Bimetallic Catalysts Containing Gold and Palladium for Environmentally Important Reactions
Abstract
:1. Introduction
1.1. General Introduction
1.2. Structures of Bimetallic Nanocatalysts
1.3. Factors Affecting the Catalytic Activity of a Bimetallic Catalyst
1.3.1. Effect of Particle Size and Shape
1.3.2. Structure Effect
1.3.3. Composition Effects
1.3.4. Surface area and Porosity
2. Preparation and Characterization Methods of Bimetallic Catalysts
2.1. Preparation Methods
2.1.1. Impregnation Method
2.1.2. Deposition-Precipitation Method
2.1.3. Chemical and Physical Vapor Deposition Methods
2.1.4. Co-Precipitation Method
2.1.5. Liquid Preparation Method
2.1.6. Catalyst Synthesis of Pd and Au Based Bimetallic Catalysts and Their Catalytic Testing Procedure
Synthesis and Testing Procedure for Pd Based Catalysts
Catalyst Synthesis and Catalytic Testing Procedure for Au Based Catalysts
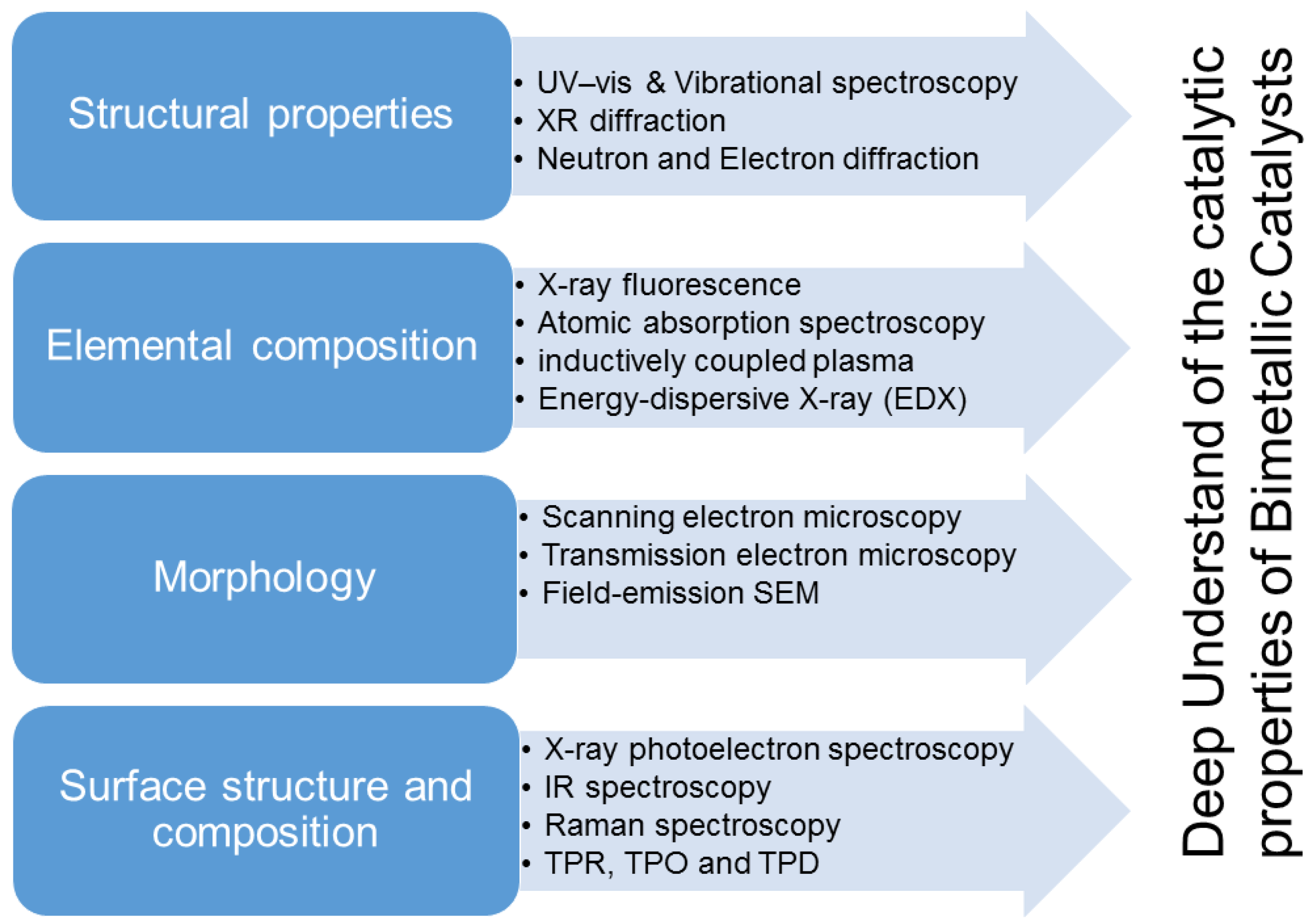
2.2. Characterization of Bimetallic Catalysts
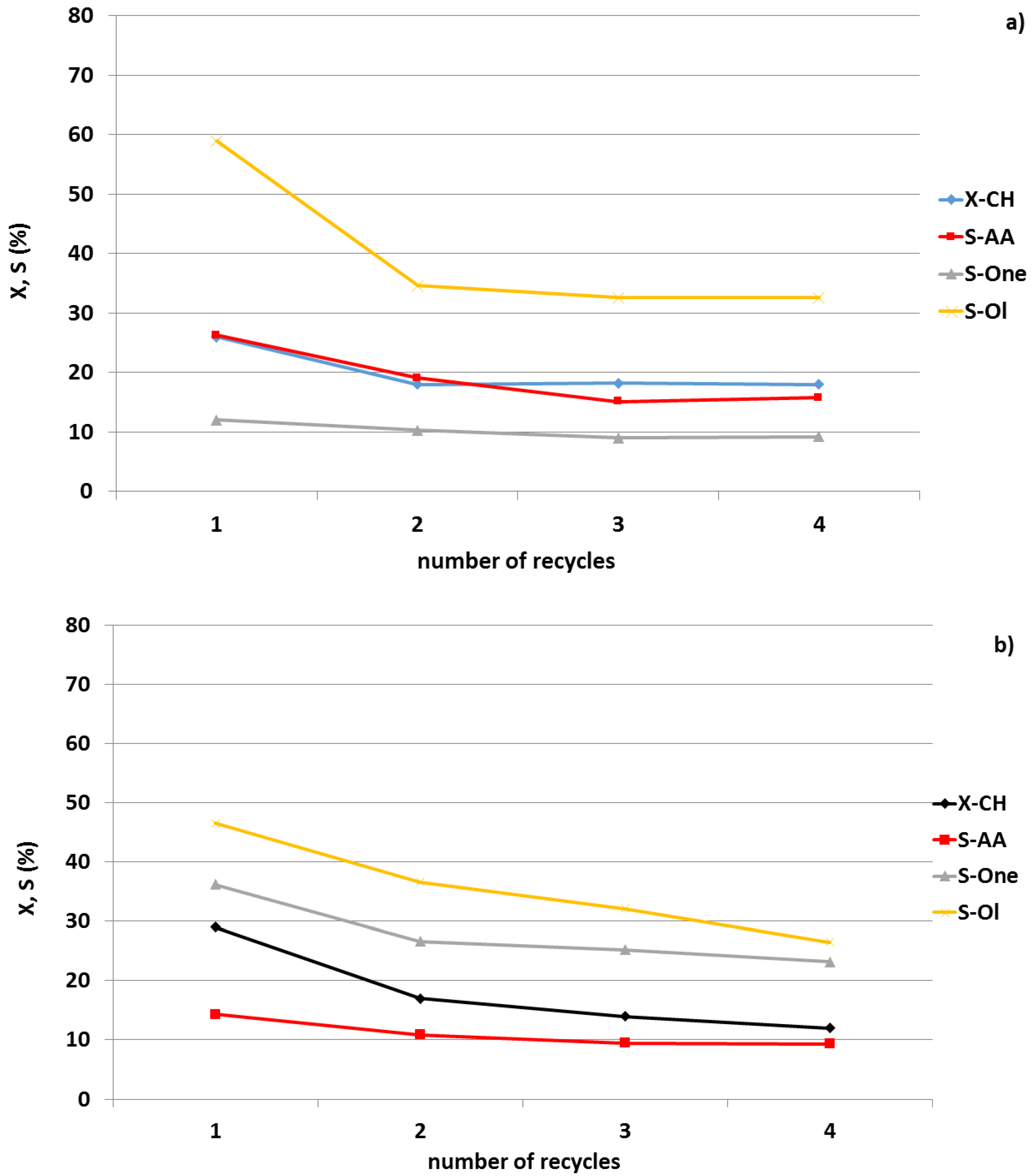
3. Own Results
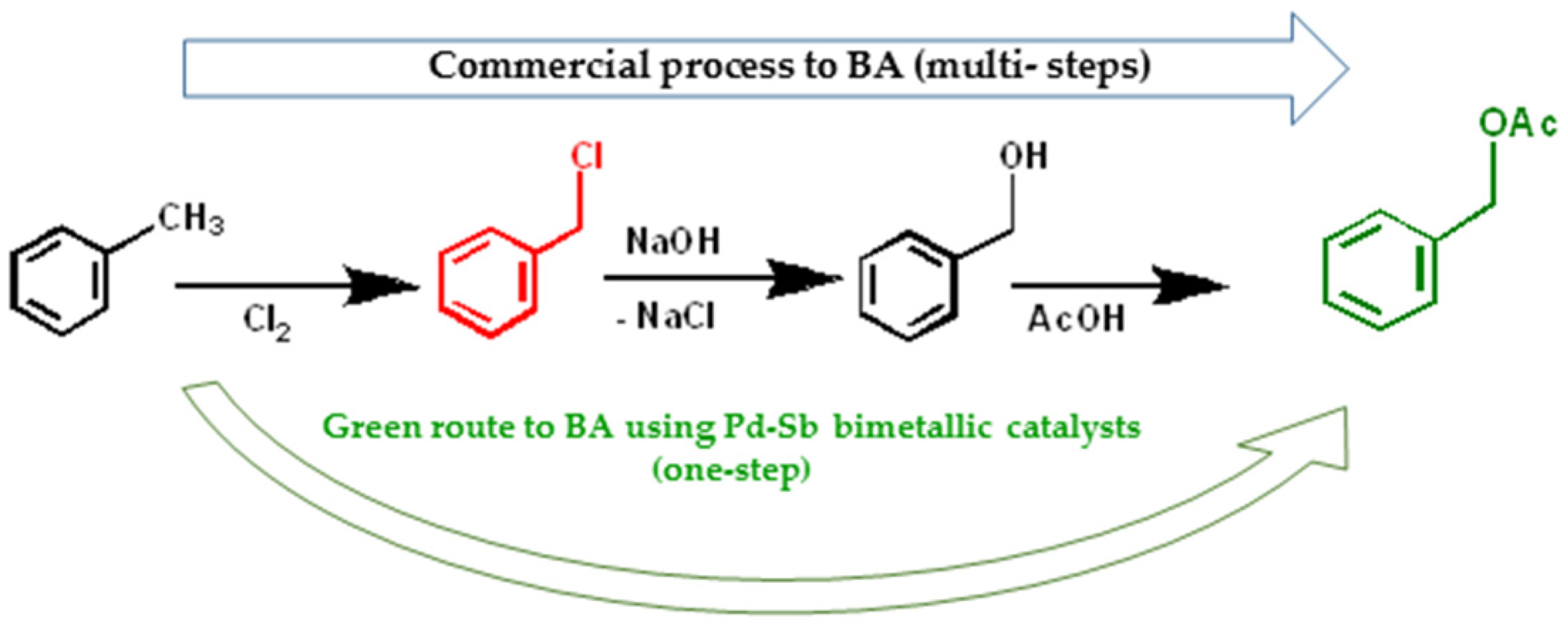
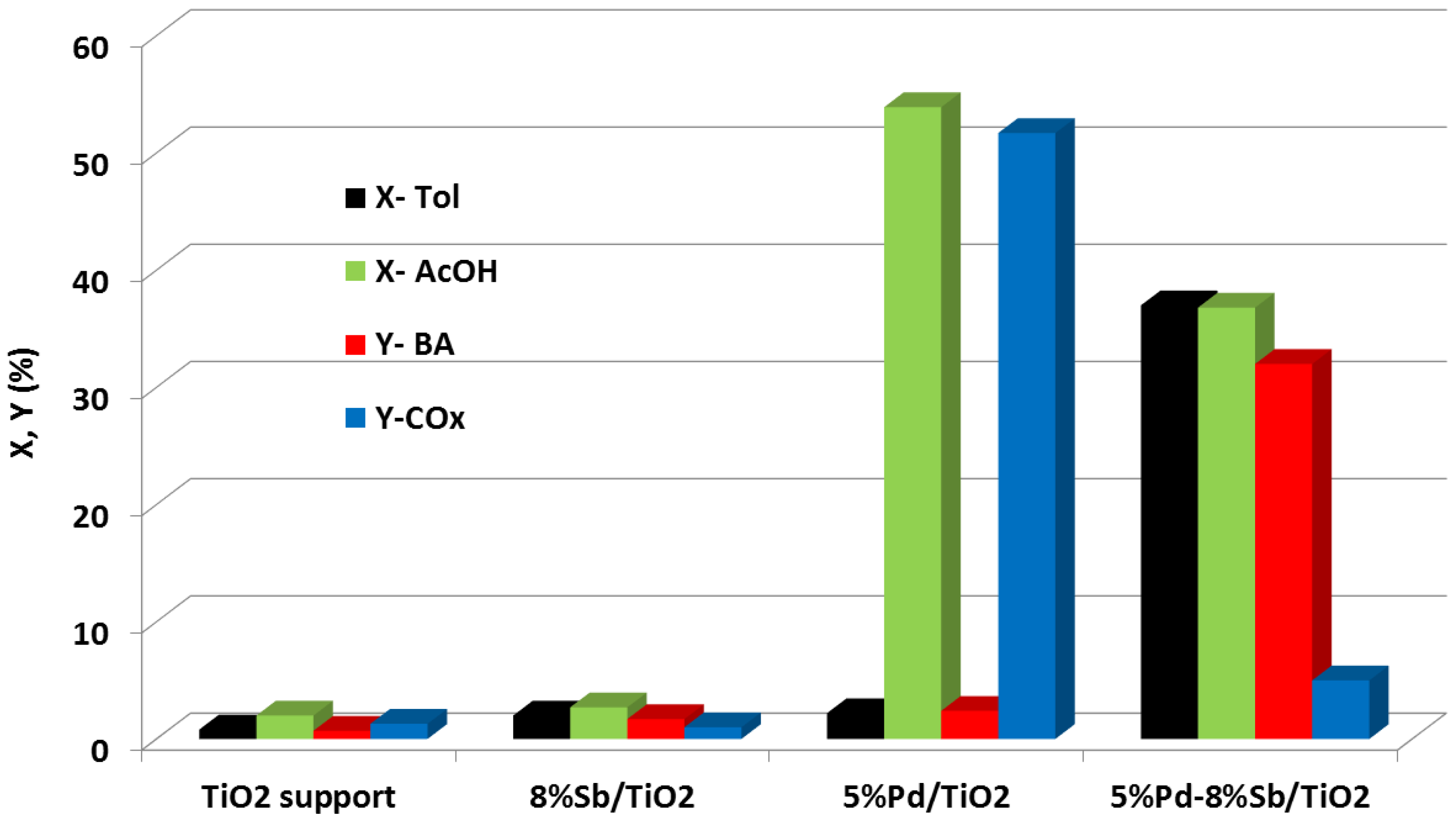
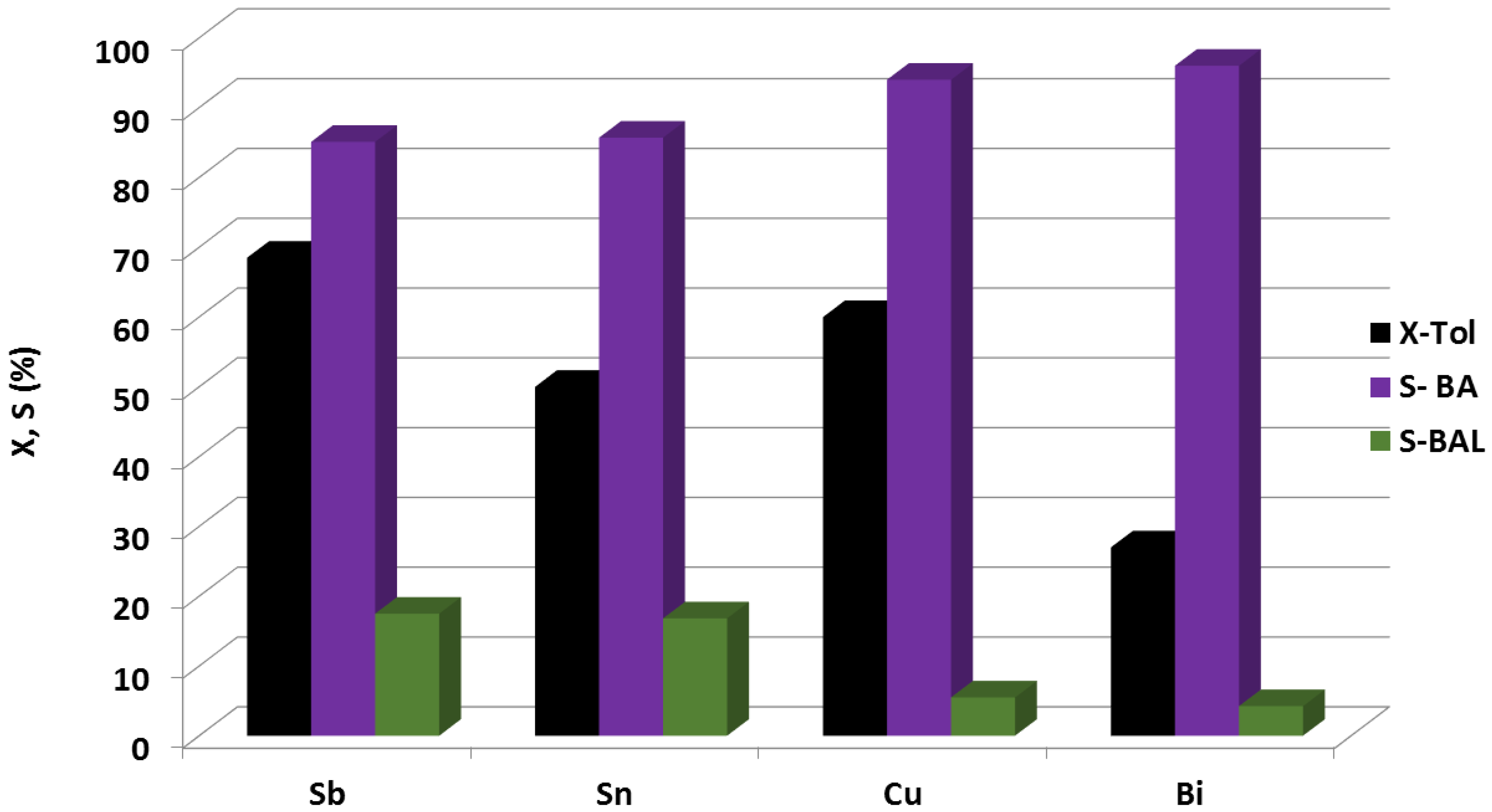
3.1. Palladium Based Bimetallic Catalysts
3.2. Gold Based Bimetallic Catalysts
