Detoxification of Olive Mill Wastewaters by Fenton’s Process
Abstract
1. Introduction
2. Homogeneous Fenton’s Process
3. Heterogeneous Fenton
4. Photo-Fenton
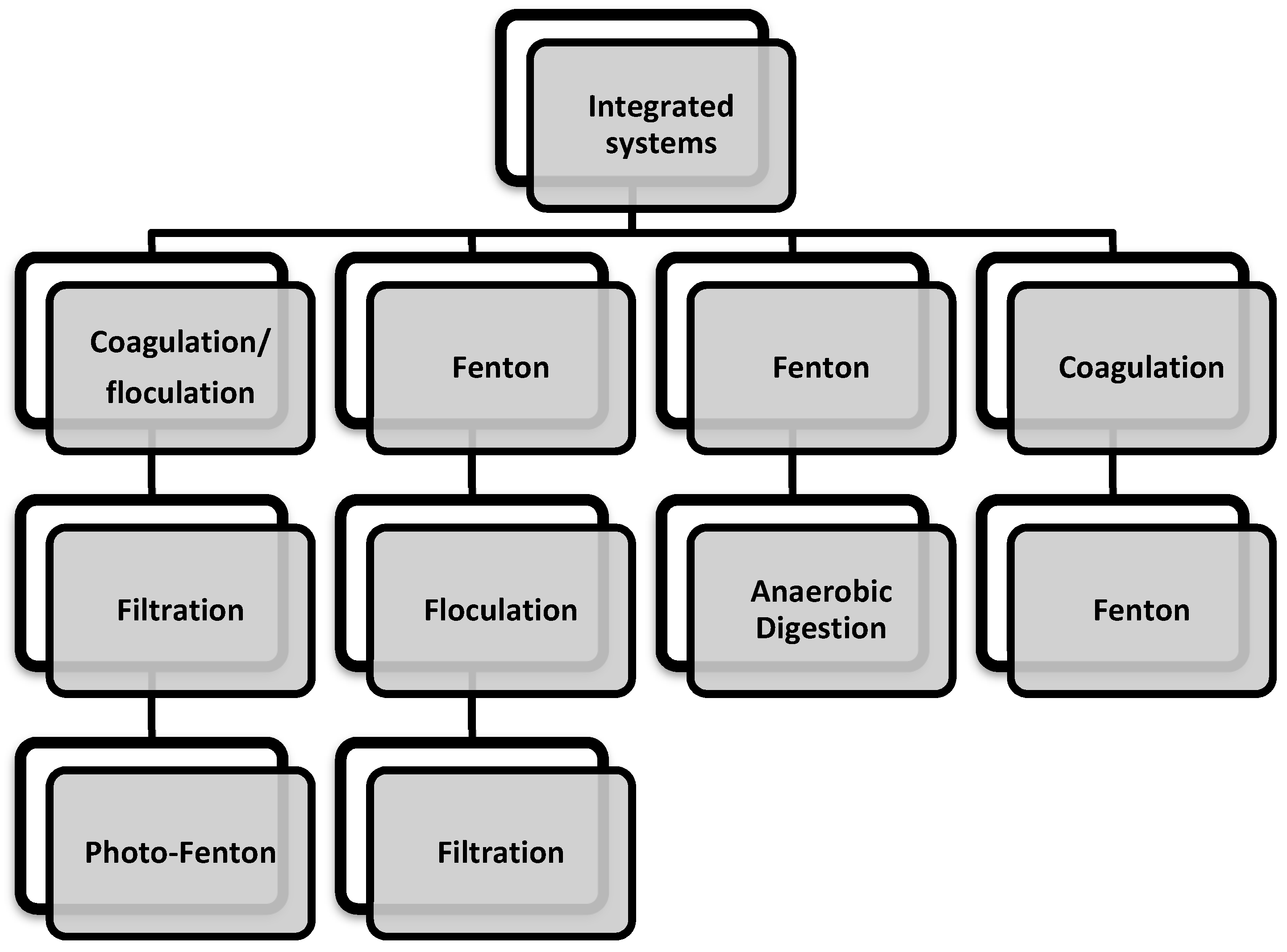
5. Integrated Systems
6. Industrial Applications
7. Concluding Remarks
Author Contributions
Funding
Conflicts of Interest
References
- Bourgin, M.; Beck, B.; Boehler, M.; Borowska, E.; Fleiner, J.; Salhi, E.; Teichler, R.; Von Gunten, U.; Siegrist, H.; Mcardell, C.S. Evaluation of a full-scale wastewater treatment plant upgraded with ozonation and biological post-treatments: Abatement of micropollutants, formation of transformation products and oxidation by-products. Water Res. 2018, 129, 486–498. [Google Scholar] [CrossRef] [PubMed]
- Pulido, J.M.O. A review on the use of membrane technology and fouling control for olive mill wastewater treatment. Sci. Total Environ. 2016, 563–564, 664–675. [Google Scholar] [CrossRef]
- Mert, B.K.; Yonar, T.; Kiliç, M.Y.; Kestioglu, K. Pre-treatment studies on olive mill efluent using physicochemical, Fenton and Fenton-like oxidations processes. J. Hazard. Mater. 2010, 174, 122–128. [Google Scholar] [CrossRef] [PubMed]
- Dias, A.A.; Bezerra, R.M.; Pereira, A.N. Activity and elution profile of laccase during biological decolorization and dephenolization of olive mill wastewater. Bioresour. Technol. 2004, 92, 7–13. [Google Scholar] [CrossRef] [PubMed]
- Cañizares, P.; Martinez, L.; Paz, R.; Sáez, C.; Lobato, J.; Rodrigo, M.A. Treatment of fenton-refractory olive mill wastes by electrochemical oxidation with boron-doped diamond anodes. J. Chem. Technol. Biotechnol. 2006, 81, 1331–1337. [Google Scholar] [CrossRef]
- Guido Greco, J.; Colarieti, M.; Toscano, G.; Iamarino, G.; Rao, M.; Gianfreda, L. Mitigation of Olive Mill Wastewater Toxicity. J. Agric. Food Chem. 2006, 54, 6776–6782. [Google Scholar]
- Saadi, I.; Laor, Y.; Raviv, M.; Medina, S. Land spreading of olive mill wastewater: Effects on soil microbial activity and potential phytotoxicity. Chemosphere 2007, 66, 75–83. [Google Scholar] [CrossRef]
- Azbar, N.; Bayram, A.; Filibeli, A.; Muezzinoglu, A.; Sengul, F.; Ozer, A. A Review of Waste Management Options in Olive Oil Production. Crit. Rev. Environ. Sci. Technol. 2004, 34, 209–247. [Google Scholar] [CrossRef]
- Fabrizio, D.C.; Altimare, P.; Pagnanelli, F. Integrated microalgae production and olive mill wastewater biodegradation: Optimization of the wastewater supply strategy. Chem. Eng. J. 2018, 349, 539–546. [Google Scholar]
- Amaral-Silva, N.; Martins, R.C.; Nunes, P.; Castro-Silva, S.; Quinta-Ferreira, R.M. From a lab test to industrial application: Scale-up of Fenton process for real olive mill wastewater treatment. J. Chem. Technol. Biotechnol. 2017, 92, 1336–1342. [Google Scholar] [CrossRef]
- Gomes, J.; Costa, R.; Quinta-Ferreira, R.M.; Martins, R.C. Application of ozonation for pharmaceuticals and personal care products removal from water. Sci. Total Environ. 2017, 586, 265–283. [Google Scholar] [CrossRef] [PubMed]
