Promotion of Ca-Co Bifunctional Catalyst/Sorbent with Yttrium for Hydrogen Production in Modified Chemical Looping Steam Methane Reforming Process
Abstract
1. Introduction
2. Results and Discussion
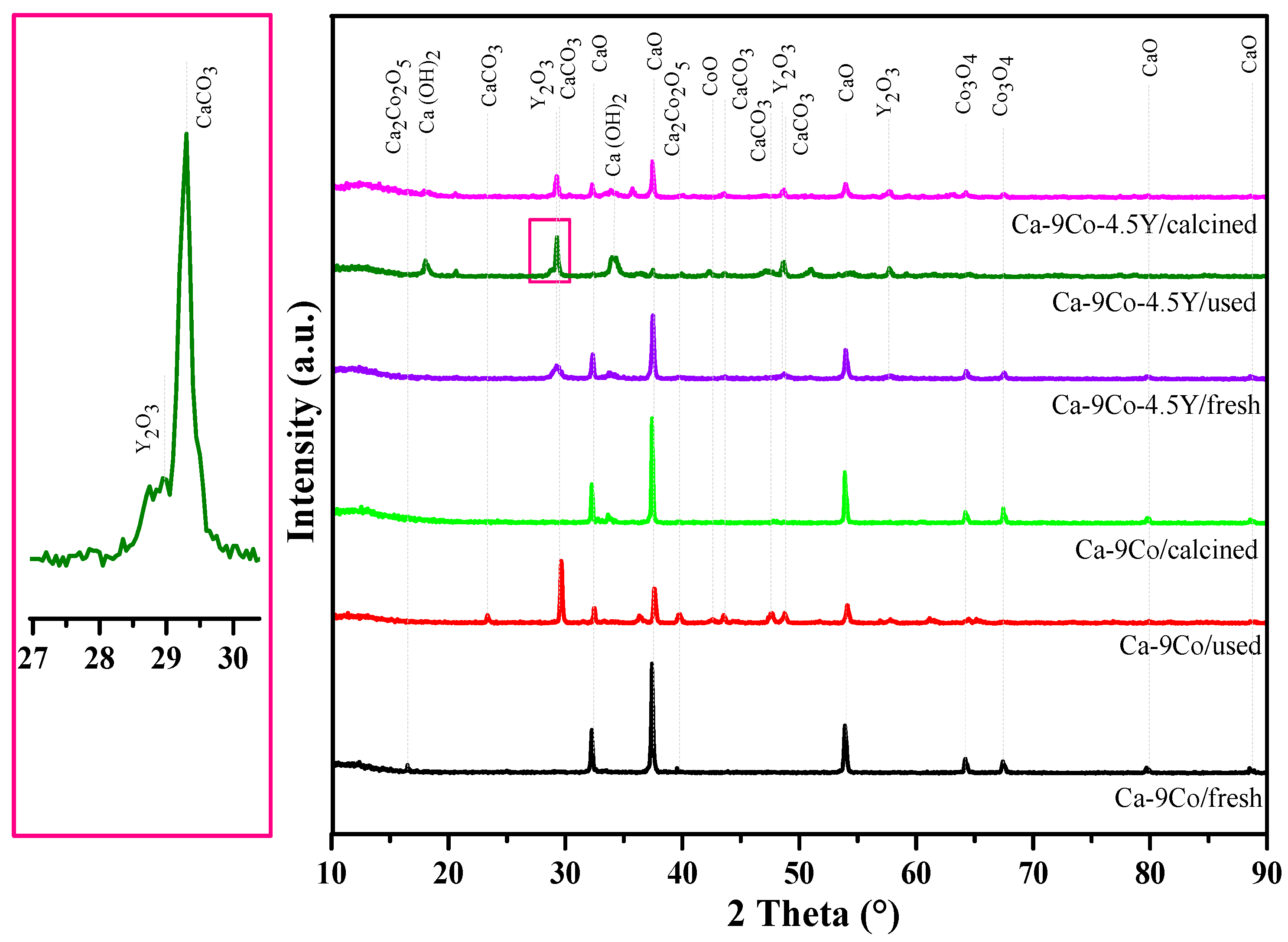
2.1. Sample Characterization
2.2. Catalytic Activity of Ca-mCo
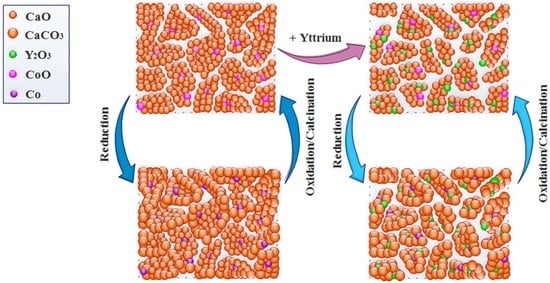
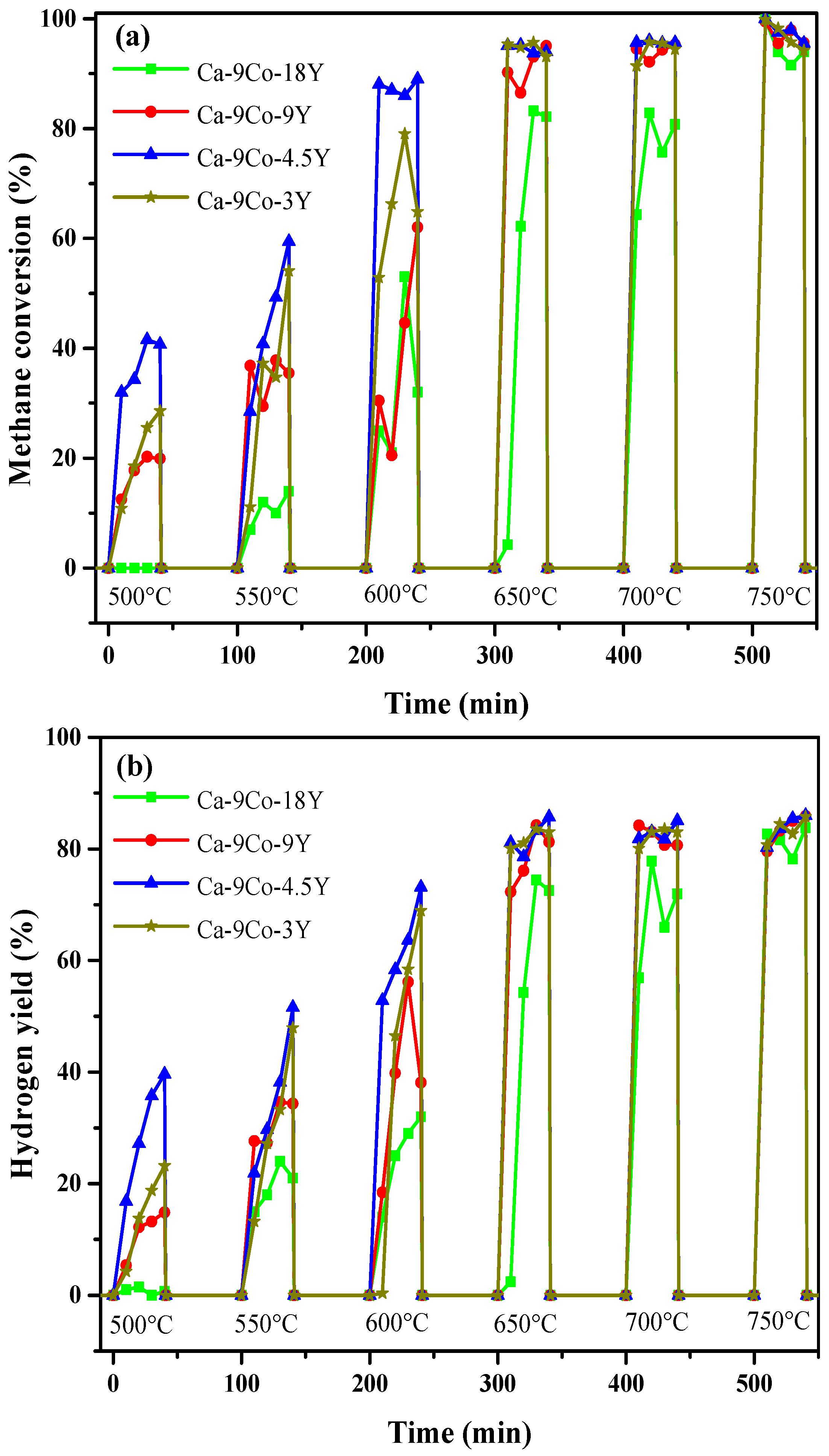
2.3. Effect of Yttrium Promoter on Catalytic Activity of Ca-9Co-nY Catalyst/Sorbent
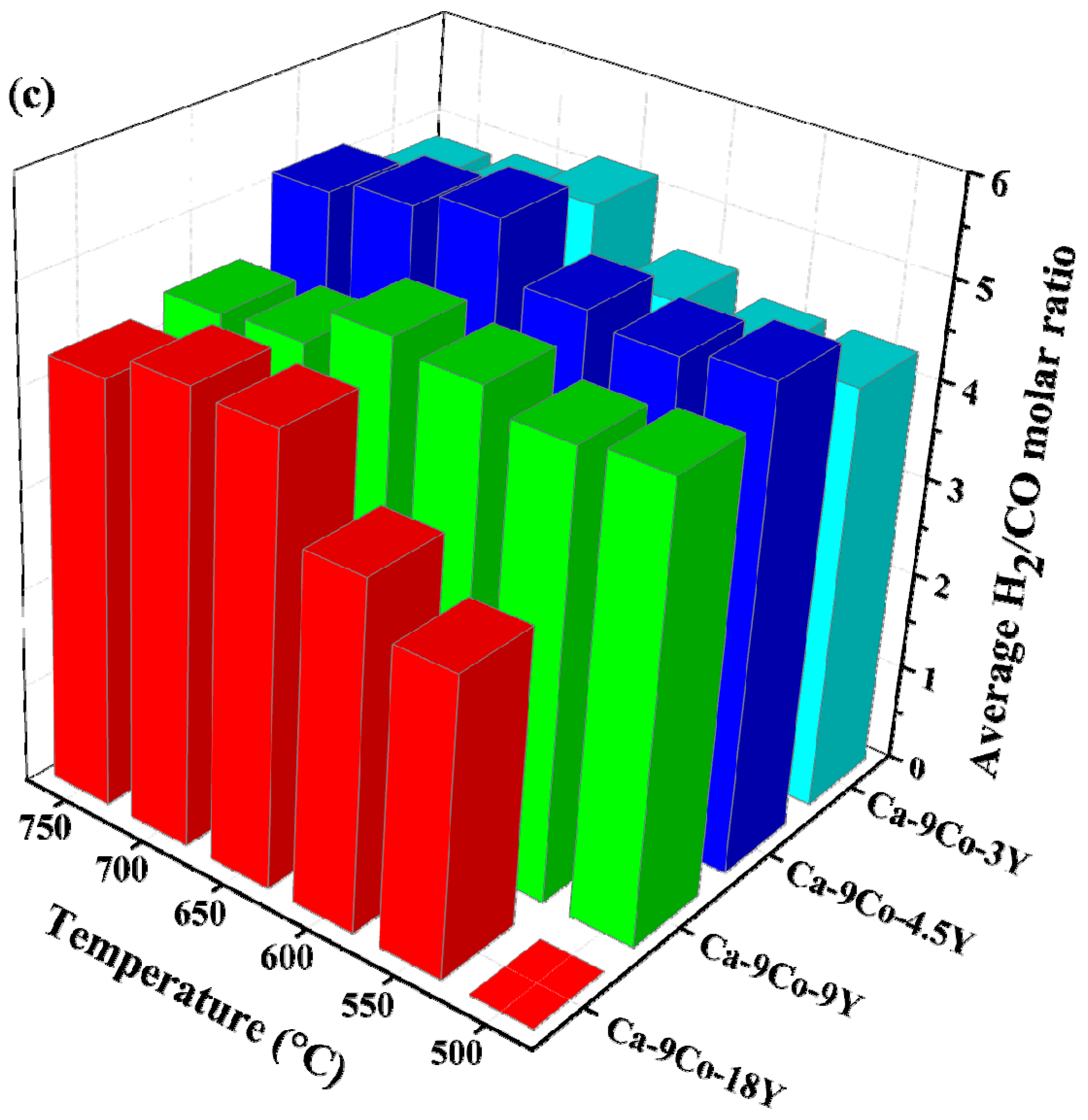
2.4. CO2 Concentration in the Reaction Media
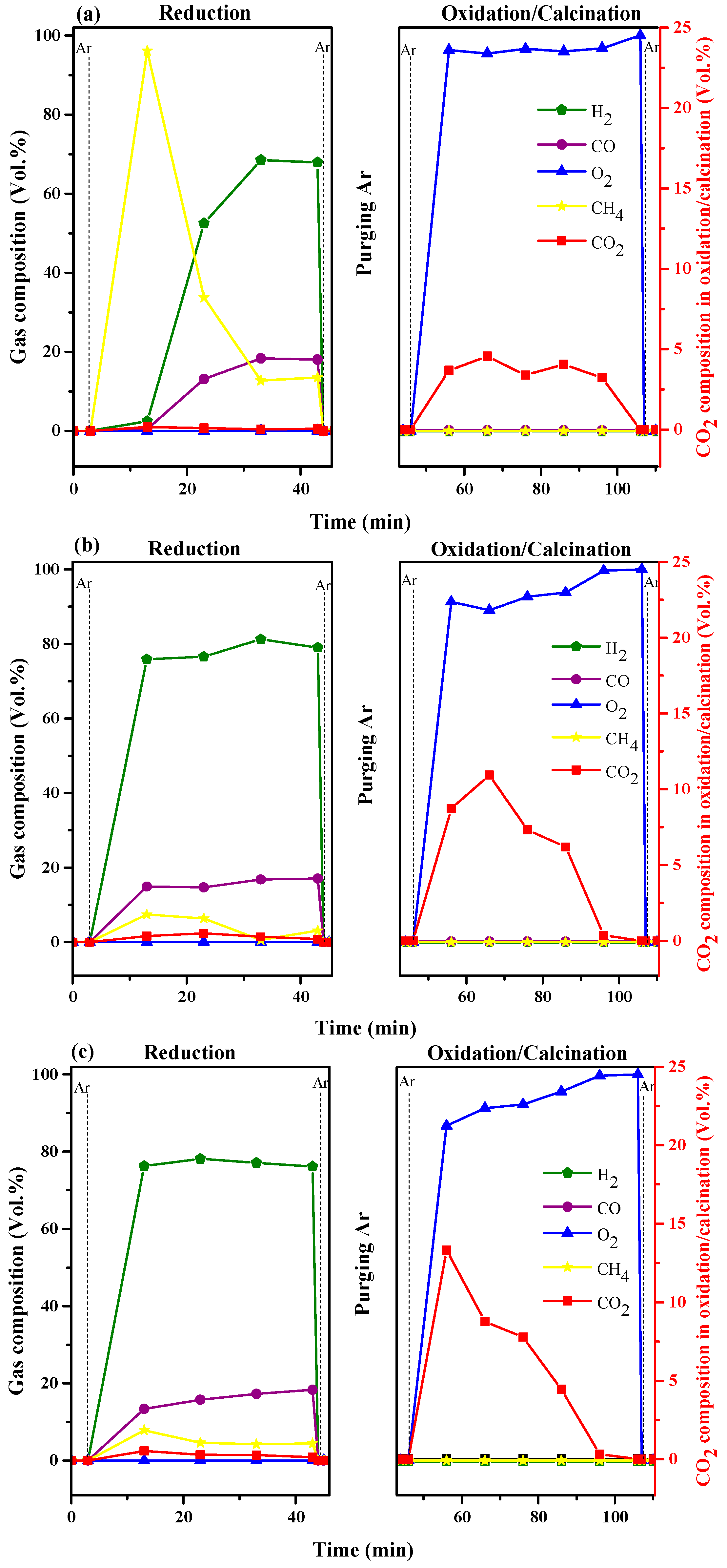
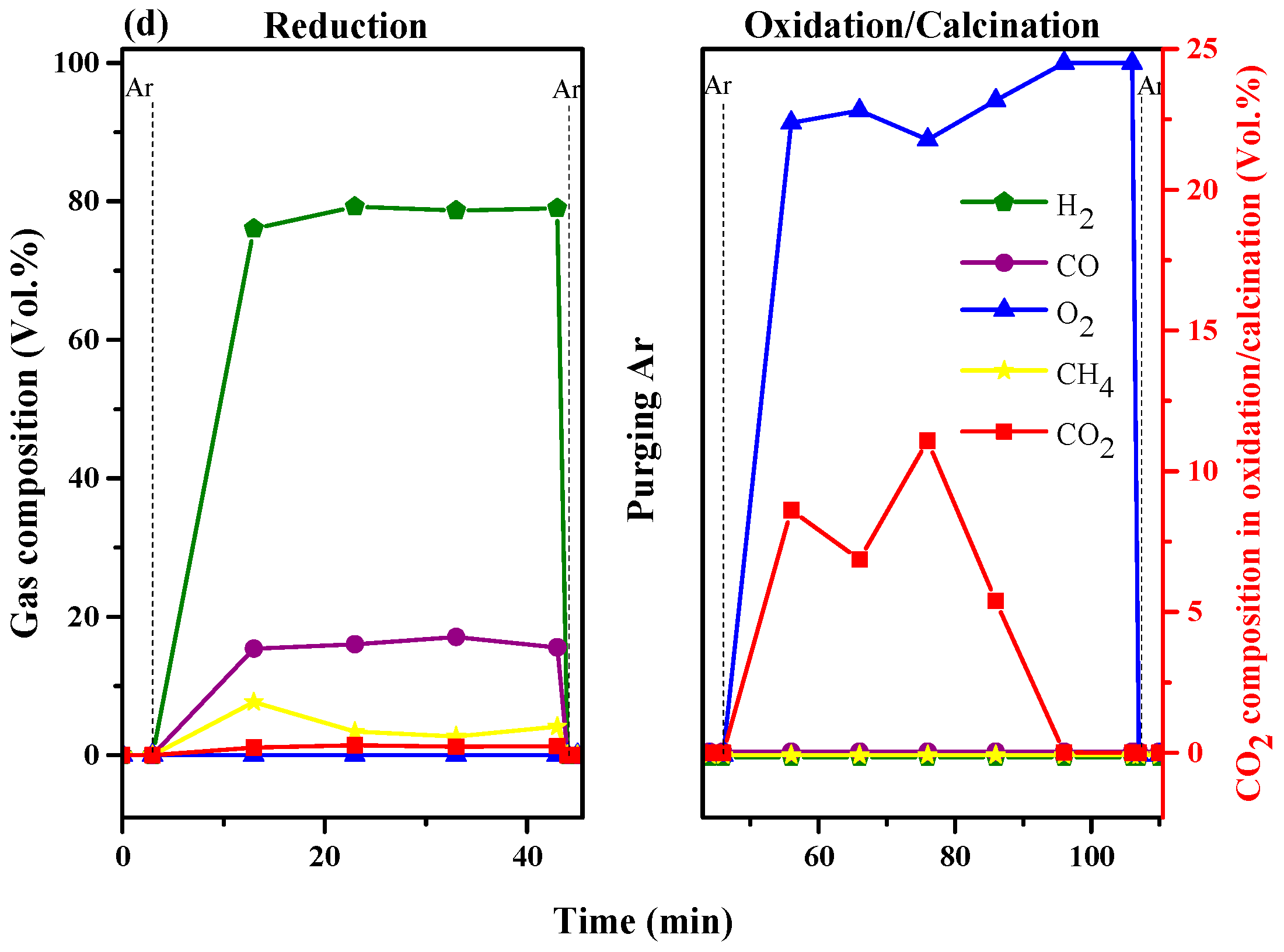
2.5. Life Time of Ca-9Co and Ca-9Co-4.5Y Bifunctional Catalyst/Sorbents
2.6. Characterization of Used and Regenerated Catalyst/Sorbents
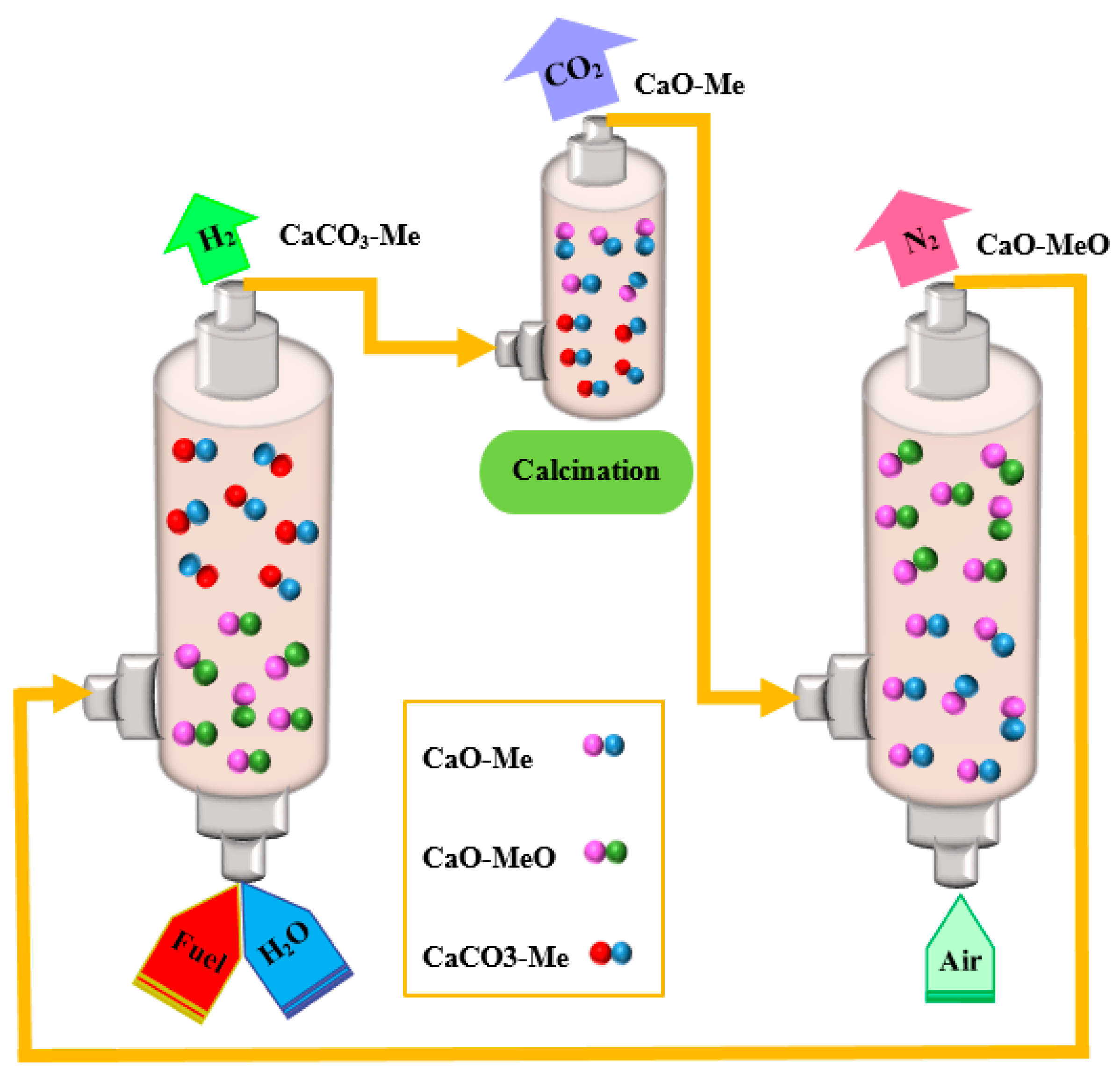
3. Experimental Methods
3.1. Preparation of Bifunctional Catalyst/Sorbent
3.2. Samples Characterization
3.3. Catalytic Evaluation
4. Conclusions
Author Contributions
Conflicts of Interest
References
- Abánades, A.; Ruiz, E.; Ferruelo, E.; Hernández, F.; Cabanillas, A.; Martínez-Val, J.; Rubio, J.A.; López, C.; Gavela, R.; Barrera, G.; et al. Experimental analysis of direct thermal methane cracking. Int. J. Hydrogen Energy 2011, 36, 12877–12886. [Google Scholar] [CrossRef]
- Jabbour, K.; Massiani, P.; Davidson, A.; Casale, S.; El Hassan, N. Ordered mesoporous “one-pot” synthesized Ni-Mg(Ca)-Al2O3 as effective and remarkably stable catalysts for combined steam and dry reforming of methane (csdrm). Appl. Catal. B 2016. [Google Scholar] [CrossRef]
- Abbasi, M.; Farniaei, M.; Rahimpour, M.R.; Shariati, A. Enhancement of hydrogen production and carbon dioxide capturing in a novel methane steam reformer coupled with chemical looping combustion and assisted by hydrogen perm-selective membranes. Energy Fuels 2013, 27, 5359–5372. [Google Scholar] [CrossRef]