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Alexeev, O.S.; Gates, B.C. Supported Bimetallic Cluster Catalysts. Ind. Eng. Chem. Res. 2003, 42, 1571–1587. [Google Scholar] [CrossRef]
- Campbell, C.T. Bimetallic Surface Chemistry. Annu. Rev. Phys. Chem. 1990, 41, 775–837. [Google Scholar] [CrossRef]
- Sinfelt, J.H. Bimetallic Catalysts: Discoveries, Concepts; Wiley-Interscience: Hoboken, NJ, USA, 1983. [Google Scholar]
- Somorjai, G.A.; Li, Y. Introduction to Surface Chemistry and Catalysis; John Wiley & Sons: Hoboken, NJ, USA, 2010. [Google Scholar]
- Tao, F.F. Synthesis, catalysis, surface chemistry and structure of bimetallic nanocatalysts. Chem. Soc. Rev. 2012, 41, 7977–7979. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Somorjai, G.A. Nanoscale advances in catalysis and energy applications. Nano Lett. 2010, 10, 2289–2295. [Google Scholar] [CrossRef] [PubMed]
- Bolz, F. Advanced Materials in Catalysis; Elsevier: London, UK, 2013. [Google Scholar]
- Alonso, D.M.; Wettstein, S.G.; Dumesic, J.A. Bimetallic catalysts for upgrading of biomass to fuels and chemicals. Chem. Soc. Rev. 2012, 41, 8075–8098. [Google Scholar] [CrossRef] [PubMed]