- Oller, I.; Malato, S.; Sánchez-Pérez, J.A. Combination of Advanced Oxidation Processes and biological treatments for wastewater decontamination—A review. Sci. Total Environ. 2011, 409, 4141–4166. [Google Scholar] [CrossRef] [PubMed]
- Martins, R.C.; Quinta-Ferreira, R.M. A Review on the Applications of Ozonation for the Treatment of Real Agro-Industrial Wastewaters. Ozone Sci. Eng. 2014, 36, 3–35. [Google Scholar] [CrossRef]
- Amaral-Silva, N.; Martins, R.C.; Castro-Silva, S.; Quinta-Ferreira, R.M. Integration of traditional systems and advanced oxidation process technologies for the industrial treatment of olive mill wastewaters. Environ. Technol. 2016, 37, 2524–2535. [Google Scholar] [CrossRef] [PubMed]
- Ochando-Pulido, J.M.; Oimentel-Mora, S.; Verardo, V.; Martines-Ferez, A. A focus on advanced physico-chemical processes for olive mill wastewater treatment. Separ. Purif. Technol. 2017, 179, 161–174. [Google Scholar] [CrossRef]
- Bautista, P.; Mohedano, A.F.; Casas, J.A.; Zazo, J.A.; Rodriguez, J.J. An overview of the application of Fenton oxidation to industrial wastewaters treatment. J. Chem. Technol. Biotechnol. 2008, 83, 1323–1338. [Google Scholar] [CrossRef]
- Badawy, M.I.; Ali, M.E.M. Fenton’s peroxidation and coagulation processes for the treatment of combined industrial and domestic wastewater. J. Hazard. Mater. 2006, 136, 961–966. [Google Scholar] [CrossRef]
- Burbano, A.A.; Dionysiou, D.D.; Suidan, M.T.; Richardson, T.L. Oxidation kinetics and effect of pH on the degradation of MTBE with Fenton reagent. Water Res. 2005, 39, 107–118. [Google Scholar] [CrossRef] [PubMed]
- Amaral-Silva, N.; Martins, R.C.; Castro-Silva, S.; Quinta Ferreira, R.M. Fenton’s treatment as an effective treatment for elderberry effluents: Economical evaluation. Environ. Technol. 2016, 37, 1208–1219. [Google Scholar] [CrossRef]
- Benzaquén, T.B.; Isla, M.A.; Alfano, O.M. Fenton and photo-Fenton processes for the degradation of atrazine: A kinetic study. J. Chem. Technol. Biotechnol. 2015, 90, 459–467. [Google Scholar] [CrossRef]
- Cao, G.; Sheng, M.; Niu, W.; Fei, Y.; Li, D. Regeneration and reuse of iron catalyst for Fenton-like reactions. J. Hazard. Mater. 2009, 172, 1446–1449. [Google Scholar] [CrossRef] [PubMed]
- Martins, R.C.; Rossi, A.F.; Quinta-Ferreira, R.M. Fenton’s oxidation process for phenolic wastewater remediation and biodegradability enhancement. J. Hazard. Mater. 2010, 180, 716–721. [Google Scholar] [CrossRef] [PubMed]
- Bolobajev, J.; Kattel, E.; Viisimaa, M.; Goi, A.; Trapido, M.; Tenno, T.; Dulova, N. Reuse of ferric sludge as an iron source for the Fenton-based process in wastewater treatment. Chem. Eng. J. 2014, 255, 8–13. [Google Scholar] [CrossRef]
- Rossi, A.F.; Martins, R.C.; Quinta-Ferreira, R.M. Reuse of homogeneous Fenton’s sludge from detergent industry as Fenton’s catalyst. J. Adv. Oxid. Technol. 2013, 16. [Google Scholar] [CrossRef]