- Wang, Y.; Chen, M.; Liang, T.; Yang, Z.; Yang, J.; Liu, S. Hydrogen generation from catalytic steam reforming of acetic acid by Ni/attapulgite catalysts. Catalysts 2016, 6, 172. [Google Scholar] [CrossRef]
- Antzara, A.; Heracleous, E.; Lemonidou, A.A. Energy efficient sorption enhanced-chemical looping methane reforming process for high-purity H2 production: Experimental proof-of-concept. Appl. Energy 2016, 180, 457–471. [Google Scholar] [CrossRef]
- Antzara, A.; Heracleous, E.; Silvester, L.; Bukur, D.B.; Lemonidou, A.A. Activity study of NiO-based oxygen carriers in chemical looping steam methane reforming. Catal. Today 2016, 272, 32–41. [Google Scholar] [CrossRef]
- Khan, M.N.; Shamim, T. Investigation of hydrogen generation in a three reactor chemical looping reforming process. Appl. Energy 2016, 162, 1186–1194. [Google Scholar] [CrossRef]
- Ni, M.; Leung, M.K.; Sumathy, K.; Leung, D.Y. Potential of renewable hydrogen production for energy supply in Hong Kong. Int. J. Hydrogen Energy 2006, 31, 1401–1412. [Google Scholar] [CrossRef]
- Forutan, H.R.; Karimi, E.; Hafizi, A.; Rahimpour, M.R.; Keshavarz, P. Expert representation chemical looping reforming: A comparative study of Fe, Mn, Co and Cu as oxygen carriers supported on Al2O3. J. Ind. Eng. Chem. 2015, 21, 900–911. [Google Scholar] [CrossRef]
- Zhou, C.; Shah, K.; Doroodchi, E.; Moghtaderi, B. Equilibrium thermodynamic analyses of methanol production via a novel chemical looping carbon arrestor process. Energy Convers. Manag. 2015, 96, 392–402. [Google Scholar] [CrossRef]
- Yahom, A.; Powell, J.; Pavarajarn, V.; Onbhuddha, P.; Charojrochkul, S.; Assabumrungrat, S. Simulation and thermodynamic analysis of chemical looping reforming and CO2 enhanced chemical looping reforming. Chem. Eng. Res. Des. 2014, 92, 2575–2583. [Google Scholar] [CrossRef]
