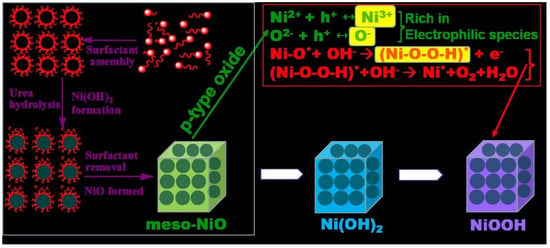
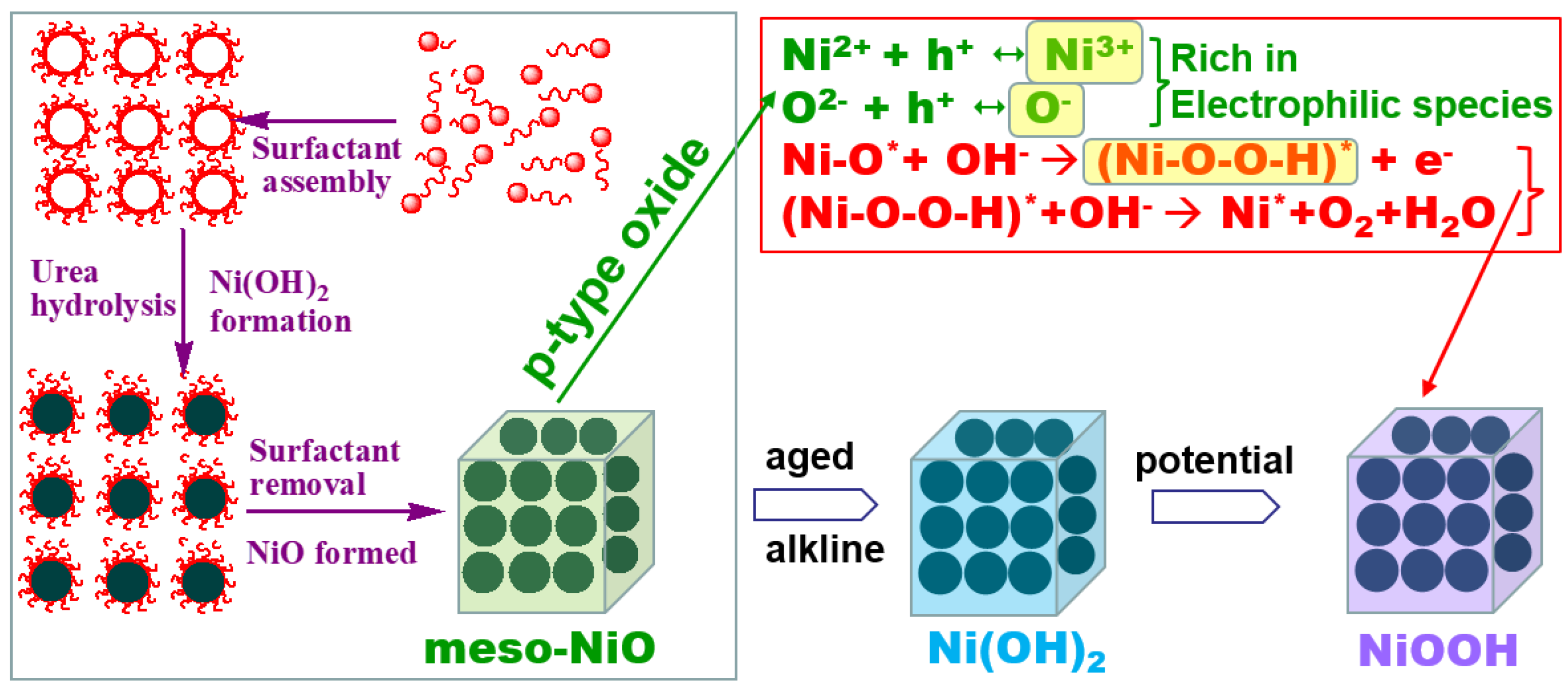
Engineering Mesoporous NiO with Enriched Electrophilic Ni3+ and O− toward Efficient Oxygen Evolution
Abstract
1. Introduction
2. Results and Discussion
2.1. Texture Characterizations
2.2. OER Activity
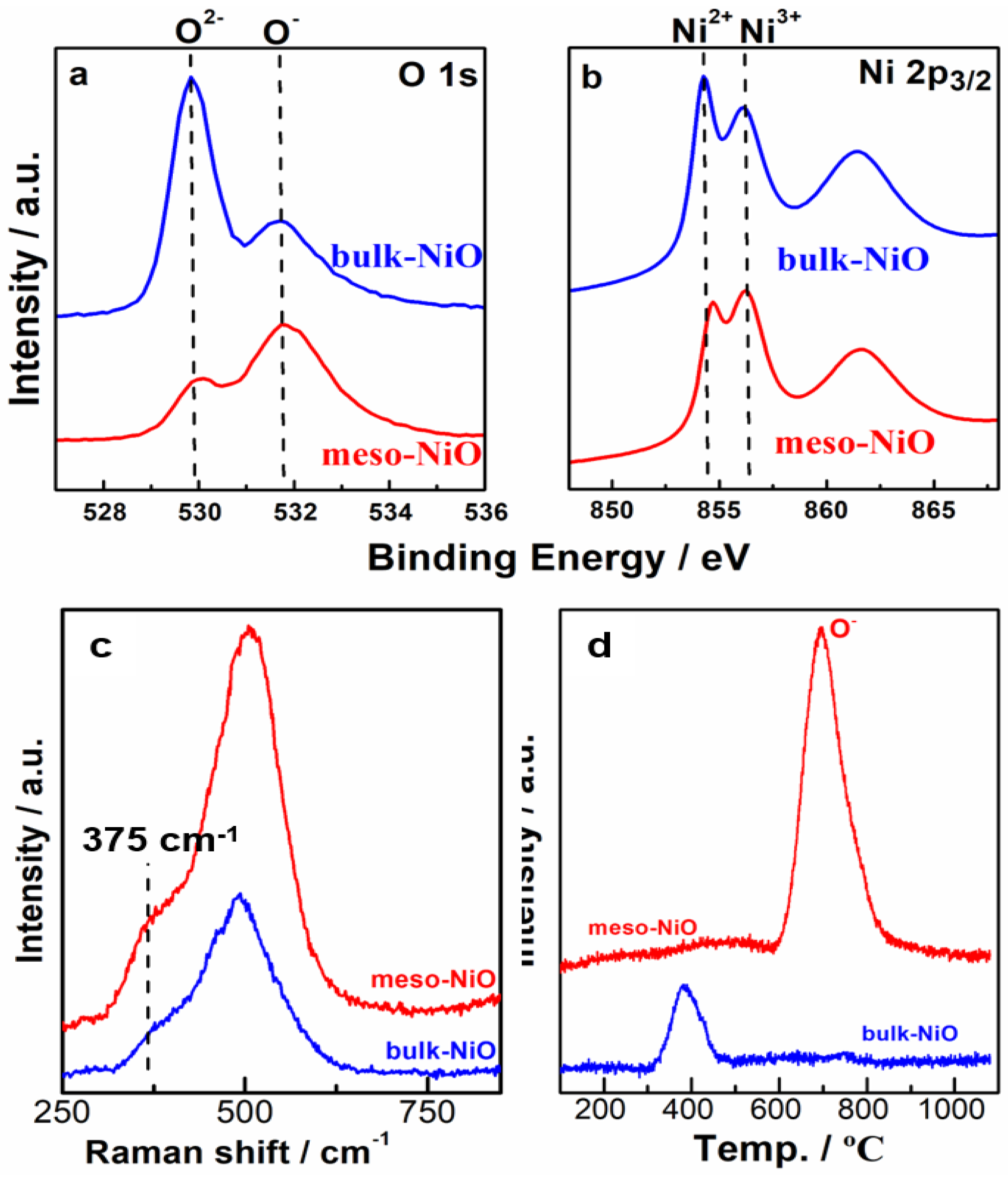
2.3. Characterizations
2.4. Discussion
3. Materials and Methods
3.1. Materials
3.2. Analytical Equipment
3.3. Synthesis of Nano-Nickel Oxide
3.4. Synthesis of Meso-Nickel Oxide
3.5. Characterizations
3.6. Electrode Preparation
3.7. Electrochemical Characterization
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Kim, J.; Jin, H.; Oh, A.; Baik, H.; Joo, S.H.; Lee, K. Synthesis of compositionally tunable, hollow mixed metal sulphide coxniysz octahedral nanocages and their composition-dependent electrocatalytic activities for oxygen evolution reaction. Nanoscale 2017, 13, 3437–3444. [Google Scholar] [CrossRef] [PubMed]
- Michalska-Domanska, M.; Norek, M.; Jóźwik, P.; Jankiewicz, B.; Stępniowski, W. Catalytic stability and surface analysis of microcrystalline Ni3Al thin foils in methanol decomposition. Appl. Surf. Sci. 2014, 293, 169–176. [Google Scholar] [CrossRef]
- Tang, C.; Asiri, A.M.; Sun, X. Highly-active oxygen evolution electrocatalyzed by a Fe-doped NiSe nanoflake array electrode. Chem. Commun. 2016, 52, 4529–4532. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Cui, W.; Liu, Q.; Xing, Z.; Asiri, A.M.; Sun, X. Recent progress in cobalt-based heterogeneous catalysts for electrochemical water splitting. Adv. Mater. 2016, 28, 215–230. [Google Scholar] [CrossRef] [PubMed]
- Trotochaud, L.; Ranney, J.K.; Williams, K.N.; Boettcher, S.W. Solution-cast metal oxide thin film electrocatalysts for oxygen evolution. J. Am. Chem. Soc. 2012, 134, 17253–17261. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.Y.; Hsu, Y.Y.; Chen, R.; Chan, T.S.; Chen, H.M.; Liu, B. Ni3+-induced formation of active NiOOH on the spinel Ni-Co oxide surface for efficient oxygen evolution reaction. Adv. Energy Mater. 2015, 5, 395–417. [Google Scholar] [CrossRef]
