Preparation of Manganese Lignosulfonate and Its Application as the Precursor of Nanostructured MnOx for Oxidative Electrocatalysis
Abstract
:1. Introduction
2. Results and Discussion
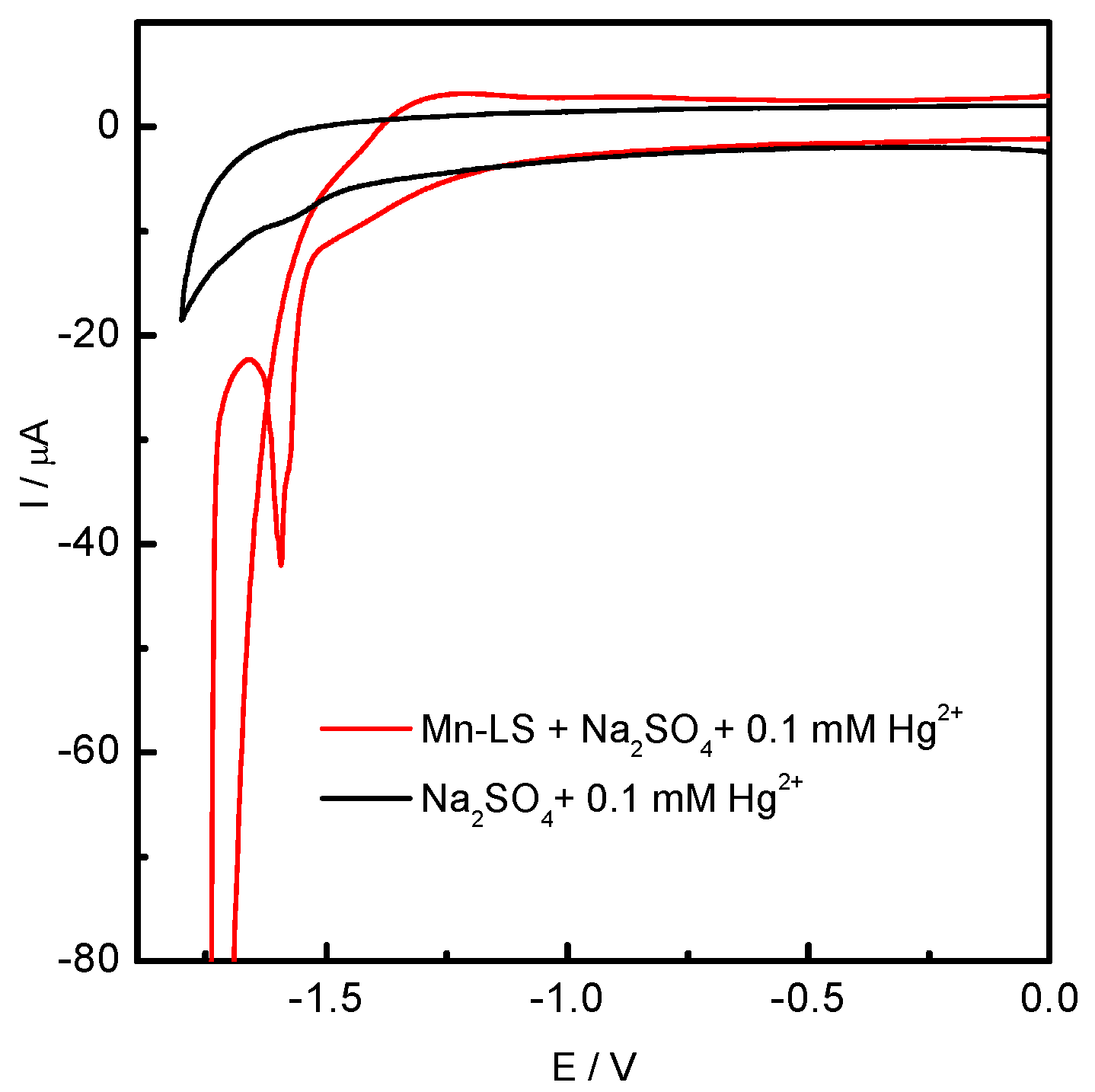
2.1. Cathodic Behavior of Mn-Lignosulfonate
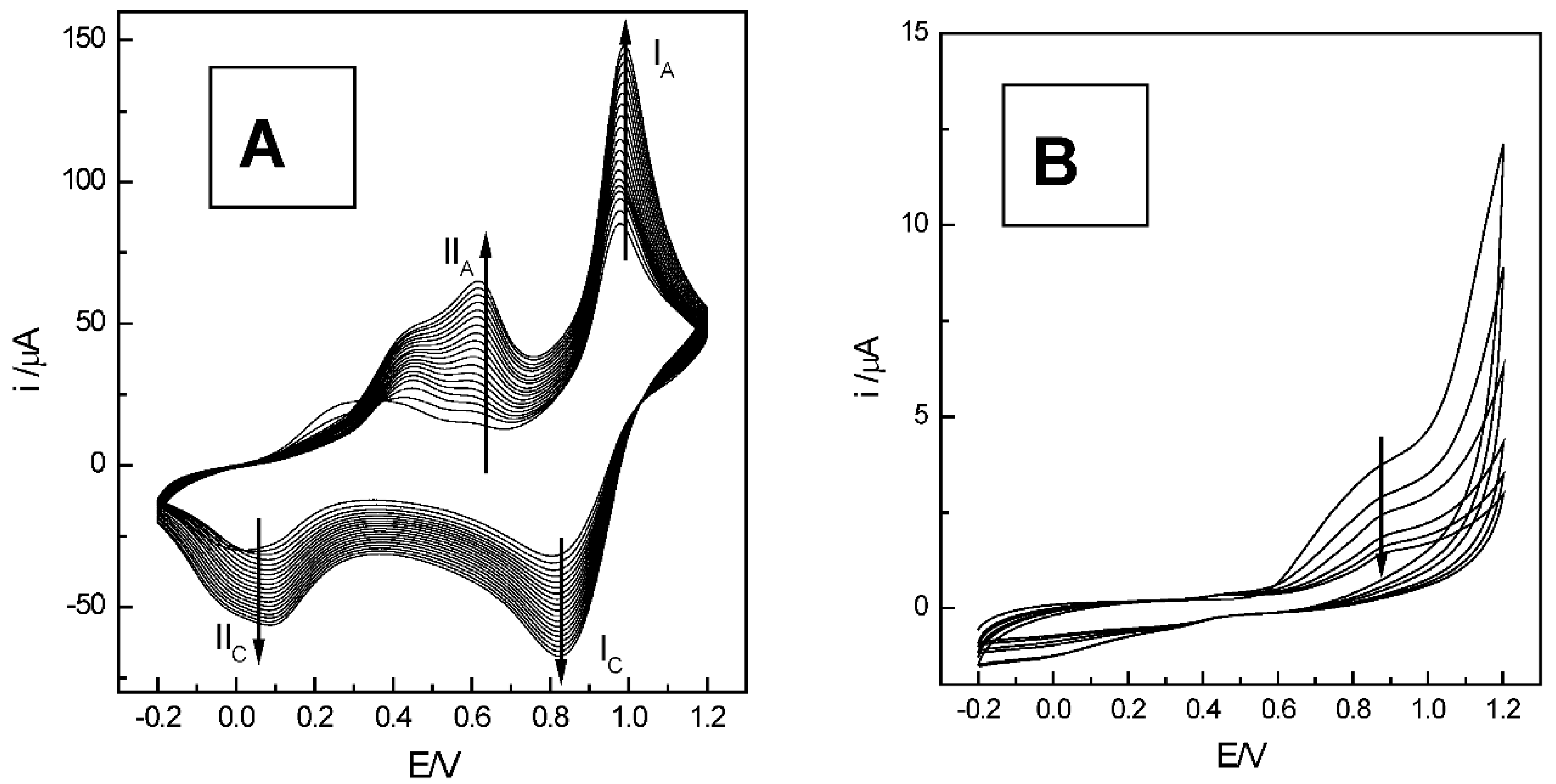
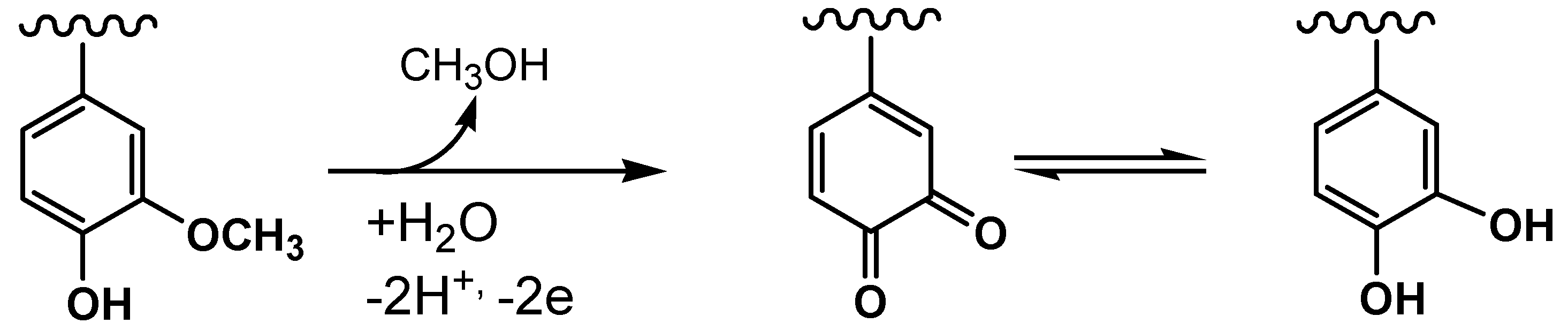
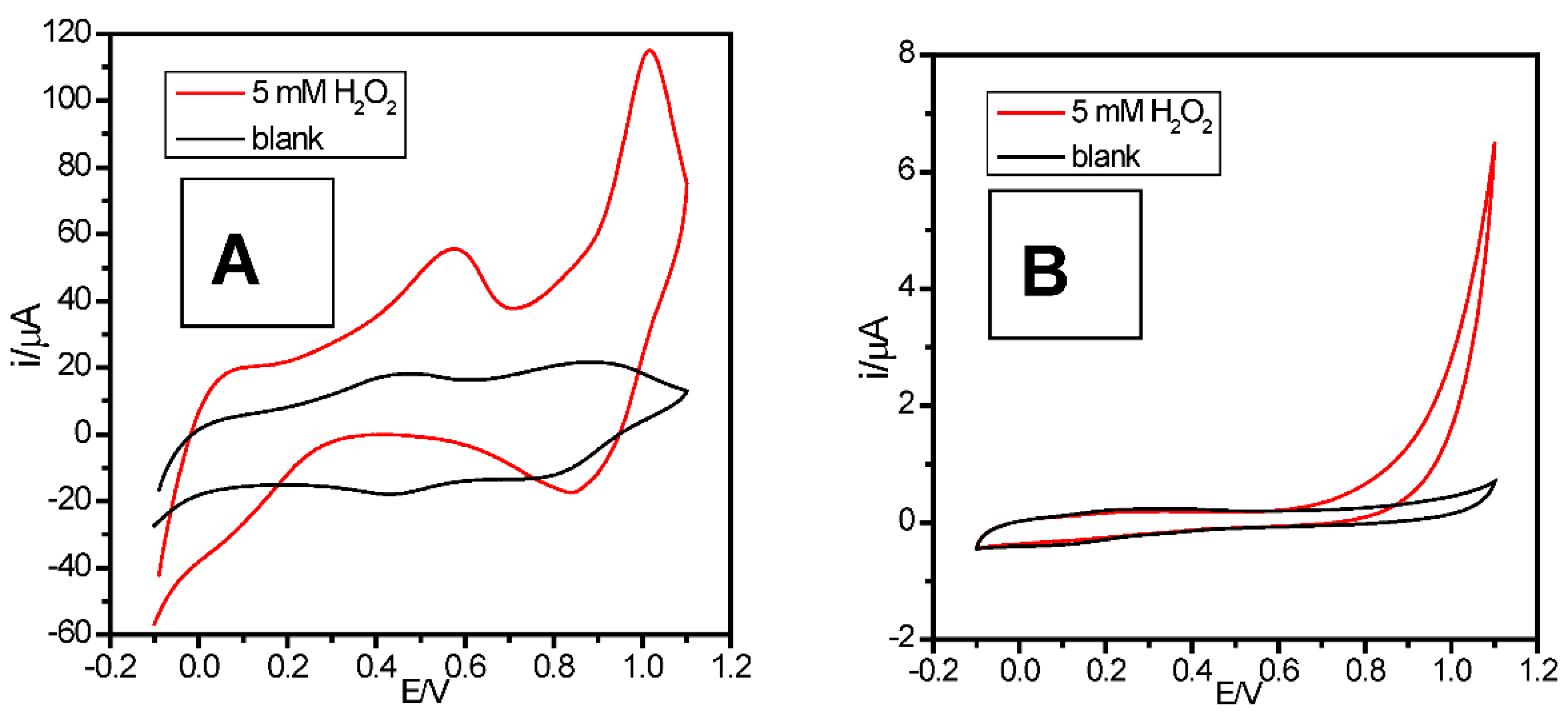
2.2. Anodic Behavior of Mn-Lignosulfonate
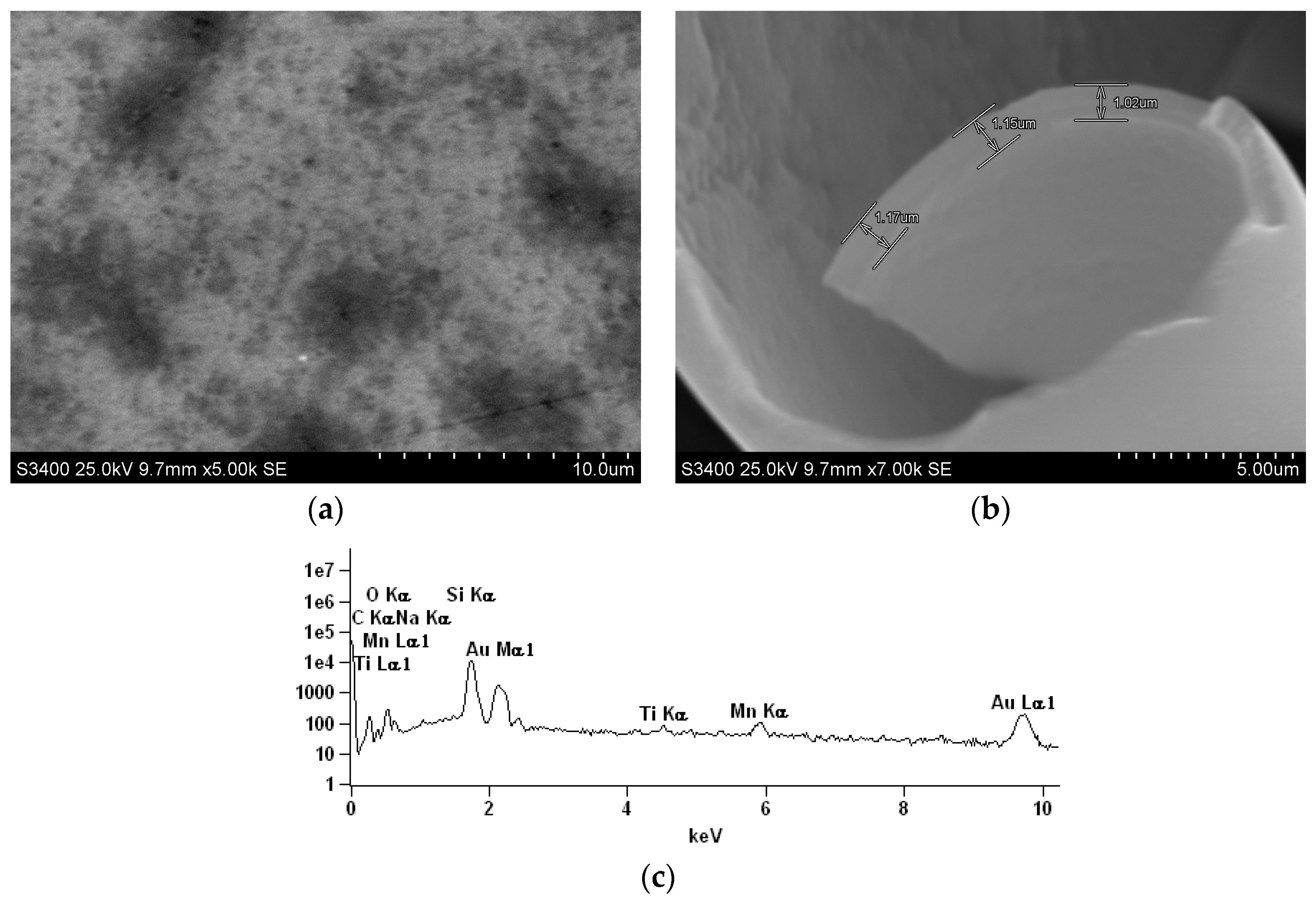
2.3. SEM/Energy Dispersive Spectroscopy EDS Characterization of Electrodeposited MnOx-Lignosulfonate Film
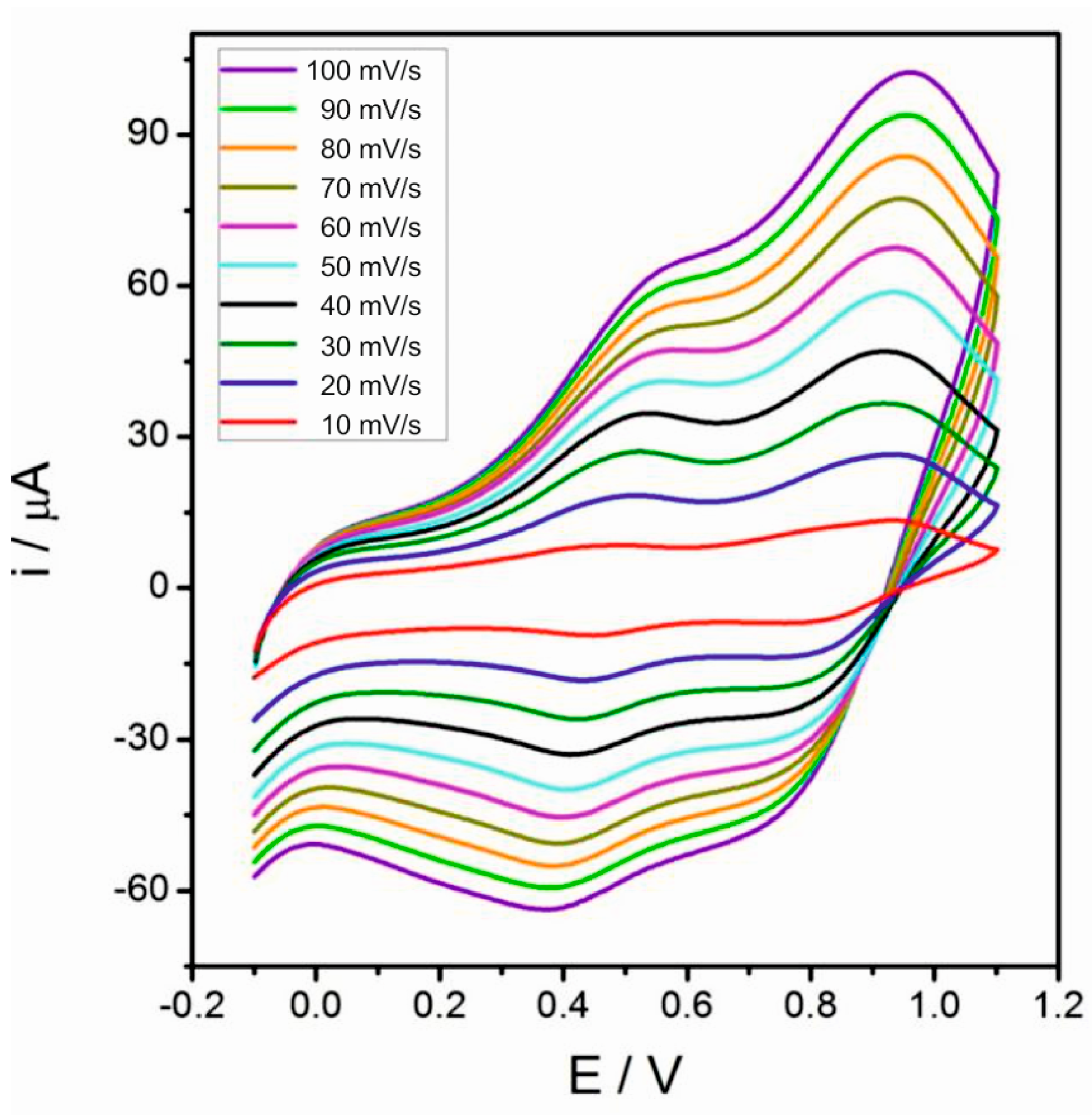
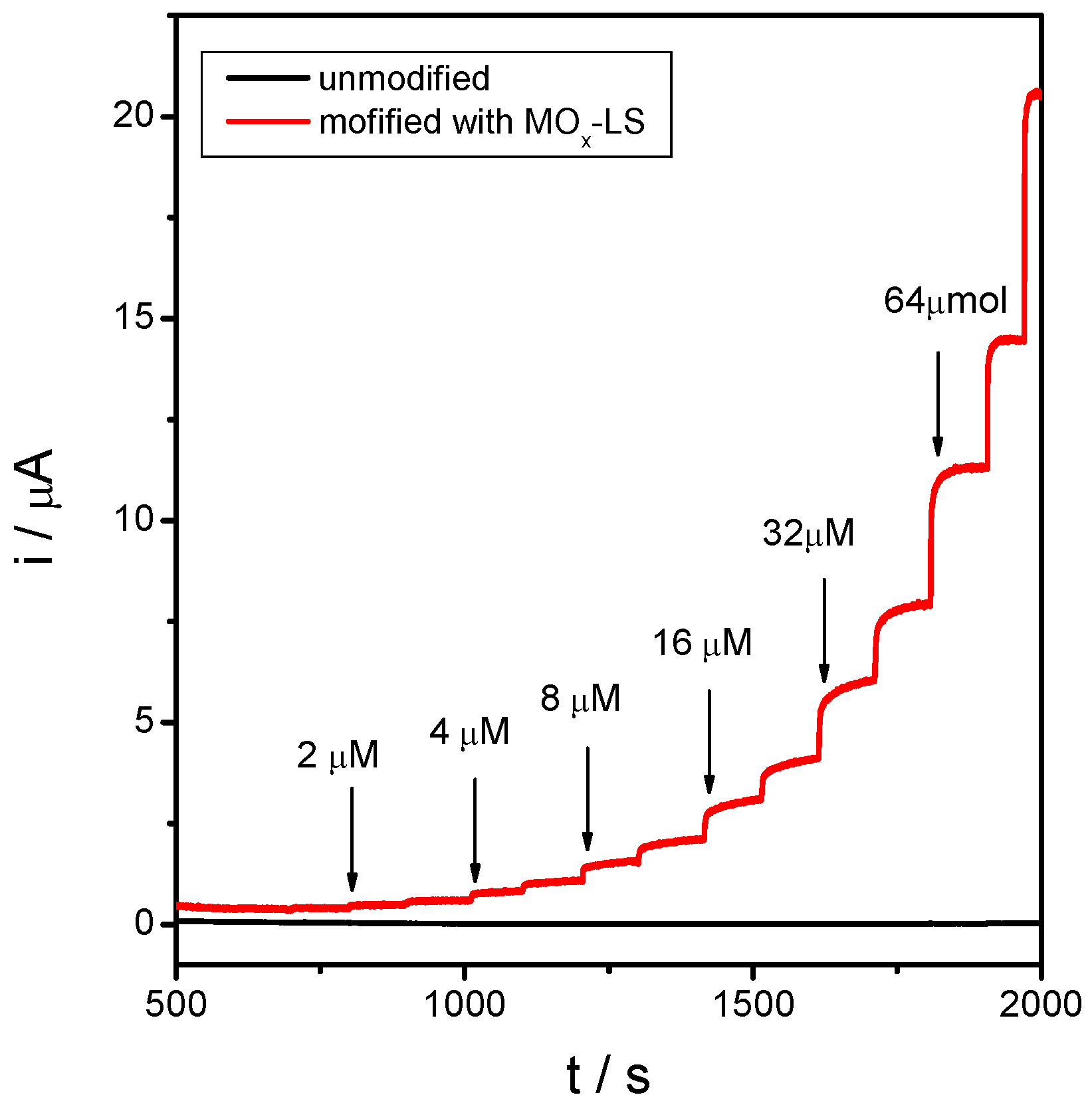
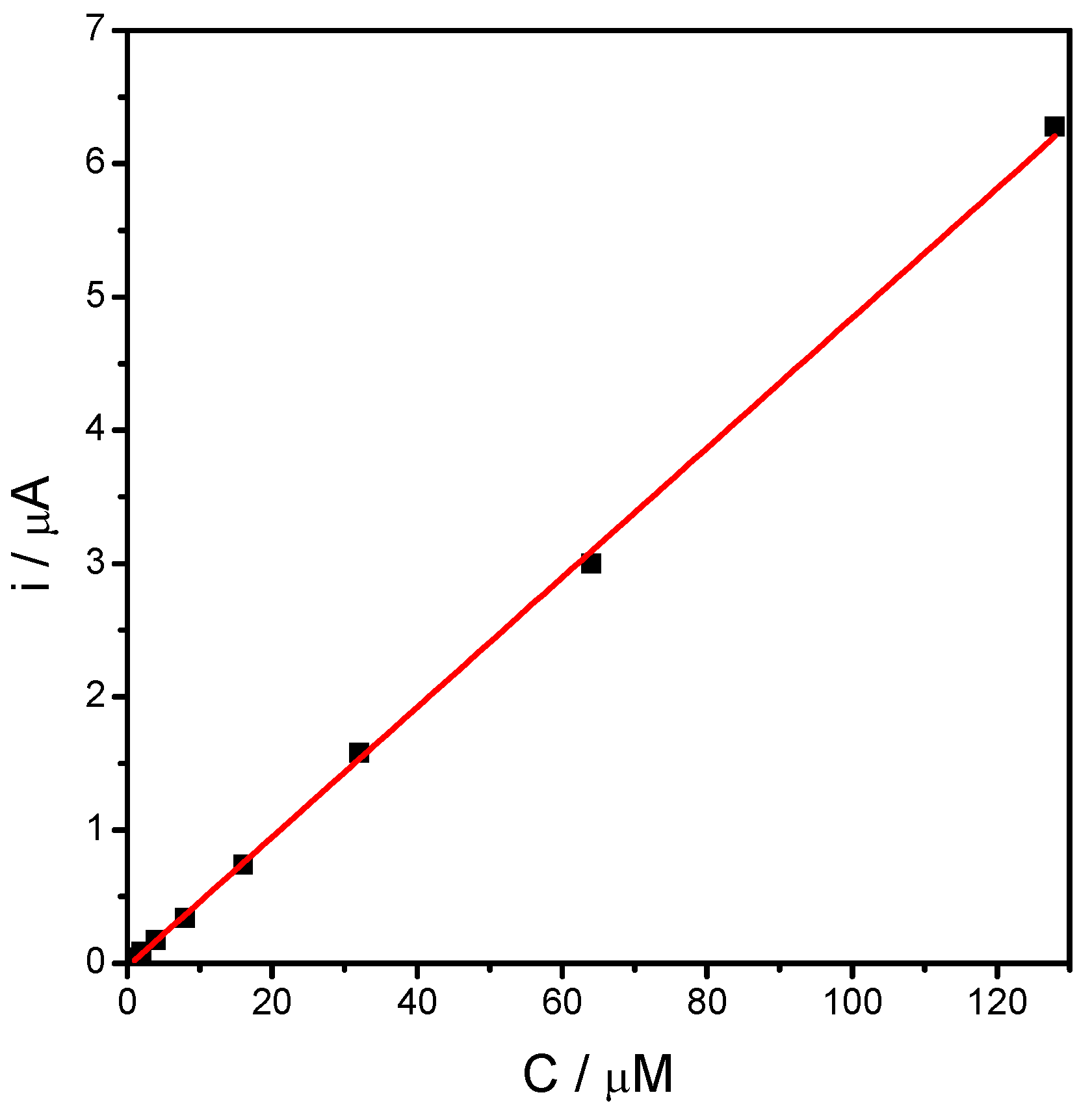
2.4. Electrocatalytic Properties of MnOx-Lignosulfonate Film
3. Materials and Methods
3.1. Chemicals
3.2. Apparatus
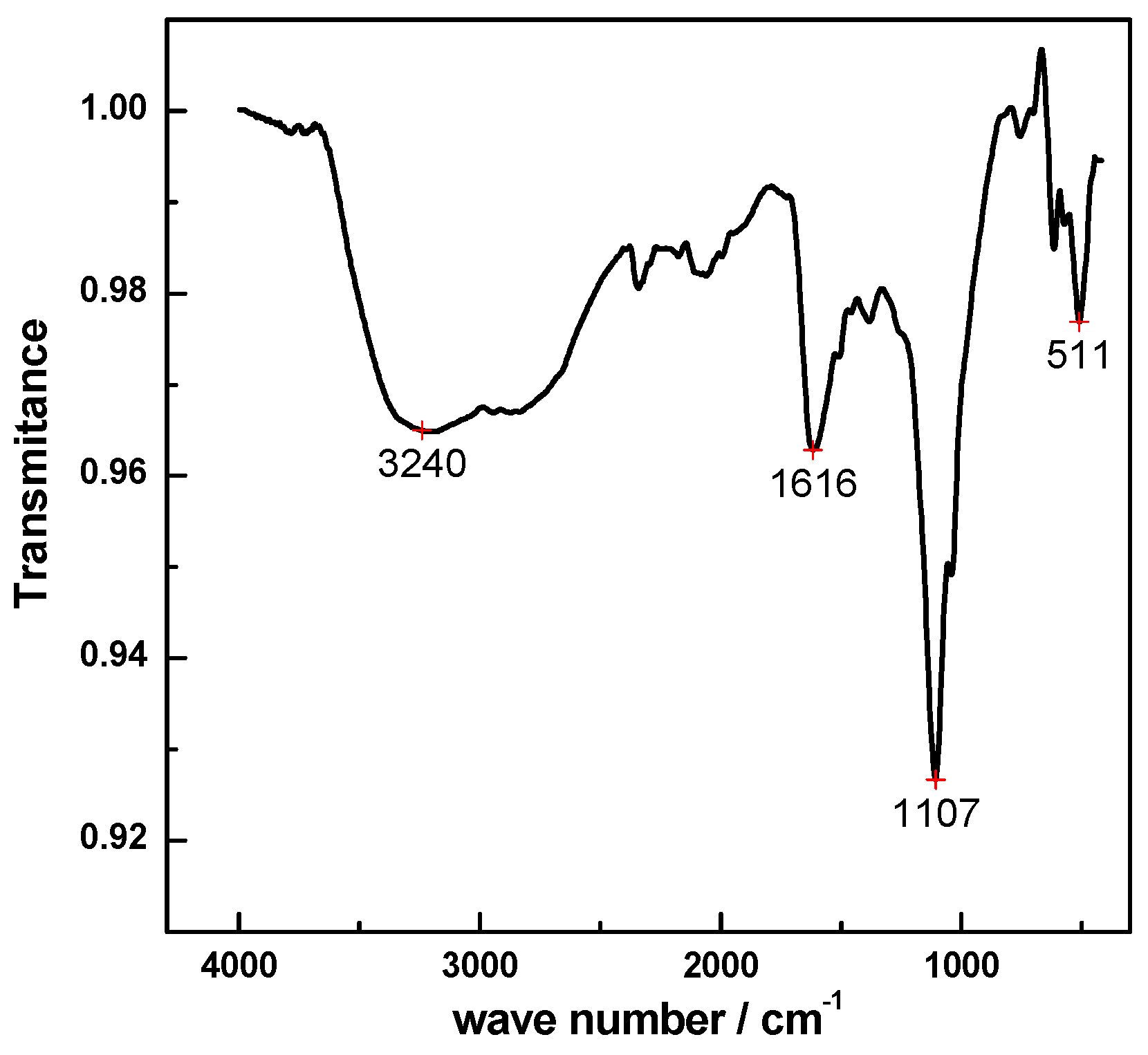
3.3. Preparation of Manganese Lignosulfonate Complex
3.4. Electrodeposition of the MnOx-LS Film
