Microwave-Assisted Synthesis of Co3(PO4)2 Nanospheres for Electrocatalytic Oxidation of Methanol in Alkaline Media
Abstract
:1. Introduction
2. Results and Discussion
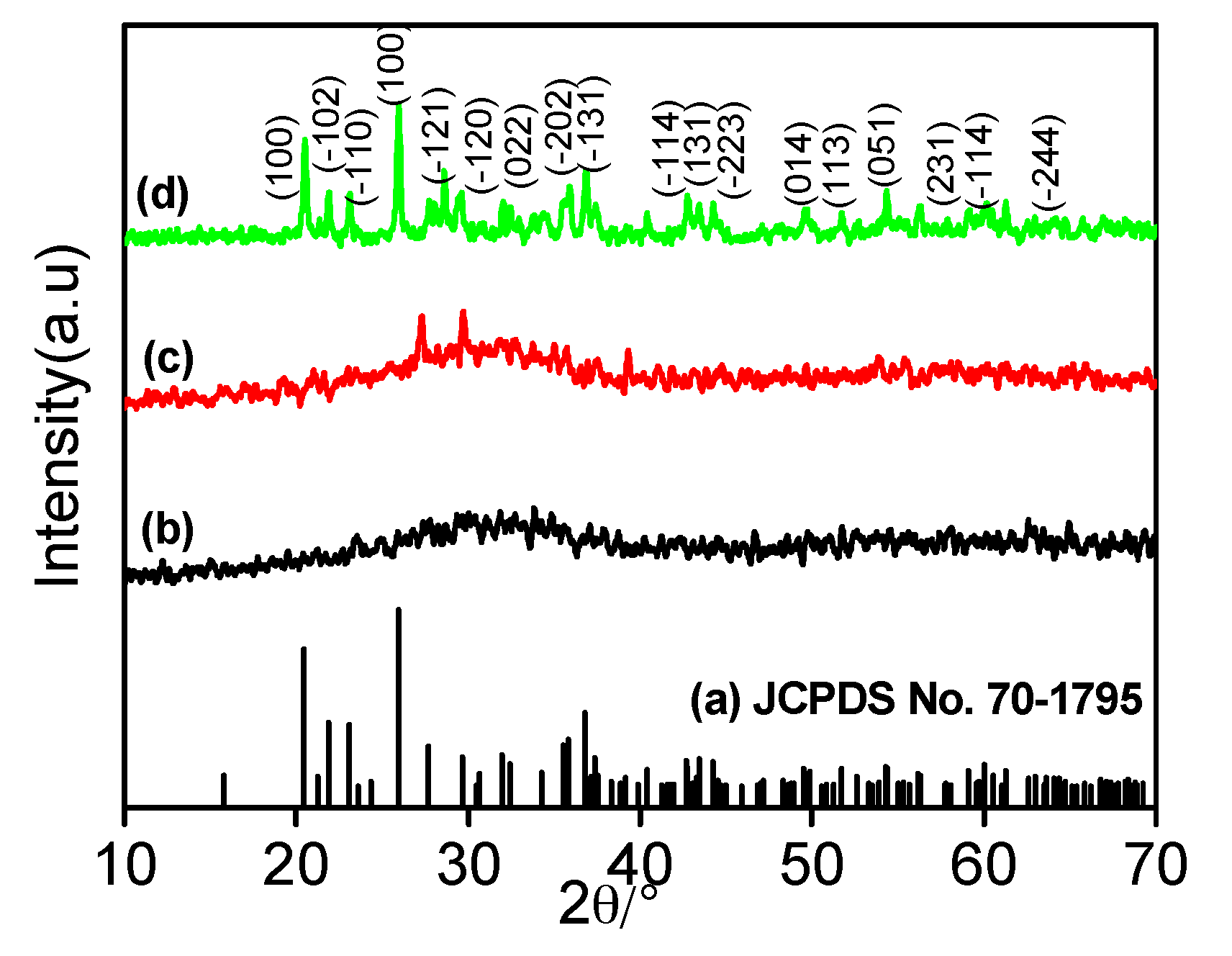
2.1. Powder XRD Analysis
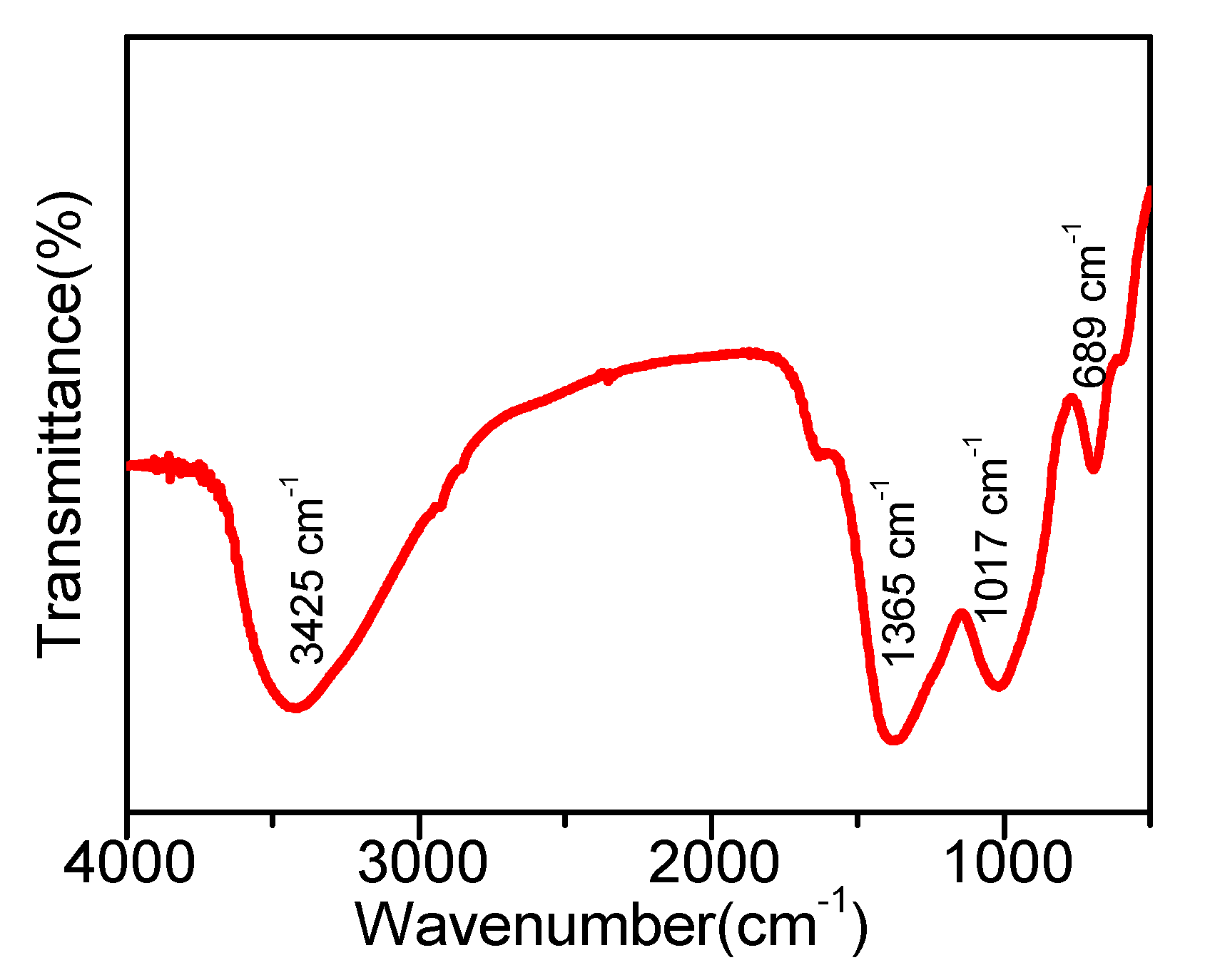
2.2. FT-IR Spectroscopy Analysis
2.3. Morphological Analysis
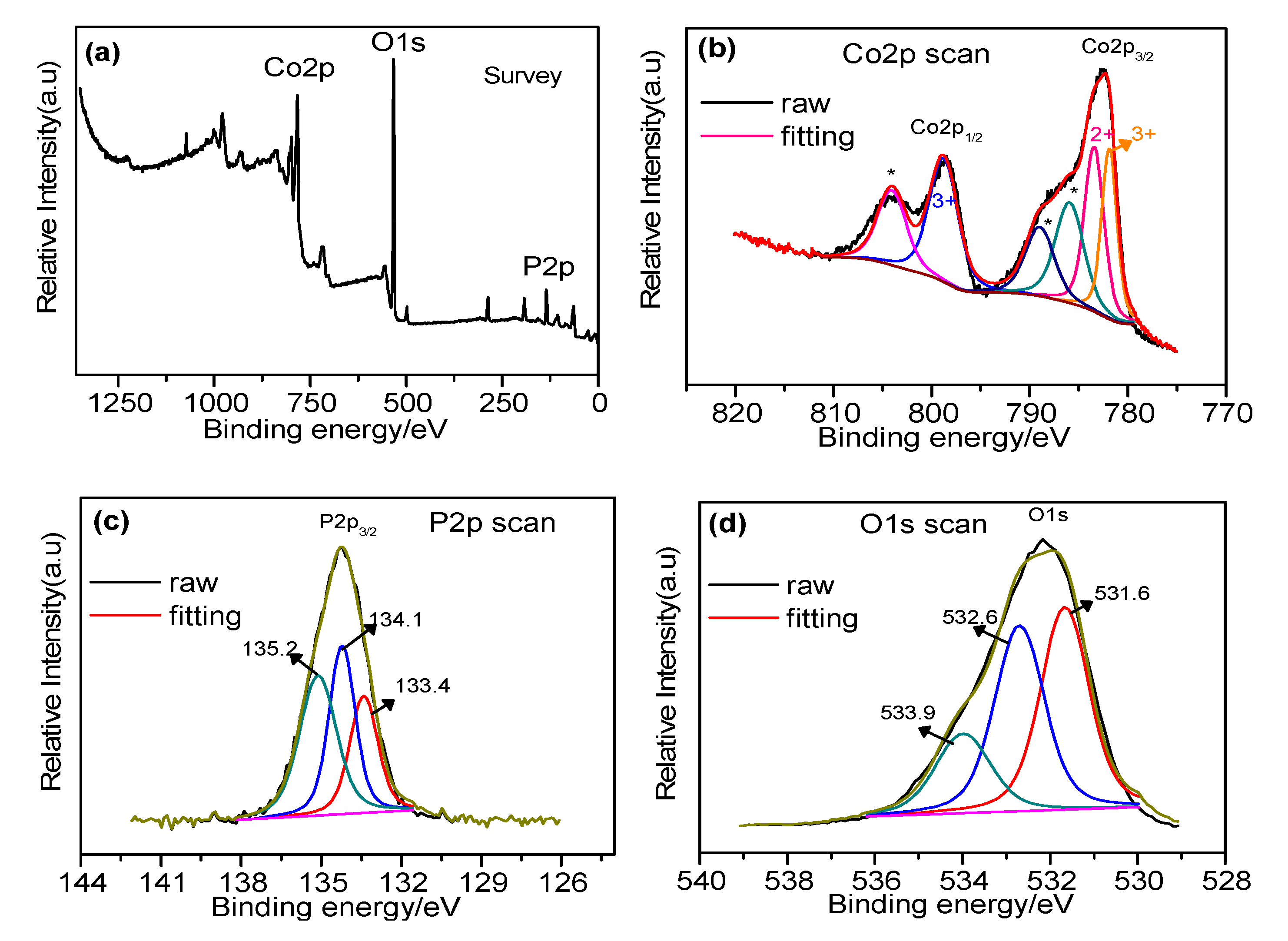
2.4. XPS Analysis
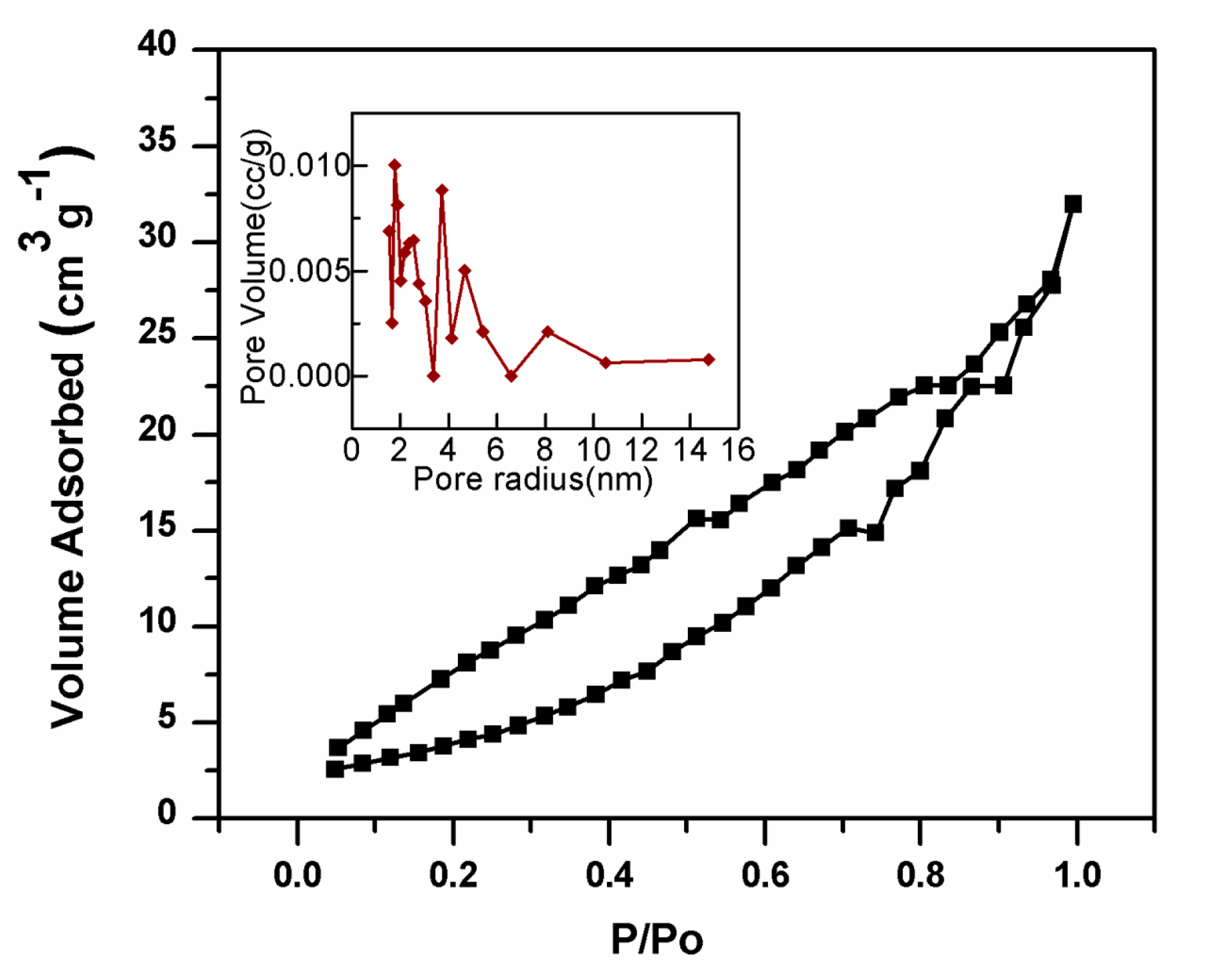
2.5. Textural Properties
2.6. Electrocatalytic Methanol Oxidation over Co3(PO4)2 Nanospheres
3. Experimental
3.1. Microwave-Assisted Synthesis of Co3(PO4)2 Nanospheres
3.2. Instrumental Characterization
3.3. Electrochemical Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Peng, L.; Gan, L.; Wei, Y.; Yang, H.; Li, J.; Du, H.; Kang, F. Pt submonolayers on au nanoparticles: coverage-dependent atomic structures and electrocatalytic stability on methanol oxidation. J. Phys. Chem. C 2016, 120, 28664–28671. [Google Scholar] [CrossRef]
- Arunachalam, P.; Zhang, S.; Abe, T.; Komura, M.; Iyoda, T.; Nagai, K. Weak visible light (~mW/cm2) organophotocatalysis for mineralization of amine, thiol and aldehyde by biphasic cobalt phthalocyanine/fullerene nanocomposites prepared by wet process. Appl. Catal. B 2016, 193, 240–247. [Google Scholar] [CrossRef]
- Puthiyapura, V.K.; Brett, D.J.L.; Russell, A.E.; Lin, W.F.; Hardacre, C. Biobutanol as fuel for direct alcohol fuel cells—Investigation of Sn-modified Pt catalyst for butanol electro-oxidation. ACS Appl. Mater. Interfaces 2016, 8, 12859–12870. [Google Scholar] [CrossRef] [PubMed]
