DeNOx Abatement Modelling over Sonically Prepared Copper USY and ZSM5 Structured Catalysts
Abstract
:1. Introduction
2. Results
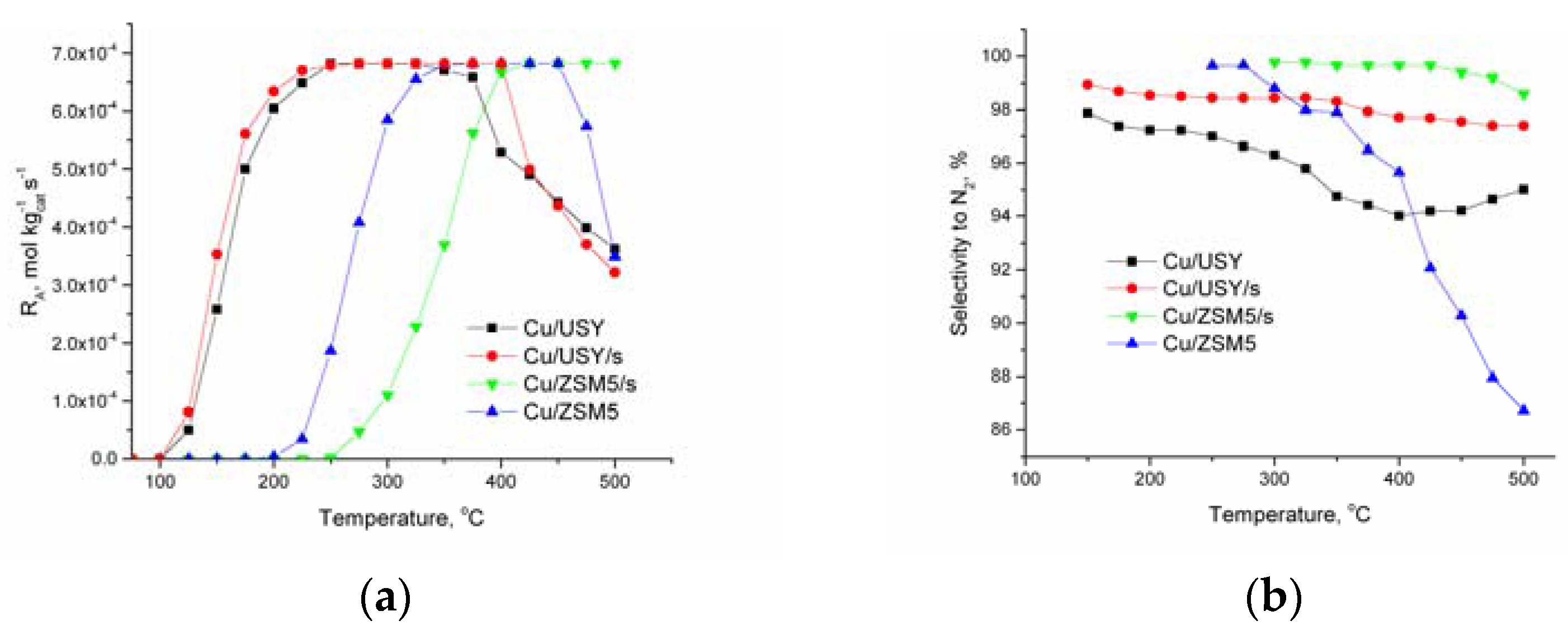
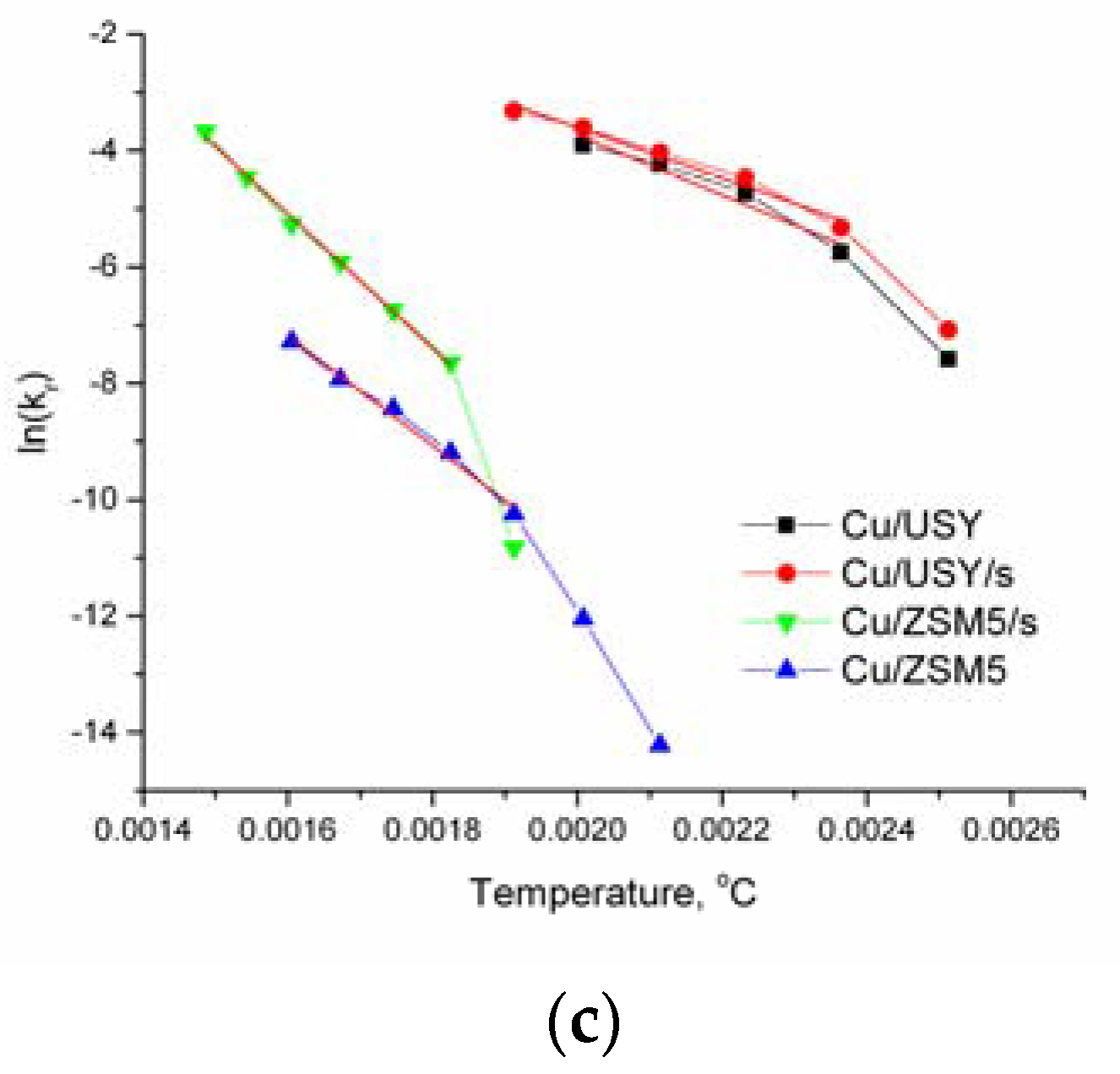
2.1. Kinetic Results
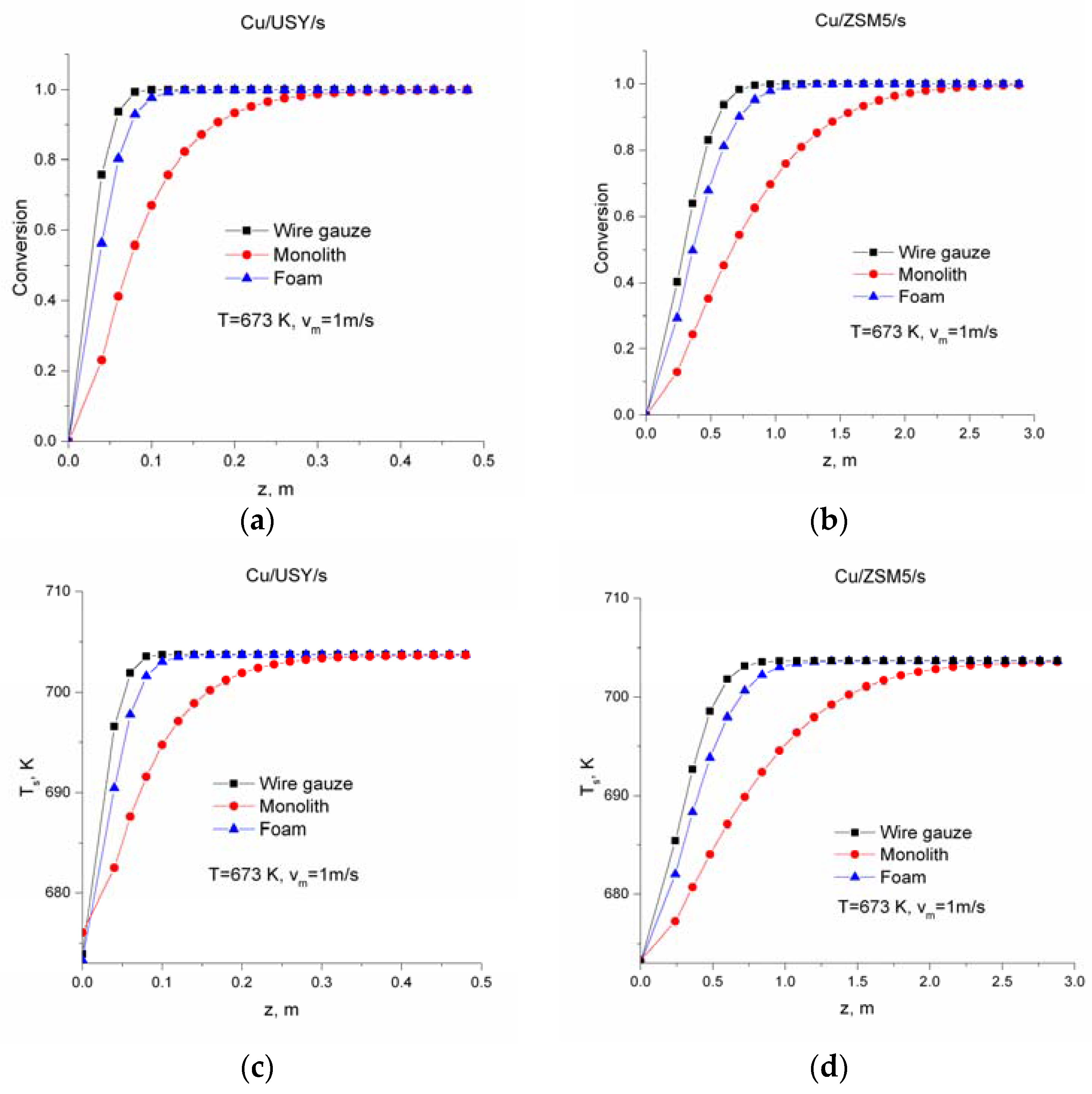
