Chlorination of Toluene to o-Chlorotoluene Catalyzed by Ionic Liquids
Abstract
:1. Introduction
2. Results and Discussion
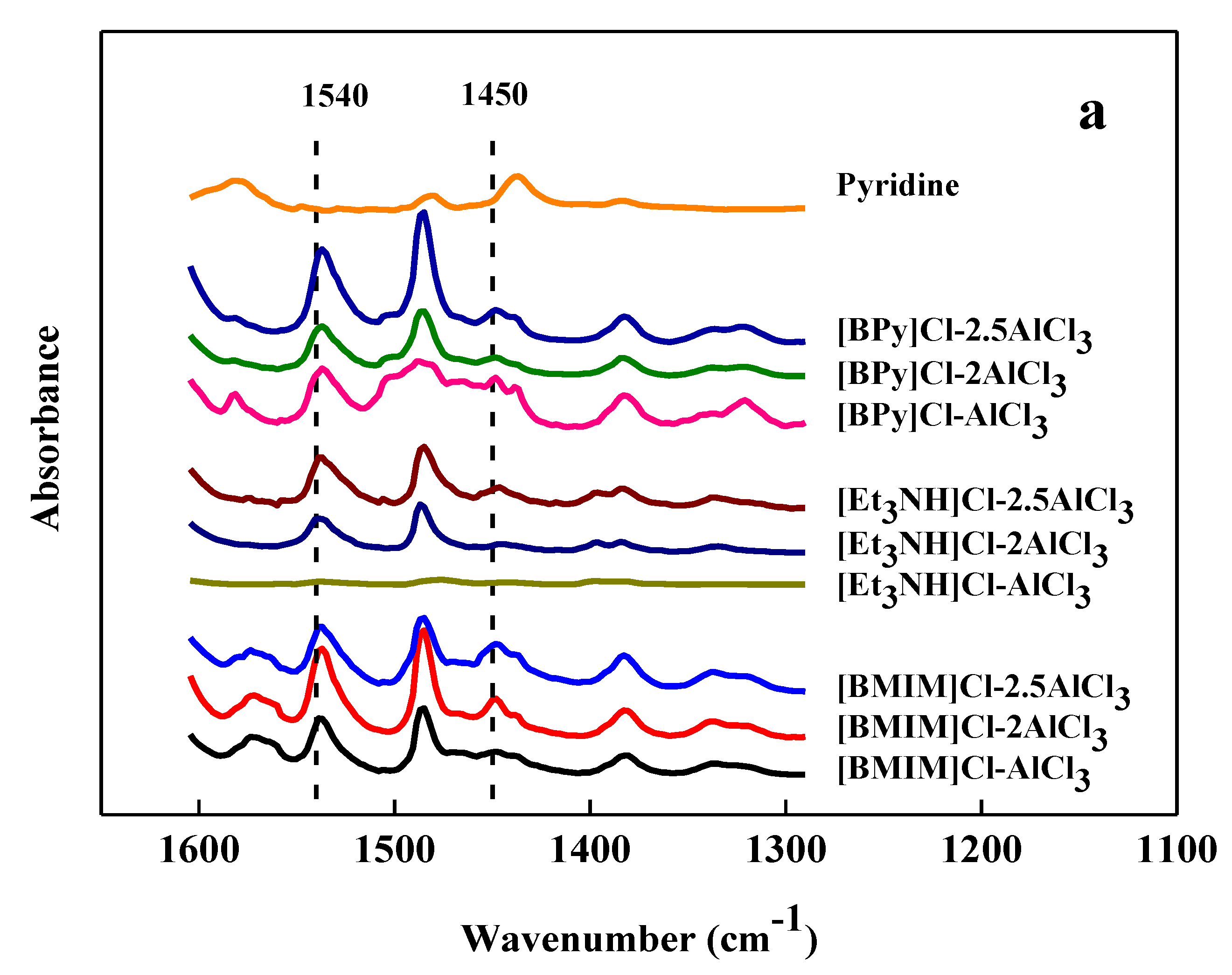
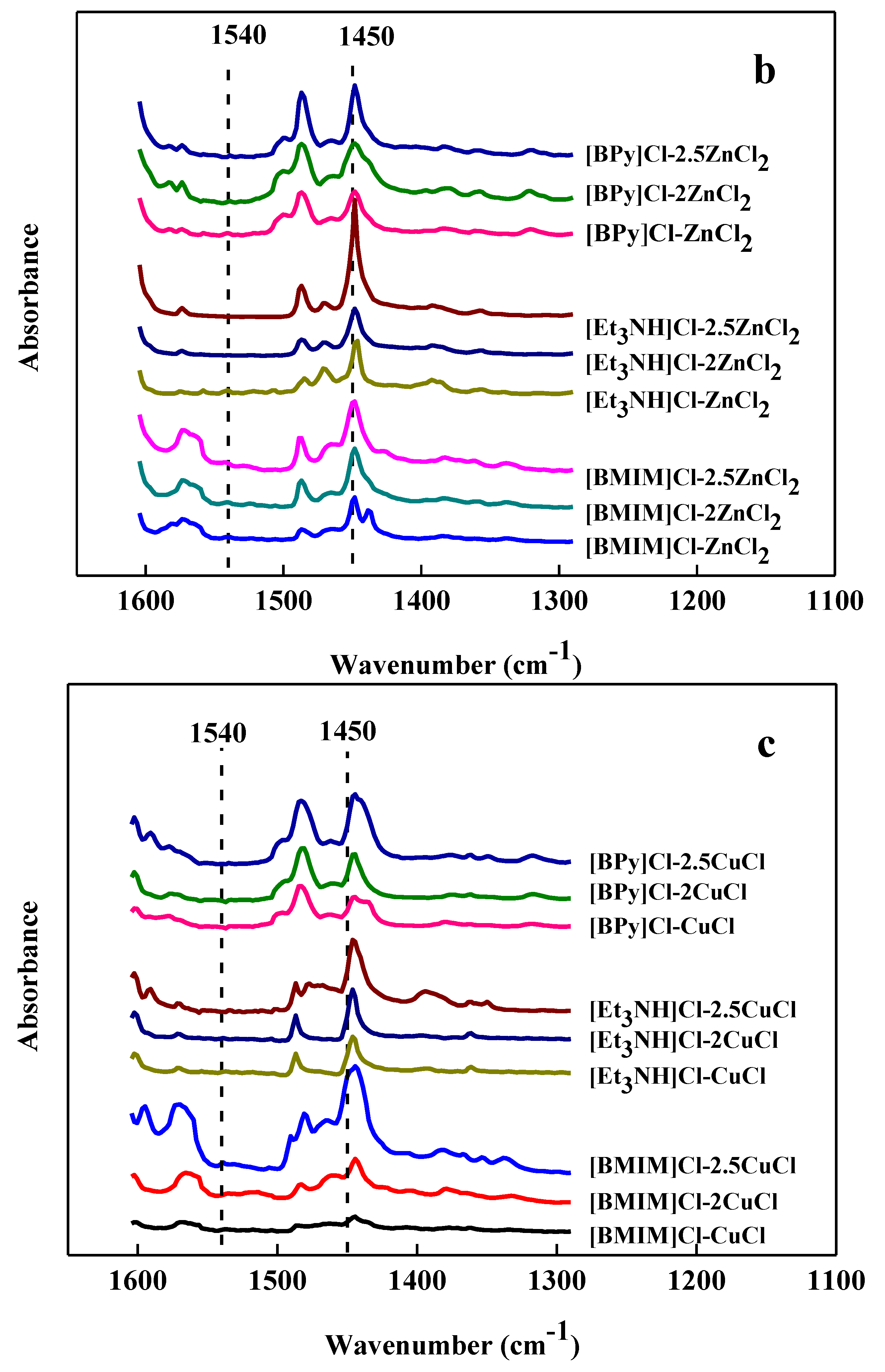
2.1. Acidity of Ionic Liquid
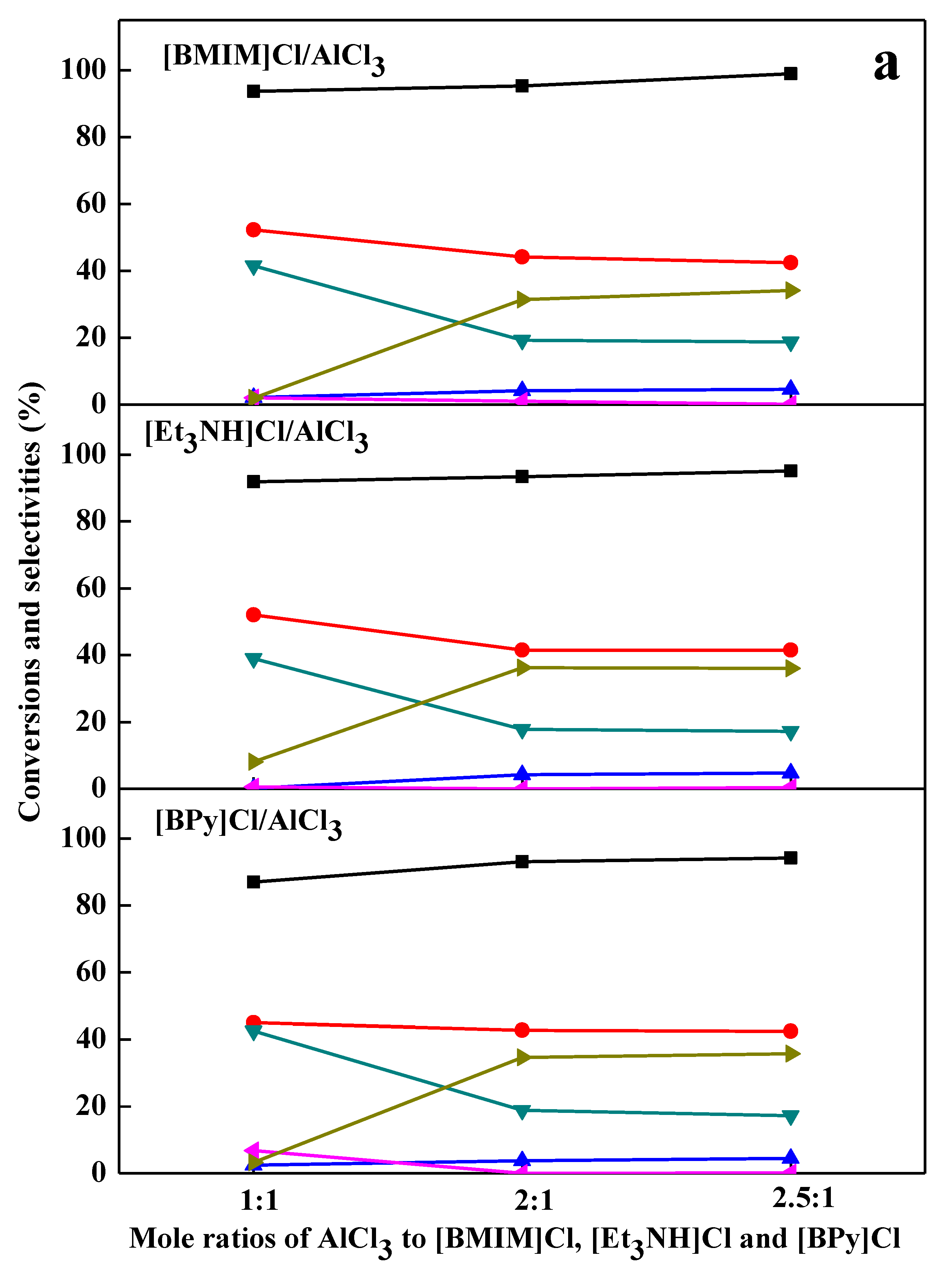
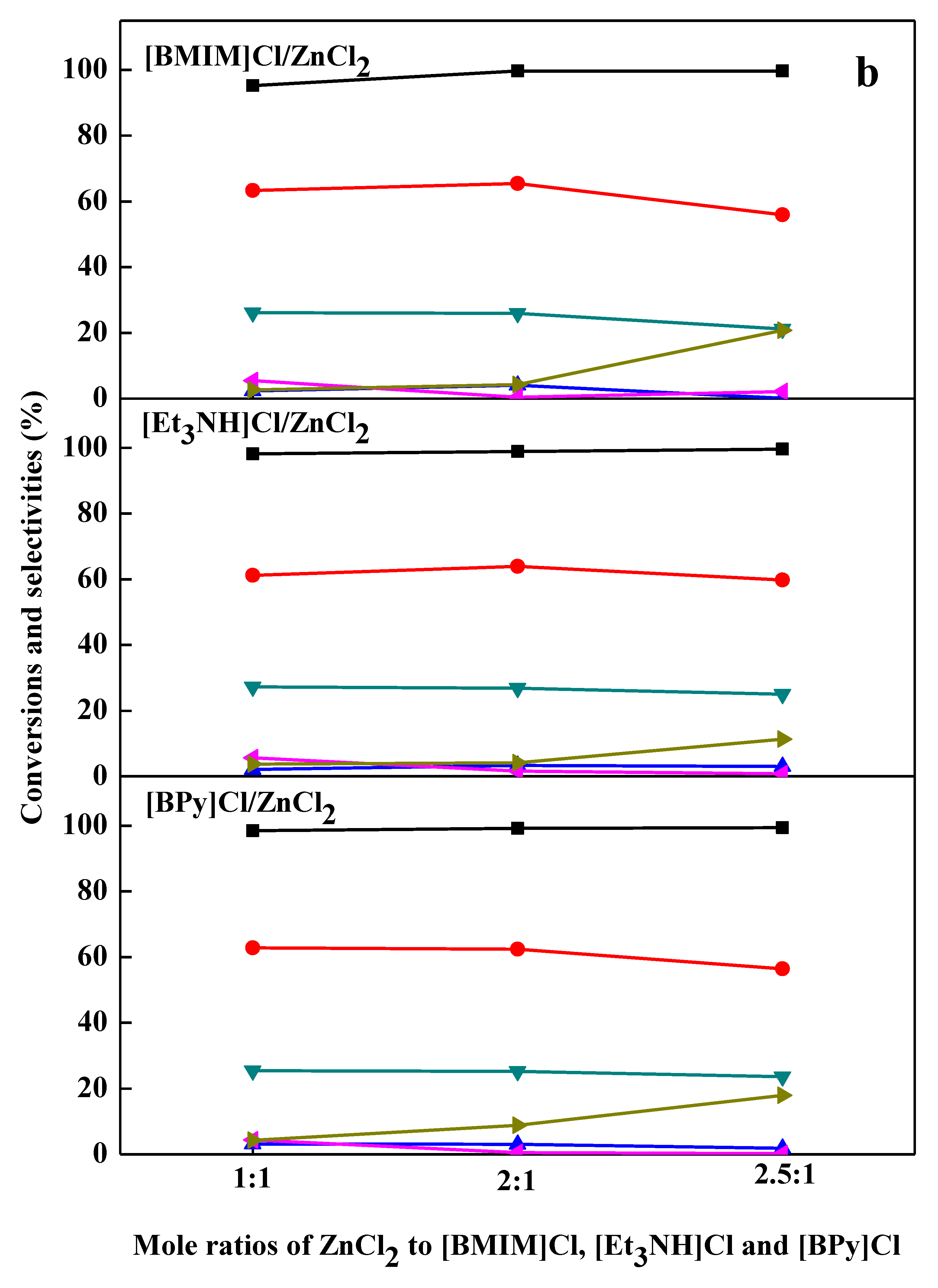
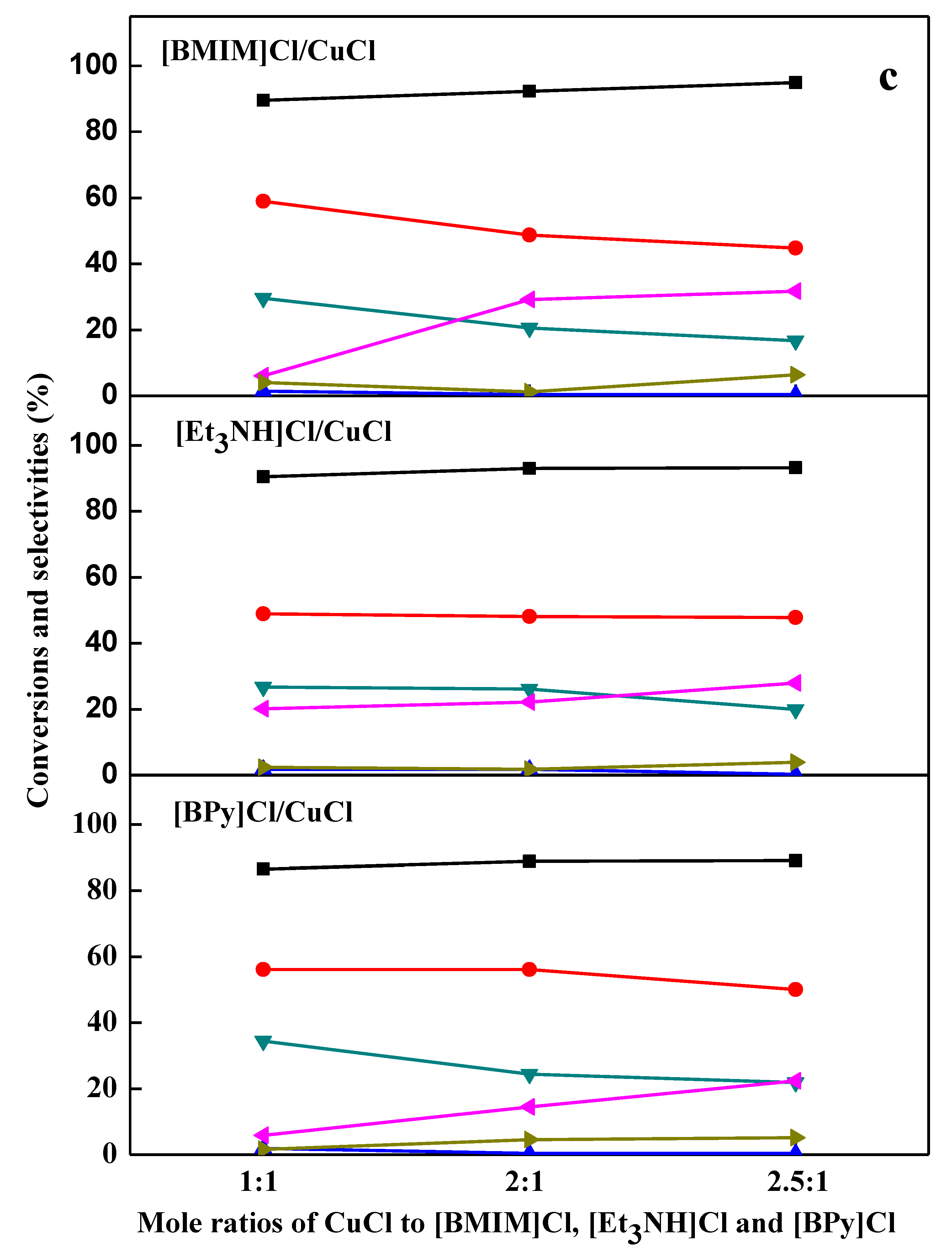
2.2. Chlorination Reaction Catalyzed by Ionic Liquids
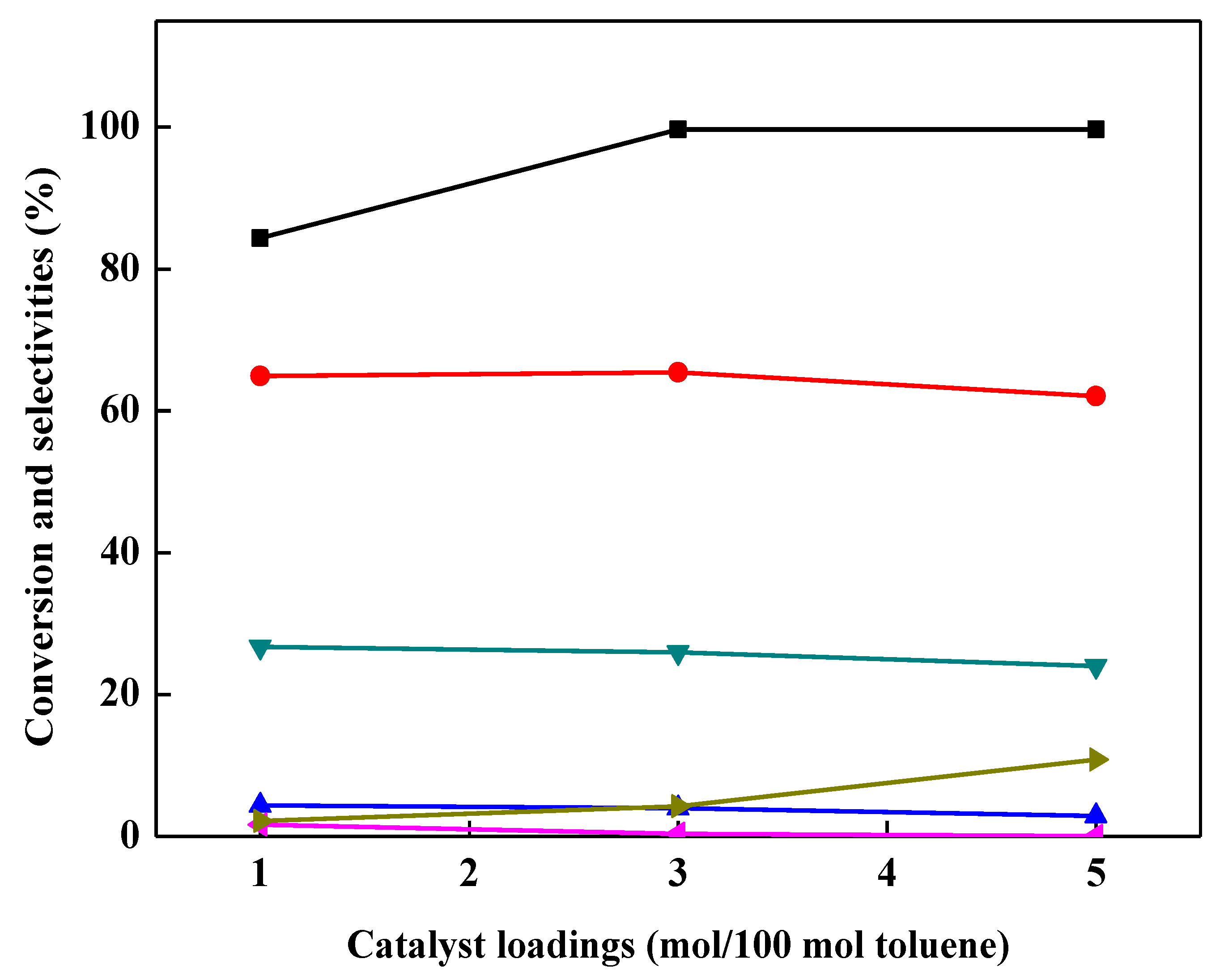
2.3. Effect of Catalyst Loading
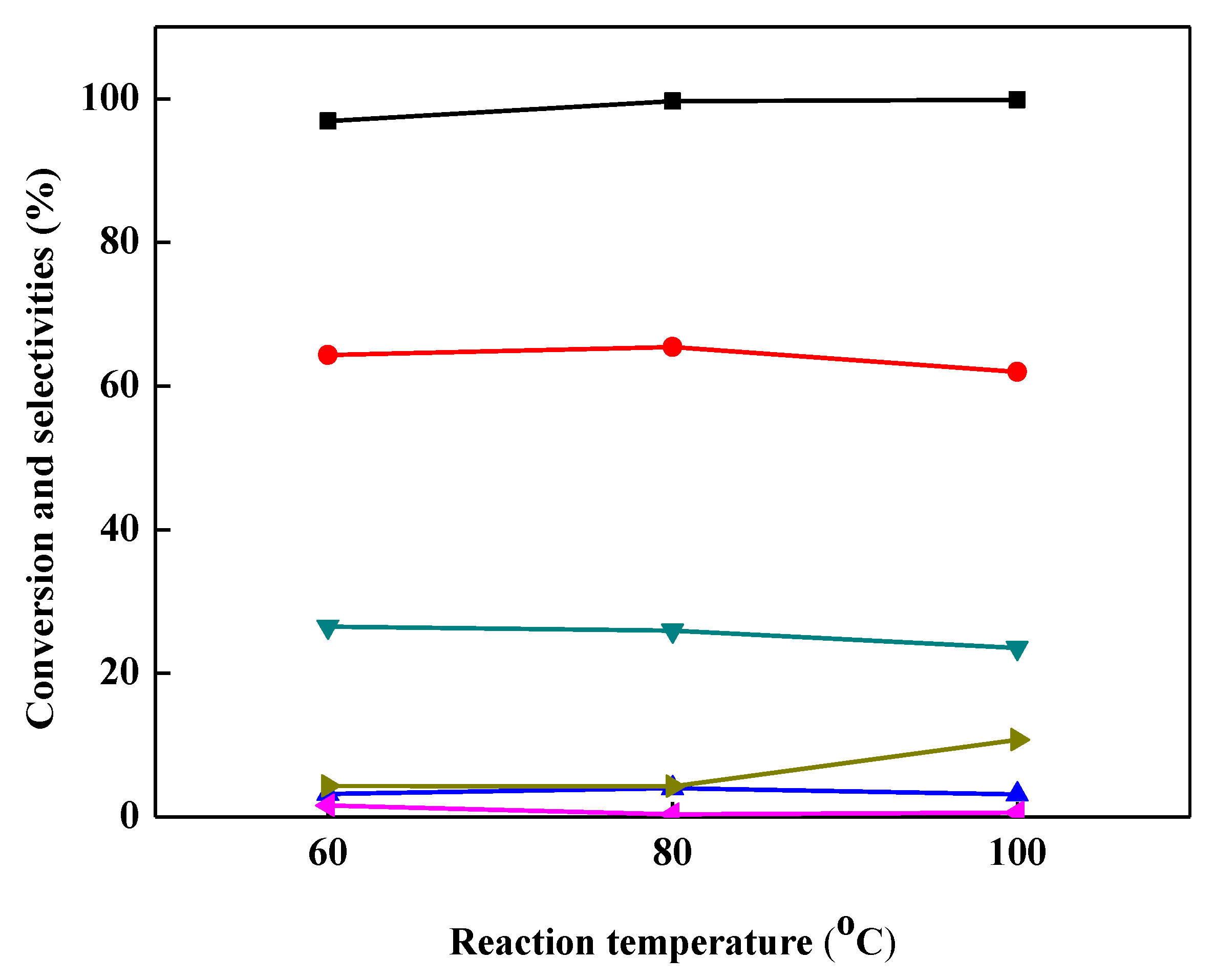
2.4. Effect of Reaction Temperature
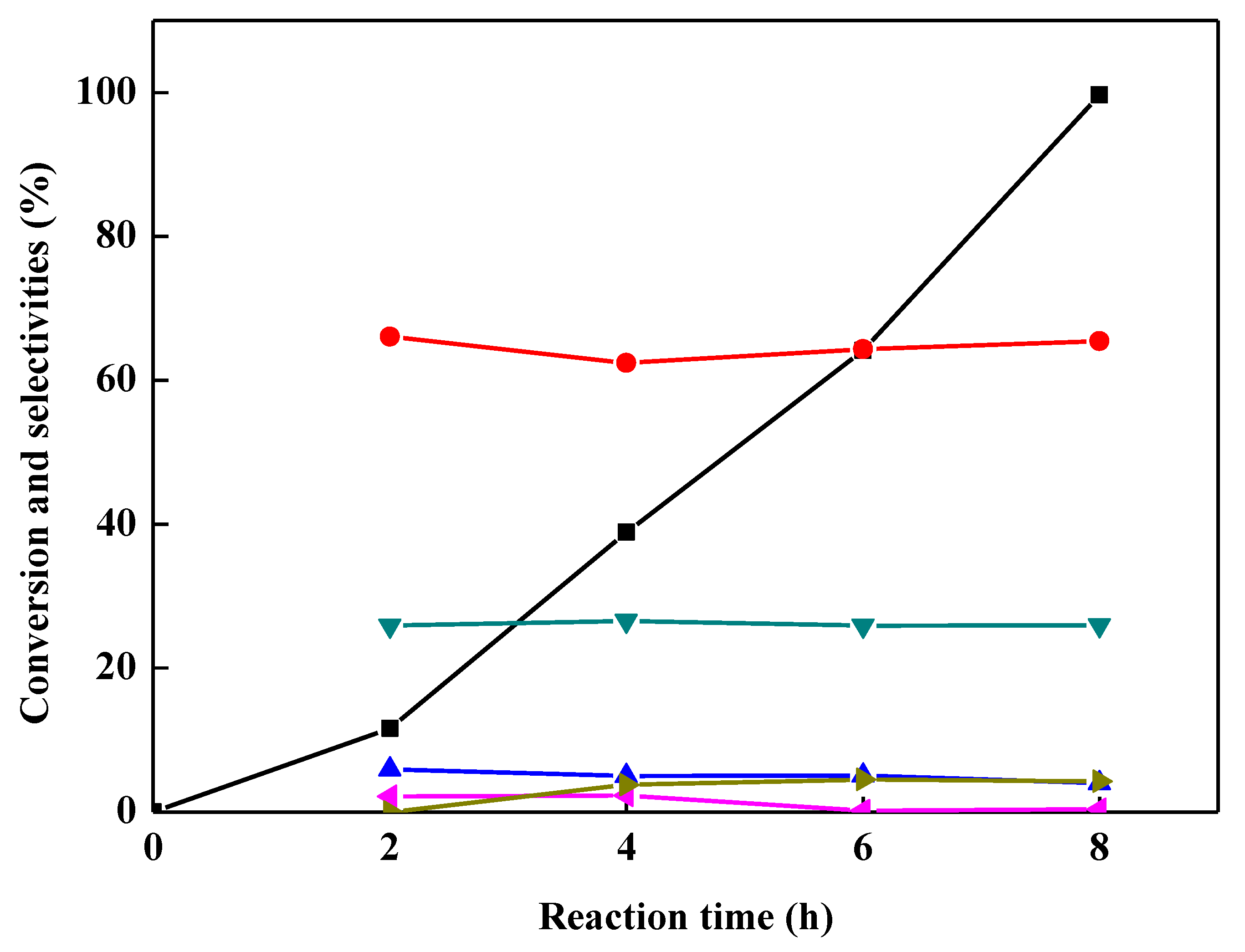
2.5. Effect of Reaction Time
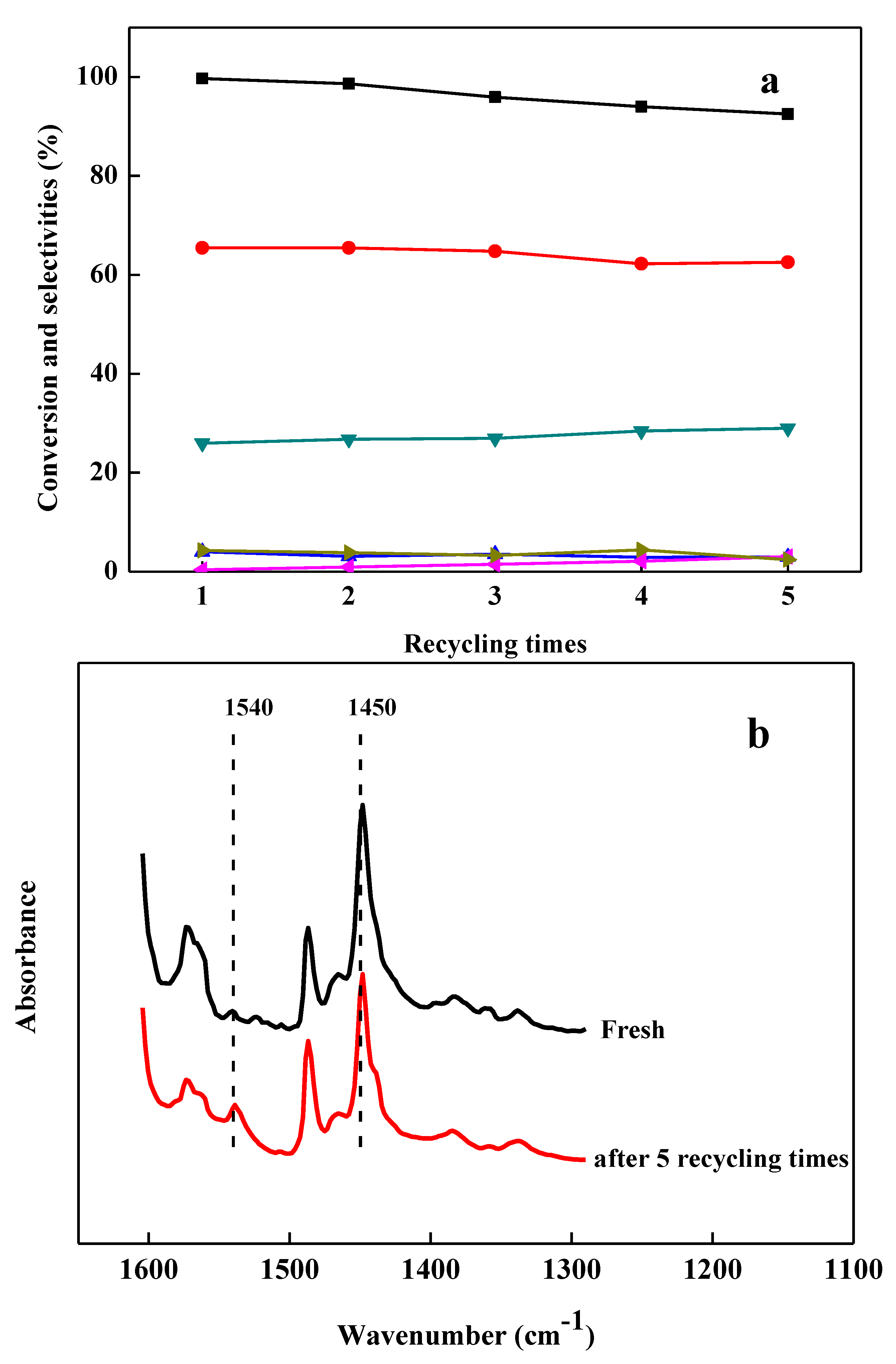
2.6. Recycling Performance
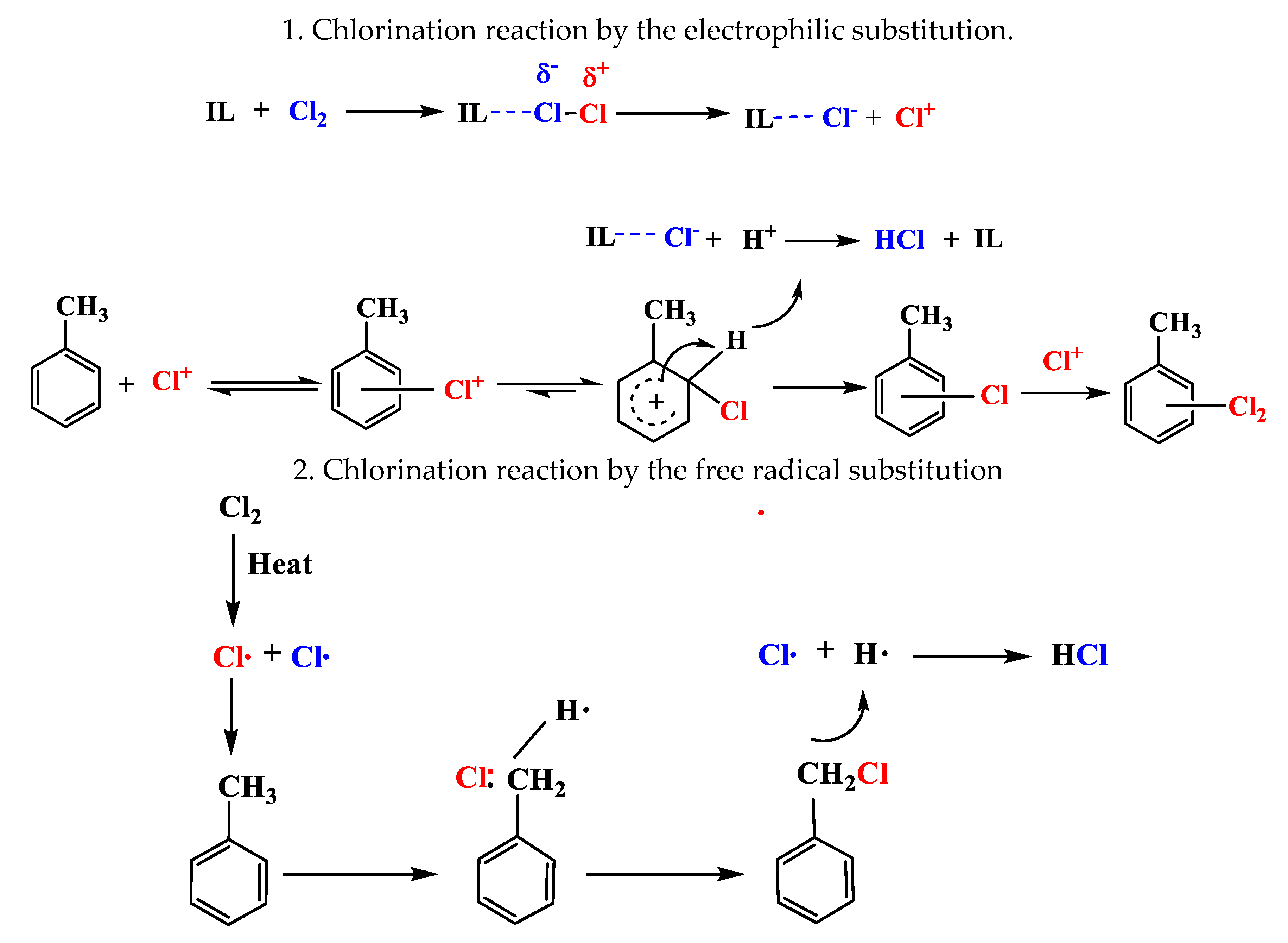
2.7. Reaction Routes
3. Experimental
3.1. Materials
3.2. Synthesis of Ionic Liquids
3.2.1. Synthesis of 1-Butyl-3-methylimidazolium Chloride ([BMIM]Cl)
3.2.2. Synthesis of Triethylammonium Chloride ([Et3NH]Cl)
