Ammonia Plasma-Catalytic Synthesis Using Low Melting Point Alloys
Abstract
1. Introduction
2. Results and Discussion
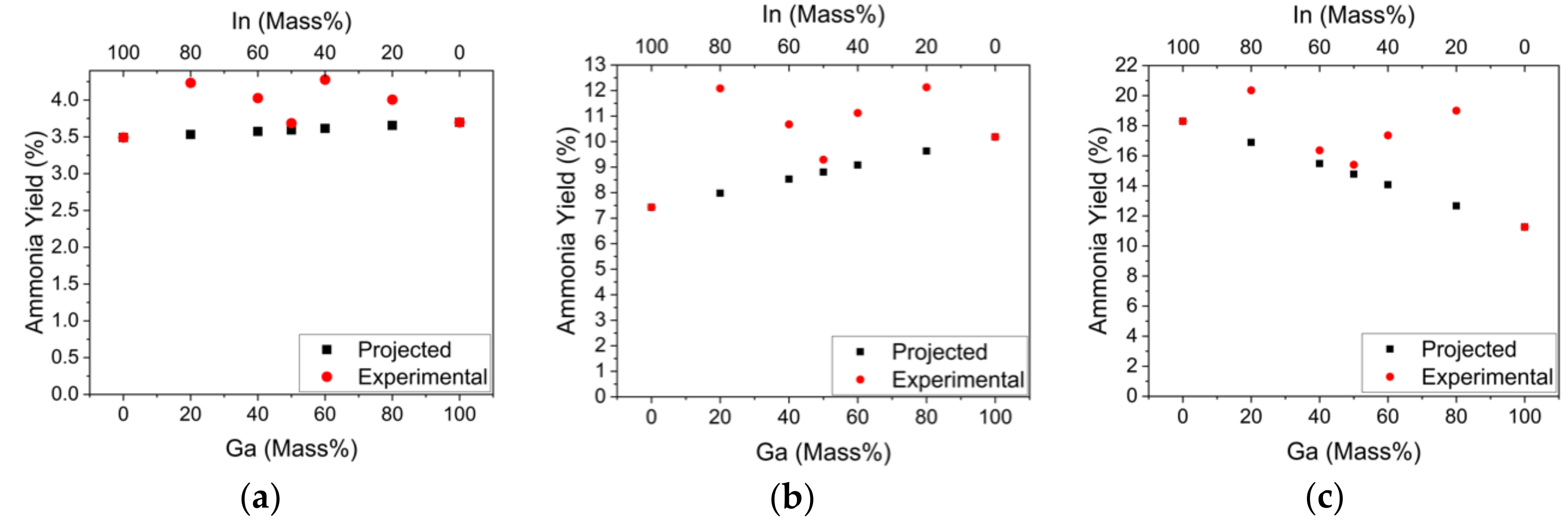
2.1. Catalytic Activity for Ammonia Synthesis
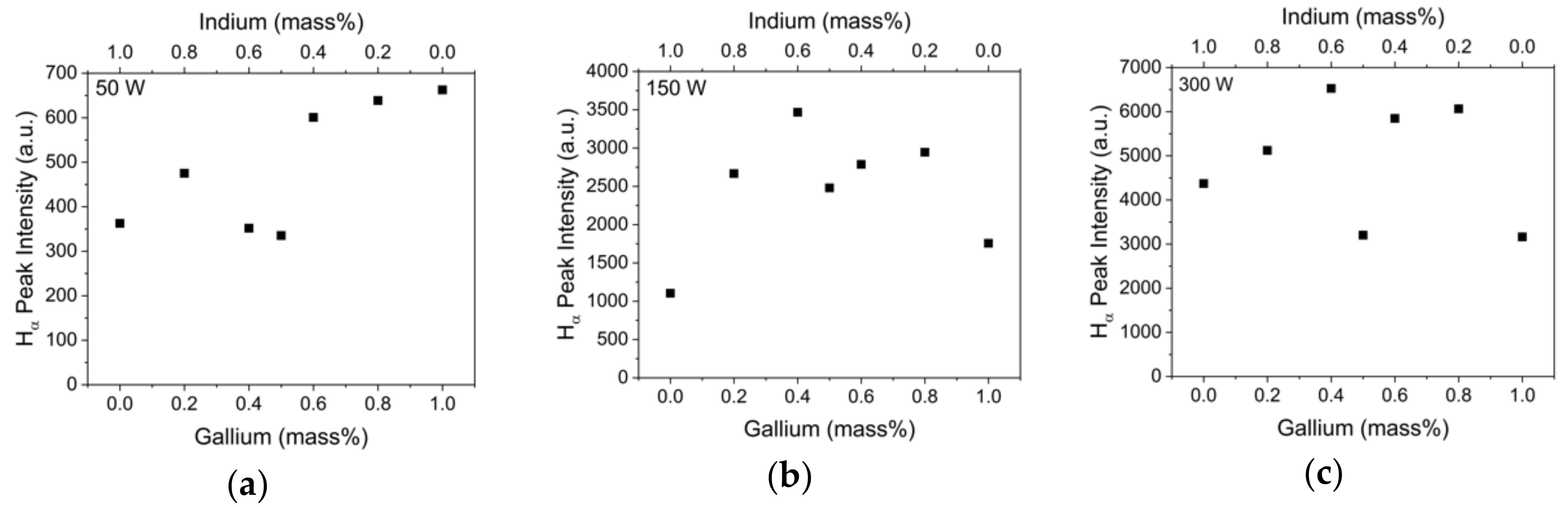
2.2. Emission Spectroscopy of Plasma
2.3. Characterization of Alloys
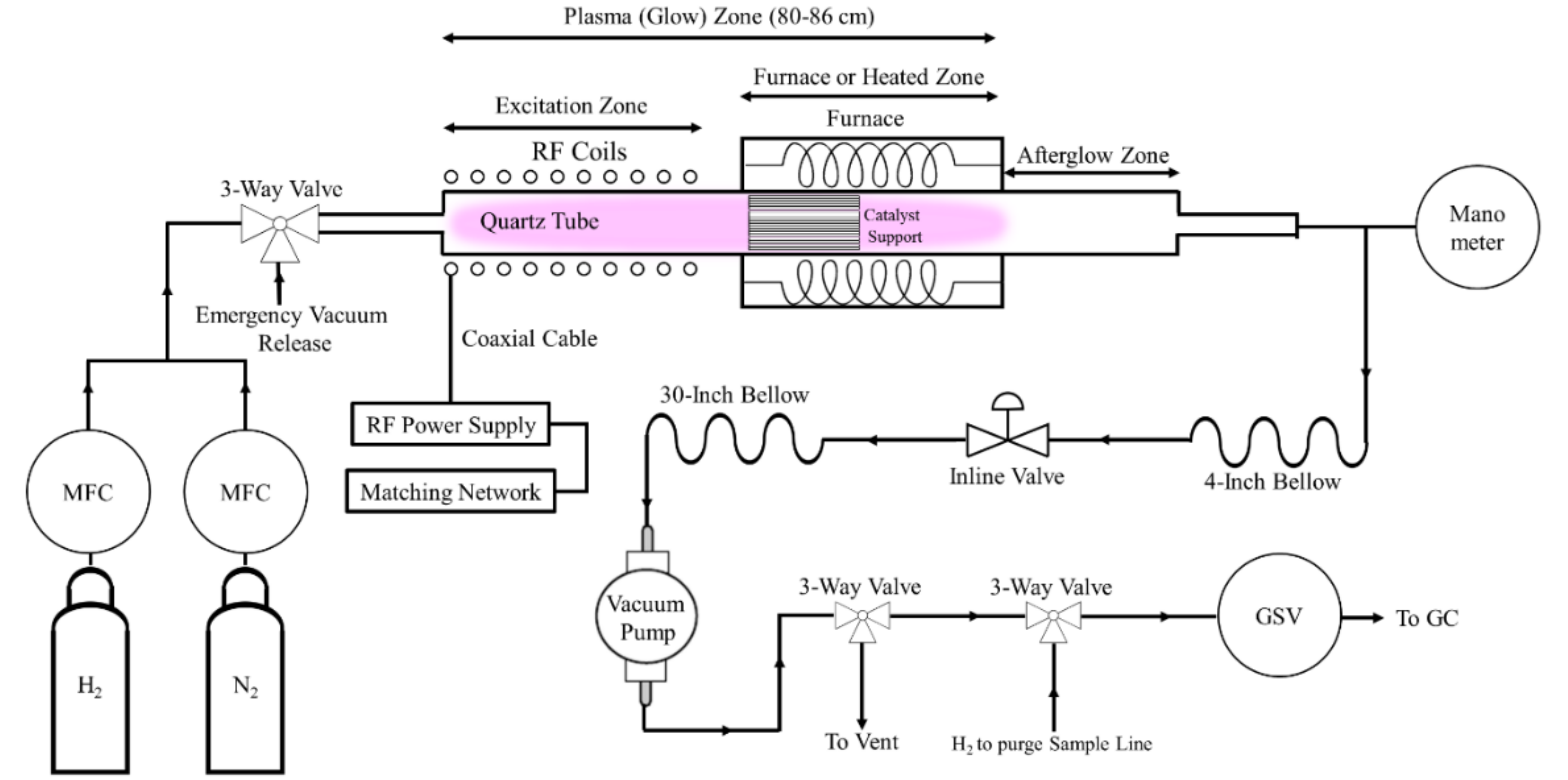
3. Materials and Methods
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Tanabe, Y.; Nishibayashi, Y. Developing more sustainable processes for ammonia synthesis. Coord. Chem. Rev. 2013, 257, 2551–2564. [Google Scholar] [CrossRef]
- Global Ammonia Capacity to Reach Almost 250 Million Tons per Year by 2018, says GlobalData. Available online: https://energy.globaldata.com/media-center/press-releases/oil-and-gas/global-ammonia-capacity-to-reach-almost-250-million-tons-per-year-by-2018-says-globaldata (accessed on 15 July 2018).
- Smil, V. Enriching the Earth: Fritz Haber, Carl Bosch, and the Transformation of World Food Production; MIT Press: Cambridge, MA, USA, 2004. [Google Scholar]
- Kiefer, D.M. Capturing Nitrogen Out of the Air Fritz Haber’s high-pressure process for combining nitrogen with hydrogen was a major milestone. Todays Chem. Work 2001, 10, 117–122. [Google Scholar]
- Jennings, J.R. Catalytic Ammonia Synthesis: Fundamentals and Practice; Springer: Berlin, Germany, 2013. [Google Scholar]
- Modak, J.M. Haber process for ammonia synthesis. Resonance 2002, 7, 69–77. [Google Scholar] [CrossRef]
- Emmett, P.; Brunauer, S. Accumulation of alkali promoters on surfaces of iron synthetic ammonia catalysts. J. Am. Chem. Soc. 1937, 59, 310–315. [Google Scholar] [CrossRef]
- Hong, J.; Prawer, S.; Murphy, A.B. Plasma catalysis as an alternative route for ammonia production: Status, mechanisms, and prospects for progress. ACS Sustain. Chem. Eng. 2017, 6, 15–31. [Google Scholar] [CrossRef]
