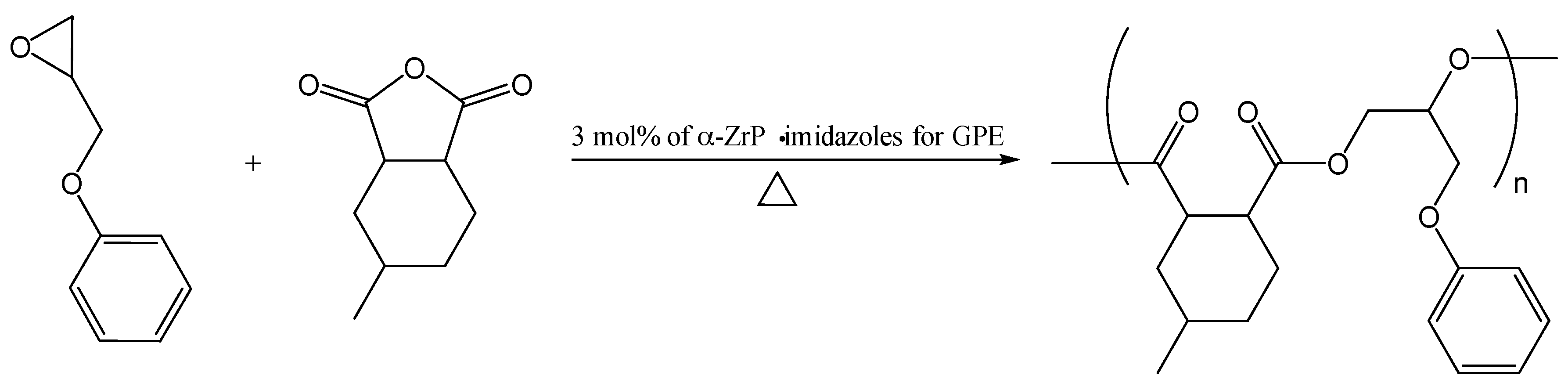1. Introduction
There has been considerable interest in the development of latent polymerization catalysts that are inert in the monomer or prepolymer under storage conditions. These catalysts can initiate polymerization under external impetus such as thermal or photo irradiation. Various kinds of latent catalysts are used in the polymerization reaction such as sulfonium salt [
1,
2,
3,
4], phosphonium salt [
5], pyrazinium salt [
6],
N-heterocyclic carbene [
7], aminimide [
8], and sulfonate [
9]. We have previously reported that primary alkylamines intercalated with α-zirconium phosphate (α-ZrP) can serve as latent thermal initiators in the reaction of glycidyl phenyl ether (GPE) [
10] and that intercalation compounds of 1,4-diazabicyclo(2,2,2)octane (DABCO) and 1,8-diazabicyclo(5,4,0)undec-7-ene (DBU) with α-ZrP: α-ZrP·DABCO and α-ZrP·DBU show good performance as latent thermal catalysts in the reaction of GPE with hexahydro-4-methylphthalic anhydride (MHHPA) [
11].
Aside from DBU and DABCO, imidazoles have been widely used for curing epoxy resins as cured epoxy resins give good thermal resistance and physical properties because of the reactivity in chain polymerization with epoxide [
12,
13]. Latent thermal initiators of imidazoles have been developed. Romanchick prepared 1,3-dialkylimidazolium salts in the curing of epoxy resin [
14]. In this case, the activation of catalyst needs a high temperature around 200 °C. Shin reported using microencapsulated imidazoles with polycaprolactone [
15]. Arimitsu et al. proposed using the Michel addition products of imidazoles and fumarate ester to improve the low miscibility in epoxy resin [
16]. However, these catalysts remain the blocking residues following the reaction to epoxy groups. These residues might be attributed to the deterioration of the resin. Amine-intercalated α-ZrP can act as latent thermal initiators [
17,
18] and the blocking residues of α-ZrP can act as additives for reinforcement of resins [
19,
20,
21].
A series of imidazoles are expected to have different p
Ka due to their substituents and to have different reactivity as the intercalation compounds of α-ZrP. We prepared intercalation compounds of imidazole (Im), 2-methylimidazole (2MIm), and 2-ethyl-4-methylimidazole (2E4MIm) with α-ZrP (hereafter abbreviated as α-ZrP·Im, α-ZrP·2MIm, and α-ZrP·2E4MIm) and examined the capabilities of α-ZrP·Im, α-ZrP·2MIm, and α-ZrP·2E4MIm as latent thermal initiators in the curing of GPE with hexahydro-4-methylphthalic anhydride (MHHPA). The p
Ka value of Im, 2MIm, and 2E4MIm were 6.99 [
22], 7.75 [
22], and 8.3 [
23], respectively. Latent thermal initiators containing imidazoles have been developed and the polymer-encapsulated imidazoles HX-3088 and HX-3722 are now commercially available. For comparison, the commercially available latent thermal initiators HX-3088 and HX-3722 were tested in the polymerization of GPE with MHHPA.