- Prathap, M.U.A.; Satpati, B.; Srivastava, R. Facile preparation of β-Ni(OH)2-NiCo2O4 hybrid nanostructure and its application in the electro-catalytic oxidation of methanol. Electrochim. Acta 2014, 130, 368–380. [Google Scholar] [CrossRef]
- Noroozifar, M.; Yavari, Z.; Motlagh, M.K.; Ghasemi, T.; Yazdi, S.H.R.; Mohammadi, M. Fabrication and performance evaluation of a novel membrane electrode assembly for DMFCs. RSC Adv. 2016, 6, 563–574. [Google Scholar] [CrossRef]
- Tong, Y.Y.; Gu, C.D.; Zhang, J.L.; Tang, H.; Li, Y.; Wang, X.L.; Tu, J.P. Urchin-like Ni-Co-P-O nanocomposite as novel methanol electro-oxidation materials in alkaline environment. Electrochim. Acta 2016, 187, 11–19. [Google Scholar] [CrossRef]
- Ghanem, M.A.; Al-Mayouf, A.M.; Arunachalam, P.; Abiti, T. Mesoporous cobalt hydroxide prepared using liquid crystal template for efficient oxygen evolution in alkaline media. Electrochim. Acta 2016, 207, 177–186. [Google Scholar] [CrossRef]
- Atar, N.; Eren, T.; Yola, M.L.; Karimi-Maleh, H.; Demirdögen, B. Magnetic iron oxide and iron oxide@gold nanoparticle anchored nitrogen and sulfur-functionalized reduced graphene oxide electrocatalyst for methanol oxidation. RSC Adv. 2015, 5, 26402–26409. [Google Scholar] [CrossRef]
