Continuous Dimethyl Carbonate Synthesis from CO2 and Methanol Using Cu-Ni@VSiO as Catalyst Synthesized by a Novel Sulfuration Method
Abstract
:1. Introduction
2. Results and Discussion
2.1. Chemical Structure and Morphology of Synthesized Catalysts
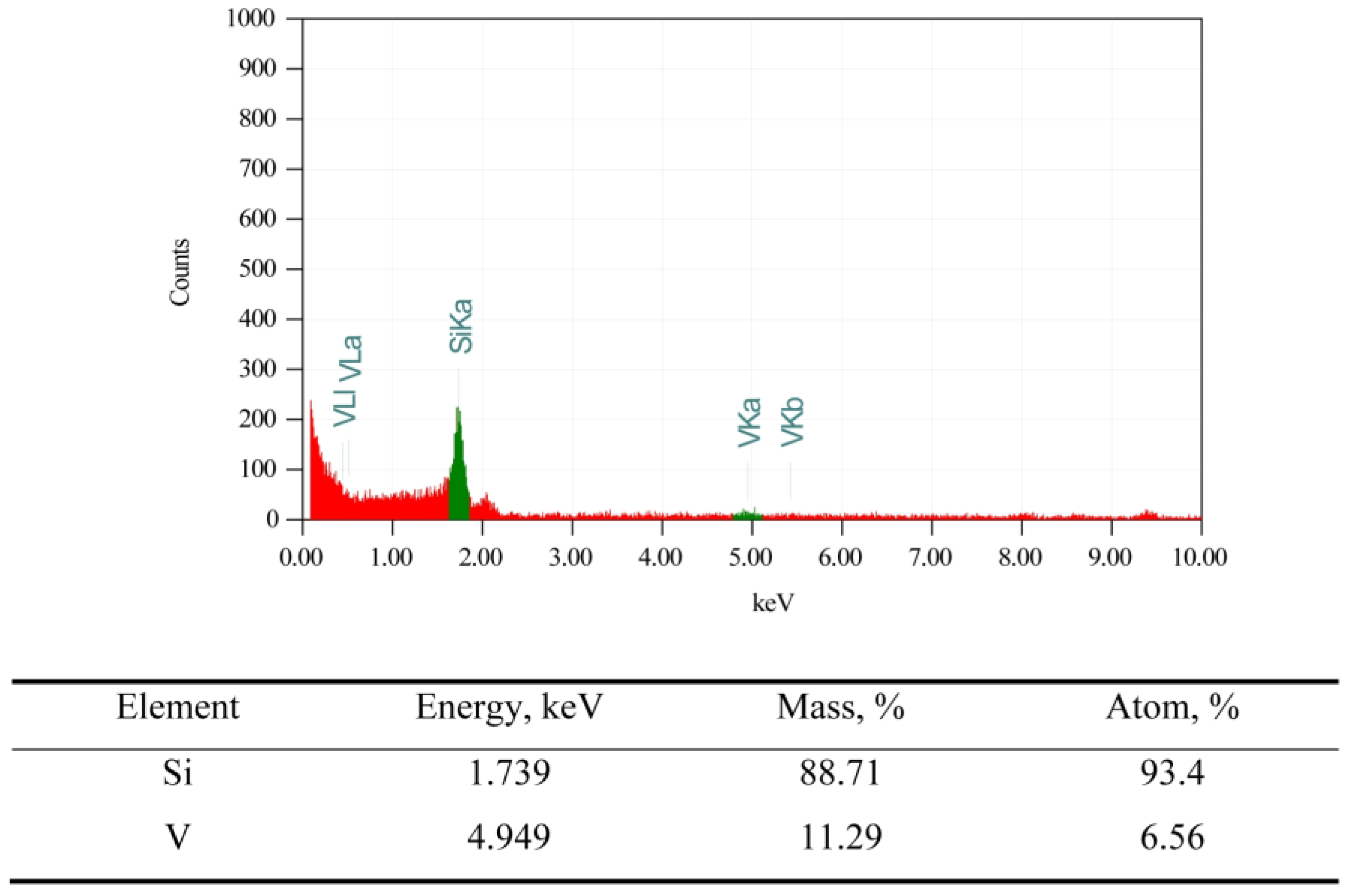
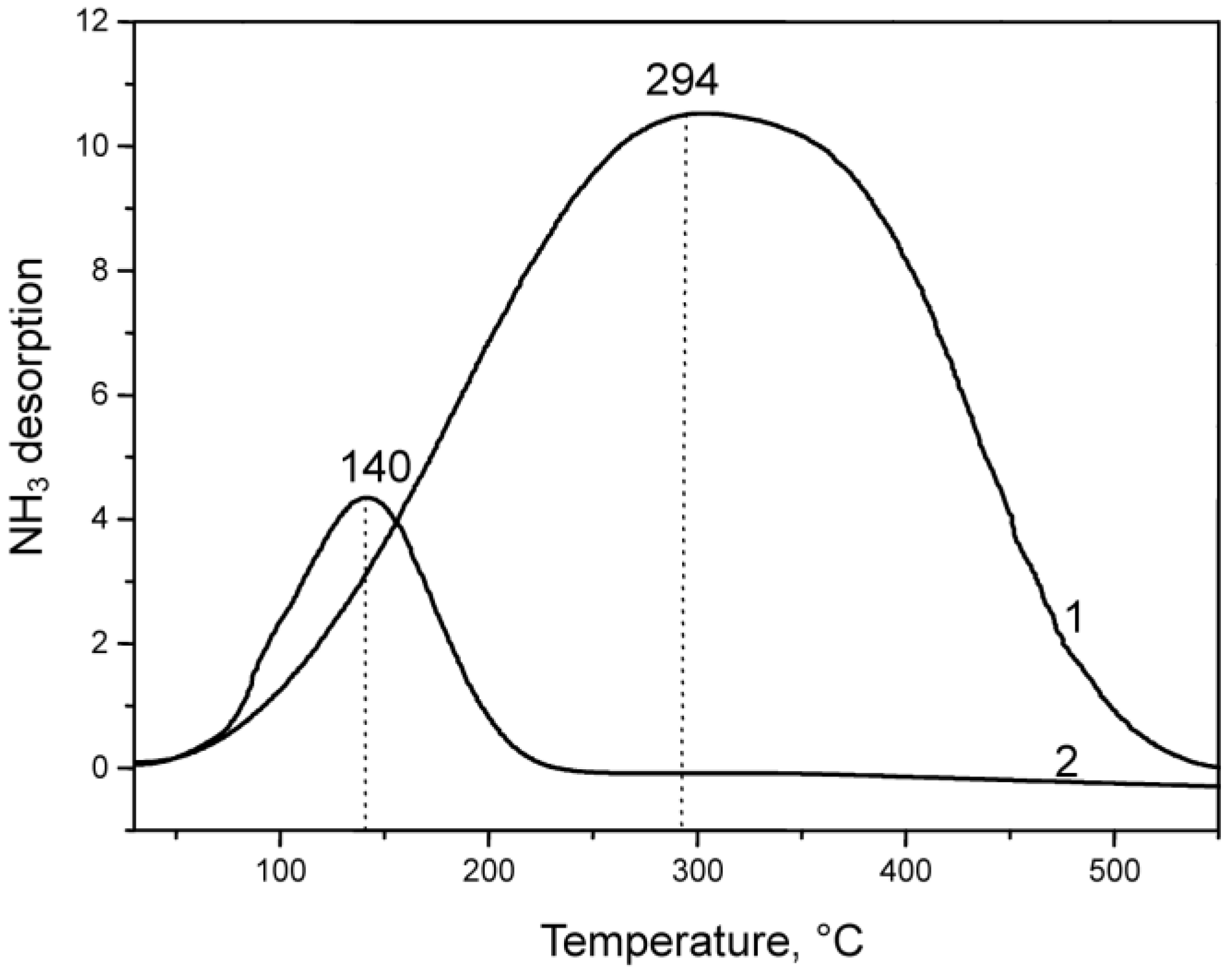
2.1.1. Characterization of Catalyst Support VSiO Microstructure
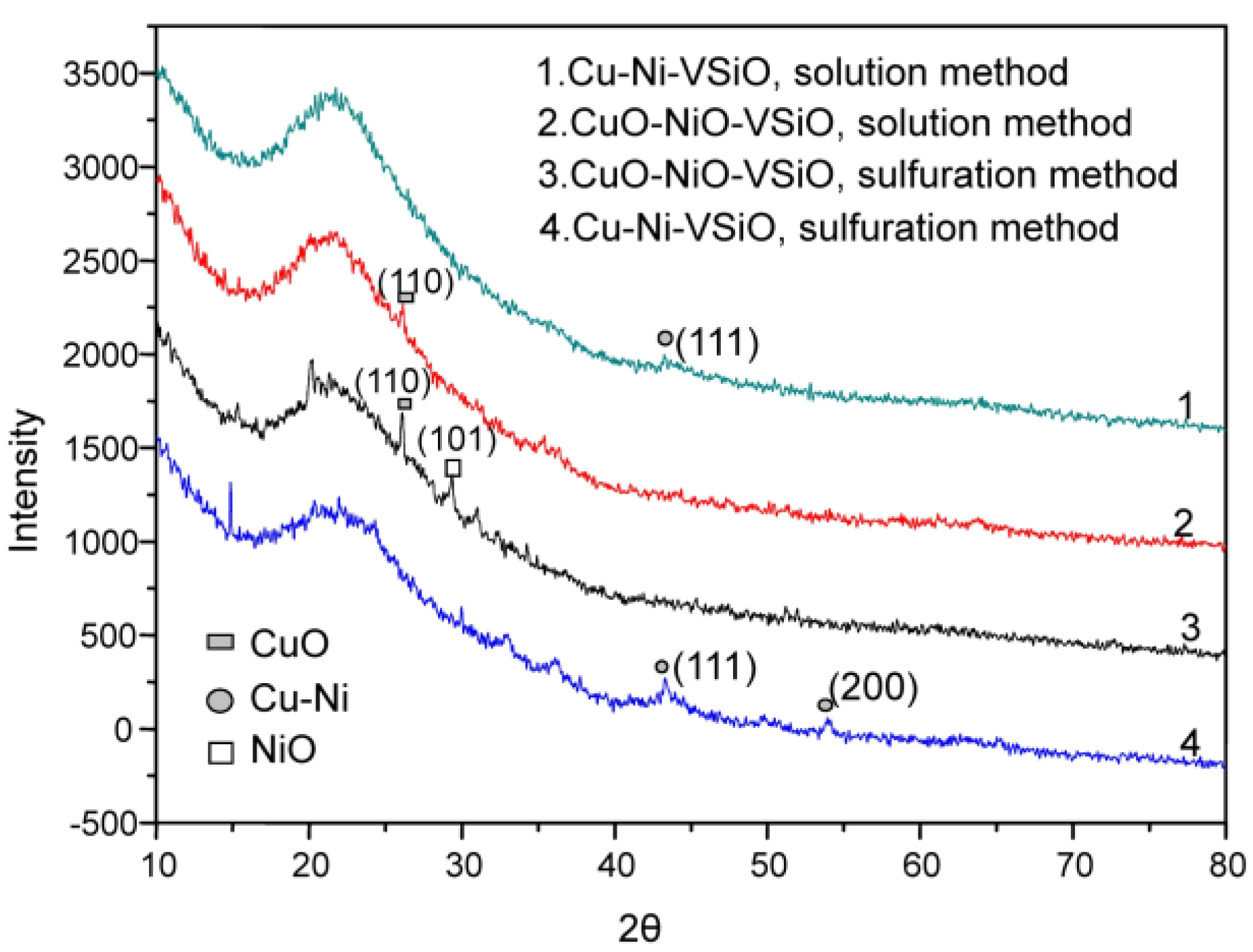
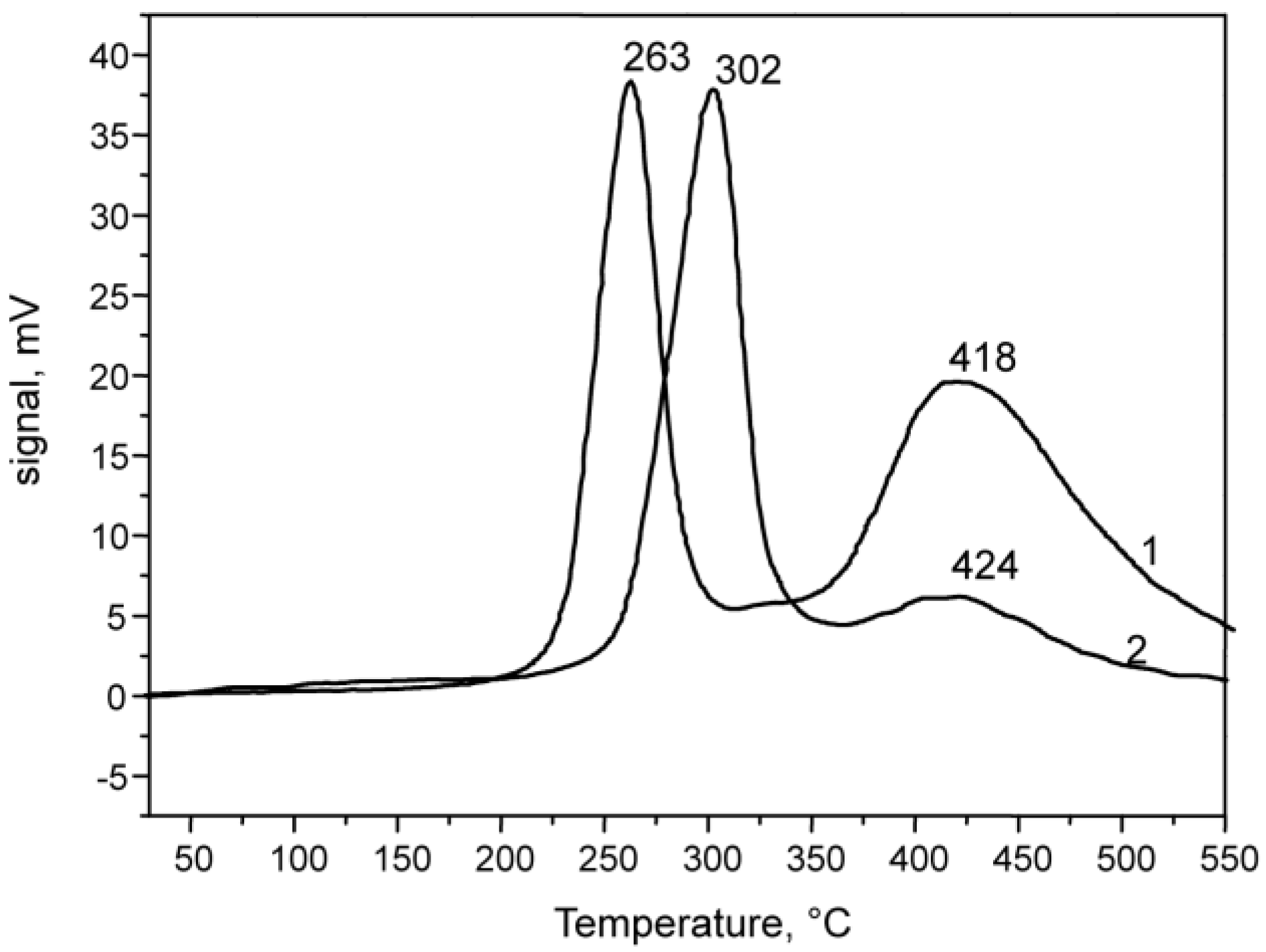
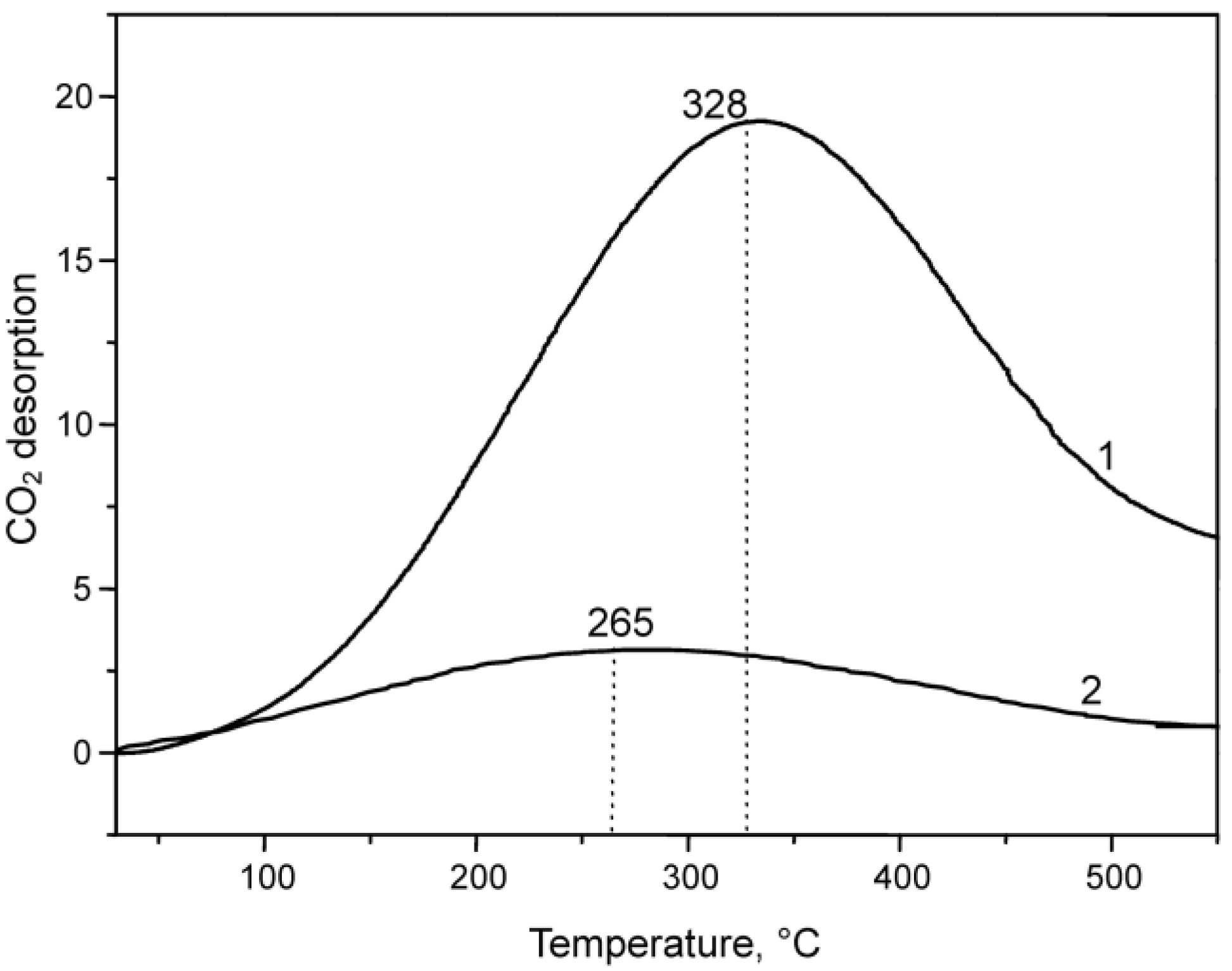
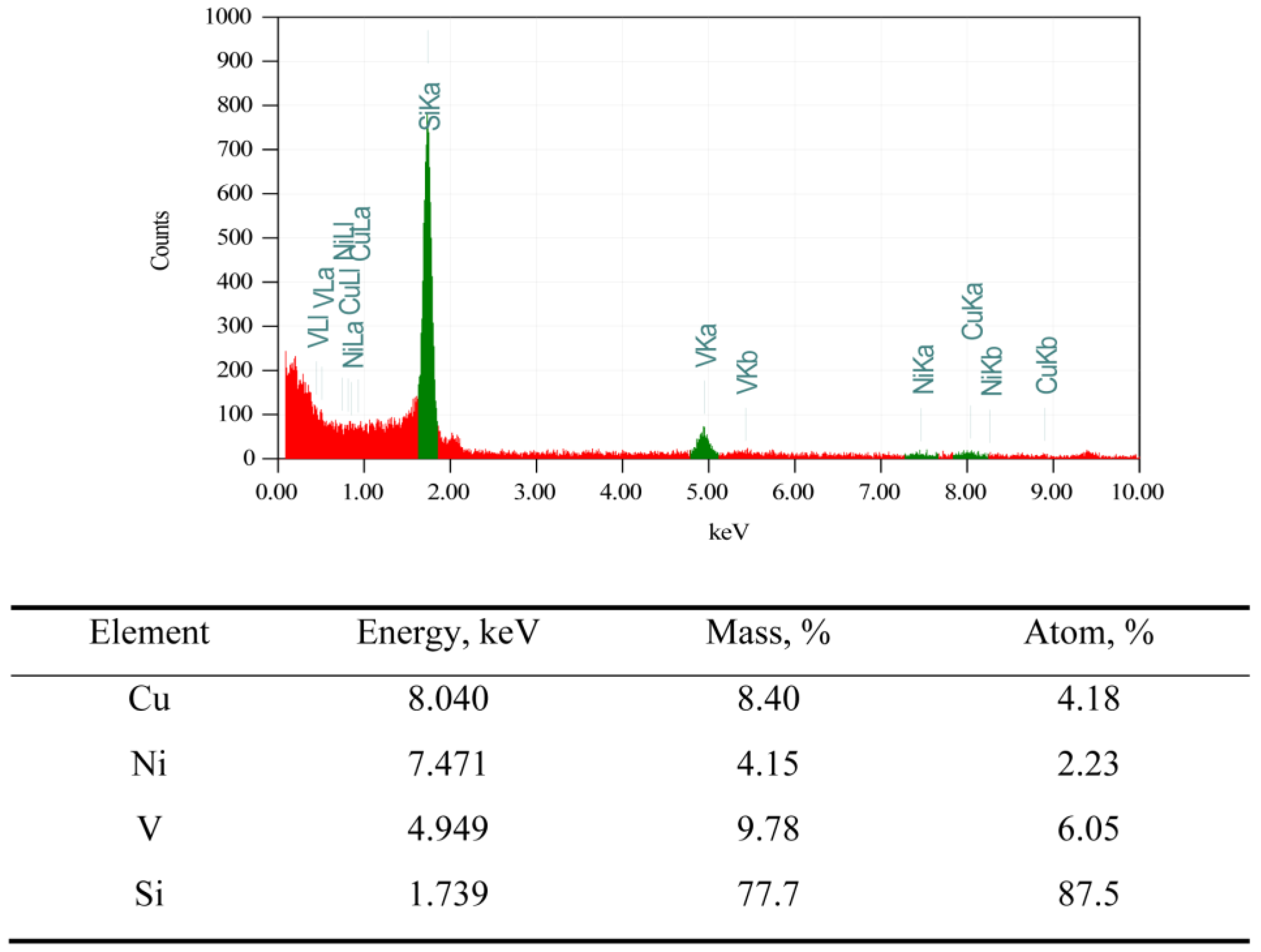
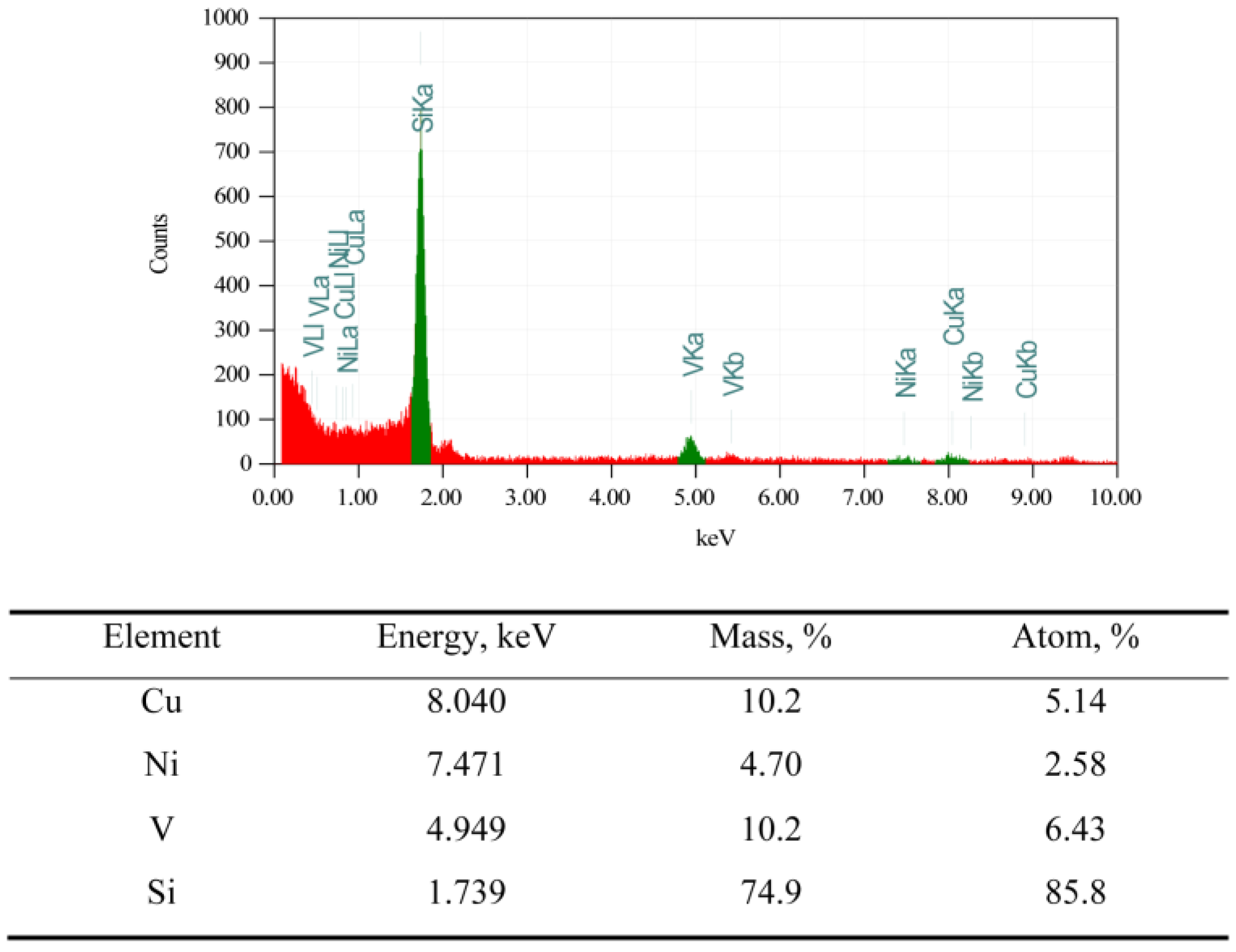
2.1.2. CuO-NiO@VSiO and Cu-Ni@VSiO Microstructure Characterization
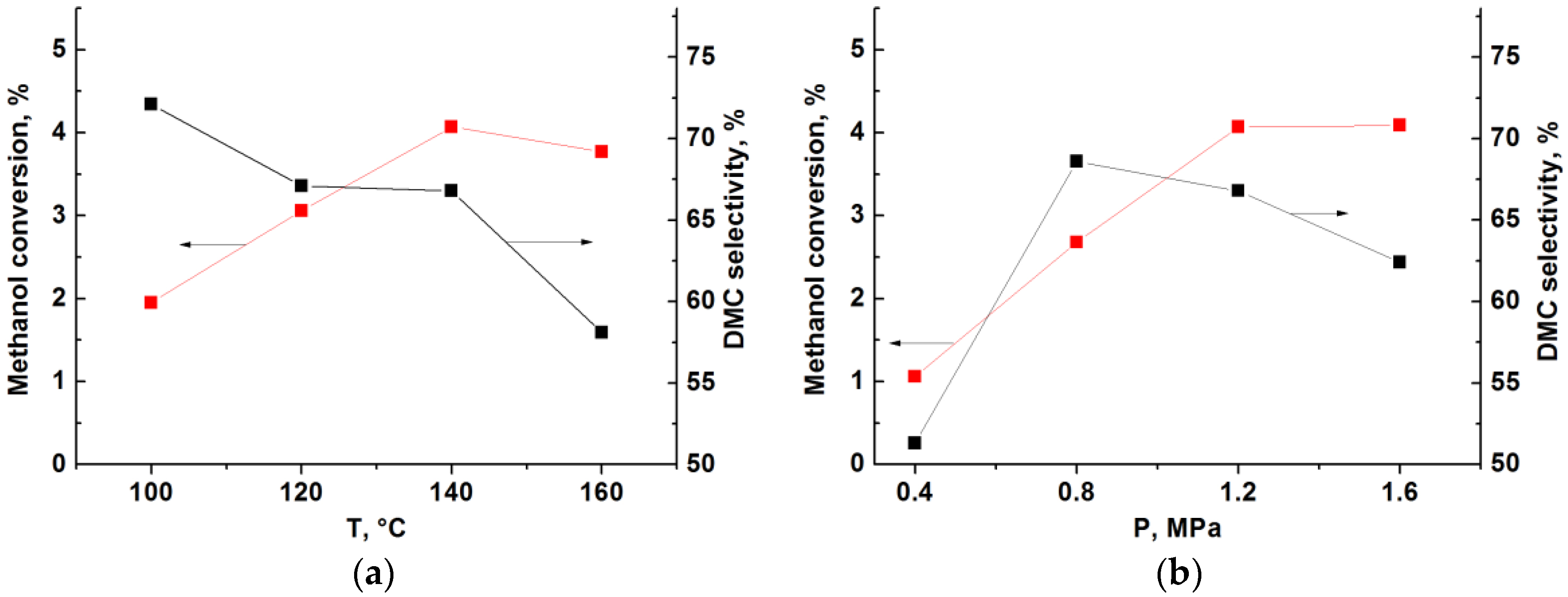
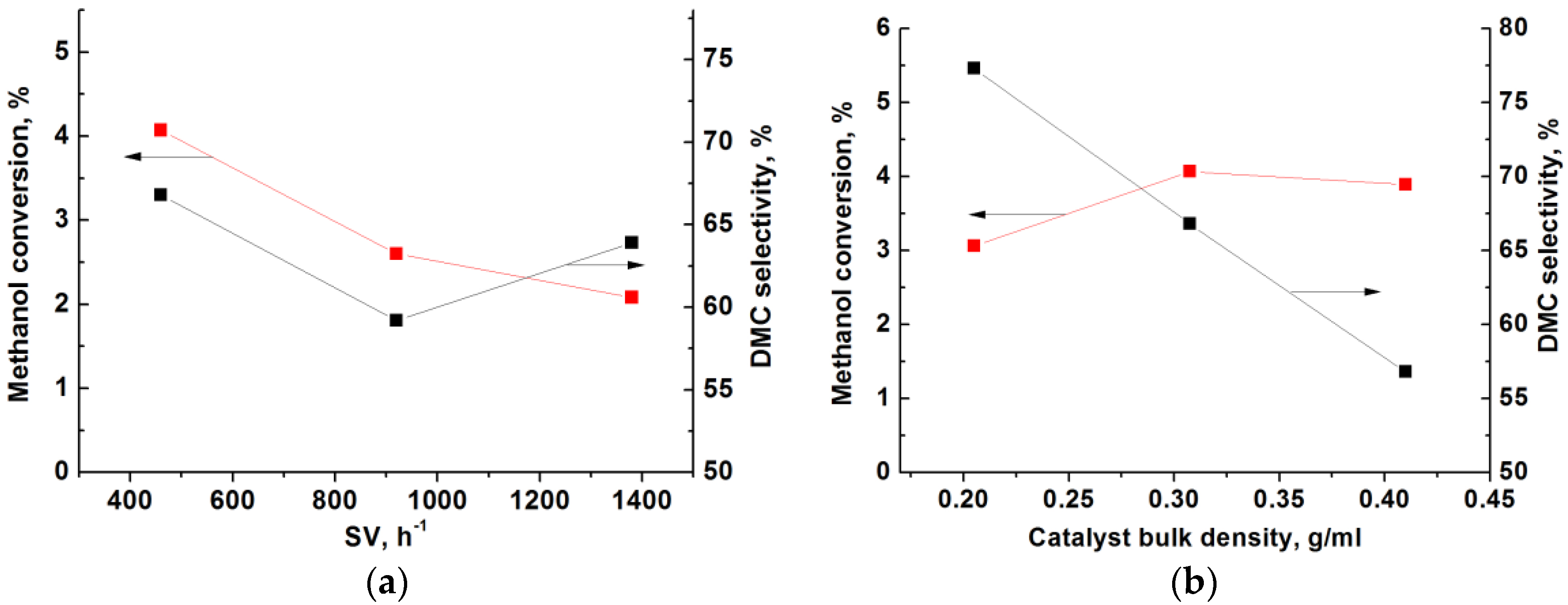
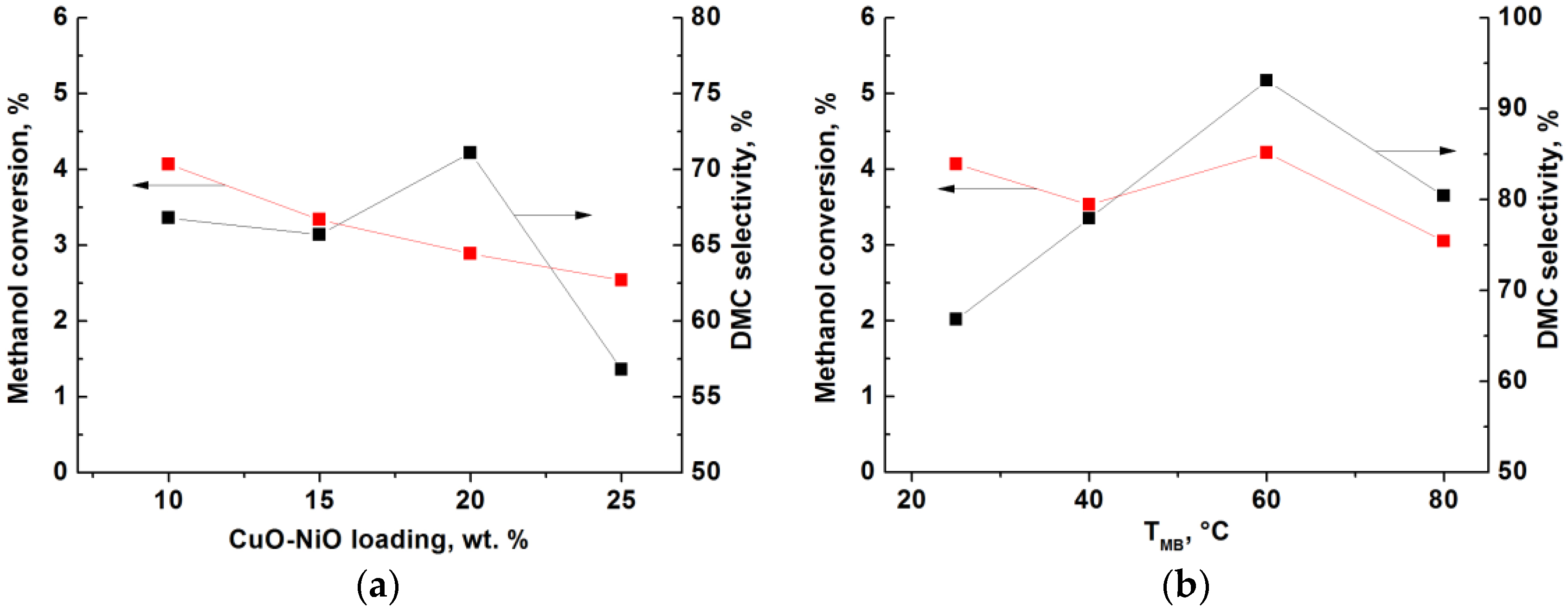
2.2. Catalytic Performance Characterization
3. Materials and Methods
3.1. Catalyst Synthesis
3.1.1. Traditional Solution Synthetic Method of Cu-Ni@VSiO
3.1.2. Novel Sulfuration Method to Synthesize Cu-Ni@VSiO
3.2. Catalysts Characterization
3.3. Evaluation of Catalytic Performance
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Marin, C.M.; Li, L.; Bhalkikar, A.; Doyle, J.E.; Zeng, X.C.; Cheung, C.L. Kinetic and mechanistic investigations of the direct synthesis of dimethyl carbonate from carbon dioxide over ceria nanorod catalysts. J. Catal. 2016, 340, 295–301. [Google Scholar] [CrossRef]
- Stoian, D.; Bansode, A.; Medina, F.; Urakawa, A. Catalysis under microscope: Unraveling the mechanism of catalyst de- and re-activation in the continuous dimethyl carbonate synthesis from CO2 and methanol in the presence of a dehydrating agent. Catal. Today 2017, 283, 2–10. [Google Scholar] [CrossRef]
- Ghorbel, S.B.; Medina, F.; Ghorbel, A.; Segarra, A.M. Phosphoric acid intercalated Mg–Al hydrotalcite-like compounds for catalytic carboxylation reaction of methanol in a continuous system. Appl. Catal. A Gen. 2015, 493, 142–148. [Google Scholar] [CrossRef]
