Unprecedented Proline-Based Heterogeneous Organocatalyst for Selective Production of Vanillin
Abstract
:1. Introduction
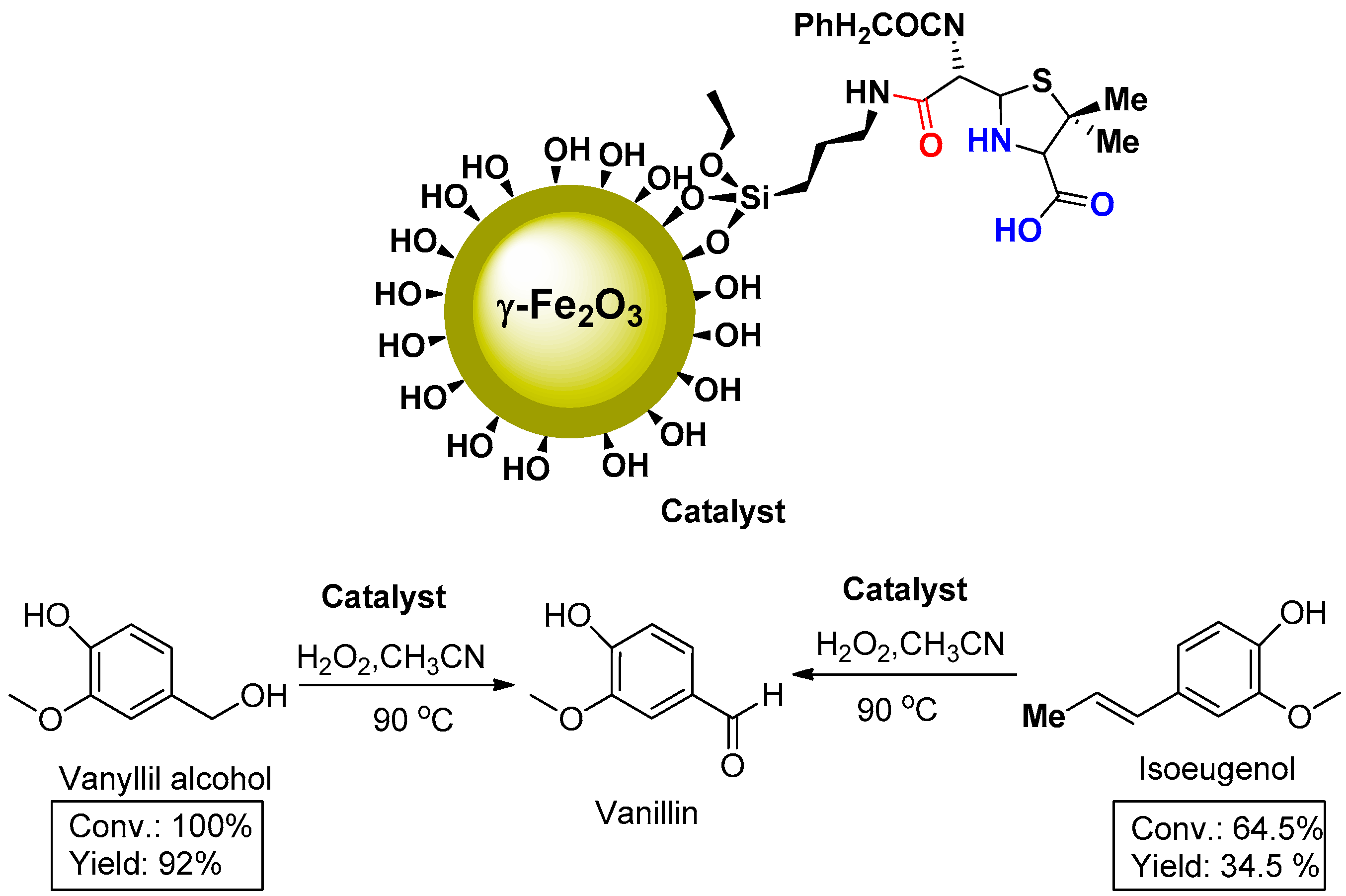
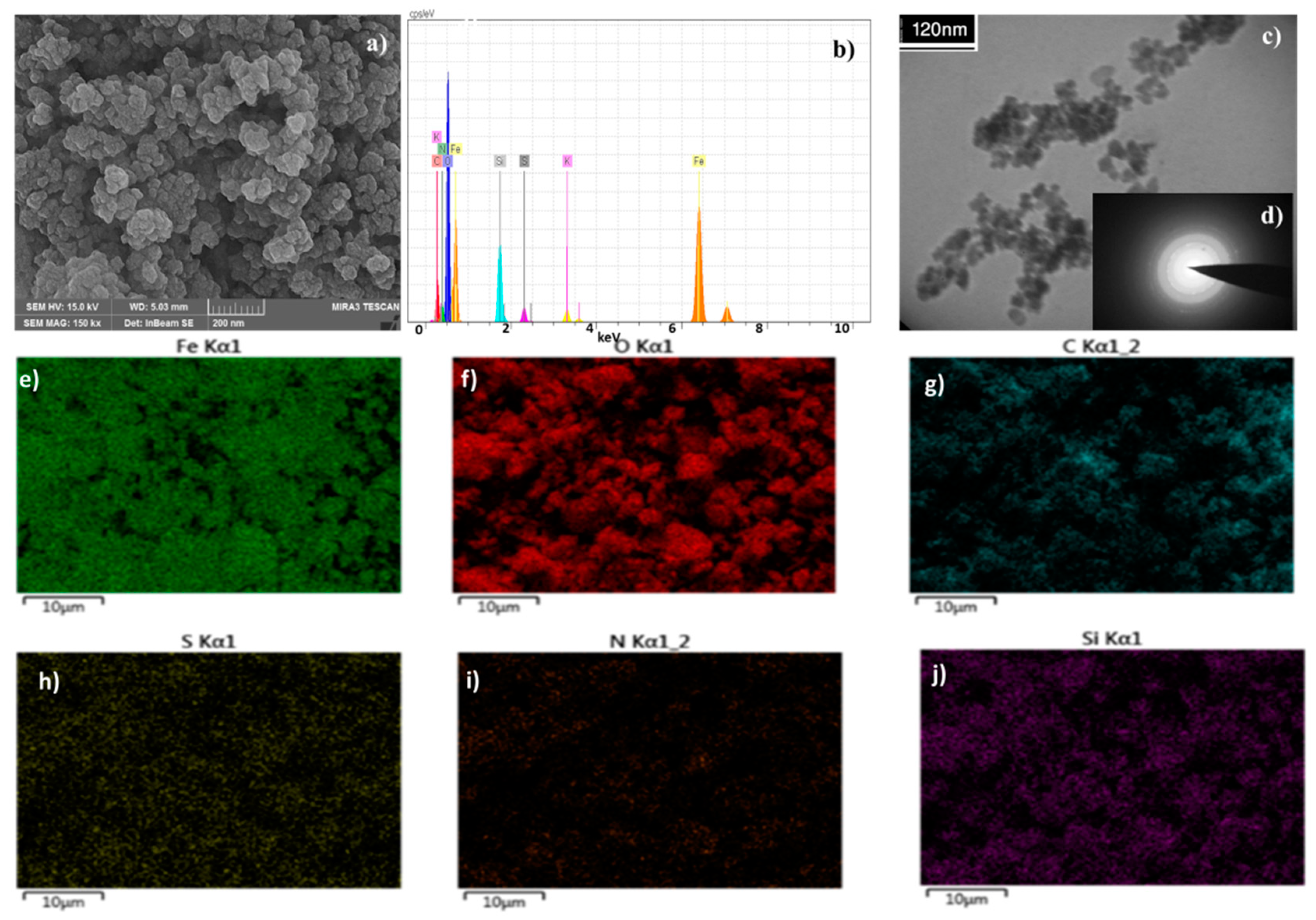
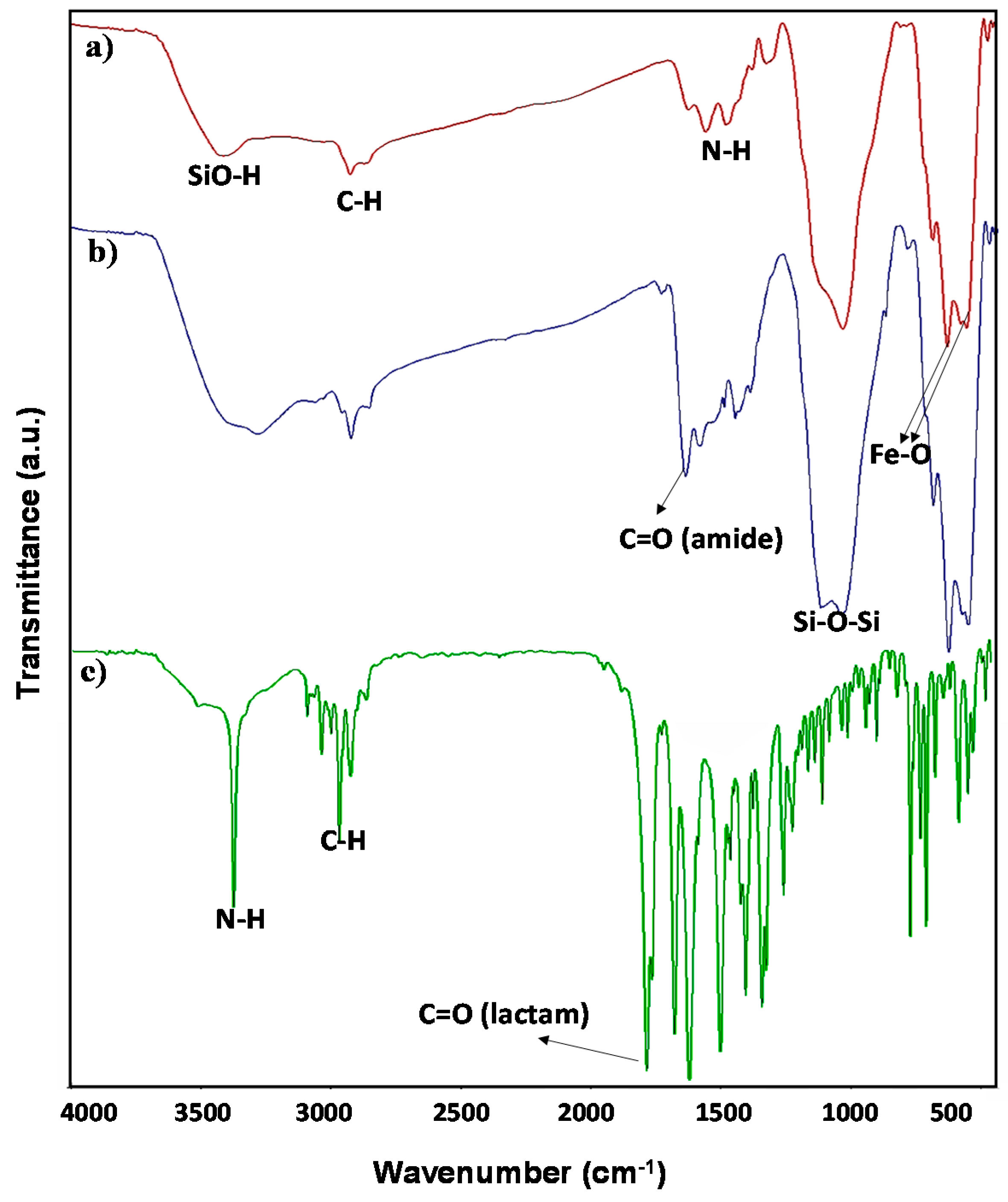
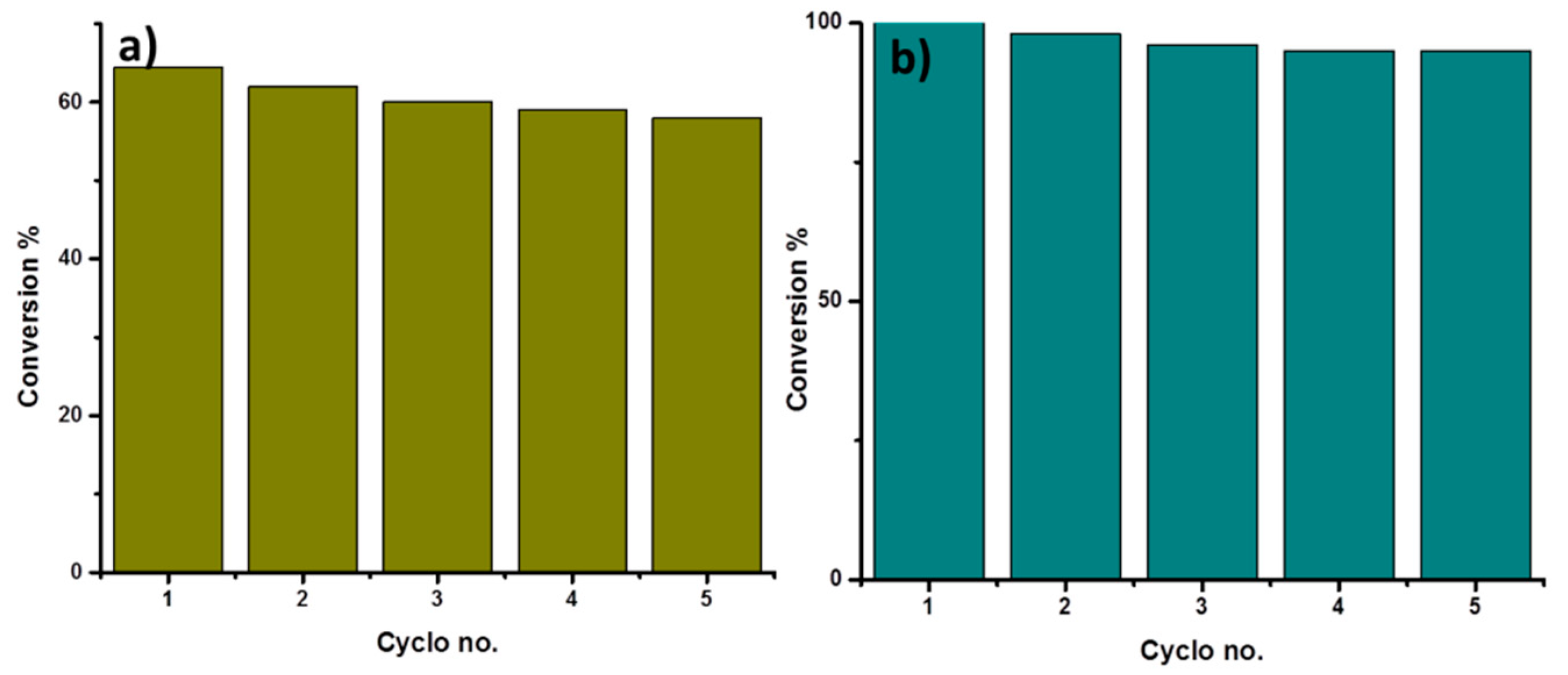
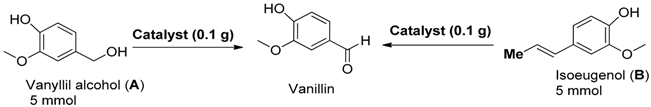
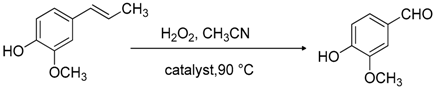
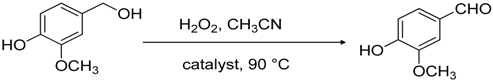
2. Results and Discussion
3. Materials and Methods
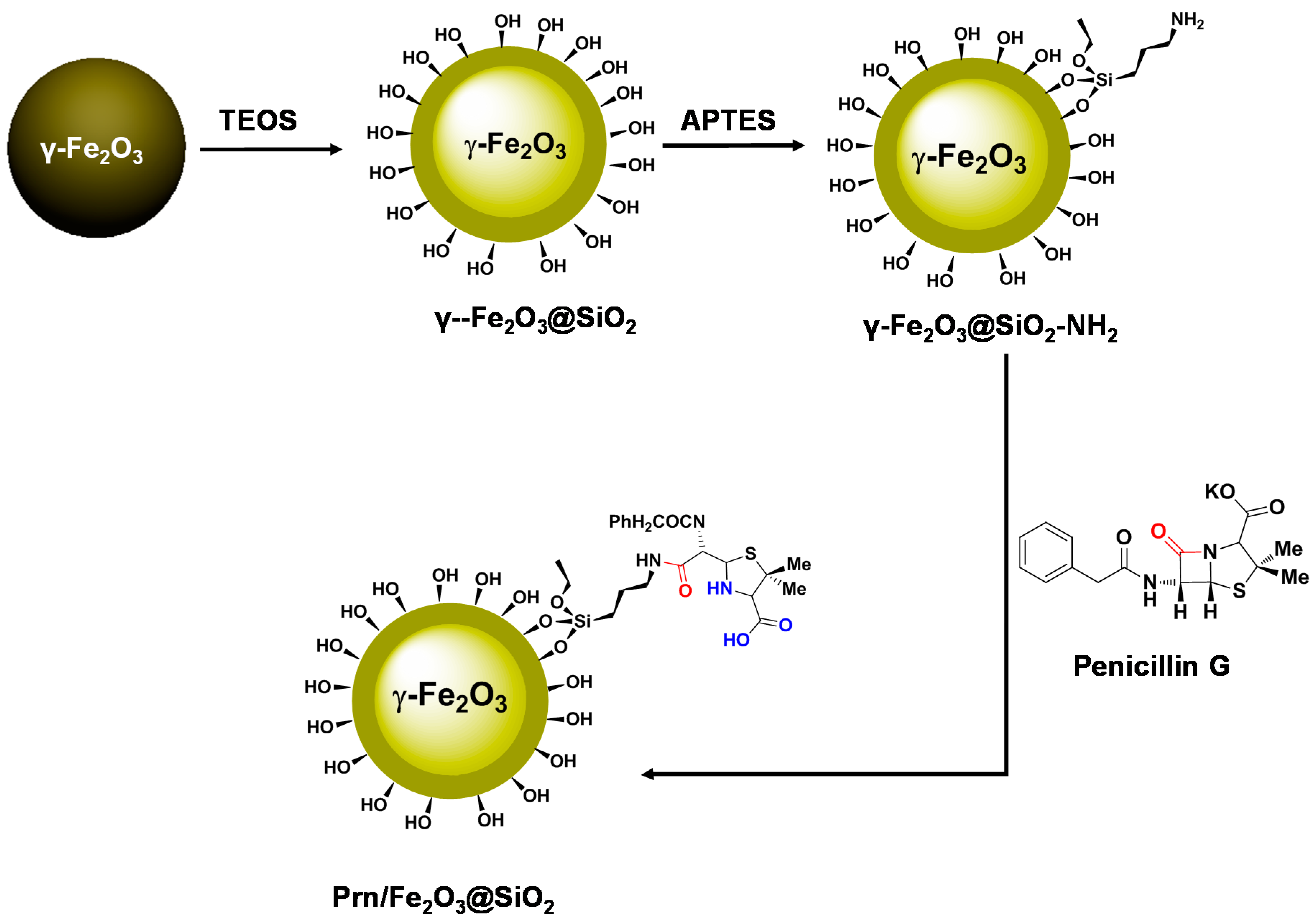
3.1. Synthesis of γ-Fe2O3@SiO2
3.2. Synthesis of Prn/γ-Fe2O3@SiO2
3.3. Catalytic Experiments
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Gusevskaya, E.V.; Menini, L.; Parreira, L.A.; Mesquita, R.A.; Kozlov, Y.N.; Shulpin, G.B. Oxidation of isoeugenol to vanillin by the “H2O2–vanadate–pyrazine-2-carboxylic acid. J. Mol. Catal. A Chem. 2012, 363–364, 140–147. [Google Scholar] [CrossRef]
- Huang, W.-B.; Du, C.-Y.; Jiang, J.-A.; Ji, Y.-F. Concurrent synthesis of vanillin and isovanillin. Res. Chem. Intermed. 2013, 39, 2849–2856. [Google Scholar] [CrossRef]
- Lampman, G.M.; Sharpe, S.D. A phase transfer catalyzed permanganate oxidation: Preparation of vanillin from isoeugenol acetate. J. Chem. Educ. 1983, 60, 503. [Google Scholar] [CrossRef]
- Franco, A.; De, S.; Balu, A.M.; Romero, A.A.; Luque, R. Selective Oxidation of Isoeugenol to Vanillin over Mechanochemically Synthesized Aluminosilicate Supported Transition Metal Catalysts. ChemistrySelect 2017, 2, 9546–9551. [Google Scholar] [CrossRef]
