Design of Specific Acid-Base-Properties in CeO2-ZrO2-Mixed Oxides via Templating and Au Modification
Abstract
1. Introduction
2. Results and Discussion
2.1. Catalyst Characterization
2.1.1. Atomic Absorption Spectroscopy (AAS)
2.1.2. Brunauer-Emmet-Teller Surface Analysis (N2-BET)
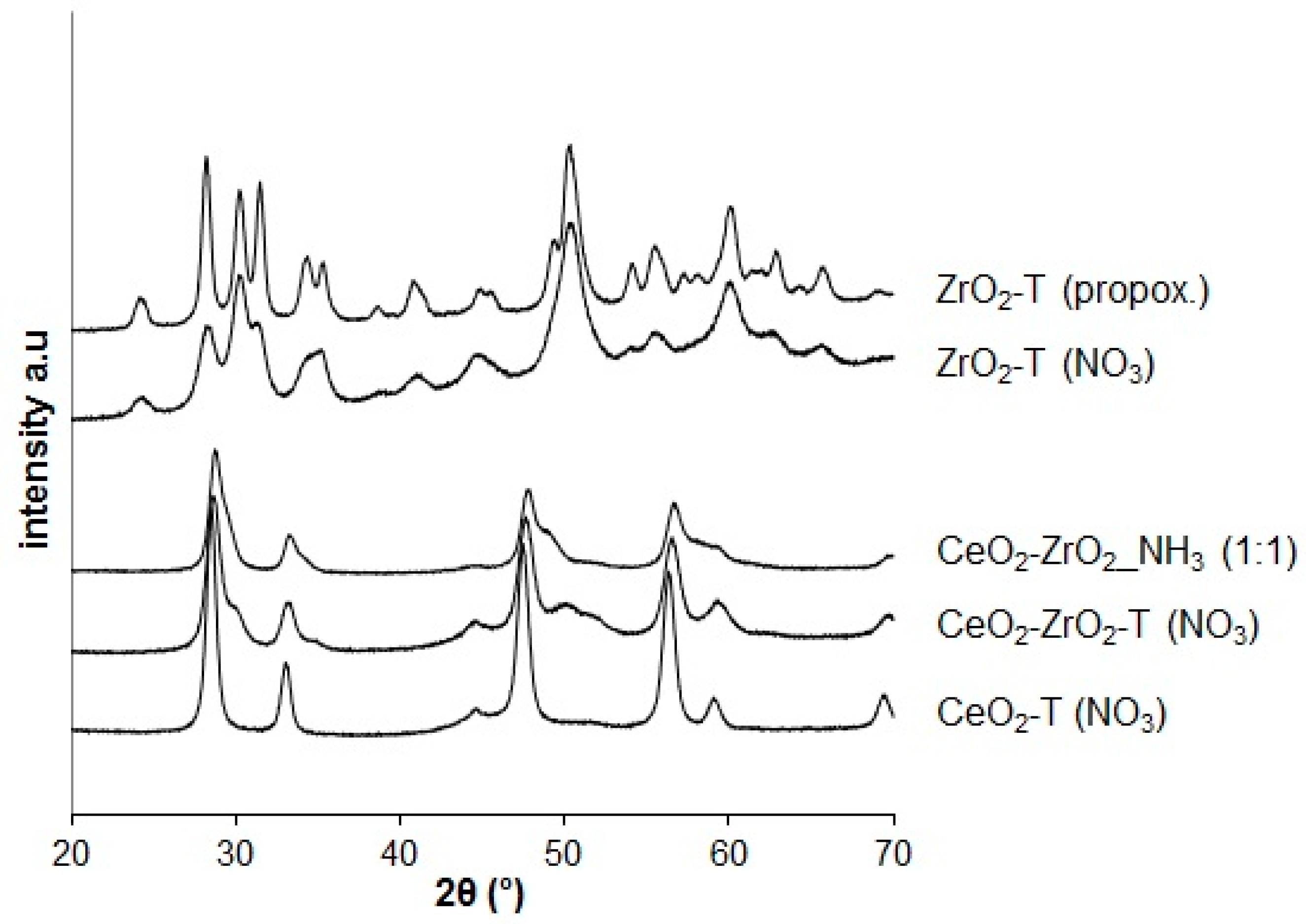
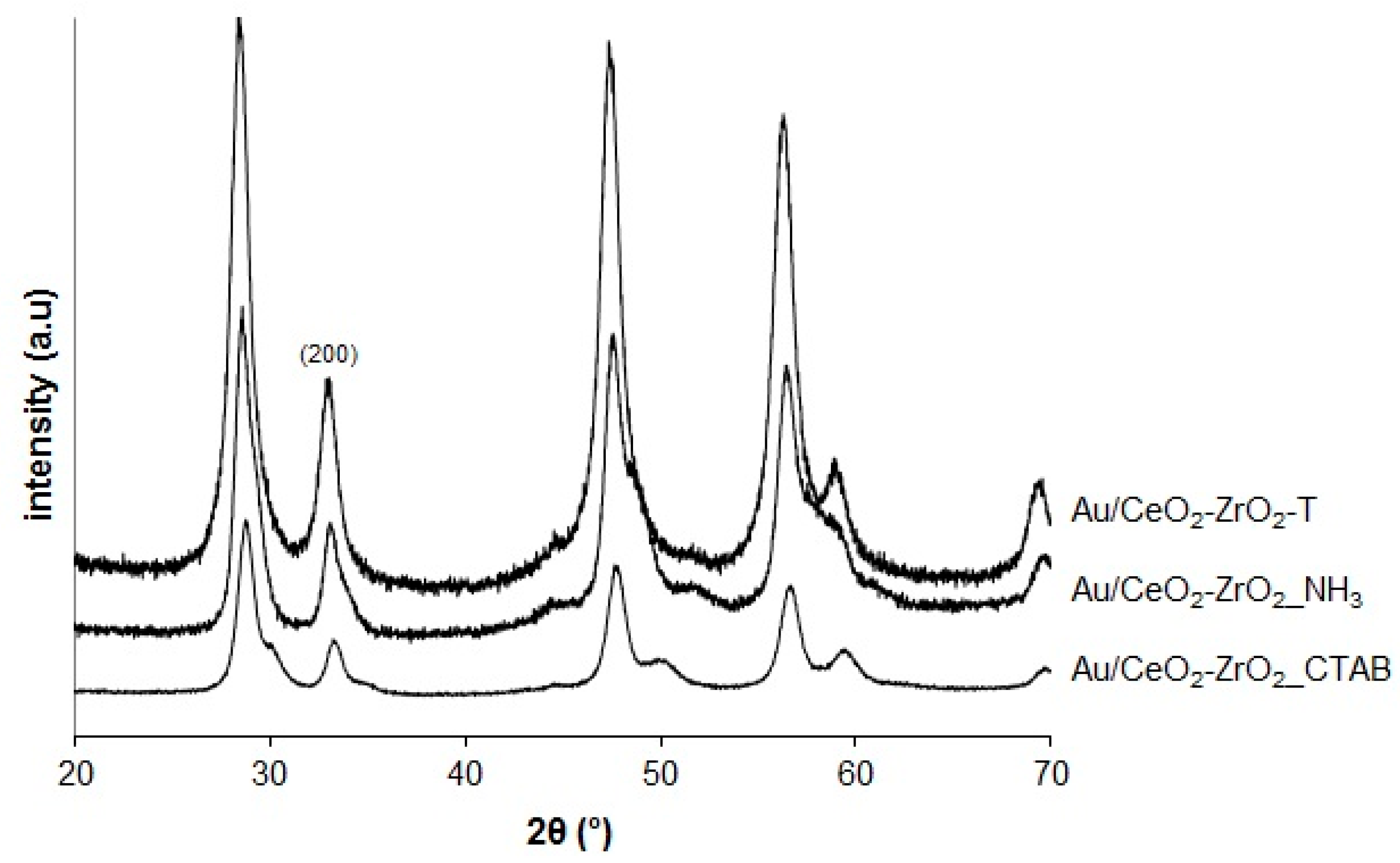
2.1.3. X-Ray Diffraction (XRD)
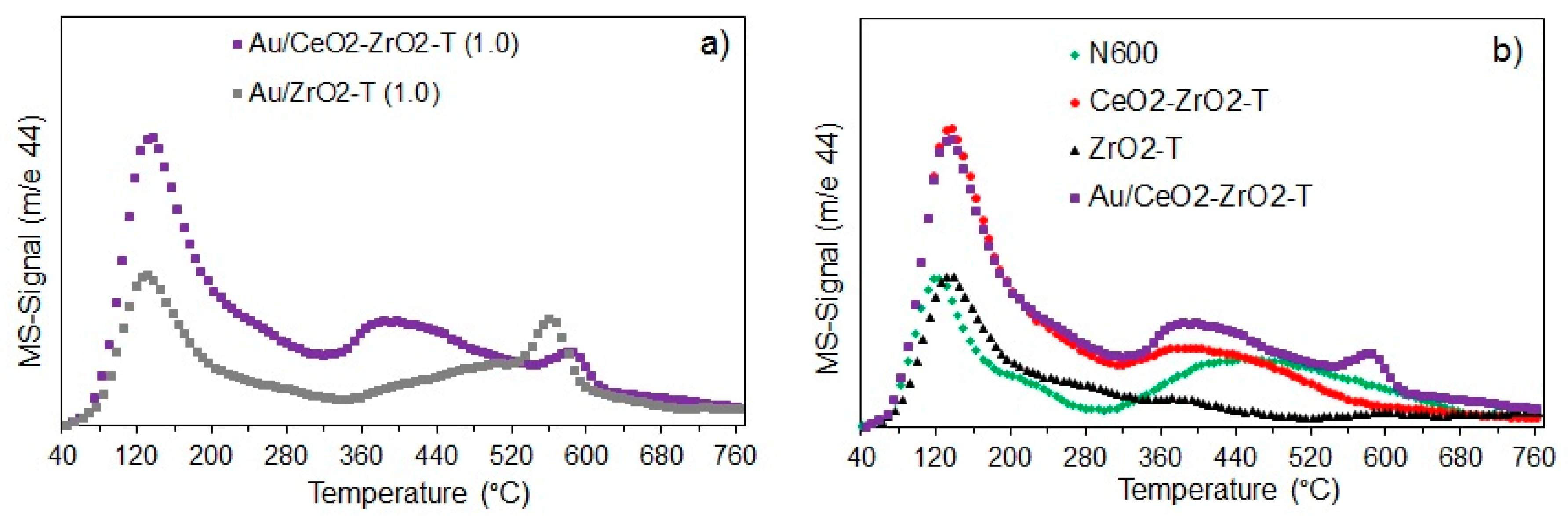
2.1.4. Temperature Programmed Desorption (TPD)
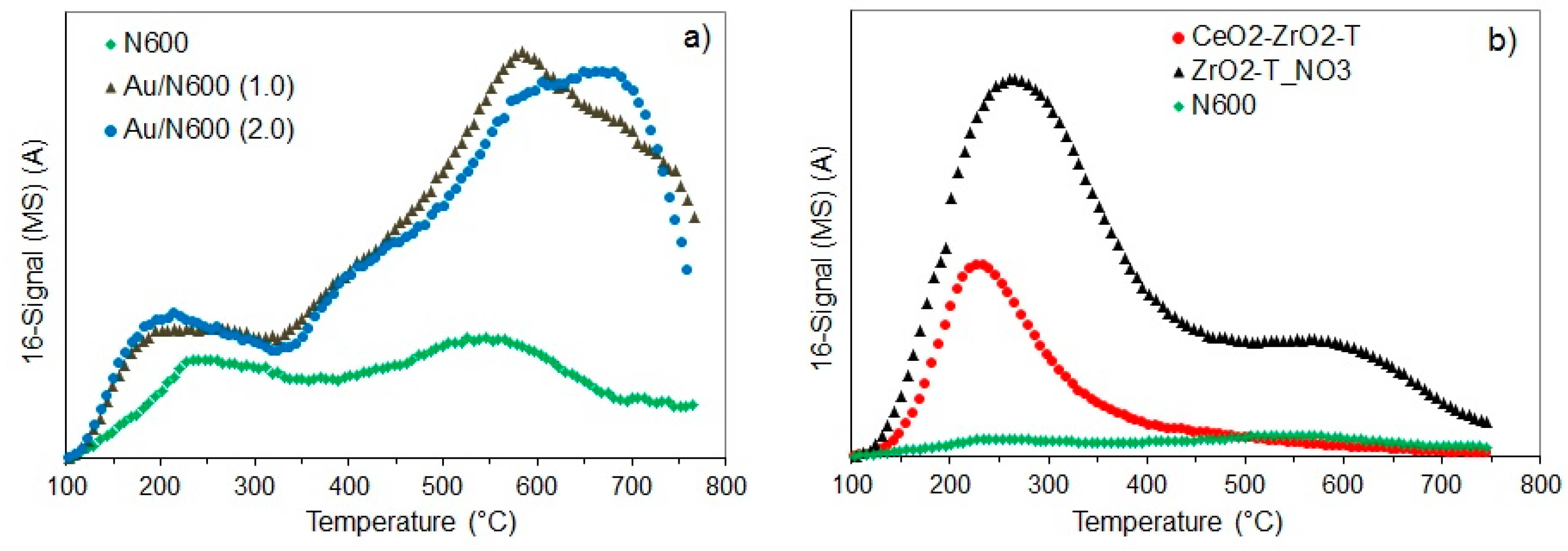
