Study of the V2O5-WO3/TiO2 Catalyst Synthesized from Waste Catalyst on Selective Catalytic Reduction of NOx by NH3 †
Abstract
:1. Introduction
2. Results and Discussion
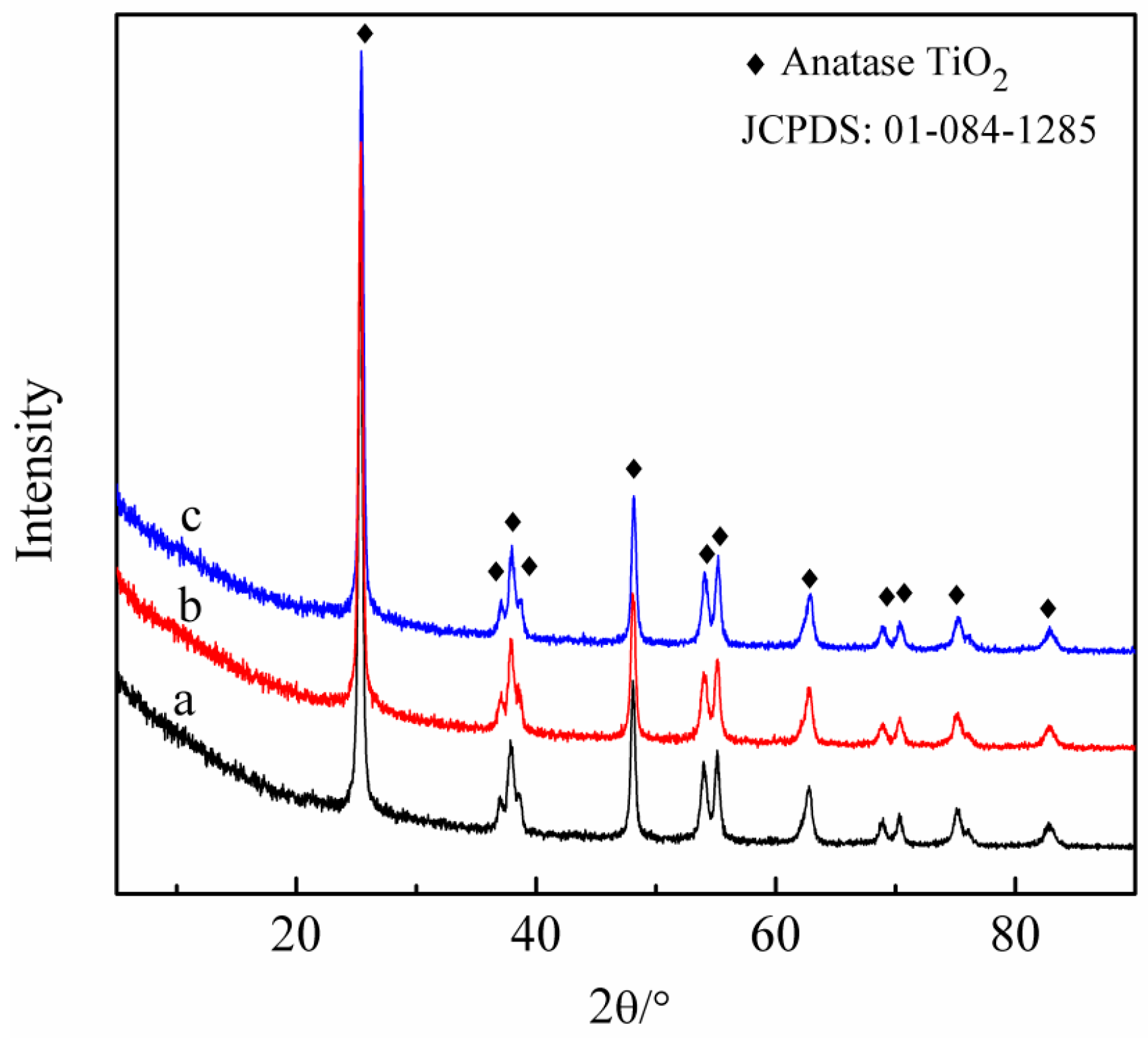
2.1. Characteristics of the Fresh and Waste SCR Catalyst
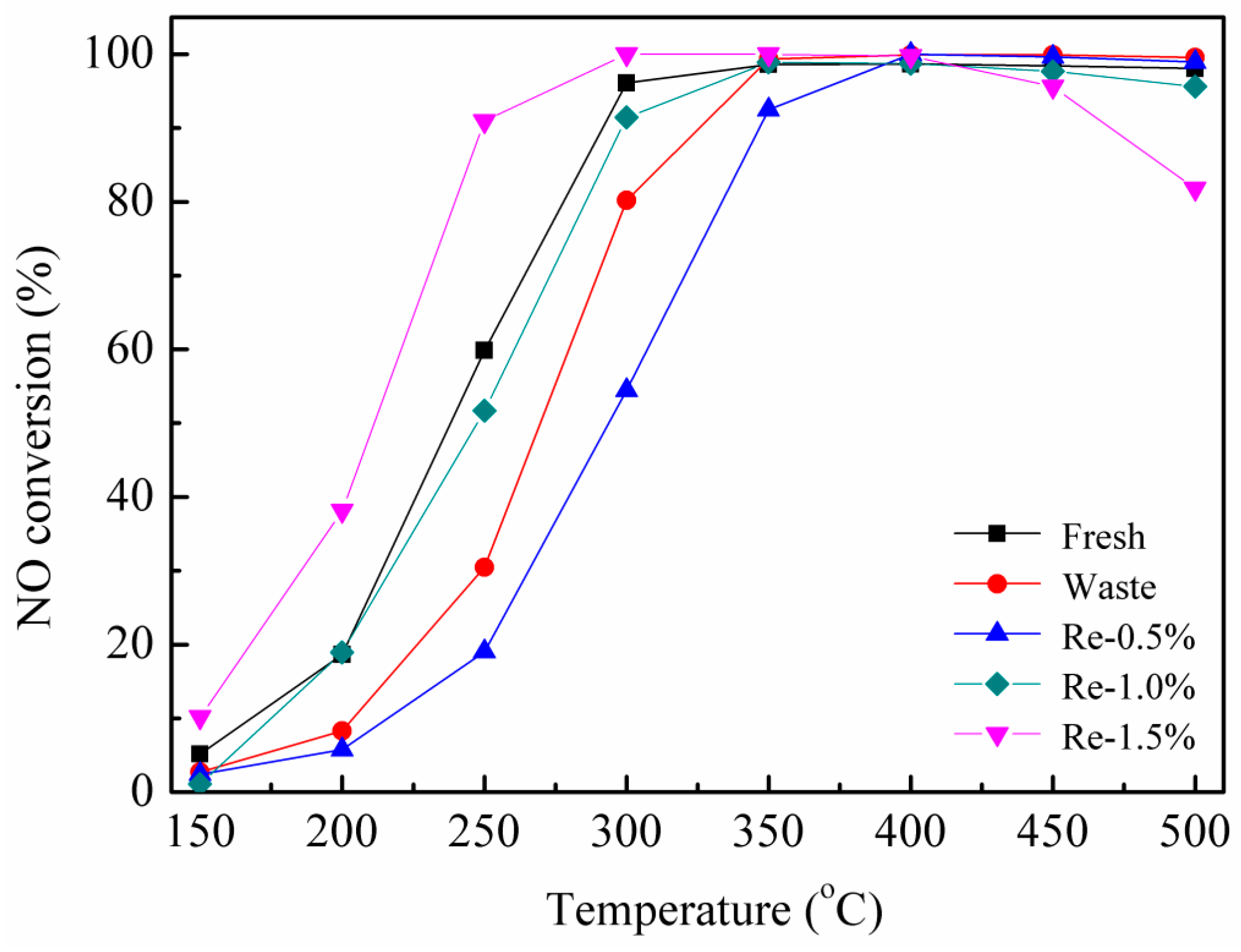
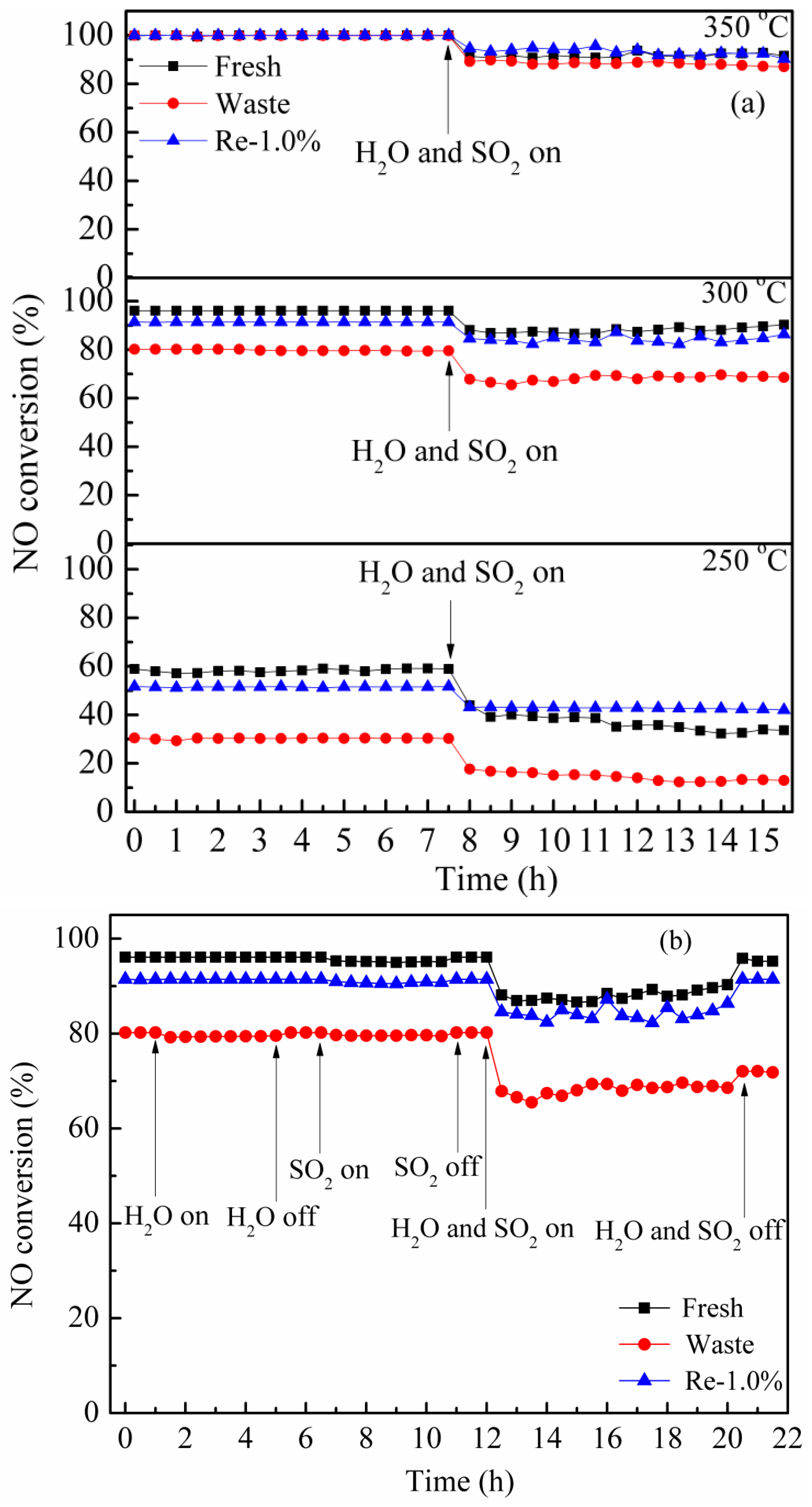
2.2. Catalytic Activity Test
2.3. Textural Properties
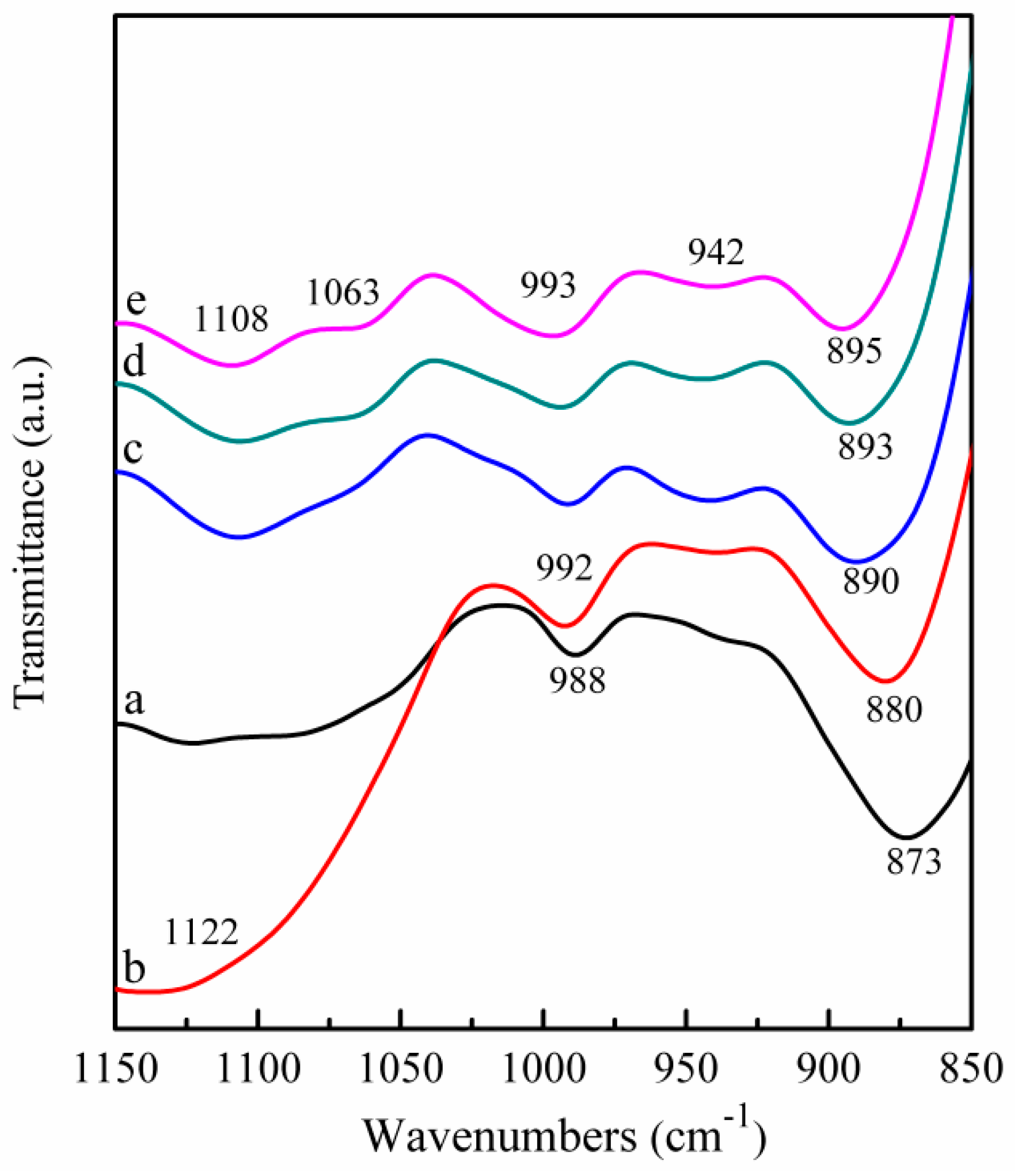
2.4. FT-IR Spectra
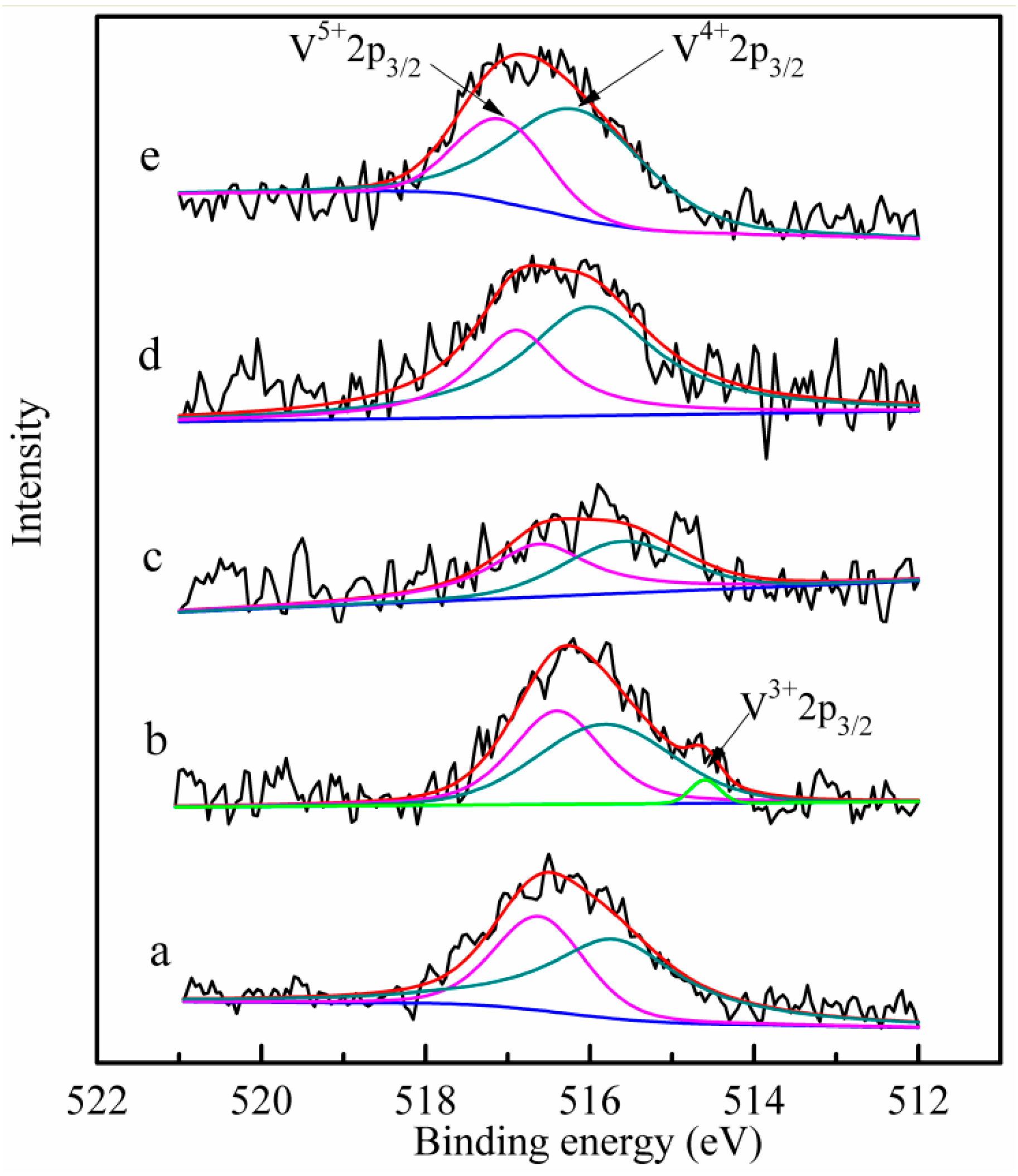
2.5. XPS Analysis
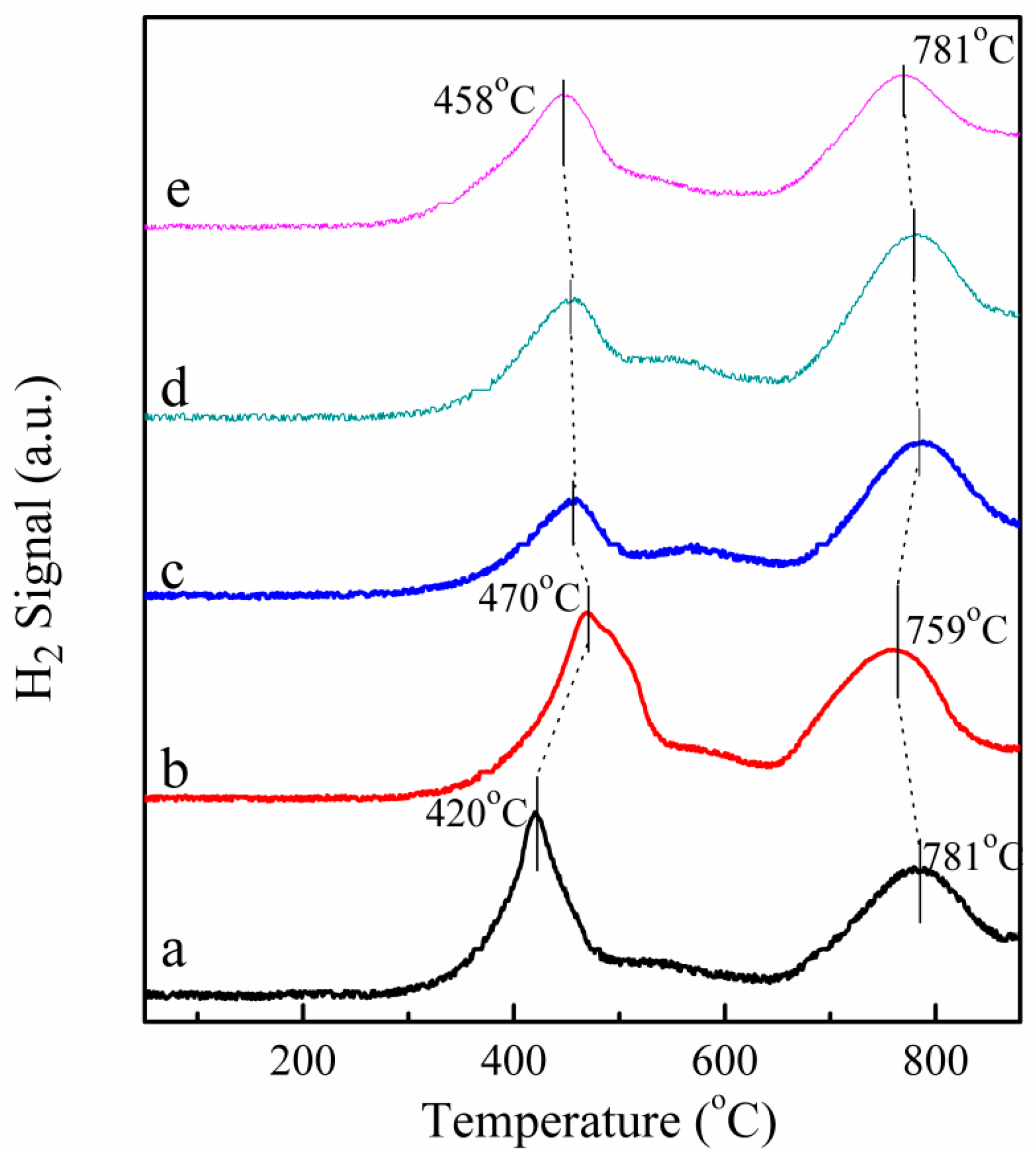
2.6. H2-TPR
3. Experimental
3.1. Raw Materials and Resynthesis Protocols
3.2. Catalytic Activity Test
3.3. Catalyst Characterization
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Forzatti, P. Environmental catalysis for stationary applications. Catal. Today 2000, 62, 51–65. [Google Scholar] [CrossRef]
- Parvulescu, V.; Grange, P.; Delmon, B. Catalytic removal of NO. Catal. Today 1998, 46, 233–316. [Google Scholar] [CrossRef]
- Nicosia, D.; Elsener, M.; Kröcher, O.; Jansohn, P. Basic investigation of the chemical deactivation of V2O5/WO3-TiO2 SCR catalysts by potassium, calcium, and phosphate. Top. Catal. 2007, 42–43, 333–336. [Google Scholar] [CrossRef]
- Klimczak, M.; Kern, P.; Heinzelmann, T.; Lucas, M.; Claus, P. High-throughput study of the effects of inorganic additives and poisons on NH3-SCR catalysts—Part I: V2O5-WO3/TiO2 catalysts. Appl. Catal. B 2010, 95, 39–47. [Google Scholar] [CrossRef]
- Kong, M.; Liu, Q.; Wang, X.; Ren, S.; Yang, J.; Zhao, D.; Xi, W.; Yao, L. Performance impact and poisoning mechanism of arsenic over commercial V2O5-WO3/TiO2 SCR catalyst. Catal. Commun. 2015, 72, 121–126. [Google Scholar] [CrossRef]
