Influence of an Electronic Structure of N-TiO2 on Its Photocatalytic Activity towards Decomposition of Acetaldehyde under UV and Fluorescent Lamps Irradiation
Abstract
:1. Introduction
2. Results
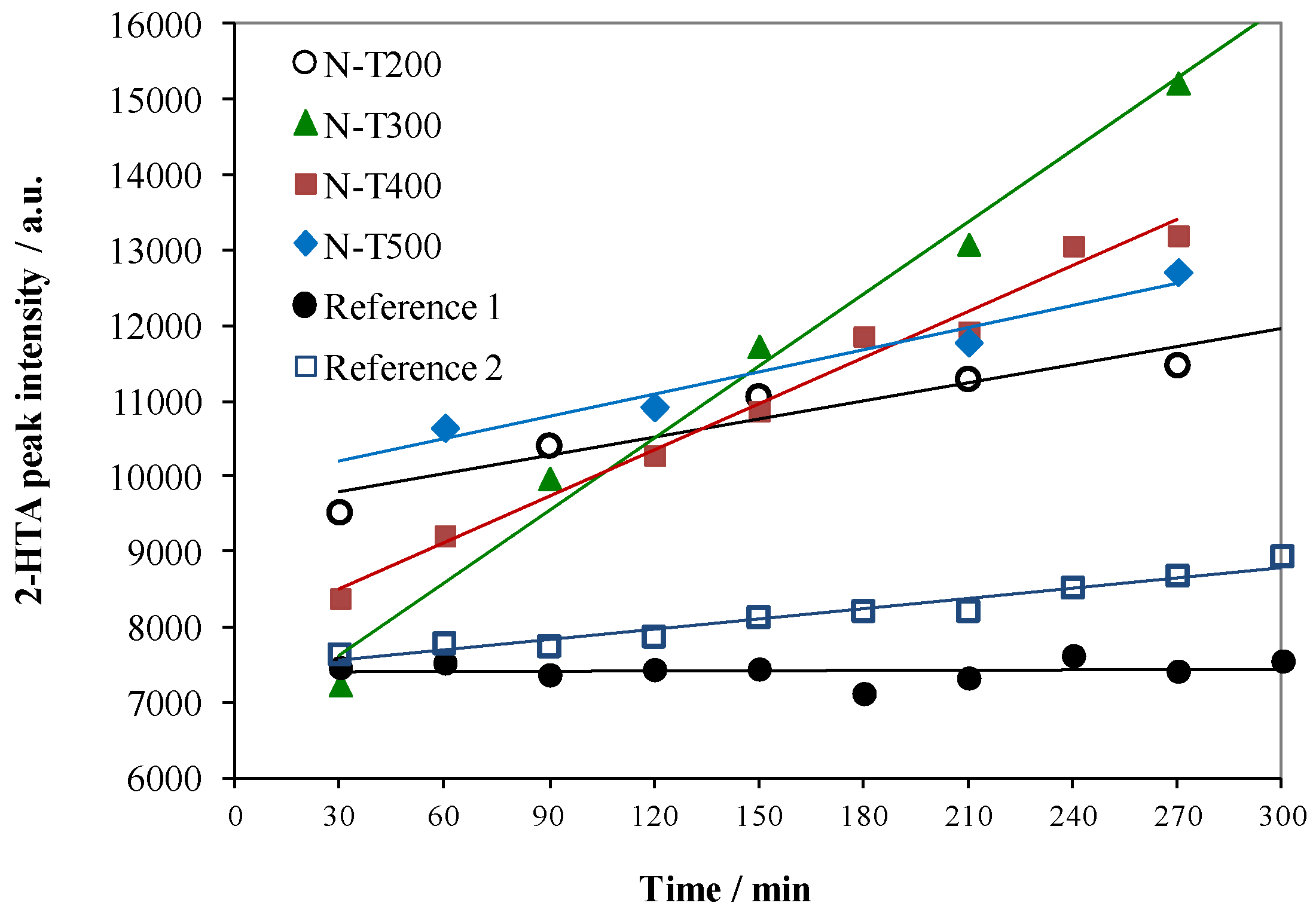
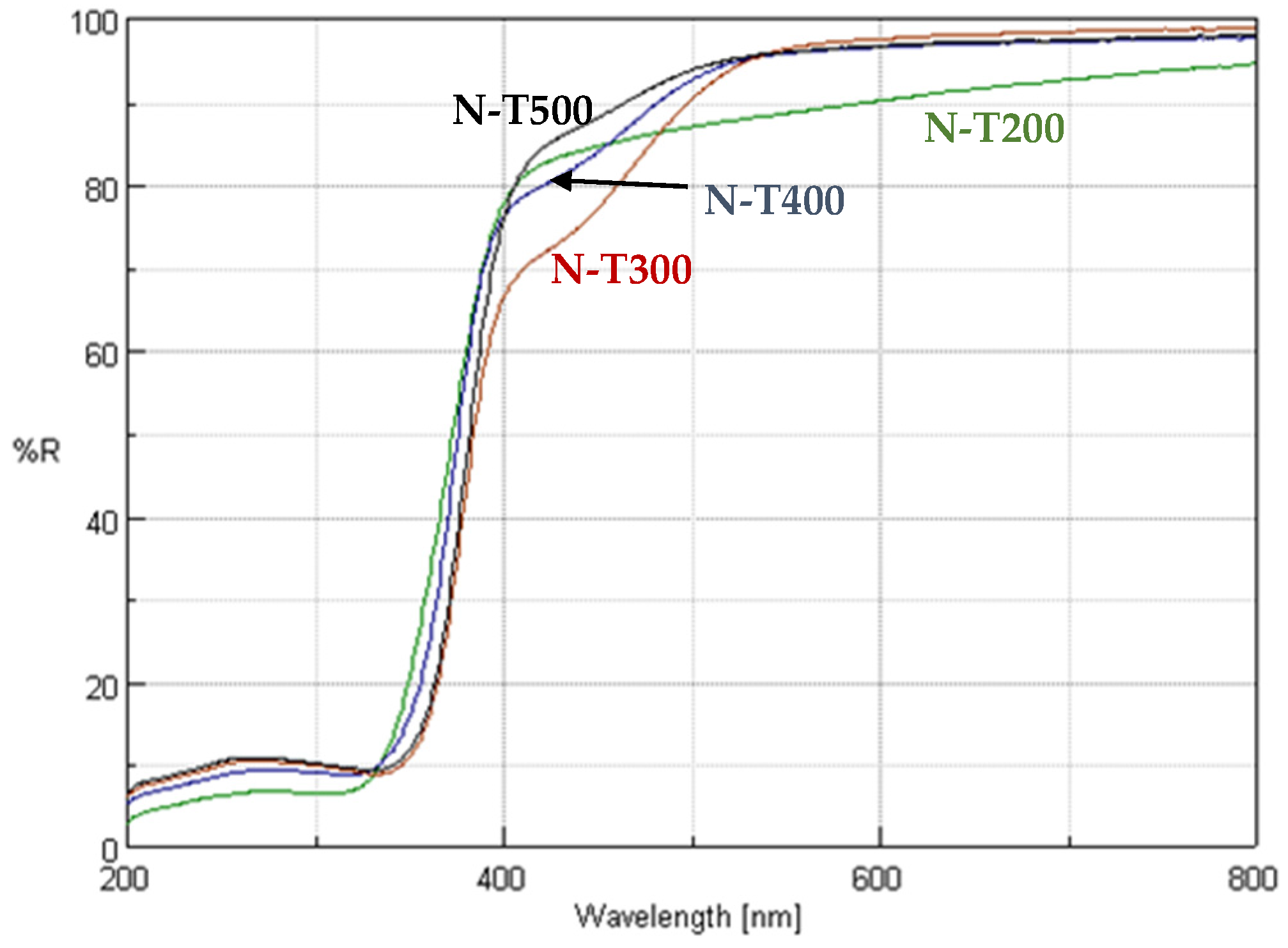
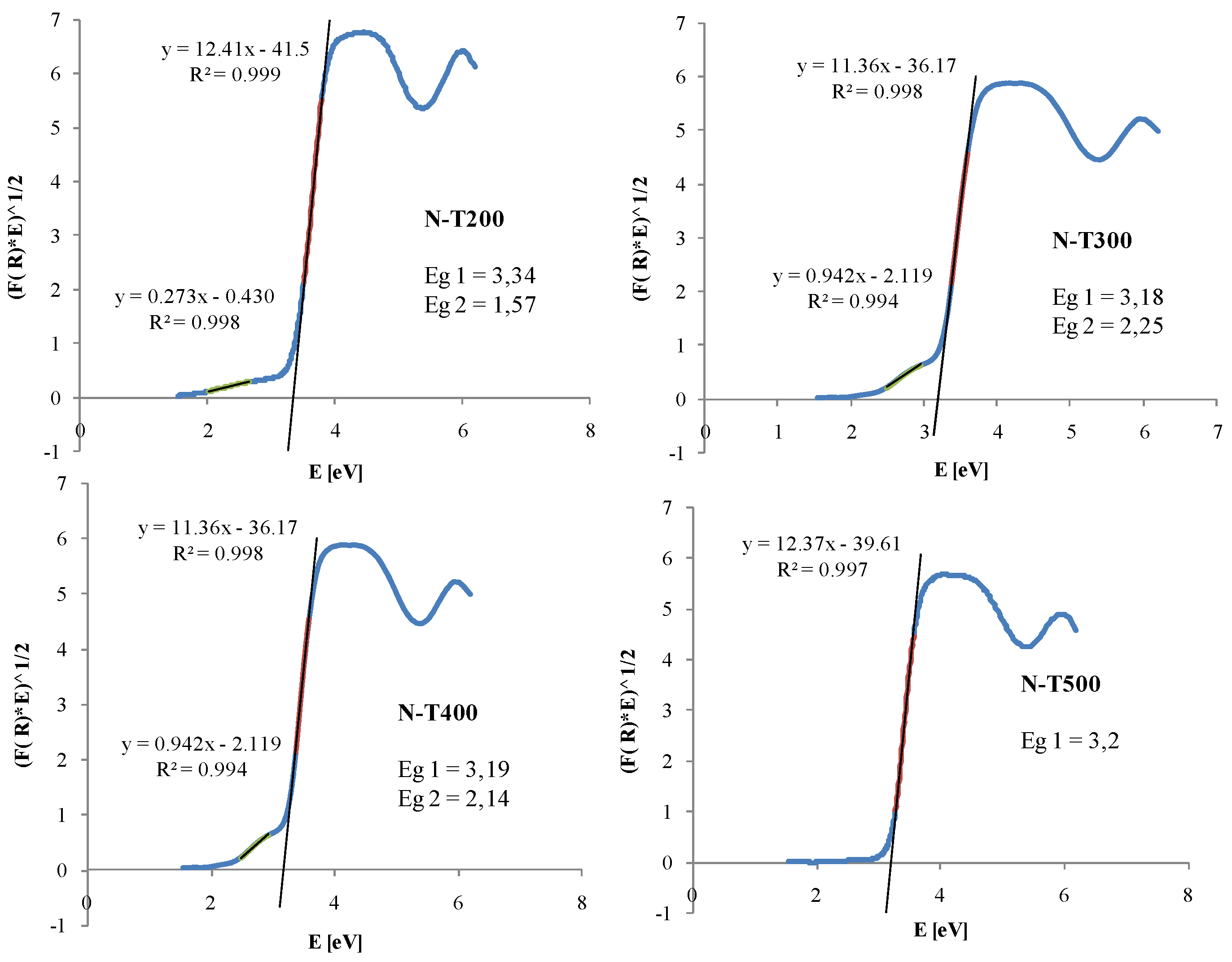
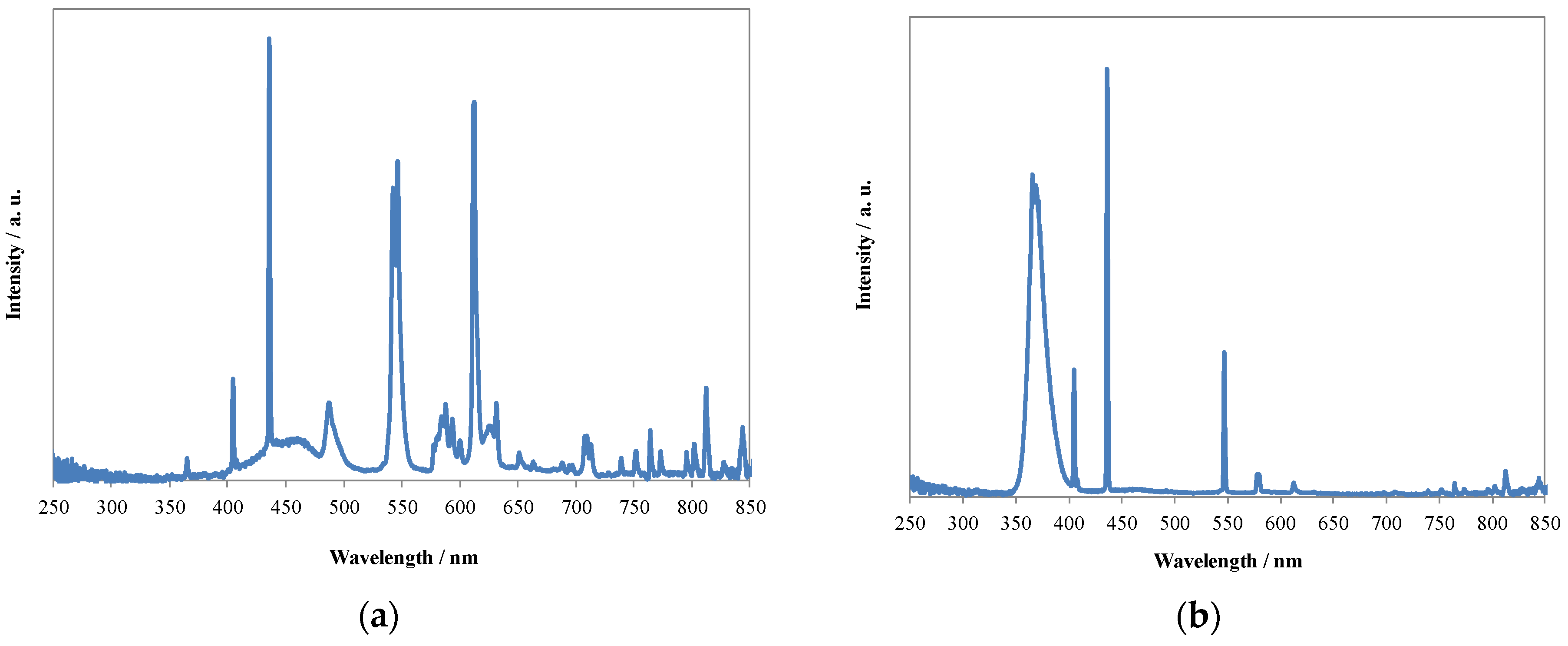
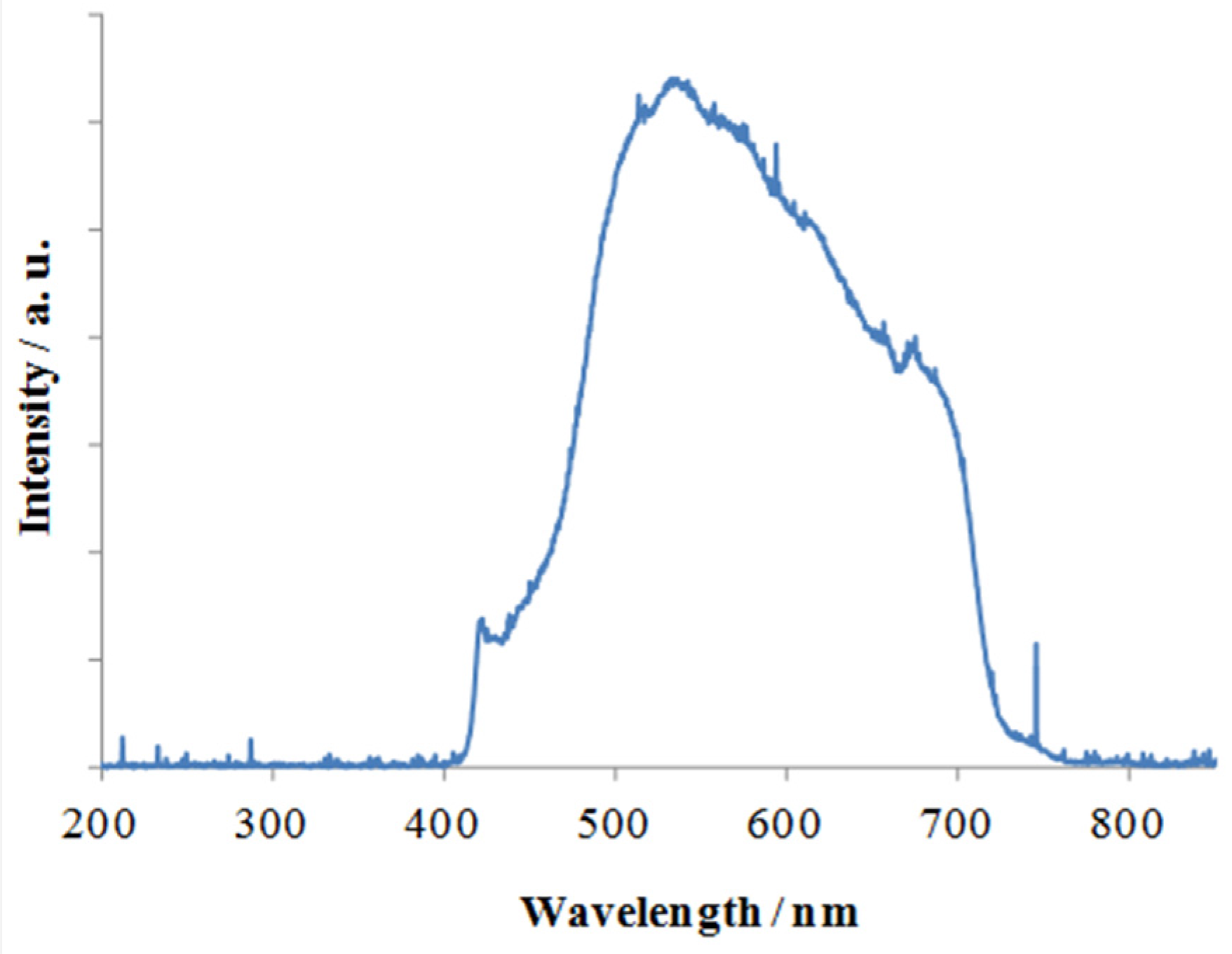
2.1. Visible Light Activity of N-Doped TiO2
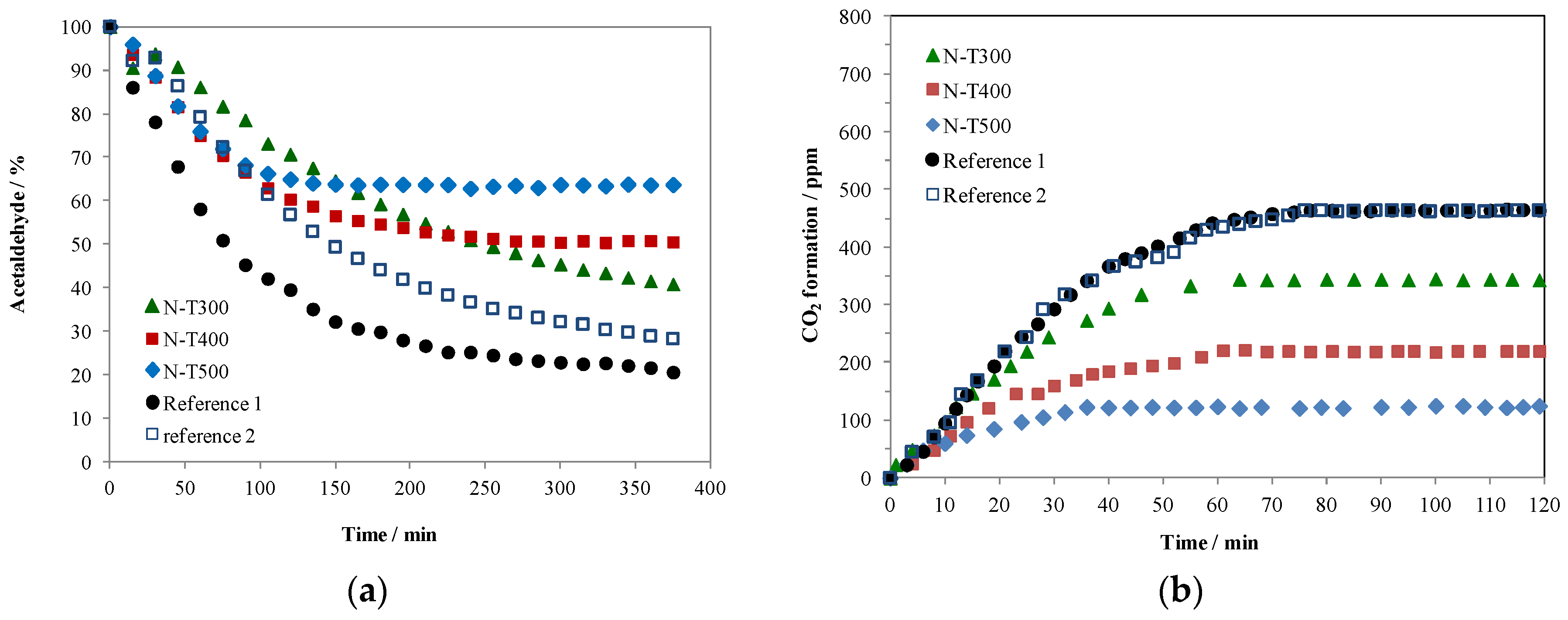
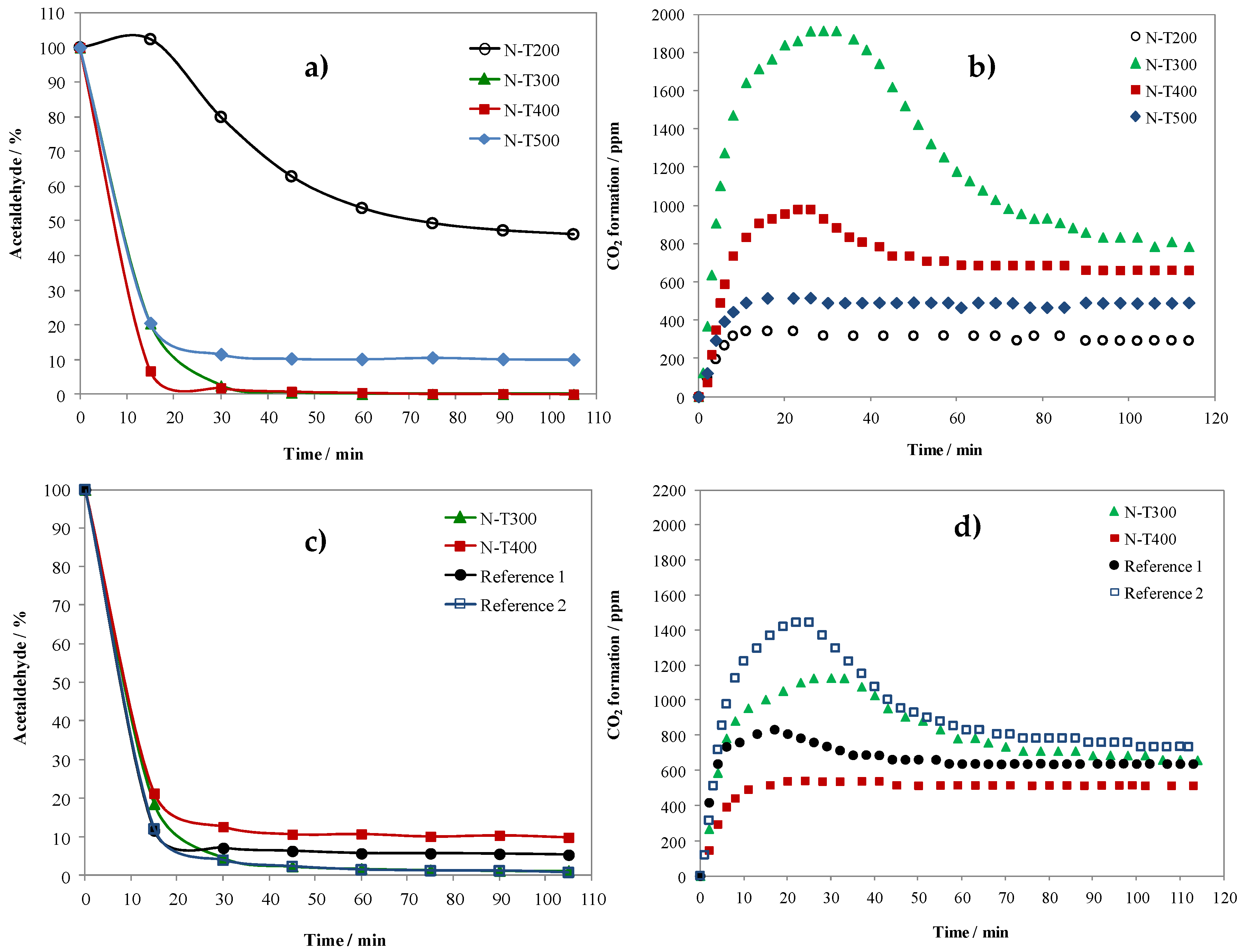
2.2. Photocatalytic Decomposition of Acetaldehyde on N-TiO2 Samples
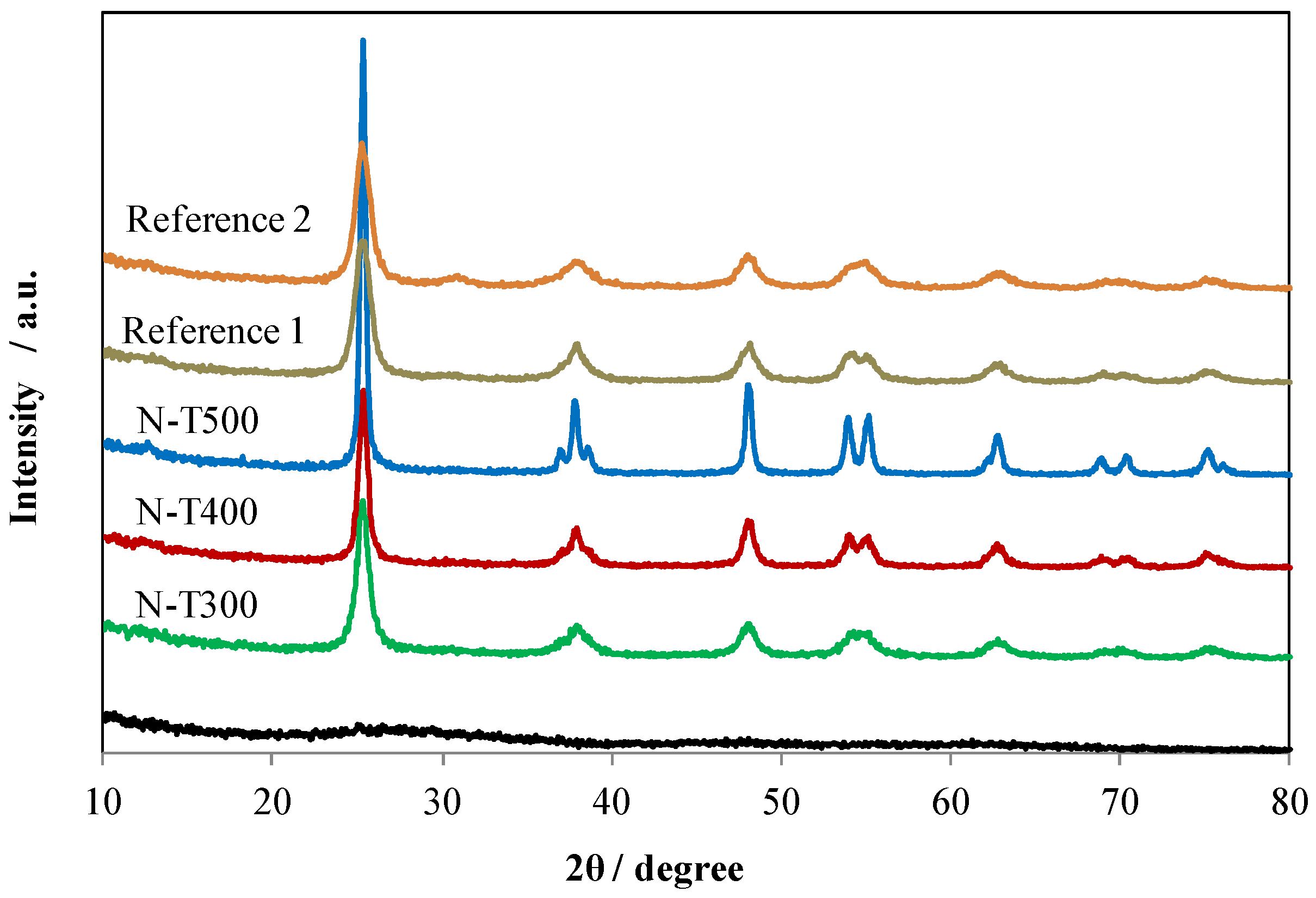
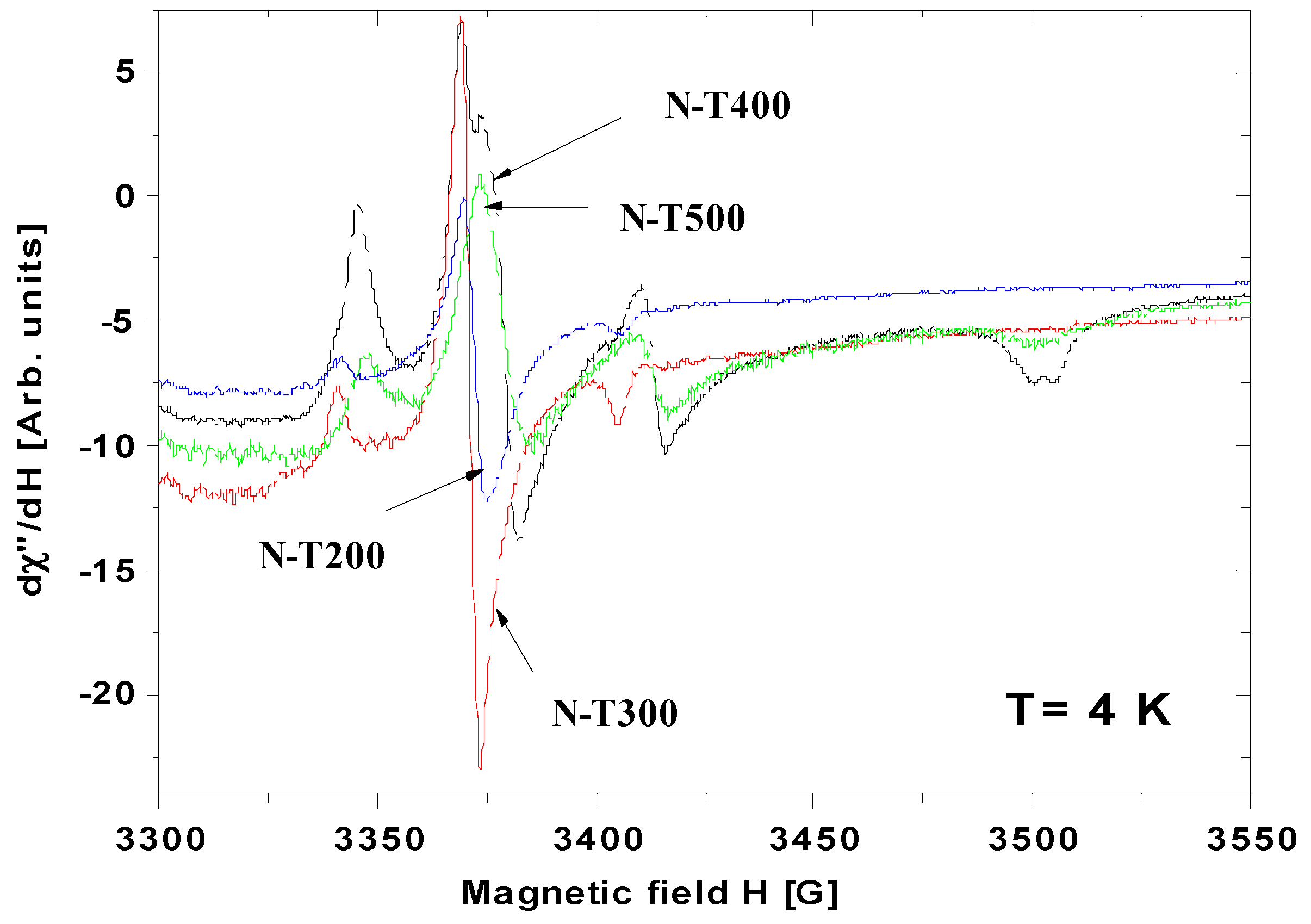
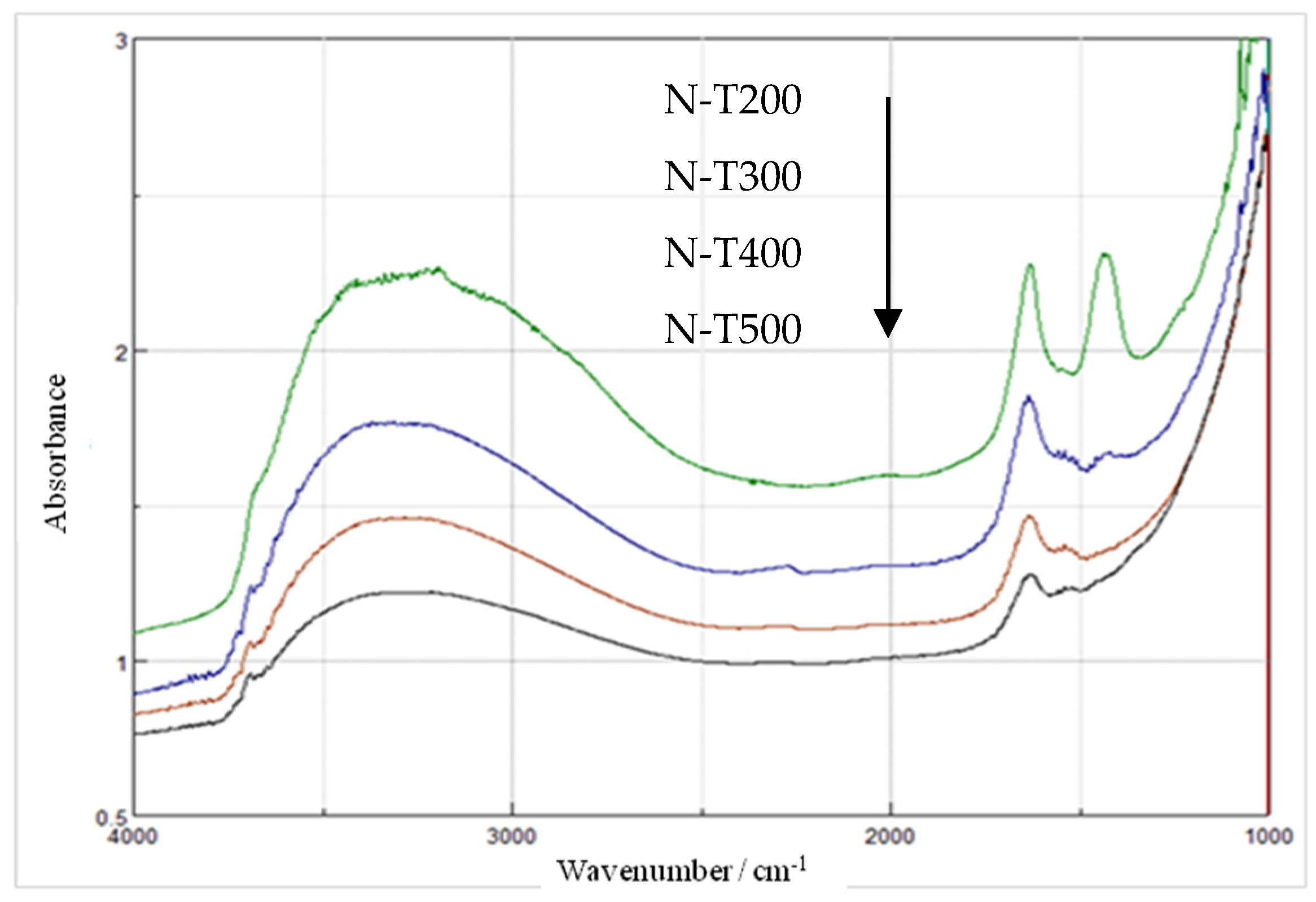
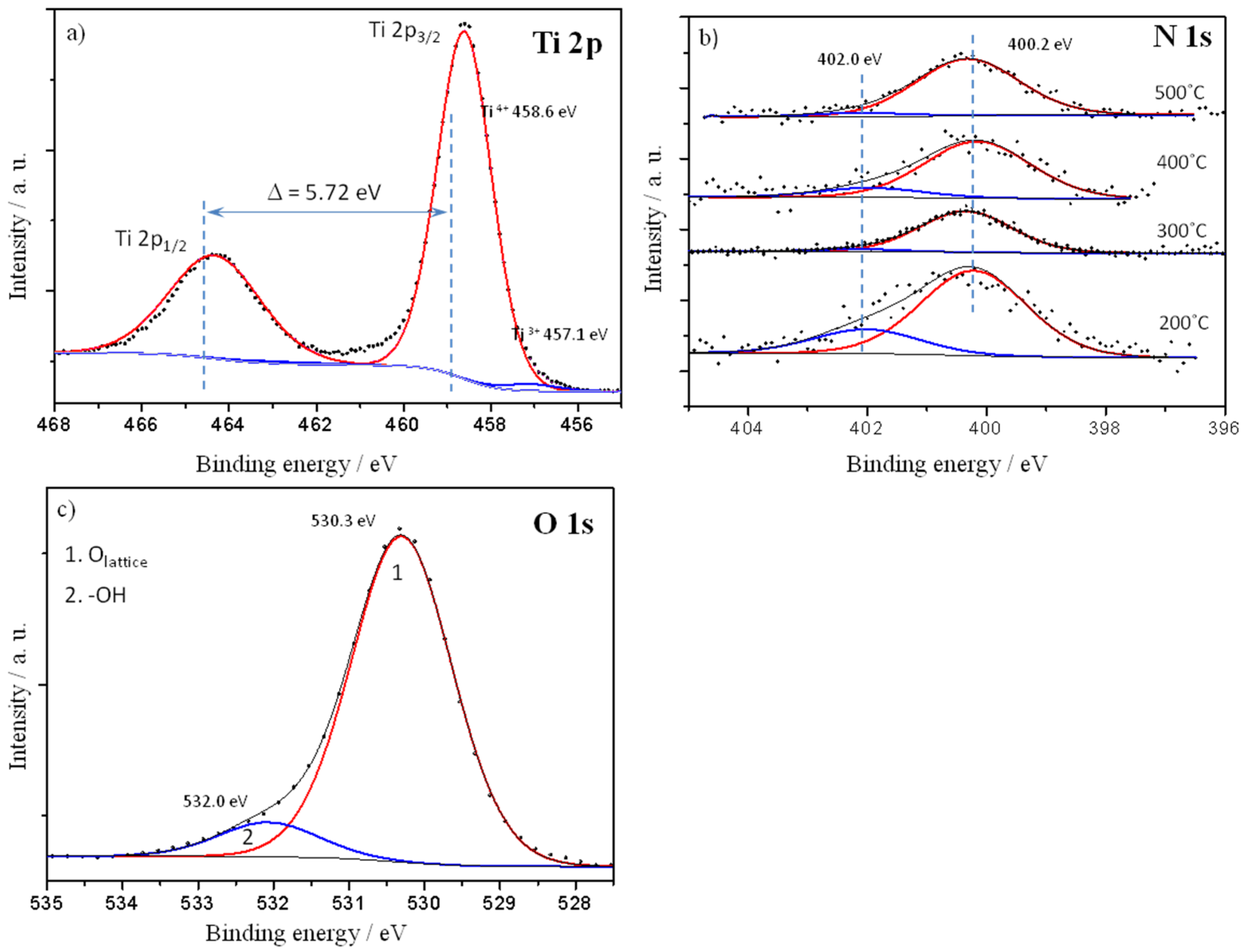
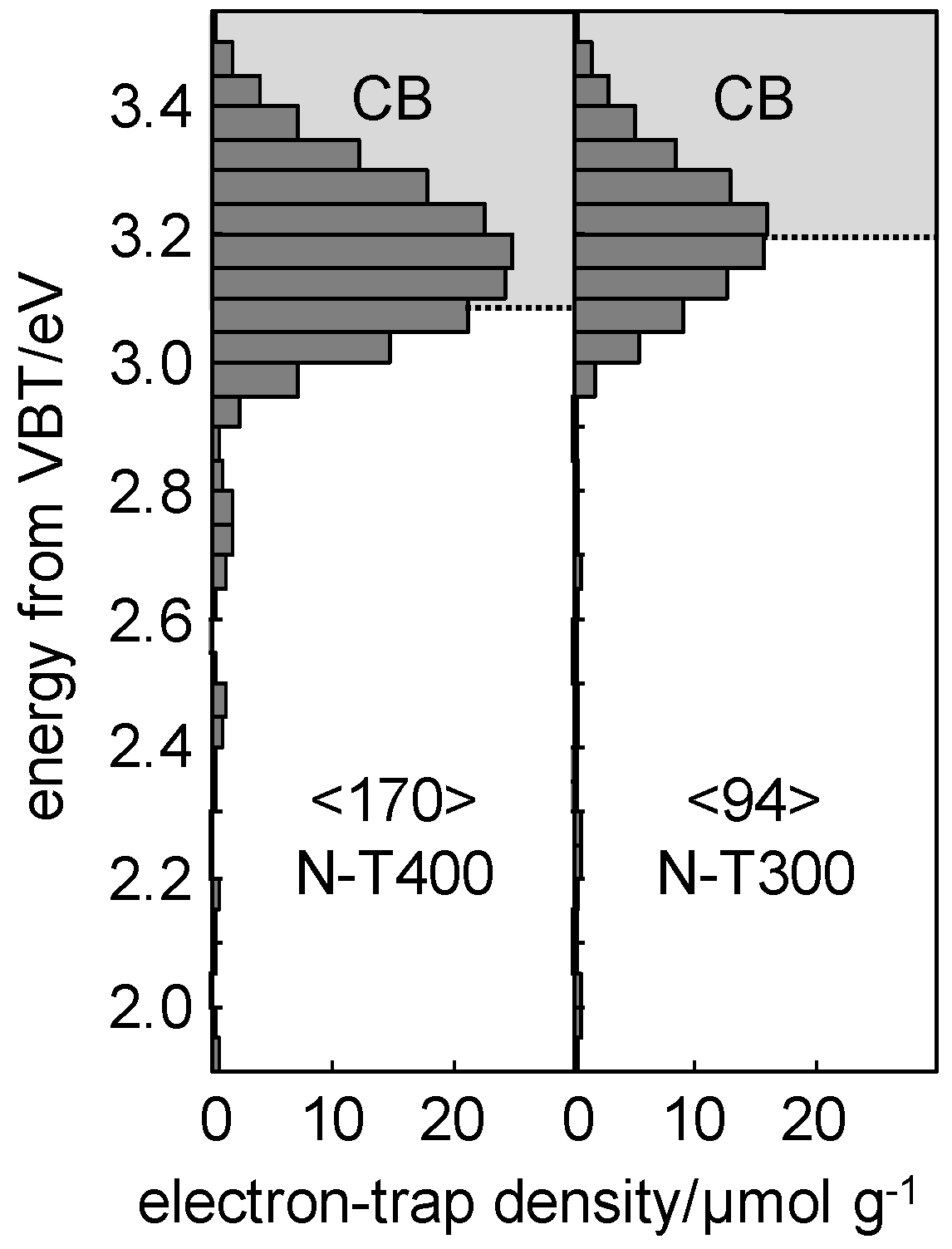
2.3. Characteristics of N-TiO2 Photocatalysts
3. Discussion
4. Materials and Methods
4.1. Preparation of Nitrogen Modified TiO2 Photocatalyst
4.2. Material Characteristics
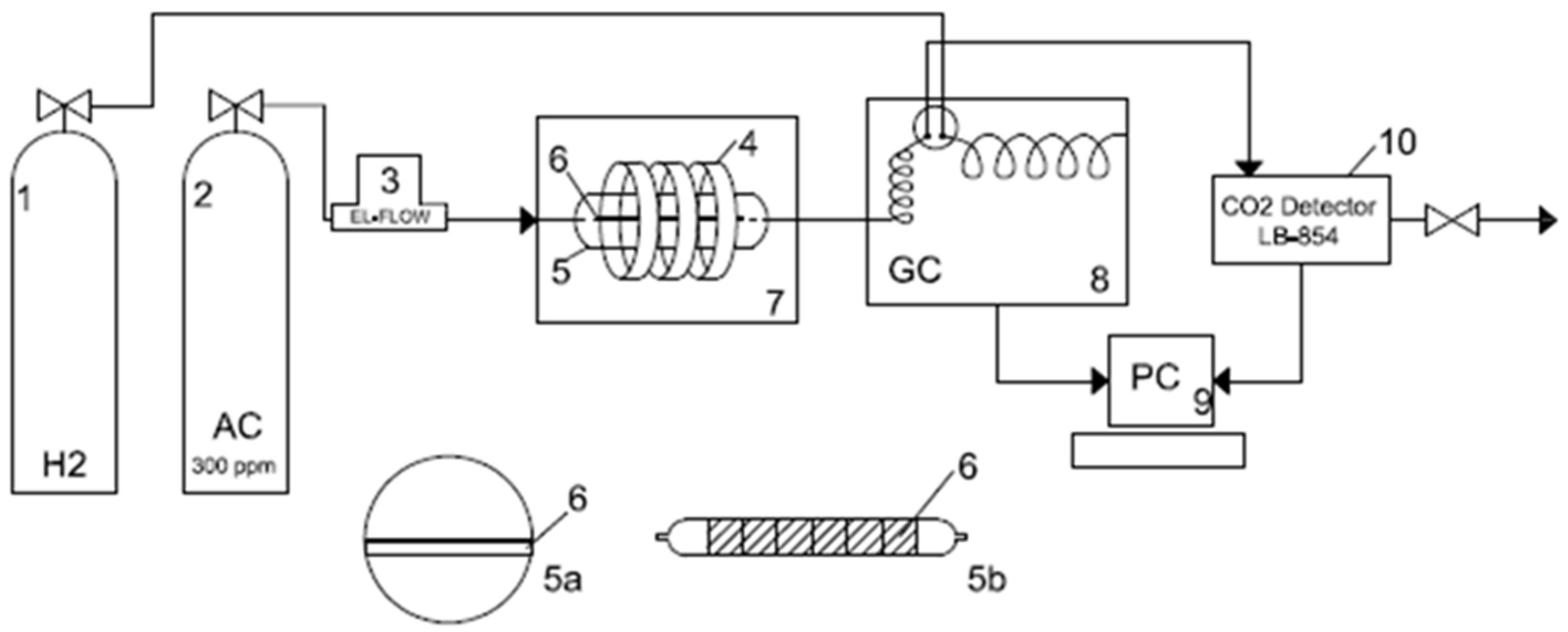
4.3. Photocatalytic Decomposition of Acetaldehyde
4.4. Photocatalytic Decomposition of Acetaldehyde
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Fujishima, A.; Zhang, X.; Tryk, A. TiO2 photocatalysis and related surface phenomena. Surf. Sci. Rep. 2008, 63, 515–582. [Google Scholar] [CrossRef]