- Huynh, T.M.; Armbruster, U.; Pohl, M.-M.; Schneider, M.; Radnik, J.; Hoang, D.L.; Phan, B.M.Q.; Nguyen, D.A.; Martin, A. Hydrodeoxygenation of phenol as a model compound for bio-oil over non-noble bimetallic catalysts based on Ni. ChemCatChem 2014, 6, 1940–1951. [Google Scholar] [CrossRef]
- Sankar, M.; Dimitratos, N.; Miedziak, P.J.; Wells, P.P.; Kiely, C.J.; Hutchings, G.J. Designing bimetallic catalysts for a green and sustainable future. Chem. Soc. Rev. 2012, 41, 8099–8139. [Google Scholar] [CrossRef] [PubMed]
- Wei, Z.; Sun, J.; Li, Y.; Datye, A.K.; Wang, Y. Bimetallic catalysts for hydrogen generation. Chem. Soc. Rev. 2012, 41, 7994–8008. [Google Scholar] [CrossRef] [PubMed]
- Jiang, T.; Zhou, Y.; Liang, S.; Liu, H.; Han, B. Hydrogenolysis of glycerol catalyzed by Ru-Cu bimetallic catalysts supported on clay with the aid of ionic liquids. Green Chem. 2009, 11, 1000–1006. [Google Scholar] [CrossRef]
- Wettstein, S.G.; Bond, J.Q.; Alonso, D.M.; Pham, H.N.; Datye, A.K.; Dumesic, J.A. RuSn bimetallic catalysts for selective hydrogenation of levulinic acid to γ-valerolactone. Appl. Catal. B 2012, 117, 321–329. [Google Scholar] [CrossRef]
- Ponec, V. On the role of promoters in hydrogenations on metals; α, β-unsaturated aldehydes and ketones. Appl. Catal. A 1997, 149, 27–48. [Google Scholar] [CrossRef]
