Thermo-Catalytic Treatment of Vapors in the Recycling Process of Carbon Fiber-Poly (Benzoxazine) Composite Waste by Pyrolysis
Abstract
1. Introduction
2. Results and Discussion
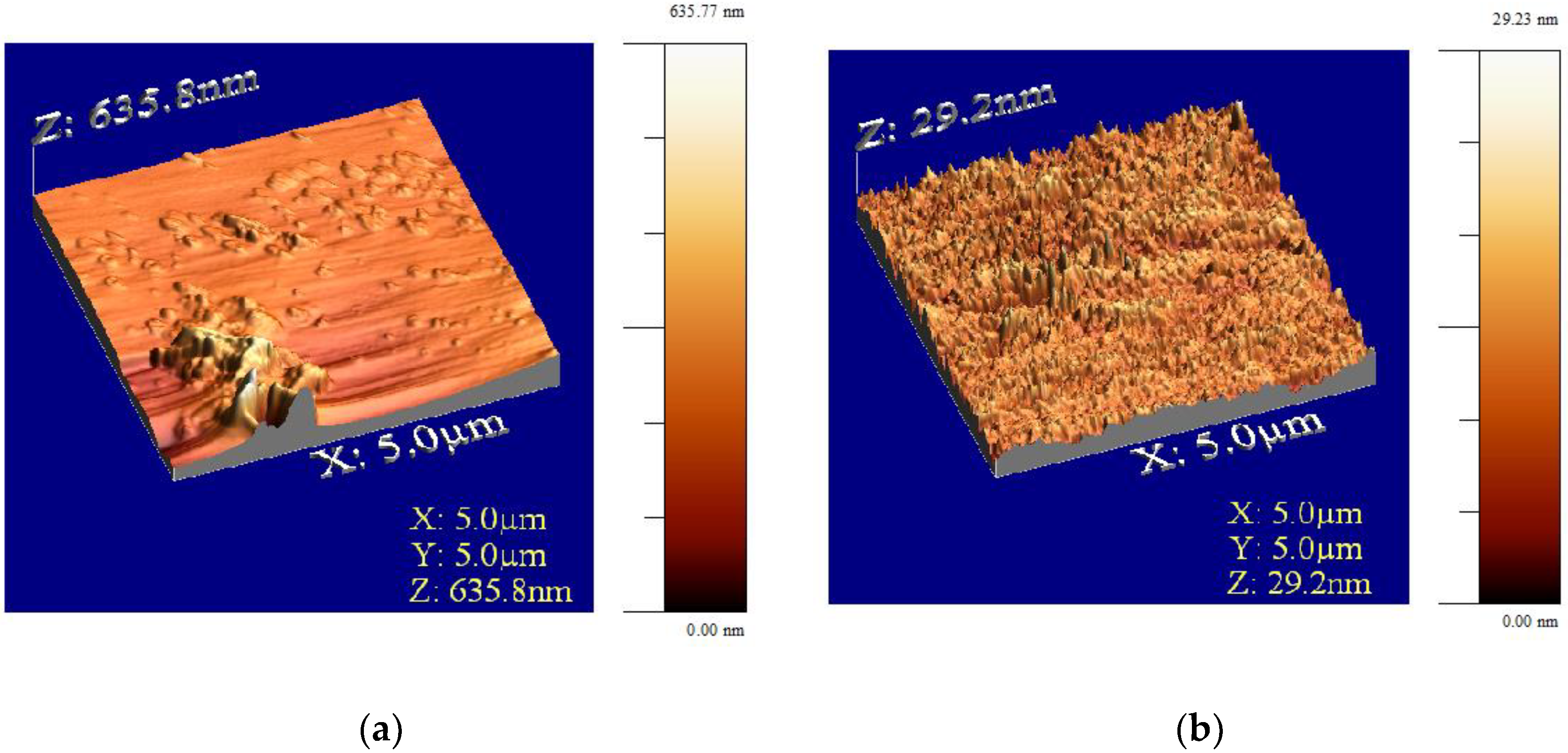
2.1. Characterization of the CFRP Waste
2.2. Pyrolysis Experiments and Gas/Liquid Composition
2.3. Pyrolysis Solids
3. Materials and Methods
3.1. CFRP Sample and Catalysts
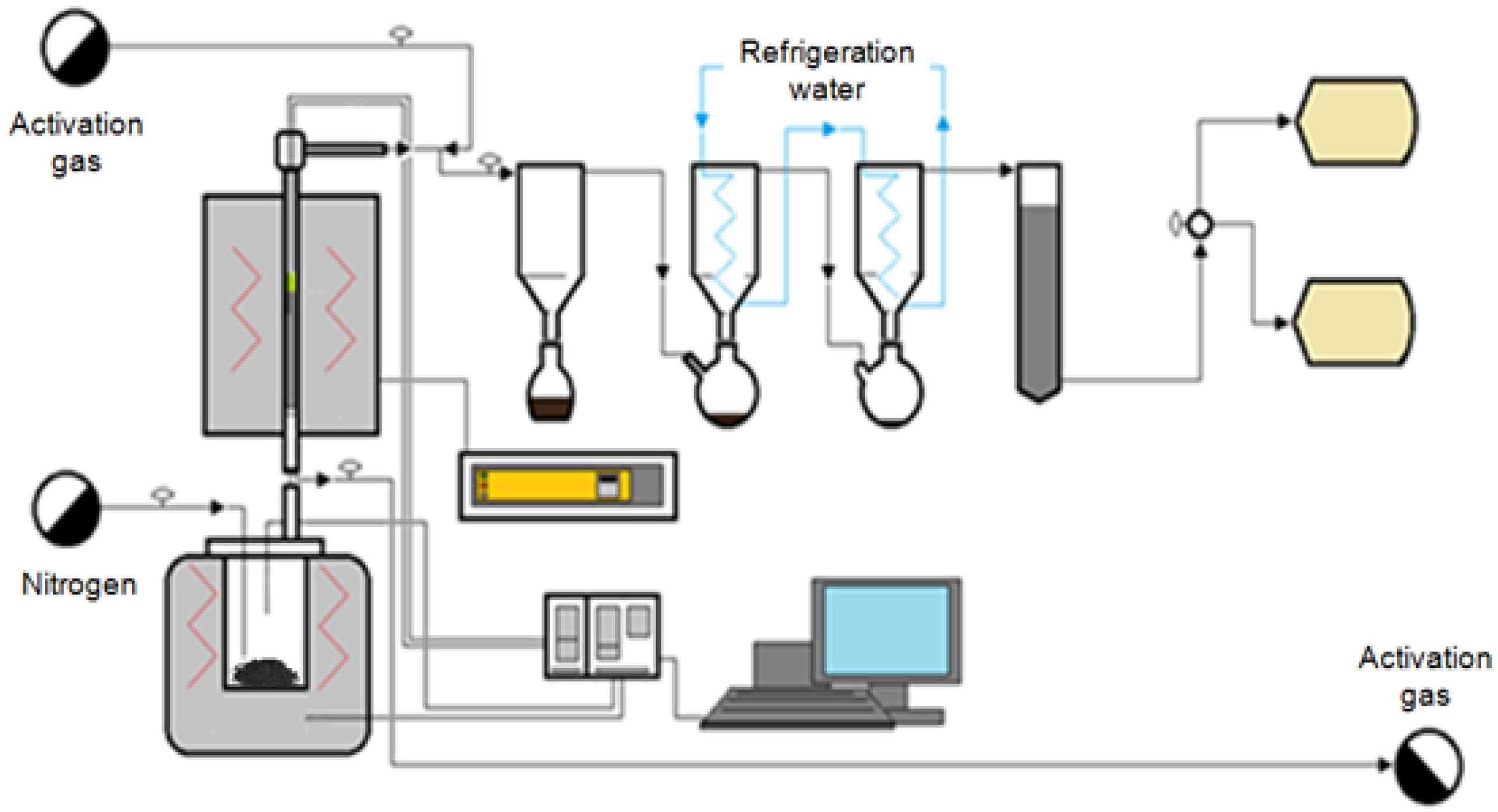
3.2. Pyrolysis Installation
3.3. Analytical Techniques
4. Conclusions
5. Patents
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Callister, W.D.; Rethwisch, D.G. Properties of Selected Engineering Materials (appendix B). In Materials Science and Engineering. An Introduction, 7th ed.; John Wiley & Sons, Inc.: New York, NY, USA, 2007; pp. A3–A30. ISBN 978-0-471-73696-7. [Google Scholar]
- Newcomb, B.A. Processing, structure, and properties of carbon fibers. Compos. Part A-Appl. Sci. Manuf. 2016, 91, 262–282. [Google Scholar] [CrossRef]
- Meng, F.; McKechnie, J.; Tuner, T.A.; Pickering, S.J. Energy and environmental assessment and reuse of fluidised bed recycled carbon fibres. Compos. Part A-Appl. Sci. Manuf. 2018, 100, 206–214. [Google Scholar] [CrossRef]
- Sloan, J. Carbon Fiber 2016 Report. Compos. World 2017, 3, 24–25. Available online: www.compositesworld.com/articles/carbon-fiber-2016-report (accessed on 28 September 2018).