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Vanholme, R.; Demedts, B.; Morreel, K.; Ralph, J.; Boerjan, W. Lignin biosynthesis and structure. Plant Physiol. 2010, 153, 895–905. [Google Scholar] [CrossRef] [PubMed]
- Khvan, A.M.; Abduazimov, K.A. Interaction of lignosulfonate with certain metal ions. Chem. Nat. Compd. 1990, 26, 575–577. [Google Scholar] [CrossRef]
- Milczarek, G. Lignosulfonate-modified electrode for electrocatalytic reduction of acidic nitrite. Electroanalysis 2008, 20, 211–214. [Google Scholar] [CrossRef]
- Milczarek, G. Lignosulfonate-modified electrodes: Electrochemical properties and electrocatalysis of NADH oxidation. Langmuir 2009, 25, 10345–10353. [Google Scholar] [CrossRef] [PubMed]
- Milczarek, G.; Nowicki, M. Carbon nanotubes/kraft lignin composite: Characterization and charge storage properties. Mater. Res. Bull. 2013, 48, 4032–4038. [Google Scholar] [CrossRef]
- Milczarek, G.; Inganäs, O. Renewable cathode materials from biopolymer/conjugated polymer interpenetrating networks. Science 2012, 335, 1468–1471. [Google Scholar] [CrossRef] [PubMed]