2. Results and Discussion
The intercalation compounds were prepared by mixing imidazole (Im), 2-methylimidazole (2MIm), or 2-ethyl-4-methylimidazole (2E4MIm) with α-ZrP by a similar procedure of another reported method [
24]. The compositions determined by elemental analyses were Zr(HPO
4)
2(C
3H
4N
2)
0.78·1.1H
2O (α-ZrP·Im), Zr(HPO
4)
2(C
4H
6N
2)
0.96·1.2H
2O (α-ZrP·2MIm), and Zr(HPO
4)
2(C
6H
10N
2)
0.65·0.6H
2O (α-ZrP·2E4MIm), respectively. Assuming that the weight loss after the weight loss of water was attributed to that of imidazoles, each of the compositional formula was calculated; α-ZrP·Im: Zr(HPO
4)
2(C
3H
4N
2)
0.87·0.92H
2O, α-ZrP·2MIm: Zr(HPO
4)
2(C
4H
6N
2)
0.94·1.5H
2O, and α-ZrP·2E4MIm, Zr(HPO
4)
2(C
6H
10N
2)
0.72·0.13H
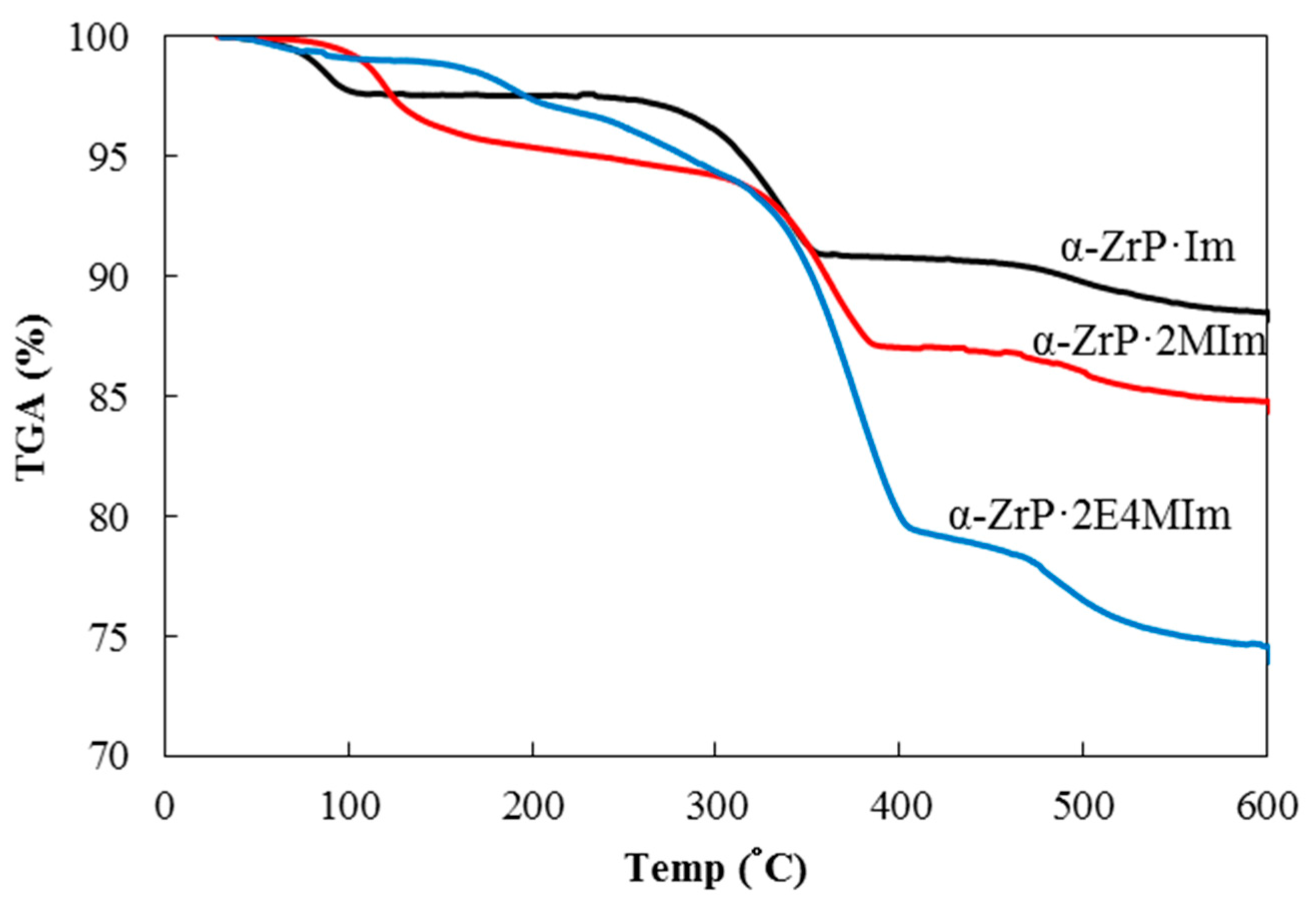
2O. The basal distances of 10.7 Å (2θ = 8.2°) for α-ZrP·Im, 12.1 Å (2θ = 7.3°) for α-ZrP·2MIm and 12.9 Å (2θ = 6.9°) for α-ZrP·2E4MIm expanded compared with that of pristine α-ZrP (7.6 Å, 2θ = 11.7°). The thermal properties of these α
-ZrP-imidazoles intercalation compounds were examined by thermogravimetric analysis (TGA) shown in
Figure 1. The thermal weight loss around 100 °C showed a decrease of water. After the weight loss of water, α-ZrP·Im was stable up to 250 °C and weight decreased until 600 °C. In α-ZrP·2MIm and α-ZrP·2E4MIm, the weights gradually decreased from 175 °C until 600 °C. Up to 140 °C of the curing temperature (discussed later), these α
-ZrP-imidazoles intercalation compounds thermally lost H
2O rather than the imidazoles. Polymerization of GPE with MHHPA in the presence of α-ZrP·Im, α-ZrP·2MIm, and α-ZrP·2E4MIm containing 3 mol % of each intercalation compound for GPE at 140 °C was performed to produce poly(GPE-co-MHHPA) as shown in
Scheme 1. The molecular weights and the molecular weight distribution of the resulting poly(GPE-co-MHHPA) were determined by GPC:
Mn = 2000,
Mw/Mn = 1.4 for α-ZrP·Im at 140 °C for 2 h,
Mn = 2100,
Mw/Mn = 1.4 for α-ZrP·2MIm at 140 °C for 1 h, and
Mn = 3100,
Mw/Mn = 1.3 for α-ZrP·2E4MIm at 140 °C for 1 h, respectively. The conversion of GPE for α-ZrP·Im was 72% at 140 °C for 1h, the reaction time was extended to 2 h to obtain the quantitative conversion. The molecular weight and molecular weight distribution of poly(GPE-co-MHHPA) were previously reported as
Mn = 2600,
Mw/Mn = 1.4 for α-ZrP·DABCO, and
Mn = 720 and
Mw/Mn = 1.2 for α-ZrP·DBU [
11].
