Heterogeneous Biocatalysts Prepared by Immuring Enzymatic Active Components inside Silica Xerogel and Nanocarbons-In-Silica Composites
Abstract
:1. Introduction
2. Results and Discussion
2.1. Procedure for Immuring Enzymatic Active Substances Inside Silica Xerogel and Nanocarbons-In-SilicaComposites
2.2. Selection of the Optimal Compositions of the Biocatalysts
3. Texture of the Biocatalysts
4. Heterogeneous Whole-Cell and Lysates-Based Biocatalysts
4.1. Glucose Isomerase-Active Biocatalyst
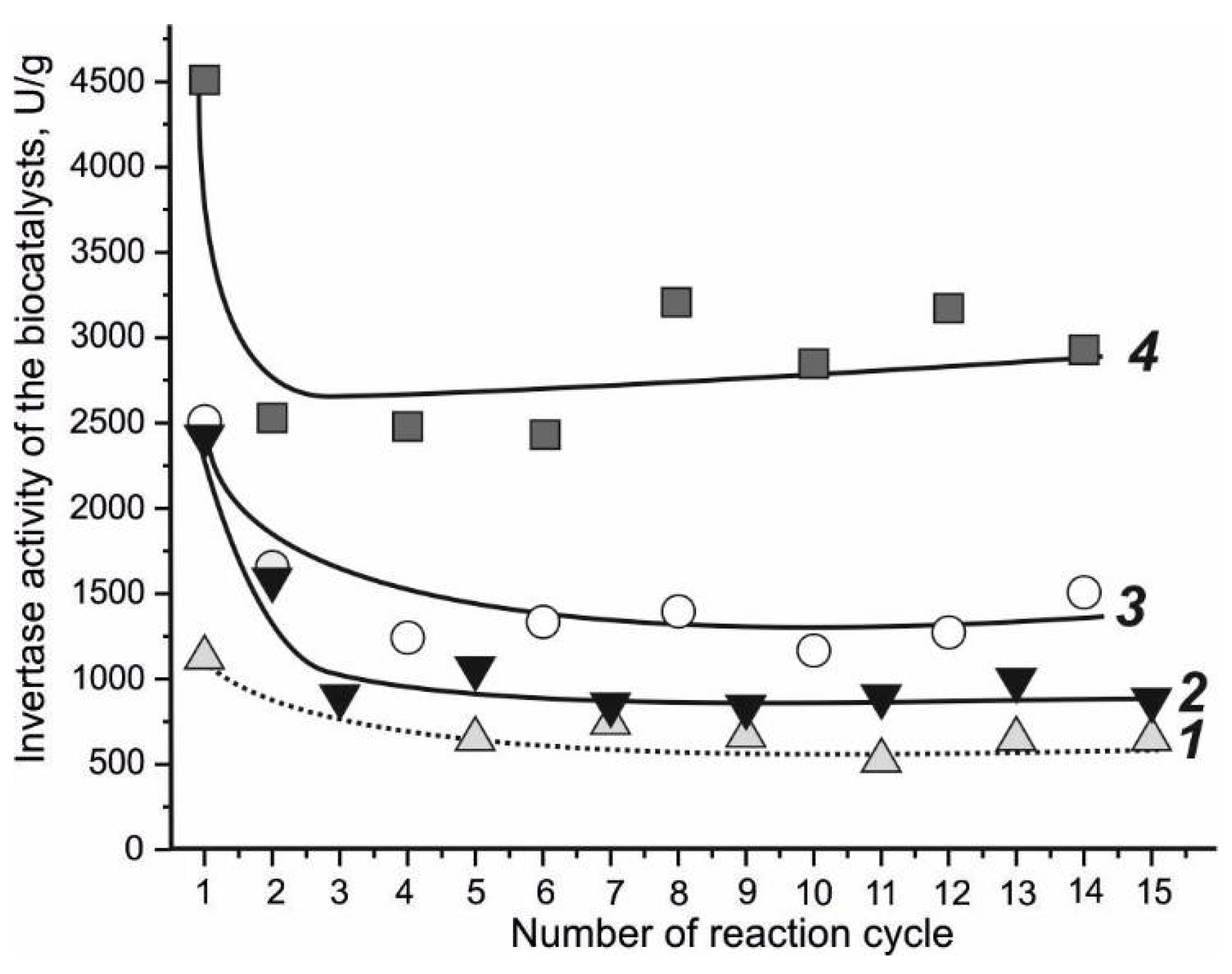
4.2. Invertase-Active Biocatalyst
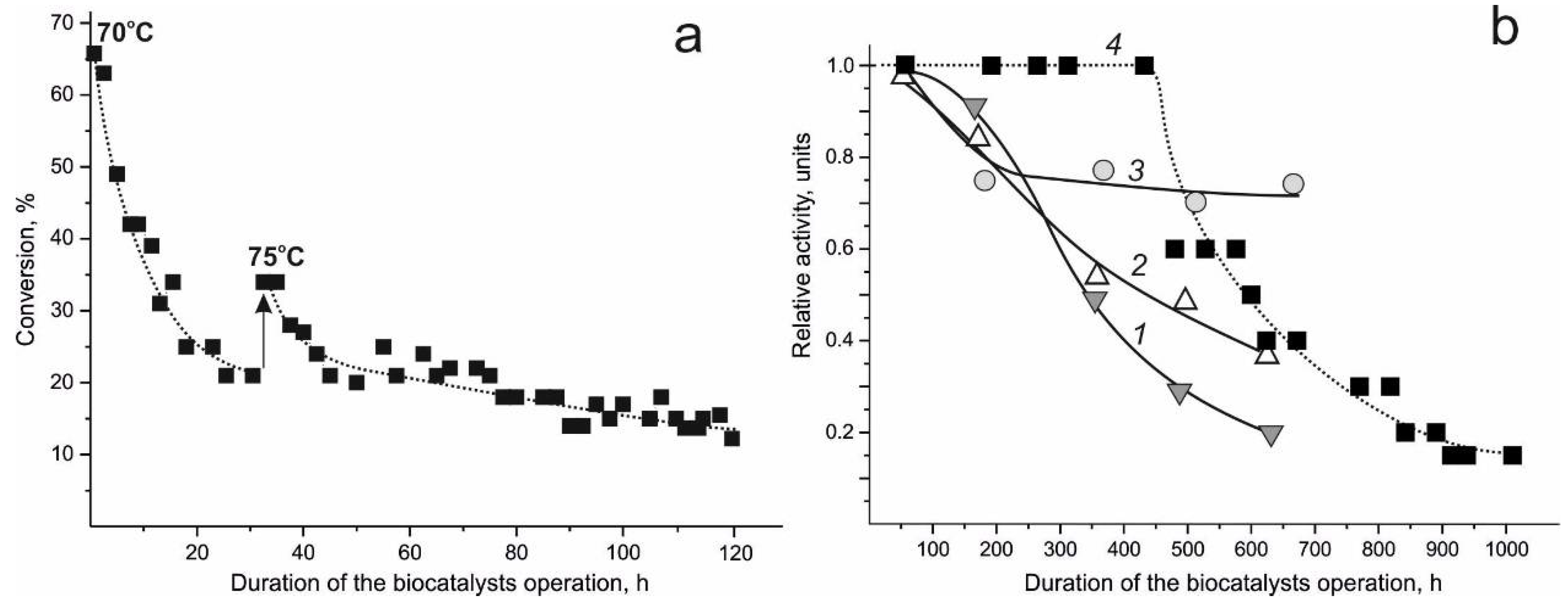
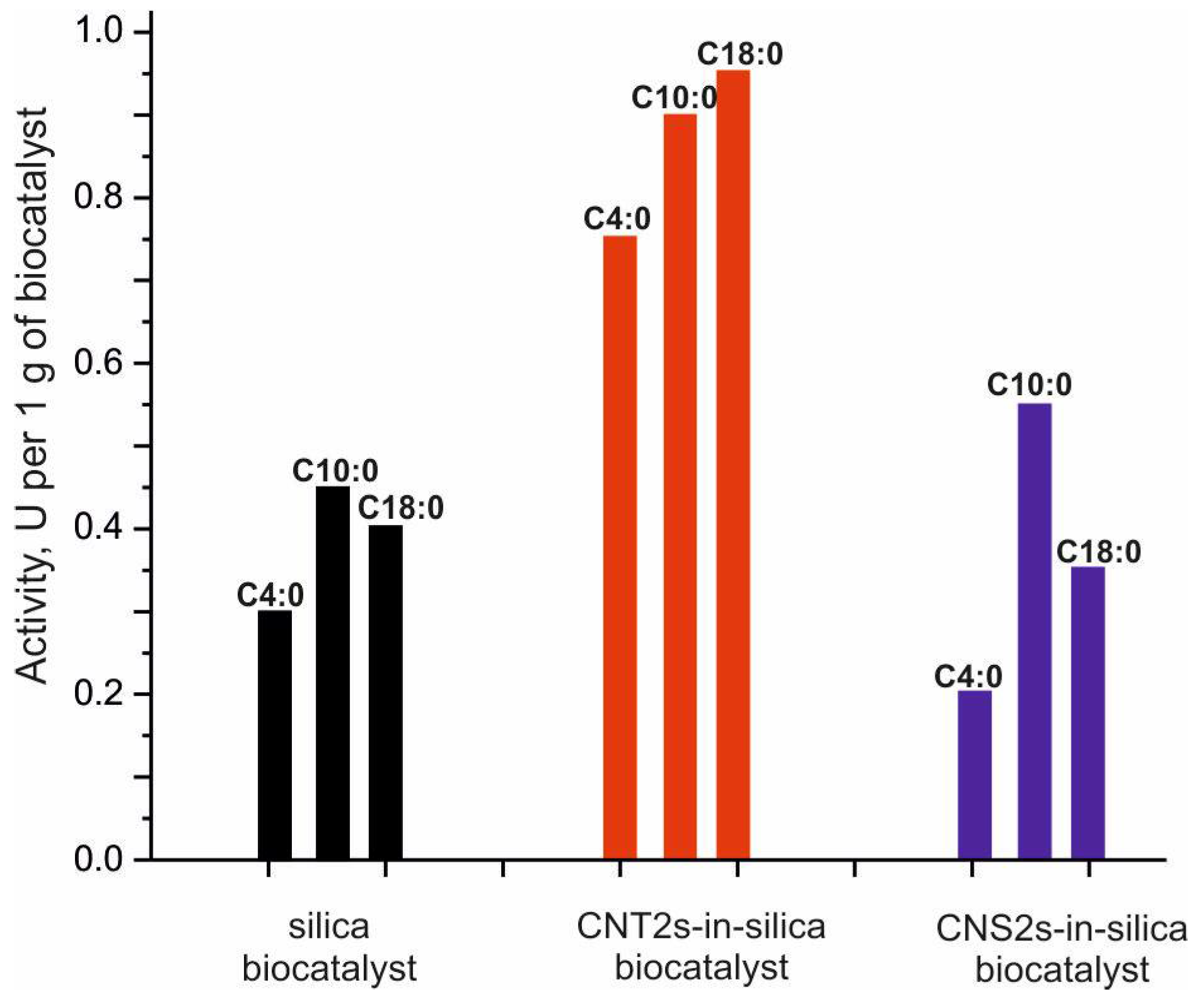
4.3. Lipase-Active Biocatalyst
5. Materials and Methods
5.1. Enzymatic Active Substances
5.1.1. Glucose Isomerase Active Component of the Biocatalysts
5.1.2. Invertase Active Component of the Biocatalysts
5.1.3. Lipase Active Component of the Biocatalysts
5.2. Silica Hydrogel and Nanocarbon Materials
5.3. Biocatalytic Processes of Substrate Conversion
5.3.1. Substrates and Reagents
5.3.2. Monosaccharide (Glucose, Fructose) Isomerization
5.3.3. Sucrose Inversion
5.3.4. Interesterification of Oil-Fat Blends
5.3.5. Interesterification of Vegetable Oil with Ethyl Acetate
5.3.6. Esterification of Fatty Acids with Aliphatic Alcohols
6. Main Equipment
7. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
Abbreviations
| CNT | carbon nanotube, multi-walled |
| CNF | carbon nanofiber |
| CNS | carbon nanosphere, onion-like |
| EAC | enzymatic active component of the heterogeneous biocatalyst |
| GI | glucose isomerase |
| INV | invertase |
| LIP | lipase |
| U | Unit of enzyme activity defined as μmols of conversed substrate per minute under the conditions described |
References
- Bommarius, A.S.; Reidel, B.R. Biocatalysis; WILEY-VCH Veglar GmbH& Co. RGaA: Weiheim, Germany, 2004; pp. 2–611. ISBN 3-527-30344-8. [Google Scholar]
- Grunwald, P. Biocatalysis; Imperial College Press: London, UK, 2009; pp. 2–1035. ISBN 10-1-86094-744-1. [Google Scholar]
- Buchholz, K.; Kasche, V.; Bornscheuer, U.T. Biocatalysts and Enzyme Technology; WILEY-VCH Veglar GmbH& Co. RGaA: Weiheim, Germany, 2005; pp. 2–431. ISBN 10-3-527-30497-5. [Google Scholar]