- Matsumura, Y.; Tanaka, K.; Tode, N.; Yazawa, T.; Haruta, M. Catalytic methanol decomposition to carbon monoxide and hydrogen over nickel supported on silica. J. Mol. Catal. A Chem. 2000, 152, 157–165. [Google Scholar] [CrossRef]
- Taguchi, A.; Schüth, F. Ordered mesoporous materials in catalysis. Micropor. Mesopor. Mater. 2005, 77, 1–45. [Google Scholar] [CrossRef]
- Chang, J.; Park, M.; Ham, D.; Ogale, S.B.; Mane, R.S.; Han, S.H. Liquid-phase synthesized mesoporous electrochemical supercapacitors of nickel hydroxide. Electrochim. Acta 2008, 53, 5016–5021. [Google Scholar] [CrossRef]
- Yuan, C.; Gao, B.; Su, L.; Zhang, X. Interface synthesis of mesoporous MnO2 and its electrochemical capacitive behaviors. J. Colloid Interface Sci. 2008, 322, 545–550. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.G.; Xia, Y.Y. Electrochemical capacitance characterization of NiO with ordered mesoporous structure synthesized by template SBA-15. Electrochim. Acta 2006, 51, 3223–3227. [Google Scholar] [CrossRef]
- Xing, W.; Li, F.; Yan, Z.F.; Lu, G.Q. Synthesis and electrochemical properties of mesoporous nickel oxide. J. Power Sources 2004, 134, 324–330. [Google Scholar] [CrossRef]
- Shaju, K.M.; Jiao, F.; Débart, A.; Bruce, P.G. Mesoporous and nanowire Co3O4 as negative electrodes for rechargeable lithium batteries. Phys. Chem. Chem. Phys. 2007, 9, 1837–1842. [Google Scholar] [CrossRef] [PubMed]
- Li, J.-H.; Wang, C.-C.; Huang, C.-J.; Sun, Y.-F.; Weng, W.-Z.; Wan, H.-L. Mesoporous nickel oxides as effective catalysts for oxidative dehydrogenation of propane to propene. Appl. Catal. A Gen. 2010, 382, 99–105. [Google Scholar] [CrossRef]