- Bogaerts, A.; Neyts, E.C. Plasma Technology: An Emerging Technology for Energy Storage. ACS Energy Lett. 2018, 3, 1013–1027. [Google Scholar] [CrossRef]
- Patil, B. Plasma (Catalyst)-Assisted Nitrogen Fixation: Reactor Development for Nitric Oxide and Ammonia Production. Ph.D. Thesis, Eindhoven University of Technology, Eindhoven, The Netherlands, 2017. [Google Scholar]
- Aihara, K.; Akiyama, M.; Deguchi, T.; Tanaka, M.; Hagiwara, R.; Iwamoto, M. Remarkable catalysis of a wool-like copper electrode for NH3 synthesis from N2 and H2 in non-thermal atmospheric plasma. Chem. Commun. 2016, 52, 13560–13563. [Google Scholar] [CrossRef] [PubMed]
- Iwamoto, M.; Akiyama, M.; Aihara, K.; Deguchi, T. Ammonia synthesis on wool-like Au, Pt, Pd, Ag, or Cu electrode catalysts in nonthermal atmospheric-pressure plasma of N2 and H2. ACS Catal. 2017, 7, 6924–6929. [Google Scholar] [CrossRef]
- Kim, H.H.; Teramoto, Y.; Ogata, A.; Takagi, H.; Nanba, T. Atmospheric-pressure nonthermal plasma synthesis of ammonia over ruthenium catalysts. Plasma Process. Polym. 2017, 14, 1600157. [Google Scholar] [CrossRef]
- Hong, J.; Pancheshnyi, S.; Tam, E.; Lowke, J.J.; Prawer, S.; Murphy, A.B. Kinetic modelling of NH3 production in N2–H2 non-equilibrium atmospheric-pressure plasma catalysis. J. Phys. D Appl. Phys. 2017, 50, 154005. [Google Scholar] [CrossRef]
- Mizushima, T.; Matsumoto, K.; Ohkita, H.; Kakuta, N. Catalytic effects of metal-loaded membrane-like alumina tubes on ammonia synthesis in atmospheric pressure plasma by dielectric barrier discharge. Plasma Chem. Plasma Process. 2007, 27, 1–11. [Google Scholar] [CrossRef]
- Akay, G.; Zhang, K. Process Intensification in Ammonia Synthesis Using Novel Coassembled Supported Microporous Catalysts Promoted by Nonthermal Plasma. Ind. Eng. Chem. Res. 2017, 56, 457–468. [Google Scholar] [CrossRef]
- Peng, P.; Cheng, Y.; Hatzenbeller, R.; Addy, M.; Zhou, N.; Schiappacasse, C.; Chen, D.; Zhang, Y.; Anderson, E.; Liu, Y. Ru-Based Multifunctional Mesoporous Catalyst for Low-Pressure and Non-Thermal Plasma Synthesis of Ammonia. Int. J. Hydrogen Energy 2017, 42, 19056–19066. [Google Scholar] [CrossRef]