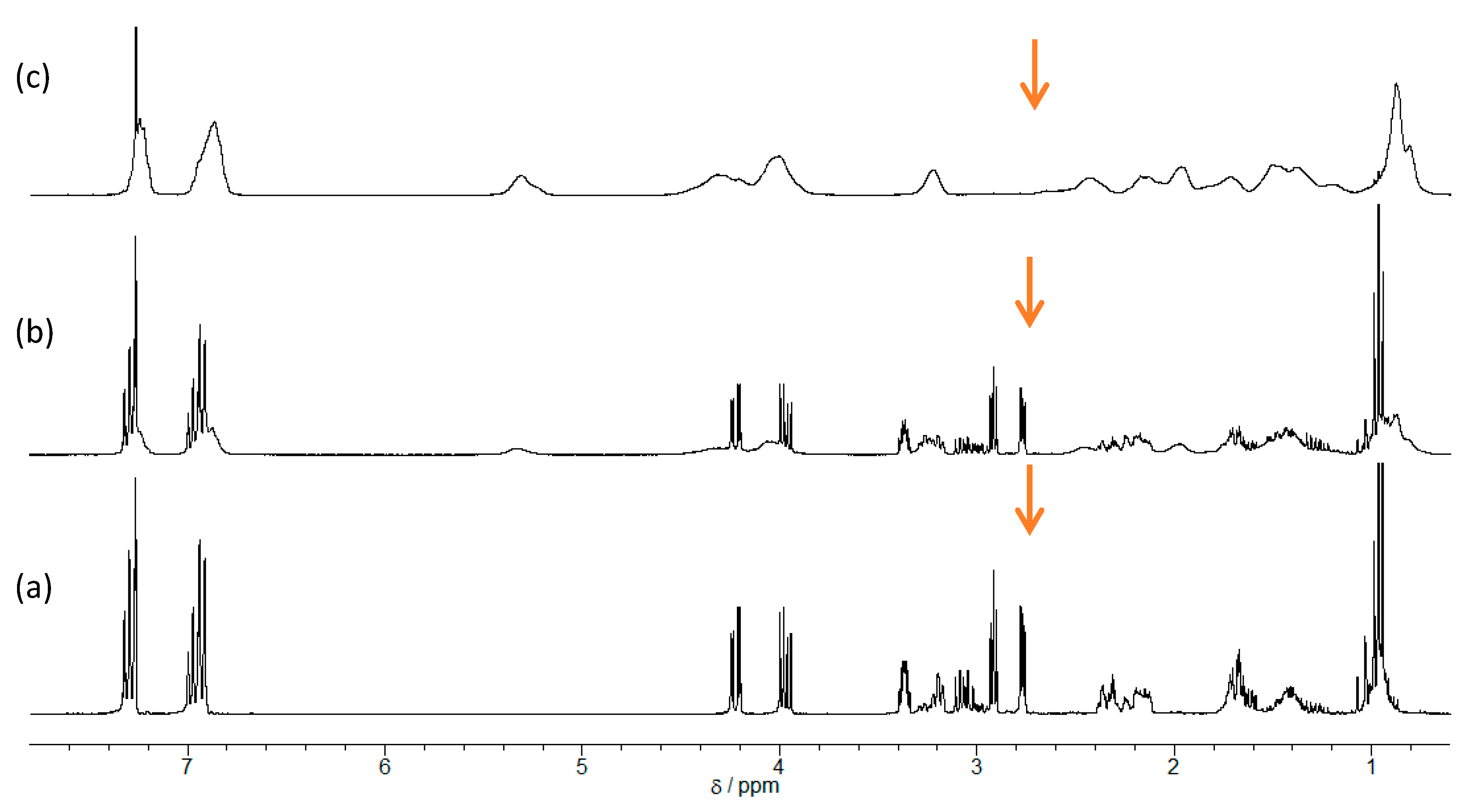
The capabilities of the α-ZrP·Im, α-ZrP·2MIm, and α-ZrP·2E4MIm as latent initiators were examined in the reactions of GPE with MHHPA containing 3 mol % of each intercalation compound for GPE at varying temperatures for 1 h periods by
1H-NMR technique as shown in
Figure 2. The conversions of GPEs were measured from the area ratio of the aromatic protons at δ 6.9 and the methylene protons of δ 2.92 (indicated by arrows), which relatively decreased with an increase in the reaction temperature from 80 to 140 °C (
Figure 2).