- He, Y.; Wu, Y.; Chen, T.; Weng, W.; Wan, H. Low-temperature catalytic performance for oxidative dehydrogenation of propane on nanosized Ti(Zr)-Ni-O prepared by modified sol-gel method. Catal. Commun. 2006, 7, 268–271. [Google Scholar] [CrossRef]
- Alberto, N.; Vigario, C.; Duarte, D.; Almeida, D.; Gonçalves, G. Characterization of graphene oxide coatings onto optical fibers for sensing applications. Mater. Today Proc. 2015, 2, 171–177. [Google Scholar] [CrossRef]
- Schenk, A.S.; Eiben, S.; Goll, M.; Reith, L.; Kulak, A.N.; Meldrum, F.C.; Jeske, H.; Wege, C.; Ludwigs, S. Virus-directed formation of electrocatalytically active nanoparticle-based Co3O4 tubes. Nanoscale 2017, 9, 6334–6345. [Google Scholar] [CrossRef] [PubMed]
- Han, L.; Dong, C.; Zhang, C.; Gao, Y.; Zhang, J.; Gao, H.; Wang, Y.; Zhang, Z. Dealloying-directed synthesis of efficient mesoporous CoFe-based catalysts towards the oxygen evolution reaction and overall water splitting. Nanoscale 2017, 9, 16467–16475. [Google Scholar] [CrossRef] [PubMed]
- Lyons, M.E.; Brandon, M.P. The oxygen evolution reaction on passive oxide covered transition metal electrodes in aqueous alkaline solution. Part 1-nickel. Int. J. Electrochem. Sci. 2008, 3, 1386–1424. [Google Scholar]
- Li, W.; Gao, X.; Wang, X.; Xiong, D.; Huang, P.P.; Song, W.G.; Bao, X.; Liu, L. From water reduction to oxidation: Janus Co-Ni-P nanowires as high-efficiency and ultrastable electrocatalysts for over 3000h water splitting. J. Power Sources 2016, 330, 156–166. [Google Scholar] [CrossRef]
- Zhou, W.; Wu, X.; Cao, X.; Huang, X.; Tan, C.; Tian, J.; Liu, H.; Wang, J.; Zhang, H. Ni3S2 nanorods/Ni foam composite electrode with low overpotential for electrocatalytic oxygen evolution. Energ. Environ. Sci. 2013, 6, 2921–2924. [Google Scholar] [CrossRef]
- Sun, H.; Zhao, Y.; Mølhave, K.; Zhang, M.; Zhang, J. Simultaneous modulation of surface composition, oxygen vacancies and assembly in hierarchical Co3O4 mesoporous nanostructures for lithium storage and electrocatalytic oxygen evolution. Nanoscale 2017, 9, 14431–14441. [Google Scholar] [CrossRef] [PubMed]