- Mäki-Arvela, P.; Hajek, J.; Salmi, T.; Murzin, D.Y. Chemoselective hydrogenation of carbonyl compounds over heterogeneous catalysts. Appl. Catal. A 2005, 292, 1–49. [Google Scholar] [CrossRef]
- Lisowski, P.; Colmenares, J.C.; Lomot, D.; Chernyayeva, O.; Lisovytskiy, D. Preparation by sonophotodeposition method of bimetallic photocatalysts Pd-Cu/TiO2 for sustainable gaseous selective oxidation of methanol to methyl formate. J. Mol. Catal. A Chem. 2016, 411, 247–256. [Google Scholar] [CrossRef]
- Sobczak, I.; Dembowiak, E. The effect of AuAg-MCF and AuAg-NbMCF catalysts pretreatment on the gold-silver alloy formation and the catalytic behavior in selective methanol oxidation with oxygen. J. Mol. Catal. A Chem. 2015, 409, 137–148. [Google Scholar] [CrossRef]
- Mo, X.; Hu, H.; Tan, D.; Guo, T. Bimetallic gold-palladium supported on 5A zeolite and their catalytic activity for vinyl acetate synthesis. Appl. Mech. Mate. 2014, 513–517. [Google Scholar] [CrossRef]
- Zaleska-Medynska, A.; Marchelek, M.; Diak, M.; Grabowska, E. Noble metal-based bimetallic nanoparticles: The effect of the structure on the optical, catalytic and photocatalytic properties. Adv. Colloid Interface Sci. 2016, 229, 80–107. [Google Scholar] [CrossRef] [PubMed]