- Meroni, D.; Ardizzone, S.; Cappelletti, G.; Oliva, C.; Ceotto, M.; Poelman, D.; Poelman, H. Photocatalytic removal of ethanol and acetaldehyde by N-promoted TiO2 films: The role of the different nitrogen sources. Catal. Today 2011, 161, 169–174. [Google Scholar] [CrossRef]
- Zhang, J.; Wu, Y.; Xing, M.; Leghari, S.A.K.; Sajjad, S. Development of modified N doped TiO2 photocatalyst with metals, nonmetals and metal oxides. Energy Environ. Sci. 2010, 3, 715–726. [Google Scholar] [CrossRef]
- Lin, Y.-H.; Weng, C.-H.; Tzeng, J.-H.; Lin, Y.-T. Adsorption and photocatalytic kinetics of visible-light response N-doped TiO2 nanocatalyst for indoor acetaldehyde removal under dark and light conditions. Intern. J. Photoen. 2016, 2016, 1–9. [Google Scholar] [CrossRef]
- Kitano, M.; Funatsu, K.; Matsuoka, M.; Ueshima, M.; Anpo, M. Preparation of nitrogen-substituted TiO2 thin film photocatalysts by the radio frequency magnetron sputtering deposition method and their photocatalytic reactivity under visible light irradiation. J. Phys. Chem. B 2006, 110, 25266–25272. [Google Scholar] [CrossRef] [PubMed]
- Dunnill, C.W.; Parkin, I.P. Nitrogen-doped TiO2 thin films: Photocatalytic applications for healthcare environments. Dalton Trans. 2011, 40, 1635–1640. [Google Scholar] [CrossRef] [PubMed]
- Miyauchi, M.; Ikezawa, A.; Tobimatsu, H.; Irie, H.; Hashimoto, K. Zeta potential and photocatalytic activity of nitrogen doped TiO2 thin films. Phys. Chem. Chem. Phys. 2004, 6, 865–870. [Google Scholar] [CrossRef]