- Hou, C.H. Handbook of Industrial Biocatalysis; Tailor & Francis Group, LLC: Abingdon, UK, 2005; pp. 2–900. ISBN 0-8247-2423-2. [Google Scholar]
- Tao, J.; Kazlauskas, R. (Eds.) Biocatalysis for Green Chemistry and Chemical Process Development; WILEY, John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2011; pp. 1–479. ISBN 978-0-470-43778-0. [Google Scholar]
- Rodgers, L.E.; Holden, P.J.; Hanna, J.V.; Foster, L.J.R.; Knott, R.B.; Pike, K.J.; Bartlett, J.R. Structural evolution and stability of sol–gel biocatalysts. Physica B 2006, 385–386, 508–510. [Google Scholar] [CrossRef]
- Illanes, A.; Wilson, L.; Vera, C. Problem Solving in Enzyme Biocatalysis; WILEY, John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2014; pp. 141–180. ISBN 978-1-118-34171-1. [Google Scholar]
- Kovalenko, G.A.; Sukhinin, S.V.; Perminova, L.V. Vortex Reactors for Heterogeneous Biocatalytical Processes. In Industrial Application of Biotechnology; Krylov, I.A., Zaikov, G.E., Eds.; NOVA Science Publisher, Inc.: New York, NY, USA, 2006; pp. 45–53. [Google Scholar]
- Kovalenko, G.A.; Perminova, L.V. Immobilization of glucoamylase by adsorption on carbon supports and its application for heterogeneous hydrolysis of dextrin. Carbohydr. Res. 2008, 343, 1202–1211. [Google Scholar] [CrossRef] [PubMed]
- Kovalenko, G.A.; Perminova, L.V.; Terentyeva, T.G.; Sapunova, L.I.; Lobanok, A.G.; Chuenko, T.V.; Rudina, N.A.; Cherhyak, E.I. Glucose isomerase activity in suspensions of Arthrobacter nicotianae cells and adsorption immobilization of the microorganisms on inorganic carriers. Appl. Biochem. Microbiol. 2008, 44, 174–181. [Google Scholar] [CrossRef]
- Kovalenko, G.A.; Perminova, L.V.; Rudina, N.A.; Maksimova, Y.G.; Maksimov, A.Y. Sapropel-based supports as novel macroporous carbon-mineral adsorbents for enzymatic active substances. Resour. Effic. Technol. 2016, 2, 159–167. [Google Scholar] [CrossRef]
- Perminova, L.V.; Kovalenko, G.A.; Rudina, N.A.; Sapunova, L.I.; Tamkovic, I.O.; Lobanok, A.G. Catalytical properties of Arthrobacter nicotianae cells, a producer of glucose isomerase, immobilized inside xerogel of silicium dioxide. Appl. Biochem. Microbiol. 2009, 45, 389–394. [Google Scholar] [CrossRef]