- Filiciotto, L.; Balu, A.M.; Romero, A.A.; Rodríguez-Castellón, E.; van der Waal, J.C.; Luque, R. Benign-by-design preparation of humin-based iron oxide catalytic nanocomposites. Green Chem. 2017, 19, 4423–4434. [Google Scholar] [CrossRef]
- Adilina, I.B.; Hara, T.; Ichikuni, N.; Shimazu, S. Oxidative cleavage of isoeugenol to vanillin under molecular oxygen catalysed by cobalt porphyrin intercalated into lithium taeniolite clay. J. Mol. Catal. A 2012, 361, 72–79. [Google Scholar] [CrossRef]
- Behling, R.; Valange, S.; Chatel, G. Heterogeneous catalytic oxidation for lignin valorization into valuable chemicals: What results? What limitations? What trends? Green Chem. 2016, 18, 1839–1854. [Google Scholar] [CrossRef]
- Behera, G.C.; Parida, K. Liquid phase catalytic oxidation of benzyl alcohol to benzaldehyde over vanadium phosphate catalyst. Appl. Catal. A 2012, 413, 245–253. [Google Scholar] [CrossRef]
- Marquez-Medina, M.D.; Prinsen, P.; Li, H.; Shih, K.; Romero, A.A.; Luque, R. Continuous-Flow Synthesis of Supported Magnetic Iron Oxide Nanoparticles for Efficient Isoeugenol Conversion into Vanillin. ChemSusChem 2018, 11, 389–396. [Google Scholar] [CrossRef] [PubMed]
- Watson, S.; Beydoun, D.; Amal, R. Synthesis of a novel magnetic photocatalyst by direct deposition of nanosized TiO2 crystals onto a magnetic core. J. Photochem. Photobiol. A 2002, 148, 303–313. [Google Scholar] [CrossRef]