- Etacheri, V.; Di Valentin, C.; Schneider, J.; Bahnemann, D.; Pillai, S.C. Visible-light activation of TiO2 photocatalysts: Advances in theory and experiments. J. Photochem. Photobiol. C 2015, 25, 1–29. [Google Scholar] [CrossRef]
- Kubacka, A.; Fernandez-García, M.; Colon, G. Advanced nanoarchitectures for solar photocatalytic applications. Chem. Rev. 2012, 112, 1555–1614. [Google Scholar] [CrossRef] [PubMed]
- Asahi, R.; Morikawa, T.; Ohwaki, T.; Aoki, K.; Taga, Y. Visible-light photocatalysis in nitrogen-doped titanium oxides. Science 2001, 293, 269–271. [Google Scholar] [CrossRef] [PubMed]
- Irie, H.; Watanabe, Y.; Hashimoto, K. Nitrogen-concentration dependence on photocatalytic activity of TiO2−xNx powders. J. Phys. Chem. B 2003, 107, 5483–5486. [Google Scholar] [CrossRef]
- Diwald, O.; Thompson, T.L.; Goralski, E.G.; Walck, S.D.; Yates, J.T. The effect of nitrogen ion implantation on the photoactivity of TiO2 rutile single crystals. J. Phys. Chem. B 2004, 108, 52–57. [Google Scholar] [CrossRef]
- Valentin, C.D.; Pacchioni, G.; Selloni, A.; Livraghi, S.; Giamello, E. Characterization of paramagnetic species in N-doped TiO2 powders by EPR Spectroscopy and DFT calculations. J. Phys. Chem. B 2005, 109, 11414–11419. [Google Scholar] [CrossRef] [PubMed]
- Ihara, T.; Miyoshi, M.; Iriyama, Y.; Matsumoto, O.; Sugihara, S. Visible-light-active titanium oxide photocatalyst realized by an oxygen-deficient structure and by nitrogen doping. Appl. Catal. B 2003, 42, 403–409. [Google Scholar] [CrossRef]