- Zhang, M.; Xiao, M.; Wang, S.J.; Han, D.M.; Lu, Y.X.; Meng, Y.Z. Cerium oxide-based catalysts made by template-precipitation for the dimethyl carbonate synthesis from Carbon dioxide and methanol. J. Clean. Prod. 2015, 103, 847–853. [Google Scholar] [CrossRef]
- Yang, Z.-Z.; Zhao, Y.-N.; He, L.-N.; Gao, J.; Yin, Z.-S. Highly efficient conversion of carbon dioxide catalyzed by polyethylene glycol-functionalized basic ionic liquids. Green Chem. 2012, 14, 519–527. [Google Scholar] [CrossRef]
- Drake, N.L.; Carter, R.M. Some representative carbonates and carbo-ethoxy derivatives related to ethylene glycol1. J. Am. Chem. Soc. 1930, 52, 3720–3724. [Google Scholar] [CrossRef]
- Curnutt, G.L. Catalytic Vapor Phase Process for Producing Dihydrocarbyl Carbonates. U.S. Patent 5,004,827, 2 April 1991. [Google Scholar]
- Feng, X.-J.; Lu, X.-B.; He, R. Tertiary amino group covalently bonded to MCM-41 silica as heterogeneous catalyst for the continuous synthesis of dimethyl carbonate from methanol and ethylene carbonate. Appl. Catal. A Gen. 2004, 272, 347–352. [Google Scholar] [CrossRef]
- Aresta, M. Perspectives in the use of carbon dioxide. Quim. Nova 1999, 22, 269–272. [Google Scholar] [CrossRef]
- Yamanaka, I.; Funakawa, A.; Otsuka, K. Electrocatalytic synthesis of DMC over the Pd/VGCF membrane anode by gas–liquid–solid phase-boundary electrolysis. J. Catal. 2004, 221, 110–118. [Google Scholar] [CrossRef]
- Bansode, A.; Urakawa, A. Continuous DMC Synthesis from CO2 and Methanol over a CeO2 Catalyst in a Fixed Bed Reactor in the Presence of a Dehydrating Agent. ACS Catal. 2014, 4, 3877–3880. [Google Scholar] [CrossRef]
- Tamboli, A.H.; Chaugule, A.A.; Kim, H. Catalytic developments in the direct dimethyl carbonate synthesis from carbon dioxide and methanol. Chem. Eng. J. 2017, 323, 530–544. [Google Scholar] [CrossRef]