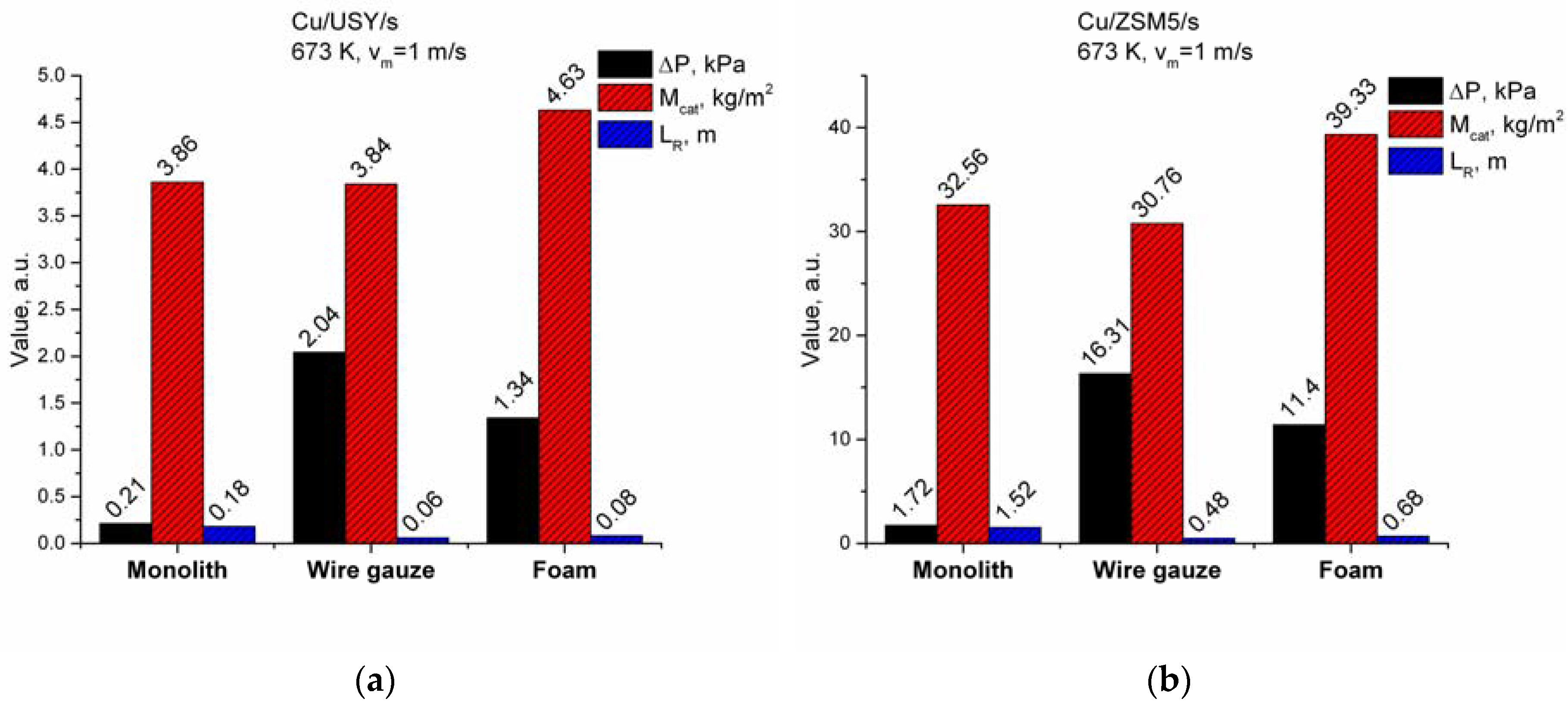
2.2. Modelling Results
3. Discussion
4. Materials and Methods
4.1. Catalyst Preparation and Characterization
4.2. Kinetic Tests
4.3. Reactor Modelling
4.3.1. Mass and Energy Balances
4.3.2. Pressure Drop
4.3.3. Reaction Kinetics
4.3.4. Modelling Conditions
- for the reaction: 4NO + 4NH3 + O2 → 4N2 + 6H2O the heat of reaction at the catalyst surface was determined to be, ∆H°R = −407 kJ/mol;
- properties along the channel change as a function of the local temperature;
- the inlet NO concentration equals 2500 ppm (complete conversion obtained for prepared catalysts);
- the inlet gas temperature equals 573 K;
- the gas superficial velocity equals 1 m/s; the corresponding Reynolds numbers are given in Table 2.