NH3-TPD
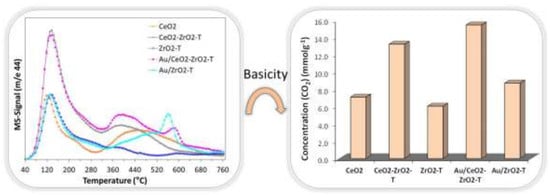
CO2-TPD
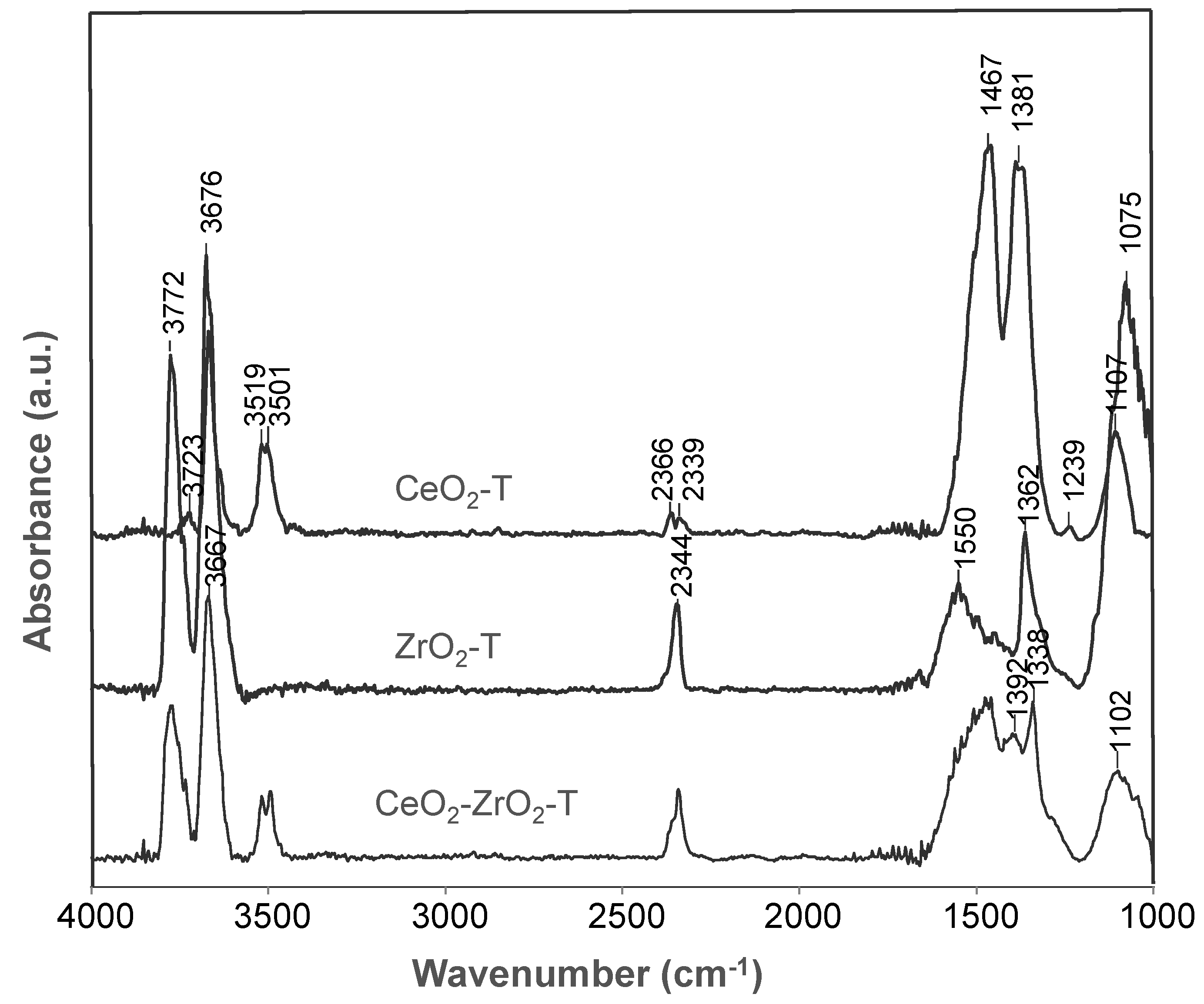
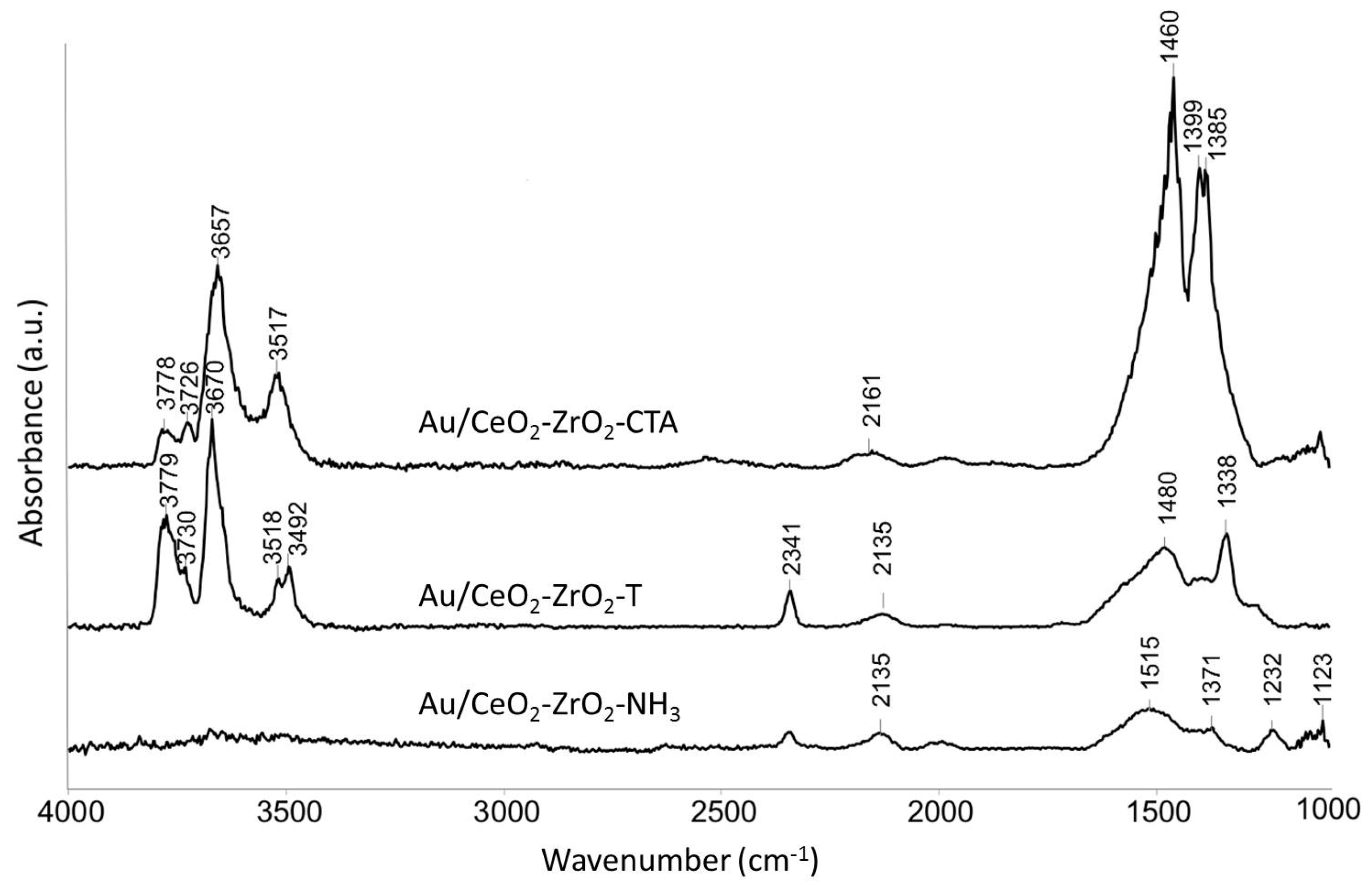
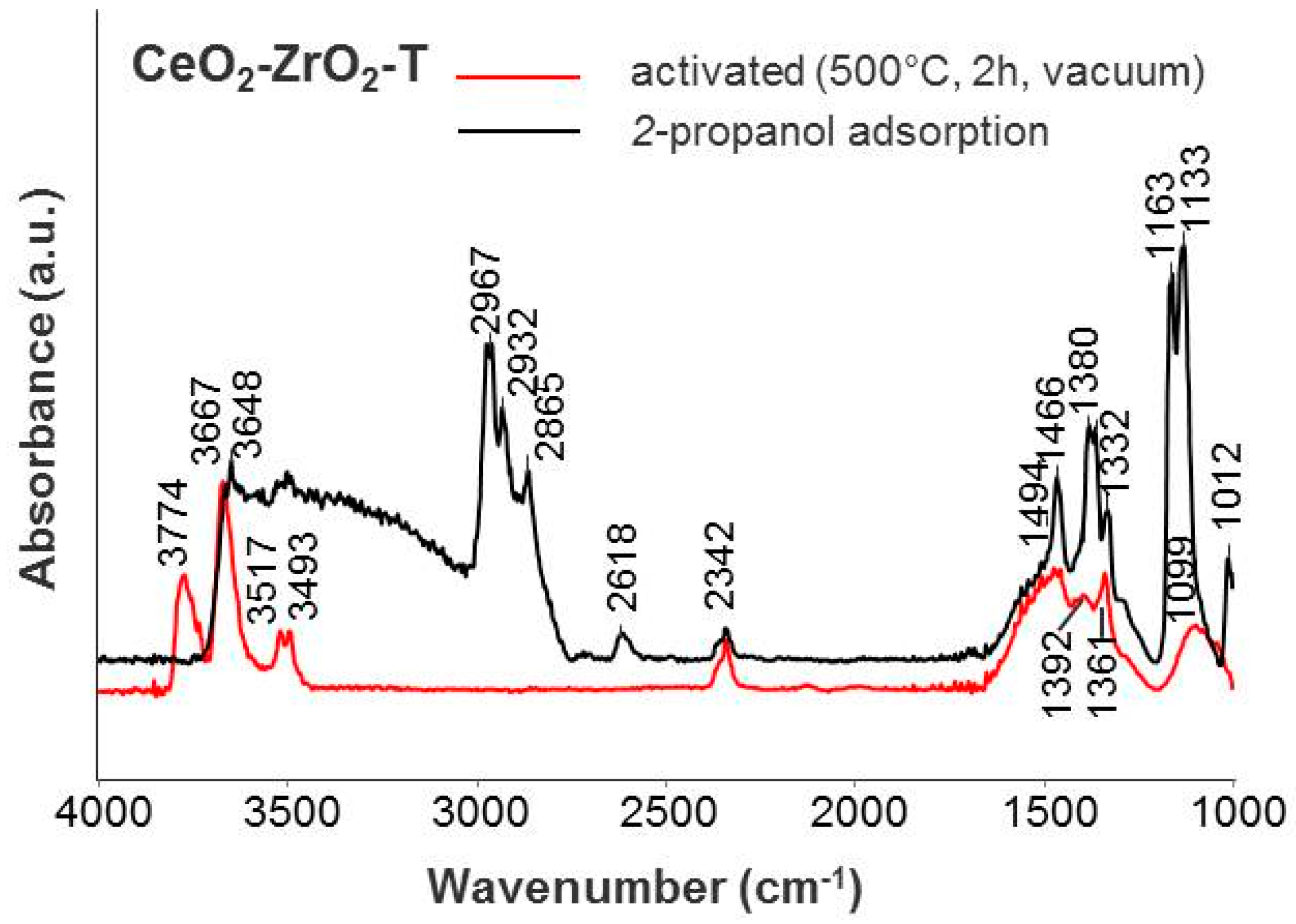
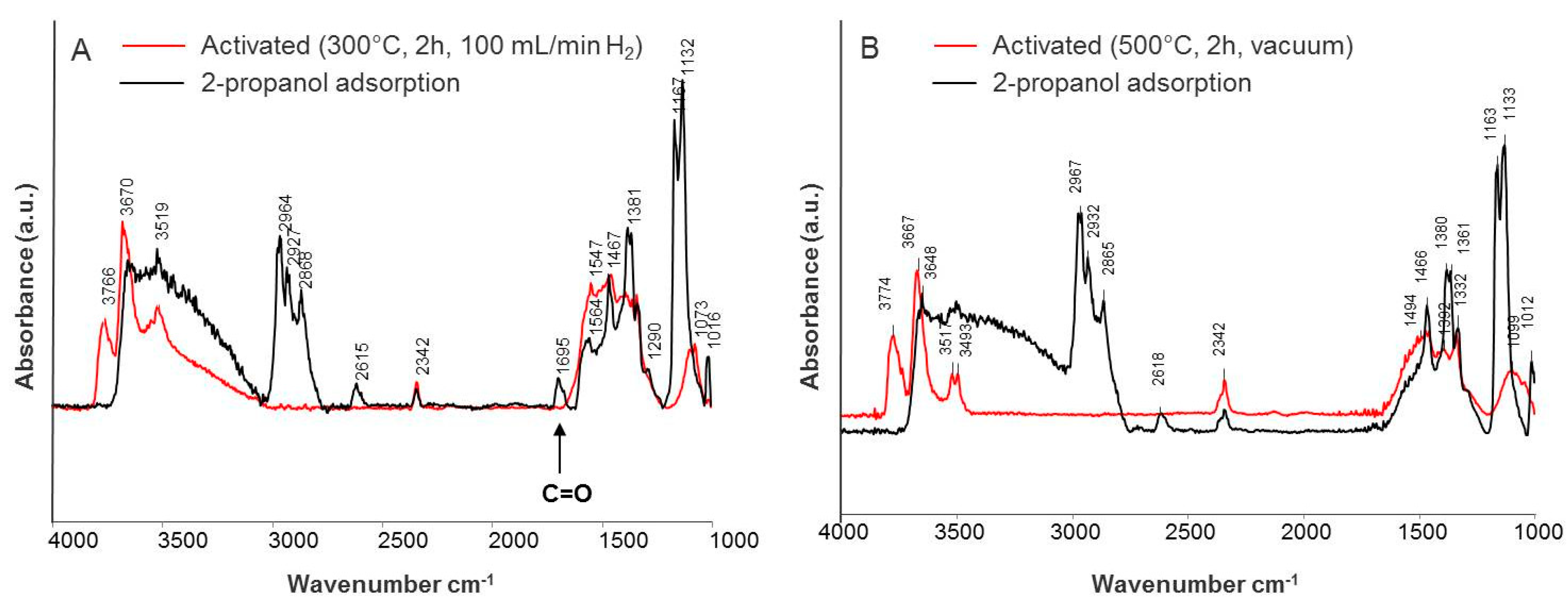
2.2.1. Fourier Transform Infrared Spectroscopy (FTIR)
Catalyst Characterization
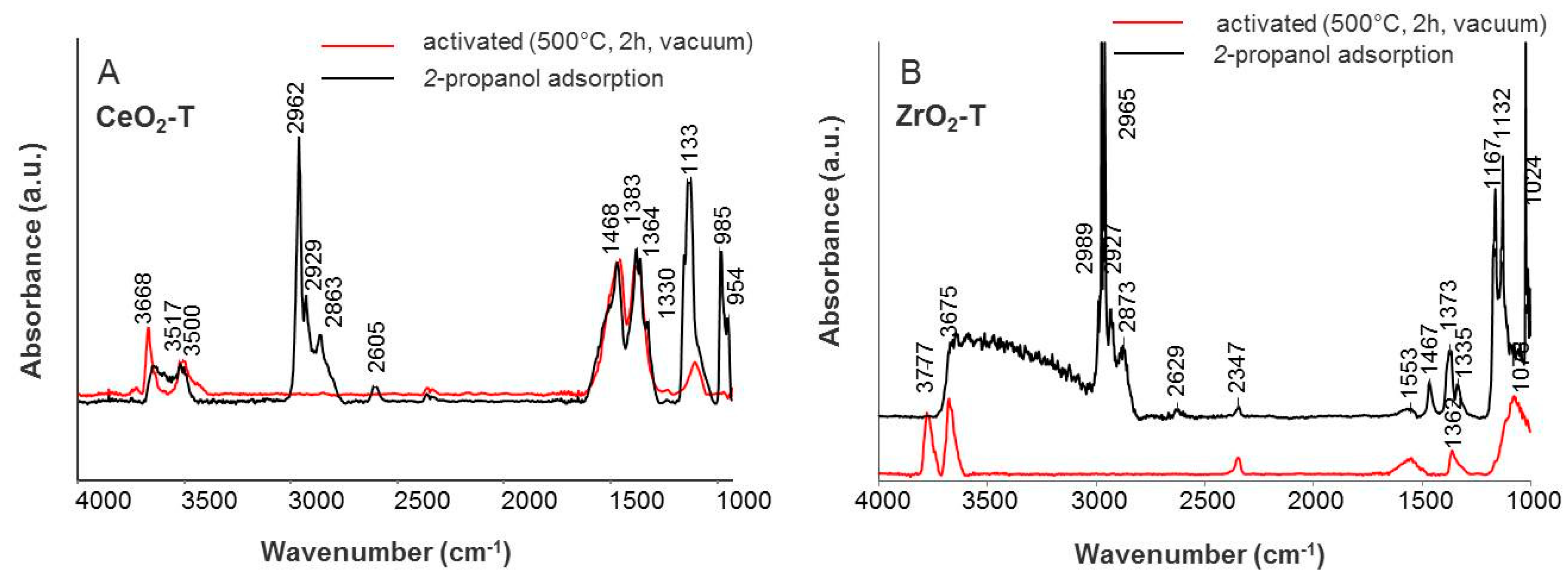
Iso-Propanol Adsorption
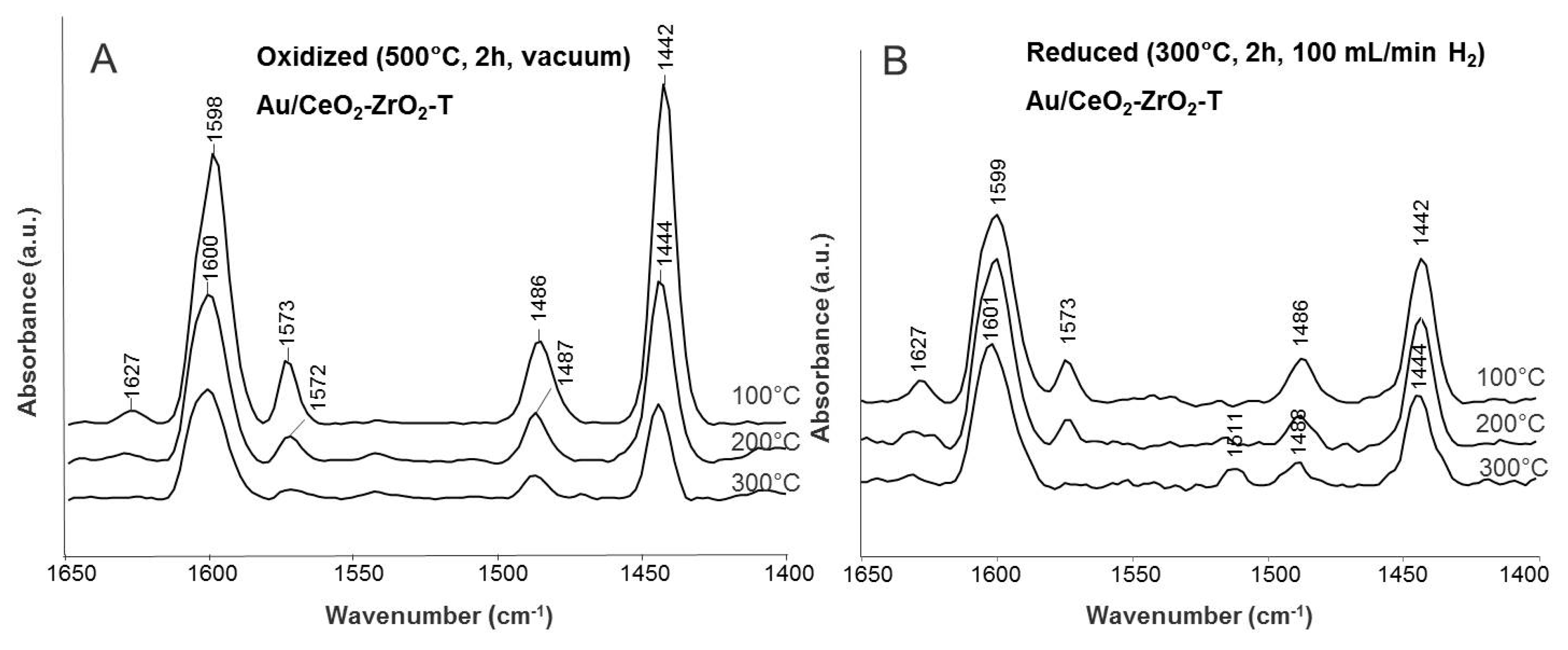
Pyridine Adsorption
2.3. Catalytic Test Reactions
2.3.1. Blind Test
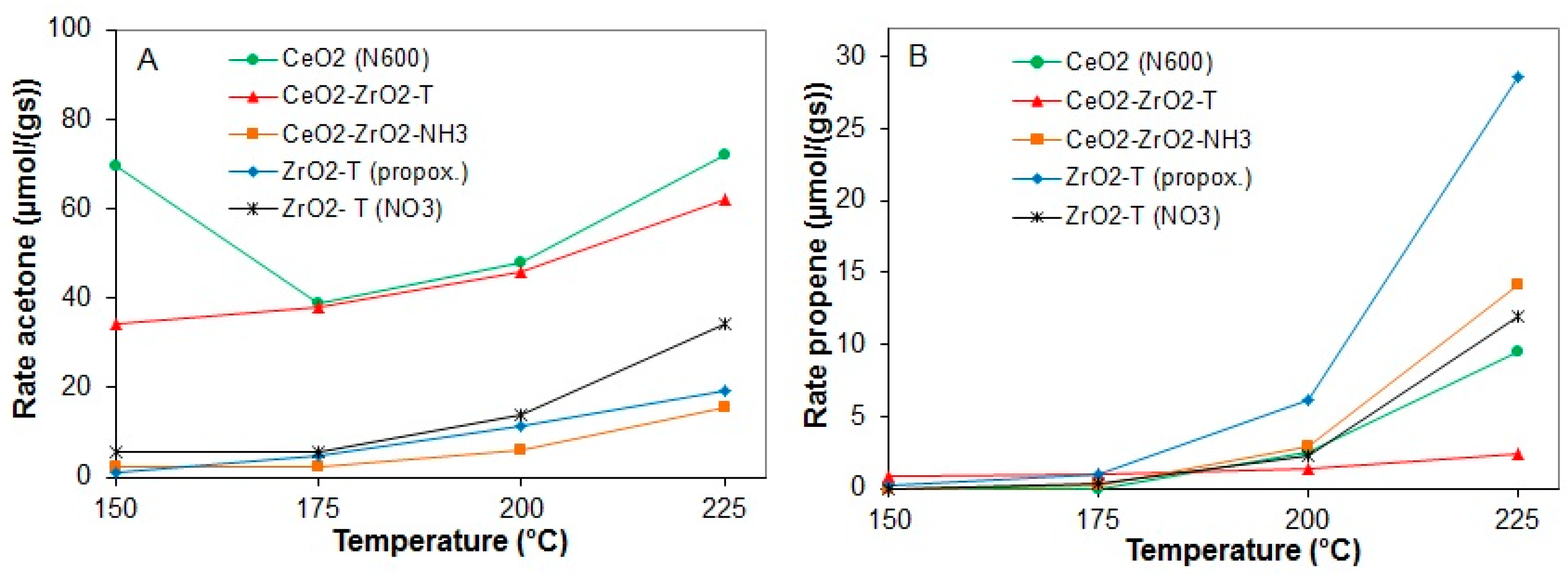