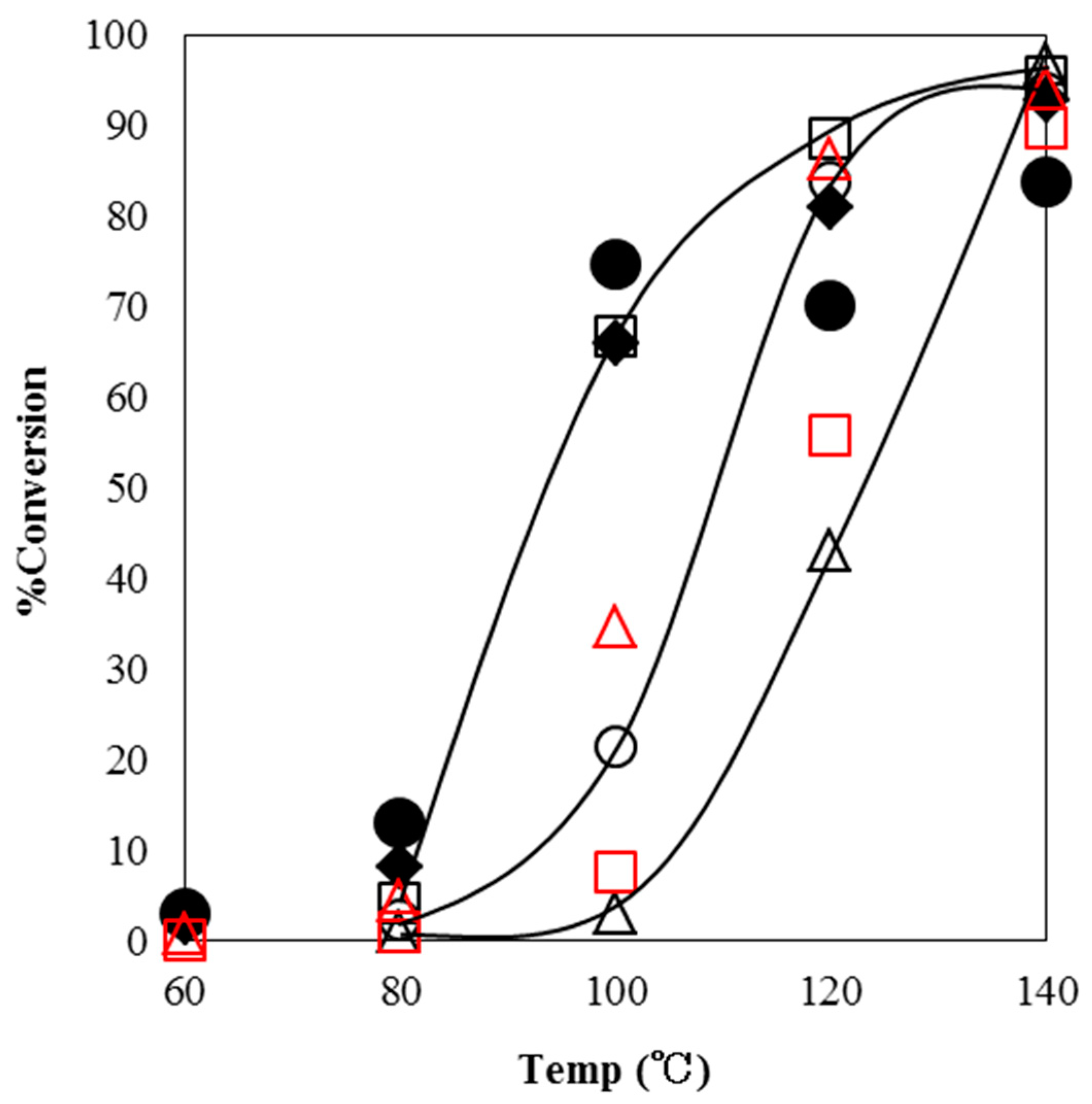
Figure 3 shows the conversions of GPE in the reaction with α-ZrP·Im, α-ZrP·2MIm, or α-ZrP·2E4MIm at these temperatures over a 1 h period. In each case, GPE did not substantially change below 80 °C. Particularly in the case of α-ZrP·Im, the reaction did not proceed at 100 °C. With an increase in temperature, the reactions with any α-ZrP intercalation compound gradually proceeded and reached over 94% of conversions at 140 °C. These α-ZrP intercalation compounds were thermally stable and required 140 °C of curing temperature as initiators. Commercially available latent thermal curing agents such as the microencapsulated imidazoles of HX-3088 and HX-3722 also did not effectively initiate the reaction up to 80 °C. When the temperature was increased, the reaction fairly proceeded at 100 °C with 66% conversion for HX-3088 and 75% conversion for HX-3722. However, these initiators required 140 °C of curing temperature as well as the imidazoles-intercalated α-ZrP. Arimitsu et al. reported that the imidazole derivatives prepared from fumarate ester as latent thermal initiators of butyl glycidyl ether. The reaction completely proceeded at 150 °C for 30 min [
16].