- Cui, C.-H.; Li, H.-H.; Yu, S.-H. Large scale restructuring of porous Pt-Ni nanoparticle tubes for methanol oxidation: A highly reactive, stable, and restorable fuel cell catalyst. Chem. Sci. 2011, 2, 1611–1614. [Google Scholar] [CrossRef]
- Bin Saiman, M.I.; Brett, G.L.; Tiruvalam, R.; Forde, M.M.; Sharples, K.; Thetford, A. Involvement of Surface-Bound Radicals in the Oxidation of Toluene Using Supported Au-Pd Nanoparticles. Angew. Chem. Int. Ed. 2012, 51, 5981–5986. [Google Scholar] [CrossRef] [PubMed]
- Jirkovský, J.S.; Panas, I.; Ahlberg, E.; Halasa, M.; Romani, S.; Schiffrin, D.J. Single atom hot-spots at Au-Pd nanoalloys for electrocatalytic H2O2 Production. J. Am. Chem. Soc. 2011, 133, 19432–19441. [Google Scholar] [CrossRef] [PubMed]
- Mohl, M.; Dobo, D.; Kukovecz, A.; Konya, Z.; Kordas, K.; Wei, J. Formation of CuPd and CuPt bimetallic nanotubes by galvanic replacement reaction. J. Phys. Chem. C. 2011, 115, 9403–9409. [Google Scholar] [CrossRef]
- Huang, Q.; Yang, H.; Tang, Y.; Lu, T.; Akins, D.L. Carbon-supported Pt-Co alloy nanoparticles for oxygen reduction reaction. Electrochem. Commun. 2006, 8, 1220–1224. [Google Scholar] [CrossRef]