- Polshettiwar, V.; Luque, R.; Fihri, A.; Zhu, H.; Bouhrara, M.; Basset, J.-M. Magnetically recoverable nanocatalysts. Chem. Rev. 2011, 111, 3036–3075. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Padrón, D.; Jodlowski, A.D.; de Miguel, G.; Puente-Santiago, A.R.; Balu, A.M.; Luque, R. Synthesis of carbon-based fluorescent polymers driven by catalytically active magnetic bioconjugates. Green Chem. 2018, 20, 225–229. [Google Scholar] [CrossRef]
- Lu, A.H.; Salabas, E.E.L.; Schüth, F. Magnetic nanoparticles: Synthesis, protection, functionalization, and application. Angew. Chem. Int. Ed. 2007, 46, 1222–1244. [Google Scholar] [CrossRef] [PubMed]
- Doustkhah, E.; Rostamnia, S.; Gholipour, B.; Zeynizadeh, B.; Baghban, A.; Luque, R. Design of chitosan-dithiocarbamate magnetically separable catalytic nanocomposites for greener aqueous oxidations at room temperature. Mol. Catal. 2017, 434, 7–15. [Google Scholar] [CrossRef]
- Kuhn, L.T.; Bojesen, A.; Timmermann, L.; Nielsen, M.M.; Mørup, S. Structural and magnetic properties of core–shell iron–iron oxide nanoparticles. J. Phys. Condens. Matter 2002, 14, 13551. [Google Scholar] [CrossRef]
- Rafiee, E.; Eavani, S. H3PW12O40 supported on silica-encapsulated γ-Fe2O3 nanoparticles: A novel magnetically-recoverable catalyst for three-component Mannich-type reactions in water. Green Chem. 2011, 13, 2116–2122. [Google Scholar] [CrossRef]
- Khalafi-Nezhad, A.; Divar, M.; Panahi, F. Magnetic nanoparticles-supported tungstic acid (MNP-TA): An efficient magnetic recyclable catalyst for the one-pot synthesis of spirooxindoles in water. RSC Adv. 2015, 5, 2223–2230. [Google Scholar] [CrossRef]