- Zhang, H.; Liu, J.; Ou, C.; Faheem; Shen, J.; Yu, H.; Jiao, Z.; Han, W.; Sun, X.; Li, J.; et al. Reuse of Fenton sludge as an iron source for NiFe2O4 synthesis and its application in the Fenton-based process. J. Environ. Sci. 2017, 53, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Xue, G.; Chen, H.; Li, X. Magnetic biochar catalyst derived from biological sludge and ferric sludge using hydrothermal carbonization: Preparation, characterization and its circulation in Fenton process for dyeing wastewater treatment. Chemosphere 2018, 191, 64–71. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, C.S.D.; Soares, O.S.G.P.; Pinho, M.T.; Pereira, M.F.R.; Madeira, L.M. p-Nitrophenol degradation by heterogeneous Fenton’s oxidation over activated carbon-based catalysts. Appl. Catal. B Environ. 2017, 219, 109–122. [Google Scholar] [CrossRef]
- Malato, S.; Blanco, J.; Maldonado, M.I.; Oller, I.; Gernjak, W.; Pérez-Estrada, L. Coupling solar photo-Fenton and biotreatment at industrial scale: Main results of a demonstration plant. J. Hazard. Mater. 2007, 146, 440–446. [Google Scholar] [CrossRef]
- González, O.; Sans, C.; Esplugas, S. Sulfamethoxazole abatement by photo-Fenton: Toxicity, inhibition and biodegradability assessment of intermediates. J. Hazard. Mater. 2007, 146, 459–464. [Google Scholar] [CrossRef]
- Poza-Nogueiras, V.; Rosales, E.; Pazos, M.; Sanromán, M.Á. Current advances and trends in electro-Fenton process using heterogeneous catalysts—A review. Chemosphere 2018, 201, 399–416. [Google Scholar] [CrossRef]
- Liu, X.; Zhou, Y.; Zhang, J.; Luo, L.; Yang, Y.; Huang, H.; Peng, H.; Tang, L.; Mu, Y. Insight into electro-Fenton and photo-Fenton for the degradation of antibiotics: Mechanism study and research gaps. Chem. Eng. J. 2018, 347, 379–397. [Google Scholar] [CrossRef]
- Walling, C. Fenton’s reagent revisited. Acc. Chem. Res. 1975, 8, 125–131. [Google Scholar] [CrossRef]
- Neyens, E.; Baeyens, J. A review of classic Fenton’s peroxidation as an advanced oxidation technique. J. Hazard. Mater. 2003, 98, 33–50. [Google Scholar] [CrossRef]
- Lucas, M.S.; Peres, J.A. Removal of COD from olive mill wastewater by Fenton’s reagent: Kinetic study. J. Hazard. Mater. 2009, 168, 1253–1259. [Google Scholar] [CrossRef] [PubMed]
- Vlyssides, A.G.; Loukakis, H.N.; Karlis, P.K.; Barampouti, E.M.P.; Mai, S.T. Olive mill wastewater detoxification by applying pH related Fenton oxidation process. Fresenius Environ. Bull. 2004, 13, 501–504. [Google Scholar]
- Gomec, C.Y.; Erdim, E.; Turan, I.; Aydin, A.F.; Ozturk, I. Advanced oxidation treatment of physico-chemically pre-treated olive mill industry effluent. J. Environ. Sci. Health Part B 2007, 42, 741–747. [Google Scholar] [CrossRef]
- Rivas, F.; Beltrán, F.; Gimeno, O.; Frades, J. Treatment of Olive Oil Mill Wastewater by Fenton’s Reagent. J. Agric. Food Chem. 2001, 49, 1873–1880. [Google Scholar] [CrossRef]
