Recent Progress in Asymmetric Catalysis and Chromatographic Separation by Chiral Metal–Organic Frameworks
Abstract
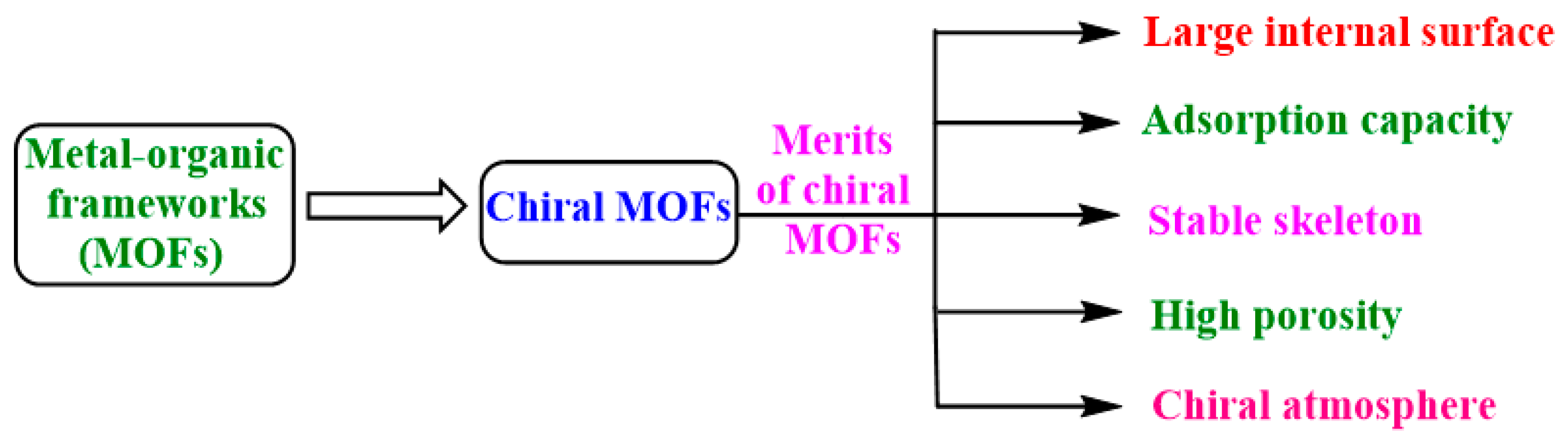
1. Introduction
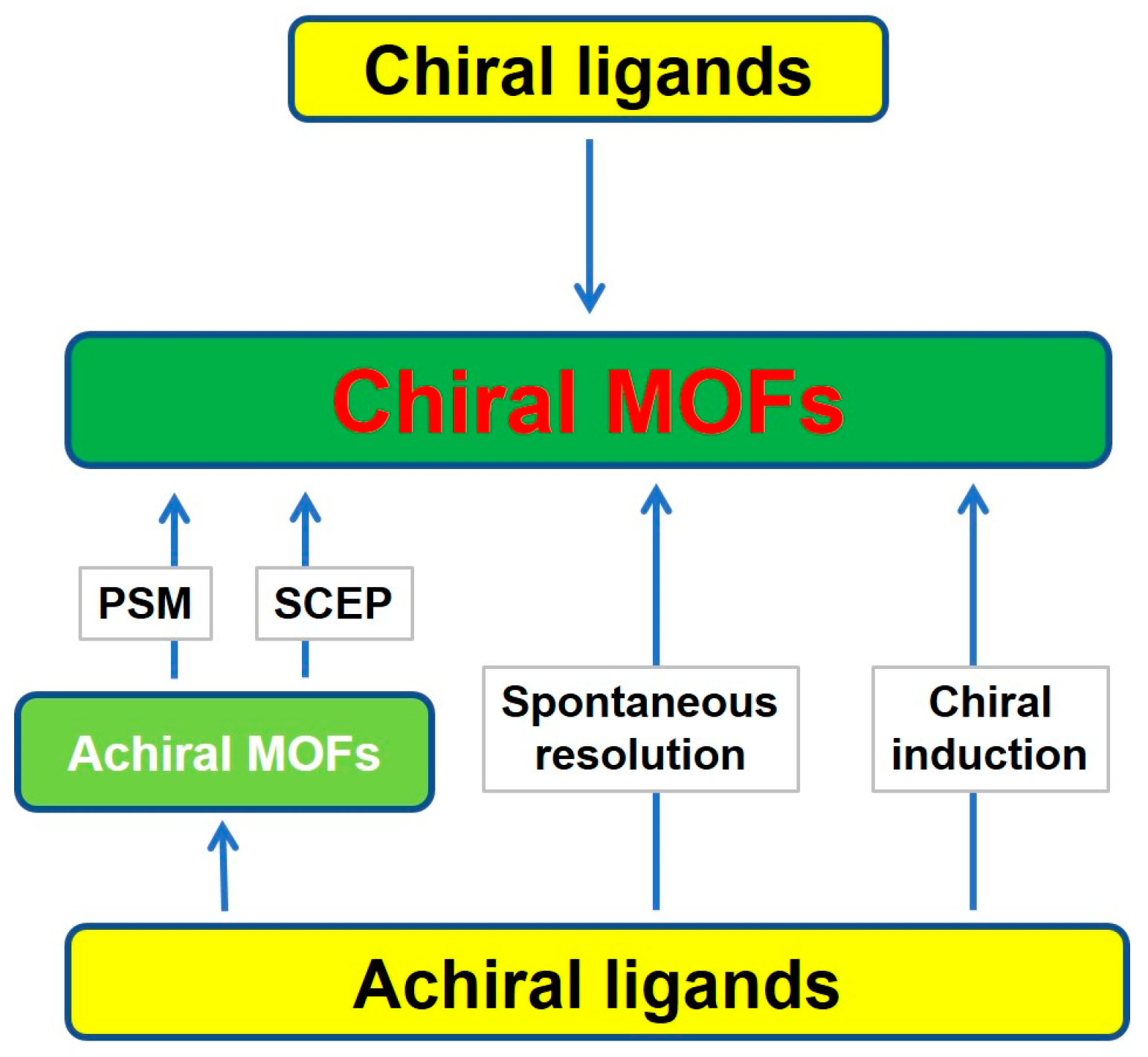
2. Synthetic Strategies of Chiral MOFs
2.1. Straightforward Method
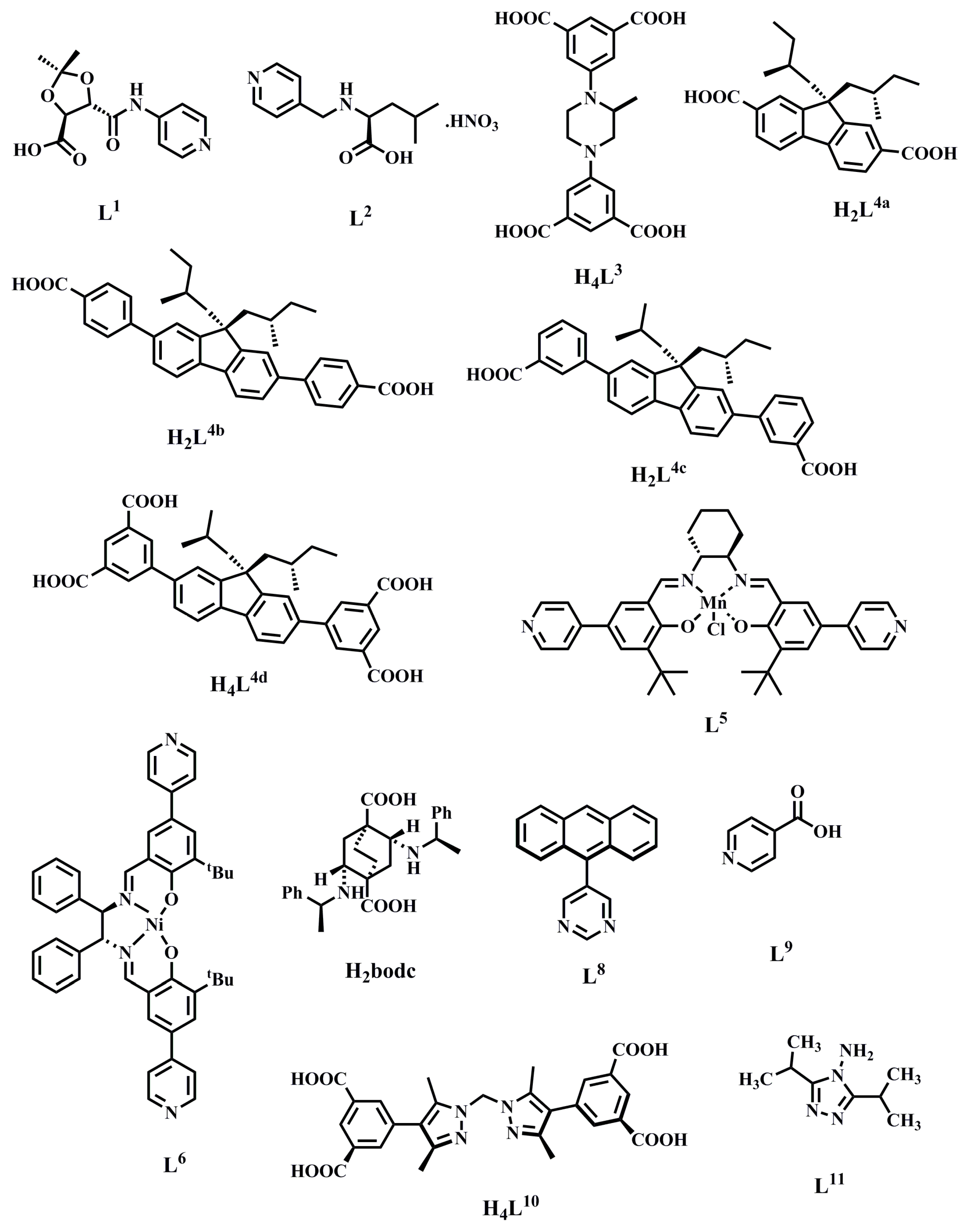
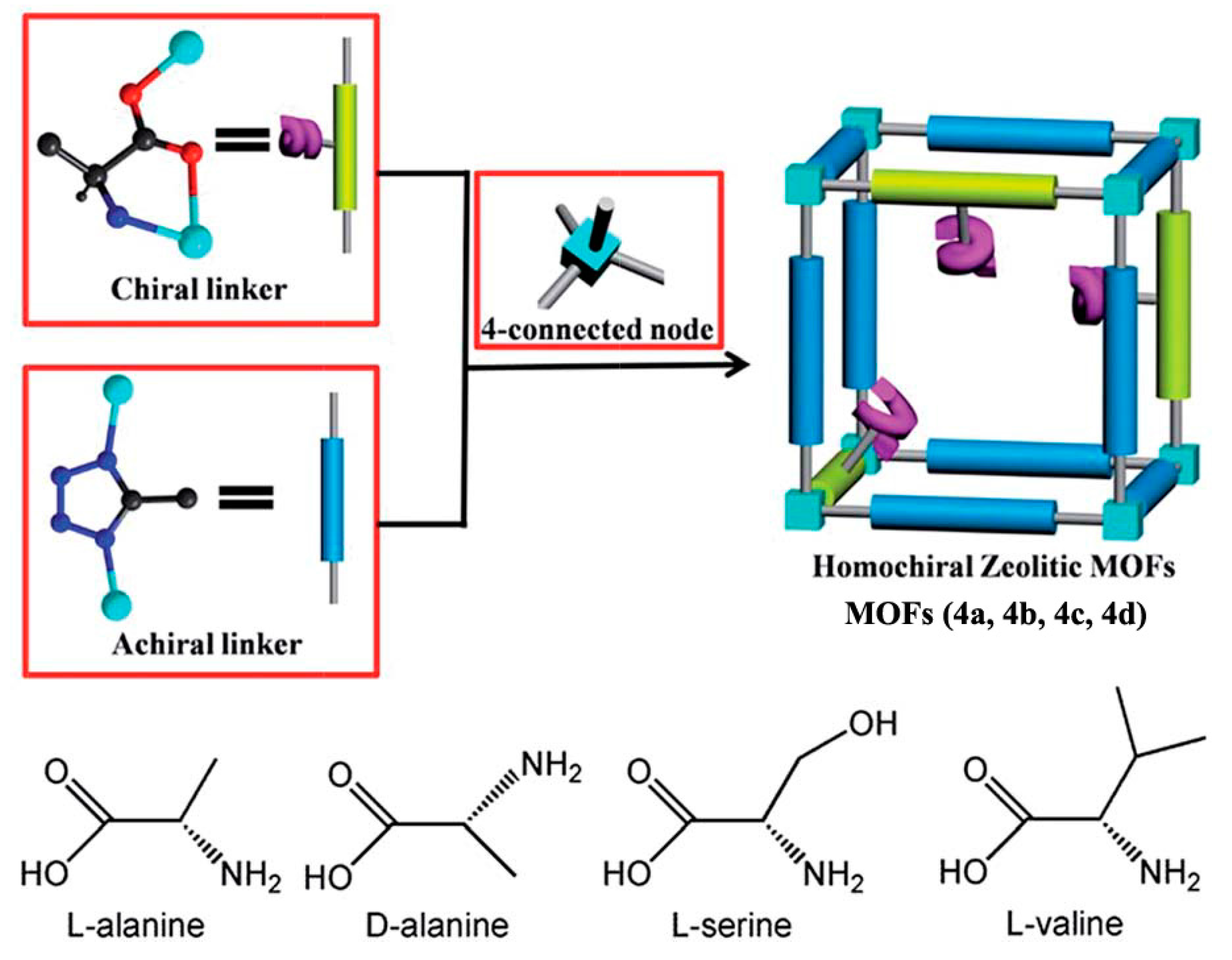
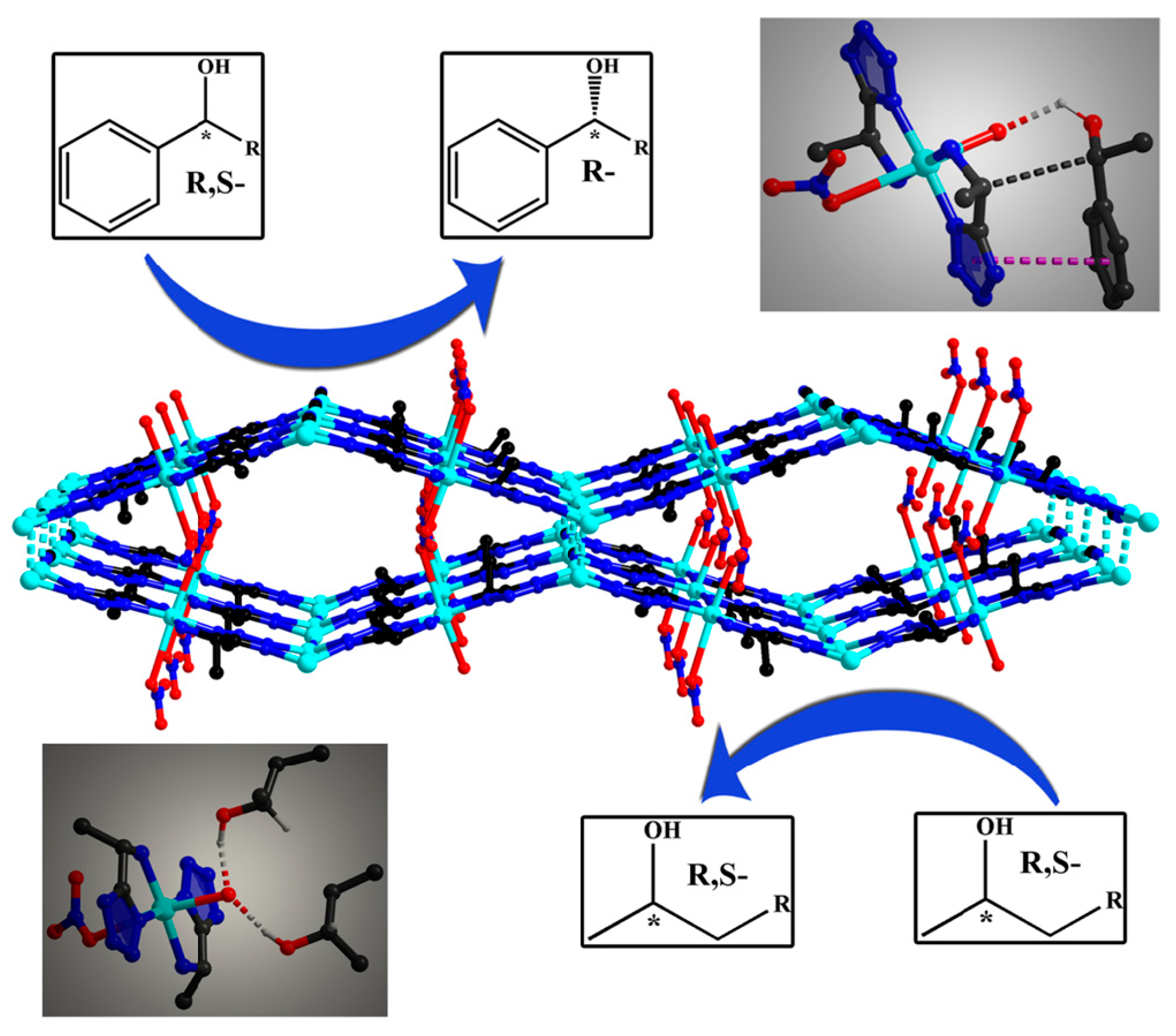
2.1.1. Chiral MOFs Prepared from Chiral Ligands
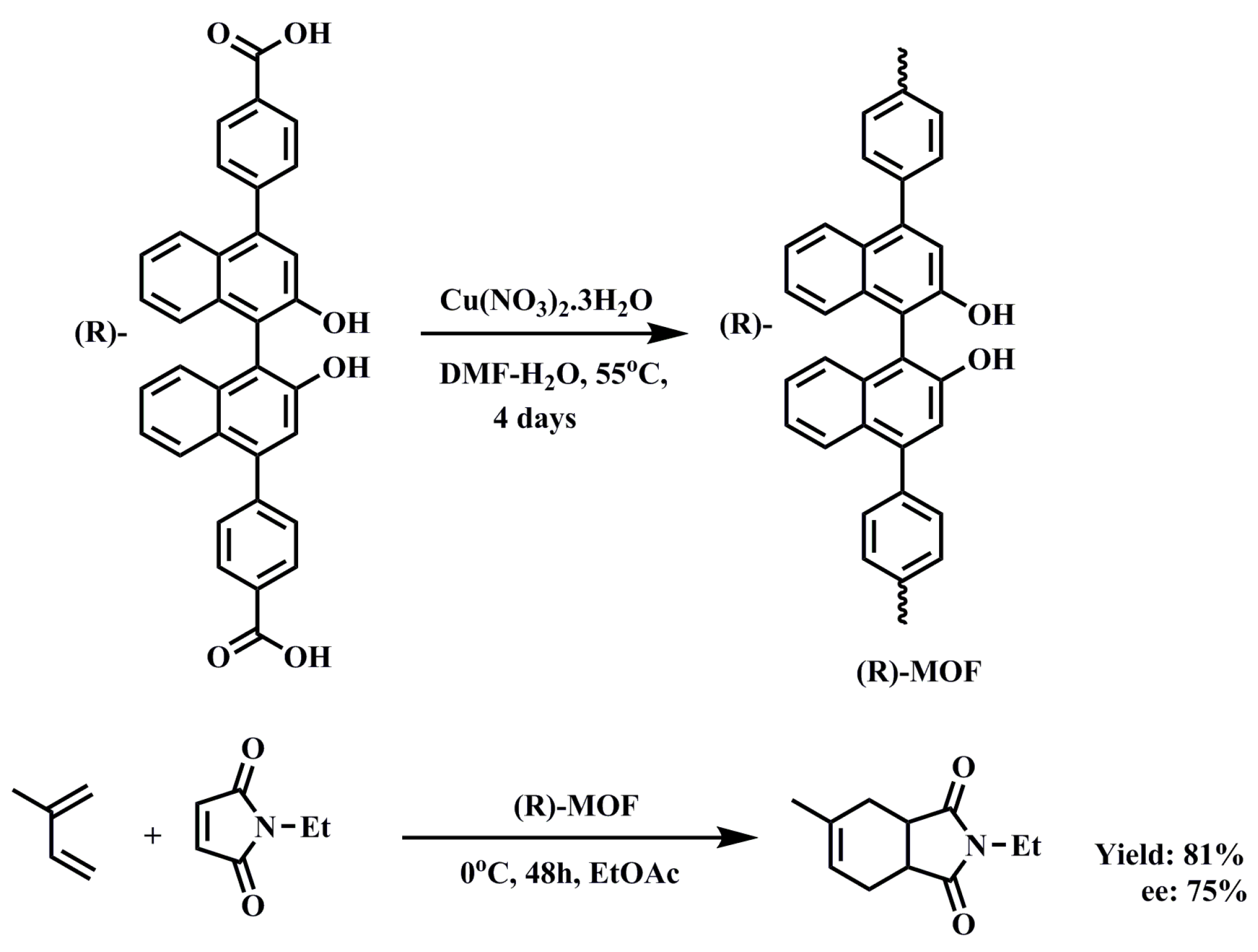
2.1.2. Chiral MOFs from Chiral Salen Ligands
2.2. Indirect Method
2.2.1. Post-Synthetic Modification (PSM)
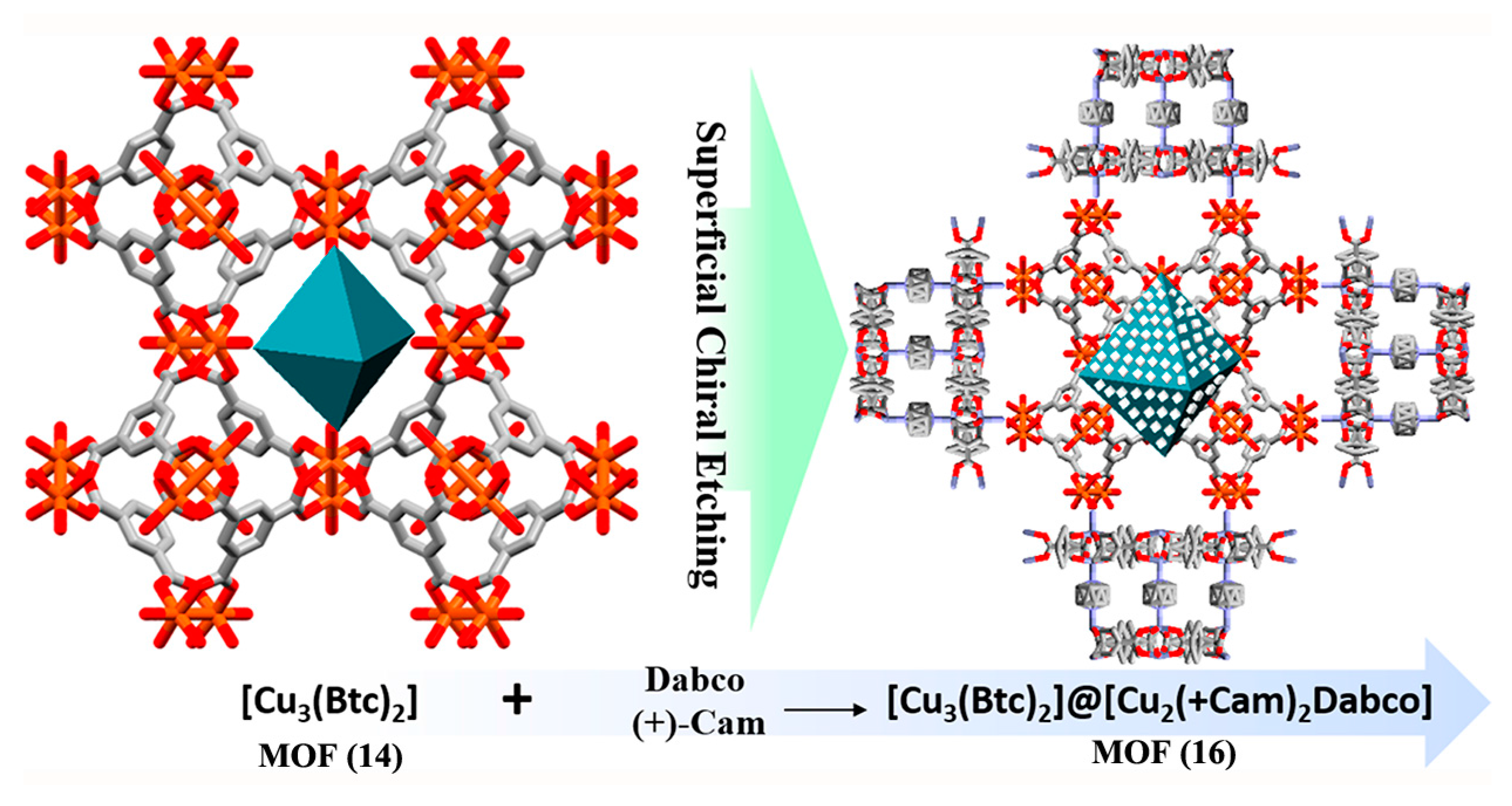
2.2.2. Superficial Chiral Etching Process
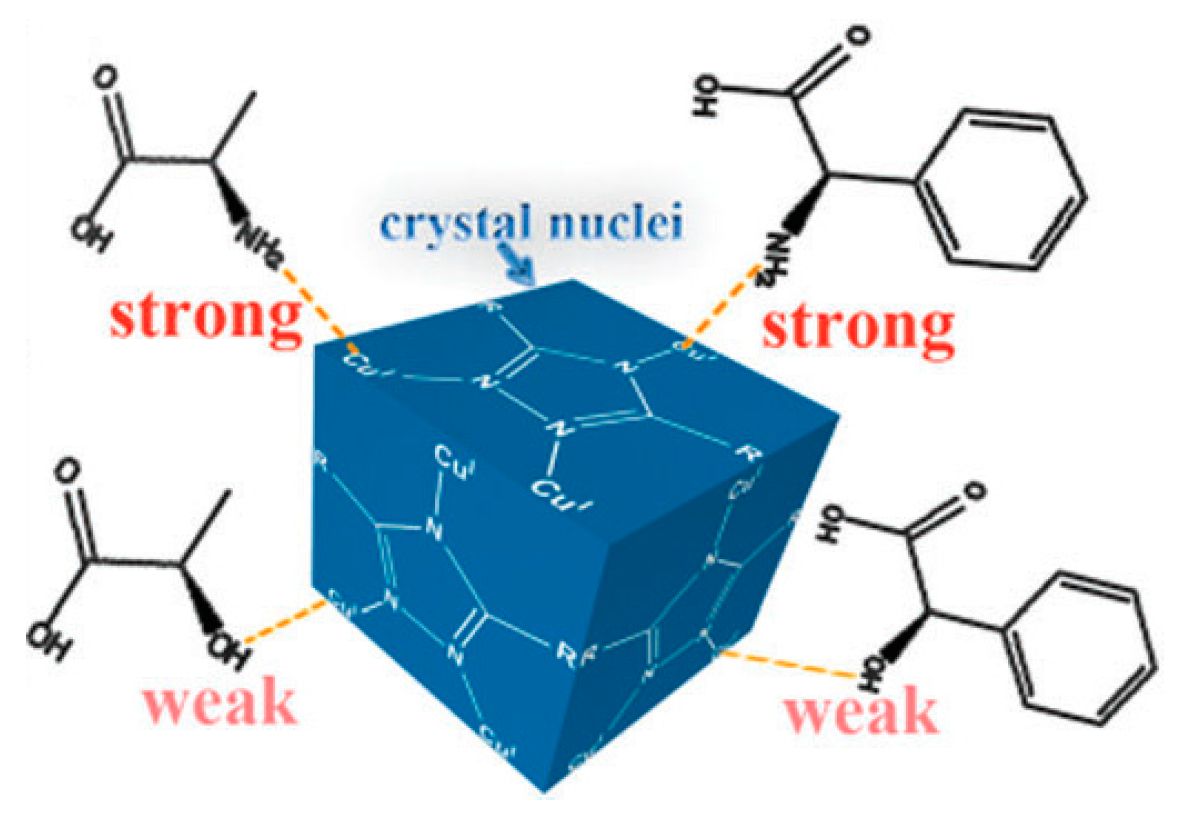
2.2.3. Spontaneous Resolution
2.2.4. Chiral Induction
3. Applications of Chiral MOFs
3.1. Asymmetric Catalysis
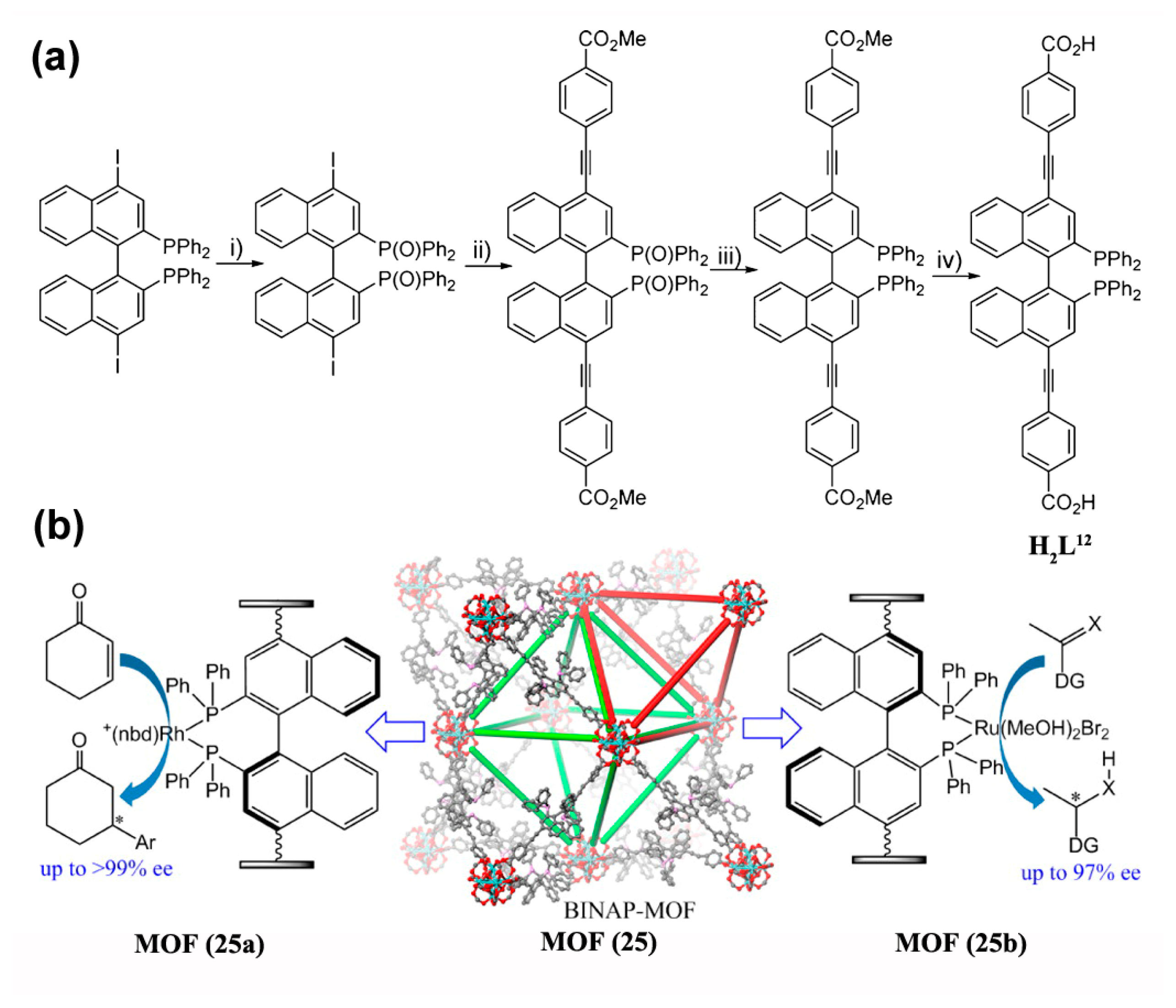
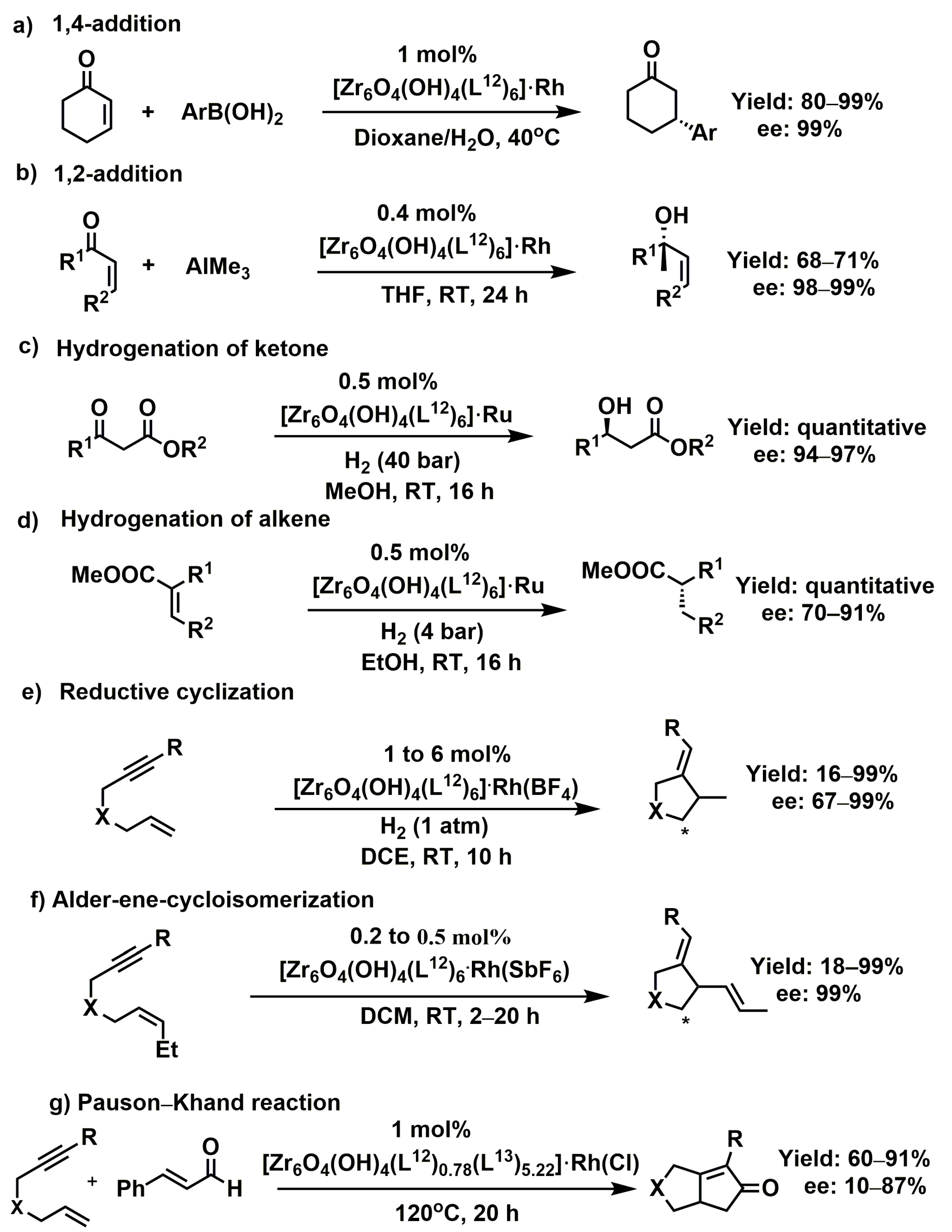
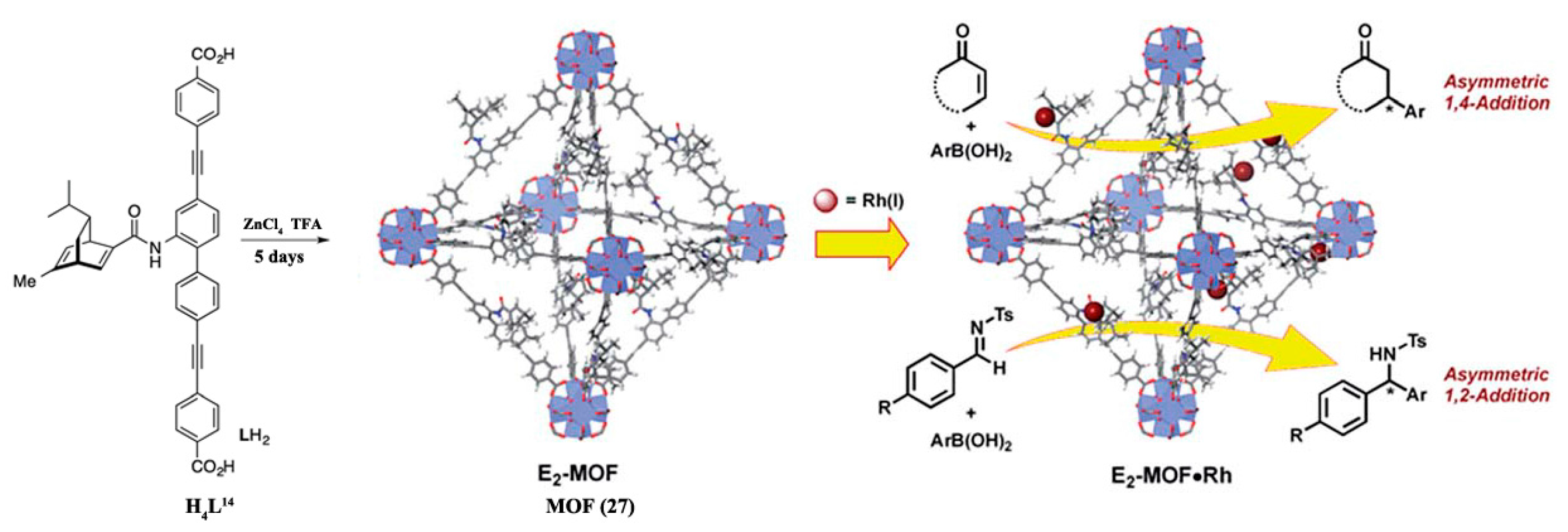
3.1.1. 1,4-, 1,2-, and Cyclo-Addition Reactions
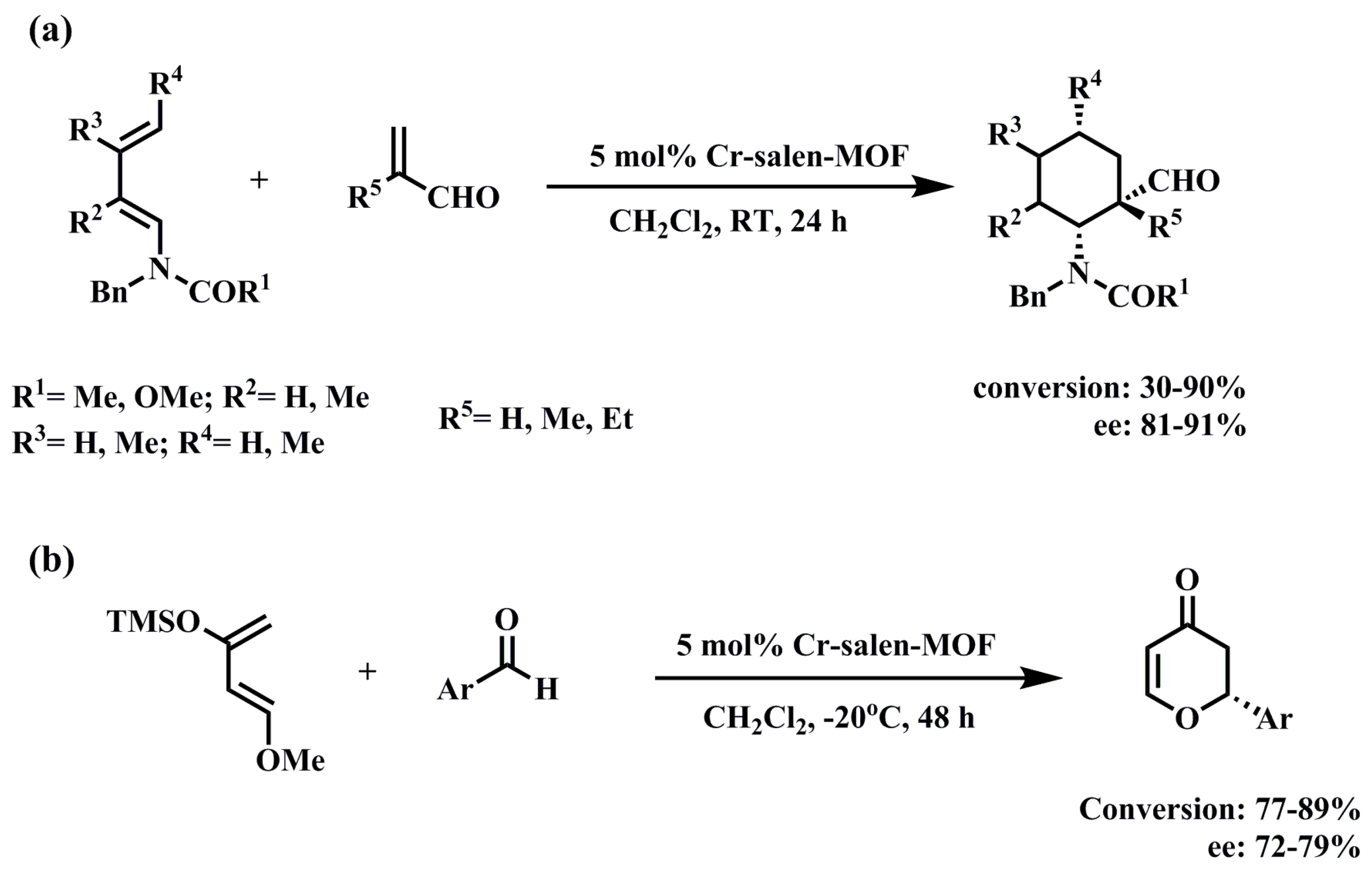
3.1.2. Diels–Alder Reaction
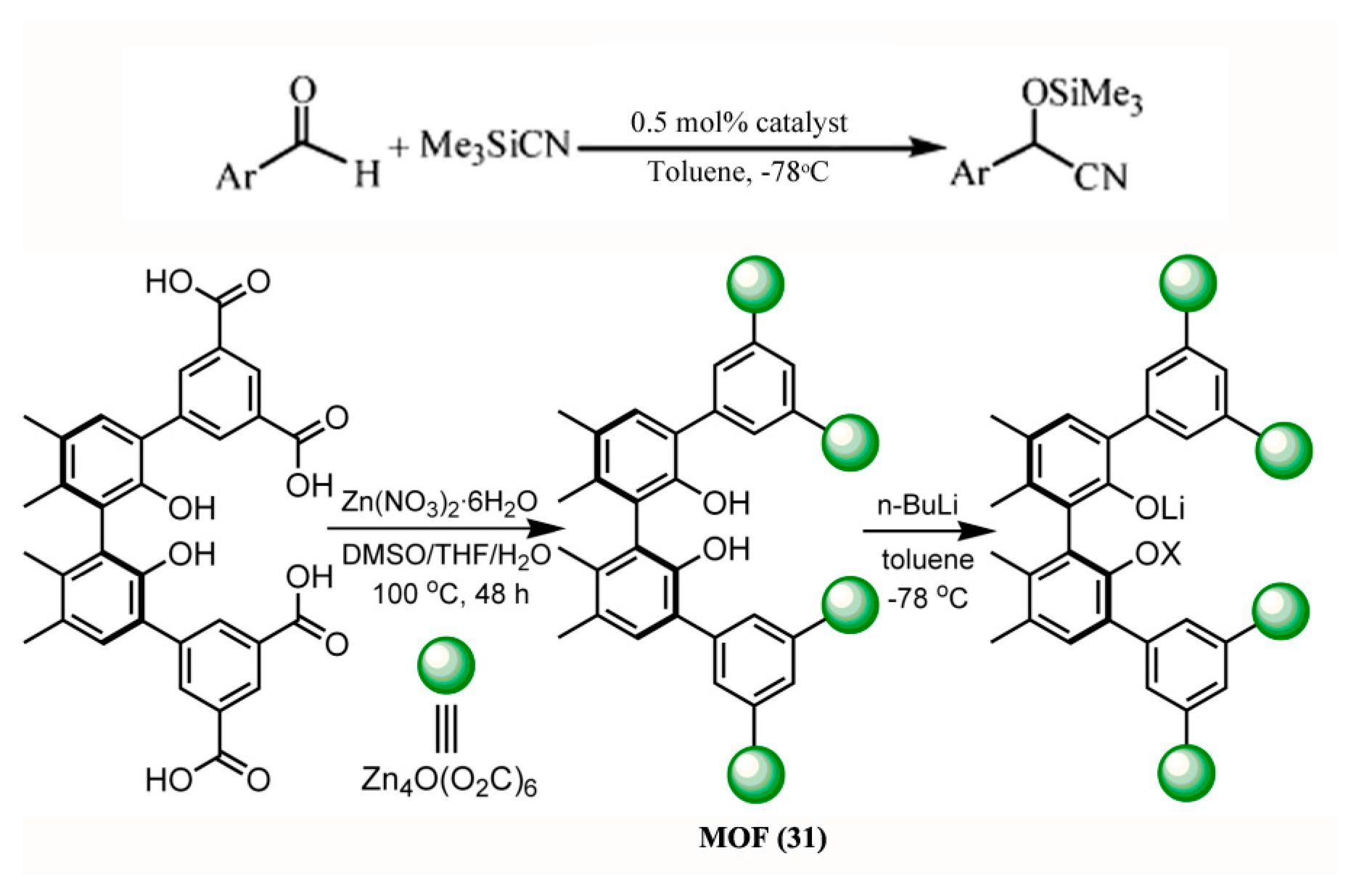
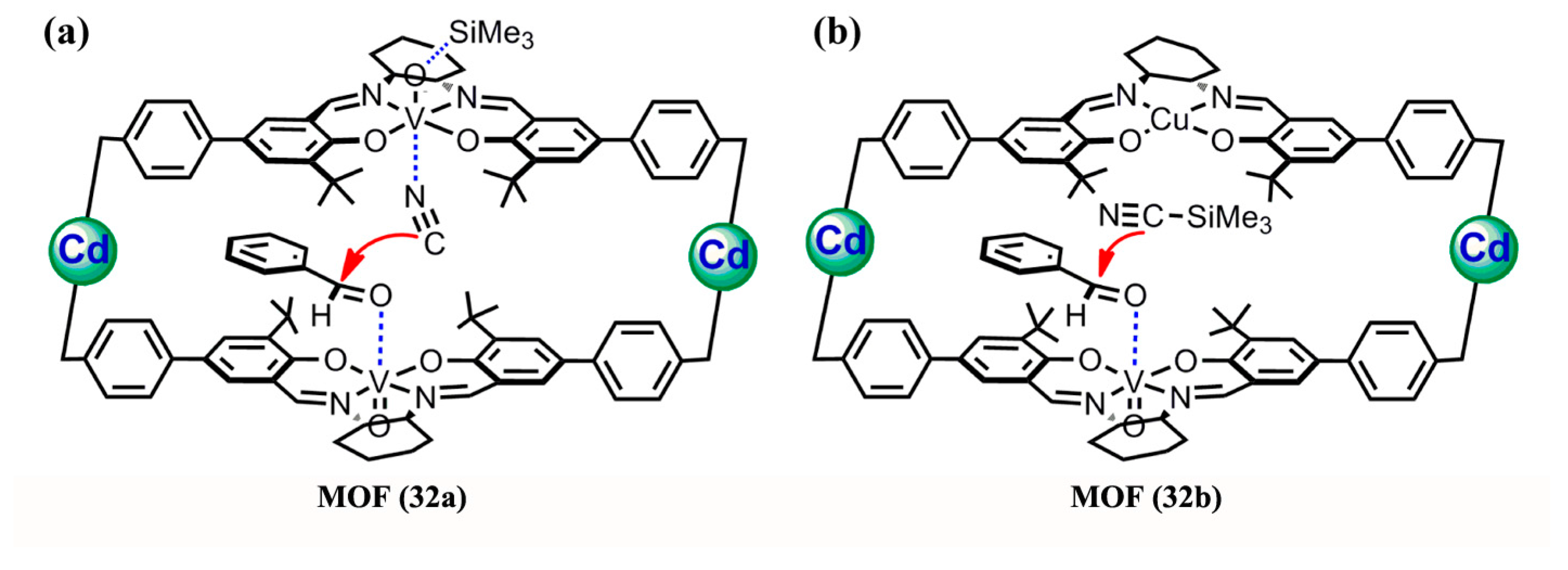
3.1.3. Cyanosilylation of Aldehydes
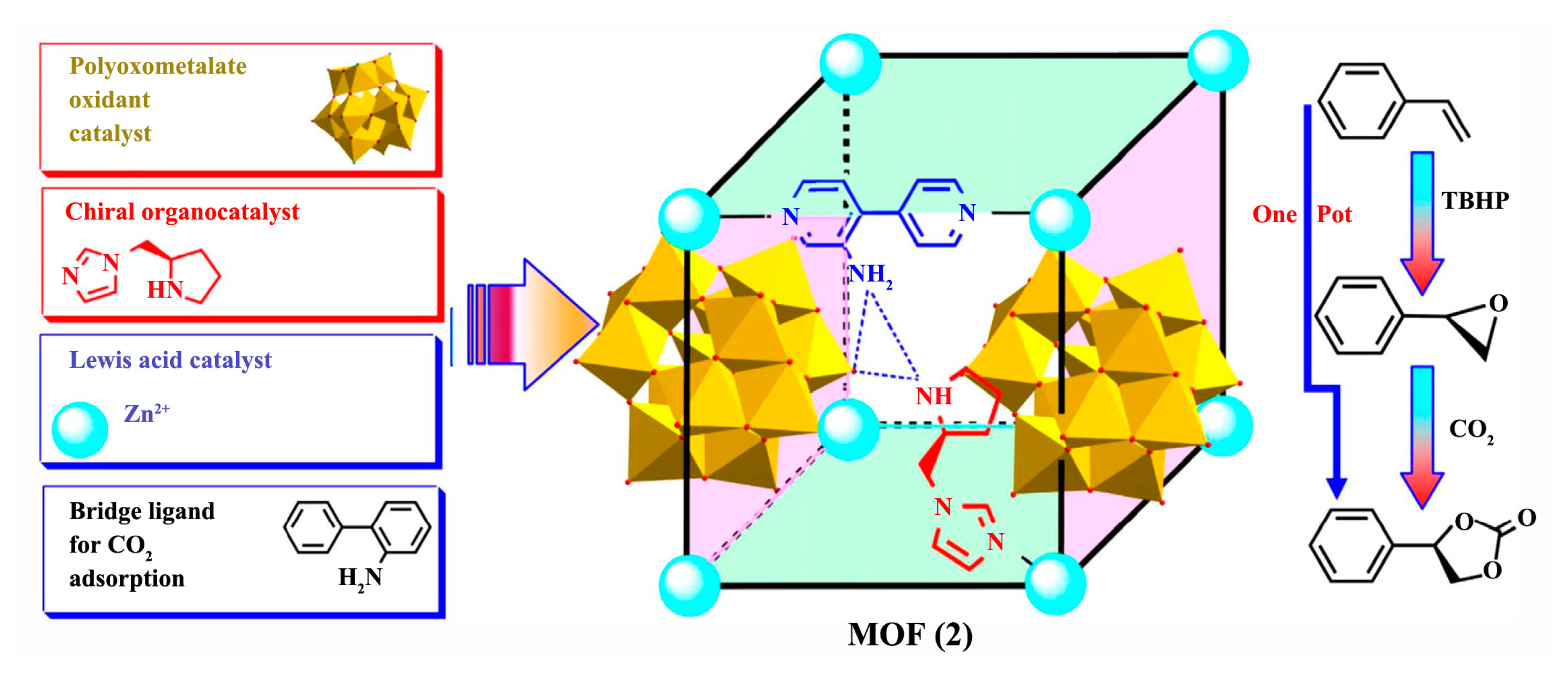
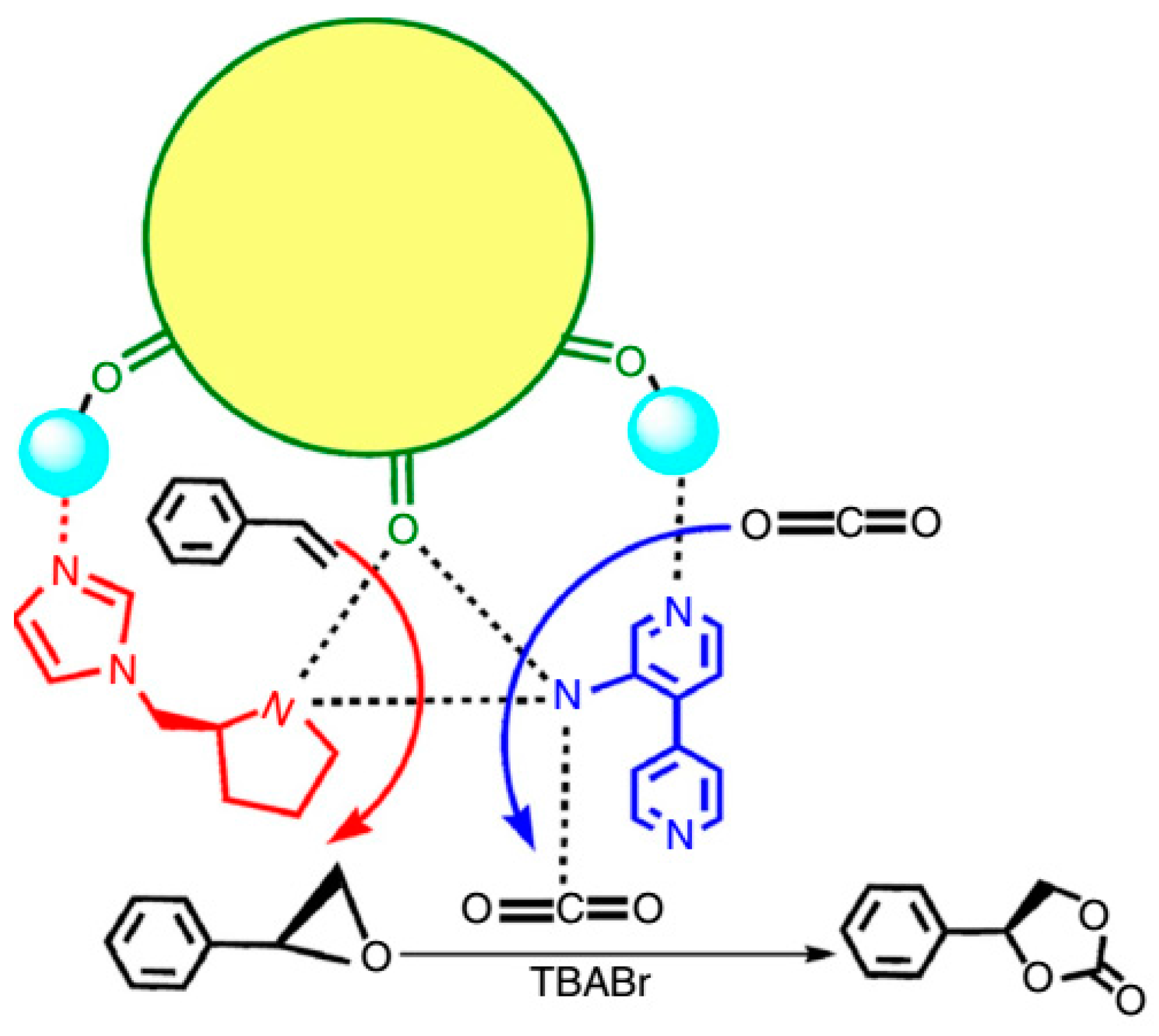
3.1.4. Epoxidation of Alkenes and Cleavage of Epoxide Ring
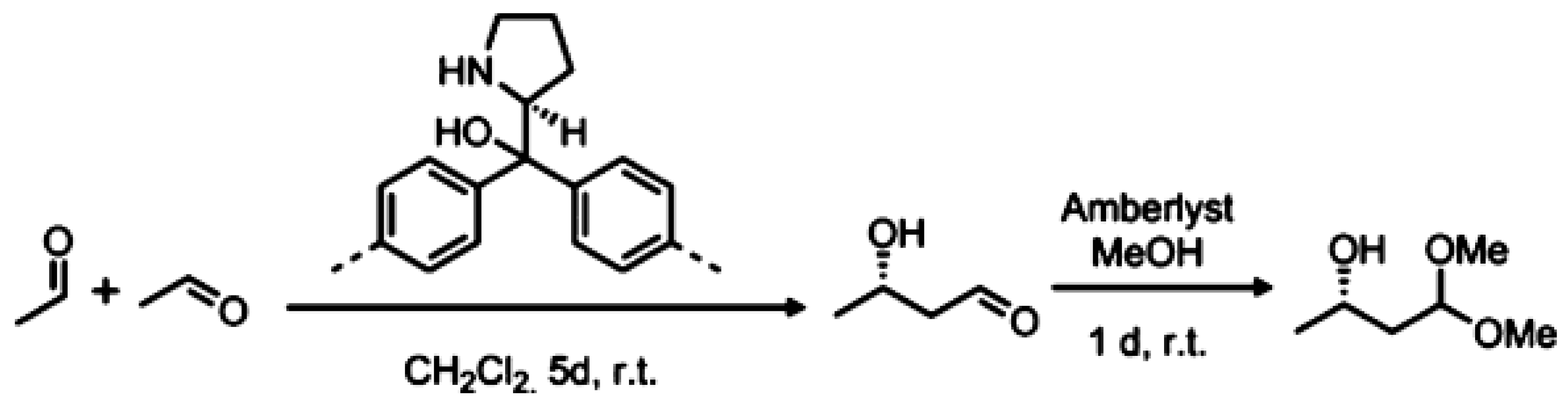
3.1.5. Aldol Condensation
3.1.6. Imine Reduction
3.2. Enantioselective Separation
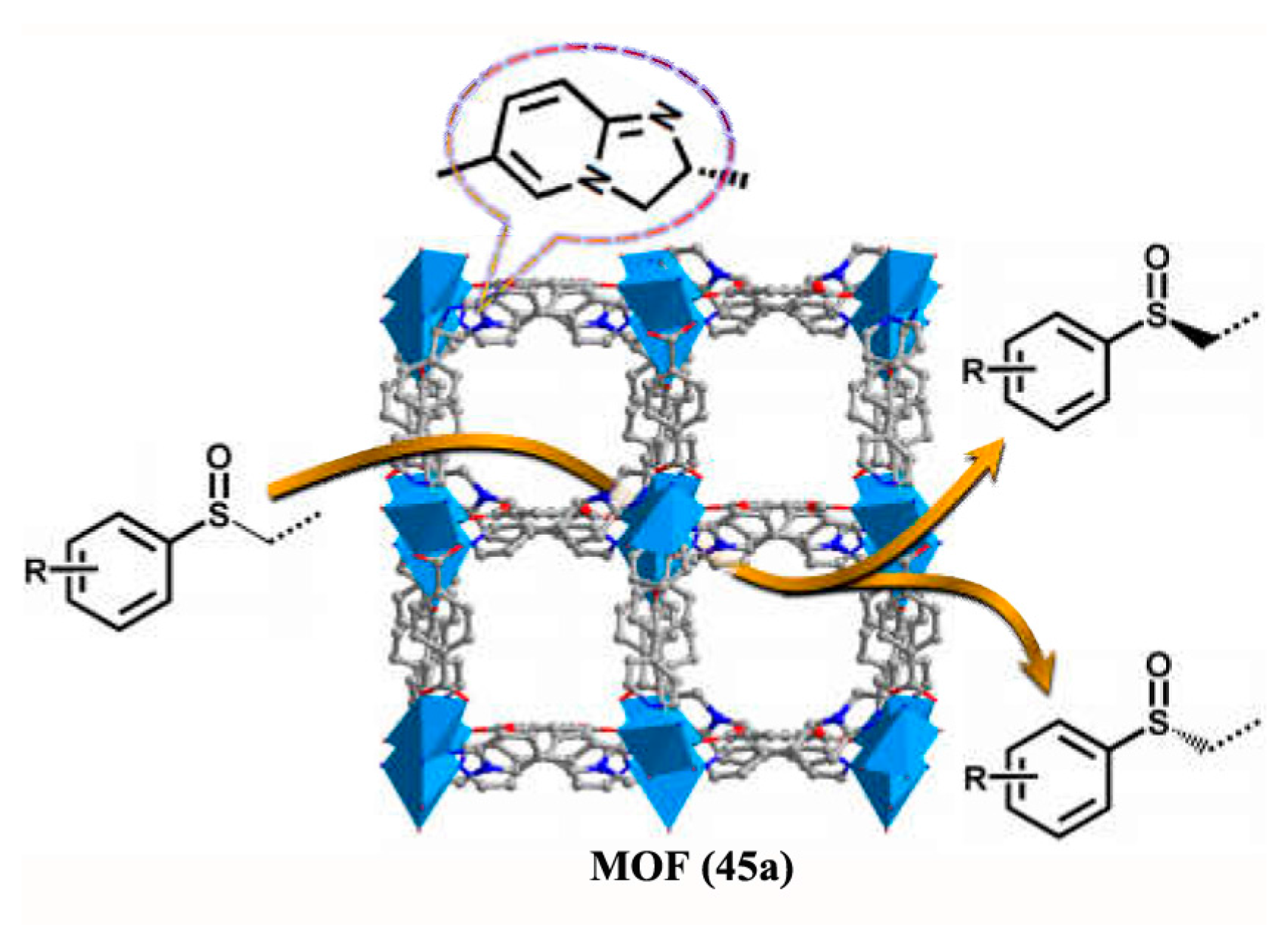
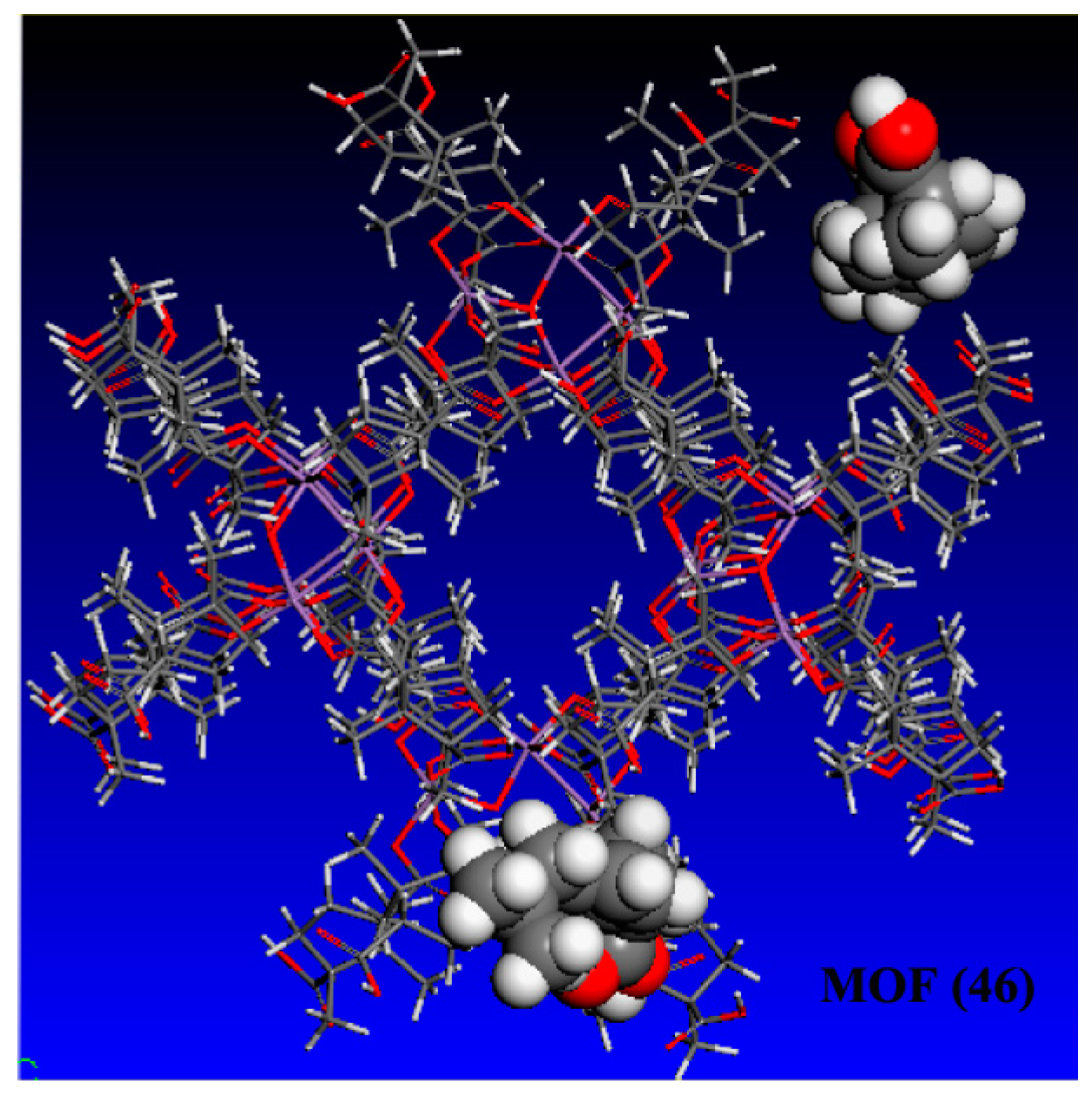
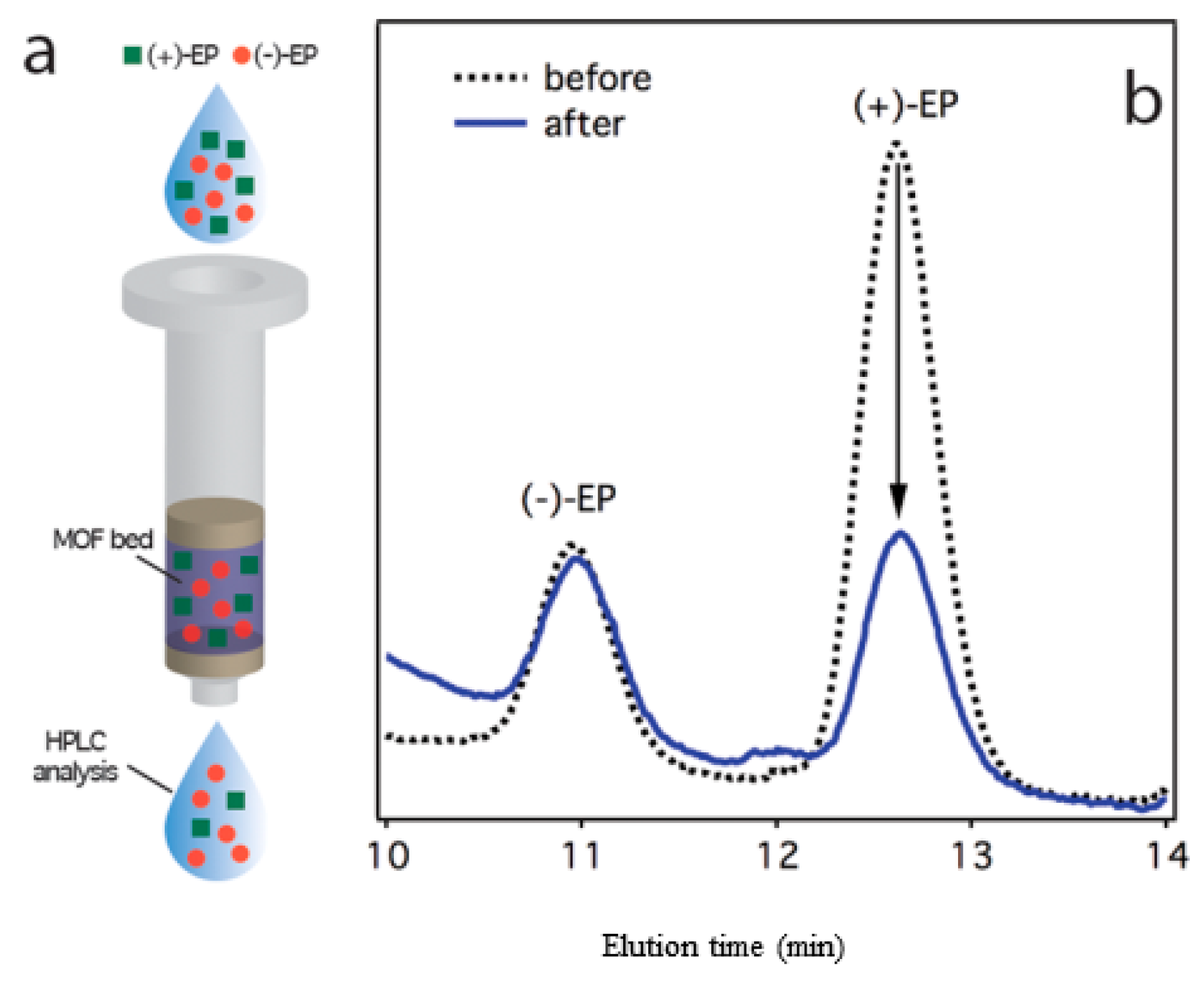
3.2.1. High-performance Liquid Chromatographic (HPLC) Analysis
3.2.2. Gas Chromatographic (GC) Analysis
4. Conclusions and Prospects
Acknowledgments
Conflicts of Interest
References
- Furukawa, H.; Cordova, K.E.; O’Keeffe, M.; Yaghi, O.M. The Chemistry and Applications of Metal–Organic Frameworks. Science 2013, 341, 1230444. [Google Scholar] [CrossRef] [PubMed]
- Kim, C.R.; Uemura, T.; Kitagawa, S. Inorganic nanoparticles in porous coordination polymers. Chem. Soc. Rev. 2016, 45, 3828–3845. [Google Scholar] [CrossRef] [PubMed]
- Zhao, D.; Timmons, D.J.; Yuan, D.; Zhou, H.-C. Tuning the Topology and Functionality of Metal−Organic Frameworks by Ligand Design. Acc. Chem. Res. 2011, 44, 123–133. [Google Scholar] [CrossRef] [PubMed]
- Li, B.; Wen, H.-M.; Cui, Y.; Zhou, W.; Qian, G.; Chen, B. Emerging Multifunctional Metal–Organic Framework Materials. Adv. Mater. 2016, 28, 8819–8860. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Q.-L.; Xu, Q. Metal–organic framework composites. Chem. Soc. Rev. 2014, 43, 5468–5512. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Q.-L.; Li, J.; Xu, Q. Immobilizing Metal Nanoparticles to Metal–Organic Frameworks with Size and Location Control for Optimizing Catalytic Performance. J. Am. Chem. Soc. 2013, 135, 10210–10213. [Google Scholar] [CrossRef] [PubMed]
- Yang, Q.; Xu, Q.; Jiang, H.-L. Metal–Organic Frameworks Meet Metal Nanoparticles: Synergistic Effect for Enhanced Catalysis. Chem. Soc. Rev. 2017, 46, 4774–4808. [Google Scholar] [CrossRef] [PubMed]
- Li, B.; Wen, H.-M.; Zhou, W.; Xu, J.Q.; Chen, B. Porous Metal–Organic Frameworks: Promising Materials for Methane Storage. Chem 2016, 1, 557–580. [Google Scholar] [CrossRef]
- Suh, M.P.; Park, H.J.; Prasad, T.K.; Lim, D.-W. Hydrogen Storage in Metal–Organic Frameworks. Chem. Rev. 2012, 112, 782–835. [Google Scholar] [CrossRef] [PubMed]
- Li, J.-R.; Sculley, J.; Zhou, H.-C. Metal–Organic Frameworks for Separations. Chem. Rev. 2012, 112, 869–932. [Google Scholar] [CrossRef] [PubMed]
- Van de Voorde, B.; Bueken, B.; Denayer, J.; De Vos, D. Adsorptive separation on metal–organic frameworks in the liquid phase. Chem. Soc. Rev. 2014, 43, 5766–5788. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Chang, C.; Wang, X.; Bai, Y.; Liu, H. Applications of homochiral metal‒organic frameworks in enantioselective adsorption and chromatography separation. Electrophoresis 2014, 35, 2733–2743. [Google Scholar] [CrossRef] [PubMed]