- Milczarek, G.; Rebis, T.; Fabianska, J. One-step synthesis of lignosulfonate-stabilized silver nanoparticles. Colloids Surf. B 2013, 105, 335–341. [Google Scholar] [CrossRef] [PubMed]
- Konował, E.; Modrzejewska-Sikorska, A.; Milczarek, G. Synthesis and multifunctional properties of lignosulfonate-stabilized gold nanoparticles. Mater. Lett. 2015, 159, 451–454. [Google Scholar] [CrossRef]
- Ciszewski, A.; Śron, K.; Stępniak, I.; Milczarek, G. Nickel(II) lignosulfonate as precursor for the deposition of nickel hydroxide nanoparticles on a glassy carbon electrode for oxidative electrocatalysis. Electrochim. Acta 2014, 134, 355–362. [Google Scholar] [CrossRef]
- Bai, W.; Zhang, X.; Zhang, S.; Sheng, Q.; Zheng, J. Acidification of manganese dioxide for ultrasensitive electrochemical sensing of hydrogen peroxide in living cells. Sens. Actuator B Chem. 2017, 242, 718–727. [Google Scholar] [CrossRef]
- Lima, F.H.B.; Calegaro, M.L.; Ticianelli, E.A. Investigations of the catalytic properties of manganese oxides for the oxygen reduction reaction in alkaline media. J. Electroanal. Chem. 2006, 590, 152–160. [Google Scholar] [CrossRef]
- Yamaguchi, R.; Sato, A.; Iwai, S.; Tomono, K.; Nakayama, M. A novel formaldehyde sensor based on the pseudocapacitive catalysis of birnessite. Electrochem. Commun. 2013, 29, 55–58. [Google Scholar] [CrossRef]
- Majd, S.M.; Teymourian, H.; Salimi, A. Fabrication of an electrochemical l-cysteine sensor based on graphene nanosheets decorated manganese oxide nanocomposite modified glassy carbon electrode. Electroanalysis 2013, 25, 2201–2210. [Google Scholar] [CrossRef]
- Salimi, A.; Pourbahram, B.; Majd, S.M.; Hallaj, R. Manganese oxide nanoflakes/multi-walled carbon nanotubes/chitosan nanocomposite modified glassy carbon electrode as a novel electrochemical sensor for chromium(III) detection. Electrochim. Acta 2015, 156, 207–215. [Google Scholar] [CrossRef]
- Massa, A.; Hernández, S.; Lamberti, A.; Galletti, C.; Russo, N.; Fino, D. Electro-oxidation of phenol over electrodeposited MnOx nanostructures and the role of a TiO2 nanotubes interlayer. Appl. Catal. B Environ. 2017, 203, 270–281. [Google Scholar] [CrossRef]