- Beltrán-Heredia, J.; Torregrosa, J.; García, J.; Dominguez, J.R.; Tierno, J.C. Degradation of olive mill wastewater by the combination of Fenton’s reagent and ozonation processes with an aerobic biological treatment. Water Sci. Technol. 2001, 44, 103–108. [Google Scholar] [CrossRef]
- Maamir, W.; Ouahabi, Y.; Poncin, S.; Li, H.-Z.; Bensadok, K. Effect of Fenton pretreatment on anaerobic digestion of olive mill wastewater and olive mill solid waste in mesophilic conditions. Int. J. Green Energy 2017, 14, 555–560. [Google Scholar] [CrossRef]
- Nieto, L.M.; Hodaifa, G.; Rodríguez, S.; Giménez, J.A.; Ochando, J. Degradation of organic matter in olive-mill wastewater through homogenous Fenton-like reaction. Chem. Eng. J. 2001, 173, 503–510. [Google Scholar] [CrossRef]
- Esteves, B.M.; Rodrigues, C.S.D.; Madeira, L.M. Synthetic olive mill wastewater treatment by Fenton’s process in batch and continuous reactors operation. Environ. Sci. Pollut. Res. 2017, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Reis, P.M.; Martins, P.J.M.; Martins, R.C.; Gando-Ferreira, L.M.; Quinta-Ferreira, R.M. Integrating Fenton’s process and ion exchange for olive mill wastewater treatment and iron recovery. Environ. Technol. 2017, 39, 308–316. [Google Scholar] [CrossRef] [PubMed]
- Víctor-Ortega, M.D.; Ochando-Pulido, J.M.; Martínez-Ferez, A. Iron removal and reuse from Fenton-like pretreated olive mill wastewater with novel strong-acid cation exchange resin fixed-bed column. J. Ind. Eng. Chem. 2016, 36, 298–305. [Google Scholar]
- Ochando-Pulido, J.M.; Víctor-Ortega, M.D.; Martínez-Férez, A.; Martínez-Férez, A. Recovery of iron after Fenton-like secondary treatment of olive mill wastewater by nano-filtration and low-pressure reverse osmosis membranes. Grasas y Aceites 2016, 67, e147. [Google Scholar] [CrossRef]
- Qiang, Z.; Chang, J.; Huang, C. Electrochemcial regeneration of Fe3+ in Fenton oxidation processes. Water Res. 2003, 37, 1308–1319. [Google Scholar] [CrossRef]
- Li, C.; Cen, Y.; Chiou, Y.; Liu, C. Dye wastewater treated by Fenton process with ferrous ions electrocatalytically generated from iron containg sludge. J. Hazard. Mater. 2007, 144, 570–576. [Google Scholar] [CrossRef] [PubMed]
- Martins, R.C.; Rossi, A.F.; Castro-Silva, S.; Quinta-Ferreira, R.M. Fentons process for post-biologically treated cheese production wastewaters final remediation. Toxicity assessemnt. Int. J. Chem. React. Eng. 2010, 8, A84. [Google Scholar]
- Rossi, A.F.; Martins, R.C.; Quinta-Ferreira, R.M. Composition effect of iron-copper composite catalysts in the fenton heterogeneous process efficiency and cooxidation synergy assessment. Ind. Eng. Chem. Res. 2014, 53, 15369–15373. [Google Scholar] [CrossRef]
- Imamura, S.; Doi, A.; Ishida, S. Wet oxidation of ammonia catalyzed by cerium-based composite oxides. Ind. Eng. Chem. Prod. Res. Dev. 1985, 24, 75–80. [Google Scholar] [CrossRef]