3.2.3. Synthesis of 1-Butylpyridinium Chloride ([BPy]Cl)
3.2.4. Synthesis of Ionic Liquids
3.3. Determination of Lewis and Brönsted Acid Strengths of Ionic Liquids
3.4. Chlorination Reaction
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Janiak, T. Kinetics of o-chlorotoluene hydrogenolysis in the presence of 3%, 5% and 10% Pd/C catalysts. Appl. Catal. A Gen. 2008, 335, 7–14. [Google Scholar] [CrossRef]
- Norris Shreve, R.; Marsel, C.J. Hydrolysis and isomerization of chlorotoluenes to m-cresol. J. Ind. Eng. Chem. 1946, 38, 254–261. [Google Scholar] [CrossRef]
- Yang, S.C.; Wang, J.Q. Catalytic oxidation of o-chlorotoluene to o-chlorobenzaldehyde by vanadium doped anatase mesoporous TiO2. Adv. Mater. Res. 2013, 781–784, 182–185. [Google Scholar] [CrossRef]
- Vaze, A.S.; Sawant, S.B.; Pangarkar, V.G. Indirect oxidation of o-chlorotoluene to o-chlorobenzaldehyde. J. Appl. Electrochem. 1997, 29, 7–10. [Google Scholar] [CrossRef]