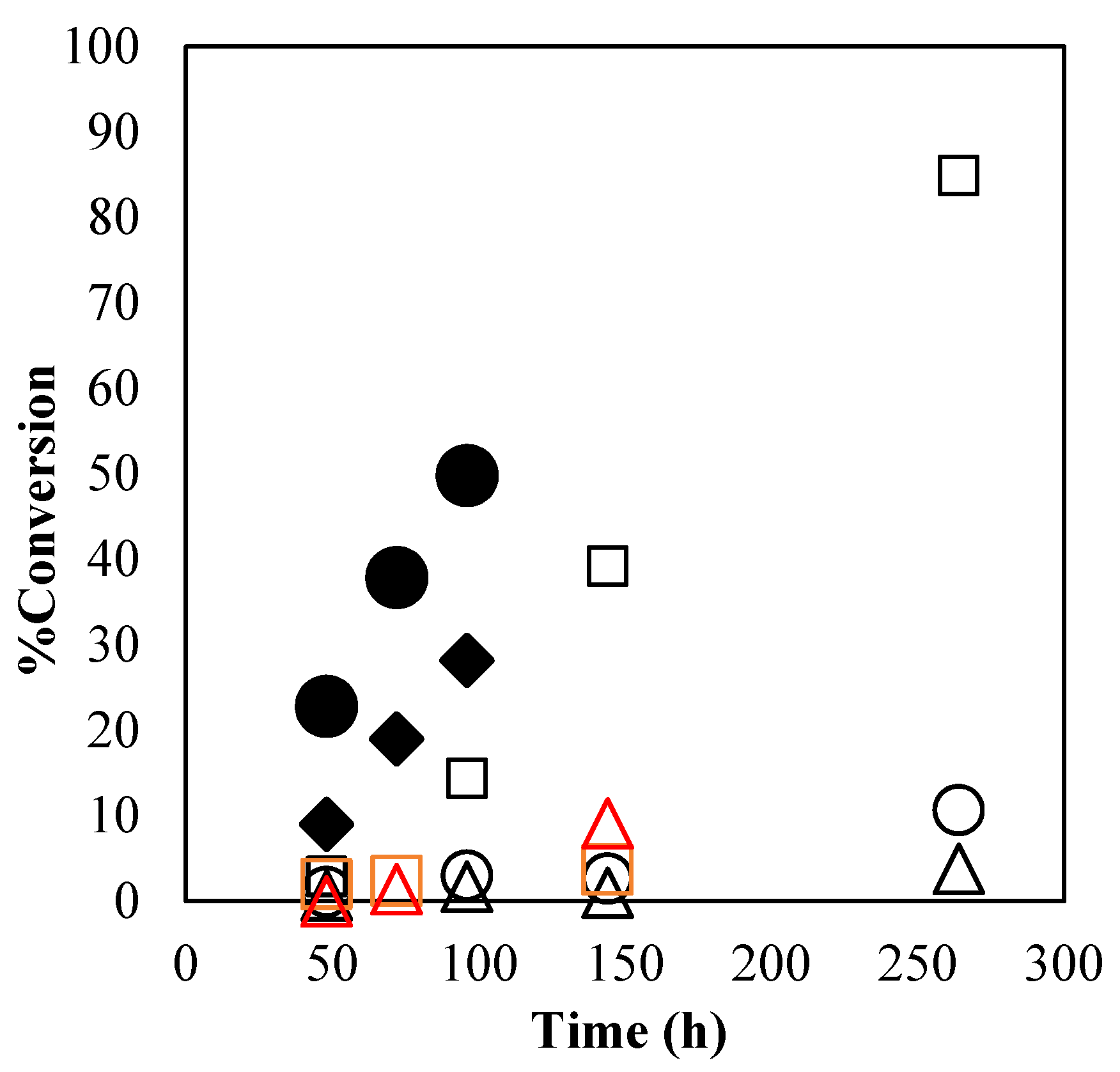
The thermal stabilities of α-ZrP·Im, α-ZrP·2MIm, α-ZrP·2E4MIm, HX-3088, and HX-3722 were tested with the following conversion of GPE at 40 °C as shown in
Figure 4. With α-ZrP·2MIm, the conversion was less than 15% up to 96 h. In the cases of α-ZrP·Im and α-ZrP·2E4MIm, the conversion reached less than 15% at 264 h. The thermal stabilities of α-ZrP·Im, α-ZrP·2MIm, and α-ZrP·2E4MIm were superior to those of HX-3088 and HX-3722.
This reaction system of GPE-MHHPA has an advantage for characterization of the resulting imidazoles-intercalated α-ZrPs after the reaction. Thus, the resulting ones can be easily isolated by simply washing the products with an organic solvent. After the reaction of GPE with MHHPA in the presence of imidazoles-intercalated α-ZrPs at 140 °C for 2 h or 1 h, the resulting imidazoles-intercalated α-ZrPs (hereafter abbreviated as α-ZrP·Im-RXN, α-ZrP·2MIm-RXN, and α-ZrP·2E4MIm-RXN) were isolated and characterized by XRD, NMR, and elemental analyses. Basal distances, chemical shifts of the main signals, and elemental analyses are listed in
Table 1.
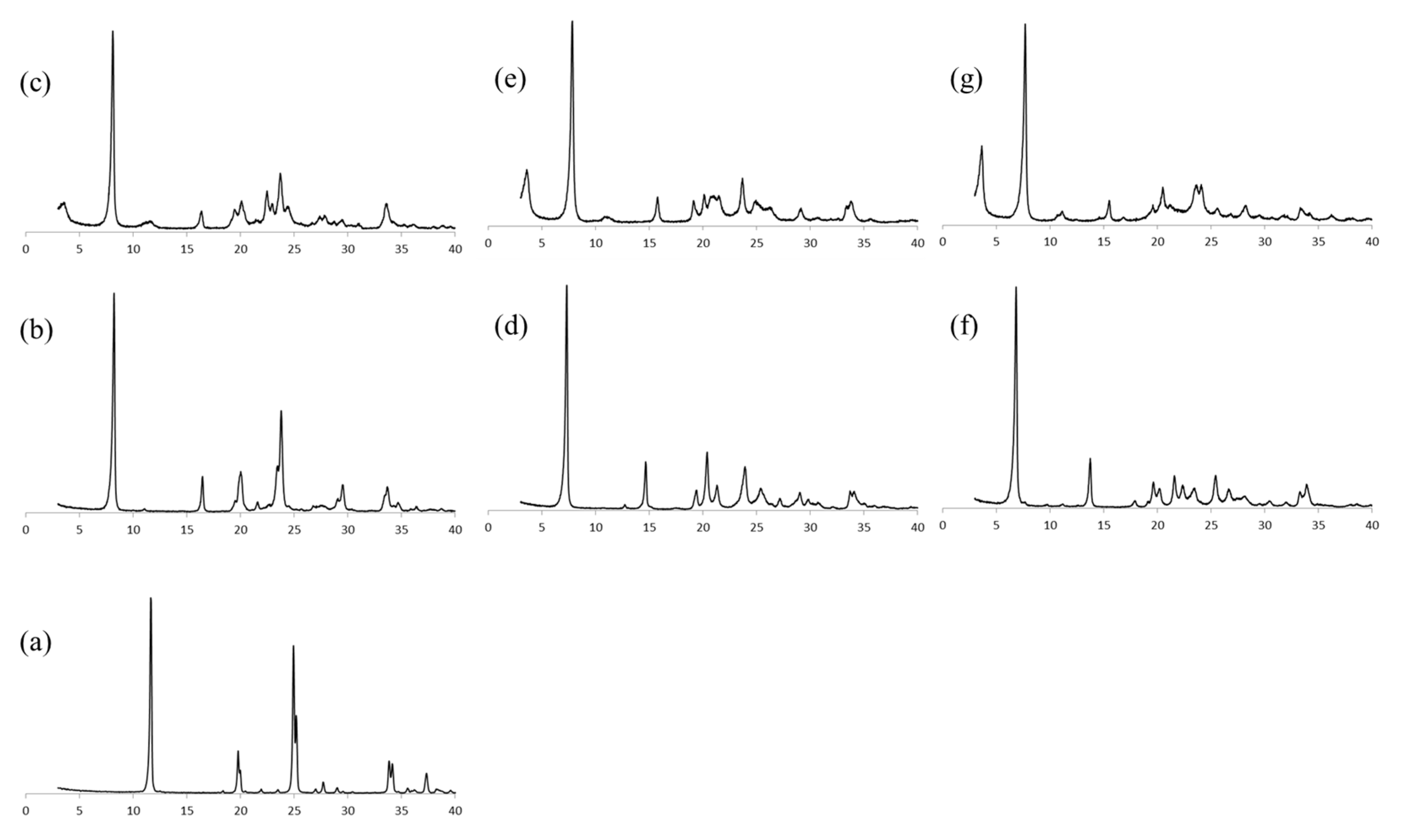
The XRD patterns of pristine α-ZrP is shown in
Figure 5a. The peak corresponding to the basal distance of 7.6 Å was expanded with the intercalation of imidazoles. The XRD patterns of α-ZrP·Im and α-ZrP·Im-RXN are shown in
Figure 5b,c. In addition to the peak corresponding to 10.7 Å of α-ZrP·Im, a small broad peak was observed at 24.4 Å (2θ = 3.6) in that of α-ZrP·Im-RXN. In the
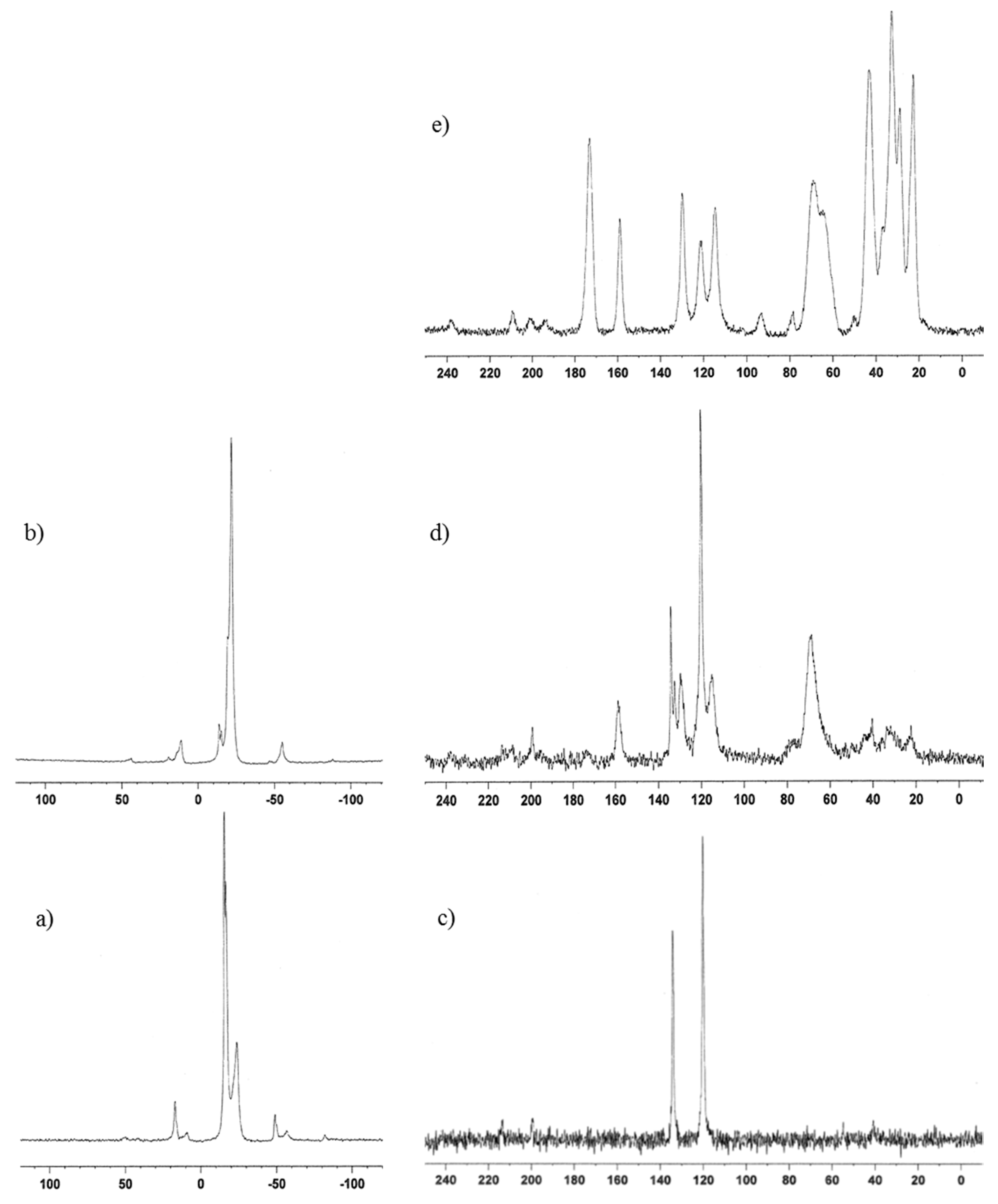
31P MAS NMR spectra of α-ZrP·Im and α-ZrP·Im-RXN, the main signals at −15.9, −16.8, and −23.7 ppm shifted to −16.1 and −22.0 ppm as shown in
Figure 6a,b. In the
31P MAS NMR spectra of intercalation compounds of α-ZrP·Im and α-ZrP·2E4MIm, the peak of deprotonated phosphate groups were observed at δ value of −15.9 and −16.8 for α-ZrP·Im and −16.1 for α-ZrP·2E4MIm. The single peak of pristine α-ZrP is shown at a δ value of −20.1 [