- Xi, Y.; Ottinger, N.A.; Liu, Z.G. New insights into sulfur poisoning on a vanadia SCR catalyst under simulated diesel engine operating conditions. Appl. Catal. B 2014, 160–161, 1–9. [Google Scholar] [CrossRef]
- Li, Q.; Chen, S.; Liu, Z.; Liu, Q. Combined effect of KCl and SO2 on the selective catalytic reduction of NO by NH3 over V2O5/TiO2 catalyst. Appl. Catal. B 2015, 164, 475–482. [Google Scholar] [CrossRef]
- Nova, I.; Acqua, L.D.; Lietti, L.; Giamello, E.; Forzatti, P. Study of thermal deactivation of a de-NOx commercial catalyst. Appl. Catal. B 2001, 35, 31–42. [Google Scholar] [CrossRef]
- Khodayari, R.; Odenbrand, C.I. Regeneration of commercial SCR catalysts by washing and sulphation: Effect of sulphate groups on the activity. Appl. Catal. B 2001, 33, 277–291. [Google Scholar] [CrossRef]
- Shang, X.; Hu, G.; He, C.; Zhao, J.; Zhang, F.; Xu, Y.; Zhang, Y.; Li, J.; Chen, J. Regeneration of full-scale commercial honeycomb monolith catalyst (V2O5-WO3/TiO2) used in coal-fired power plant. J. Ind. Eng. Chem. 2012, 18, 513–519. [Google Scholar] [CrossRef]
- Gao, F.; Tang, X.; Yi, H.; Zhao, S.; Zhang, T.; Li, D.; Ma, D. The poisoning and regeneration effect of alkali metals deposed over commercial V2O5-WO3/TiO2 catalysts on SCR of NO by NH3. Chin. Sci. Bull. 2014, 59, 3966–3972. [Google Scholar] [CrossRef]
- Huo, Y.; Chang, Z.; Li, W.; Liu, S.; Dong, B. Reuse and valorization of vanadium and tungsten from waste V2O5-WO3/TiO2 SCR catalyst. Waste Biomass Valoriz. 2014, 6, 159–165. [Google Scholar] [CrossRef]
- Siemens, R.; Jong, B.; Russell, J. Potential of spent catalysts as a source of critical metals. Conserv. Recycl. 1986, 9, 189–196. [Google Scholar] [CrossRef]