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Appendix A
List of Symbols:
| A | channel cross-sectional surface area, m2 |
| A | specific surface area, m−1 |
| CA, CAS | mean reactant A concentration in bulk gas phase, at catalyst external surface, respectively, mol/m3 |
| Cp | heat capacity, J/mol∙K |
| dp | sphere diameter, m |
| Dh | hydraulic diameter, m |
| DA, DAi | molecular diffusivity; internal diffusivity (in porous catalyst), respectively; m2/s |
| ds | strut diameter (foam), m |
| dw | wire diameter (wire gauze), m |
| EA | apparent activation energy, J/mol |
| f | Fanning friction factor |
| h | heat transfer coefficient, W/m2∙K |
| ∆H°R | standard heat of reaction, J/mol |
| kr | reaction rate constant, m/s |
| kC | mass transfer coefficient, m/s |
| k∞ | pre-exponential coefficient in Arrhenius equation, m/s |
| keff - | effective reaction rate constant, m/s |
| L, LR | channel or reactor length, respectively, m |
| L*H, L*M | heat or mass dimensionless channel length, respectively |
| L+ | dimensional channel length, |
| L | diffusion path length in the catalytic phase, m |
| Mcat | Mass of catalyst, kg/m2 |
| Nu | Nusselt number, hDh/λ |
| Pr | Prandtl number, Cpνρ/λ |
| P | channel perimeter, m |
| ∆P | pressure drop, Pa |
| (-RA) | reaction rate, mol/(m2·s) |
| Re | Reynolds number, vmDh/ν |
| Sc | Schmidt number, ν/DA |
| Sh | Sherwood number, kCDh/DA |
| T | temperature, K |
| vm | superficial gas velocity, m/s |
| z | reactor axis, m |
| ε | bed or structure void volume (external packing porosity), dimensionless |
| η | effectiveness factor for catalyst |
| λ | heat conductivity, W/mK |
| φ | Thiele modulus, Equation (3) |
| ν | kinematic viscosity |
Subscripts
| A | relating to species A |
| AS | relating to adsorbed species A, surface value |
| 0 | refers to reactor inlet |
| S | refers to catalyst surface |
References
- Chen, H.; Wei, Z.; Kollar, M.; Gao, F.; Wang, Y.; Szanyi, J.; Peden, C.H.F. A comparative study of N2O formation during the selective catalytic reduction of NOx with NH3 on zeolite supported Cu catalysts. J. Catal. 2015, 329, 490–498. [Google Scholar] [CrossRef]
- Li, S.; Chen, Z.; He, E.; Jiang, B.; Li, Z.; Wang, Q. Combustion characteristics and NOx formation of a retrofitted low-volatile coal-fired 330 MW utility boiler under various loads with deep-air-staging. Appl. Therm. Eng. 2017, 110, 223–233. [Google Scholar] [CrossRef]
- Roy, S.; Hegde, M.S.; Madras, G. Catalysis for NOx abatement. Appl. Energy 2009, 86, 2283–2297. [Google Scholar] [CrossRef]
- Carabineiro, S.A.C.; Lobo, L.S. Understanding the Reactions of CO2, NO, and N2O with Activated Carbon Catalyzed by Binary Mixtures. Energy Fuels 2016, 30, 6881–6891. [Google Scholar] [CrossRef]
- Carabineiro, S.A.; Fernandes, F.B.; Vital, J.S.; Ramos, A.M.; Silva, I.F. Uncatalyzed and catalyzed NO and N2O reaction using various catalysts and binary barium mixtures supported on activated carbon. Catal. Today 1999, 54, 559–567. [Google Scholar] [CrossRef]
- BTG Biomass Technology Group BV. Handbook of Biomass Gasification; Knoef, H.A.M., Ed.; BTG Biomass Technology Group BV: Enschede, The Netherlands, 2005. [Google Scholar]