- Tanaka, S.; Uyama, H.; Matsumoto, O. Synergistic effects of catalysts and plasmas on the synthesis of ammonia and hydrazine. Plasma Chem. Plasma Process. 1994, 14, 491–504. [Google Scholar] [CrossRef]
- Mingdong, B.; Xiyao, B.; Zhitao, Z.; Mindi, B. Synthesis of Ammonia in a Strong Electric Field Discharge at Ambient Pressure. Plasma Chem. Plasma Process. 2000, 20, 511–520. [Google Scholar] [CrossRef]
- Sugiyama, K.; Akazawa, K.; Oshima, M.; Miura, H.; Matsuda, T.; Nomura, O. Ammonia Synthesis by Means of Plasma over MgO Catalyst. Plasma Chem. Plasma Process. 1986, 6, 179–193. [Google Scholar] [CrossRef]
- Peng, P.; Li, Y.; Cheng, Y.; Deng, S.; Chen, P.; Ruan, R. Atmospheric Pressure Ammonia Synthesis using Non-Thermal Plasma Assisted Catalysis. Plasma Chem. Plasma Process. 2016, 36, 1201–1210. [Google Scholar] [CrossRef]
- Uyama, H.; Matsumoto, O. Synthesis of ammonia in high-frequency discharges. II. Synthesis of ammonia in a microwave discharge under various conditions. Plasma Chem. Plasma Process. 1989, 9, 421–432. [Google Scholar] [CrossRef]
- Uyama, H.; Matsumoto, O. Synthesis of ammonia in high-frequency discharges. Plasma Chem. Plasma Process. 1989, 9, 13–24. [Google Scholar] [CrossRef]
- Uyama, H.; Nakamura, T.; Tanaka, S.; Matsumoto, O. Catalytic effect of iron wires on the syntheses of ammonia and hydrazine in a radio-frequency discharge. Plasma Chem. Plasma Process. 1993, 13, 117–131. [Google Scholar] [CrossRef]
- Mehta, P.; Barboun, P.; Herrera, F.A.; Kim, J.; Rumbach, P.; Go, D.B.; Hicks, J.C.; Schneider, W.F. Overcoming ammonia synthesis scaling relations with plasma-enabled catalysis. Nat. Catal. 2018, 1, 269–275. [Google Scholar] [CrossRef]
- Singh, A.R.; Montoya, J.H.; Rohr, B.A.; Tsai, C.; Vojvodic, A.; Nørskov, J.K. Computational Design of Active Site Structures with Improved Transition-State Scaling for Ammonia Synthesis. ACS Catal. 2018, 8, 4017–4024. [Google Scholar] [CrossRef]