- Martins, R.C.; Amaral-Silva, N.; Quinta-Ferreira, R.M. Ceria based solid catalysts for Fenton’s depuration of phenolic wastewatrs, biodegradability enhancement and toxicity removal. Appl. Catal. B 2010, 99, 135–144. [Google Scholar] [CrossRef]
- Rossi, A.F.; Amaral-Silva, N.; Martins, R.C.; Quinta-Ferreira, R.M. Heterogeneous Fenton using ceria bsed catalysts: Effects of the calcination temperature in the process efficiency. Appl. Catal. B 2012, 111, 254–263. [Google Scholar] [CrossRef]
- Kallel, M.; Belaid, C.; Boussahel, R.; Ksibi, M.; Montiel, A.; Elleuch, B. Olive mill wastewater degradation by Fenton oxidation with zero-valent iron and hydrogen peroxide. J. Hazard. Mater. 2009, 163, 550–554. [Google Scholar] [CrossRef] [PubMed]
- Martins, R.C.; Henriques, L.R.; Quinta-Ferreira, R.M. Catalytic activity of low cost materials for pollutants abatment by Fenton’s process. Chem. Eng. Sci. 2013, 100, 225–233. [Google Scholar] [CrossRef]
- Martins, R.C.; Gomes, T.; Quinta-Ferreira, R.M. Fenton’s depuration of weathered olive mill wastewaters over a Fe-Ce-O solid catalyst. Ind. Eng. Chem. Res. 2010, 49, 9043–9051. [Google Scholar] [CrossRef]
- Martins, R.C.; Quinta-Ferreira, R.M. Remediation of phenolic wastewaters by advanced oxidation processes (AOPs) at ambient conditions: Comparative studies. Chem. Eng. Sci. 2011, 66, 3243–3250. [Google Scholar] [CrossRef]
- Babuponnusami, A.; Muthukumar, K. A review on Fenton and improvements to Fenton process for wastewater treatment. J. Environ. Chem. Eng. 2014, 2, 557–572. [Google Scholar] [CrossRef]
- García, C.; Hodaifa, G. Real olive mill wastewater treatment by photo-fenton system using artificial ultraviolet light lamps. J. Clean. Prod. 2017, 162, 743–753. [Google Scholar] [CrossRef]
- Monteagudo, J.M.; Durán, A.; Aguirre, M.; San Martín, I. Optimization of the mineralization of a mixture of phenolic pollutants under a ferrioxalate-induced solar photo-Fenton process. J. Hazard. Mater. 2011, 185, 131–139. [Google Scholar] [CrossRef]
- Davididou, K.; Chatzisymeon, E.; Perez-Estrada, L.; Oller, I.; Malato, S. Photo-Fenton treatment of saccharin in a solar pilot compound parabolic collector: Use of olive mill wastewater as iron chelating agent, preliminary results. J. Hazard. Mater. 2018. [Google Scholar] [CrossRef]
- Ltaïef, A.H.; Pastrana-Martínez, L.M.; Ammar, S.; Gadri, A.; Faria, J.L.; Silva, A.M. Mined pyrite and chalcopyrite as catalysts for spontaneous acidic pH adjustment in Fenton and LED photo-Fenton-like processes. J. Chem. Technol. Biotechnol. 2018, 93, 1137–1146. [Google Scholar] [CrossRef]
- Ioannou-Ttofa, L.; Michael-Kordatou, I.; Fattas, S.C.; Eusebio, A.; Ribeiro, B.; Rusan, M.; Amer, A.R.B.; Zuraiqi, S.; Waismand, M.; Linder, C.; et al. Treatment efficiency and economic feasibility of biological oxidation, membrane filtration and separation processes, and advanced oxidation for the purification and valorization of olive mill wastewater. Water Res. 2017, 114, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Obied, H.; Allen, M.; Bedgood, D.; Prenzel, P.; Robards, K.; Stockmann, R. Bioactivity and analysis of biophenols recovered from olive mill wastes. J. Agric. Food Chem. 2005, 53, 823–837. [Google Scholar] [CrossRef] [PubMed]