- Bai, Y.-H.; Zhang, H.; Xu, J.-J.; Chen, H.-Y. Relationship between nanostructure and electrochemical/biosensing properties of MnO2 nanomaterials for H2O2/Choline. J. Phys. Chem. C 2008, 112, 18984–18990. [Google Scholar] [CrossRef]
- Zeng, F.; Pan, Y.; Yang, Y.; Li, Q.; Li, G.; Hou, Z.; Gu, G. Facile construction of Mn3O4-MnO2 hetero-nanorods/graphene nanocomposite for highly sensitive electrochemical detection of hydrogen peroxide. Electrochim. Acta 2016, 196, 587–596. [Google Scholar] [CrossRef]
- Lu, J.; Dreisinger, D.; Glück, T. Manganese electrodeposition—A literature review. Hydrometallurgy 2014, 141, 105–116. [Google Scholar] [CrossRef]
- O’Halloran, R.J. Anodic stripping voltammetry of manganese in seawater at a mercury film electrode. Anal. Chim. Acta 1982, 140, 51–58. [Google Scholar] [CrossRef]
- Liu, Y.-H.; His, H.-C.; Li, K.-C.; Hou, C.-H. Electrodeposited manganese dioxide/activated carbon composite as a high-performance electrode material for capacitive deionization. ACS Sustain. Chem. Eng. 2016, 4, 4762–4770. [Google Scholar] [CrossRef]
- Hosseini-Benhangi, P.; Kung, C.H.; Alfantazi, A.; Gyenge, E.L. Controlling the interfacial environment in the electrosynthesis of MnOx nanostructures for high-performance oxygen reduction/evolution electrocatalysis. ACS Appl. Mater. Interfaces 2017, 9, 26771–26785. [Google Scholar] [CrossRef] [PubMed]
- Hao, C.; Feng, F.; Wang, X.; Zhou, M.; Zhao, Y.; Ge, C.; Wang, K. The preparation of Fe2O3 nanoparticles by liquid phase-based ultrasonic-assisted method and its application as enzyme-free sensor for the detection of H2O2. RSC Adv. 2015, 5, 21161–21169. [Google Scholar] [CrossRef]
- Pang, B.; Yan, J.; Yao, L.; Liu, H.; Guan, J.; Wang, H.; Liu, H. Preparation and antibacterial paper coated with sodium lignosulfonates stabilized ZnO nanoparticles. RSC Adv. 2016, 6, 9753–9759. [Google Scholar] [CrossRef]