- Kreno, L.E.; Leong, K.; Farha, O.K.; Allendorf, M.; Van Duyne, R.P.; Hupp, J.T. Metal–Organic Framework Materials as Chemical Sensors. Chem. Rev. 2012, 112, 1105–1125. [Google Scholar] [CrossRef] [PubMed]
- Cao, K.-L.; Xia, Y.; Wang, G.-X.; Feng, Y.-L. A robust luminescent Ba (II) metal–organic framework based on pyridine carboxylate ligand for sensing of small molecules. Inorg. Chem. Commun. 2015, 53, 42–45. [Google Scholar] [CrossRef]
- Zhao, X.-L.; Tian, D.; Gao, Q.; Sun, H.-W.; Xu, J.; Bu, X.-H. The international journal for high quality, original research in inorganic and organometallic chemistry. Dalton Trans. 2016, 45, 845. [Google Scholar]
- Wanderley, M.M.; Wang, C.; Wu, C.-D.; Lin, W. A Chiral Porous Metal‒Organic Framework for Highly Sensitive and Enantioselective Fluorescence Sensing of Amino Alcohols. J. Am. Chem. Soc. 2012, 134, 9050–9053. [Google Scholar] [CrossRef] [PubMed]
- Qiu, S.; Xue, M.; Zhu, G. Metal–organic framework membranes: From synthesis to separation application. Chem. Soc. Rev. 2014, 43, 6116–6140. [Google Scholar] [CrossRef] [PubMed]
- Duerinck, T.; Denayer, J.F.M. Metal‒organic frameworks as stationary phases for chiral chromatographic and membrane separations. Chem. Eng. Sci. 2015, 124, 179–187. [Google Scholar] [CrossRef]
- Horcajada, P.; Gref, R.; Baati, T.; Allan, P.K.; Maurin, G.; Couvreur, P.; Férey, G.; Morris, R.E.; Serre, C. Metal–Organic Frameworks in Biomedicine. Chem. Rev. 2012, 112, 1232–1268. [Google Scholar] [CrossRef] [PubMed]
- Sha, J.-Q.; Zhong, X.-H.; Wu, L.-H.; Liu, G.-D.; Sheng, N. Nontoxic and renewable metal–organic framework based on [α]-cyclodextrin with efficient drug delivery. RSC Adv. 2016, 6, 82977–82983. [Google Scholar] [CrossRef]
- Liu, M.; Zhang, L.; Wang, T. Supramolecular Chirality in Self-Assembled Systems. Chem. Rev. 2015, 115, 7304–7397. [Google Scholar] [CrossRef] [PubMed]
- Kumar, J.; Thomas, K.G.; Liz-Marzan, L.M. Nanoscale chirality in metal and semiconductor nanoparticles. Chem. Commun. 2016, 52, 12555–12569. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.-M.; Duan, X.; Yao, S.; Wang, Z.; Lin, Z.; Li, Y.-G.; Long, L.-S.; Wang, E.-B.; Lin, W. Cation-mediated optical resolution and anticancer activity of chiral polyoxometalates built from entirely achiral building blocks. Chem. Sci. 2016, 7, 4220–4229. [Google Scholar] [CrossRef]
- Burneo Saavedra, I.P. Metal‒Organic Frameworks Made of Amino Acids and Adenine: Chirality and Hydrochromism. Available online: http://hdl.handle.net/10803/457357 (accessed on 18 March 2018).
- Yoon, M.; Srirambalaji, R.; Kim, K. Homochiral Metal–Organic Frameworks for Asymmetric Heterogeneous Catalysis. Chem. Rev. 2012, 112, 1196–1231. [Google Scholar] [CrossRef] [PubMed]
- Song, F.; Wang, C.; Lin, W. A chiral metal–organic framework for sequential asymmetric catalysis. Chem. Commun. 2011, 47, 8256–8258. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Zheng, M.; Lin, W. Asymmetric Catalysis with Chiral Porous Metal‒Organic Frameworks: Critical Issues. J. Phys. Chem. Lett. 2011, 2, 1701–1709. [Google Scholar] [CrossRef]
- Leus, K.; Liu, Y.Y.; Voort, P.V.D. Metal‒Organic Frameworks as Selective or Chiral Oxidation Catalysts. Catal. Rev. Sci. Eng. 2014, 56, 1–56. [Google Scholar] [CrossRef]
- Liu, S.; Shinde, S.; Pan, J.; Ma, Y.; Yan, Y.; Pan, G. Interface-induced growth of boronate-based metal‒organic framework membrane on porous carbon substrate for aqueous phase molecular recognition. Chem. Eng. J. 2017, 324, 216–227. [Google Scholar] [CrossRef]