- Hafizi, A.; Jafari, M.; Rahimpour, M.R.; Hassanajili, S. Experimental investigation of sorption enhanced chemical looping reforming for high purity hydrogen production using CeO2-CaO CO2 sorbent and 15Fe-5Ca/Al2O3 oxygen carrier. J. Taiwan Inst. Chem. Eng. 2016. [Google Scholar] [CrossRef]
- Ghasemzadeh, K.; Liguori, S.; Morrone, P.; Iulianelli, A.; Piemonte, V.; Babaluo, A.A.; Basile, A. H2 production by low pressure methanol steam reforming in a dense Pd-Ag membrane reactor in CO-current flow configuration: Experimental and modeling analysis. Int. J. Hydrogen Energy 2013, 38, 16685–16697. [Google Scholar] [CrossRef]
- Sengupta, S.; Deo, G. Modifying alumina with CaO or MgO in supported Ni and Ni-CO catalysts and its effect on dry reforming of CH4. J. CO2 Util. 2015, 10, 67–77. [Google Scholar] [CrossRef]
- Agrawal, V.; Srivastava, V.C. Simulation of fluidized bed reactor for producing synthesis gas by catalytic CH4-CO2 reforming. J. CO2 Util. 2014, 5, 10–16. [Google Scholar] [CrossRef]
- Hafizi, A.; Rahimpour, M.R.; Hassanajili, S. Hydrogen production by chemical looping steam reforming of methane over Mg promoted iron oxygen carrier: Optimization using design of experiments. J. Taiwan Inst. Chem. Eng. 2016. [Google Scholar] [CrossRef]
- LeValley, T.L.; Richard, A.R.; Fan, M. Development of catalysts for hydrogen production through the integration of steam reforming of methane and high temperature water gas shift. Energy 2015, 90, 748–758. [Google Scholar] [CrossRef]
- Hafizi, A.; Rahimpour, M.R.; Hassanajili, S. High purity hydrogen production via sorption enhanced chemical looping reforming: Application of 22Fe2O3/MgAl2O4 and 22Fe2O3/Al2O3 as oxygen carriers and cerium promoted CaO as CO2 sorbent. Appl. Energy 2016, 169, 629–641. [Google Scholar] [CrossRef]