- Di Bella, E.P.; Rochelle Park, N.J. Process for the Production of 2,4-Dichlorotoluene. U.S. Patent 3,366,698, 30 January 1968. [Google Scholar]
- Singh, A.P.; Kumar, S.B. Para selective chlorination of toluene with an L-zeolite catalyst. Appl. Catal. A Gen. 1995, 126, 27–38. [Google Scholar] [CrossRef]
- Huasladen, M.C.; Cyganovich, R.C.; Huang, H.Y.; Lund, C.R.F. Zeolite-catalyzed chlorination of toluene by sulfuryl chloride: Activity, selectivity and deactivation of NaK-L zeolite. Appl. Catal. A Gen. 2001, 219, 1–12. [Google Scholar] [CrossRef]
- Zhu, X.Y.; Fu, Y.J.; Yin, H.B.; Feng, Y.H.; Shen, L.Q.; Wang, A.L.; Li, J.T.; Ni, W.X.; Xie, X.L. Selective chlorination of toluene to p-chlorotoluene catalyzed by nanosized zeolite K-L catalysts. J. Nanosci. Nanotechnol. 2015, 15, 1–10. [Google Scholar] [CrossRef]
- Lei, Z.; Chen, B.; Koo, Y.M. Introduction: Ionic liquids. Chem. Rev. 2017, 117, 6633–6635. [Google Scholar] [CrossRef] [PubMed]