- Arunachalam, P.; Ghanem, M.A.; Al-Mayouf, A.M.; Al-shalwi, M.; Abd-Elkader, O.H. Microwave assisted synthesis and characterization of Ni/NiO nanoparticles as electrocatalyst for methanol oxidation in alkaline solution. Mater. Res. Express 2017, 4, 025035. [Google Scholar] [CrossRef]
- Sun, J.; Dou, M.; Zhang, Z.; Ji, J.; Wang, F. Carbon nanotubes supported Pt-Co-P ultrafine nanoparticle electrocatalysts with superior activity and stability for methanol electro-oxidation. Electrochim. Acta 2016, 215, 447–454. [Google Scholar] [CrossRef]
- Gao, M.R.; Gao, Q.; Jiang, J.; Cui, C.H.; Yao, W.T.; Yu, S.H. A methanol-tolerant Pt/CoSe2 nanobelt cathode catalyst for direct methanol fuel cells. Angew. Chem. 2011, 123, 5007–5010. [Google Scholar] [CrossRef]
- Shi, M.; Zhang, W.; Li, Y.; Chu, Y.; Ma, C. Tungsten carbide-reduced graphene oxide intercalation compound as co-catalyst for methanol oxidation. Chin. J. Catal. 2016, 37, 1851–1859. [Google Scholar] [CrossRef]
- Cui, P.; He, H.; Liu, H.; Zhang, S.; Yang, J. Heterogeneous nanocomposites of silver selenide and hollow platinum nanoparticles toward methanol oxidation reaction. J. Power Sources 2016, 327, 432–437. [Google Scholar] [CrossRef]
- Yang, C.; Srinivasan, S.; Aricò, A.S.; Cretı, P.; Baglio, V.; Antonucci, V. Composite nafion/zirconium phosphate membranes for direct methanol fuel cell operation at high temperature. Electrochem. Solid-State Lett. 2001, 4, A31–A34. [Google Scholar] [CrossRef]