- Murray, R. Method of Producing Finely-Divided Nickel. U.S. Patents 1,628,190, 10 May 1927. [Google Scholar]
- Raney, M. Catalysts from alloys. Ind. Eng. Chem. 1940, 32, 1199–1203. [Google Scholar] [CrossRef]
- Schwab, G.M. Catalysis on Liquids Metals. Berichte der Bunsengesellschaft für physikalische Chemie 1976, 80, 746–749. [Google Scholar] [CrossRef]
- Ozawa, S.; Sasaki, K.; Ogino, Y. Reaction of benzyl phenyl ether over various molten metal catalysts. Fuel 1986, 65, 707–710. [Google Scholar] [CrossRef]
- Okano, K.; Saito, Y.; Ogino, Y. The Dehydrogenation of Amines by Molten Metal Catalysts. Bull. Chem. Soc. Jpn. 1972, 45, 69–73. [Google Scholar] [CrossRef]
- Saito, Y.; Hiramatsu, N.; Kawanami, N.; Ogino, Y. Dehydrogenation of Some Alcohols by the Molten Metal Catalysts. Bull. Jpn. Pet. Inst. 1972, 14, 169–173. [Google Scholar] [CrossRef]
- Saito, Y.; Miyashita, F.; Ogino, Y. Studies on catalysis by molten metal: VI. Kinetics and the reaction scheme for the dehydrogenation of isopropyl alcohol over the liquid indium catalyst. J. Catal. 1975, 36, 67–73. [Google Scholar] [CrossRef]
- Stelmachowski, M. Thermal conversion of waste polyolefins to the mixture of hydrocarbons in the reactor with molten metal bed. Energy Convers. Manag. 2010, 51, 2016–2024. [Google Scholar] [CrossRef]
- Stelmachowski, M. Conversion of waste rubber to the mixture of hydrocarbons in the reactor with molten metal. Energy Convers. Manag. 2009, 50, 1739–1745. [Google Scholar] [CrossRef]
- Upham, D.C.; Agarwal, V.; Khechfe, A.; Snodgrass, Z.R.; Gordon, M.J.; Metiu, H.; McFarland, E.W. Catalytic molten metals for the direct conversion of methane to hydrogen and separable carbon. Science 2017, 358, 917–921. [Google Scholar] [CrossRef] [PubMed]