- Kovalenko, G.A.; Perminova, L.V.; Chernyak, E.I.; Sapunova, L.I. Investigation on macrokinetics of heterogeneous process of monosaccharide isomerization using non-growing cells of a glucose isomerase producer Arthrobacter nicotianae immobilized inside SiO2-xerogel. Appl. Biochem. Microbiol. 2010, 46, 697–705. [Google Scholar] [CrossRef]
- Kovalenko, G.A.; Perminova, L.V.; Sapunova, L.I. A peculiar method for immobilization of non-growing microbial cells by entrapment into silica xerogel. In Biotechnology in Medicine, Foodstuffs, Biocatalysis, Environment and Biogeotechnology; Varfolomeev, S.D., Zaikov, G.E., Krylova, L.P., Eds.; NOVA Science Publisher, Inc.: New York, NY, USA, 2010; pp. 41–49. [Google Scholar]
- Kovalenko, G.A.; Simonova, L.G.; Perminova, L.V.; Yakushko, R.I. Biocatalyst, Method of Its Preparation and Method of Production of Glucose-Fructose Syrups. Patent RU 2341560 C1, 10 June 2008. [Google Scholar]
- Kovalenko, G.A.; Beklemishev, A.B.; Perminova, L.V.; Mamaev, A.L.; Rudina, N.A.; Moseenkov, S.I.; Kuznetsov, V.L. Immobilization of recombinant E. coli thermostable lipase by entrapment inside silica xerogel and nanocarbon-in-silica composites. J. Mol. Catal. B Enzym. 2013, 98, 78–86. [Google Scholar] [CrossRef]
- Kovalenko, G.A.; Perminova, L.V.; Beklemishev, A.B.; Tkachenko, V.I. Study on physicochemical properties of biocatalysts with thermostable lipase activity and final products of triglycerides’ interesterification. Appl. Biochem. Microbiol. 2014, 50, 709–721. [Google Scholar] [CrossRef]
- Kovalenko, G.A.; Perminova, L.V.; Rudina, N.A.; Mazov, I.N.; Moseenkov, S.I.; Kuznetsov, V.L. Immobilization of enzymatic active substances by immuring inside nanocarbon-in-silica composites. J. Mol. Catal. B Enzym. 2012, 76, 116–124. [Google Scholar] [CrossRef]
- Kovalenko, G.A.; Beklemishev, A.B.; Perminova, L.V.; Chuenko, T.V.; Mamaev, A.L.; Ivanov, I.D.; Moseenkov, S.I.; Kuznetsov, V.L. Recombinant strain producing thermostable lipase from Thermomyces lanuginosus immobilized into nanocarbon-in-silica matrixes and properties of prepared biocatalysts. Appl. Biochem. Microbiol. 2013, 49, 296–305. [Google Scholar] [CrossRef]