- Riente, P.; Mendoza, C.; Pericás, M.A. Functionalization of Fe3O4 magnetic nanoparticles for organocatalytic Michael reactions. J. Mater. Chem. 2011, 21, 7350–7355. [Google Scholar] [CrossRef]
- Doustkhah, E.; Rostamnia, S.; Imura, M.; Ide, Y.; Mohammadi, S.; Hyland, C.J.; You, J.; Tsunoji, N.; Zeynizadeh, B.; Yamauchi, Y. Thiourea bridged periodic mesoporous organosilica with ultra-small Pd nanoparticles for coupling reactions. RSC Adv. 2017, 7, 56306–56310. [Google Scholar] [CrossRef]
- Rostamnia, S.; Doustkhah, E. Nanoporous silica-supported organocatalyst: A heterogeneous and green hybrid catalyst for organic transformations. RSC Adv. 2014, 4, 28238–28248. [Google Scholar] [CrossRef]
- Polshettiwar, V.; Baruwati, B.; Varma, R.S. Magnetic nanoparticle-supported glutathione: A conceptually sustainable organocatalyst. Chem. Commun. 2009, 1837–1839. [Google Scholar] [CrossRef] [PubMed]
- Rostamnia, S.; Doustkhah, E. Increased SBA-15-SO3H catalytic activity through hydrophilic/hydrophobic fluoroalkyl-chained alcohols (RFOH/SBA-15–Pr-SO3H). Synlett 2015, 26, 1345–1347. [Google Scholar] [CrossRef]
- Rodríguez-Padrón, D.; Balu, A.M.; Romero, A.A.; Luque, R. New bio-nanocomposites based on iron oxides and polysaccharides, applied to oxidation and alkylation reactions. Beilstein J. Org. Chem. 2017, 13, 1982–1993. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.; Li, S.; Wang, X.; Zhang, F.; Zhong, X.; Dong, Z.; Ma, J. Core–shell silica magnetic microspheres supported proline as a recyclable organocatalyst for the asymmetric aldol reaction. J. Mol. Catal. A 2012, 363, 404–410. [Google Scholar] [CrossRef]