- Santos, B.A.V.; Silva, V.M.T.M.; Loureiro, J.M.; Rodrigues, A.E. Review for the Direct Synthesis of Dimethyl Carbonate. ChemBioEng Rev. 2014, 1, 214–229. [Google Scholar] [CrossRef]
- Sakakura, T.; Kohno, K. The synthesis of organic carbonates from carbon dioxide. Chem. Commun. 2009, 1312–1330. [Google Scholar] [CrossRef] [PubMed]
- Fu, Z.; Meng, Y. Research Progress in the Phosgene-Free and Direct Synthesis of Dimethyl Carbonate from CO2 and Methanol. In Chemistry beyond Chlorine; Tundo, P., He, L.-N., Lokteva, E., Mota, C., Eds.; Springer International Publishing: Cham, Switzerland, 2016; pp. 363–385. ISBN 978-3-319-30071-9. [Google Scholar]
- Cao, Y.; Cheng, H.; Ma, L.; Liu, F.; Liu, Z. Research Progress in the Direct Synthesis of Dimethyl Carbonate from CO2 and Methanol. Catal. Surv. Asia 2012, 16, 138–147. [Google Scholar] [CrossRef]
- Zhou, Y.J.; Wang, S.J.; Xiao, M.; Han, D.M.; Lu, Y.X.; Meng, Y.Z. Novel Cu-Fe bimetal catalyst for the formation of dimethyl carbonate from carbon dioxide and methanol. RSC Adv. 2012, 17, 6831–6837. [Google Scholar] [CrossRef]
- Choi, J.C.; He, L.N.; Yasuda, H.; Sakakura, T. Selective and high yield synthesis of dimethyl carbonate directly from carbon dioxide and methanol. Green Chem. 2002, 230–234. [Google Scholar] [CrossRef]
- Fang, S.N.; Fujimoto, K. Direct synthesis of dimethyl carbonate from carbon dioxide and methanol catalyzed by base. Appl. Catal. A Gen. 1996, 142, L1–L3. [Google Scholar] [CrossRef]
- Kumar, P.; With, P.; Srivastava, V.C.; Glaser, R.; Mishra, I.M. Conversion of carbon dioxide along with methanol to dimethyl carbonate over ceria catalyst. J. Environ. Chem. Eng. 2015, 3, 2943–2947. [Google Scholar] [CrossRef]