- Kovalenko, G.A.; Perminova, L.V.; Lenskaya, V.M.; Sapunova, L.I.; Tamkovich, I.O.; Lobanok, A.G. Biocatalyst, Method of Its Preparation and Method of Production of Glucose-Fructose Syrups. Patent Eurasia 01766 B1, 30 November 2012. [Google Scholar]
- Kovalenko, G.A.; Perminova, L.V.; Chuenko, T.V.; Sapunova, L.I.; Shlyakhotko, E.A.; Lobanok, A.G. Immobilization of a recombinant strain producing glucose isomerase inside SiO2-xerogel and properties of prepared biocatalysts. Appl. Biochem. Microbiol. 2011, 47, 151–157. [Google Scholar] [CrossRef]
- D’Souza, S.F.; Melo, J.S. Immobilization of bakers yeast on jute fabric through adhesion using polyethyleneimine: Application in an annular column reactor for the inversion of sucrose. Process. Biochem. 2001, 36, 677–681. [Google Scholar] [CrossRef]
- Sungur, S.; Al-Taweel, R.; Yildirim, O.; Logoglu, E. Immobilization of Saccharomyces cerevisiae in gelatin cross-linked with chromium ions for conversion of sucrose by intracellular invertase. Polym. Plast. Technol. Eng. 2006, 45, 929–934. [Google Scholar] [CrossRef]
- Hasal, P.; Vojtisek, V.; Lejkova, A.; Kleczek, P.; Kofronova, O. An immobilized whole yeast cell biocatalyst for enzymatic sucrose hydrolysis. Enzym. Microb. Technol. 1992, 14, 221–229. [Google Scholar] [CrossRef]
- Nassif, N.; Bouvet, O.; Rager, M.N.; Roux, C.; Coradin, T.; Livage, J. Living bacteria in silica gels. Nat. Mater. 2002, 1, 42–44. [Google Scholar] [CrossRef] [PubMed]
- Carturan, G.; Campostrini, R.; Dir, S.; Scardi, V.; De Alteriis, E. Inorganic gels for immobilization of biocatalysts: Inclusion of invertase-active whole cells of yeast (Saccharomyces cerevisiae) into thin layers of SiO2 gel deposited on glass sheets. J. Mol. Catal. 1989, 57, L13–L16. [Google Scholar] [CrossRef]
- Kovalenko, G.A.; Perminova, L.V. Biocatalyst, Method of Its Preparation and Method of Production of Invert Syrup Using This Biocatalyst. Patent RU 2372403 C1, 10 November 2009. [Google Scholar]
- Kovalenko, G.A.; Perminova, L.V.; Plaksin, G.V.; Komova, O.V.; Chuenko, T.V.; Rudina, N.A. Immobilized yeast membranes as biocatalysts for sucrose inversion. Appl. Biochem. Microbiol. 2005, 41, 399–403. [Google Scholar] [CrossRef]
- Mateo, C.; Palomo, J.M.; Lorente, G.; Guisan, J.M.; Fernandez-Lafuente, R. Improvement of enzyme activity, stability and selectivity via immobilization techniques. Enzym. Microb. Technol. 2007, 40, 1451–1463. [Google Scholar] [CrossRef]