10]. The peak δ value of higher than that of −20.1 for α-ZrP·2MIm doesn’t observed. That might show the separation of 2MIm from phosphate group. In the
13C CPMAS NMR spectra presented in
Figure 6d,e, the aromatic carbons associated with the GPE at δ = 158.8, 132.4, 129.8, and 116.7 ppm and the structural main chain methylene groups at δ = 67.9 were observed. However, the carbonyl carbon of the resulting poly(GPE-co-MHHPA) at δ = 172.9 in
Figure 6e was not observed in that of α-ZrP·Im-RXN. The resulting products were derived from GPE. A possible phosphate ester formed by PO
4 of α-ZrP with GPE may be denied by the chemical shifts in the
13C CP MAS NMR spectra. Phosphate esters produced by the reaction of α-ZrP and 1,2-epoxydodecane, the carbons generated from epoxide were observed at δ = 61.6 and 71.2 for β-cleavage and δ = 79.6 and 71.2 for α-cleavage in 13C CP MAS NMR spectra [
25]. Epoxy-ring opening products, such as the homo-oligomer of GPE, exist in the interlayer. In the following cases, the epoxy-ring opening products are formed in the interlayer.
In the XRD patterns of α-ZrP·2MIm and α-ZrP·2MIm-RXN (shown in
Figure 5d,e), the basal distances were shortened from 12.1 to 11.3 Å and a broad peak was observed at 24.4 Å (2θ = 3.6). In the
31P MAS NMR spectra, the main signals at −21.0, −22.2, and −23.4 ppm shifted to −21.5 ppm. Similarly, the aromatic carbons and methylene groups derived from GPE were observed in the
13C CPMAS NMR spectra.
In the case of α-ZrP·2E4MIm, similar to α-ZrP·2MIm, the basal distances were shortened from 12.9 to 11.5 Å and a broad peak was observed at 24.1 Å (2θ = 3.7). In the 31P MAS NMR spectrum, the main signals at −16.1, and −22.2 ppm shifted to −14.8 and −20.6 ppm. In the 13C CPMAS NMR spectrum, the aromatic carbons and methylene groups derived from GPE were similarly observed.
In any case, with the expansion of the basal distances, an increase in C content was recognized. Moreover, the
13C CPMAS NMR suggested the presence of substances derived from GPE in the interlayer. Based on the elemental analyses, the compositions were calculated to give GPE/Im = 0.7, Zr(HPO
4)
2(C
3H
4N
2)