- Mendiratta, S.; Lee, C.-H.; Lee, S.-Y.; Kao, Y.-C.; Chang, B.-C.; Lo, Y.-H.; Lu, K.-L. Structural Characteristics and Non-Linear Optical Behaviour of a 2-Hydroxynicotinate-Containing Zinc-Based Metal–Organic Framework. Molecules 2015, 20, 8941–8951. [Google Scholar] [CrossRef] [PubMed]
- Li, H.-Y.; Xu, H.; Zang, S.-Q.; Mak, T.C.W. A viologen-functionalized chiral Eu-MOF as a platform for multifunctional switchable material. Chem. Commun. 2016, 52, 525–528. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.-Q.; Liu, Q.; Liu, L.-H.; Ding, L.; Chen, Y.-P. Two 3D nonlinear optical and luminescent lanthanide–organic frameworks with multidirectional helical intersecting channels. New J. Chem. 2017, 41, 6736–6741. [Google Scholar] [CrossRef]
- Kong, J.; Zhang, M.; Duan, A.-H.; Zhang, J.-H.; Yang, R.; Yuan, L.-M. Homochiral metal–organic framework used as a stationary phase for high-performance liquid chromatography. J. Sep. Sci. 2015, 38, 556–561. [Google Scholar] [CrossRef] [PubMed]
- Li, P.; He, Y.; Guang, J.; Weng, L.; Zhao, J.C.-G.; Xiang, S.; Chen, B. A Homochiral Microporous Hydrogen-Bonded Organic Framework for Highly Enantioselective Separation of Secondary Alcohols. J. Am. Chem. Soc. 2014, 136, 547–549. [Google Scholar] [CrossRef] [PubMed]
- Okamoto, Y.; Ikai, T. Chiral HPLC for efficient resolution of enantiomers. Chem. Soc. Rev. 2008, 37, 2593–2608. [Google Scholar] [CrossRef] [PubMed]
- Cavazzini, A.; Pasti, L.; Massi, A.; Marchetti, N.; Dondi, F. Recent applications in chiral high performance liquid chromatography: A review. Anal. Chim. Acta 2011, 706, 205–222. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.; Wang, X.; Bai, Y.; Liu, H. Applications of nanomaterials in enantioseparation and related techniques. Trends Anal. Chem. 2012, 39, 195–206. [Google Scholar] [CrossRef]
- Lin, W. Asymmetric Catalysis with Chiral Porous Metal–Organic Frameworks. Top. Catal. 2010, 53, 869–875. [Google Scholar] [CrossRef]
- Ma, L.; Abney, C.; Lin, W. Enantioselective catalysis with homochiral metal–organic frameworks. Chem. Soc. Rev. 2009, 38, 1248–1256. [Google Scholar] [CrossRef] [PubMed]
- Gu, Z.-G.; Zhan, C.; Zhang, J.; Bu, X. Chiral chemistry of metal–camphorate frameworks. Chem. Soc. Rev. 2016, 45, 3122–3144. [Google Scholar] [CrossRef] [PubMed]
- Nickerl, G.; Henschel, A.; Grünker, R.; Gedrich, K.; Kaskel, S. Chiral Metal–Organic Frameworks and Their Application in Asymmetric Catalysis and Stereoselective Separation. Chem. Ing. Tech. 2011, 83, 90–103. [Google Scholar] [CrossRef]
- Bisht, K.K.; Parmar, B.; Rachuri, Y.; Kathalikattil, A.C.; Suresh, E. Progress in the synthetic and functional aspects of chiral metal–organic frameworks. CrystEngComm 2015, 17, 5341–5356. [Google Scholar] [CrossRef]
- Han, Z.; Shi, W.; Cheng, P. Synthetic strategies for chiral metal–organic frameworks. Chin. Chem. Lett. 2017. [Google Scholar] [CrossRef]
- Seo, J.S.; Whang, D.; Lee, H.; Jun, S.I.; Oh, J.; Jeon, Y.J.; Kim, K. A homochiral metal–organic porous material for enantioselective separation and catalysis. Nature 2000, 404, 982. [Google Scholar] [CrossRef] [PubMed]
- Ingleson, M.J.; Barrio, J.P.; Bacsa, J.; Dickinson, C.; Park, H.; Rosseinsky, M.J. Generation of a solid Bronsted acid site in a chiral framework. Chem. Commun. 2008, 1287–1289. [Google Scholar] [CrossRef] [PubMed]
- Rebilly, J.-N.; Bacsa, J.; Rosseinsky, M.J. 1 D Tubular and 2 D Metal–Organic Frameworks Based on a Flexible Amino Acid Derived Organic Spacer. Chem. Asian J. 2009, 4, 892–903. [Google Scholar] [CrossRef] [PubMed]
- Sartor, M.; Stein, T.; Hoffmann, F.; Fröba, M. A New Set of Isoreticular, Homochiral Metal–Organic Frameworks with ucp Topology. Chem. Mater. 2016, 28, 519–528. [Google Scholar] [CrossRef]
- Zhu, Q.; Sheng, T.; Fu, R.; Tan, C.; Hu, S.; Wu, X. Two luminescent enantiomorphic 3D metal–organic frameworks with 3D homochiral double helices. Chem. Commun. 2010, 46, 9001–9003. [Google Scholar] [CrossRef] [PubMed]