- Selective Catalytic Reduction Catalyst Recycle and Re-Use Options; 1019710; Technical Report for Post-Combustion NOx Control; EPRI: Palo Alto, CA, USA, December 2010.
- W. S. Hinton & Associates. Recycling and Disposal of Spent Selective Catalytic Reduction Catalyst; 1004888; Technical Report for Post-Combination NOx Control; EPRI: Palo Alto, CA, USA, October 2003. [Google Scholar]
- Kim, J.W.; Lee, W.G.; Hwang, I.S.; Lee, J.Y.; Han, C. Recovery of tungsten from spent selective catalytic reduction catalysts by pressure leaching. J. Ind. Eng. Chem. 2015, 28, 73–77. [Google Scholar] [CrossRef]
- Li, Q.; Liu, Z.; Liu, Q. Kinetics of vanadium leaching from a spent industrial V2O5/TiO2 catalyst by sulfuric acid. Ind. Eng. Chem. Res. 2014, 53, 2956–2962. [Google Scholar] [CrossRef]
- Liu, W.; Liu, X.B.; Liu, H.H.; Zhou, L.; Xi, W.C.; Zhang, H.K.; Cheng, D.; Zeng, Q.; Li, Y.T.; Fan, Y.S.; et al. Recycling Method of Denitration Catalyst Waste and Denitration Catalyst Prepared from Denitration Catalyst Waste. CN102049317 A, 11 May 2011. [Google Scholar]
- Raziyeh, K.; Ingemar, O.C. Regeneration of commercial TiO2-V2O5-WO3 SCR catalysts used in bio fuel plants. Appl. Catal. B 2001, 30, 87–99. [Google Scholar]
- Zheng, Y.J.; Jensen, A.D.; Johnsson, J.E. Laboratory investigation of selective catalytic reduction catalysts deactivationby potassium compounds and catalyst regeneration. Ind. Eng. Chem. Res. 2004, 43, 941–947. [Google Scholar] [CrossRef]
- Yu, Y.; He, C.; Chen, J.; Yin, L.; Qiu, T.; Meng, X. Regeneration of deactivated commercial SCR catalyst by alkali washing. Catal. Commun. 2013, 39, 78–81. [Google Scholar] [CrossRef]