0.51·(GPE)
0.34 for α-ZrP·Im-RXN, GPE/2MIm = 0.8, Zr(HPO
4)
2(C
4H
6N
2)
0.50·(GPE)
0.42 for α-ZrP·2MIm-RXN, and GPE/2E4MIm = 1.5, Zr(HPO
4)
2(C
6H
10N
2)
0.41·(GPE)
0.61 for α-ZrP·2E4MIm-RXN. Thus, 35% of imidazole, 48% of 2-methylimidazole, or 37% of 2-ethyl-4-methylimidazole was deintercalated in each case. These deintercalated imidazoles initiated the copolymerization of GPE and MHHPA. Since there was not a substantial difference among the catalytic activities of imidazole, 2-methylimidazole, and 2-ethyl-4-methyl imidazole in this reaction, the amount of deintercalated imidazoles must affect reactivity (as shown in
Figure 3) and thermal stability (as shown in
Figure 4). In fact, the order of thermal instability and that of reactivity were α-ZrP·2MIm > α-ZrP·2E4MIm > α-ZrP·Im, which corresponded to the amount of the deintercalated imidazoles.
In these cases, while the imidazoles were deintercalated, the intercalation of GPE and successive reactions occurred. In the preceding paper, the interlayer polymerization of GPE- MHHPA with α-ZrP·DBU; in contrast, the lack of interlayer polymerization with α-ZrP·DABCO were observed [
11]. The compositions after the polymerization of α-ZrP·DBU and α-ZrP·DABCO were calculated based on the contents of N% and H% of the elemental analyses. The results are as follows: α-ZrP·DBU (basal distance: 20.0A): Zr(HPO
4)
2(C
9H
16N
2)
0.83·2.8H
2O, α-ZrP·DBU-RXN: GPE-MHHPA/DBU = 3.7, Zr(HPO
4)
2(C
9H
16N
2)
0.24·(GPE-MHHPA)
0.89, α-ZrP·DABCO (basal distance: 16.1A): Zr(HPO
4)
2(C
6H
12N
2)
0.69·1.5H
2O, α-ZrP·DABCO-RXN: Zr(HPO
4)
2(C
6H
12N
2)
0.63·(GPE-MHHPA)
0.03. Thus, 72% of DBU and 9% of DABCO were deintercalated in these cases; at the same time, in TG-DTA data of imidazoles-, DBU-, and DABCO-intercalated ZrPs, these amines appeared not to be deintercalated up to 140 °C, and sufficient deintercalation of the amines in the reactants took place at 140 °C. Accordingly, when the intercalated amine was sufficiently deintercalated to afford enough space for GPE, the interlayer reaction occurred to expand the basal distance.