- Sakatani, Y.; Nunoshige, J.; Ando, H.; Okusako, K.; Koike, H.; Takata, T.; Kondo, J.N.; Hara, M.; Domen, K. Photocatalytic decomposition of acetaldehyde under visible light irradiation over La3+ and N co-doped TiO2. Chem. Lett. 2003, 32, 1156–1157. [Google Scholar] [CrossRef]
- Nitta, A.; Takase, M.; Takashima, M.; Murakami, N.; Ohtani, B. A fingerprint of metal-oxide powders: Energy-resolved distribution of electron traps. Chem. Commun. 2016, 52, 12096–12099. [Google Scholar] [CrossRef] [PubMed]
- Colbeau-Justin, C.; Kunst, M.; Huguenin, D. Structural influence on charge-carrier lifetimes in TiO2 powders studied by microwave absorption. J. Mater. Sci. 2003, 38, 2429–2437. [Google Scholar] [CrossRef]
- Luna, A.L.; Novoseltceva, E.; Louarn, E.; Beaunier, P.; Kowalska, E.; Ohtani, B.; Valenzuela, M.A.; Remita, H.; Colbeau-Justin, C. Synergetic effect of Ni and Au nanoparticles synthesized on titania particles for efficient photocatalytic hydrogen production. Appl. Catal. B 2016, 191, 18–28. [Google Scholar] [CrossRef]
- Chesalov, Y.A.; Chernobay, G.B.; Andrushkevich, T.V. FTIR study of the surface complexes of β-picoline, 3-pyridine-carbaldehyde and nicotinic acid on sulfated TiO2 (anatase). J. Mol. Catal. A 2013, 373, 96–107. [Google Scholar] [CrossRef]