- Yu, Y.; Meng, X.; Chen, J.; Yin, L.; Qiu, T.; He, C. Deactivation mechanism and feasible regeneration approaches for the used commercial NH3-SCR catalysts. Environ. Technol. 2016, 37, 828–836. [Google Scholar] [CrossRef] [PubMed]
- Shang, X.; Li, J.; Yu, X.; Chen, J.; He, C. Effective regeneration of thermally deactivated commercial V-W-Ti catalysts. Front. Chem. Sci. Eng. 2012, 6, 38–46. [Google Scholar] [CrossRef]
- Christou, S.Y.; Birgersson, H.; Fierro, J.G.; EfstathIou, A.M. Reactivation of an aged commercial three-way catalyst by oxalic and citric acid washing. Environ. Sci. Technol 2006, 40, 2020–2036. [Google Scholar] [CrossRef]
- Yu, Y.; Wang, J.; Chen, J.; He, X.; Wang, Y.; Song, K.; Xie, Z. Regeneration of commercial selective catalyst reduction catalysts deactivated by Pb and other inorganic elements. J. Environ. Sci. 2016, 47, 100–108. [Google Scholar] [CrossRef] [PubMed]
- Christou, S.; Birgersson, H.; Efstathiou, A. Reactivation of severely aged commercial three-way catalysts by washing with weak EDTA and oxalic acid solutions. Appl. Catal. B 2007, 71, 185–198. [Google Scholar] [CrossRef]
- Marcel, F. Method for Regenerating Iron-Loaded deNOx Catalysts. U.S. Patent 7,569,506, 4 August 2009. [Google Scholar]
- Zeng, L.; Cheng, C.Y. A literature review of the recovery of molybdenum and vanadium from spent hydrodesulphurisation catalysts. Hydrometallurgy 2009, 98, 1–9. [Google Scholar] [CrossRef]
- Mazurek, K. Recovery of vanadium, potassium and iron from a spent vanadium catalyst by oxalic acid solution leaching, precipitation and ion exchange processes. Hydrometallurgy 2013, 134–135, 26–31. [Google Scholar] [CrossRef]