- Tan, J.; Yang, J.H.; Liu, X.; Yang, F.; Li, X.; Ma, D. Electrochemical oxidation of methanol on mesoporous nickel phosphates and Si-incorporated mesoporous nickel phosphates. Electrochem. Commun. 2013, 27, 141–143. [Google Scholar] [CrossRef]
- Kiyani, R.; Rowshanzamir, S.; Parnian, M.J. Nitrogen doped graphene supported palladium-cobalt as a promising catalyst for methanol oxidation reaction: Synthesis, characterization and electrocatalytic performance. Energy 2016, 113, 1162–1173. [Google Scholar] [CrossRef]
- Kepeniene, V.; Tamasiunait, L.T.; Jablonskiene, J.; Semasko, M.; Vaiciunien, J.; Vaitkus, R.; Norkus, E. One-pot synthesis of graphene supported platinum cobalt nanoparticles as electrocatalysts for methanol oxidation. Mater. Chem. Phys. 2016, 171, 145–152. [Google Scholar] [CrossRef]
- Poon, K.C.; Khezri, B.; Li, Y.; Webster, R.D.; Suc, H.; Sato, H. A highly active Pd–P nanoparticle electrocatalyst for enhanced formic acid oxidation synthesized via stepwise electroless deposition. Chem. Commun. 2016, 52, 3556–3559. [Google Scholar] [CrossRef] [PubMed]
- Tan, D.C.L.; Khezri, B.; Amatyakul, W.; Webster, R.D.; Sato, H. A facilely synthesized highly active Pd nanoparticle electrocatalyst for electroless deposition process. RSC Adv. 2015, 5, 88805–88808. [Google Scholar] [CrossRef]
- Li, T.; Fu, G.; Su, J.; Wang, Y.; Lv, Y.; Zou, X.; Zhu, X.; Xu, L.; Sun, D.; Tang, Y. Carbon supported ultrafine gold phosphorus nanoparticles as highly efficient electrocatalyst for alkaline ethanol oxidation reaction. Electrochim. Acta 2017, 231, 13–19. [Google Scholar] [CrossRef]