- Rodrigues, R.C.; Fernandez-Lafuente, R. Lipase from Rhizomucor miehei as an industrial biocatalyst in chemical process. J. Mol. Catal. B Enzym. 2010, 64, 1–22. [Google Scholar] [CrossRef]
- Rodrigues, R.C.; Fernandez-Lafuente, R. Lipase from Rhizomucor miehei as a biocatalyst in fats and oils modification. J. Mol. Catal. B Enzym. 2010, 66, 15–32. [Google Scholar] [CrossRef]
- Contesini, F.J.; Lopes, D.B.; Macedo, G.A.; da G. Nascimento, M.; de O. Carvalho, P. Aspergillus sp. lipase: Potential biocatalyst for industrial use. J. Mol. Catal. B Enzym. 2010, 67, 163–171. [Google Scholar] [CrossRef]
- Fernandez-Lafuente, R. Lipase from Thermomyces lanuginosus: Uses and prospects as an industrial biocatalyst. J. Mol. Catal. B Enzym. 2010, 62, 197–212. [Google Scholar] [CrossRef]
- Itabaiana, I., Jr.; Miranda, L.S.M.; Mendon, R.O.; de Souza, R.O.M.A. Towards a continuous flow environment for lipase-catalyzed reactions. J. Mol. Catal. B Enzym. 2013, 85–86, 1–9. [Google Scholar] [CrossRef]
- Bezborodov, A.M.; Zagustina, N.A. Lipases in catalytic reactions of organic synthesis (review). Appl. Biochem. Microbiol. 2014, 50, 313–337. [Google Scholar] [CrossRef]
- Perminova, L.V.; Kovalenko, G.A.; Beklemishev, A.B.; Mamaev, A.L.; Pykhtina, M.B.; Rudina, N.A. Catalytic properties of lipase entrapped as lysates of recombinant strain-producer rEscherichia coli/lip into nanocarbon-in-silica composites in the bioconversion of triglycerides and fatty acids. Appl. Biochem. Microbiol. 2018, 54, 38–44. [Google Scholar] [CrossRef]
- Sokolovskii, V.D.; Kovalenko, G.A. Immobilization of oxydoreductases on inorganic supports based on alumina: The role of mutual correspondence of enzyme-support hydrophobic-hydrophilic characters. Biotechnol. Bioeng. 1988, 32, 916–919. [Google Scholar] [CrossRef] [PubMed]
- Kovalenko, G.A.; Sokolovskii, V.D. Immobilization of oxydoreductases on inorganic supports based on alumina: Immobilization of alcohol dehydrogenase on nonmodified and modified alumina. Biotechnol. Bioeng. 1983, 25, 3177–3184. [Google Scholar] [CrossRef] [PubMed]