- Tamboli, A.H.; Chaugule, A.A.; Kim, H. Highly selective and multifunctional chitosan/ionic liquids catalyst for conversion of CO2 and methanol to dimethyl carbonates at mild reaction conditions. Fuel 2016, 166, 495–501. [Google Scholar] [CrossRef]
- Saada, R.; Kellici, S.; Heil, T.; Morgan, D.; Saha, B. Greener synthesis of dimethyl carbonate using a novel ceria-zirconia oxide/graphene nanocomposite catalyst. Appl. Catal. B Environ. 2015, 168, 353–362. [Google Scholar] [CrossRef]
- Kumar, S.; Kumar, P.; Jain, S.L. Graphene oxide immobilized copper phthalocyanine tetrasulphonamide: The first heterogenized homogeneous catalyst for dimethylcarbonate synthesis from CO2 and methanol. J. Mater. Chem. A 2014, 18861–18866. [Google Scholar] [CrossRef]
- Eta, V.; Maki-Arvela, P.; Leino, A.R.; Kordas, K.; Salmi, T.; Murzin, D.Y.; Mikkola, J.P. Synthesis of Dimethyl Carbonate from Methanol and Carbon Dioxide: Circumventing Thermodynamic Limitations. Ind. Eng. Chem. Res. 2010, 49, 9609–9617. [Google Scholar] [CrossRef]
- Fan, B.B.; Li, H.Y.; Fan, W.B.; Zhang, J.L.; Li, R.F. Organotin compounds immobilized on mesoporous silicas as heterogeneous catalysts for direct synthesis of dimethyl carbonate from methanol and carbon dioxide. Appl. Catal. A Gen. 2010, 372, 94–102. [Google Scholar] [CrossRef]
- Kohno, K.; Choi, J.C.; Ohshima, Y.; Yasuda, H.; Sakakura, T. Synthesis of dimethyl carbonate from carbon dioxide catalyzed by titanium alkoxides with polyether-type ligands. ChemSusChem 2008, 1, 186–188. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.L.; Shu, D.; Meng, Y.Z. Direct synthesis of dimethyl carbonate from CO2 and methanol. Res. J. Chem. Environ. 2005, 9, 74–79. [Google Scholar] [CrossRef]
- Ikeda, Y.; Sakaihori, T.; Tomishige, K.; Fujimoto, K. Promoting effect of phosphoric acid on zirconia catalysts in selective synthesis of dimethyl carbonate from methanol and carbon dioxide. Catal. Lett. 2000, 66, 59–62. [Google Scholar] [CrossRef]