- Yang, G.; Zhou, L. Mechanisms and reactivity differences of proline-mediated catalysis in water and organic solvents. Catal. Sci. Technol. 2016, 6, 3378–3385. [Google Scholar] [CrossRef]
- Bhattacharjee, D.; Sutradhar, D.; Chandra, A.K.; Myrboh, B. l-proline as an efficient asymmetric induction catalyst in the synthesis of chromeno[2,3-d]pyrimidine-triones, xanthenes in water. Tetrahedron 2017, 73, 3497–3504. [Google Scholar] [CrossRef]
- Akceylan, E.; Uyanik, A.; Eymur, S.; Sahin, O.; Yilmaz, M. Calixarene-proline functionalized iron oxide magnetite nanoparticles (Calix-Pro-MN): An efficient recyclable organocatalyst for asymmetric aldol reaction in water. Appl. Catal. A 2015, 499, 205–212. [Google Scholar] [CrossRef]
- Zhang, H.; Han, M.; Chen, T.; Xu, L.; Yu, L. Poly (N-isopropylacrylamide-co-l-proline)-catalyzed Claisen–Schmidt and Knoevenagel condensations: Unexpected enhanced catalytic activity of the polymer catalyst. RSC Adv. 2017, 7, 48214–48221. [Google Scholar] [CrossRef]
- Aghahosseini, H.; Ramazani, A.; Ślepokura, K.; Lis, T. The first protection-free synthesis of magnetic bifunctional l-proline as a highly active and versatile artificial enzyme: Synthesis of imidazole derivatives. J. Colloid Interface Sci. 2018, 511, 222–232. [Google Scholar] [CrossRef] [PubMed]