- Vozniuk, O.; Agnoli, S.; Artiglia, L.; Vassoi, A.; Tanchoux, N.; Di Renzo, F.; Granozzi, G.; Cavani, F. Towards an improved process for hydrogen production: The chemical-loop reforming of ethanol. Green Chem. 2016, 18, 1038–1050. [Google Scholar] [CrossRef]
- Dewoolkar, K.D.; Vaidya, P.D. Improved hydrogen production by sorption-enhanced steam methane reforming over hydrotalcite-and calcium-based hybrid materials. Energy Fuels 2015, 29, 3870–3878. [Google Scholar] [CrossRef]
- Freitas, A.C.D.; Guirardello, R. Thermodynamic analysis of methane reforming with CO2, CO2 + H2O, CO2 + O2 and CO2 + air for hydrogen and synthesis gas production. J. CO2 Util. 2014, 7, 30–38. [Google Scholar] [CrossRef]
- Du, Y.-L.; Wu, X.; Cheng, Q.; Huang, Y.-L.; Huang, W. Development of Ni-based catalysts derived from hydrotalcite-like compounds precursors for synthesis gas production via methane or ethanol reforming. Catalysts 2017, 7, 70. [Google Scholar] [CrossRef]
- Xu, P.; Zhou, Z.; Zhao, C.; Cheng, Z. Catalytic performance of Ni/CaO-Ca5Al6O14 bifunctional catalyst extrudate in sorption-enhanced steam methane reforming. Catal. Today 2016, 259, 347–353. [Google Scholar] [CrossRef]
- Farniaei, M.; Abbasi, M.; Rahnama, H.; Rahimpour, M.R.; Shariati, A. Syngas production in a novel methane dry reformer by utilizing of tri-reforming process for energy supplying: Modeling and simulation. J. Nat. Gas Sci. Eng. 2014, 20, 132–146. [Google Scholar] [CrossRef]
- Hafizi, A.; Rahimpour, M.R.; Hassanajili, S. Calcium promoted Fe/Al2O3 oxygen carrier for hydrogen production via cyclic chemical looping steam methane reforming process. Int. J. Hydrogen Energy 2015, 40, 16159–16168. [Google Scholar] [CrossRef]