- Yang, G.; Jiang, Z.; Shi, H.; Xiao, T.; Yan, Z. Preparation of highly visible-light active N-doped TiO2 photocatalyst. J. Mater. Chem. 2010, 20, 5301–5309. [Google Scholar] [CrossRef]
- Lopez, R.; Gomez, R. Band-gap energy estimation from diffuse reflectance measurements on sol–gel and commercial TiO2: A comparative study. J. Sol-Gel Sci. Technol. 2012, 61, 1–7. [Google Scholar] [CrossRef]
- Biesinger, M.C.; Lau, L.W.M.; Gerson, A.R.; Smart, R.St.C. Resolving surface chemical states in XPS analysis of first row transition metals, oxides and hydroxides: Sc, Ti, V, Cu and Zn. Appl. Surf. Sci. 2010, 257, 887–898. [Google Scholar] [CrossRef]
- Ketteler, G.; Yamamoto, S.; Bluhm, H.; Andersson, K.; Starr, D.E.; Ogletree, D.F.; Ogasawara, H.; Nilsson, A.; Salmeron, M. The nature of water nucleation sites on TiO2 (110) surfaces revealed by ambient pressure X-ray photoelectron spectroscopy. J. Phys. Chem. C 2007, 111, 8278–8282. [Google Scholar] [CrossRef]
- Diesen, V.; Dunnill, C.W.; Bear, J.C.; Firth, S.; Jonsson, M.; Parkin, I.P. Visible light photocatalytic activity in AACVD-prepared N-modified TiO2 thin films. Chem. Vap. Deposition 2014, 20, 91–97. [Google Scholar] [CrossRef]
- Quesada-Cabrera, R.; Sotelo Vazquez, C.; Darr, J.A.; Parkin, I.P. Critical influence of surface nitrogen species on the activity of N-doped TiO2 thin-films during photodegradation of stearic acid under UV light irradiation. Appl. Catal. B 2014, 160–161, 582–588. [Google Scholar] [CrossRef]