- Zhou, C.Y.; Zahng, L.N.; Deng, Y.; Ma, S.C. Research progress on ammonium bisulfate formation and control in the process of selective catalytic reduction. Environ. Prog. Sustain. 2016, 35, 1664–1672. [Google Scholar] [CrossRef]
- Lee, J.B.; Kim, S.K.; Kim, D.W.; Kim, K.H.; Chun, S.N.; Hur, K.B.; Jeong, S.M. Effect of H2SO4 concentration in washing solution on regeneration of commercial selective catalytic reduction catalyst. Korean J. Chem. Eng. 2012, 29, 270–276. [Google Scholar] [CrossRef]
- Tang, F.; Zhuang, K.; Yang, F.; Yang, L.; Xu, B.; Qiu, J.; Fan, Y. Effect of dispersion state and surface properties of supported vanadia on the activity of V2O5/TiO2 catalysts for the selective catalytic reduction of NO by NH3. Chin. J. Catal. 2012, 33, 933–940. [Google Scholar] [CrossRef]
- Baiker, A.; Handy, B.; Nickl, J.; Schraml-Marth, M.; Wokaun, A. Selective catalytic reduction of nitric oxide over vanadia grafted on titania. Influence of vanadia loading on structural and catalytic properties of catalysts. Catal. Lett. 1992, 14, 89–99. [Google Scholar] [CrossRef]
- Marberger, A.; Elsener, M.; Ferri, D.; Kröcher, O. VOx surface coverage optimization of V2O5/WO3-TiO2 SCR catalysts by variation of the V loading and by aging. Catalysts 2015, 5, 1704–1720. [Google Scholar] [CrossRef]
- Orsenigo, C.; Lietti, L.; Tronconi, E.; Forzatti, P.; Bregani, F. Dynamic investigation of the role of the surface sulfates in NOx reduction and SO2 oxidation over V2O5-WO3/TiO2 catalysts. Ind. Eng. Chem. Res. 1998, 37, 2350–2359. [Google Scholar] [CrossRef]
- Chen, J.P.; Yang, R.T. Selective catalytic reduction of NO with NH3 on SO42−/TiO2 superacid catalyst. J. Catal. 1993, 139, 277–288. [Google Scholar] [CrossRef]