- Jessop, P.G. Fundamental properties and practical applications of ionic liquids: Concluding remarks. Faraday Discuss. 2018, 206, 587–601. [Google Scholar] [CrossRef] [PubMed]
- Saielli, G. Computational spectroscopy of ionic liquids for bulk structure elucidation. Adv. Theory Simul. 2018, 1, 1800084. [Google Scholar] [CrossRef]
- Li, Z.H.; Jia, Z.; Luan, Y.X.; Mu, T.C. Ionic liquids for synthesis of inorganic nanomaterials. Curr. Opin. Solid State Mater. Sci. 2008, 12, 1–8. [Google Scholar] [CrossRef]
- Duan, Z.Y.; Gu, Y.L.; Zhang, J.; Zhu, L.Y.; Deng, Y.Q. Protic pyridinium ionic liquids: Synthesis, acidity determination and their performances for acid catalysis. J. Mol. Catal. A Chem. 2006, 250, 163–168. [Google Scholar] [CrossRef]
- Olivier-Bourbigou, H.; Magna, L. Ionic liquids: Perspectives for organic and catalytic reactions. J. Mol. Catal. A Chem. 2002, 182–183, 419–437. [Google Scholar] [CrossRef]
- Chen, W.Q.; Yin, H.B.; Zhang, Y.S.; Lu, Z.Z.; Wang, A.L.; Shen, Y.T.; Jiang, T.S.; Yu, L.B. Acylation of salicylamide to 5-acetylsalicylamide using ionic liquids as dual catalyst and solvent. J. Ind. Eng. Chem. 2010, 16, 800–804. [Google Scholar] [CrossRef]
- Wang, A.L.; Jiang, Y.Q.; Chen, W.G.; Yin, H.B.; Liu, Y.J.; Shen, Y.T.; Jiang, T.S.; Wu, Z.N. [BMIM]Cl-nAlCl3 ionic liquid-catalyzed redistribution reaction between methyltrichlorosilane and low-boiling residue to dimethyldichlorosilane. J. Ind. Eng. Chem. 2012, 18, 237–242. [Google Scholar] [CrossRef]
- Zhao, H.; Xia, S.Q.; Ma, P.S. Use of ionic liquids as ‘green’ solvents for extractions. J. Chem. Technol. Biotechnol. 2005, 80, 1089–1096. [Google Scholar] [CrossRef]
- Jain, N.; Kumar, A.; Chauhan, S.; Chauhan, S.M.S. Chemical and biochemical transformations in ionic liquids. Tetrahedron 2005, 61, 1015–1060. [Google Scholar] [CrossRef]