- Nolan, N.T.; Synnott, D.W.; Seery, M.K.; Hinder, S.J.; Van Wassenhovend, A.; Pillai, S.C. Effect of N-doping on the photocatalytic activity of sol-gel TiO2. J. Hazard. Mater. 2012, 211–212, 88–94. [Google Scholar] [CrossRef] [PubMed]
- Cheng, X.; Yu, X.; Xing, Z.; Wan, J. Enhanced photocatalytic activity of nitrogen doped TiO2 anatase nano-particle under simulated sunlight irradiation. Energy Procedia 2012, 16, 598–605. [Google Scholar] [CrossRef]
- Dunnill, C.W.H.; Aiken, Z.A.; Pratten, J.; Wilson, M.; Morgan, D.J.; Parkin, I.P. Enhanced photocatalytic activity under visible light in N-doped TiO2 thin films produced by APCVD preparations using t-butylamine as a nitrogen source and their potential for antibacterial films. J. Photochem. Photobiol. A 2009, 207, 244–253. [Google Scholar] [CrossRef]
- Ohtani, B. Titania photocatalysis beyond recombination: A Critical Review. Catalysts 2013, 3, 942–953. [Google Scholar] [CrossRef]
- Tryba, B.; Tygielska, M.; Colbeau-Justin, C.; Kusiak-Nejman, E.; Kapica-Kozar, J.; Wróbel, R.; Żołnierkiewicz, G.; Guskos, N. Influence of pH of sol-gel solution on phase composition and photocatalytic activity of TiO2 under UV and visible light. Mater. Res. Bull. 2016, 84, 152–161. [Google Scholar] [CrossRef]
| Sample | HTT (°C) | BET (m2/g) | Phase Composition | Mean Crystallites Size (nm) | Potential Zeta (mV) |