- Mukanova, A.; Tussupbayev, R.; Sabitov, A.; Bondarenko, I.; Nemkaeva, R.; Aldamzharov, B.; Bakenov, Z. CVD graphene growth on a surface of liquid gallium. Mater. Today Proc. 2017, 4, 4548–4554. [Google Scholar] [CrossRef]
- Fujita, J.-I.; Hiyama, T.; Hirukawa, A.; Kondo, T.; Nakamura, J.; Ito, S.-I.; Araki, R.; Ito, Y.; Takeguchi, M.; Pai, W.W. Near room temperature chemical vapor deposition of graphene with diluted methane and molten gallium catalyst. Sci. Rep. 2017, 7, 12371. [Google Scholar] [CrossRef] [PubMed]
- Fujita, J.-I.; Miyazawa, Y.; Ueki, R.; Sasaki, M.; Saito, T. Fabrication of large-area graphene using liquid gallium and its electrical properties. Jpn. J. Appl. Phys. 2010, 49, 06GC01. [Google Scholar] [CrossRef]
- Lee, M.V.; Hiura, H.; Tyurnina, A.V.; Tsukagoshi, K. Controllable gallium melt-assisted interfacial graphene growth on silicon carbide. Diam. Relat. Mater. 2012, 24, 34–38. [Google Scholar] [CrossRef]
- Lugstein, A.; Steinmair, M.; Hyun, Y.J.; Bertagnolli, E.; Pongratz, P. Ga/Au alloy catalyst for single crystal silicon-nanowire epitaxy. Appl. Phys. Lett. 2007, 90, 023109. [Google Scholar] [CrossRef]
- Gewalt, A.; Kalkofen, B.; Lisker, M.; Burte, E.P. Gallium Assisted PECVD Synthesis of Silicon Nanowires. ECS Trans. 2010, 28, 45–56. [Google Scholar]
- Pan, Z.W.; Dai, Z.R.; Ma, C.; Wang, Z.L. Molten gallium as a catalyst for the large-scale growth of highly aligned silica nanowires. J. Am. Chem. Soc. 2002, 124, 1817–1822. [Google Scholar] [CrossRef] [PubMed]
- Carreon, M.L.; Jasinski, J.; Sunkara, M. Low temperature synthesis of silicon nanowire arrays. Mater. Res. Express 2014, 1, 045006. [Google Scholar] [CrossRef]
- Carreon, M. Plasma catalysis using low melting point metals. Ph.D. Thesis, University of Louisville, Louisville, KY, USA, 2015. [Google Scholar]
- Carreon, M.L.; Jaramillo-Cabanzo, D.F.; Chaudhuri, I.; Menon, M.; Sunkara, M.K. Synergistic interactions of H2 and N2 with molten gallium in the presence of plasma. J. Vac. Sci. Technol. A 2018, 36, 021303. [Google Scholar] [CrossRef]