- Karimi, E.; Forutan, H.; Saidi, M.; Rahimpour, M.R.; Shariati, A. Experimental study of chemical-looping reforming in a fixed-bed reactor: Performance investigation of different oxygen carriers on Al2O3 and TiO2 support. Energy Fuels 2014, 28, 2811–2820. [Google Scholar] [CrossRef]
- Martini, M.; van den Berg, A.; Gallucci, F.; van Sint Annaland, M. Investigation of the process operability windows for Ca-Cu looping for hydrogen production with CO2 capture. Chem. Eng. J. 2016, 303, 73–88. [Google Scholar] [CrossRef]
- Dou, B.; Wang, C.; Song, Y.; Chen, H.; Jiang, B.; Yang, M.; Xu, Y. Solid sorbents for in-situ CO2 removal during sorption-enhanced steam reforming process: A review. Renew. Sustain. Energy Rev. 2016, 53, 536–546. [Google Scholar] [CrossRef]
- Yancheshmeh, M.S.; Radfarnia, H.R.; Iliuta, M.C. High temperature CO2 sorbents and their application for hydrogen production by sorption enhanced steam reforming process. Chem. Eng. J. 2016, 283, 420–444. [Google Scholar] [CrossRef]
- Li, C.-C.; Cheng, J.-Y.; Liu, W.-H.; Huang, C.-M.; Hsu, H.-W.; Lin, H.-P. Enhancement in cyclic stability of the CO2 adsorption capacity of CaO-based sorbents by hydration for the calcium looping cycle. J. Taiwan Inst. Chem. Eng. 2014, 45, 227–232. [Google Scholar] [CrossRef]
- Zhong, X.; Xie, W.; Wang, N.; Duan, Y.; Shang, R.; Huang, L. Dolomite-derived ni-based catalysts with Fe modification for hydrogen production via auto-thermal reforming of acetic acid. Catalysts 2016, 6, 85. [Google Scholar] [CrossRef]
- Buelens, L.C.; Galvita, V.V.; Poelman, H.; Detavernier, C.; Marin, G.B. Super-dry reforming of methane intensifies CO2 utilization via Le Chatelier’s principle. Science 2016, 354, 449–452. [Google Scholar] [CrossRef] [PubMed]