- Khalafi-Nezhad, A.; Shahidzadeh, E.S.; Sarikhani, S.; Panahi, F. A new silica-supported organocatalyst based on l-proline: An efficient heterogeneous catalyst for one-pot synthesis of spiroindolones in water. J. Mol. Catal. A 2013, 379, 1–8. [Google Scholar] [CrossRef]
- Lee, S.; Shin, K.Y.; Jang, J. Enhanced magnetorheological performance of highly uniform magnetic carbon nanoparticles. Nanoscale 2015, 7, 9646–9654. [Google Scholar] [CrossRef] [PubMed]
- Yepez, A.; Hidalgo, J.M.; Pineda, A.; Černý, R.; Jíša, P.; Garcia, A.; Luque, R. Mechanistic insights into the hydroconversion of cinnamaldehyde using mechanochemically-synthesized Pd/Al-SBA-15 catalysts. Green Chem. 2015, 17, 565–572. [Google Scholar] [CrossRef]
- Rajpure, K.Y. Exploring structural and magnetic properties of nanocrystalline iron oxide synthesized by autocombustion method. Superlattices Microstruct. 2015, 77, 181–195. [Google Scholar] [CrossRef]
- Hyeon, T.; Lee, S.S.; Park, J.; Chung, Y.; Na, H.B. Synthesis of highly crystalline and monodisperse maghemite nanocrystallites without a size-selection process. J. Am. Chem. Soc. 2001, 123, 12798–12801. [Google Scholar] [CrossRef] [PubMed]
- Choudhury, A.J.; Gogoi, D.; Kandimalla, R.; Kalita, S.; Chaudhari, Y.B.; Khan, M.R.; Chutia, J. Penicillin impregnation on oxygen plasma surface functionalized chitosan/Antheraea assama silk fibroin: Studies of antibacterial activity and antithrombogenic property. Mater. Sci. Eng. C 2016, 60, 475–484. [Google Scholar] [CrossRef] [PubMed]