- Giorgini, L.; Benelli, T.; Mazzocchetti, L.; Leonardi, C.; Zattini, G.; Minak, G.; Dolcini, E.; Cavazzoni, M.; Montanari, I.; Tosi, C. Recovery of Carbon Fibers From Cured and Uncured Carbon Fiber Reinforced Composites Wastes and Their Use as Feedstock for a New Composite Production. Polym. Compos. 2015, 36, 1084–1095. [Google Scholar] [CrossRef]
- Vo Dong, P.A.; Azzaro-Pantel, C.; Cadene, A.L. Economic and environmental assessment of recovery and disposal pathways for CFRP waste management. Resour. Conserv. Recycl. 2018, 133, 63–75. [Google Scholar] [CrossRef]
- Pimenta, S.; Pinho, S.T. Recycling carbon fibre reinforced polymers for structural applications: Technology review and market outlook. Waste Manag. 2011, 31, 378–392. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Boom, R.; Irion, B.; van Heerden, D.J.; Kuiper, P.; de Wit, H. Recycling of composite materials. Chem. Eng. Process. 2012, 51, 53–68. [Google Scholar] [CrossRef]
- Pimenta, S.; Pinho, S.T. Recycling of Carbon Fibers. In Handbook of Recycling, 1st ed.; Worrell, E., Reuter, M.A., Eds.; Elsevier: Waltham, MA, USA, 2014; pp. 269–283. ISBN 978-0-12-396459-5. [Google Scholar]
- Black, S. Composites recycling: Gaining traction. Compos. World 2017, 3, 46–55. Available online: www.compositesworld.com/articles/composites-recycling-gaining-traction (accessed on 28 September 2018).
- Wang, F.C.Y.; Dettloff, M.L.; Null, M.J.; White, J.E. Qualitative and quantitative analysis of a thermoset polymer, poly(benzoxazine), by pyrolysis-gas chromatography. J. Chromatogr. A 2000, 886, 217–244. [Google Scholar] [CrossRef]
- Cunliffe, A.M.; Jones, N.; Williams, P.T. Recycling of fibre reinforced polymeric waste by pyrolysis: Thermo-gravimetric and bench-scale investigations. J. Anal. Appl. Pyrolysis 2003, 70, 315–338. [Google Scholar] [CrossRef]
- Yang, J.; Liu, J.; Liu, W.; Wang, J.; Tang, T. Recycling of carbon fibre reinforced epoxy resin composites under various oxygen concentrations in nitrogen-oxygen atmosphere. J. Anal. Appl. Pyrolysis 2015, 112, 253–261. [Google Scholar] [CrossRef]
- Nahil, M.A.; Williams, P.T. Recycling of carbon fibre reinforced polymeric waste for the production of activated carbon fibres. J. Anal. Appl. Pyrolysis 2011, 91, 67–75. [Google Scholar] [CrossRef]
- López, F.A.; Rodríguez, O.; Alguacil, F.J.; García-Díaz, I.; Centeno, T.A.; García-Fierro, J.L.; González, C. Recovery of carbon fibres by the thermolysis and gasification of waste prepreg. J. Anal. Appl. Pyrolysis 2013, 104, 675–683. [Google Scholar] [CrossRef]
- Zhang, J. Different Surface Treatments of Carbon Fibers and Their Influence on the Interfacial Properties of Carbon Fiber/Epoxy Composites. Ph.D. Thesis, National Center for Scientific Research, Paris, France, 2012. [Google Scholar]
- Ghosh, N.N.; Kiskan, B.; Yagci, Y. Polybenzoxazines—New high performance thermosetting resins: Synthesis and properties. Prog. Polym. Sci. 2007, 32, 1344–1391. [Google Scholar] [CrossRef]