- Wang, F.; Xu, C.Z.; Li, Z.; Xia, C.G.; Chen, J. Mechanism and origins of enantioselectivity for [BMIM]Cl ionic liquids and ZnCl2 co-catalyzed coupling reaction of CO2 with epoxides. J. Mol. Catal. A Chem. 2014, 385, 133–140. [Google Scholar] [CrossRef]
- Jiang, Y.Q.; Chen, W.G.; Liu, Y.J.; Yin, H.B.; Shen, Y.T.; Wang, A.L.; Yu, L.B.; Jiang, T.S. Synthesis of trimethylchlorosilane by [BMIM]Cl-nAlCl3 ionic liquids-catalyzed redistribution between methyltrichlorosilane and low-boiling products from the direct synthesis of methylchlorosilanes. Ind. Eng. Chem. Res. 2011, 50, 1893–1898. [Google Scholar] [CrossRef]
- Liu, S.W.; Xie, C.X.; Yu, S.T.; Liu, F.S. Polymerization of α-pinene using Lewis acidic ionic liquid as catalyst. Catal. Commun. 2009, 10, 986–988. [Google Scholar] [CrossRef]
- Yang, Y.L.; Kou, Y. Determination of the Lewis acidity of ionic liquids by means an IR spectroscopic probe. Chem. Commun. 2004, 2, 226–227. [Google Scholar] [CrossRef] [PubMed]