- Poon, K.C.; Tan, D.C.L.; Vo, T.D.T.; Khezri, B.; Su, H.; Webster, R.D.; Sato, H. Newly developed stepwise electroless deposition enables a remarkably facile synthesis of highly active and stable amorphous Pd nanoparticle electrocatalysts for oxygen reduction reaction. J. Am. Chem. Soc. 2014, 136, 5217–5220. [Google Scholar] [CrossRef] [PubMed]
- Zhang, K.; Wang, C.; Bin, D.; Wang, J.; Yan, B.; Shiraishi, Y.; Du, Y. Fabrication of Pd/P nanoparticle networks with high activity for methanol oxidation. Catal. Sci. Technol. 2016, 6, 6441–6447. [Google Scholar] [CrossRef]
- Zhang, M.; Yan, Z.; Li, Y.; Jing, J.; Xie, J. Preparation of cobalt silicide on graphene as Pt electrocatalyst supports for highly efficient and stable methanol oxidation in acidic media. Electrochim. Acta 2015, 161, 48–54. [Google Scholar] [CrossRef]
- Theerthagiri, J.; Thiagarajan, K.; Senthilkumar, B.; Khan, Z.; Senthil, R.A.; Arunachalam, P.; Madhavan, J.; Ashokkumar, M. Synthesis of hierarchical cobalt phosphate nanoflakes and their enhanced electrochemical performances for supercapacitor applications. ChemistrySelect 2016, 1, 1–11. [Google Scholar] [CrossRef]