- Tomishige, K.; Kunimori, K. Catalytic and direct synthesis of dimethyl carbonate starting from carbon dioxide using CeO2-ZrO2 solid solution heterogeneous catalyst: Effect of H2O removal from the reaction system. Appl. Catal. A Gen. 2002, 237, 103–109. [Google Scholar] [CrossRef]
- Aresta, M.; Dibenedetto, A.; Pastore, C.; Cuocci, C.; Aresta, B.; Cometa, S.; De Giglio, E. Cerium(IV)oxide modification by inclusion of a hetero-atom: A strategy for producing efficient and robust nano-catalysts for methanol carboxylation. Catal. Today 2008, 137, 125–131. [Google Scholar] [CrossRef]
- Lee, H.J.; Park, S.; Song, I.K.; Jung, J.C. Direct Synthesis of Dimethyl Carbonate from Methanol and Carbon Dioxide over Ga2O3/Ce0.6Zr0.4O2 Catalysts: Effect of Acidity and Basicity of the Catalysts. Catal. Lett. 2011, 141, 531–537. [Google Scholar] [CrossRef]
- Zhao, T.; Hu, X.; Wu, D.; Li, R.; Yang, G.; Wu, Y. Direct synthesis of dimethyl carbonate from CO2 and methanol at room temperature using imidazolium hydrogen carbonate ionic liquid as recyclable catalyst and dehydrant. ChemSusChem 2017, 10, 2046–2052. [Google Scholar] [CrossRef] [PubMed]
- Verma, S.; Nasir Baig, R.B.; Nadagouda, M.N.; Varma, R.S. Fixation of carbon dioxide into dimethyl carbonate over titanium-based zeolitic thiophenebenzimidazolate framework. Sci. Rep. 2017, 7, 655. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.L.; Meng, Y.Z.; Xiao, M.; Lu, Y.X. Direct synthesis of dimethyl carbonate (DMC) using Cu-Ni/VSO as catalyst. J. Mol. Catal. A Chem. 2006, 249, 93–97. [Google Scholar] [CrossRef]
- Wang, X.J.; Xiao, M.; Wang, S.J.; Lu, Y.X.; Meng, Y.Z. Direct synthesis of dimethyl carbonate from carbon dioxide and methanol using supported copper (Ni, V, O) catalyst with photo-assistance. J. Mol. Catal. A Chem. 2007, 278, 92–96. [Google Scholar] [CrossRef]