- Mohamed, M.B.; Volkov, V.; Link, S.; El-Sayed, M.A. The ‘lightning’ gold nanorods: Fluorescence enhancement of over a million compared to the gold metal. Chem. Phys. Lett. 2000, 317, 517–523. [Google Scholar] [CrossRef]
- Lee, K.-S.; El-Sayed, M.A. Gold and silver nanoparticles in sensing and imaging: Sensitivity of plasmon response to size, shape, and metal composition. J. Phys. Chem. Lett. B 2006, 110, 19220–19225. [Google Scholar] [CrossRef] [PubMed]
- Cheng, M.-T.; Liu, S.-D.; Wang, Q.-Q. Modulating emission polarization of semiconductor quantum dots through surface plasmon of metal nanorod. Appl. Phys. Lett. 2008, 92, 162107–162110. [Google Scholar] [CrossRef]
- Wang, H.; Brandl, D.W.; Le, F.; Nordlander, P.; Halas, N.J. Nanorice: A hybrid plasmonic nanostructure. Nano Lett. 2006, 6, 827–832. [Google Scholar] [CrossRef] [PubMed]
- Kreibig, U.; Vollmer, M. Optical Properties of Metal Clusters; Springer Science & Business Media: Berlin, Germany, 2013. [Google Scholar]
- Kim, K.; Kim, K.L.; Choi, J.-Y.; Lee, H.B.; Shin, K.S. Surface enrichment of Ag atoms in Au/Ag Alloy nanoparticles revealed by surface-enhanced Raman scattering of 2, 6-dimethylphenyl isocyanide. J. Phys. Chem. C 2010, 114, 3448–3453. [Google Scholar] [CrossRef]