- Ismail, R.A.; Sulaiman, G.M.; Abdulrahman, S.A.; Marzoog, T.R. Antibacterial activity of magnetic iron oxide nanoparticles synthesized by laser ablation in liquid. Mater. Sci. Eng. C 2015, 53, 286–297. [Google Scholar] [CrossRef] [PubMed]
- Azizi, K.; Heydari, A. Vitamin B1 supported on silica-encapsulated γ-Fe2O3 nanoparticles: Design, characterization and application as a greener biocatalyst for highly efficient acylation. RSC Adv. 2014, 4, 8812–8816. [Google Scholar] [CrossRef]
- Sobhani, S.; Ghasemzadeh, M.S.; Honarmand, M. Piperidine and piperazine immobilized on iron oxide nanoparticles as magnetically recyclable heterogeneous catalysts for one-pot synthesis of β-phosphonomalonates. Catal. Lett. 2014, 144, 1515–1523. [Google Scholar] [CrossRef]
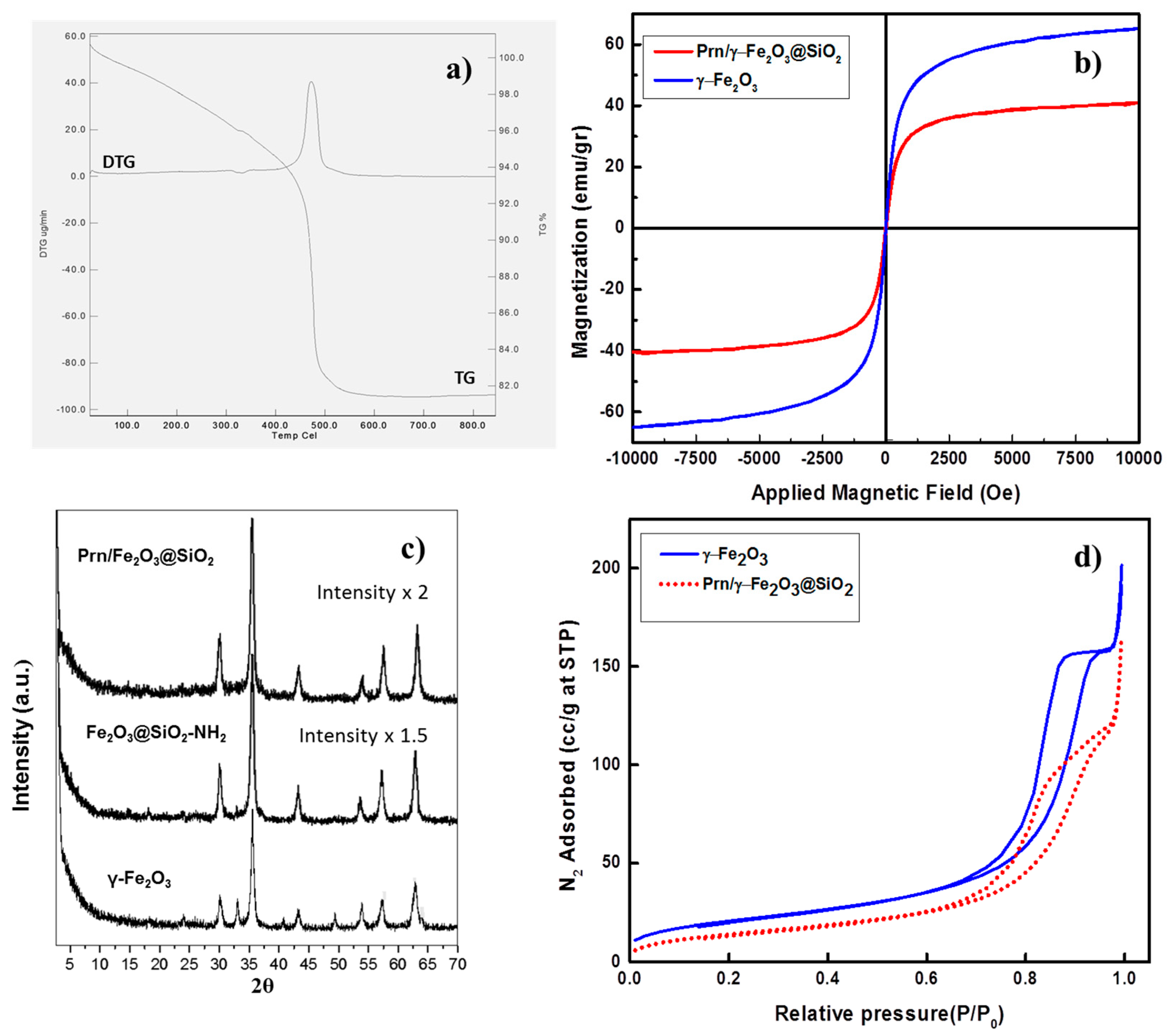
| Material | SBET [a] (m2 g−1) | DBJH [b] (nm) | VBJH [c] (cm3 g−1) |
|---|---|---|---|
| γ-Fe2O3 | 75 | 14.5 | 0.27 |
| Prn/Fe2O3@SiO2 | 53 | 15.2 | 0.2 |
 | ||||||||
| Entry | Solvent | Oxidant | Time (h) | T (°C) | Conversion A (%) | Selectivity A (%) | Conversion B (%) | Selectivity B (%) |
| 1 | CH3CN | - | 15 | 90 | traces | - | traces | - |
| 2 | CH3CN | O2 | 15 | 90 | 25 | 92 | 13 | 32 |
| 3 | CH3CN | tBHP | 4 | 90 | 65 | 88 | 33 | 43 |
| 4 | CH3CN | UHP | 4 | 90 | 85 | 86 | 52 | 48 |
| 5 | CH3CN | H2O2 | 4 | 90 | 100 | 92 | 83 | 51 |
| 6 | Toluene | H2O2 | 10 | 90 | 88 | 88 | 60 | 42 |
 | ||||||
| Entry | Catalyst | Time (h) | Conversion (mol %) | Selectivity (mol %) | ||
| Vanillin | Diphenyl ether | Others [b] | ||||