- Zúñiga, C.J. Polybenzoxazine Materials from Renewable Diphenolic Acid. Ph.D. Thesis, Universitat Rovira I Virgili, Tarragona, Spain, 2013. [Google Scholar]
- Beyazkilic, Z.; Kahveci, M.U.; Aydogan, B.; Kiskan, B.; Yagci, Y. Synthesis of Polybenzoxazine Precursors Using Thiols: Simultaneous Thiol–Ene and Ring-Opening Reactions. J. Polym. Sci. 2012, 50, 4029–4036. [Google Scholar] [CrossRef]
- Wang, Q.; Ning, H.; Vaidya, U.; Pillay, S.; Nolen, L.A. Development of a carbonization-in-nitrogen method for measuring the fiber content of carbon fiber reinforced thermoset composites. Compos. Part A-Appl. Sci. Manuf. 2015, 73, 80–84. [Google Scholar] [CrossRef]
- Irisawa, T.; Inagaki, R.; Iida, J.; Iwamura, R.; Ujihara, K.; Kobayashi, S.; Tanabe, Y. The influence of oxygen containing functional groups on carbon fibers for mechanical properties and recyclability of CFRTPs made with in-situ polymerizable polyamide 6. Compos. Part A-Appl. Sci. Manuf. 2018, 112, 91–99. [Google Scholar] [CrossRef]
- Meyer, L.O.; Schulte, K. CFRP-Recycling Following a Pyrolysis Route: Process Optimization and Potentials. J. Compos. Mater. 2009, 43, 1121–1132. [Google Scholar] [CrossRef]
- Tranchard, P.; Duquesne, S.; Samyn, F.; Estèbe, B.; Bourbigot, S. Kinetic analysis of the thermal decomposition of a carbon fibre-reinforced epoxy resin laminate. J. Anal. Appl. Pyrolysis 2017, 126, 14–21. [Google Scholar] [CrossRef]
- Onwudili, J.A.; Miskolczi, N.; Nagy, T.; Lipóczi, G. Recovery of glass fibre and carbon fibres from reinforced thermosets by batch pyrolysis and investigation of fibre re-using as reinforcement in LDPE matrix. Compos. Part B-Eng. 2016, 91, 154–161. [Google Scholar] [CrossRef]
- Solar, J.; de Marco, I.; Caballero, B.M.; Lopez-Urionabarrenechea, A.; Rodriguez, N.; Agirre, I.; Adrados, A. Influence of temperature and residence time in the pyrolysis of woody biomass waste in a continuous screw reactor. Biomass Bioenerg. 2016, 95, 416–423. [Google Scholar] [CrossRef]
- Solar, J.; Hernandez, A.; Lopez-Urionabarrenechea, A.; de Marco, I.; Adrados, A.; Caballero, B.M.; Gastelu, N. From woody biomass Waste to biocoke: Influence of the proportion of different tree components. Eur J. Wood Prod. 2017, 75, 485–497. [Google Scholar] [CrossRef]
- Adrados, A.; de Marco, I.; Lopez-Urionabarrenechea, A.; Solar, J.; Caballero, B. Avoiding tar formation in biocoke production from waste biomass. Biomass Bioenerg. 2015, 74, 172–179. [Google Scholar] [CrossRef]
- Adrados, A.; Lopez-Urionabarrenechea, A.; Solar, J.; Requies, J.; de Marco, I.; Cambra, J.F. Upgrading of pyrolysis vapours from biomass carbonization. J. Anal. Appl. Pyrolysis 2013, 103, 293–299. [Google Scholar] [CrossRef]
- Solar, J.; Caballero, B.M.; de Marco, I.; Lopez-Urionabarrenechea, A.; Gastelu, N. Optimization of charcoal production process from woody biomass waste: Effect of Ni-containing catalysts on pyrolysis vapors. Catalysts 2018, 8, 191. [Google Scholar] [CrossRef]