- Ramakritinan, C.M.; Kaarunya, E.; Shankar, S.; Kumaraguru, A.K. Antibacterial effects of Ag, Au and bimetallic (Ag-Au) nanoparticles synthesized from red algae. Solid State Phenom. 2013, 201, 211–230. [Google Scholar] [CrossRef]
- Liu, X.; Wang, D.; Li, Y. Synthesis and catalytic properties of bimetallic nanomaterials with various architectures. Nano Today 2012, 7, 448–466. [Google Scholar] [CrossRef]
- Kyriakou, G.; Boucher, M.B.; Jewell, A.D.; Lewis, E.A.; Lawton, T.J.; Baber, A.E. Isolated metal atom geometries as a strategy for selective heterogeneous hydrogenations. Science 2012, 335, 1209–1212. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Watanabe, T.; Okumura, M.; Haruta, M.; Toshima, N. Catalytically highly active top gold atom on palladium nanocluster. Nat. Mater. 2012, 11, 49–52. [Google Scholar] [CrossRef] [PubMed]
- Cheng, K.; Sun, S. Recent advances in syntheses and therapeutic applications of multifunctional porous hollow nanoparticles. Nano Today 2010, 5, 183–196. [Google Scholar] [CrossRef]
- An, K.; Hyeon, T. Synthesis and biomedical applications of hollow nanostructures. Nano Today 2009, 4, 359–373. [Google Scholar] [CrossRef]