- Bian, J.; Xiao, M.; Wang, S.J.; Lu, Y.X.; Meng, Y.Z. Novel application of thermally expanded graphite as the support of catalysts for direct synthesis of DMC from CH3OH and CO2. J. Colloid Interface Sci. 2009, 334, 50–57. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Wang, S.; Xiao, M.; Han, D.; Lu, Y.; Meng, Y. Formation of dimethyl carbonate on nature clay supported bimetallic copper-nickel catalysts. J. Clean. Prod. 2015, 103, 925–933. [Google Scholar] [CrossRef]
- Chen, H.; Wang, S.; Xiao, M.; Han, D.; Lu, Y.; Meng, Y. Direct synthesis of dimethyl carbonate from CO2 and CH3OH using 0.4 nm molecular sieve supported Cu-Ni bimetal catalyst. Chin. J. Chem. Eng. 2012, 20, 906–913. [Google Scholar] [CrossRef]
- Bian, J.; Xiao, M.; Wang, S.J.; Lu, Y.X.; Meng, Y.Z. Highly effective direct synthesis of DMC from CH3OH and CO2 using novel Cu-Ni/C bimetallic composite catalysts. Chin. Chem. Lett. 2009, 20, 352–355. [Google Scholar] [CrossRef]
- Bian, J.; Xiao, M.; Wang, S.; Wang, X.; Lu, Y.; Meng, Y. Highly effective synthesis of dimethyl carbonate from methanol and carbon dioxide using a novel copper-nickel/graphite bimetallic nanocomposite catalyst. Chem. Eng. J. 2009, 147, 287–296. [Google Scholar] [CrossRef]
- Bian, J.; Xiao, M.; Wang, S.; Lu, Y.; Meng, Y. Direct synthesis of DMC from CH3OH and CO2 over V-doped Cu-Ni/AC catalysts. Catal. Commun. 2009, 10, 1142–1145. [Google Scholar] [CrossRef]
- Bian, J.; Xiao, M.; Wang, S.J.; Lu, Y.X.; Meng, Y.Z. Carbon nanotubes supported Cu-Ni bimetallic catalysts and their properties for the direct synthesis of dimethyl carbonate from methanol and carbon dioxide. Appl. Surf. Sci. 2009, 255, 7188–7196. [Google Scholar] [CrossRef]