- Hu, G.; Zhang, H.; Miao, H.; Wang, J.; Xu, Y. Synthesis, structures and properties of two new chiral rare earth–organic frameworks constructed by l/d-tartaric acid. J. Solid State Chem. 2015, 229, 208–212. [Google Scholar] [CrossRef]
- Ren, W.X.; Jian, L.Z.; Wei, G.; Yan, L.; Zhan, L.B.; Yong, C. A Novel TADDOL-based Chiral Metal–organic Framework: Synthesis, Structure and Photoluminescence Study. Chin. J. Struct. Chem. 2016, 35, 1399–1405. [Google Scholar]
- Evans, O.R.; Ngo, H.L.; Lin, W. Chiral Porous Solids Based on Lamellar Lanthanide Phosphonates. J. Am. Chem. Soc. 2001, 123, 10395–10396. [Google Scholar] [CrossRef] [PubMed]
- Ma, L.; Falkowski, J.M.; Abney, C.; Lin, W. A series of isoreticular chiral metal–organic frameworks as a tunable platform for asymmetric catalysis. Nat. Chem. 2010, 2, 838. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.D.; Hu, A.; Zhang, L.; Lin, W. A Homochiral Porous Metal−Organic Framework for Highly Enantioselective Heterogeneous Asymmetric Catalysis. J. Am. Chem. Soc. 2005, 127, 8940–8941. [Google Scholar] [CrossRef] [PubMed]
- Han, Q.; Qi, B.; Ren, W.; He, C.; Niu, J.; Duan, C. Polyoxometalate-based homochiral metal–organic frameworks for tandem asymmetric transformation of cyclic carbonates from olefins. Nat. Commun. 2015, 6, 10007. [Google Scholar] [CrossRef] [PubMed]
- Kuang, X.; Ye, S.; Li, X.; Ma, Y.; Zhang, C.; Tang, B. A new type of surface-enhanced Raman scattering sensor for the enantioselective recognition of d/l-cysteine and d/l-asparagine based on a helically arranged Ag NPs@homochiral MOF. Chem. Commun. 2016, 52, 5432–5435. [Google Scholar] [CrossRef] [PubMed]
- Li, M.-Y.; Wang, F.; Gu, Z.-G.; Zhang, J. Synthesis of homochiral zeolitic metal–organic frameworks with amino acid and tetrazolates for chiral recognition. RSC Adv. 2017, 7, 4872–4875. [Google Scholar] [CrossRef]
- Gupta, A.K.; De, D.; Katoch, R.; Garg, A.; Bharadwaj, P.K. Synthesis of a NbO Type Homochiral Cu(II) Metal–Organic Framework: Ferroelectric Behavior and Heterogeneous Catalysis of Three-Component Coupling and Pechmann Reactions. Inorg. Chem. 2017, 56, 4697–4705. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Trickett, C.A.; Alahmadi, S.B.; Alshammari, A.S.; Yaghi, O.M. Calcium l-Lactate Frameworks as Naturally Degradable Carriers for Pesticides. J. Am. Chem. Soc. 2017, 139, 8118–8121. [Google Scholar] [CrossRef] [PubMed]
- Robin, J.; Audebrand, N.; Poriel, C.; Canivet, J.; Calvez, G.; Roisnel, T.; Dorcet, V.; Roussel, P. A series of chiral metal–organic frameworks based on fluorene di- and tetra-carboxylates: Syntheses, crystal structures and luminescence properties. CrystEngComm 2017, 19, 2042–2056. [Google Scholar] [CrossRef]
- Kutzscher, C.; Janssen-Muller, D.; Notzon, A.; Stoeck, U.; Bon, V.; Senkovska, I.; Kaskel, S.; Glorius, F. Synthesis of the homochiral metal–organic framework DUT-129 based on a chiral dicarboxylate linker with 6 stereocenters. CrystEngComm 2017, 19, 2494–2499. [Google Scholar] [CrossRef]
- Cho, S.-H.; Ma, B.; Nguyen, S.T.; Hupp, J.T.; Albrecht-Schmitt, T.E. A metal–organic framework material that functions as an enantioselective catalyst for olefin epoxidation. Chem. Commun. 2006, 2563–2565. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Li, Z.; Yuan, G.; Xia, Q.; Yuan, C.; Cui, Y. Chiral Cu(salen)-Based Metal–Organic Framework for Heterogeneously Catalyzed Aziridination and Amination of Olefins. Inorg. Chem. 2016, 55, 12500–12503. [Google Scholar] [CrossRef] [PubMed]
- Fan, Y.; Li, J.; Ren, Y.; Jiang, H. A Ni(salen)-Based Metal–Organic Framework: Synthesis, Structure, and Catalytic Performance for CO2 Cycloaddition with Epoxides. Eur. J. Inorg. Chem. 2017, 4982–4989. [Google Scholar] [CrossRef]
- Li, J.; Ren, Y.; Qi, C.; Jiang, H. A chiral salen-based MOF catalytic material with high thermal, aqueous and chemical stabilities. Dalton Trans. 2017, 46, 7821–7832. [Google Scholar] [CrossRef] [PubMed]