- Wilkes, J.S.; Levisky, J.A.; Wilson, R.A.; Hussey, C.L. Dialkylimidazolium chloroaluminate melts: A new class of room-temperature ionic liquids for electrochemistry, spectroscopy, and synthesis. Inorg. Chem. 1982, 21, 1263–1264. [Google Scholar] [CrossRef]
- Chowdhury, P.K.; Halder, M.; Sanders, L.; Calhoun, T.; Anderson, J.L.; Armstrong, D.W.; Song, X.; Petrich, J.W. Dynamic solvation in room-temperature ionic liquids. J. Phys. Chem. B 2004, 108, 10245–10255. [Google Scholar] [CrossRef]
- Liu, Y.P.; Tan, Z.C.; Di, Y.Y.; Xing, Y.T.; Zhang, P. Lattice potential energies and thermochemical properties of triethylammonium halides (Et3NHX) (X = Cl, Br, and I). J. Chem. Thermodyn. 2012, 45, 100–108. [Google Scholar] [CrossRef]
- Bui, T.L.T.; Korth, W.; Jess, A. Influence of acidity of modified chloroaluminate based ionic liquid catalysts on alkylation of iso-butene with butene-2. Catal. Commun. 2012, 25, 118–124. [Google Scholar] [CrossRef]