- Busca, G.; Centi, G.; Marchetti, L.; Trifiro, F. Chemical and spectroscopic study of the nature of a vanadium oxide monolayer supported on a high-surface-area TiO2 anatase. Langmuir 1986, 2, 568–577. [Google Scholar] [CrossRef]
- Alemany, L.J.; Lietti, L.; Ferlazzo, N.; Forzatti, P.; Busca, G.; Giamello, E.; Bregani, F. Reactivity and phsicochemical characterisation of V2O5-WO3/TiO2 De-NOx catalysts. J. Catal. 1995, 155, 117–130. [Google Scholar] [CrossRef]
- Cristiani, C.; Forzatti, P.; Busca, G. On the surface structure of vanadia-titania catalysts combined laser-Raman and fourier transform-infrared investigation. J. Catal. 1989, 116, 586–589. [Google Scholar] [CrossRef]
- Seo, P.W.; Lee, J.Y.; Shim, K.S.; Hong, S.H.; Hong, S.C.; Hong, S.I. The control of valence state: How V/Ti2 catalysts is hindering the deactivation using the mechanochemical method. J. Hazard. Mater. 2009, 165, 39–47. [Google Scholar] [CrossRef] [PubMed]
- Dong, G.J.; Bai, Y.; Zhang, Y.Y.; Zhao, Y. Effect of the V4+(3+)/V5+ ratio on the denitration activity for V2O5-WO3/TiO2 catalysts. New J. Chem. 2015, 39, 3588–3596. [Google Scholar] [CrossRef]
- Xiao, Y.; Liu, Z.; Liu, Q.Y.; Wang, J.C. Meachanism of SO2 influence on NO removal over V2O5/AC catalyst. Chin. J. Catal. 2008, 29, 81–85. [Google Scholar]
- Topsoe, N.Y.; Topsoe, H.; Dumesic, J.A. Vanadia/titania catalysts for selective catalytic reduction (SCR) of nitric-oxide by ammonia I. Combined temperature-programmed in-situ FTIR and on-line mass-spectroscopy studies. J. Catal. 1995, 151, 226–240. [Google Scholar] [CrossRef]