- Kovalenko, G.A.; Perminova, L.V.; Chuenko, T.V.; Rudina, N.A. Tuning surface morphology of inorganic supports for adsorptive immobilization of enzymatic active substances. Compos. Interfaces 2009, 16, 293–305. [Google Scholar] [CrossRef]
- Kovalenko, G.A.; Beklemishev, A.B.; Perminova, L.V.; Mamaev, A.L.; Chuenko, T.V.; Kuznetsov, V.L. Biocatalyst, Method of Its Preparation and Method of Interesterification of Vegetable Oils Using This Biocatalyst. Patent RU 2539101 C2, 10 January 2014. [Google Scholar]
- Kovalenko, G.A.; Perminova, L.V.; Beklemishev, A.B.; Yakovleva, E.Y.; Pykhtina, M.B. Heterogeneous biocatalytic processes of vegetable oil interesterification to biodiesel. Catal. Ind. 2015, 7, 73–81. [Google Scholar] [CrossRef]
- Kovalenko, G.A.; Perminova, L.V.; Beklemishev, A.B.; Mamaev, A.L.; Patrushev, Y.V. Heterogeneous biocatalytic processes of esterification of saturated fatty acids with aliphatic alcohols. Catal. Ind. 2018, 10, 68–74. [Google Scholar] [CrossRef]
- Perminova, L.V.; Kovalenko, G.A.; Chukanov, N.V.; Patrushev, Y.V. Enzymatic esterification of saturated fatty acids with aliphatic alcohols as an alternative method of a low-temperature synthesis of esters. Russ. Chem. Bull. 2017, 66, 2194–2197. [Google Scholar] [CrossRef]
- Joseph, M.D.; Kasprzak, D.J.; Crouch, S.R. A stopped-flow clinical analyzer in which immobilized-enzyme reaction loops are used. Clin. Chem. 1977, 23, 1033–1036. [Google Scholar] [PubMed]
- Usoltseva, A.N.; Kuznetsov, V.L.; Rudina, N.A.; Moroz, E.M.; Haluska, M.; Roth, S. Influence of catalysts activation on their activity and selectivity in carbon nanotubes synthesis. Phys. Status Solidi 2007, 244, 3920–3924. [Google Scholar] [CrossRef]
- Kuznetsov, V.L.; Chuvilin, A.L.; Moroz, E.M.; Kolomiichuk, V.N.; Shaichutdinov, S.K.; Butenko, Y.V.; Malkov, I.Y. Effect of explosion conditions on the structure of detonation soot: Ultra disperse diamond and onion carbon. Carbon 1994, 32, 873–882. [Google Scholar] [CrossRef]
- Kuznetsov, V.L.; Chuvilin, A.L.; Butenko, Y.V.; Malkov, I.Y.; Titov, V.M. Onion-like carbon from ultra disperse diamond. Chem. Phys. Lett. 1994, 222, 343–348. [Google Scholar] [CrossRef]
- Kuvshinov, G.G.; Mogilnykh, Y.I.; Kuvshinov, D.G.; Zaikovskii, V.I.; Avdeeva, L.B. Pecularities of filamentous carbon formation in metane decomposition in Ni-containing catalysts. Carbon 1998, 36, 87–97. [Google Scholar] [CrossRef]