- Dou, B.; Jiang, B.; Song, Y.; Zhang, C.; Wang, C.; Chen, H.; Du, B.; Xu, Y. Enhanced hydrogen production by sorption-enhanced steam reforming from glycerol with in-situ CO2 removal in a fixed-bed reactor. Fuel 2016, 166, 340–346. [Google Scholar] [CrossRef]
- Rydén, M.; Ramos, P. H2 production with CO2 capture by sorption enhanced chemical-looping reforming using NiO as oxygen carrier and CaO as CO2 sorbent. Fuel Process. Technol. 2012, 96, 27–36. [Google Scholar] [CrossRef]
- Hafizi, A.; Rahimpour, M.R.; Hassanajili, S. Hydrogen production via chemical looping steam methane reforming process: Effect of cerium and calcium promoters on the performance of Fe2O3/Al2O3 oxygen carrier. Appl. Energy 2016, 165, 685–694. [Google Scholar] [CrossRef]
- Tang, M.; Xu, L.; Fan, M. Progress in oxygen carrier development of methane-based chemical-looping reforming: A review. Appl. Energy 2015, 151, 143–156. [Google Scholar] [CrossRef]
- Meshksar, M.; Daneshmand-Jahromi, S.; Rahimpour, M.R. Synthesis and characterization of cerium promoted Ni/SBA-16 oxygen carrier in cyclic chemical looping steam methane reforming. J. Taiwan Inst. Chem. Eng. 2017. [Google Scholar] [CrossRef]
- Dou, B.; Song, Y.; Wang, C.; Chen, H.; Yang, M.; Xu, Y. Hydrogen production by enhanced-sorption chemical looping steam reforming of glycerol in moving-bed reactors. Appl. Energy 2014, 130, 342–349. [Google Scholar] [CrossRef]
- Köpfle, N.; Mayr, L.; Schmidmair, D.; Bernardi, J.; Knop-Gericke, A.; Hävecker, M.; Klötzer, B.; Penner, S. A comparative discussion of the catalytic activity and CO2-selectivity of Cu-Zr and Pd-Zr (intermetallic) compounds in methanol steam reforming. Catalysts 2017, 7, 53. [Google Scholar] [CrossRef]
- Yang, X.; Wang, Y.; Wang, Y. Significantly improved catalytic performance of Ni-based MgO catalyst in steam reforming of phenol by inducing mesostructure. Catalysts 2015, 5, 1721–1736. [Google Scholar] [CrossRef]