2.3.2. Conversion of 2-Propanol
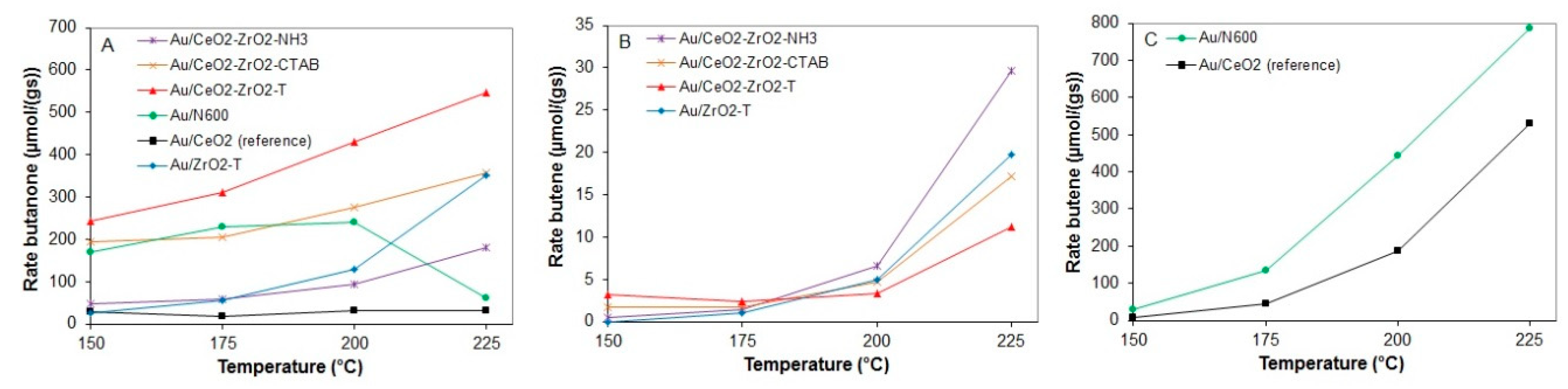
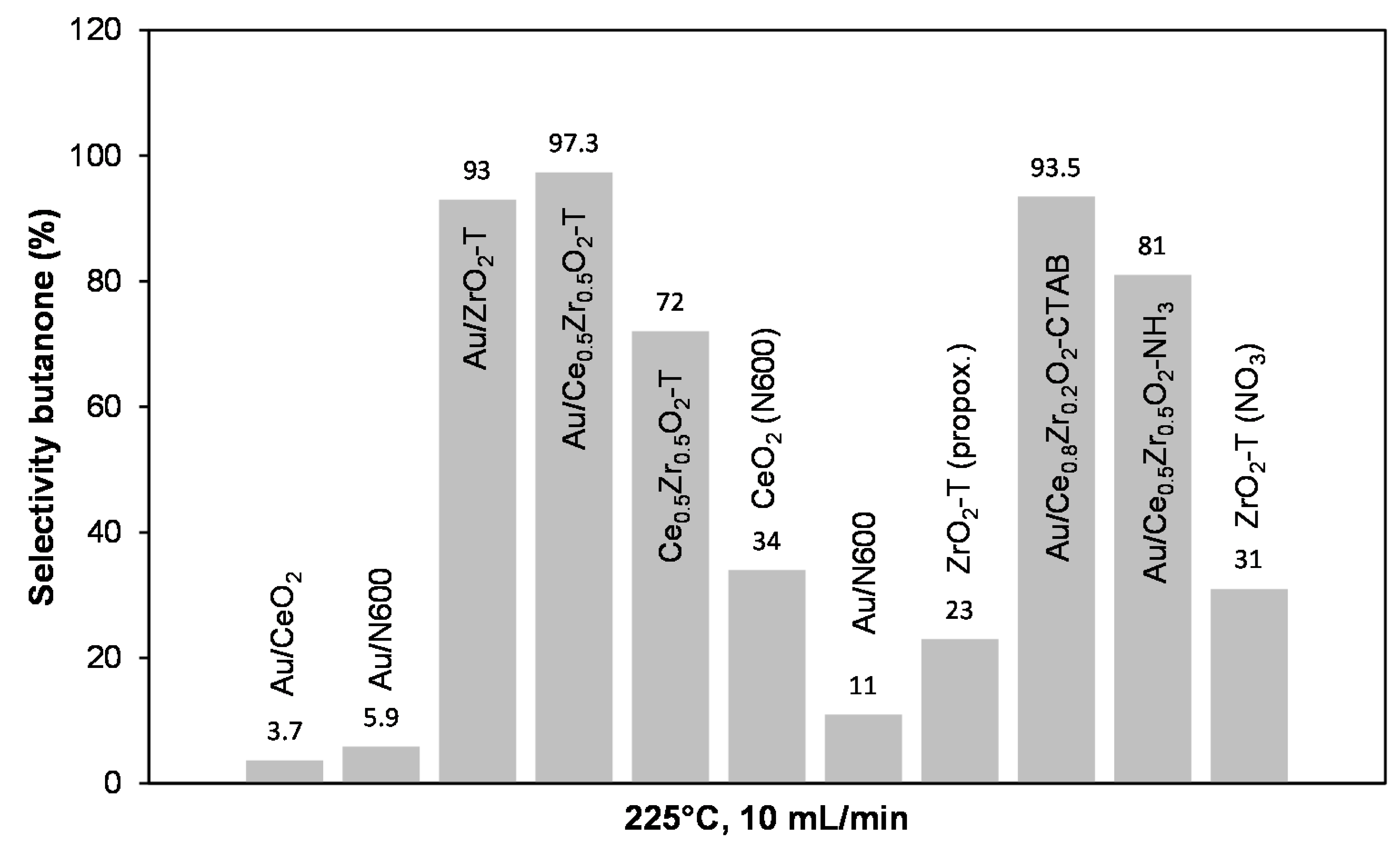
2.3.3. Conversion of 2-Butanol
3. Materials and Methods
3.1. Catalyst Preparation
3.1.1. Ceria, Zirconia and Ceria-Zirconia Mixed Oxides
Precipitation with Ammonia
Co-Precipitation Method with Ammonia
Surfactant-Assisted Method Using CTAB (Cetyltrimethylammonium Bromide)
Combined Use of Surfactant Brij-56® and Triblock-Copolymer Pluronic®123
Combined Use of Surfactant Brij-56® and Triblock-Copolymer Pluronic®123 and Precipitation with Ammonia