- Suen, N.T.; Hung, S.F.; Quan, Q.; Zhang, N.; Xu, Y.J.; Chen, H.M. Electrocatalysis for the oxygen evolution reaction: Recent development and future perspectives. Chem. Soc. Rev. 2017, 46, 337–365. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B.; Yu, H.L.; Ni, H.; Hu, S. Bimetallic (FexNi1−x)2P nanoarrays as exceptionally efficient electrocatalysts for oxygen evolution in alkaline and neutral media. Nano Energy 2017, 38, 553–560. [Google Scholar] [CrossRef]
- Stoyanova, M.; Konova, P.; Nikolov, P.; Naydenov, A.; Christoskova, S.; Mehandjiev, D. Alumina-supported nickel oxide for ozone decomposition and catalytic ozonation of CO and VOCs. Chem. Eng. J. 2006, 122, 41–46. [Google Scholar] [CrossRef]
- Cordoba-Torresi, S.I. Electrochromic behavior of nickel oxide electrodes. J. Electrochem. Soc. 1991, 138, 1554–1559. [Google Scholar] [CrossRef]
- Heracleous, E.; Lemonidou, A.A. Ni-nb-o mixed oxides as highly active and selective catalysts for ethene production via ethane oxidative dehydrogenation. Part i: Characterization and catalytic performance. J. Catal. 2006, 237, 162–174. [Google Scholar] [CrossRef]
- Fabbri, E.; Habereder, A.; Waltar, K.; Kötz, R.; Schmidt, T.J. Developments and perspectives of oxide-based catalysts for the oxygen evolution reaction. Catal. Sci. Technol. 2014, 4, 3800–3821. [Google Scholar] [CrossRef]
- Pfeifer, V.; Jones, T.E.; Wrabetz, S.; Massue, C.; Velasco Velez, J.J.; Arrigo, R.; Scherzer, M.; Piccinin, S.; Havecker, M.; Knop-Gericke, A. Reactive oxygen species in iridium-based oer catalysts. Chem. Sci. 2016, 7, 6791–6795. [Google Scholar] [CrossRef] [PubMed]
- Corrigan, D.A.; Bendert, R.M. Effect of coprecipitated metal ions on the electrochemistry of nickel hydroxide thin films: Cyclic voltammetry in 1 M KOH. J. Electrochem. Soc. 1989, 20, 723–728. [Google Scholar] [CrossRef]
