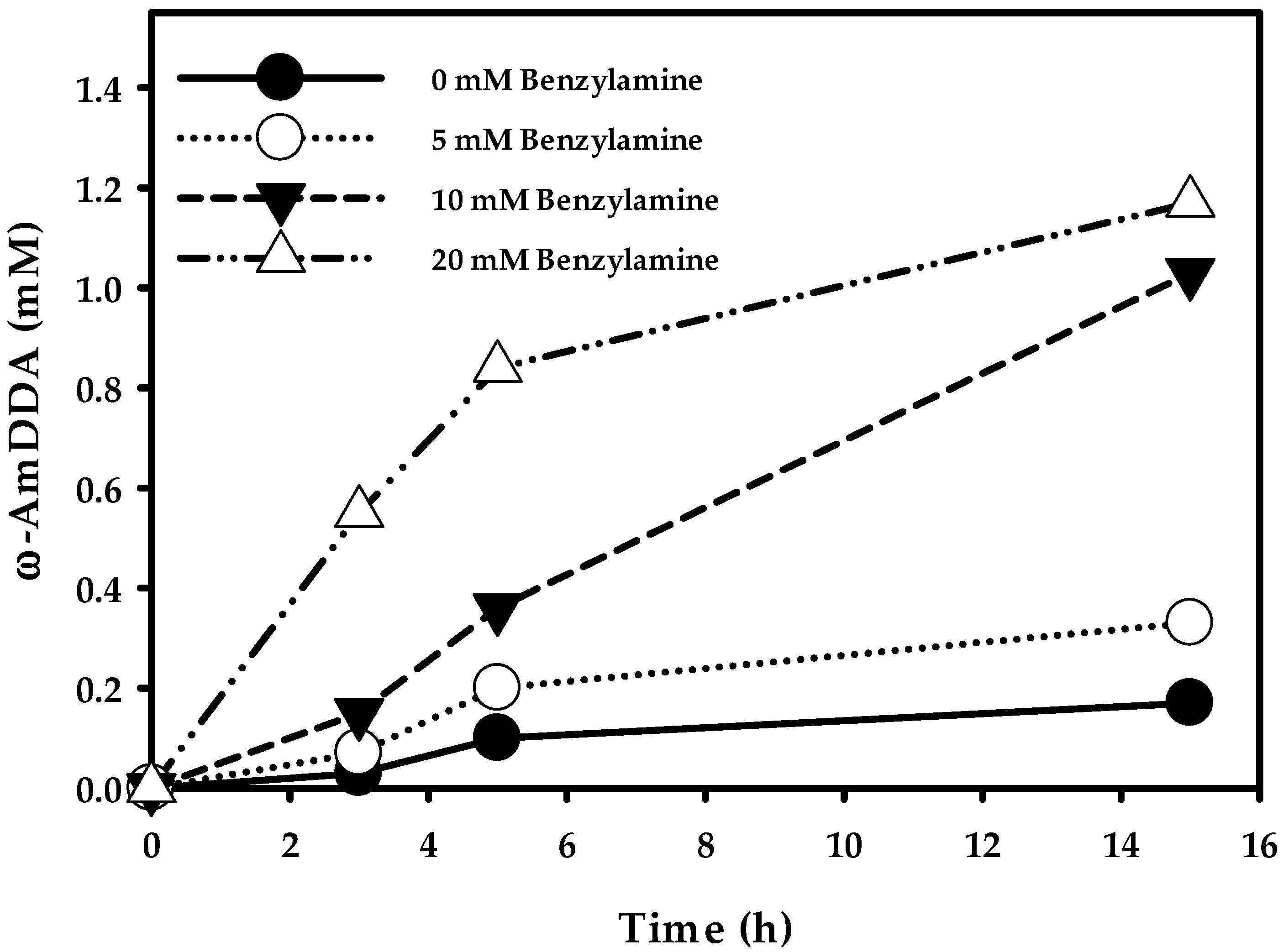
2.3.3. Optimization of ω-AmDDA Biosynthesis
3. Materials and Methods
3.1. Chemicals and Media
3.2. Artificial Self-Sufficient P450 Construction and Gene Manipulation
3.3. Expression of Enzymes
3.4. CO-Binding Assay and Gel Electrophoresis
3.5. Biotransformation
3.6. Product Identification and Quantification
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Steen, E.J.; Kang, Y.; Bokinsky, G.; Hu, Z.; Schirmer, A.; McClure, A.; del Cardayre, S.B.; Keasling, J.D. Microbial production of fatty-acid-derived fuels and chemicals from plant biomass. Nature 2010, 463, 559–562. [Google Scholar] [CrossRef] [PubMed]
- Chung, H.; Yang, J.E.; Ha, J.Y.; Chae, T.U.; Shin, J.H.; Gustavsson, M.; Lee, S.Y. Bio-based production of monomers and polymers by metabolically engineered microorganisms. Curr. Opin. Biotechnol. 2015, 36, 73. [Google Scholar] [CrossRef] [PubMed]
- Zorn, K.; Oroz-Guinea, I.; Brundiek, H.; Bornscheuer, U.T. Engineering and application of enzymes for lipid modification, an update. Prog. Lipid Res. 2016, 63, 153–164. [Google Scholar] [CrossRef] [PubMed]
- Ahsan, M.; Sung, S.; Jeon, H.; Patil, M.D.; Chung, T.; Yun, H. Biosynthesis of medium- to long-chain α,ω-diols from free fatty acids using CYP153A monooxygenase, carboxylic acid reductase, and E. coli endogenous aldehyde reductases. Catalysts 2018, 8, 4. [Google Scholar] [CrossRef]
- Metzger, J.O.; Bornscheuer, U. Lipids as renewable resources: Current state of chemical and biotechnological conversion and diversification. Appl. Microbiol. Biotechnol. 2006, 71, 13–22. [Google Scholar] [CrossRef] [PubMed]
- Lu, W.; Ness, J.E.; Xie, W.; Zhang, X.; Minshull, J.; Gross, R.A. Biosynthesis of monomers for plastics from renewable oils. J. Am. Chem. Soc. 2010, 132, 15451–15455. [Google Scholar] [CrossRef] [PubMed]
- Durairaj, P.; Malla, S.; Nadarajan, S.P.; Lee, P.-G.; Jung, E.; Park, H.H.; Kim, B.-G.; Yun, H. Fungal cytochrome P450 monooxygenases of Fusarium oxysporum for the synthesis of ω-hydroxy fatty acids in engineered Saccharomyces cerevisiae. Microb. Cell Fact. 2015, 14, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Song, J.-W.; Lee, J.-H.; Bornscheuer, U.T.; Park, J.-B. Microbial Synthesis of Medium-Chain α,ω-Dicarboxylic Acids and ω-Aminocarboxylic Acids from Renewable Long-Chain Fatty Acids. Adv. Synth. Catal. 2014, 356, 1782–1788. [Google Scholar] [CrossRef]