- Mazzocchetti, L.; Benelli, T.; D’Angelo, E.; Leonardi, C.; Zattini, G.; Giorgini, L. Validation of carbon fibers recycling by pyro-gasification: The influence of oxidation conditions to obtain clean fibers and promote fiber/matrix adhesion in epoxy composites. Compos. Part A-Appl. Sci. Manuf. 2018, 112, 504–514. [Google Scholar] [CrossRef]
- Gu, Y.; Ran, Q.C. Polybenzoxazine/Fiber Composites. In Handbook of Benzoxazine Resins, 1st ed.; Ishida, H., Agag, T., Eds.; Elsevier: Waltham, MA, USA, 2011; pp. 481–494. ISBN 9780444537911. [Google Scholar]
- Sawaryn, C.; Kreiling, S.; Schönfeld, R.; Landfester, K.; Taden, A. Benzoxazines for Industrial Applications Comparison with Other Resins, Formulation and Toughening Know-How, and Water-Based Dispersion Technology. In Handbook of Benzoxazine Resins, 1st ed.; Ishida, H., Agag, T., Eds.; Elsevier: Waltham, MA, USA, 2011; pp. 605–620. ISBN 9780444537911. [Google Scholar]
- Adrados, A.; Lopez-Urionabarrenechea, A.; Acha, E.; Solar, J.; Caballero, B.M.; de Marco, I. Hydrogen rich reducing gases generation in the production of charcoal from woody biomass carbonization. Energy Convers. Manag. 2017, 148, 352–359. [Google Scholar] [CrossRef]
- Haber, J.; Block, J.H.; Delmon, B. IUPAC Technical Reports. Manual of methods and procedures for catalyst characterization. Pure Appl. Chem. 1995, 67, 1257–1306. [Google Scholar] [CrossRef]
- Fogler, H.S. Elements of Chemical Reaction Engineering, 5th ed.; Prentice Hall: New Jersey, NJ, USA, 2016; ISBN 9780133887518. [Google Scholar]
- Ross, J. Heterogeneous Catalysis: Fundamentals and Applications, 1st ed.; Elsevier: Waltham, MA, USA, 2011; ISBN 9780444638403. [Google Scholar]



| CFRP Waste | CF | |
|---|---|---|
| Fiber content | 61.0 | 100.0 |
| Proximate analysis | ||
| Moisture | 0.5 | 0.0 |
| Volatiles | 28.4 | 6.2 |
| Fixed carbon 1 | 71.0 | 93.8 |
| Ash | 0.1 | 0.0 |
| Elemental analysis | ||
| C | 86.2 | 95.7 |
| H | 2.2 | 0.1 |
| N | 4.8 | 2.8 |
| S | 1.3 | 0.1 |
| Cl | u.d.l. 2 | u.d.l. 2 |
| Others 3 | 4.9 | 1.3 |
| E1 | E2 | E3 | E4 | E5 | |
|---|---|---|---|---|---|
| Vapor thermal treatment | No | No | 900 °C | 900 °C | 900 °C |
| Catalyst (cat) | No | No | No | Reforming cat | Ni/HZSM-5 |
| Pyrolysis heating rate (°C min−1) | 15 | 3 | 3 | 3 | 3 |
| Pyrolysis yields (wt.%) | |||||
| Solid | 74.8 | 76.9 | 75.2 | 77.1 | 76.3 |
| Total condensates | 20.2 | 16.5 | 10.5 | 10.1 | 10.5 |
| Collected liquid 1 | 10.4 | 4.4 | 1.4 | 1.5 | 1.8 |
| Aqueous fraction 2 | 66.5 | 43.8 | 72.4 | 96.0 | 94.2 |
| Organic fraction 2 | 33.5 | 56.2 | 27.6 | 4.0 | 5.8 |
| Gases 3 | 5.0 | 6.6 | 14.3 | 12.8 | 13.2 |
| Composition (vol.%) and HHV (MJ Nm−3) of pyrolysis gases | |||||
| H2 | 18.6 | <0.1 | 43.4 | 52.9 | 56.3 |
| CO | 14.4 | <0.1 | 12.9 | 21.0 | 19.1 |
| CO2 | 21.8 | 35.0 | 5.9 | 5.4 | 5.1 |
| CH4 | 34.2 | 64.1 | 24.7 | 15.1 | 15.2 |
| C2 | 3.7 | <0.1 | 6.7 | 2.9 | 2.9 |
| C3 | <0.1 | <0.1 | 0.4 | <0.1 | <0.1 |
| C4 | <0.1 | <0.1 | <0.1 | <0.1 | <0.1 |
| C5 | <0.1 | <0.1 | 0.1 | <0.1 | <0.1 |
| C6 | 7.2 | 0.8 | 6.0 | 2.8 | 1.5 |