|---|---|---|---|---|---|
| N-T200 | 200 | 354 | Amorphous | - | −23.0 |
| N-T300 | 300 | 153 | Anatase | 11.6 | −16.4 |
| N-T400 | 400 | 50 | Anatase | 16.3 | −21.4 |
| N-T500 | 500 | 16 | Anatase | 34.1 | −15.2 |
| Reference 1 | 400 | 140 | Anatase | 8.7 | −17.3 |
| Reference 2 | 400 | 124 | Anatase/Brookite = 0.87/0.13 | A(16.2); B(8.1) | −15.4 |
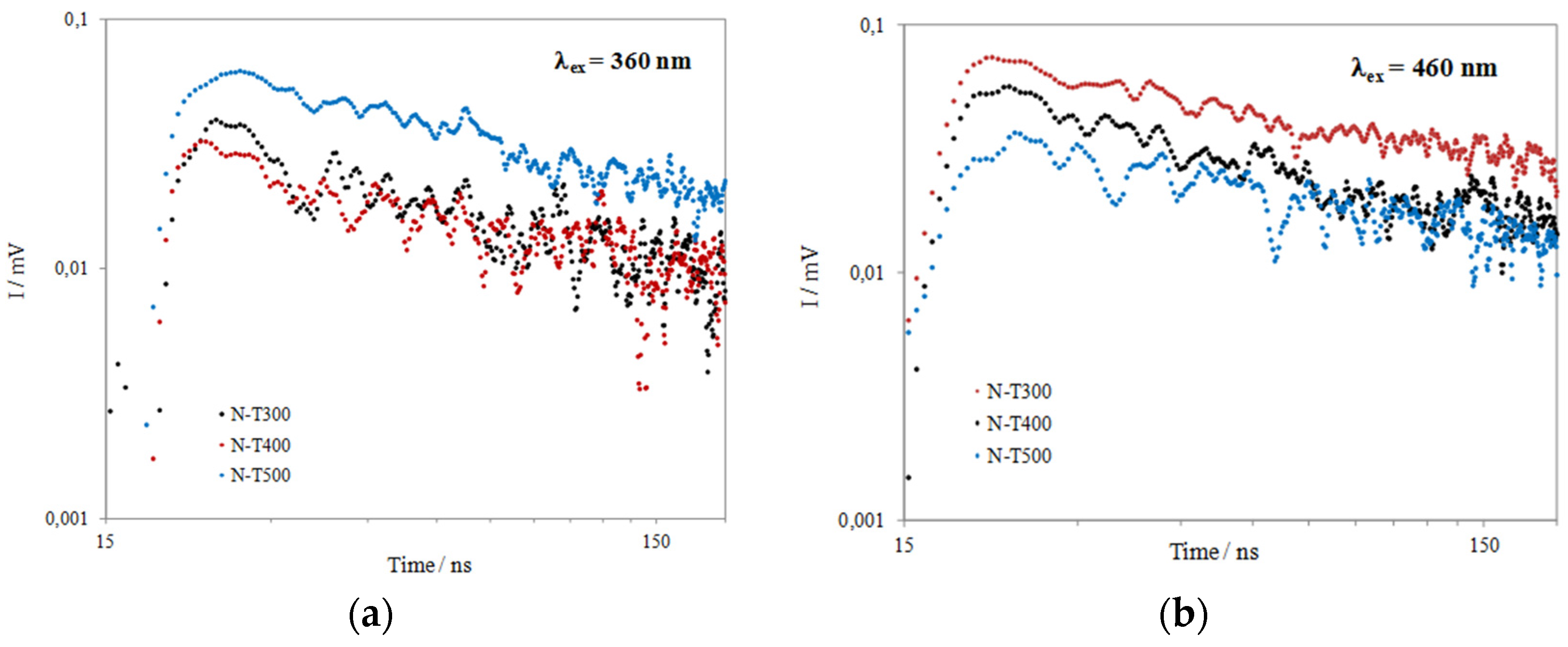
| Sample | λex (nm) | Imax | I40 | I40/Imax |
|---|---|---|---|---|
| N-T300 | 460 | 0.074 | 0.043 | 0.58 |
| N-T400 | 0.056 | 0.031 | 0.55 | |
| N-T500 | 0.039 | 0.026 | 0.67 | |
| N-T300 | 360 | 0.039 | 0.020 | 0.51 |
| N-T400 | 0.032 | 0.013 | 0.41 | |
| N-T500 | 0.062 | 0040 | 0.65 |
| Sample | N (%) | C (%) |
|---|---|---|
| N-T200 | 0.6 | 0.3 |
| N-T300 | 0.1 | 0.08 |
| N-T400 | 0.07 | 0 |
| N-T500 | 0 | 0 |
| Reference 1 | 0 | 0 |
| Reference 2 | 0 | 0 |
| Sample | Ti (at.%) | O (at.%) | N (at.%) |
|---|---|---|---|
| N-T200 | 23.2 | 75.5 | 1.4 |
| N-T300 | 26.1 | 73.5 | 0.4 |
| N-T400 | 25.3 | 74.0 | 0.7 |
| N-T500 | 23.1 | 76.5 | 0.5 |
| Sample | Ti 2p (%) | N 1s (%) | O 1s (%) | |||
|---|---|---|---|---|---|---|
| 4+ | 3+ | 400.2 eV | 402 eV | Surface | Lattice | |
| N-T200 | 98 | 2 | 78 | 22 | 10 | 90 |
| N-T300 | 98 | 2 | 94 | 6 | 10 | 91 |
| N-T400 | 98 | 2 | 86 | 14 | 8 | 92 |