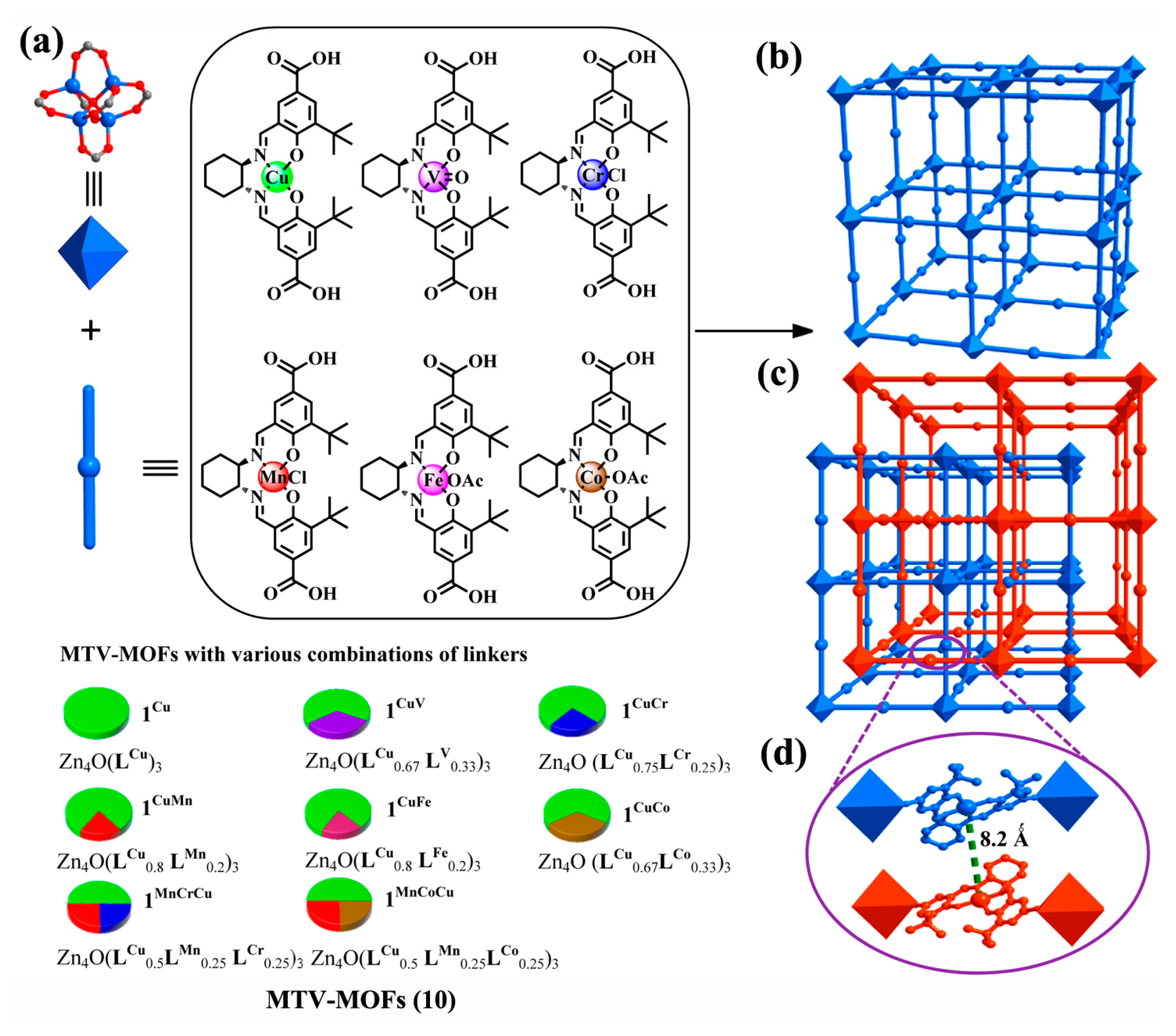
- Xia, Q.; Li, Z.; Tan, C.; Liu, Y.; Gong, W.; Cui, Y. Multivariate Metal–Organic Frameworks as Multifunctional Heterogeneous Asymmetric Catalysts for Sequential Reactions. J. Am. Chem. Soc. 2017, 139, 8259–8266. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Cohen, S.M. Postsynthetic modification of metal–organic frameworks. Chem. Soc. Rev. 2009, 38, 1315–1329. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Cohen, S.M. Tandem Modification of Metal–Organic Frameworks by a Postsynthetic Approach. Angew. Chem. Int. Ed. 2008, 47, 4699–4702. [Google Scholar] [CrossRef] [PubMed]
- Tanabe, K.K.; Cohen, S.M. Engineering a Metal–Organic Framework Catalyst by Using Postsynthetic Modification. Angew. Chem. Int. Ed. 2009, 48, 7424–7427. [Google Scholar] [CrossRef] [PubMed]
- Garibay, S.J.; Wang, Z.; Tanabe, K.K.; Cohen, S.M. Postsynthetic Modification: A Versatile Approach toward Multifunctional Metal–Organic Frameworks. Inorg. Chem. 2009, 48, 7341–7349. [Google Scholar] [CrossRef] [PubMed]
- Bonnefoy, J.; Legrand, A.; Quadrelli, E.A.; Canivet, J.; Farrusseng, D. Enantiopure Peptide-Functionalized Metal–Organic Frameworks. J. Am. Chem. Soc. 2015, 137, 9409–9416. [Google Scholar] [CrossRef] [PubMed]
- Chen, D.; Luo, R.; Li, M.; Wen, M.; Li, Y.; Chen, C.; Zhang, N. Salen(Co(III)) imprisoned within pores of a metal–organic framework by post-synthetic modification and its asymmetric catalysis for CO2 fixation at room temperature. Chem. Commun. 2017, 53, 10930–10933. [Google Scholar] [CrossRef] [PubMed]
- Hou, X.; Xu, T.; Wang, Y.; Liu, S.; Tong, J.; Liu, B. Superficial Chiral Etching on Achiral Metal–Organic Framework for Enantioselective Sorption. ACS Appl. Mater. Interfaces 2017, 9, 32264–32269. [Google Scholar] [CrossRef] [PubMed]
- Ezuhara, T.; Endo, K.; Aoyama, Y. Helical Coordination Polymers from Achiral Components in Crystals. Homochiral Crystallization, Homochiral Helix Winding in the Solid State, and Chirality Control by Seeding. J. Am. Chem. Soc. 1999, 121, 3279–3283. [Google Scholar] [CrossRef]
- Li, Z.; Du, L.; Zhou, J.; Li, L.; Hu, Y.; Qiao, Y.; Xie, M.; Zhao, Q. A chiral porous cobalt-organic framework based on reinforced sinusoidal-like SBUs involving in situ-generated formate. New J. Chem. 2013, 37, 2473–2478. [Google Scholar] [CrossRef]
- Durá, G.; Carrión, M.C.; Jalón, F.A.; Rodríguez, A.M.; Manzano, B.R. Self-Assembly of Silver(I) and Ditopic Heteroscorpionate Ligands. Spontaneous Chiral Resolution in Helices and Sequence Isomerism in Coordination Polymers. Cryst. Growth Des. 2013, 13, 3275–3282. [Google Scholar] [CrossRef]
- Liu, W.; Bao, X.; Mao, L.-L.; Tucek, J.; Zboril, R.; Liu, J.-L.; Guo, F.-S.; Ni, Z.-P.; Tong, M.-L. A chiral spin crossover metal–organic framework. Chem. Commun. 2014, 50, 4059–4061. [Google Scholar] [CrossRef] [PubMed]
- Sun, Q.; Cheng, A.-L.; Wang, K.; Yi, X.-C.; Gao, E.-Q. Chiral or achiral: Four isomeric Cd(II) coordination polymers based on phenylenediacrylate ligands. CrystEngComm 2015, 17, 1389–1397. [Google Scholar] [CrossRef]
- Mei, H.-X.; Zhang, T.; Wang, D.-F.; Huang, R.-B.; Zheng, L.-S. A Zn-oxalate helix linked by a water helix: Spontaneous chiral resolution of a Zn helical coordination polymer. New J. Chem. 2015, 39, 2075–2080. [Google Scholar] [CrossRef]
- Li, X.; Yu, Z.; Li, X.; Guo, X. Solvent-Mediated Transformation from Achiral to Chiral Nickel(II) Metal–Organic Frameworks and Reassembly in Solution. Chem. Eur. J. 2015, 21, 16593–16600. [Google Scholar] [CrossRef] [PubMed]
- Verma, A.; De, D.; Tomar, K.; Bharadwaj, P.K. Chiral Cadmium(II) Metal–Organic Framework from an Achiral Ligand by Spontaneous Resolution: An Efficient Heterogeneous Catalyst for the Strecker Reaction of Ketones. Inorg. Chem. 2017, 56, 13629–13633. [Google Scholar] [CrossRef] [PubMed]