- Klapiszewski, Ł.; Szalaty, T.J.; Kurc, B.; Stanisz, M.; Skrzypczak, A.; Jesionowski, T. Functional hybryd materials based on manganese dioxide and lignin activated by ionic liquids and their application in the production of lithium ion batteries. Int. J. Mol. Sci. 2017, 18, 1509. [Google Scholar] [CrossRef] [PubMed]
- Xu, W.; Liu, J.; Wang, M.; Chen, L.; Wang, X.; Hu, C. Direct growth of MnOOH nanorod arrays on a carbon cloth for high performance non-enzymatic hydrogen peroxide sensing. Anal. Chim. Acta 2016, 913, 128–136. [Google Scholar] [CrossRef] [PubMed]
- Jędrzak, A.; Rębiś, T.; Klapiszewski, Ł.; Zdarta, J.; Milczarek, G.; Jesionowski, T. Carbon paste electrode based on functional GOx/silica-lignin system to prepare an amperometric glucose biosensor. Sens. Actuator B Chem. 2018, 256, 176–185. [Google Scholar] [CrossRef]
| Element | Atomic % | Weight % |
|---|---|---|
| Mn | 11.9 | 32.8 |
| O | 70.6 | 56.7 |
| C | 17.5 | 10.5 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gawluk, K.; Modrzejwska-Sikorska, A.; Rębiś, T.; Milczarek, G. Preparation of Manganese Lignosulfonate and Its Application as the Precursor of Nanostructured MnOx for Oxidative Electrocatalysis. Catalysts 2017, 7, 392. https://doi.org/10.3390/catal7120392
Gawluk K, Modrzejwska-Sikorska A, Rębiś T, Milczarek G. Preparation of Manganese Lignosulfonate and Its Application as the Precursor of Nanostructured MnOx for Oxidative Electrocatalysis. Catalysts. 2017; 7(12):392. https://doi.org/10.3390/catal7120392
Chicago/Turabian StyleGawluk, Klaudia, Anna Modrzejwska-Sikorska, Tomasz Rębiś, and Grzegorz Milczarek. 2017. "Preparation of Manganese Lignosulfonate and Its Application as the Precursor of Nanostructured MnOx for Oxidative Electrocatalysis" Catalysts 7, no. 12: 392. https://doi.org/10.3390/catal7120392
APA StyleGawluk, K., Modrzejwska-Sikorska, A., Rębiś, T., & Milczarek, G. (2017). Preparation of Manganese Lignosulfonate and Its Application as the Precursor of Nanostructured MnOx for Oxidative Electrocatalysis. Catalysts, 7(12), 392. https://doi.org/10.3390/catal7120392