| N-T500 | 99 | 1 | 94 | 6 | 7 | 94 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tryba, B.; Wozniak, M.; Zolnierkiewicz, G.; Guskos, N.; Morawski, A.; Colbeau-Justin, C.; Wrobel, R.; Nitta, A.; Ohtani, B. Influence of an Electronic Structure of N-TiO2 on Its Photocatalytic Activity towards Decomposition of Acetaldehyde under UV and Fluorescent Lamps Irradiation. Catalysts 2018, 8, 85. https://doi.org/10.3390/catal8020085
Tryba B, Wozniak M, Zolnierkiewicz G, Guskos N, Morawski A, Colbeau-Justin C, Wrobel R, Nitta A, Ohtani B. Influence of an Electronic Structure of N-TiO2 on Its Photocatalytic Activity towards Decomposition of Acetaldehyde under UV and Fluorescent Lamps Irradiation. Catalysts. 2018; 8(2):85. https://doi.org/10.3390/catal8020085
Chicago/Turabian StyleTryba, Beata, Magdalena Wozniak, Grzegorz Zolnierkiewicz, Nikos Guskos, Antoni Morawski, Christophe Colbeau-Justin, Rafał Wrobel, Akio Nitta, and Bunsho Ohtani. 2018. "Influence of an Electronic Structure of N-TiO2 on Its Photocatalytic Activity towards Decomposition of Acetaldehyde under UV and Fluorescent Lamps Irradiation" Catalysts 8, no. 2: 85. https://doi.org/10.3390/catal8020085
APA StyleTryba, B., Wozniak, M., Zolnierkiewicz, G., Guskos, N., Morawski, A., Colbeau-Justin, C., Wrobel, R., Nitta, A., & Ohtani, B. (2018). Influence of an Electronic Structure of N-TiO2 on Its Photocatalytic Activity towards Decomposition of Acetaldehyde under UV and Fluorescent Lamps Irradiation. Catalysts, 8(2), 85. https://doi.org/10.3390/catal8020085