- Cybulski, A.; Moulijn, J.A. Structured Catalysts and Reactors; Chemical Industries; Taylor & Francis: Abingdon, UK, 2006. [Google Scholar]
- Busca, G.; Kennedy, P.J.F. Acidity and basicity of zeolites: A fundamental approach. Microporous Mesoporous Mater. 2017, 1–14. [Google Scholar] [CrossRef]
- Vaiciukyniene, D.; Kantautas, A.; Vaitkevicius, V.; Jakevicius, L.; Rudzionis, Z.; Paskevicius, M. Effects of ultrasonic treatment on zeolite NaA synthesized from by-product silica. Ultrason. Sonochem. 2015, 27, 515–521. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Wu, J.; Yuan, Z.-Y.; Li, N.; Xiang, S. Synthesis of MCM-22 zeolite by an ultrasonic-assisted aging procedure. Ultrason. Sonochem. 2008, 15, 334–338. [Google Scholar] [CrossRef] [PubMed]
- Belviso, C.; Cavalcante, F.; Lettino, A.; Fiore, S. Ultrasonics Sonochemistry Effects of ultrasonic treatment on zeolite synthesized from coal fly ash. Ultrason. Sonochem. 2011, 18, 661–668. [Google Scholar] [CrossRef] [PubMed]
- Kryca, J.; Iwaniszyn, M.; Piątek, M.; Jodłowski, P.J.; Jędrzejczyk, R.; Pędrys, R.; Wróbel, A.; Łojewska, J.; Kołodziej, A. Structured Foam Reactor with CuSSZ-13 Catalyst for SCR of NOx with Ammonia. Top. Catal. 2016, 59, 887–894. [Google Scholar] [CrossRef]
- Gancarczyk, A.; Piątek, M.; Iwaniszyn, M.; Jodłowski, P.J.; Łojewska, J.; Kowalska, J.; Kołodziej, A. In Search of Governing Gas Flow Mechanism through Metal Solid Foams. Catalysts 2017, 7, 124. [Google Scholar] [CrossRef]
- Iwaniszyn, M.; Piątek, M.; Gancarczyk, A.; Jodłowski, P.J.; Łojewska, J.; Kołodziej, A. Flow resistance and heat transfer in short channels of metallic monoliths: Experiments versus CFD. Int. J. Heat Mass Transf. 2017, 109, 778–785. [Google Scholar] [CrossRef]
- Łojewska, J.; Knapik, A.; Jodłowski, P.; Łojewski, T.; Kołodziej, A. Topography and morphology of multicomponent catalytic materials based on Co, Ce and Pd oxides deposited on metallic structured carriers studied by AFM/Raman interlaced microscopes. Catal. Today 2013, 216, 11–17. [Google Scholar] [CrossRef]
- Jodłowski, P.J.; Gołąb, R.; Kryca, J.; Kołodziej, A.; Iwaniszyn, M.; Kolaczkowski, S.T.; Łojewska, J. A Comparison Between Monolithic and Wire Gauze Structured Catalytic Reactors for CH4 and CO Removal from Biogas-Fuelled Engine Exhaust. Top. Catal. 2013, 56, 390–396. [Google Scholar] [CrossRef]
- Kryca, J.; Jodłowski, P.J.; Iwaniszyn, M.; Gil, B.; Sitarz, M.; Kołodziej, A.; Łojewska, T.; Łojewska, J. Cu SSZ-13 zeolite catalyst on metallic foam support for SCR of NOx with ammonia: Catalyst layering and characterisation of active sites. Catal. Today 2016, 268, 142–149. [Google Scholar] [CrossRef]
- Ochońska, J.; Rogulska, A.; Jodłowski, P.J.; Iwaniszyn, M.; Michalik, M.; Łasocha, W.; Kołodziej, A.; Łojewska, J. Prospective Catalytic Structured Converters for NH3-SCR of NOx from Biogas Stationary Engines: In Situ Template-Free Synthesis of ZSM-5 Cu Exchanged Catalysts on Steel Carriers. Top. Catal. 2013, 56, 56–61. [Google Scholar] [CrossRef]
- Ochońska, J.; McClymont, D.; Jodłowski, P.J.; Knapik, A.; Gil, B.; Makowski, W.; Łasocha, W.; Kołodziej, A.; Kolaczkowski, S.T.; Łojewska, J. Copper exchanged ultrastable zeolite Y—A catalyst for NH3-SCR of NOx from stationary biogas engines. Catal. Today 2012, 191, 6–11. [Google Scholar] [CrossRef]