- Abad, A.; Adánez-Rubio, I.; Gayán, P.; García-Labiano, F.; de Diego, L.F.; Adánez, J. Demonstration of chemical-looping with oxygen uncoupling (CLOU) process in a 1.5 kWth continuously operating unit using a Cu-based oxygen-carrier. Int. J. Greenh. Gas Control 2012, 6, 189–200. [Google Scholar] [CrossRef]
- Ayodele, B.V.; Khan, M.R.; Cheng, C.K. Syngas production from CO2 reforming of methane over ceria supported cobalt catalyst: Effects of reactants partial pressure. J. Nat. Gas Sci. Eng. 2015, 27, 1016–1023. [Google Scholar] [CrossRef]
- Ramírez-Hernández, G.Y.; Viveros-García, T.; Fuentes-Ramírez, R.; Galindo-Esquivel, I.R. Promoting behavior of yttrium over nickel supported on alumina-yttria catalysts in the ethanol steam reforming reaction. Int. J. Hydrogen Energy 2016, 41, 9332–9343. [Google Scholar] [CrossRef]
- Li, J.; Xia, C.; Au, C.; Liu, B. Y2O3-promoted NiO/SBA-15 catalysts highly active for CO2/CH4 reforming. Int. J. Hydrogen Energy 2014, 39, 10927–10940. [Google Scholar] [CrossRef]
- Sangwichien, C.; Aranovich, G.L.; Donohue, M.D. Density functional theory predictions of adsorption isotherms with hysteresis loops. Colloids Surf. A 2002, 206, 313–320. [Google Scholar] [CrossRef]
- Youn, M.H.; Seo, J.G.; Jung, J.C.; Park, S.; Song, I.K. Hydrogen production by auto-thermal reforming of ethanol over nickel catalyst supported on mesoporous yttria-stabilized zirconia. Int. J. Hydrogen Energy 2009, 34, 5390–5397. [Google Scholar] [CrossRef]
- Luo, L.; Songjun, L.; Zhu, Y. The effects of yttrium on the hydrogenation performance and surface properties of a ruthenium-supported catalyst. J. Serbian Chem. Soc. 2005, 70, 1419–1425. [Google Scholar] [CrossRef]
- Li, B.; Zhang, S. Methane reforming with CO2 using nickel catalysts supported on yttria-doped sba-15 mesoporous materials via sol-gel process. Int. J. Hydrogen Energy 2013, 38, 14250–14260. [Google Scholar] [CrossRef]