- Lin, Z.; Slawin, A.M.Z.; Morris, R.E. Chiral Induction in the Ionothermal Synthesis of a 3-D Coordination Polymer. J. Am. Chem. Soc. 2007, 129, 4880–4881. [Google Scholar] [CrossRef] [PubMed]
- Morris, R.E.; Bu, X. Induction of chiral porous solids containing only achiral building blocks. Nat. Chem. 2010, 2, 353. [Google Scholar] [CrossRef] [PubMed]
- Jing, X.; He, C.; Dong, D.; Yang, L.; Duan, C. Homochiral Crystallization of Metal–Organic Silver Frameworks: Asymmetric [3+2] Cycloaddition of an Azomethine Ylide. Angew. Chem. Int. Ed. 2012, 51, 10127–10131. [Google Scholar] [CrossRef] [PubMed]
- Bisht, K.K.; Suresh, E. Spontaneous Resolution to Absolute Chiral Induction: Pseudo-Kagomé Type Homochiral Zn(II)/Co(II) Coordination Polymers with Achiral Precursors. J. Am. Chem. Soc. 2013, 135, 15690–15693. [Google Scholar] [CrossRef] [PubMed]
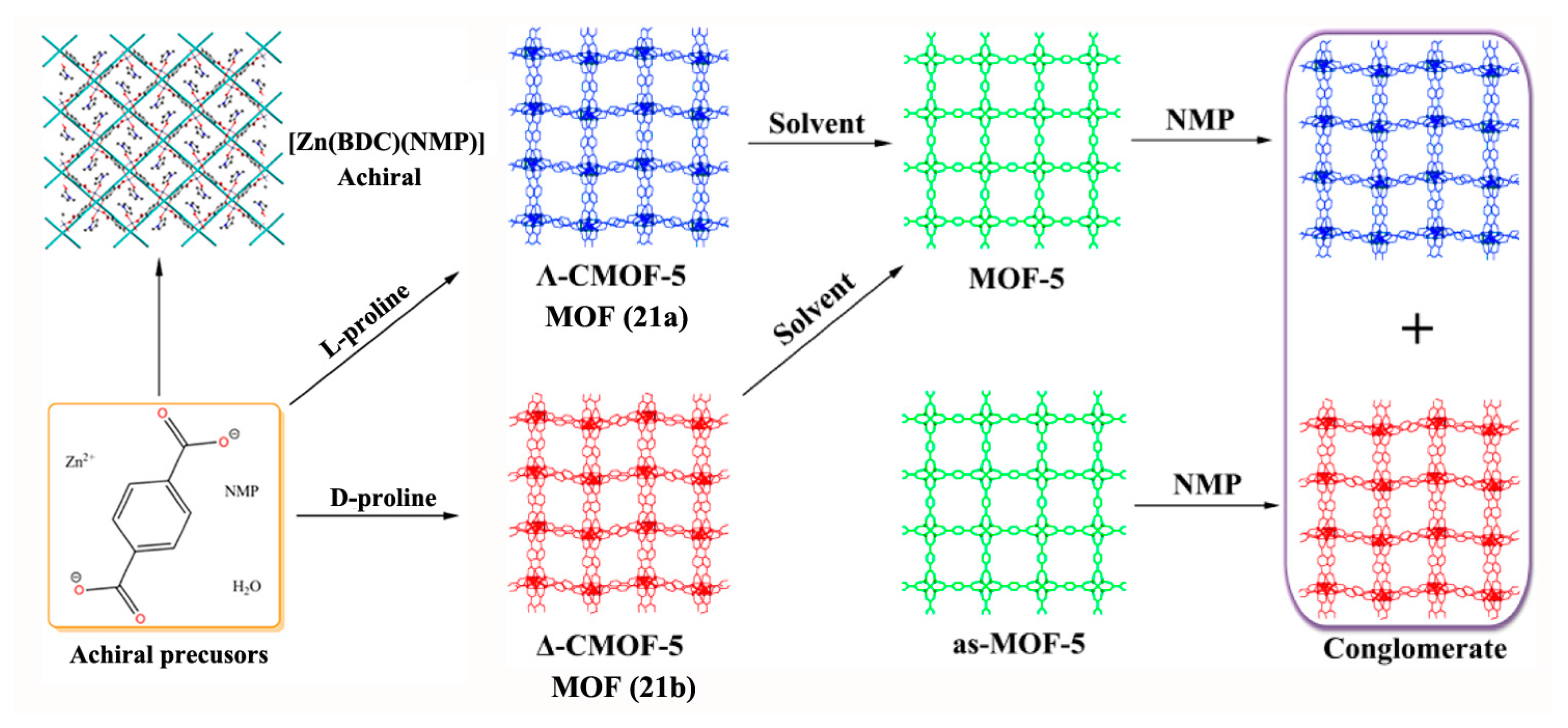
- Zhang, S.-Y.; Li, D.; Guo, D.; Zhang, H.; Shi, W.; Cheng, P.; Wojtas, L.; Zaworotko, M.J. Synthesis of a Chiral Crystal Form of MOF-5, CMOF-5, by Chiral Induction. J. Am. Chem. Soc. 2015, 137, 15406–15409. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Weng, Z.; Wang, Y.; Chen, L.; Sheng, D.; Liu, Y.; Diwu, J.; Chai, Z.; Albrecht-Schmitt, T.E.; Wang, S. Centrosymmetric and chiral porous thorium organic frameworks exhibiting uncommon thorium coordination environments. Dalton Trans. 2015, 44, 20867–20873. [Google Scholar] [CrossRef] [PubMed]
- Song, B.-Q.; Chen, D.-Q.; Ji, Z.; Tang, J.; Wang, X.-L.; Zang, H.-Y.; Su, Z.-M. Control of bulk homochirality and proton conductivity in isostructural chiral metal–organic frameworks. Chem. Commun. 2017, 53, 1892–1895. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.-T.; Cai, Z.-W.; Ye, Q.-Y.; Weng, C.-H.; Huang, X.-H.; Hu, X.-L.; Huang, C.-C.; Zhuang, N.-F. Enantioselective Synthesis of a Chiral Coordination Polymer with Circularly Polarized Visible Laser. Angew. Chem. Int. Ed. 2014, 53, 12860–12864. [Google Scholar] [CrossRef] [PubMed]
- Falkowski, J.M.; Sawano, T.; Zhang, T.; Tsun, G.; Chen, Y.; Lockard, J.V.; Lin, W. Privileged Phosphine-Based Metal–Organic Frameworks for Broad-Scope Asymmetric Catalysis. J. Am. Chem. Soc. 2014, 136, 5213–5216. [Google Scholar] [CrossRef] [PubMed]
- Sawano, T.; Thacker, N.C.; Lin, Z.; McIsaac, A.R.; Lin, W. Robust, Chiral, and Porous BINAP-Based Metal–Organic Frameworks for Highly Enantioselective Cyclization Reactions. J. Am. Chem. Soc. 2015, 137, 12241–12248. [Google Scholar] [CrossRef] [PubMed]
- Sawano, T.; Ji, P.; McIsaac, A.R.; Lin, Z.; Abney, C.W.; Lin, W. The first chiral diene-based metal–organic frameworks for highly enantioselective carbon-carbon bond formation reactions. Chem. Sci. 2015, 6, 7163–7168. [Google Scholar] [CrossRef]
- Lee, M.; Shin, S.M.; Jeong, N.; Thallapally, P.K. Chiral environment of catalytic sites in the chiral metal–organic frameworks. Dalton Trans. 2015, 44, 9349–9352. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, K.; Nagase, S.; Anami, T.; Wierzbicki, M.; Urbanczyk-Lipkowska, Z. Enantioselective Diels-Alder reaction in the confined space of homochiral metal–organic frameworks. RSC Adv. 2016, 6, 111436–111439. [Google Scholar] [CrossRef]
- Xia, Q.; Liu, Y.; Li, Z.; Gong, W.; Cui, Y. A Cr(salen)-based metal–organic framework as a versatile catalyst for efficient asymmetric transformations. Chem. Commun. 2016, 52, 13167–13170. [Google Scholar] [CrossRef] [PubMed]
- Mo, K.; Yang, Y.; Cui, Y. A Homochiral Metal–Organic Framework as an Effective Asymmetric Catalyst for Cyanohydrin Synthesis. J. Am. Chem. Soc. 2014, 136, 1746–1749. [Google Scholar] [CrossRef] [PubMed]
- Zhu, C.; Xia, Q.; Chen, X.; Liu, Y.; Du, X.; Cui, Y. Chiral Metal–Organic Framework as a Platform for Cooperative Catalysis in Asymmetric Cyanosilylation of Aldehydes. ACS Catal. 2016, 6, 7590–7596. [Google Scholar] [CrossRef]