- Wehrens-Dijksma, M.; Notten, P.H.L. Electrochemical quartz microbalance characterization of Ni(OH)2-based thin film electrodes. Electrochim. Acta 2006, 51, 3609–3621. [Google Scholar] [CrossRef]
- Michalska-Domańska, M.; Jóźwik, P.; Jankiewicz, B.J.; Bartosewicz, B.; Siemiaszko, D.; Stępniowski, W.J.; Bojar, Z. Study of Cyclic Ni3Al Catalyst Pretreatment Process for Uniform Carbon Nanotubes Formation and Improved Hydrogen Yield in Methanol Decomposition. Mater. Today Proc. 2016, 3S, S171–S177. [Google Scholar] [CrossRef]
- Chen, G.F.; Ma, T.Y.; Liu, Z.Q.; Li, N.; Su, Y.Z.; Davey, K.; Qiao, S.Z. Efficient and stable bifunctional electrocatalysts Ni/NixMy (M = P, S) for overall water splitting. Adv. Funct. Mater. 2016, 26, 3314–3323. [Google Scholar] [CrossRef]


© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, X.; Zhai, Z.-Y.; Chen, Z.; Zhang, L.-Z.; Zhao, X.-F.; Si, F.-Z.; Li, J.-H. Engineering Mesoporous NiO with Enriched Electrophilic Ni3+ and O− toward Efficient Oxygen Evolution. Catalysts 2018, 8, 310. https://doi.org/10.3390/catal8080310
Liu X, Zhai Z-Y, Chen Z, Zhang L-Z, Zhao X-F, Si F-Z, Li J-H. Engineering Mesoporous NiO with Enriched Electrophilic Ni3+ and O− toward Efficient Oxygen Evolution. Catalysts. 2018; 8(8):310. https://doi.org/10.3390/catal8080310
Chicago/Turabian StyleLiu, Xiu, Zhi-Yuan Zhai, Zhou Chen, Li-Zhong Zhang, Xiu-Feng Zhao, Feng-Zhan Si, and Jian-Hui Li. 2018. "Engineering Mesoporous NiO with Enriched Electrophilic Ni3+ and O− toward Efficient Oxygen Evolution" Catalysts 8, no. 8: 310. https://doi.org/10.3390/catal8080310
APA StyleLiu, X., Zhai, Z.-Y., Chen, Z., Zhang, L.-Z., Zhao, X.-F., Si, F.-Z., & Li, J.-H. (2018). Engineering Mesoporous NiO with Enriched Electrophilic Ni3+ and O− toward Efficient Oxygen Evolution. Catalysts, 8(8), 310. https://doi.org/10.3390/catal8080310