- Papaphilippou, P.C.; Yiannapas, C.; Politi, M.; Daskalaki, V.M.; Michael, C.; Kalogerakis, N.; Fatta-Kassinos, D. Sequential coagulation–flocculation, solvent extraction and photo-Fenton oxidation for the valorization and treatment of olive mill effluent. Chem. Eng. J. 2013, 224, 82–88. [Google Scholar] [CrossRef]
- Alver, A.; Bastuk, E.; Kiliç, A.; Karatas, M. Use of advance oxidation process to improve the biodegradability of olive oil mill effluents. Process Saf. Environ. Prot. 2015, 98, 319–324. [Google Scholar] [CrossRef]
- Ochando-Pulido, J.M.; Hodaifa, G.; Martinez-Ferez, A. Fouling inhibition upon Fenton-like oxidation pretreatment for olive mill wastewater reclamation by membrane process. Chem. Eng. Process. 2012, 62, 89–98. [Google Scholar] [CrossRef]
- El-Goahry, F.A.; Badawy, M.I.; El-Khateeb, M.A.; El-Kalliny, A.S. Integrated treatment of olive mill wastewater (OMW) by the combination of Fenton´s reaction and anaerobic treatment. J. Hazard. Mater. 2009, 162, 1536–1541. [Google Scholar] [CrossRef]
- Amor, C.; Lucas, M.S.; Garcia, J.; Dominguez, J.R.; De Heredia, J.B.; Peres, J.A. Combined treatment of olive mill wastewater by Fenton´s reagent and anaerobic biological process. J. Environ. Sci. Health Part A 2017, 50, 161–168. [Google Scholar] [CrossRef]
- Rizzo, L.; Lofrano, G.; Grassi, M.; Belgiorno, V. Pre-treatment of olive mill wastewater by chitosan coagulation and advanced oxidation processes. Sep. Pur. Technol. 2008, 63, 648–653. [Google Scholar] [CrossRef]
- Martins, R.C.; Silva, A.; Castro-Silva, S.; Garção-Nunes, P.; Quinta-Ferreira, R.M. Adopting strategies to improve the efficiency of ozonation in the real-scale treatment of olive mill wastewaters. Env. Technol. 2010, 31, 1459–1469. [Google Scholar] [CrossRef]
- Hodaifa, G.; Ochando-Pulido, J.M.; Rodriguez-Vives, S.; Martinez-Ferez, A. Optimization of continuous reactor at pilot sacale for olive-oil mill wastemater treatment by Fenton-like process. Chem. Eng. J. 2013, 220, 117–124. [Google Scholar] [CrossRef]
- Pinho, I.; Lopes, D.; Martins, R.; Quina, M. Phytotoxicity assessment of olive mil solid wastes and influence of phenolic compounds. Chemosphere 2017, 185, 258–267. [Google Scholar] [CrossRef] [PubMed]
- Quina, M.; Lopes, D.; Cruz, L.; Andrade, J.; Martins, R.C.; Gando-Ferreira, L.; Dias-Ferreira, C.; Quinta-Ferreira, R.M. Studies on the chemical stabilisation of digestate from mechanically recovered organic fraction of municipal solid waste. Waste Biomass Valor 2015, 6, 711–721. [Google Scholar] [CrossRef]

| Homogeneous Fenton’s Process | |
|---|---|
| Advantages | Disadvantages |
| - Easy operation - Chemical oxygen demand (COD) removal - Biodegradability improvement - Established technology | - Sludge formation - Operation in acidic conditions (pH ~3) - Requires neutralization of pH |