The same procedure was done with 0.03 mol of ZrO(NO3)2·xH2O aqueous solution. The sample was labeled ZrO2-T. The same was also done for 0.015 mol of Ce(NO3)3·6H2O and 0.015 mol of ZrO(NO3)2·xH2O aqueous solution. The sample was labeled CeO2-ZrO2-T.Synthesis of Mesoporous CexZr1-xO2
3.1.2. Au/CeO2, Au/ZrO2 and Au/CeO2-ZrO2 (1 wt.%)
3.2. Catalyst Characterization
3.2.1. AAS (Atomic Absorption Spectroscopy)
3.2.2. N2-BET (Brunauer-Emmet-Teller Surface Analysis)
3.2.3. XRD (X-Ray Diffraction)
3.2.4. NH3- and CO2-TPD (Temperature Programmed Desorption)
3.2.5. FTIR (Fourier Transform Infrared Spectroscopy)
3.3. Catalytic Tests
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Trovarelli, A. Catalytic Properties of Ceria and CeO2-Containing Materials. Catal. Rev. 1996, 38, 439–520. [Google Scholar] [CrossRef]
- Minh, N.Q.; Takahashi, T. Science and Technology of Ceramic Fuel Cells. Available online: http://www.123library.org/book_details/?id=39537 (accessed on 27 August 2018).