- Combes, C.; Rey, C. Amorphous calcium phosphates: Synthesis, properties and uses in biomaterials. Acta Biomater. 2010, 6, 3362–3378. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.H.; Jeong, J.M.; Lee, S.J.; Choi, B.G.; Lee, K.G. Protein-directed assembly of cobalt phosphate hybrid nanoflowers. J. Colloid Interface Sci. 2016, 484, 44–50. [Google Scholar] [CrossRef] [PubMed]
- Theerthagiri, J.; Senthil, R.A.; Buraidah, M.H.; Raghavender, M.; Madhavan, J.; Arof, A.K. Synthesis and characterization of (Ni1−xCox)Se2 based ternary selenides as electrocatalyst for triiodide reduction in dye-sensitized solar cells. J. Solid State Chem. 2016, 238, 113–120. [Google Scholar] [CrossRef]
- Arunachalam, P.; Ghanem, M.A.; Al-Mayouf, A.M.; Al-shalwi, M. Enhanced electrocatalytic performance of mesoporous nickel-cobalt oxide electrode for methanol oxidation in alkaline solution. Mater. Lett. 2017, 196, 365–368. [Google Scholar] [CrossRef]
- Al-Enizia, A.M.; Ghanem, M.A.; El-Zatahry, A.A.; Al-Deyab, S.S. Nickel oxide/nitrogen doped carbon nanofibers catalyst for methanol oxidation in alkaline media. Electrochim. Acta 2014, 137, 774–780. [Google Scholar] [CrossRef]
- Hameed, R.M.A.; El-Khatib, K.M. Ni–P and Ni–Cu–P modified carbon catalysts for methanol electro-oxidation in KOH solution. Int. J. Hydrog. Energy 2010, 35, 2517–2529. [Google Scholar] [CrossRef]
- Xu, C.; Cheng, L.; Shen, P.; Liu, Y. Methanol and ethanol electrooxidation on Pt and Pd supported on carbon microspheres in alkaline media. Electrochem. Commun. 2007, 9, 997–1001. [Google Scholar] [CrossRef]