| Effluent | Process | Optimal Conditions | Results | Reference |
|---|---|---|---|---|
| OMW collected in Portugal and diluted to initial COD ~2 g/L | Fenton | pH = 3.5 T = 30 oC H2O2/Fe2+ = 15 H2O2/COD = 1.75 | COD removal of 70% | [34] |
| OMW after cracking and cationic polyelectrolite coagulation. | Fenton | 2000 mg/L of H2O2 and Fe2+ | COD removal of 89% | [36] |
| OMW | Fenton | pH = 3 [H2O2] = 3500 mg/L [FeSO4] = 3000 mg/L | 81% COD removal 86% phenol removal 350 mL/L sludge volume | [3] |
| OMW | Fenton-like | pH = 3 [H2O2] = 3000 mg/L [FeSO4] = 2500 mg/L | 88% COD removal 91% phenol removal 300 mL/L sludge volume | [3] |
| OMW | Fenton | pH = 3 [H2O2]/[Fe2+] = 1000 (mol/mol) [Fe2+] = 1.5 mM t = 120 min | improve methane yield up to 24% | [39] |
| OMW coming from a two-phase olive-oil process industry as well as the wastewaters from olives and olive-oil washing | Fenton-like | pH = 3 ratio of catalyst to hydrogen between 0.02 and 0.04 | 90% in the degradation of organic compounds and phenolic compounds Ea = 8.7 kJ mol−1 | [40] |
| Phenolic mixture (vanillic, caffeic, gallic, 3,4-dihydroxyphenylecetic, coumeric acids and tyrosol) | Fenton | t = 120 min [Fe2+] = 100 ppm [H2O2] = 2.0 g/L T = 30 oC | TOC removal 47.5% phenolic content removals of 96.9% | [41] |
| OMW | Fenton | t = 1 h pH = 3.5 [Fe2+] = 50 mg/L [Fe2+]/[H2O2] = 0.002 (w/w) | 81% of COD removal | [42] |
| Effluent | Process | Optimal Conditions | Results | Reference |
|---|---|---|---|---|
| OMW | Heterogenous Fenton: H2O2 and zero-valent iron | pH = 2.0–4.0 5% of H2O2 Fe0 = 20 g/L t = 3 h | Total color removal. 50% of phenolic content removal | [52] |
| Weathered OMW | Heterogenous Fenton: H2O2 and Fe–Ce–O | pH = 4 [H2O2] = 115 mM [Fe-Ce-O] = 1.5 g/L | 25% of COD removal Final effluent biodegradable (BOD5/COD) = 0.54 and low toxicity | [54] |
| OMW | Heterogenous Fenton: H2O2 and Fe–Ce–O | pH = 3 [H2O2] = 224 mM [Fe-Ce-O] = 1.0 g/L | 31% of TOC removed 48% of COD removed Total TPh degradation after 120 min | [55] |
| Effluent | Process | Optimal conditions | Results | Reference |
|---|---|---|---|---|
| OMW | Photo-Fenton: different medium pressure commercial UV-lamps | pH = 3 [H2O2] >8 g/L FeCl3 solution 3 g/L t = 5–30 min T = 20 oC | 90% of COD, total organic carbon, total phenolic compounds, and turbidity removals | [57] |
| OMW (tyrosol, vanillic acid, caffeic acid, coumaric acid, and ferulic acid) | Photo-Fenton: FeS2 and CuFeS2; UV LED (maximum emission wavelength of 395 nm) | CuFeS2 led to total phenolic compounds depletion; 98% of TOC removal. pH autocorrection (~3). High toxicity towards Aliivibrio fischeri due to Fe and Cu leaching. | [60] | |
| OMW | Solar photo-Fenton after coagulation/flocculation | 95% of COD removal Removed OMW phytotoxicity High dilution required. | [61] |
| Effluent | Process | Optimal conditions | Results | Reference |