- Brandenberger, S.; Kröcher, O.; Tissler, A.; Althoff, R. The State of the Art in Selective Catalytic Reduction of NOx by Ammonia Using Metal-Exchanged Zeolite Catalysts. Catal. Rev. 2008, 50, 492–531. [Google Scholar] [CrossRef]
- Komatsu, T.; Nunokawa, M.; Moon, I.L.; Takahara, T.; Namba, S.; Yashima, T. Kinetic Studies of Reduction of Nitric Oxide with Ammonia on Cu2+-Exchanged Zeolites. J. Catal. 1994, 148, 427–437. [Google Scholar] [CrossRef]
- Yu, C.; Huang, B.; Dong, L.; Chen, F.; Liu, X. In situ FT-IR study of highly dispersed MnOx/SAPO-34 catalyst for low-temperature selective catalytic reduction of NOx by NH3. Catal. Today 2016, 281, 610–620. [Google Scholar] [CrossRef]
- Rutkowska, M.; Pacia, I.; Basąg, S.; Kowalczyk, A.; Piwowarska, Z.; Duda, M.; Tarach, K.A.; Góra-Marek, K.; Michalik, M.; Díaz, U. Catalytic performance of commercial Cu-ZSM-5 zeolite modified by desilication in NH3-SCR and NH3-SCO processes. Microporous Mesoporous Mater. 2017, 246, 193–206. [Google Scholar] [CrossRef]
- Sanz, O.; Velasco, I.; Reyero, I.; Legorburu, I.; Arzamendi, G.; Gandía, L.M.; Montes, M. Effect of the thermal conductivity of metallic monoliths on methanolsteam reforming. Catal. Today 2016, 273, 131–139. [Google Scholar] [CrossRef]
- Aghaei, P.; Visconti, C.G.; Groppi, G.; Tronconi, E. Development of a heat transport model for open-cell metal foams with high cell densities. Chem. Eng. J. 2017, 321, 432–446. [Google Scholar] [CrossRef]
- Bianchi, E.; Heidig, T.; Visconti, C.G.; Groppi, G.; Freund, H.; Tronconi, E. Heat transfer properties of metal foam supports for structured catalysts: Wall heat transfer coefficient. Catal. Today 2013, 216, 121–134. [Google Scholar] [CrossRef]
- Kołodziej, A.; Łojewska, J. Mass transfer for woven and knitted wire gauze substrates: Experiments and modelling. Catal. Today 2009, 147S, 120–124. [Google Scholar] [CrossRef]
- Hayes, R.; Kolaczkowski, S.T. A study of Nusselt and Sherwood numbers in a monolith reactor. Catal. Today 1999, 47, 295–303. [Google Scholar] [CrossRef]
- Hawthorn, R.D. Afterburner catalysts effects of heat and mass transfer between gas and catalyst surface. AIChE Symp. Ser. 1974, 70, 428. [Google Scholar]
- Giani, L.; Groppi, G.; Tronconi, E. Heat Transfer Characterisation of Metallic Foams. Ind. Eng. Chem. Res. 2005, 44, 9078–9085. [Google Scholar] [CrossRef]
- Shah, R.K.; London, A.L. Laminar Flow Forced Convection in Ducts: A Source Book for Compact Heat Exchanger Analytical Data; Academic Press: New York, NY, USA, 1978. [Google Scholar]
- Sharratt, P.N.; Mann, R. Some observations on the variation of tortuosity with Thiele modulus and pore size distribution. Chem. Eng. Sci. 1987, 42, 1565–1576. [Google Scholar] [CrossRef]
- Piątek, M.; Garncarczyk, A.; Iwaniszyn, M.; Jodłowski, P.J.; Łojewska, J.; Kołodziej, A. Gas-Phase Flow Resistance of Metal Foams: Experiments and Modeling. AIChE J. 2017. [Google Scholar] [CrossRef]
- Kołodziej, A.; Łojewska, J.; Jaroszyński, M.; Gancarczyk, A.; Jodłowski, P. Heat transfer and flow resistance for stacked wire gauzes: Experiments and modelling. Int. J. Heat Fluid Flow 2012, 33, 101–108. [Google Scholar] [CrossRef]