| HHV 4 | 30.0 | 24.6 | 29.6 | 20.3 | 18.4 |
| Composition of pyrolysis liquids (area%)—aqueous phase | |||||
| Water | 82.4 | 86.2 | 79.4 | 83.8 | 83.3 |
| Aniline | 6.7 | 13.8 | 13.7 | 16.2 | 16.7 |
| Phenol | 5.9 | n.d. 5 | n.d. 5 | n.d. 5 | n.d. 5 |
| Unidentified | 5.0 | 0.0 | 6.9 | 0.0 | 0.0 |
| Composition of pyrolysis liquids (area%)—organic phase | |||||
| Aniline | 28.4 | 35.3 | 19.3 | 27.5 | 23.1 |
| Other N-aromatics | 11.8 | 2.2 | 29.8 | 33.5 | 38.8 |
| Phenol | 24.7 | 24.3 | n.d. 5 | n.d.5 | n.d. 5 |
| Other O-aromatics | 28.0 | 12.9 | n.d. 5 | 1.6 | 3.7 |
| PAH 6 | 0.0 | 0.0 | 28.1 | 35.5 | 29.7 |
| Others 7 | 4.3 | 1.9 | 22.8 | 1.9 | 3.2 |
| Unidentified | 2.8 | 23.4 | 0.0 | 0.0 | 1.5 |
| E1 | E2 | |
|---|---|---|
| Proximate analysis | ||
| Moisture | 0.8 | 0.9 |
| Volatiles | 7.2 | 7.0 |
| Fixed carbon 1 | 91.9 | 92.0 |
| Ash | 0.1 | 0.1 |
| Elemental analysis | ||
| C | 91.2 | 91.2 |
| H | 0.8 | 0.4 |
| N | 5.4 | 5.3 |
| S | <0.1 | <0.1 |
| Others 2 | 2.5 | 3.0 |
| Parameter | E1 Solid | E2 Solid | Initial CFRP |
|---|---|---|---|
| Arithmetical mean deviation, Ra (µm) | 1.69 ± 0.27 | 1.84 ± 0.19 | 1.23 ± 0.34 |
| Maximum peak height, Rp (µm) | 5.60 ± 1.03 | 6.88 ± 1.46 | 7.82 ± 3.83 |
| Maximum valley depth, Rv (µm) | −6.52 ± 1.27 | −6.98 ± 1.80 | −6.46 ± 2.65 |
| Property | Reforming Catalyst | Ni/HZSM-5 |
|---|---|---|
| BET surface area (m2/g) | 29.0 | 273.9 |
| Total pore volume (cm3/g) | 0.1312 | 0.3887 |
| Average pore diameter (Å) | 179.3 | 70.7 |
| Measured Ni content (wt.%) | 12.5 | 11.0 |
| Ni crystal size (nm) | 5.0 | 4.5 |
| Main reduction peak temperature (°C) | ≈700 | ≈350 |
| Total acidity (mmol NH3/g cat) | 0.205 | 0.471 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gastelu, N.; Lopez-Urionabarrenechea, A.; Solar, J.; Acha, E.; Caballero, B.M.; López, F.A.; De Marco, I. Thermo-Catalytic Treatment of Vapors in the Recycling Process of Carbon Fiber-Poly (Benzoxazine) Composite Waste by Pyrolysis. Catalysts 2018, 8, 523. https://doi.org/10.3390/catal8110523
Gastelu N, Lopez-Urionabarrenechea A, Solar J, Acha E, Caballero BM, López FA, De Marco I. Thermo-Catalytic Treatment of Vapors in the Recycling Process of Carbon Fiber-Poly (Benzoxazine) Composite Waste by Pyrolysis. Catalysts. 2018; 8(11):523. https://doi.org/10.3390/catal8110523
Chicago/Turabian StyleGastelu, Naia, Alexander Lopez-Urionabarrenechea, Jon Solar, Esther Acha, Blanca María Caballero, Félix A. López, and Isabel De Marco. 2018. "Thermo-Catalytic Treatment of Vapors in the Recycling Process of Carbon Fiber-Poly (Benzoxazine) Composite Waste by Pyrolysis" Catalysts 8, no. 11: 523. https://doi.org/10.3390/catal8110523
APA StyleGastelu, N., Lopez-Urionabarrenechea, A., Solar, J., Acha, E., Caballero, B. M., López, F. A., & De Marco, I. (2018). Thermo-Catalytic Treatment of Vapors in the Recycling Process of Carbon Fiber-Poly (Benzoxazine) Composite Waste by Pyrolysis. Catalysts, 8(11), 523. https://doi.org/10.3390/catal8110523