- Ladkau, N.; Assmann, M.; Schrewe, M.; Julsing, M.K.; Schmid, A.; Bühler, B. Efficient production of the Nylon 12 monomer ω-aminododecanoic acid methyl ester from renewable dodecanoic acid methyl ester with engineered Escherichia coli. Metab. Eng. 2016, 36, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Ahsan, M.M.; Jeon, H.; Nadarajan, S.P.; Chung, T.; Yoo, H.-W.; Kim, B.-G.; Patil, M.D.; Yun, H. Biosynthesis of the Nylon 12 monomer, ω-aminododecanoic acid with novel CYP153A, AlkJ, and ω-TA enzymes. Biotechnol. J. 2018, 13, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Jung, E.; Park, B.G.; Ahsan, M.M.; Kim, J.; Yun, H.; Choi, K.Y.; Kim, B.G. Production of ω-hydroxy palmitic acid using CYP153A35 and comparison of cytochrome P450 electron transfer system in vivo. Appl. Microbiol. Biotechnol. 2016, 100, 10375–10384. [Google Scholar] [CrossRef] [PubMed]
- Jung, E.; Park, B.G.; Yoo, H.-W.; Kim, J.; Choi, K.-Y.; Kim, B.-G. Semi-rational engineering of CYP153A35 to enhance ω-hydroxylation activity toward palmitic acid. Appl. Microbiol. Biotechnol. 2018, 102, 269–277. [Google Scholar] [CrossRef] [PubMed]
- Patil, M.D.; Grogan, G.; Yun, H. Biocatalyzed C-C bond formation for the production of alkaloids. ChemCatChem 2018. [Google Scholar] [CrossRef]
- Patil, M.D.; Dev, M.J.; Shinde, A.S.; Bhilare, K.D.; Patel, G.; Chisti, Y.; Banerjee, U.C. Surfactant-mediated permeabilization of Pseudomonas putida KT2440 and use of the immobilized permeabilized cells in biotransformation. Process Biochem. 2017, 63, 113–121. [Google Scholar] [CrossRef]
- Lin, B.; Tao, Y. Whole-cell biocatalysts by design. Microb. Cell Fact. 2017, 16, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Patil, M.D.; Grogan, G.; Bommarius, A.; Yun, H. Recent advances in ω-transaminase-mediated biocatalysis for the enantioselective synthesis of chiral amines. Catalysts 2018, 8, 254. [Google Scholar] [CrossRef]
- Honda Malca, S.; Scheps, D.; Kuhnel, L.; Venegas-Venegas, E.; Seifert, A.; Nestl, B.M.; Hauer, B. Bacterial CYP153A monooxygenases for the synthesis of omega-hydroxylated fatty acids. Chem. Commun. 2012, 48, 5115–5117. [Google Scholar] [CrossRef] [PubMed]
- Scheps, D.; Honda Malca, S.; Richter, S.M.; Marisch, K.; Nestl, B.M.; Hauer, B. Synthesis of ω-hydroxydodecanoic acid based on an engineered CYP153A fusion construct. Microb. Biotechnol. 2013, 6, 694–707. [Google Scholar] [PubMed]
- Chowdhary, P.K.; Alemseghed, M.; Haines, D.C. Cloning, expression and characterization of a fast self-sufficient P450: CYP102A5 from Bacillus cereus. Arch. Biochem. Biophys. 2007, 468, 32–43. [Google Scholar] [CrossRef] [PubMed]