- Anderson, T.; Ansara, I. The Ga-In (gallium-indium) system. J. Phase Equilib. 1991, 12, 64–72. [Google Scholar] [CrossRef]
- Daeneke, T.; Khoshmanesh, K.; Mahmood, N.; de Castro, I.; Esrafilzadeh, D.; Barrow, S.; Dickey, M.; Kalantar-Zadeh, K. Liquid metals: Fundamentals and applications in chemistry. Chem. Soc. Rev. 2018, 47, 4073–4111. [Google Scholar] [CrossRef] [PubMed]
- Shah, J.; Wang, W.; Bogaerts, A.; Carreon, M.L. Ammonia synthesis by radio frequency plasma catalysis: Revealing the underlying mechanisms. ACS Appl. Energy Mater. 2018, 1, 4824–4839. [Google Scholar] [CrossRef]
- Nabi, G.; Cao, C.; Khan, W.S.; Hussain, S.; Usman, Z.; Mahmood, T.; Khattak, N.A.D.; Zhao, S.; Xin, X.; Yu, D. Synthesis, characterization, photoluminescence and field emission properties of novel durian-like gallium nitride microstructures. Mater. Chem. Phys. 2012, 133, 793–798. [Google Scholar] [CrossRef]

| Gallium Weight % | Indium Weight % | Melting Point (°C) 1 |
|---|---|---|
| 100 | 0 | 29.8 |
| 80 | 20 | 16.3 |
| 60 | 40 | 50.2 |
| 50 | 50 | 61 |
| 40 | 60 | 65 |
| 20 | 80 | 90 |
| 0 | 100 | 156.6 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shah, J.R.; Harrison, J.M.; Carreon, M.L. Ammonia Plasma-Catalytic Synthesis Using Low Melting Point Alloys. Catalysts 2018, 8, 437. https://doi.org/10.3390/catal8100437
Shah JR, Harrison JM, Carreon ML. Ammonia Plasma-Catalytic Synthesis Using Low Melting Point Alloys. Catalysts. 2018; 8(10):437. https://doi.org/10.3390/catal8100437
Chicago/Turabian StyleShah, Javishk R., Joshua M. Harrison, and Maria L. Carreon. 2018. "Ammonia Plasma-Catalytic Synthesis Using Low Melting Point Alloys" Catalysts 8, no. 10: 437. https://doi.org/10.3390/catal8100437
APA StyleShah, J. R., Harrison, J. M., & Carreon, M. L. (2018). Ammonia Plasma-Catalytic Synthesis Using Low Melting Point Alloys. Catalysts, 8(10), 437. https://doi.org/10.3390/catal8100437