- Zhong, S.H.; Wang, J.W.; Xiao, X.F.; Li, H.S. Dimethyl carbonate synthesis from carbon dioxide and methanol over Cu-Ni/MoSiO(VSiO) catalysts. In 12th International Congress on Catalysis; Corma, A., Melo, F.V., Mendioroz, S., Fierro, J.L.G., Eds.; Studies in Surface Science and Catalysis; Elsevier: Amsterdam, The Netherlands, 2000; Volume 130, pp. 1565–1570. [Google Scholar]
- Wang, D.; Zhou, L.; Feng, X.; Zhao, N.; Yang, B. Polysilicic acid gel method derived V2O5/SiO2 composite materials: Synthesis and characterization. AIP Conf. Proc. 2017, 1794, 020021. [Google Scholar] [CrossRef]
- Khatab, T.K.; Abdelghany, A.M.; Soliman, H.A. V2O5/SiO2 as a Heterogeneous Catalyst in the Synthesis of bis(indolyl)methanes under Solvent Free Condition. Silicon 2017, 1–6. [Google Scholar] [CrossRef]
- Musić, S.; Filipović-Vinceković, N.; Sekovanić, L. Precipitation of amorphous SiO2 particules and their properties. Braz. J. Chem. Eng. 2011, 28, 89–94. [Google Scholar] [CrossRef]
- Poungsombate, A.; Imyen, T.; Dittanet, P.; Embley, B.; Kongkachuichay, P. Direct synthesis of dimethyl carbonate from CO2 and methanol by supported bimetallic Cu–Ni/ZIF-8 MOF catalysts. J. Taiwan Inst. Chem. Eng. 2017, 80, 16–24. [Google Scholar] [CrossRef]
- Pimprom, S.; Sriboonkham, K.; Dittanet, P.; Föttinger, K.; Rupprechter, G.; Kongkachuichay, P. Synthesis of copper-nickel/SBA-15 from rice husk ash catalyst for dimethyl carbonate production from methanol and carbon dioxide. J. Ind. Eng. Chem. 2015, 31, 156–166. [Google Scholar] [CrossRef]
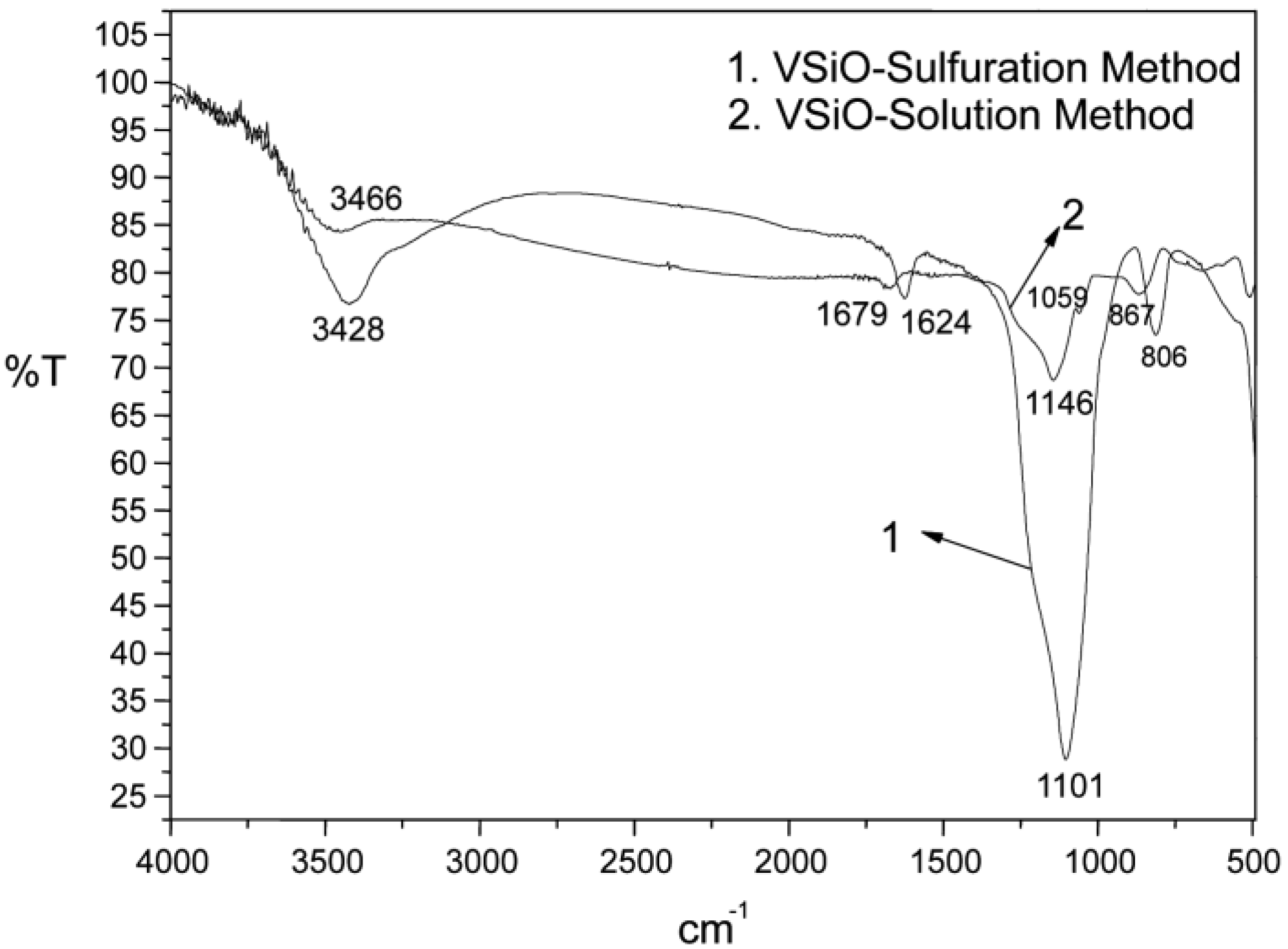
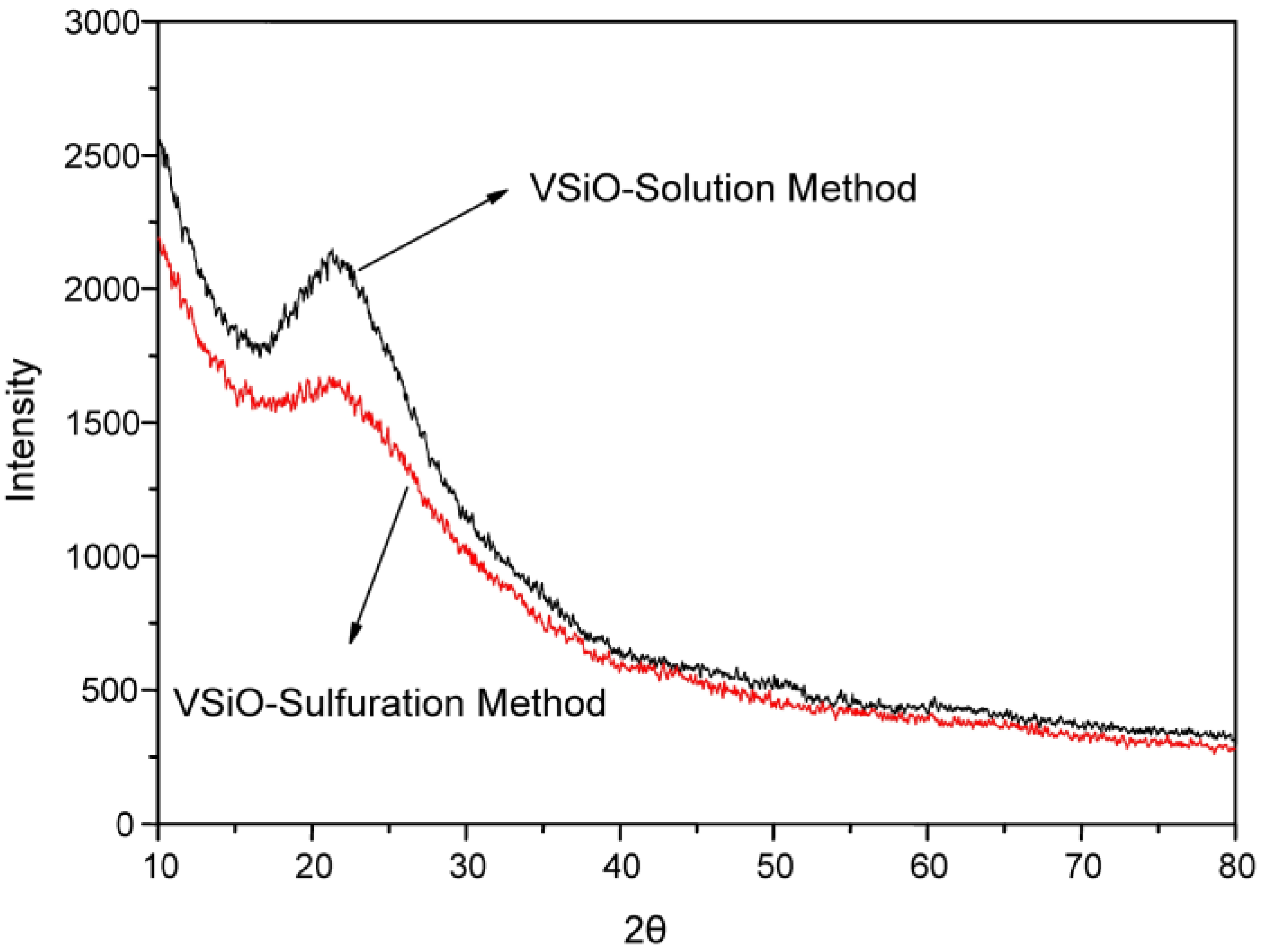
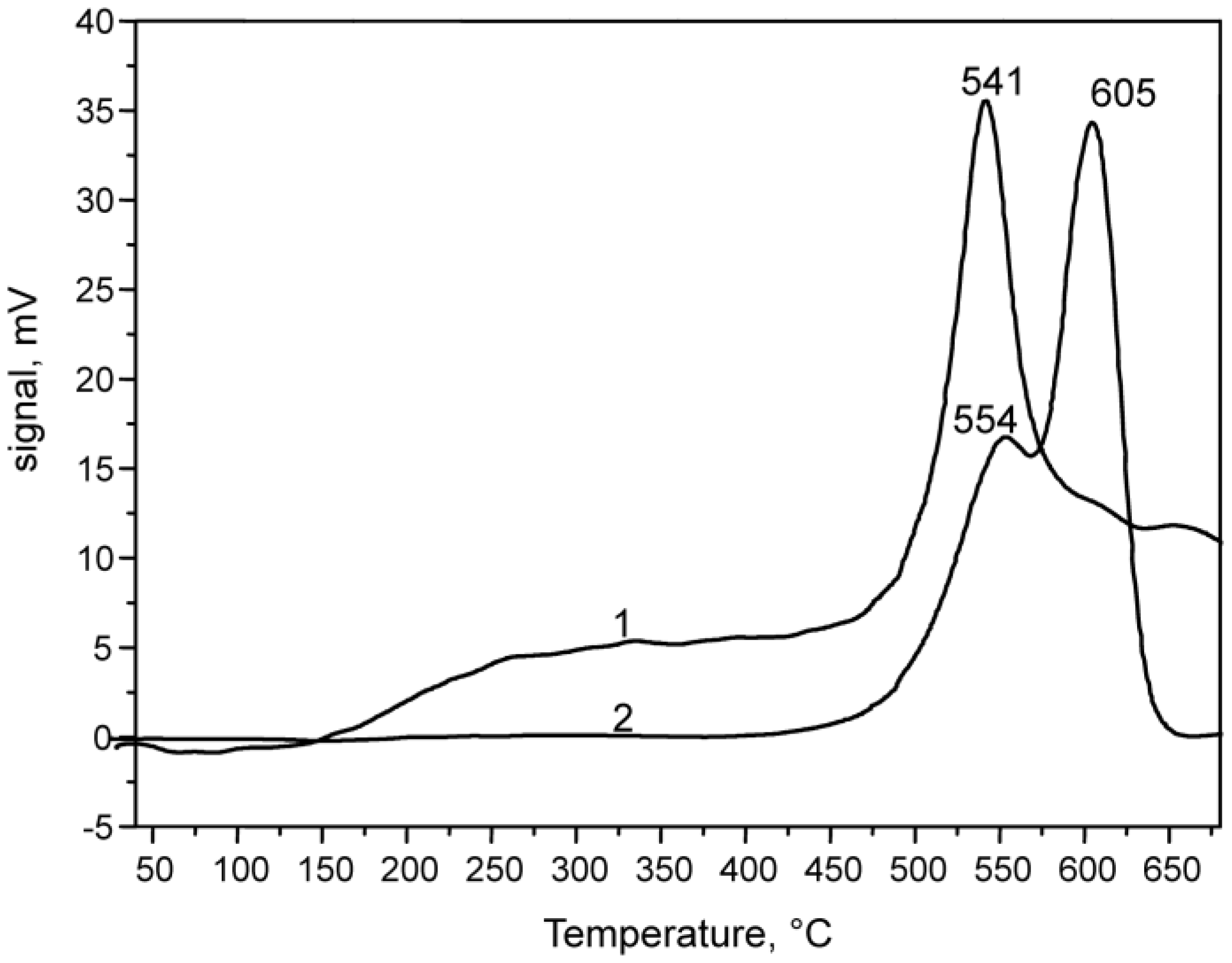

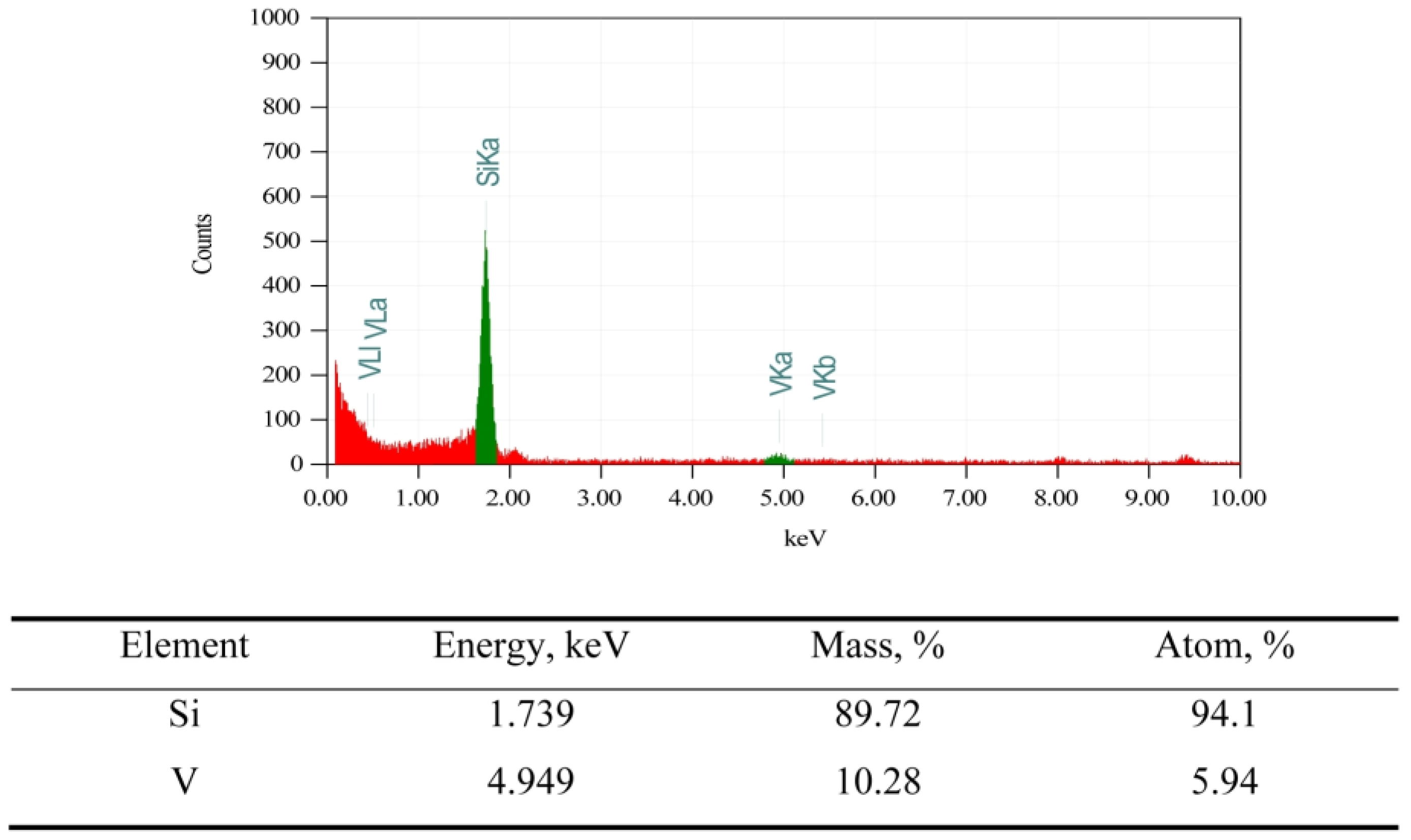

| Temperature, °C | Methanol Conversion, % | DMC Selectivity, % |
|---|---|---|
| 100 | 0.47 | 96.3 |
| 120 | 0.84 | 91.5 |
| 140 | 1.69 | 86.3 |
| 160 | 1.73 | 80.6 |
| Catalyst | T, °C | P, MPa | Reactor Type | MeOH Conversion, % | DMC Selectivity | Ref. |
|---|---|---|---|---|---|---|
| Cu-Ni@VSiO (sulfuration) | 140 | 1.2 | C 1 | 4.2 | 93.1 | present work |