- Choi, K.Y.; Jung, E.; Yun, H.; Yang, Y.H.; Kim, B.G. Engineering class I cytochrome P450 by gene fusion with NADPH-dependent reductase and S. avermitilis host development for daidzein biotransformation. Appl. Microbiol. Biotechnol. 2014, 98, 8191. [Google Scholar] [CrossRef] [PubMed]
- Hausjell, J.; Halbwirth, H.; Spadiut, O. Recombinant production of eukaryotic cytochrome P450s in microbial cell factories. Biosci. Rep. 2018, 38, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Mathew, S.; Jeong, S.-S.; Chung, T.; Lee, S.-H.; Yun, H. Asymmetric synthesis of aromatic β-amino acids using ω-transaminase: Optimizing the lipase concentration to obtain thermodynamically unstable β-keto acids. Biotechnol. J. 2016, 11, 185–190. [Google Scholar] [CrossRef] [PubMed]
- Tavanti, M.; Mangas-Sanchez, J.; Montgomery, S.L.; Thompson, M.P.; Turner, N.J. A biocatalytic cascade for the amination of unfunctionalised cycloalkanes. Org. Biomol. Chem. 2017, 15, 9790–9793. [Google Scholar] [CrossRef] [PubMed]
- Kirmair, L.; Skerra, A. Biochemical analysis of recombinant AlkJ from Pseudomonas putida reveals a membrane-associated, FAD-dependent dehydrogenase suitable for the biosynthetic production of aliphatic aldehydes. Appl. Environ. Microbiol. 2014, 80, 2468–2477. [Google Scholar] [CrossRef] [PubMed]
- Tan, Z.; Black, W.; Yoon, J.M.; Shanks, J.V.; Jarboe, L.R. Improving Escherichia coli membrane integrity and fatty acid production by expression tuning of FadL and OmpF. Microb. Cell Fact. 2017, 16, 38. [Google Scholar] [CrossRef] [PubMed]
- Jeon, E.Y.; Song, J.W.; Cha, H.J.; Lee, S.M.; Lee, J.; Park, J.B. Intracellular transformation rates of fatty acids are influenced by expression of the fatty acid transporter FadL in Escherichia coli cell membrane. J. Biotechnol. 2018, 281, 161–167. [Google Scholar] [CrossRef] [PubMed]
- Rosano, G.L.; Ceccarelli, E.A. Recombinant protein expression in Escherichia coli: Advances and challenges. Front. Microbiol. 2014, 5, 172. [Google Scholar] [CrossRef] [PubMed]
- Janßen, H.; Steinbüchel, A. Fatty acid synthesis in escherichia coli and its applications towards the production of fatty acid based biofuels. Biotechnol. Biofuels 2014, 7, 7. [Google Scholar] [CrossRef] [PubMed]
- Hanahan, D. Studies on transformation of Escherichia coli with plasmids. J. Mol. Biol. 1983, 166, 557–580. [Google Scholar] [CrossRef]