- Asgari, M.; Maragheh, M.G.; Davarkhah, R.; Lohrasbi, E. Methanol electrooxidation on the nickel oxide nanoparticles/multi-walled carbon nanotubes modified glassy carbon electrode prepared using pulsed electrodeposition. J. Electrochem. Soc. 2011, 158, K225–K229. [Google Scholar] [CrossRef]
- Barakat, N.A.M.; El-Newehy, M.; Al-Deyab, S.S.; Kim, H.Y. Cobalt/copper-decorated carbon nanofibers as novel non-precious electrocatalyst for methanol electrooxidation. Nanoscale Res. Lett. 2014, 9, 2. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.; Wang, H.; Zhou, J.; Wang, J.; Duchesne, P.N.; Muir, D.; Zhang, P.; Han, N.; Zhao, F.; Zeng, M.; et al. Highly active and durable methanol oxidation electrocatalyst based on the synergy of platinum–nickel hydroxide–grapheme. Nat. Commun. 2015, 6, 10035. [Google Scholar] [CrossRef] [PubMed]


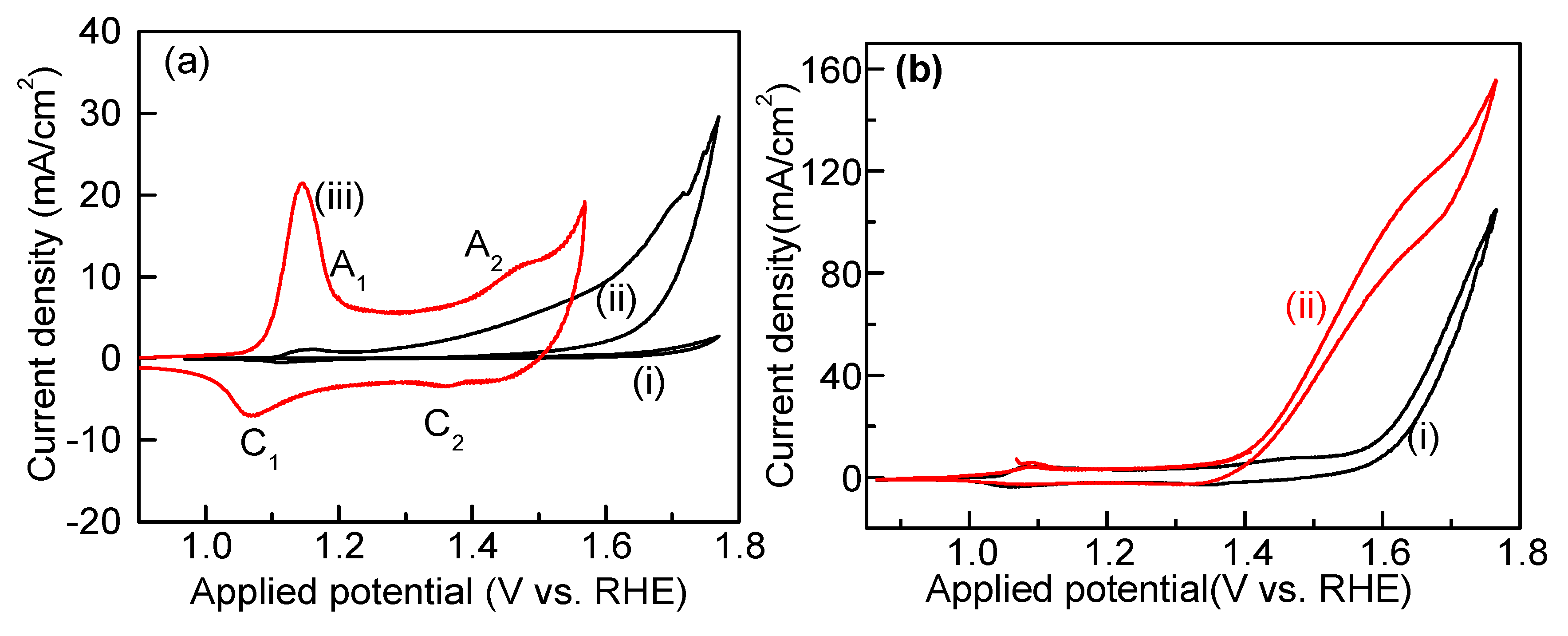
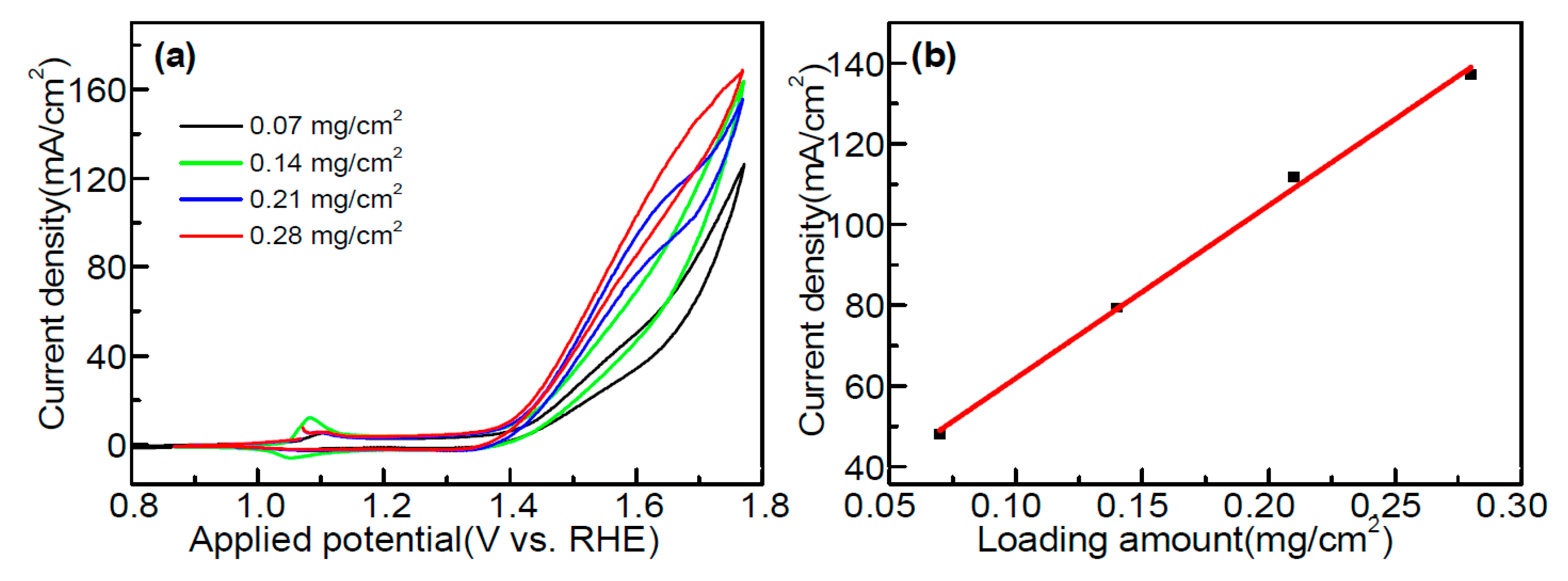
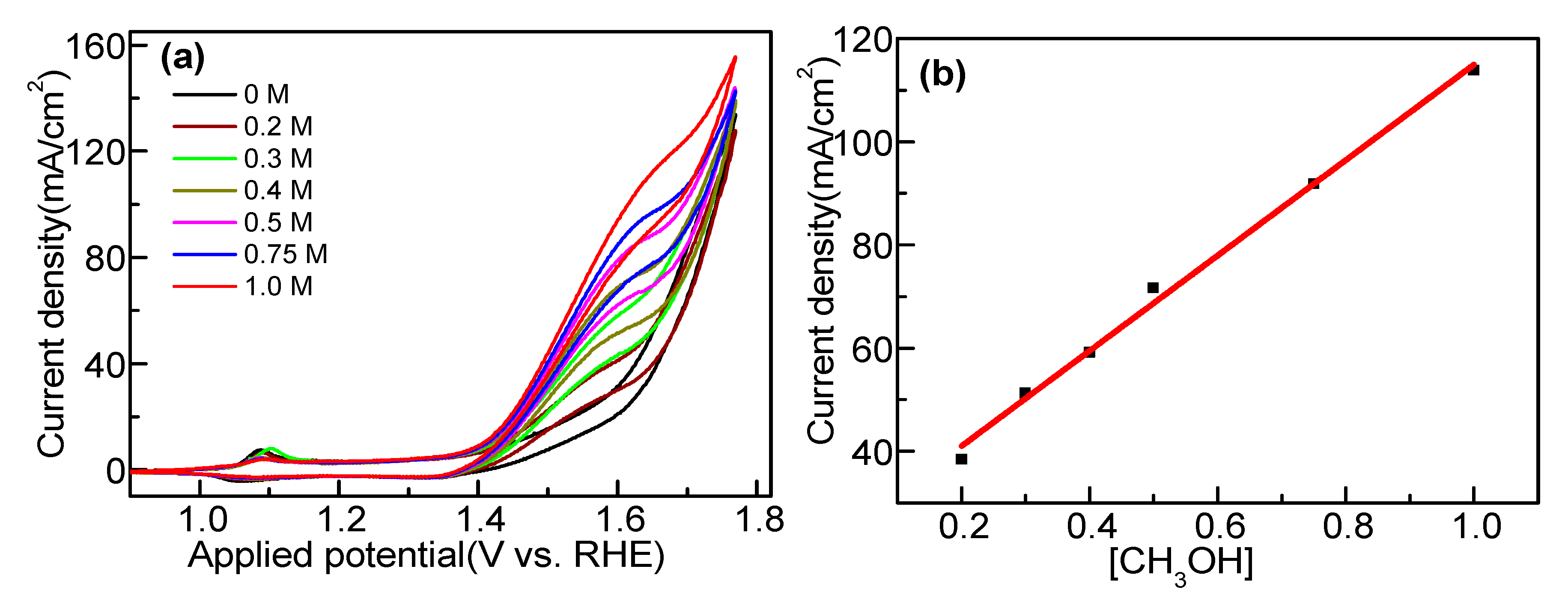
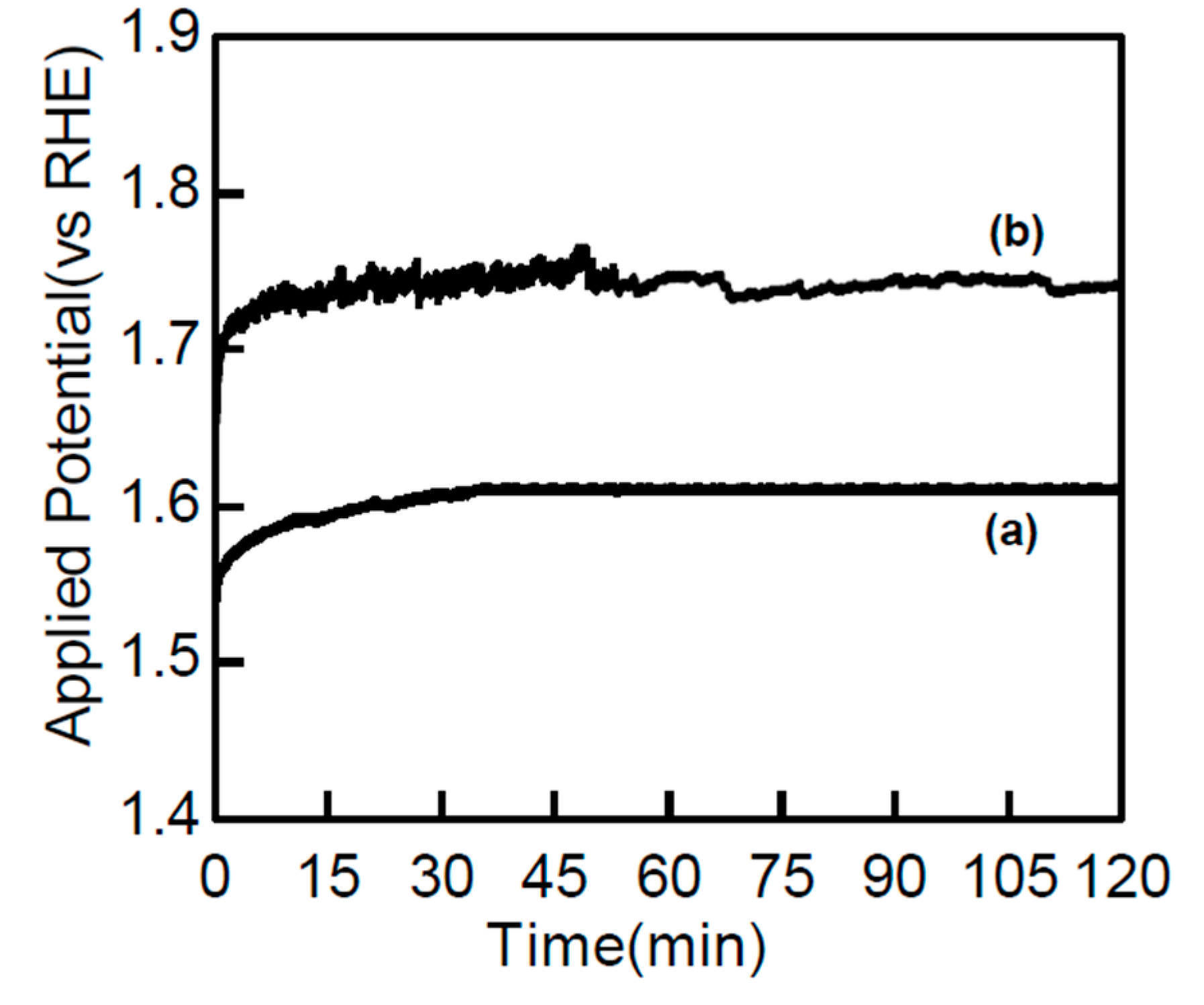
| Sr. No. | Catalyst | Method | Electrolyte (M) | Current Density mA mg−1/mA cm−2 | Ref. |
|---|---|---|---|---|---|
| 1 | Ni-Cu-P/C | Electroless deposition | KOH: 0.1 CH3OH: 0.5 | -/9.5 | [30] |
| 2 | Pt/C | Chemical reduction | KOH: 1 CH3OH: 1 | -/40 | [31] |
| 3 | Pd/C | Hydrothermal | KOH: 1 CH3OH: 1 | -/24 | [31] |
| 4 | NiO/MWCNT | Pulsed Electrodeposition | NaOH: 0.1 CH3OH: 0.1 | -/10 | [32] |