- Li, J.; Ren, Y.; Qi, C.; Jiang, H. The first porphyrin-salen based chiral metal–organic framework for asymmetric cyanosilylation of aldehydes. Chem. Commun. 2017, 53, 8223–8226. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Liu, Y.; Xia, Q.; Cui, Y. Chiral binary metal–organic frameworks for asymmetric sequential reactions. Chem. Commun. 2017, 53, 12313–12316. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Chen, X.; Zhang, Z.; Bao, Z.; Xing, H.; Yang, Q.; Ren, Q. MIL-101(Cr) as a synergistic catalyst for the reduction of imines with trichlorosilane. Mol. Catal. 2018, 445, 163–169. [Google Scholar] [CrossRef]
- Ran, R.; You, L.; Di, B.; Hao, W.; Su, M.; Yan, F.; Huang, L. A novel chiral mesoporous binaphthyl-silicas: Preparation, characterization, and application in HPLC. J. Sep. Sci. 2012, 35, 1854–1862. [Google Scholar] [CrossRef] [PubMed]
- Nuzhdin, A.L.; Dybtsev, D.N.; Bryliakov, K.P.; Talsi, E.P.; Fedin, V.P. Enantioselective Chromatographic Resolution and One-Pot Synthesis of Enantiomerically Pure Sulfoxides over a Homochiral Zn-Organic Framework. J. Am. Chem. Soc. 2007, 129, 12958–12959. [Google Scholar] [CrossRef] [PubMed]
- Peng, Y.; Gong, T.; Zhang, K.; Lin, X.; Liu, Y.; Jiang, J.; Cui, Y. Engineering chiral porous metal–organic frameworks for enantioselective adsorption and separation. Nat. Commun. 2014, 5, 4406. [Google Scholar] [CrossRef] [PubMed]
- Kuang, X.; Ma, Y.; Su, H.; Zhang, J.; Dong, Y.-B.; Tang, B. High-Performance Liquid Chromatographic Enantioseparation of Racemic Drugs Based on Homochiral Metal–Organic Framework. Anal. Chem. 2014, 86, 1277–1281. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Zhang, J.-H.; Zhang, Y.; Wang, B.-J.; Xie, S.-M.; Yuan, L.-M. Chromatographic study on the high performance separation ability of a homochiral [Cu2(d-Cam)2(4,4′-bpy)]n based-column by using racemates and positional isomers as test probes. J. Chromatogr. A 2014, 1325, 163–170. [Google Scholar] [CrossRef] [PubMed]
- Kutzscher, C.; Hoffmann, H.C.; Krause, S.; Stoeck, U.; Senkovska, I.; Brunner, E.; Kaskel, S. Proline Functionalization of the Mesoporous Metal–Organic Framework DUT-32. Inorg. Chem. 2015, 54, 1003–1009. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, K.; Muraoka, T.; Otubo, Y.; Takahashi, H.; Ohnishi, A. HPLC enantioseparation on a homochiral MOF-silica composite as a novel chiral stationary phase. RSC Adv. 2016, 6, 21293–21301. [Google Scholar] [CrossRef]
- Zhang, M.; Chen, X.; Zhang, J.; Kong, J.; Yuan, L. A 3D Homochiral MOF [Cd2(d-cam)3]·2Hdma·4dma for HPLC Chromatographic Enantioseparation. Chirality 2016, 28, 340–346. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.-H.; Nong, R.-Y.; Xie, S.-M.; Wang, B.-J.; Ai, P.; Yuan, L.-M. Homochiral metal–organic frameworks based on amino acid ligands for HPLC separation of enantiomers. Electrophoresis 2017, 38, 2513–2520. [Google Scholar] [CrossRef] [PubMed]
- Hartlieb, K.J.; Holcroft, J.M.; Moghadam, P.Z.; Vermeulen, N.A.; Algaradah, M.M.; Nassar, M.S.; Botros, Y.Y.; Snurr, R.Q.; Stoddart, J.F. CD-MOF: A Versatile Separation Medium. J. Am. Chem. Soc. 2016, 138, 2292–2301. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Li, Z.; Gong, W.; Han, X.; Liu, Y.; Cui, Y. Chiral DHIP-Based Metal–Organic Frameworks for Enantioselective Recognition and Separation. Inorg. Chem. 2016, 55, 7229–7232. [Google Scholar] [CrossRef] [PubMed]
- Hailili, R.; Wang, L.; Qv, J.; Yao, R.; Zhang, X.-M.; Liu, H. Planar Mn4O Cluster Homochiral Metal–Organic Framework for HPLC Separation of Pharmaceutically Important (±)-Ibuprofen Racemate. Inorg. Chem. 2015, 54, 3713–3715. [Google Scholar] [CrossRef] [PubMed]