- Topsoe, N.Y.; Dumesic, J.A.; Topsoe, H. Vanadia/titania catalysts for selective catalytic reduction of nitric oxide by ammonia II: Studies of active sites and formulation of catalytic cycles. J. Catal. 1995, 151, 241–252. [Google Scholar] [CrossRef]
- Tang, F.; Xu, B.; Shi, H.; Qiu, J.; Fan, Y. The poisoning effect of Na+ and Ca2+ ions doped on the V2O5/TiO2 catalysts for selective catalytic reduction of NO by NH3. Appl. Catal. B 2010, 94, 71–76. [Google Scholar] [CrossRef]
- Busca, G.; Lietti, L.; Ramis, G.; Berti, F. Chemical and mechanistic aspects of the selective catalytic reduction of NOx by ammonia over oxide catalysts: A review. Appl. Catal. B 1998, 18, 1–36. [Google Scholar] [CrossRef]
- Wang, C.; Yang, S.; Chang, H.; Peng, Y.; Li, J. Dispersion of tungsten oxide on SCR performance of V2O5-WO3/TiO2: Acidity, surface species and catalytic activity. Chem. Eng. J. 2013, 225, 520–527. [Google Scholar] [CrossRef]
- Chen, L.; Li, J.; Ge, M. The poisoning effect of alkali metals doping over nano V2O5–WO3/TiO2 catalysts on selective catalytic reduction of NOx by NH3. Chem. Eng. J. 2011, 170, 531–537. [Google Scholar] [CrossRef]
| Sample | Composition (wt %) | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Ti | V | W | Al | Si | Ca | Fe | K | Na | |
| Fresh | 55.04 | 0.39 | 3.48 | 0.44 | 0.87 | 0.84 | 0.04 | 0.02 | 0.07 |
| Waste | 51.88 | 0.31 | 4.57 | 0.92 | 0.92 | 0.90 | 0.24 | 0.24 | 0.13 |
| OA leaching residue | 52.42 | 0.12 | 3.94 | 0.52 | 1.02 | 0.75 | 0.07 | 0.04 | 0.03 |
| Catalyst | V2O5 Content a (wt %) | SBET (m2/g) | VOx Coverage | |
|---|---|---|---|---|
| mmol V/100 m2 TiO2 b | % c | |||
| Fresh | 0.71 | 65.6 | 0.139 | 21 |
| Waste | 0.53 | 66.3 | 0.104 | 16 |