| 1 | Blank (no catalyst) | 2 | <15 | 7 | 84 | <10 |
| 2 | γ-Fe2O3 | 2 | 49 | 44 | 15 | <40 |
| 3 | proton-unexchanged catalyst | 2 | 21 | 14 | - | <10 |
| 4 | Prn/Fe2O3@SiO2 | 2 | 65 | 55 | <10 | <40 |
 | ||||
| Entry | Catalyst | Time (h) | Conversion (mol %) | Selectivity Vanillin (mol %) |
| 1 | Blank (no cat.) | 4 | <10 | >85 |
| 2 | γ-Fe2O3 | 4 | 60 | 90 |
| 3 | proton-unexchanged catalyst | 4 | 72 | 91 |
| 4 | Prn/Fe2O3@SiO2 | 4 | >99 | 92 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Saberi, F.; Rodriguez-Padrón, D.; Garcia, A.; Shaterian, H.R.; Luque, R. Unprecedented Proline-Based Heterogeneous Organocatalyst for Selective Production of Vanillin. Catalysts 2018, 8, 167. https://doi.org/10.3390/catal8040167
Saberi F, Rodriguez-Padrón D, Garcia A, Shaterian HR, Luque R. Unprecedented Proline-Based Heterogeneous Organocatalyst for Selective Production of Vanillin. Catalysts. 2018; 8(4):167. https://doi.org/10.3390/catal8040167
Chicago/Turabian StyleSaberi, Farveh, Daily Rodriguez-Padrón, Araceli Garcia, Hamid Reza Shaterian, and Rafael Luque. 2018. "Unprecedented Proline-Based Heterogeneous Organocatalyst for Selective Production of Vanillin" Catalysts 8, no. 4: 167. https://doi.org/10.3390/catal8040167
APA StyleSaberi, F., Rodriguez-Padrón, D., Garcia, A., Shaterian, H. R., & Luque, R. (2018). Unprecedented Proline-Based Heterogeneous Organocatalyst for Selective Production of Vanillin. Catalysts, 8(4), 167. https://doi.org/10.3390/catal8040167