|---|---|---|---|---|
| OMW | Integrated treatment scheme involving coagulation-flocculation for the removal of solids followed by extraction with ethyl acetate for the recovery of phenolic compounds (caffeic acid, tyrosol, gallic acid, and oleuropein). Thus, a solar photo-Fenton step | 200 mg/L of Fe2+ 5000 mg/L H2O2 pH 3 | 73% of COD removal High toxicity (Daphnia magna) because of the intermediates formed. | [63] |
| OMW | Integrating coagulation and Fenton oxidation | pH = 3 Fe2+ = 2.5 g/L H2O2 = 1 g/L Fe2+/H2O2 = 2.5 | Pre-coagulation had a good impact in the degradation of organic matter through the following Fenton’s process step. | [64] |
| OMW | Pre-treatment process involved Fenton-like oxidation, flocculation-sedimentation and filtration through olive stones and after membrane process | The pre-treatment decreased COD and polyphenols concentration and acts in the particle size distribution reducing membrane fouling | [65] | |
| OMW | Integrated system of wet hydrogen peroxide catalytic oxidation (WHPCO) followed by two stage upflow anaerobic sludge blanket (UASB) reactor | Fenton´s reaction: diluted wastewater (1OMW:1 tap water) pH = 3.0 COD:H2O2 = 1.0:1.1 Fe2+:H2O2 = 1:50 | Fenton´s reaction improves the efficiency of anaerobic digestion | [66] |
| OMW | Combined treatment by Fenton´s reagent and anaerobic biological process | H2O2/COD ratio of 0.20 pH 3.5 H2O2/Fe2+ molar ratio of 15:1 | Pre-treatment: 17.6% of COD and 82.5% total polyphenols removal. Biodegradability was improved. | [67] |
| OMW | Combined treatment by coagulation with natural organic coagulant, chitosan and advanced oxidation processes, namely, photocatalysis, Fenton, and photo-Fenton | ---- | Coagulation: removed 81% of total suspended solids Photo-Fenton: efficient in COD and aromaticity removal, (93% after 0.5 h) Fenton process reduced 81% of COD after 1 h reaction TiO2 photocatalysis: no significant COD removalChitosan coagulation combined with Fenton or photo-Fenton could be a good alternative. | [68] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Domingues, E.; Gomes, J.; Quina, M.J.; Quinta-Ferreira, R.M.; Martins, R.C. Detoxification of Olive Mill Wastewaters by Fenton’s Process. Catalysts 2018, 8, 662. https://doi.org/10.3390/catal8120662
Domingues E, Gomes J, Quina MJ, Quinta-Ferreira RM, Martins RC. Detoxification of Olive Mill Wastewaters by Fenton’s Process. Catalysts. 2018; 8(12):662. https://doi.org/10.3390/catal8120662
Chicago/Turabian StyleDomingues, Eva, João Gomes, Margarida J. Quina, Rosa M. Quinta-Ferreira, and Rui C. Martins. 2018. "Detoxification of Olive Mill Wastewaters by Fenton’s Process" Catalysts 8, no. 12: 662. https://doi.org/10.3390/catal8120662
APA StyleDomingues, E., Gomes, J., Quina, M. J., Quinta-Ferreira, R. M., & Martins, R. C. (2018). Detoxification of Olive Mill Wastewaters by Fenton’s Process. Catalysts, 8(12), 662. https://doi.org/10.3390/catal8120662