- Abdel-Mageed, A.M.; Kučerová, G.; Bansmann, J.; Behm, R.J. Active Au Species During the Low-Temperature Water Gas Shift Reaction on Au/CeO2: A Time-Resolved Operando XAS and DRIFTS Study. ACS Catal. 2017, 7, 6471–6484. [Google Scholar] [CrossRef]
- Campo, B.; Santori, G.; Petit, C.; Volpe, M. Liquid phase hydrogenation of crotonaldehyde over Au/CeO2 catalysts. Appl. Catal. Gen. 2009, 359, 79–83. [Google Scholar] [CrossRef]
- Karpenko, A.; Denkwitz, Y.; Plzak, V.; Cai, J.; Leppelt, R.; Schumacher, B.; Behm, R.J. Low-temperature water-gas shift reaction on Au/CeO2 catalysts—The influence of catalyst pre-treatment on the activity and deactivation in idealized reformate. Catal. Lett. 2007, 116, 105–115. [Google Scholar] [CrossRef]
- Meunier, F.C.; Reid, D.; Goguet, A.; Shekhtman, S.; Hardacre, C.; Burch, R.; Deng, W.; Flytzani-Stephanopoulos, M. Quantitative analysis of the reactivity of formate species seen by DRIFTS over a Au/Ce(La)O2 water–gas shift catalyst: First unambiguous evidence of the minority role of formates as reaction intermediates. J. Catal. 2007, 247, 277–287. [Google Scholar] [CrossRef]
- Rodriguez, J.A.; Hanson, J.C.; Stacchiola, D.; Senanayake, S.D. In situ/operando studies for the production of hydrogen through the water-gas shift on metal oxide catalysts. Phys. Chem. Chem. Phys. 2013, 15, 12004–12025. [Google Scholar] [CrossRef] [PubMed]
- Damyanova, S.; Pawelec, B.; Arishtirova, K.; Huerta, M.V.M.; Fierro, J.L.G. Study of the surface and redox properties of ceria–zirconia oxides. Appl. Catal. Gen. 2008, 337, 86–96. [Google Scholar] [CrossRef]
- Mori, K.; Miyauchi, Y.; Kuwahara, Y.; Yamashita, H. Shape Effect of MnOx-Decorated CeO2 Catalyst in Diesel Soot Oxidation. Bull. Chem. Soc. Jpn. 2017, 90, 556–564. [Google Scholar] [CrossRef]
- Pol, V.G.; Pol, S.V.; Gedanken, A.; Kessler, V.G.; Seisenbaeva, G.A.; Sung, M.-G.; Asai, S. Applied Magnetic Field Rejects the Coating of Ferromagnetic Carbon from the Surface of Ferromagnetic Cobalt: RAPET of CoZr2(acac)2(OiPr)8. J. Phys. Chem. B 2005, 109, 6121–6125. [Google Scholar] [CrossRef] [PubMed]
- Shanmugam, V.; Zapf, R.; Neuberg, S.; Hessel, V.; Kolb, G. Effect of ceria and zirconia promotors on Ni/SBA-15 catalysts for coking and sintering resistant steam reforming of propylene glycol in microreactors. Appl. Catal. B Environ. 2017, 203, 859–869. [Google Scholar] [CrossRef]
- Kubo, H.; Ohshima, Y.; Yamashita, T.; Kikuhara, S.; Saitoh, N.; Yoshizawa, N.; Nakagoe, O.; Tanabe, S. Mechanism of Soot Oxidation over CeO2–ZrO2 under O2 Flow. Bull. Chem. Soc. Jpn. 2017, 91, 437–443. [Google Scholar] [CrossRef]
- Dong, Y.; Wang, H.; Chen, I.-W. Electrical and hydrogen reduction enhances kinetics in doped zirconia and ceria: I. grain growth study. J. Am. Ceram. Soc. 2016, 100, 876–886. [Google Scholar] [CrossRef]
- Azambre, B.; Atribak, I.; Bueno-López, A.; García-García, A. Probing the Surface of Ceria−Zirconia Catalysts Using NOx Adsorption/Desorption: A First Step Toward the Investigation of Crystallite Heterogeneity. J. Phys. Chem. C 2010, 114, 13300–13312. [Google Scholar] [CrossRef]
- de Rivas, B.; López-Fonseca, R.; González-Velasco, J.R.; Gutiérrez-Ortiz, J.I. On the mechanism of the catalytic destruction of 1,2-dichloroethane over Ce/Zr mixed oxide catalysts. J. Mol. Catal. Chem. 2007, 278, 181–188. [Google Scholar] [CrossRef]
- Monte, R.D.; Kašpar, J. Nanostructured CeO2–ZrO2 mixed oxides. J. Mater. Chem. 2005, 15, 633–648. [Google Scholar] [CrossRef]
- Moretti, E.; Storaro, L.; Talon, A.; Chitsazan, S.; Garbarino, G.; Busca, G.; Finocchio, E. Ceria-zirconia based catalysts for ethanol steam reforming. Fuel 2015, 153, 166–175. [Google Scholar] [CrossRef]