In our system, control of the deintercalation of amines was an important factor in the design of the latent thermal initiator. In addition to the pKa of the amines, the basal distances related to the molecular shape and size of the amines, the shape and size of α-zirconium phosphate, the structural design of the intercalation compounds, and the equilibrium of the reactants should be thermodynamically considered.
3. Experimental Section
3.1. Materials
Zr(HPO4)2·H2O (CZP-100) was purchased from Daiichi Kigenso Kagaku Kogyo Co., Ltd. (Osaka, Japan); GPE from Aldrich Chemical Co., Inc. (Tokyo, Japan); imidazole, 2-methylimidazole, 2-ethyl-4-methylimidazole, and MHHPA from Tokyo Chemical Industries, Co., Ltd. (Tokyo, Japan); HX-3088 and HX-3722 from Asahi Kasei Co. Ltd. (Tokyo, Japan). Solvents were used as received without further purification.
3.2. Measurements
X-ray diffraction (XRD) patterns were obtained using a Rigaku RINT2200 (Rigaku CO., Ltd., Tokyo, Japan) with Cu Kα radiation over a scan range of 3–40° at a rate of 2° min−1. NMR spectra in solution were recorded on a Varian Unity-300 spectrometer (Varian Co., Inc., Palo Alto, CA, USA) using tetramethylsilane (TMS) as an internal standard. The contents of imidazoles and water in the intercalation compounds of α-ZrP were measured using a Yanaco CHN Corder MT-5 (Yanako Co., Ltd., Kyoto, Japan). 31P MAS and 13C CPMAS NMR spectra were recorded on a Bruker AVANCEIII600WB NMR spectrometer (Bruker Co. Inc., Billerica, MA, USA). Thermogravimetric analyses (TGA) were carried out with a Shimadzu DTG-60 (Shimadzu Co., Ltd., Kyoto, Japan) at a heating rate of 10 °C/min under nitrogen. Gel permeation chromatographic analyses (GPC) were carried out on a Shodex GPC-101 (LF804*3 and KF-800RF*3, THF as eluent) (Showa Denko Co. Ltd., Tokyo, Japan) using polystyrene standards.
3.3. Preparation of Imidazole-Intercalated α-ZrP (α-ZrP·Im)
The intercalation of imidazole into the layers of Zr(HPO
4)
2·H
2O (α-ZrP) was carried out using a previously reported method [
16]. α-ZrP (10 g) was added to 142 mL of a 0.7 mol dm
−3 imidazole aqueous solution. The reaction mixture was then allowed to stand at 40 °C for 24 h, before the product was collected by centrifugation and washed with water several times. The resulting residue was dried under vacuum. The intercalation of 2-methylimidazole (2MIm) and 2-ethyl-4-methylimidazole (2E4MIm) were carried out by the same procedure for the preparation of α-ZrP·Im.
3.4. Typical Polymerization Procedure
A mixture of GPE (151 mg, 1.0 mmol), MHHPA (172 mg, 1.0 mmol), and intercalation compound of imidazole with α-ZrP (α-ZrP·Im) (11.0 mg, 0.031 mmol, content of imidazole: 0.024 mmol) was heated at 140 °C for 1 h. A small aliquot of the reaction mixture was dissolved in CDCl3, and its 1H-NMR spectra was acquired to determine the extent of the conversion of GPE and MHHPA. In the case of 40 °C, the small aliquot of sample was collected every determined time. The polymerization with intercalation compound of 2-methylimidazole with α-ZrP (α-ZrP·2MIm) (11.7 mg, 0.031 mmol, content of 2-methylimidazole: 0.029 mmol) and intercalation compound of 2-ethyl-4-methylimidazole with α-ZrP (α-ZrP·2E4MIm) (11.2 mg, 0.031 mmol, content of 2-ethyl-4-methylimidazole: 0.020 mmol) were carried out by the same procedure for the preparation of poly(GPE-co-MHHPA) with α-ZrP·Im. Commercial agents of the encapsulation of imidazoles, ca. 10 mg of HX-3088 and HX-3722 were used by the same procedure.
3.5. Polymerization Procedure for Preparation of Poly(GPE-co-MHHPA) with α-ZrP·Im
A mixture of GPE (7.51 g, 50.0 mmol), MHHPA (8.41 g, 50.0 mmol), and α-ZrP·Im (535 mg, 1.50 mmol, content of imidazole: 1.17 mmol) was heated at 140 °C for 2 h. After the reaction, tetrahydrofurane (THF) was added to the mixture and filtered off, and the residue, α-ZrP·Im-RXN, was rinsed to wash the polymer. The obtained α-ZrP·Im-RXN was dried under vacuum and served to measure the XRD analysis. The obtained polymer was isolated by the precipitation with methanol and dried under vacuum with 50% yield. The copolymer was identified as poly(GPE-co-MHHPA) by 1H-NMR spectra. The number-average molecular weight (Mn) and the molecular weight distribution (Mw/Mn) were found to be 1990 and 1.4, respectively (GPC, calibrated by polystyrene standards). The polymerization with α-ZrP·2MIm and α-ZrP·2E4MIm was carried out at 140 °C for 1 h to obtain the corresponding poly(GPE-co-MHHPA). α-ZrP·2MIm: Mn = 2080, Mw/Mn = 1.4, α-ZrP·2E4MIm: Mn = 3120, Mw/Mn = 1.3.