- Kuvshinov, G.G.; Mogilnykh, Y.I.; Kuvshinov, D.G. Kinetics of carbon formation from CH4-H2 mixtures over a nickel containing catalyst. Catal. Today. 1998, 42, 357–360. [Google Scholar] [CrossRef]
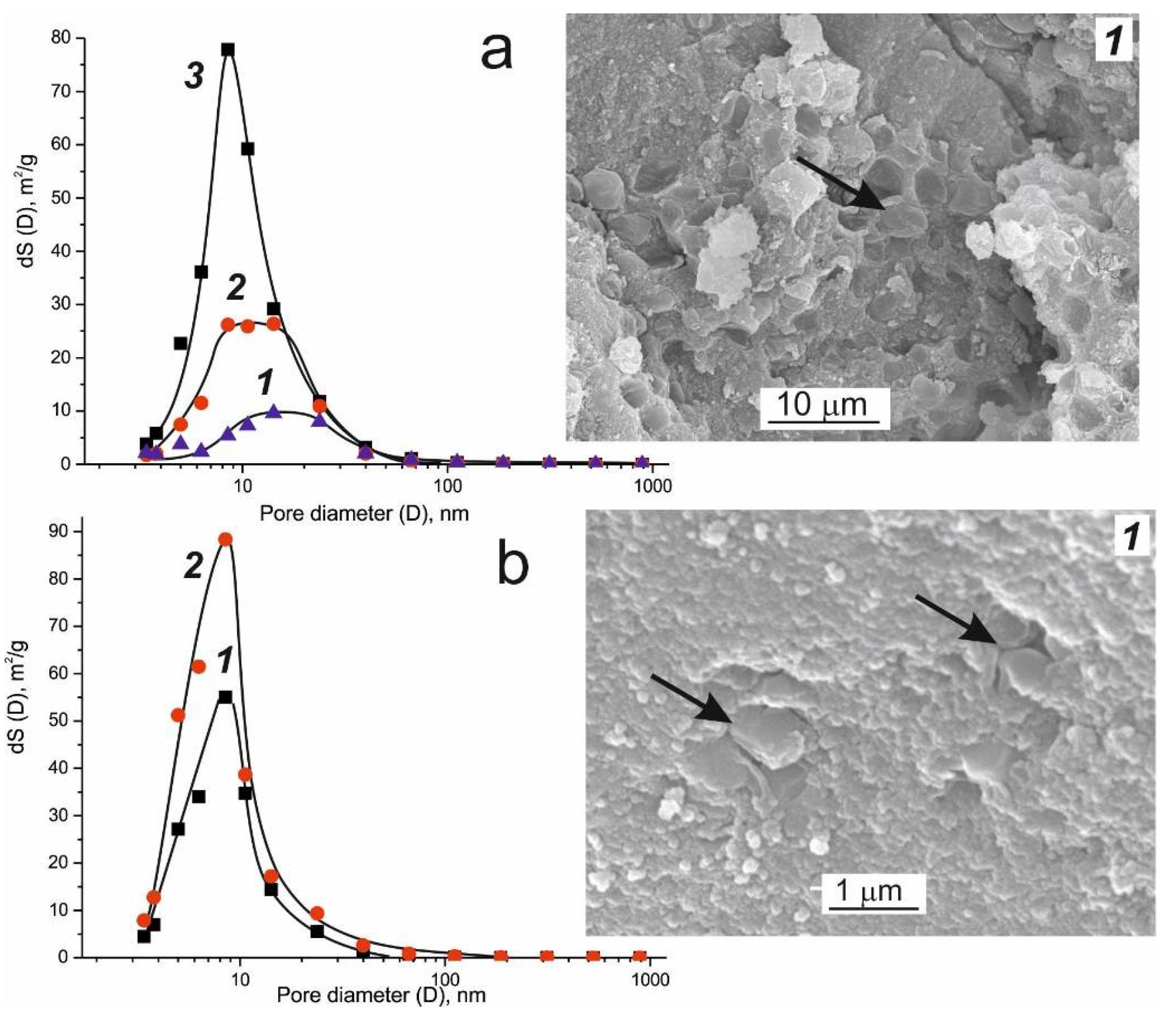
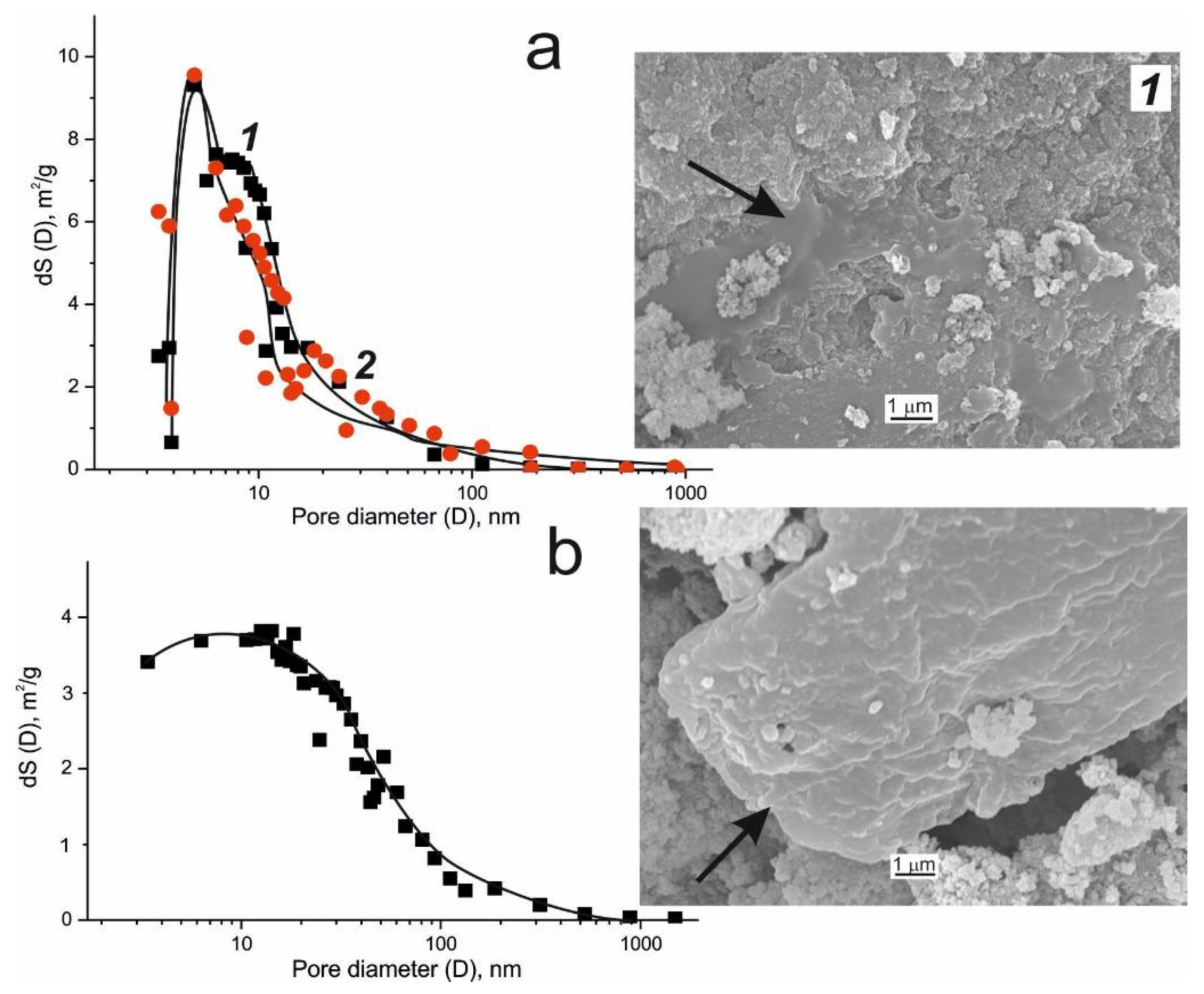
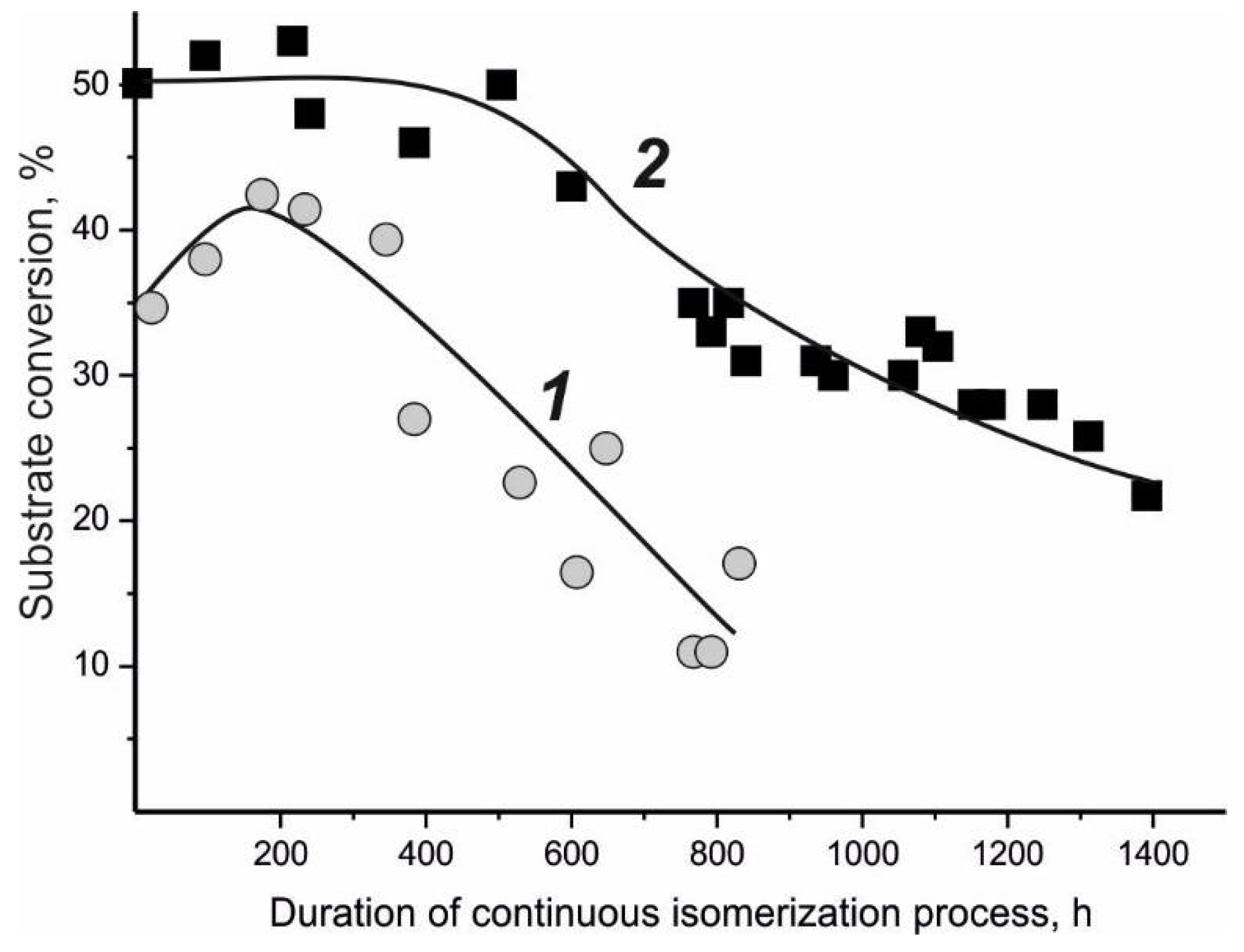
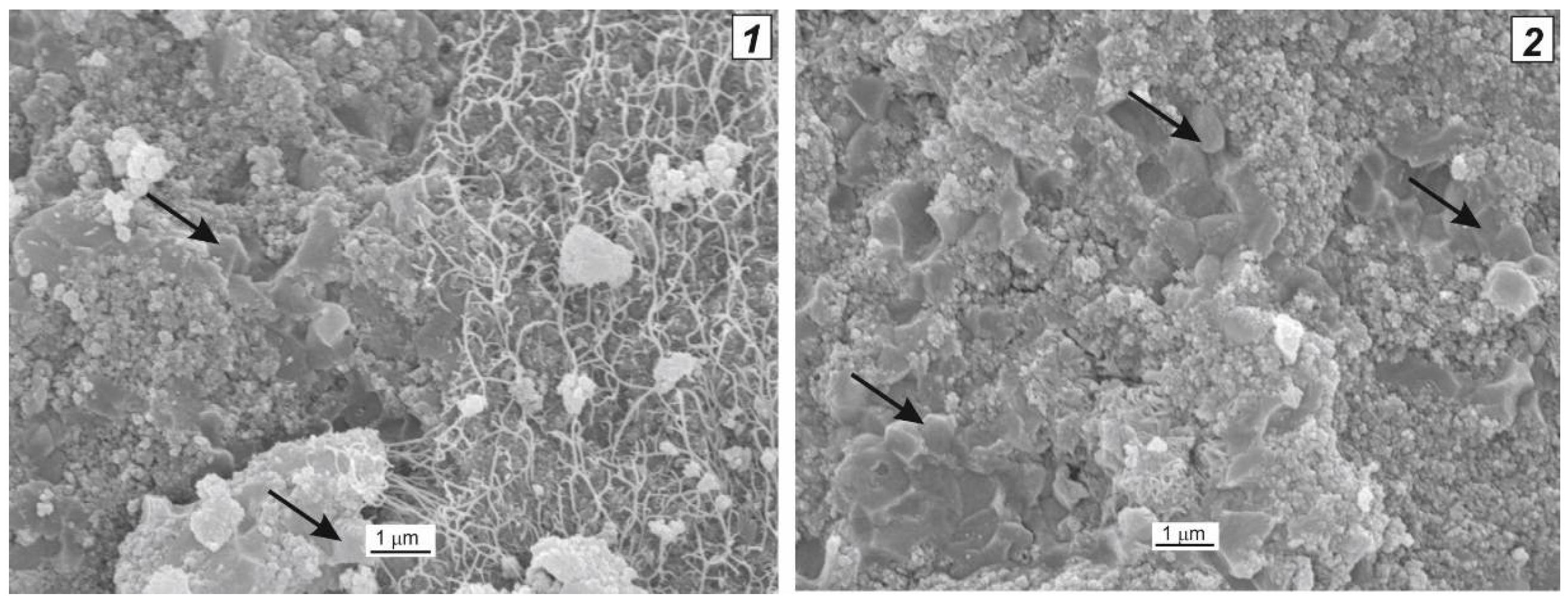


| Microorganism | Biocatalytical Process | Optimal Composition, w/w % | ||||
|---|---|---|---|---|---|---|
| Microbial Biomass | СоxOy | Maltodextrin | Carbon Nanotubes | SiO2 | ||
| S. cerevisiae | Sucrose inversion | 60–80 | 0 | 0 | 5–10 | 20–30 |
| A. nicotianae | Glucose/fructose isomerization | 10–15 | 20–40 | 0 | 0 | 45–70 |
| rE. coli/xyl | 35–40 | 10–40 | 0 | 0–5 | 20–40 | |
| rE. coli/lip | Tributyrin hydrolysis | 35–40 | 0 | 0 | 5–10 | 50–55 |
| Interesterification | 35–40 | 0 | 10–20 | 0 | 40–55 | |
| Microorganism | Biocatalyst Composition, w/w % of Dry Substances | Ssp.BET, m2/g | VΣ, cm3/g | Dpore, nm | |||
|---|---|---|---|---|---|---|---|
| Biomass | SiO2 | CoSO4 | CoxOy | ||||
| S. cerevisiae | 15 | 85 | 250 | 0.6 | 22 | ||
| 20 | 80 | 220 | 0.9 | 15 | |||
| 60 | 40 | 100 | 0.6 | 22 | |||
| 80 | 20 | 35 | 0.4 | 36 | |||
| A. nicotianae | 15 | 85 | 0.06 | 230 | 0.9 | 15 | |
| 10 | 50 | 40 | 175 | 0.6 | 13 | ||
| rE. coli/xyl | 40 | 40 | 20 | 75 | 0.35 | 19 | |
| rE. coli/lip | 40 | 60 | 90 | 0.6 | 25 | ||
| Type of Included Nanocarbon * (Ssp.BET of Nanocarbons) | Baker Yeast Autolysate-Based Biocatalysts | rE. coli/lip Lysate-Based Biocatalysts | ||
|---|---|---|---|---|
| Ssp.BET, m2/g | Dpore, nm | Ssp.BET, m2/g | Dpore, nm | |
| Without nanocarbons | 110 | 11 | 105 | 31 |
| Carbon nanotubes CNT1 (320 m2/g) | 105 | 19 | 90 | 25 |
| Carbon nanofibers CNF (160 m2/g) | 60 | 12 | ||
| Carbon nanospheres CNS1 (485 m2/g) | 95 | 11 | 110 | 22 |
| Nanodiamond ND (325 m2/g) | 80 | 14 | ||
| Type of Nanocarbon Included in Content of 10 w/v % | Enzymatic Active Component | ||
|---|---|---|---|
| Partially Disrupted rE. сoli/lip Cells | Lysates of rE. сoli/lip Cells | His6×Lipase Purified * | |
| Without nanocarbons | 210 | 870 | 510 |