- Rydén, M.; Lyngfelt, A.; Mattisson, T. Synthesis gas generation by chemical-looping reforming in a continuously operating laboratory reactor. Fuel 2006, 85, 1631–1641. [Google Scholar] [CrossRef]

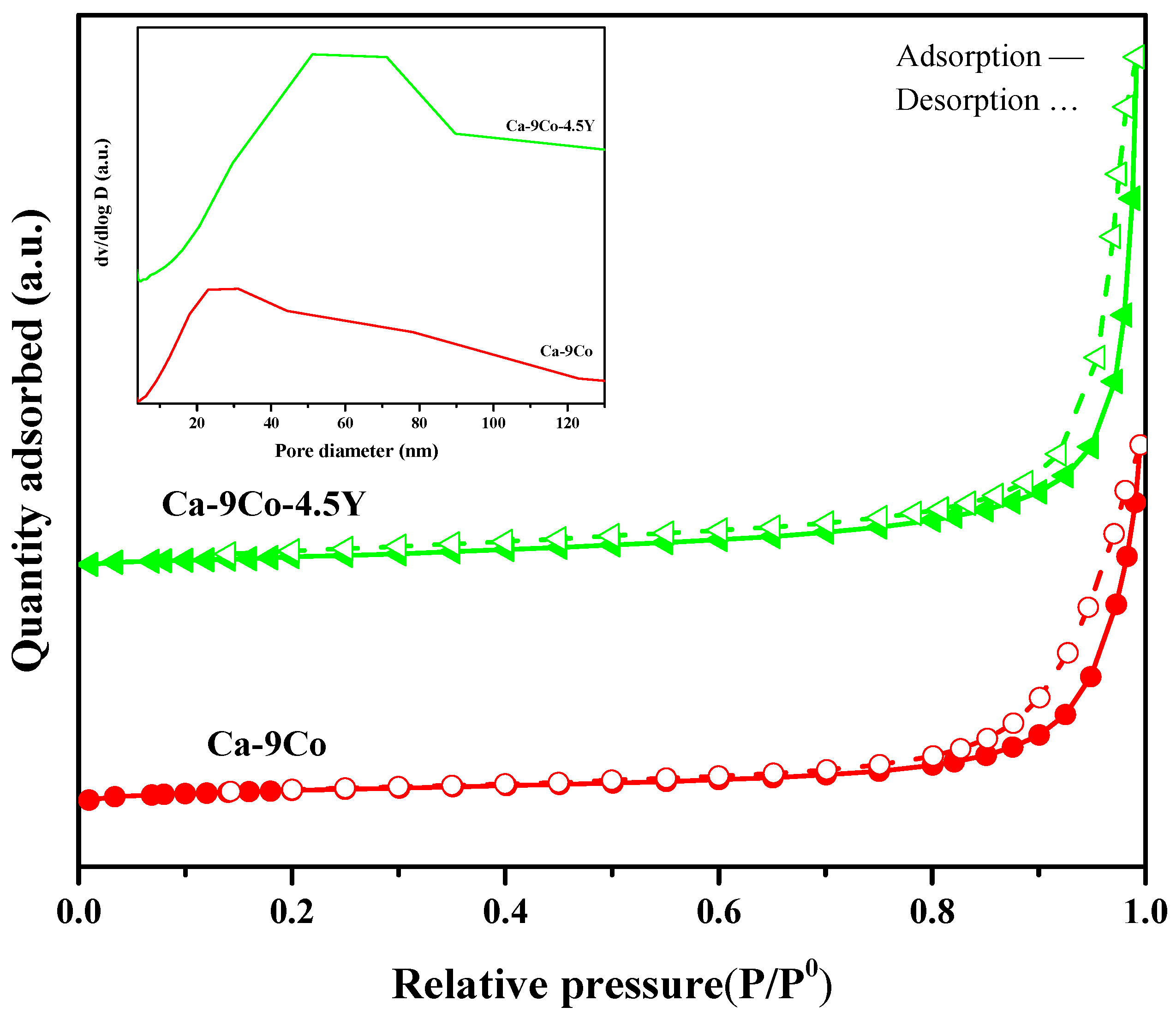






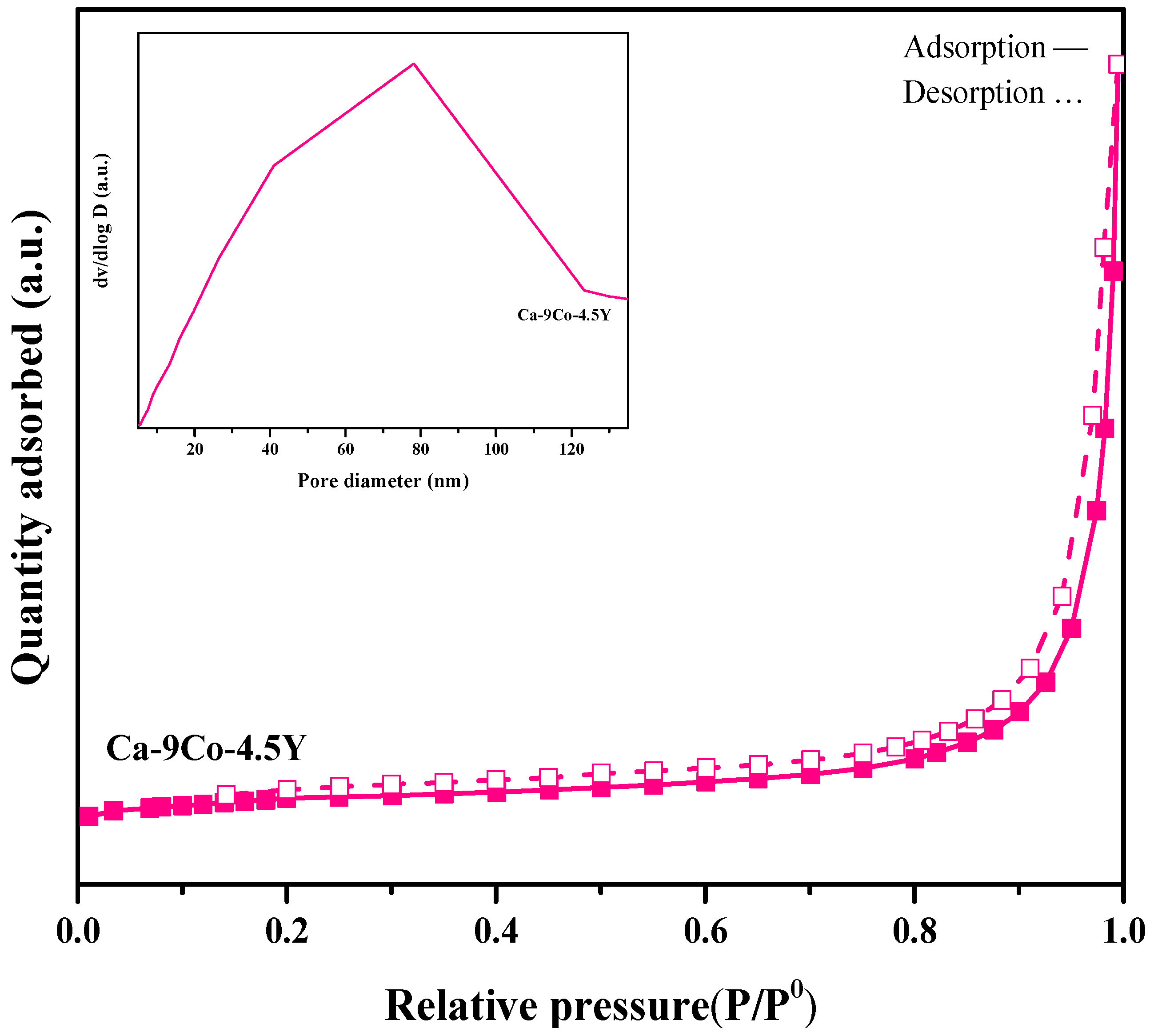
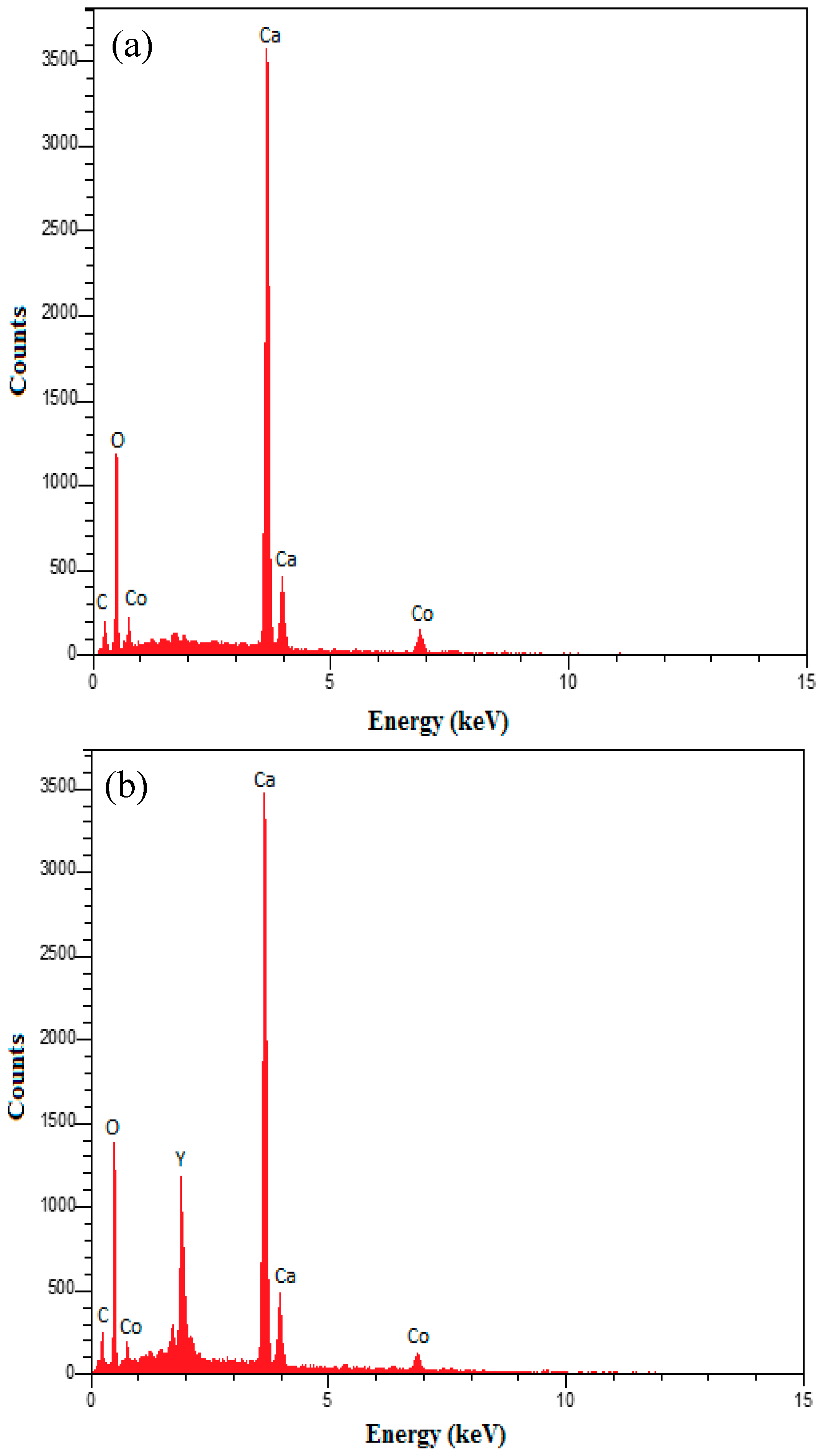

| Sample | BET Surface Area (m2/g) | Pore Diameter (nm) a | Pore Volume (cm3/g) b |
|---|---|---|---|
| Ca-9Co/fresh | 14.0 | 25.55 | 0.111 |
| Ca-9Co-4.5Y/fresh | 17.9 | 26.41 | 0.161 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Akbari-Emadabadi, S.; Rahimpour, M.R.; Hafizi, A.; Keshavarz, P. Promotion of Ca-Co Bifunctional Catalyst/Sorbent with Yttrium for Hydrogen Production in Modified Chemical Looping Steam Methane Reforming Process. Catalysts 2017, 7, 270. https://doi.org/10.3390/catal7090270
Akbari-Emadabadi S, Rahimpour MR, Hafizi A, Keshavarz P. Promotion of Ca-Co Bifunctional Catalyst/Sorbent with Yttrium for Hydrogen Production in Modified Chemical Looping Steam Methane Reforming Process. Catalysts. 2017; 7(9):270. https://doi.org/10.3390/catal7090270
Chicago/Turabian StyleAkbari-Emadabadi, Samira, Mohammad Reza Rahimpour, Ali Hafizi, and Peyman Keshavarz. 2017. "Promotion of Ca-Co Bifunctional Catalyst/Sorbent with Yttrium for Hydrogen Production in Modified Chemical Looping Steam Methane Reforming Process" Catalysts 7, no. 9: 270. https://doi.org/10.3390/catal7090270
APA StyleAkbari-Emadabadi, S., Rahimpour, M. R., Hafizi, A., & Keshavarz, P. (2017). Promotion of Ca-Co Bifunctional Catalyst/Sorbent with Yttrium for Hydrogen Production in Modified Chemical Looping Steam Methane Reforming Process. Catalysts, 7(9), 270. https://doi.org/10.3390/catal7090270