- Navarro-Sánchez, J.; Argente-García, A.I.; Moliner-Martínez, Y.; Roca-Sanjuán, D.; Antypov, D.; Campíns-Falcó, P.; Rosseinsky, M.J.; Martí-Gastaldo, C. Peptide Metal–Organic Frameworks for Enantioselective Separation of Chiral Drugs. J. Am. Chem. Soc. 2017, 139, 4294–4297. [Google Scholar] [CrossRef] [PubMed]
- Xie, S.-M.; Zhang, Z.-J.; Wang, Z.-Y.; Yuan, L.-M. Chiral Metal–Organic Frameworks for High-Resolution Gas Chromatographic Separations. J. Am. Chem. Soc. 2011, 133, 11892–11895. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Xie, S.-M.; Ai, P.; Zhang, J.-H.; Zhang, M.; Yuan, L.-M. Metal–Organic Framework Co(D-Cam)1/2(bdc)1/2(tmdpy) for Improved Enantioseparations on a Chiral Cyclodextrin Stationary Phase in Gas Chromatography. ChemPlusChem 2014, 79, 1103–1108. [Google Scholar] [CrossRef]
- Yang, J.-R.; Xie, S.-M.; Liu, H.; Zhang, J.-H.; Yuan, L.-M. Metal–Organic Framework InH(d-C10H14O4)2 for Improved Enantioseparations on a Chiral Cyclodextrin Stationary Phase in GC. Chromatographia 2015, 78, 557–564. [Google Scholar] [CrossRef]
- Xue, X.; Zhang, M.; Xie, S.; Yuan, L. Homochiral metal–organic framework [Zn2(d-Cam)2(4,4′-bpy)]n for high-resolution gas chromatographic separations. Acta Chromatogr. 2015, 27, 15–26. [Google Scholar] [CrossRef]
- Yang, J.-R.; Xie, S.-M.; Zhang, J.-H.; Chen, L.; Nong, R.-Y.; Yuan, L.-M. Metal–Organic Framework [Cd(LTP)2]n for Improved Enantioseparations on a Chiral Cyclodextrin Stationary Phase in GC. J. Chromatogr. Sci. 2016, 54, 1467–1474. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Wang, L.; Yao, R.-X.; Zhang, X.-M. Fourfold-Interpenetrated MOF [Ni(pybz)2] as Coating Material in Gas Chromatographic Capillary Column for Separation. Inorg. Chem. 2017, 56, 8912–8919. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Wang, F.; Liu, L.-Y.; Zhang, J. Interpenetrated Three-Dimensional Copper–Iodine Cluster-Based Framework with Enantiopure Porphyrin-like Templates. Inorg. Chem. 2016, 55, 1358–1360. [Google Scholar] [CrossRef] [PubMed]
- Lang, L.I.; Shengming, X.I.E.; Junhui, Z.; Ling, C.; Pengjing, Z.; Liming, Y. A Gas Chromatographic Stationary of Homochiral Metal-peptide Framework Material and Its Applications. Chem. Res. Chin. Univ. 2017, 33, 24–30. [Google Scholar]
- Zhang, J.; Han, X.; Wu, X.; Liu, Y.; Cui, Y. Multivariate Chiral Covalent Organic Frameworks with Controlled Crystallinity and Stability for Asymmetric Catalysis. J. Am. Chem. Soc. 2017, 139, 8277–8285. [Google Scholar] [CrossRef] [PubMed]
- Han, X.; Xia, Q.; Huang, J.; Liu, Y.; Tan, C.; Cui, Y. Chiral Covalent Organic Frameworks with High Chemical Stability for Heterogeneous Asymmetric Catalysis. J. Am. Chem. Soc. 2017, 139, 8693–8697. [Google Scholar] [CrossRef] [PubMed]
- Lin, Z.-J.; Lü, J.; Li, L.; Li, H.-F.; Cao, R. Defect porous organic frameworks (dPOFs) as a platform for chiral organocatalysis. J. Catal. 2017, 355, 131–138. [Google Scholar] [CrossRef]


© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bhattacharjee, S.; Khan, M.I.; Li, X.; Zhu, Q.-L.; Wu, X.-T. Recent Progress in Asymmetric Catalysis and Chromatographic Separation by Chiral Metal–Organic Frameworks. Catalysts 2018, 8, 120. https://doi.org/10.3390/catal8030120
Bhattacharjee S, Khan MI, Li X, Zhu Q-L, Wu X-T. Recent Progress in Asymmetric Catalysis and Chromatographic Separation by Chiral Metal–Organic Frameworks. Catalysts. 2018; 8(3):120. https://doi.org/10.3390/catal8030120
Chicago/Turabian StyleBhattacharjee, Suchandra, Muhammad Imran Khan, Xiaofang Li, Qi-Long Zhu, and Xin-Tao Wu. 2018. "Recent Progress in Asymmetric Catalysis and Chromatographic Separation by Chiral Metal–Organic Frameworks" Catalysts 8, no. 3: 120. https://doi.org/10.3390/catal8030120
APA StyleBhattacharjee, S., Khan, M. I., Li, X., Zhu, Q.-L., & Wu, X.-T. (2018). Recent Progress in Asymmetric Catalysis and Chromatographic Separation by Chiral Metal–Organic Frameworks. Catalysts, 8(3), 120. https://doi.org/10.3390/catal8030120