| Multi-walled CNT1s (5–11 nm in diameter) aggregated | 220 | 1050 | 120 |
| Multi-walled CNT1s (5–11 nm in diameter) dispergated | 50 | 400 | 25 |
| Multi-walled CNT2s (20–22 nm in diameter) aggregated | 250 | 870 | 260 |
| Carbon onion-like nanospheres (5–6 nm in diameter) aggregated | 105 | 710 | |
| Type of Aggregated Nanocarbon Included ** | The Type of Reaction (Reaction Media) | |||
|---|---|---|---|---|
| Hydrolysis of Emulsified Tributyrin (Aqueous) | Interesterification in Oil-Fat Blends (Anhydrous) | Interesterification of Oil with Ethyl Acetate (in Hexane) | Esterification of Capric Acid with Isopentanol (in Hexane with Diethyl Ether) | |
| Without nanocarbons | 0.9 | 1.0 | 1.0 | 0.3 |
| CNT1 | 1.0 | 0.9 | 0.8 | |
| CNT2 | 0.8 | 0.9 | 1.0 | 1.0 |
| CNS1 | 0.7 | 0.5 | 0.4 | |
| CNS2 | 0.9 | 0.8 | 0.5 | |
| Type of Nanocarbon | Diameter of Primary Particle, nm | Ssp.BET, m2/g |
|---|---|---|
| CNT1 | 9–11 | 320 |
| CNT2 | 20–22 | 140 |
| CNF | 20–60 | 160 |
| CNS1 | 5–6 | 485 |
| CNS2 | 8–10 | 250 |
| ND | 4–6 | 325 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kovalenko, G.A.; Perminova, L.V.; Beklemishev, A.B.; Parmon, V.N. Heterogeneous Biocatalysts Prepared by Immuring Enzymatic Active Components inside Silica Xerogel and Nanocarbons-In-Silica Composites. Catalysts 2018, 8, 177. https://doi.org/10.3390/catal8050177
Kovalenko GA, Perminova LV, Beklemishev AB, Parmon VN. Heterogeneous Biocatalysts Prepared by Immuring Enzymatic Active Components inside Silica Xerogel and Nanocarbons-In-Silica Composites. Catalysts. 2018; 8(5):177. https://doi.org/10.3390/catal8050177
Chicago/Turabian StyleKovalenko, Galina A., Larisa V. Perminova, Anatoly B. Beklemishev, and Valentin N. Parmon. 2018. "Heterogeneous Biocatalysts Prepared by Immuring Enzymatic Active Components inside Silica Xerogel and Nanocarbons-In-Silica Composites" Catalysts 8, no. 5: 177. https://doi.org/10.3390/catal8050177
APA StyleKovalenko, G. A., Perminova, L. V., Beklemishev, A. B., & Parmon, V. N. (2018). Heterogeneous Biocatalysts Prepared by Immuring Enzymatic Active Components inside Silica Xerogel and Nanocarbons-In-Silica Composites. Catalysts, 8(5), 177. https://doi.org/10.3390/catal8050177