| OA leaching residue | 0.12 | 76.9 | 0.017 | 3 |
| Re-0.5% | 0.57 | 51.5 | 0.122 | 18 |
| Re-1.0% | 0.98 | 50.1 | 0.193 | 29 |
| Re-1.5% | 1.45 | 43.6 | 0.373 | 56 |
| Catalyst | Binding Energy (eV) | Surface Atomic Concentration (%) | Surface Atomic Ratio | |||||
|---|---|---|---|---|---|---|---|---|
| V3+ | V4+ | V5+ | V3+ | V4+ | V5+ | V/Ti | V4+/V5+ | |
| Fresh | - | 515.7 | 516.6 | - | 62.5 | 37.5 | 0.046 | 1.67 |
| Waste | 514.6 | 515.8 | 516.6 | 3.0 | 51.5 | 45.5 | 0.036 | 1.13 |
| Re-0.5% | - | 515.7 | 516.6 | - | 46.9 | 53.1 | 0.037 | 0.88 |
| Re-1.0% | - | 515.9 | 516.9 | - | 60.8 | 39.2 | 0.047 | 1.55 |
| Re-1.5% | - | 516.1 | 517.1 | - | 69.1 | 30.9 | 0.076 | 2.24 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Qi, C.; Bao, W.; Wang, L.; Li, H.; Wu, W. Study of the V2O5-WO3/TiO2 Catalyst Synthesized from Waste Catalyst on Selective Catalytic Reduction of NOx by NH3. Catalysts 2017, 7, 110. https://doi.org/10.3390/catal7040110
Qi C, Bao W, Wang L, Li H, Wu W. Study of the V2O5-WO3/TiO2 Catalyst Synthesized from Waste Catalyst on Selective Catalytic Reduction of NOx by NH3. Catalysts. 2017; 7(4):110. https://doi.org/10.3390/catal7040110
Chicago/Turabian StyleQi, Chunping, Weijun Bao, Liguo Wang, Huiquan Li, and Wenfen Wu. 2017. "Study of the V2O5-WO3/TiO2 Catalyst Synthesized from Waste Catalyst on Selective Catalytic Reduction of NOx by NH3" Catalysts 7, no. 4: 110. https://doi.org/10.3390/catal7040110
APA StyleQi, C., Bao, W., Wang, L., Li, H., & Wu, W. (2017). Study of the V2O5-WO3/TiO2 Catalyst Synthesized from Waste Catalyst on Selective Catalytic Reduction of NOx by NH3. Catalysts, 7(4), 110. https://doi.org/10.3390/catal7040110