- Zhai, C.; Yin, H.B.; Wang, A.L.; Li, J.T. Isomerizationand redistribution of 2,5-dichlorotoluene catalyzed by AlCl3 and isomerization thermodynamics. Eng. Sci. 2018, 3, 84–88. [Google Scholar]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, A.; Zhu, X.; Yin, H.; Fu, Y.; Hou, X. Chlorination of Toluene to o-Chlorotoluene Catalyzed by Ionic Liquids. Catalysts 2018, 8, 532. https://doi.org/10.3390/catal8110532
Wang A, Zhu X, Yin H, Fu Y, Hou X. Chlorination of Toluene to o-Chlorotoluene Catalyzed by Ionic Liquids. Catalysts. 2018; 8(11):532. https://doi.org/10.3390/catal8110532
Chicago/Turabian StyleWang, Aili, Xiaoyan Zhu, Hengbo Yin, Yujun Fu, and Xiangxiang Hou. 2018. "Chlorination of Toluene to o-Chlorotoluene Catalyzed by Ionic Liquids" Catalysts 8, no. 11: 532. https://doi.org/10.3390/catal8110532
APA StyleWang, A., Zhu, X., Yin, H., Fu, Y., & Hou, X. (2018). Chlorination of Toluene to o-Chlorotoluene Catalyzed by Ionic Liquids. Catalysts, 8(11), 532. https://doi.org/10.3390/catal8110532