- Sambrook, J. Molecular Cloning: A Laboratory Manual/joseph Sambrook, David w. Russell; Cold Spring Harbor Laboratory: Cold Spring Harbor, NY, USA, 2001. [Google Scholar]
- Bollag, D.M.; Rozycki, M.D.; Edelstein, S.J. Protein Methods, 2nd ed.; Wiley-Liss: Hoboken, NJ, USA, 1996; ISBN 978-0-471-11837-4. [Google Scholar]
| Plasmids/Strains | Description | Reference |
|---|---|---|
| Plasmids | ||
| pET24ma | P15A ori lacI T7 promoter, KmR | [10] |
| pETDuet-1 | pBR322 ori lacI T7 promoter, AmpR | Novagen |
| pA13 | pET24ma encoding CYP153A13 (AbkCYP153A13) | [11] |
| pA13-BM3R | pET24ma encoding AbkCYP153A13BM3CPR | This study |
| pA13-Bc21R | pET24ma encoding AbkCYP153A13Bc21CPR | This study |
| pA13-SavR | pET24ma encoding AbkCYP153A13SavCPR | This study |
| pAlkJ | pET24ma encoding AlkJ | [10] |
| pPPTA | pETDuet-1 encoding Sp ω-TA | [10] |
| pAS | pETDuet-1 encoding Sp ω-TA and AlkJ | This study |
| E. coli strains | ||
| DH5α | F−, endA1, glnV44, thi-1, recA1, relA1, gyrA96 deoR, supE44, Φ80dlacZΔM15 Δ(lacZYA−-argF)-U169, hsdR17(rK− mK+), λ− | [29] |
| BW25113(DE3) | rrnB3 ΔlacZ4787 hsdR514 Δ(araBAD)567 Δ(rhaBAD)568 rph-1 λ(DE3) | [11] |
| DL | BW25113(DE3) ΔfadD | [10] |
| A13 | DL carrying pA13 | This study |
| A13-BM3R | DL carrying pA13-BM3R | This study |
| A13-Bc21R | DL carrying pA13-Bc21R | This study |
| A13-BSavR | DL carrying pA13-BSavR | This study |
| AlkJ-PPTA | DL carrying pAlkJ and pPPTA | This study |
| BM-AS | DL carrying pA13-BM3R and pAS | This study |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ahsan, M.M.; Patil, M.D.; Jeon, H.; Sung, S.; Chung, T.; Yun, H. Biosynthesis of Nylon 12 Monomer, ω-Aminododecanoic Acid Using Artificial Self-Sufficient P450, AlkJ and ω-TA. Catalysts 2018, 8, 400. https://doi.org/10.3390/catal8090400
Ahsan MM, Patil MD, Jeon H, Sung S, Chung T, Yun H. Biosynthesis of Nylon 12 Monomer, ω-Aminododecanoic Acid Using Artificial Self-Sufficient P450, AlkJ and ω-TA. Catalysts. 2018; 8(9):400. https://doi.org/10.3390/catal8090400
Chicago/Turabian StyleAhsan, Md Murshidul, Mahesh D. Patil, Hyunwoo Jeon, Sihyong Sung, Taeowan Chung, and Hyungdon Yun. 2018. "Biosynthesis of Nylon 12 Monomer, ω-Aminododecanoic Acid Using Artificial Self-Sufficient P450, AlkJ and ω-TA" Catalysts 8, no. 9: 400. https://doi.org/10.3390/catal8090400
APA StyleAhsan, M. M., Patil, M. D., Jeon, H., Sung, S., Chung, T., & Yun, H. (2018). Biosynthesis of Nylon 12 Monomer, ω-Aminododecanoic Acid Using Artificial Self-Sufficient P450, AlkJ and ω-TA. Catalysts, 8(9), 400. https://doi.org/10.3390/catal8090400