- Kołodziej, A.; Łojewska, J. Experimental and modelling study on flow resistance of wire gauzes. Chem. Eng. Process. 2009, 48, 816–822. [Google Scholar] [CrossRef]
- Koebel, M.; Elsner, M. Selective catalytic reduction of NO over commercial DeNOx-catalysts: Experimental determination of kinetic and thermodynamic parameters. Chem. Eng. Sci. 1998, 53, 657–669. [Google Scholar] [CrossRef]
- Kołodziej, A.; Łojewska, J.; Tyczkowski, J.; Jodłowski, P.; Redzynia, W.; Iwaniszyn, M.; Zapotoczny, S.; Kuśtrowski, P. Coupled engineering and chemical approach to the design of a catalytic structured reactor for combustion of VOCs: Cobalt oxide catalyst on knitted wire gauzes. Chem. Eng. J. 2012, 200, 329–337. [Google Scholar] [CrossRef]
| Catalyst | Preparation Method | Si/Al | Copper Content *, wt % | Preexponential Factor, k∞, m3/kg s | Activation Energy, Ea, kJ/mol | η ** |
|---|---|---|---|---|---|---|
| Cu/USY | Ion-exchange | 4.52 | 4.90 ± 0.05 | 1.18 × 101 | 42.76 | - |
| Cu/USY/s | Sonication | 4.52 | 3.70 ± 0.04 | 1.66 × 102 | 36.21 | 1 |
| Cu/ZSM5 | Ion-exchange | 37 | 0.320 ± 0.003 | 2.72 × 103 | 78.39 | - |
| Cu/ZSM5/s | Sonication | 37 | 0.130 ± 0.001 | 1.10 × 104 | 96.01 | 1 |
| Superficial Gas Velocity, m/s | Temperature Reactor Support | 673 K LR, m | 673 K Mcat *, kg/m2 | 673 K ∆P, kPa |
|---|---|---|---|---|
| νm = 0.5 | Wire gauze | 0.028 | 1.79 | 0.41 |
| Metallic foam | 0.036 | 2.08 | 0.26 | |
| Monolith | 0.08 | 1.71 | 0.05 | |
| νm = 1 | Wire gauze | 0.06 | 3.84 | 2.04 |
| Metallic foam | 0.08 | 4.63 | 1.34 | |
| Monolith | 0.18 | 3.86 | 0.21 | |
| νm = 2 | Wire gauze | 0.12 | 7.69 | 10.43 |
| Metallic foam | 0.16 | 7.71 | 6.70 | |
| Monolith | 0.36 | 9.25 | 0.87 |
| Reactor Support | Dh, mm | a, m−1 | Mesh/PPI/cpsi | L, mm | Re | Heat and Mass Transfer Equation |
|---|---|---|---|---|---|---|
| Wire gauze [27] | 0.699 | 4005 | 30.48 | dw = 0.30 | 25 | |
| Metal foam (NC2733) * | 0.961 | 3615 | 27–33 | ds = 0.14 | 35 | |
| Monolith [28,29] | 2.15 | 1399 | 100 | LR | 80 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jodłowski, P.J.; Kuterasiński, Ł.; Jędrzejczyk, R.J.; Chlebda, D.; Gancarczyk, A.; Basąg, S.; Chmielarz, L. DeNOx Abatement Modelling over Sonically Prepared Copper USY and ZSM5 Structured Catalysts. Catalysts 2017, 7, 205. https://doi.org/10.3390/catal7070205
Jodłowski PJ, Kuterasiński Ł, Jędrzejczyk RJ, Chlebda D, Gancarczyk A, Basąg S, Chmielarz L. DeNOx Abatement Modelling over Sonically Prepared Copper USY and ZSM5 Structured Catalysts. Catalysts. 2017; 7(7):205. https://doi.org/10.3390/catal7070205
Chicago/Turabian StyleJodłowski, Przemysław J., Łukasz Kuterasiński, Roman J. Jędrzejczyk, Damian Chlebda, Anna Gancarczyk, Sylwia Basąg, and Lucjan Chmielarz. 2017. "DeNOx Abatement Modelling over Sonically Prepared Copper USY and ZSM5 Structured Catalysts" Catalysts 7, no. 7: 205. https://doi.org/10.3390/catal7070205
APA StyleJodłowski, P. J., Kuterasiński, Ł., Jędrzejczyk, R. J., Chlebda, D., Gancarczyk, A., Basąg, S., & Chmielarz, L. (2017). DeNOx Abatement Modelling over Sonically Prepared Copper USY and ZSM5 Structured Catalysts. Catalysts, 7(7), 205. https://doi.org/10.3390/catal7070205