- Reddy, B.M.; Reddy, G.K.; Katta, L. Structural characterization and dehydration activity of CeO2–SiO2 and CeO2–ZrO2 mixed oxides prepared by a rapid microwave-assisted combustion synthesis method. J. Mol. Catal. Chem. 2010, 319, 52–57. [Google Scholar] [CrossRef]
- Wang, B.; Wu, X.; Ran, R.; Si, Z.; Weng, D. IR characterization of propane oxidation on Pt/CeO2–ZrO2: The reaction mechanism and the role of Pt. J. Mol. Catal. Chem. 2012, 356, 100–105. [Google Scholar] [CrossRef]
- Sugiura, M.; Ozawa, M.; Suda, A.; Suzuki, T.; Kanazawa, T. Development of Innovative Three-Way Catalysts Containing Ceria–Zirconia Solid Solutions with High Oxygen Storage/Release Capacity. Bull. Chem. Soc. Jpn. 2005, 78, 752–767. [Google Scholar] [CrossRef]
- Kaspar, J.; Fornasiero, P. Structural properties and thermal stability of ceria-zirconia and related materials. In Catalysis by Ceria and Related Materials; Catalytic Science Series; Imperial College Press (distributed by World Scientific Publishing Co.): London, UK, 2002; pp. 217–241. [Google Scholar]
- Mamontov, E.; Egami, T.; Brezny, R.; Koranne, M.; Tyagi, S. Lattice Defects and Oxygen Storage Capacity of Nanocrystalline Ceria and Ceria-Zirconia. J. Phys. Chem. B 2000, 104, 11110–11116. [Google Scholar] [CrossRef]
- Corti, C.W.; Holliday, R.J.; Thompson, D.T. Commercial aspects of gold catalysis. Catal. Gold 2005, 291, 253–261. [Google Scholar] [CrossRef]
- Rynkowski, J.M.; Dobrosz-Gomez, I. Ceria -zirconia supported gold catalysts. In Annales Universitatis Marie Curie-Sklodowska Lublin; vol. LXIV, 14 Sectio AA; Maria Curie Sklodowska University: Lublin, Poland, 2009. [Google Scholar]
- Grünert, W. Oxidation Catalysis over Gold–New Insight into Active Sites and Reaction Mechanisms. In 47. Jahrestreffen Deutscher Katalytiker; Weimar, Germany, 2014. [Google Scholar]
- Grunwaldt, J.-D.; Baiker, A. Gold/Titania interfaces and their role in carbon monoxide oxidation. J. Phys. Chem. B 1999, 103, 1002–1012. [Google Scholar] [CrossRef]
- Wang, Y.; Widmann, D.; Lehnert, F.; Gu, D.; Schüth, F.; Behm, R.J. Avoiding Self-Poisoning: A Key Feature for the High Activity of Au/Mg(OH)2 Catalysts in Continuous Low-Temperature CO Oxidation. Angew. Chem. Int. Ed. 2017, 56, 9597–9602. [Google Scholar] [CrossRef] [PubMed]
- Smit, G.; Lazar, K.; Craje, M.W.J. Influence of water vapour on low-temperature CO oxidation over Au/Fe2O3 catalyst. Croat. Chem. Acta 2007, 80, 141–145. [Google Scholar]
- Xu, Q.; Kharas, K.C.C.; Datye, A.K. The Preparation of Highly Dispersed Au/Al2O3 by Aqueous Impregnation. Catal. Lett. 2003, 85, 229–235. [Google Scholar] [CrossRef]
- Pop, M. Variation of acid-base- and redoxproperties in CeO2-ZrO2- mixed oxides. Master’s Thesis, Technische Universität München, Munich, Germany, 2010. [Google Scholar]
- Brunauer, S.; Emmett, P.H.; Teller, E. Adsorption of Gases in Multimolecular Layers. J. Am. Chem. Soc. 1938, 60, 309–319. [Google Scholar] [CrossRef]
- Yuan, Q.; Duan, H.-H.; Li, L.-L.; Sun, L.-D.; Zhang, Y.-W.; Yan, C.-H. Controlled synthesis and assembly of ceria-based nanomaterials. J. Colloid Interface Sci. 2009, 335, 151–167. [Google Scholar] [CrossRef] [PubMed]
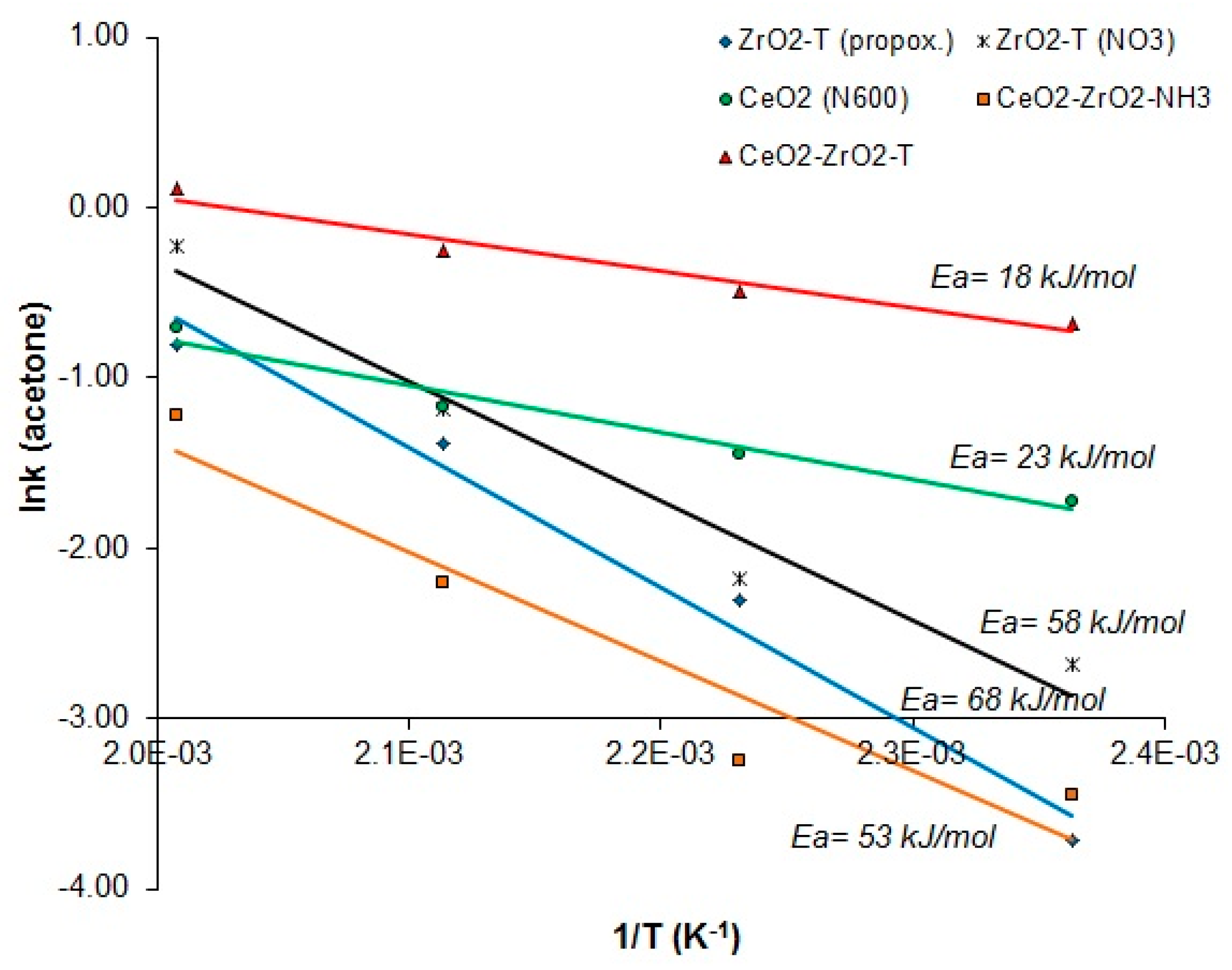

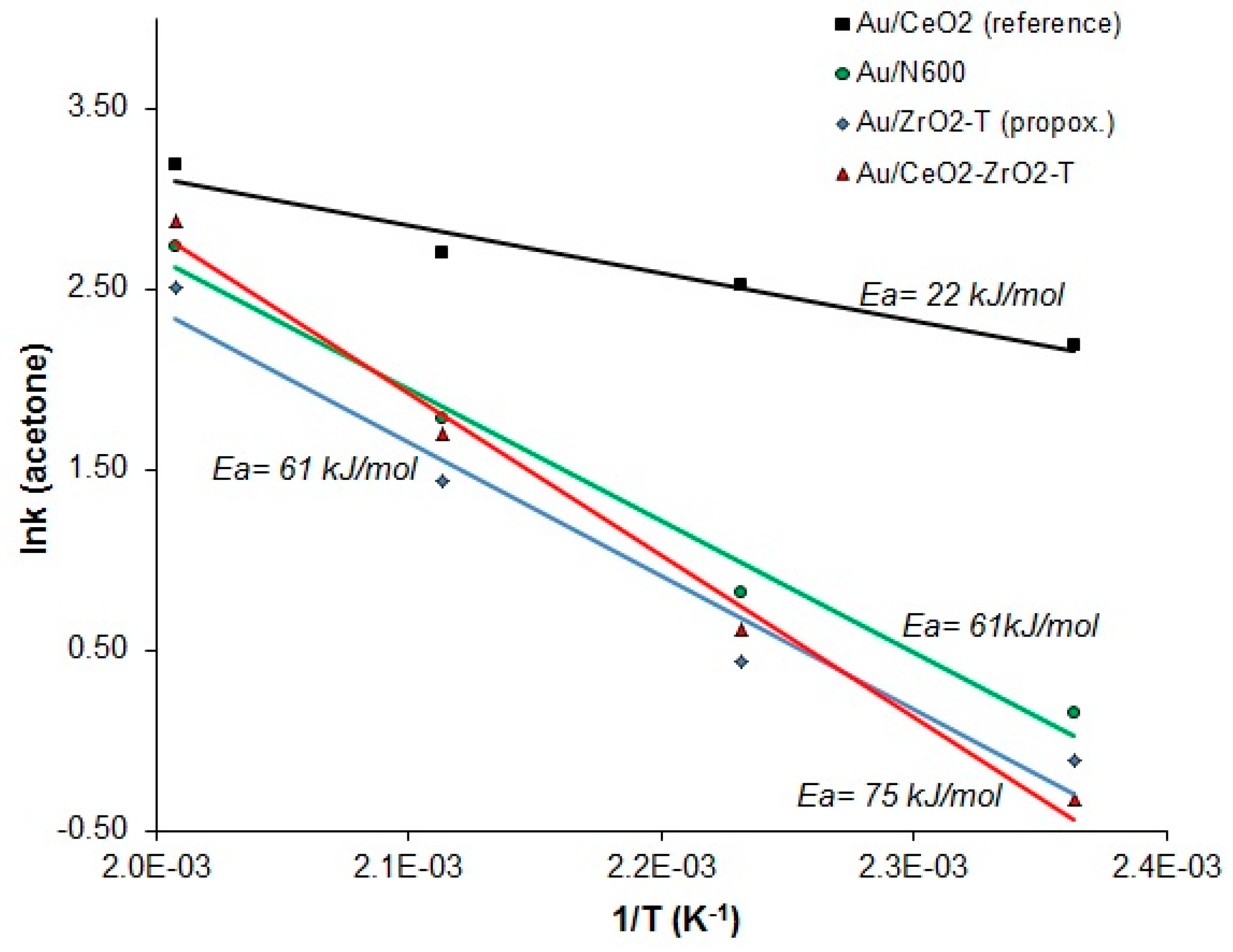
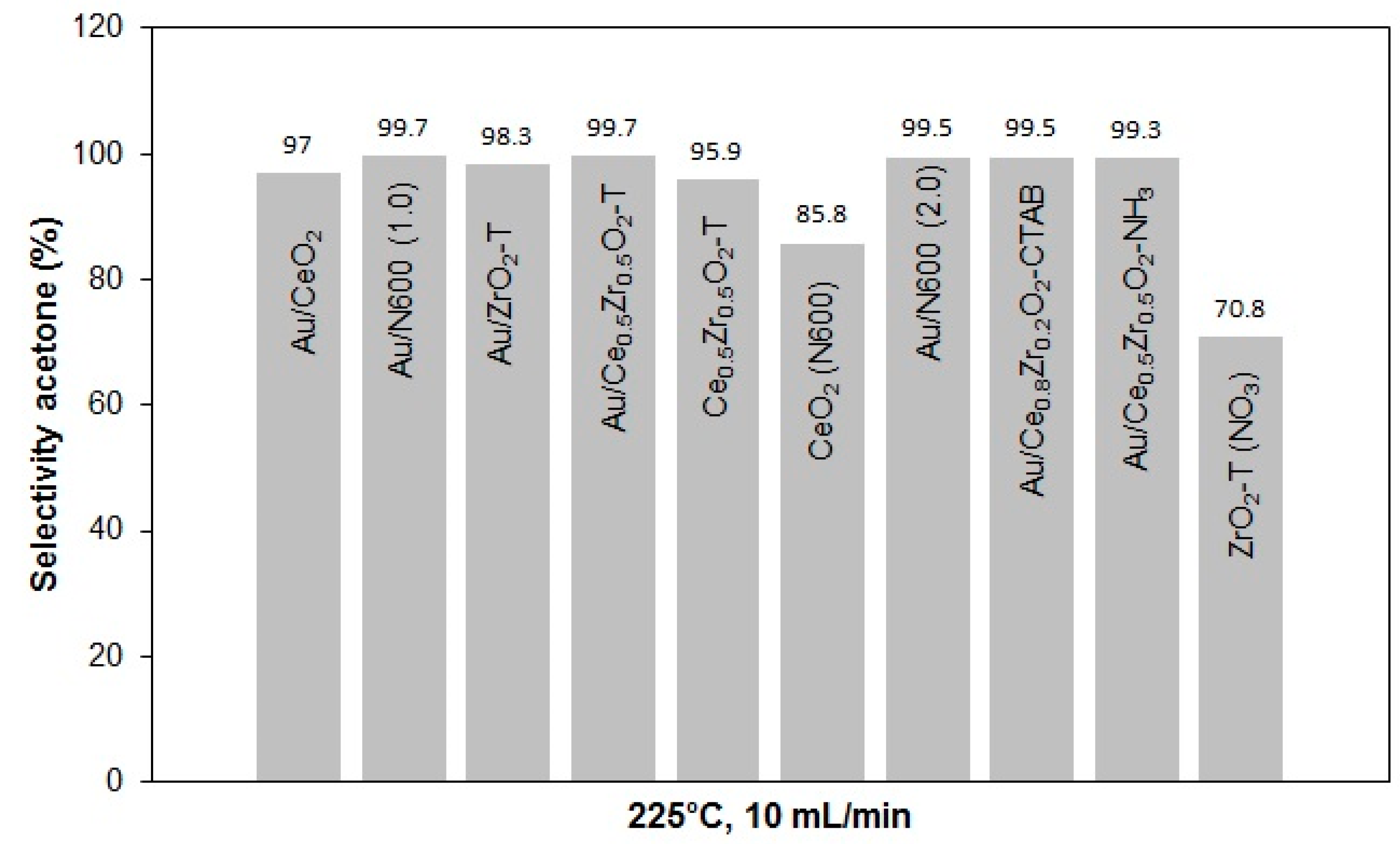
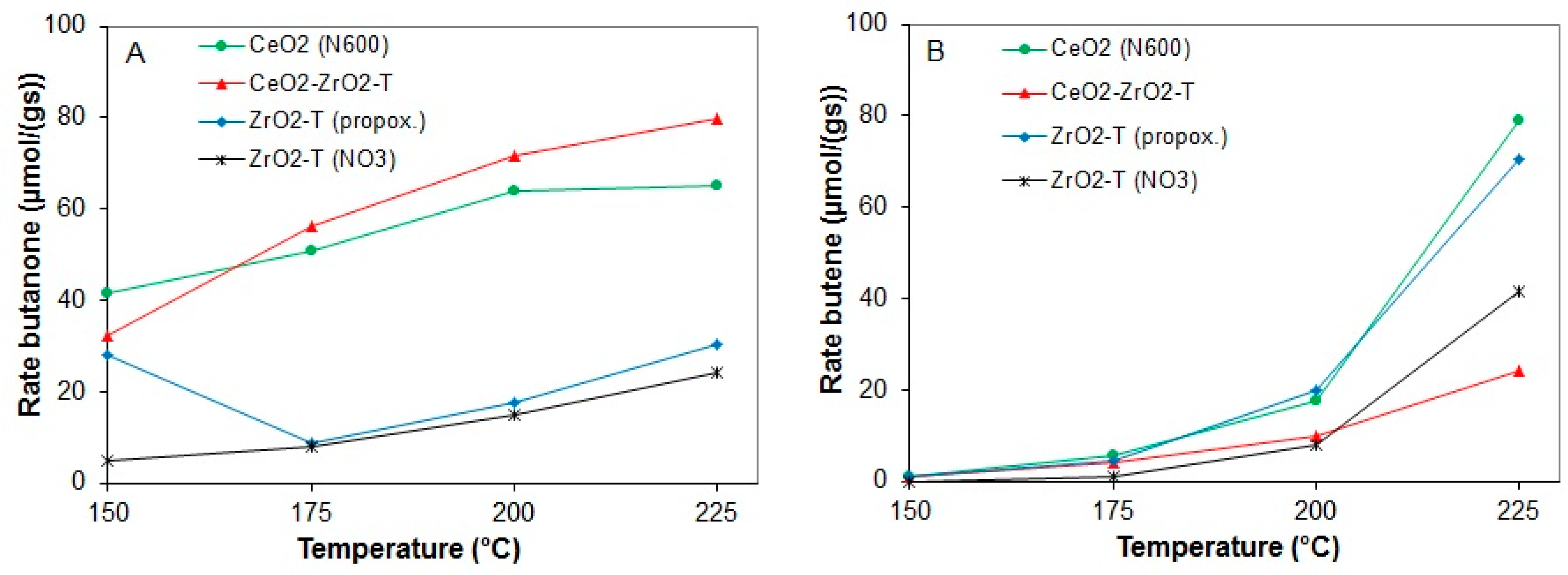
| Sample | Weight | Au-Amount | Au-Content |
|---|---|---|---|
| [mg] | [mg] | [wt.%] | |
| reference Au/CeO2 | - | - | 0.84 |
| Au/N600 | 101.7 | 0.979 | 0.96 |
| Au/N600 | 181.7 | 3.18 | 1.75 |
| Au/CeO2-T | - | - | 0.01 |
| Au/ZrO2-T | 171.2 | 2.0 | 1.17 |
| Au/CeO2-ZrO2-T | 121.5 | 1.17 | 0.97 |
| Au/CeO2-ZrO2_NH3 | 152 | 1.43 | 0.94 |