| 5 | Co-Cu-CNF | Electrospining | KOH: 1 CH3OH: 2 | -/16 | [33] |
| 6 | Pt/Ni(OH)2/Graphene | Two-step solution method | KOH: 0.1 CH3OH: 0.5 | 400/- | [34] |
| 7 | Co3O4 | Microwave | KOH: 1 CH3OH: 1 | 335/75 | [28] |
| 8 | Co3(PO4)2 nanospheres | Microwave | KOH: 1 CH3OH: 1 | 556/126 | This work |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Arunachalam, P.; Shaddad, M.N.; Alamoudi, A.S.; Ghanem, M.A.; Al-Mayouf, A.M. Microwave-Assisted Synthesis of Co3(PO4)2 Nanospheres for Electrocatalytic Oxidation of Methanol in Alkaline Media. Catalysts 2017, 7, 119. https://doi.org/10.3390/catal7040119
Arunachalam P, Shaddad MN, Alamoudi AS, Ghanem MA, Al-Mayouf AM. Microwave-Assisted Synthesis of Co3(PO4)2 Nanospheres for Electrocatalytic Oxidation of Methanol in Alkaline Media. Catalysts. 2017; 7(4):119. https://doi.org/10.3390/catal7040119
Chicago/Turabian StyleArunachalam, Prabhakarn, Maged N. Shaddad, Abdullah Salah Alamoudi, Mohamed A. Ghanem, and Abdullah M. Al-Mayouf. 2017. "Microwave-Assisted Synthesis of Co3(PO4)2 Nanospheres for Electrocatalytic Oxidation of Methanol in Alkaline Media" Catalysts 7, no. 4: 119. https://doi.org/10.3390/catal7040119
APA StyleArunachalam, P., Shaddad, M. N., Alamoudi, A. S., Ghanem, M. A., & Al-Mayouf, A. M. (2017). Microwave-Assisted Synthesis of Co3(PO4)2 Nanospheres for Electrocatalytic Oxidation of Methanol in Alkaline Media. Catalysts, 7(4), 119. https://doi.org/10.3390/catal7040119