| Cu-Ni@VSiO (solution) | 140 | 1.2 | C | 1.7 | 86.3 | present work |
| Cu-Ni@VSiO | 140 | 0.1 | C | 14.5 | 87.8 | [43] |
| Cu–Ni@ZIF-8 2 | 110 | 2 | batch | 12.8 | 50.0 | [47] |
| Cu-Ni@VSiO 3 | 120 | 0.1 | C | 4.0 | 85 | [35] |
| Cu-Ni@VSiO | 140 | 0.9 | C | n.a. | 87.1 | [34] |
| Cu-Ni@SBA-15 | 110 | 1.2 | continuous fixed-bed | ca 21 | ca 20 | [48] |
| Cu-Ni@TEG 2 | 100 | 1.4 | C | 5.0 | 91.0 | [36] |
| Cu-Ni@ KHNTs 2 | 130 | 1.2 | C | 7.8 | 89.0 | [37] |
| Cu-Ni@MS 2 | 120 | 1.1 | C | 7.1 | 87 | [38] |
| Cu-Ni@graphite | 100 | 1.2 | C | 10.1 | 90.2 | [40] |
| Cu-Ni-V@AC 2 | 110 | 1.2 | C | 7.8 | 89.9 | [41] |
| Cu-Ni@MWCNTs 2 | 120 | 1.2 | C | 4.4 | 90.5 | [42] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, M.; Alferov, K.A.; Xiao, M.; Han, D.; Wang, S.; Meng, Y. Continuous Dimethyl Carbonate Synthesis from CO2 and Methanol Using Cu-Ni@VSiO as Catalyst Synthesized by a Novel Sulfuration Method. Catalysts 2018, 8, 142. https://doi.org/10.3390/catal8040142
Zhang M, Alferov KA, Xiao M, Han D, Wang S, Meng Y. Continuous Dimethyl Carbonate Synthesis from CO2 and Methanol Using Cu-Ni@VSiO as Catalyst Synthesized by a Novel Sulfuration Method. Catalysts. 2018; 8(4):142. https://doi.org/10.3390/catal8040142
Chicago/Turabian StyleZhang, Meng, Kirill A. Alferov, Min Xiao, Dongmei Han, Shuanjin Wang, and Yuezhong Meng. 2018. "Continuous Dimethyl Carbonate Synthesis from CO2 and Methanol Using Cu-Ni@VSiO as Catalyst Synthesized by a Novel Sulfuration Method" Catalysts 8, no. 4: 142. https://doi.org/10.3390/catal8040142
APA StyleZhang, M., Alferov, K. A., Xiao, M., Han, D., Wang, S., & Meng, Y. (2018). Continuous Dimethyl Carbonate Synthesis from CO2 and Methanol Using Cu-Ni@VSiO as Catalyst Synthesized by a Novel Sulfuration Method. Catalysts, 8(4), 142. https://doi.org/10.3390/catal8040142