| Au/CeO2-ZrO2_CTAB | 163.8 | 2.03 | 1.24 |
| Sample | Specific Surface | Calcination | Micropore | Cummulative |
|---|---|---|---|---|
| Area | Temperature | Volume | Pore Volume | |
| [m2/g] | [°C] | [cm3/g] | [cm3/g] | |
| reference CeO2 | 6 | 600 | - | - |
| CeO2-T | 65 | 600 | 0.012 | 0.12 |
| CeO2 (N600) | 72 | 600 | 0.015 | 0.15 |
| ZrO2-T (propox.) | 46 | 600 | 0.0065 | 0.13 |
| ZrO2-T (NO3) | 92 | 600 | 0.015 | 0.18 |
| CeO2-ZrO2_CTAB | 125 | 400 | 0.02 | 0.16 |
| CeO2-ZrO2-T | 83 | 600 | 0.014 | 0.17 |
| CeO2-ZrO2-T_EtOH | 102 | 400 | 0.013 | 0.11 |
| CeO2-ZrO2_NH3 | 15 | 600 | - | 0.08 |
| Sample | Specific Surface | Acid Sites | Basic Sites | Basicity | Active Sites |
|---|---|---|---|---|---|
| Area [m2/g] | [mol/g] | [mol/g] | [%] | [mol/m2] | |
| commercial CeO2 | 6 | 7 | 0.5 | 7 | 1.25 |
| CeO2 (N600) | 72 | 13; 21 (H2) | 7 | 25 | 0.38 |
| ZrO2-T (propox.) | 46 | 59 (H2) | 6 | 9 | 1.41 |
| ZrO2-T (NO3) | 92 | 173 | - | - | - |
| CeO2-ZrO2-T | 83 | 93 | 13 | 12 | 1.28 |
| reference Au/CeO2 | 6 | 13 | 0.9 | 6 | 2.32 |
| Au/N600 (1.0) | 72 | 8 | - | - | - |
| Au/N600 (2.0) | 72 | 12; 19 (H2) | - | - | - |
| Au/ZrO2-T (propox) | 43 | 83 (H2) | 9 | 10 | 2.14 |
| Au/CeO2-ZrO2-T | 83 | 59 (H2) | 15 | 20 | 0.89 |
| Species | Frequencies (cm−1) | ||
|---|---|---|---|
| CeO2-T | ZrO2-T | CeO2-ZrO2-T | |
| ν (OH) Type I | 3723 | 3772 | 3772 |
| ν (OH) Type II | 3667 | 3676 | 3667 |
| ν (OH) Type III | 3519 | - | 3519 |
| ν (CO2) | 2366 | 2344 | 2344 |
| ν (CO32−) bidentate | 1467 | 1550 | 1467 |
| monodentate | 1381 | 1362 | 1392, 1338 |
| bridged | 1107 | 1075 | 1102 |
| Species | Frequencies (cm−1) | |||||
|---|---|---|---|---|---|---|
| CeO2-T | ZrO2-T | CeO2-ZrO2-T | CeO2-ZrO2-T | Au/CeO2-ZrO2-T | Au/CeO2-ZrO2-T | |
| Oxidized | Oxidized | Oxidized | Reduced | Oxidized | Reduced | |
| ν(CH3) | 2962 | 2965 | 2967 | 2964 | 2962 | 2961 |
| ν (CH2) | 2929 | 2927 | 2932 | 2927 | 2926 | 2926 |
| ν (CH) | 2863 | 2873 | 2865 | 2868 | 2866 | 2866 |
| ν (C=O) | - | - | - | 1695 | 1677 | 1692 |
| ν (OOC) | 1468 | 1467 | 1466 | 1564 | 1550 | |
| 1383 | 1373 | 1380 | 1467, 1381 | 1466, 1362 | 1464, 1381 | |
| ν (CO32-) | 1330 | 1335 | 1332 | 1332 | 1332 | 1332 |
| ν (C-O) | 1163 | 1167 | 1163 | 1167 | 1157 | 1166 |
| 1133 | 1132 | 1133 | 1132 | 1131 | 1129 | |
| ν (C-C) | 985 | 1024 | 1012 | 1016 | 1012 | 1012 |
| Ring Vibration ν(CCN) | Frequency (cm−1) | |||
|---|---|---|---|---|
| CeO2 (N600) | Au/N600 | CeO2-ZrO2-T | Au/CeO2-ZrO2-T | |
| Oxidized | Oxidized | Reduced | Oxidized | |
| L-Pyd | 1621, 1573, 1483 | 1621, 1573, 1483 | 1625, 1573, 1484 | 1626, 1573, 1485 |
| H-Pyd | 1595, 1440 | 1595, 1440, 1405 | 1596, 1440 | 1597, 1440 |
| B-Pyd | - | - | - | - |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Glorius, M.; Markovits, M.A.C.; Breitkopf, C. Design of Specific Acid-Base-Properties in CeO2-ZrO2-Mixed Oxides via Templating and Au Modification. Catalysts 2018, 8, 358. https://doi.org/10.3390/catal8090358
Glorius M, Markovits MAC, Breitkopf C. Design of Specific Acid-Base-Properties in CeO2-ZrO2-Mixed Oxides via Templating and Au Modification. Catalysts. 2018; 8(9):358. https://doi.org/10.3390/catal8090358
Chicago/Turabian StyleGlorius, Maja, Monica A. C. Markovits, and Cornelia Breitkopf. 2018. "Design of Specific Acid-Base-Properties in CeO2-ZrO2-Mixed Oxides via Templating and Au Modification" Catalysts 8, no. 9: 358. https://doi.org/10.3390/catal8090358
APA StyleGlorius, M., Markovits, M. A. C., & Breitkopf, C. (2018). Design of Specific Acid-Base-Properties in CeO2-ZrO2-Mixed Oxides via Templating and Au Modification. Catalysts, 8(9), 358. https://doi.org/10.3390/catal8090358