- Zhang, Q.; Wang, W.; Goebl, J.; Yin, Y. Self-templated synthesis of hollow nanostructures. Nano Today 2009, 4, 494–507. [Google Scholar] [CrossRef]
- Lim, B.; Jiang, M.; Yu, T.; Camargo, P.H.; Xia, Y. Nucleation and growth mechanisms for Pd-Pt bimetallic nanodendrites and their electrocatalytic properties. Nano Res. 2010, 3, 69–80. [Google Scholar] [CrossRef]
- Krylova, G.; Giovanetti, L.J.; Requejo, F.G.; Dimitrijevic, N.M.; Prakapenka, A.; Shevchenko, E.V. Study of nucleation and growth mechanism of the metallic nanodumbbells. J. Am. Chem. Soc. 2012, 134, 4384–4392. [Google Scholar] [CrossRef] [PubMed]
- Jiang, H.-L.; Akita, T.; Ishida, T.; Haruta, M.; Xu, Q. Synergistic catalysis of Au@Ag core-Shell nanoparticles stabilized on metal-organic framework. J. Am. Chem. Soc. 2011, 133, 1304–1306. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; Tang, S.; Liu, B.; Ren, B.; Zheng, N. Enhancing the Photothermal Stability of Plasmonic Metal Nanoplates by a Core-Shell Architecture. Adv. Mater. 2011, 23, 3420–3425. [Google Scholar] [CrossRef] [PubMed]
- Yoo, W.-J.; Miyamura, H.; Kobayashi, S. Polymer-Incarcerated Gold-Palladium Nanoclusters with Boron on Carbon: A Mild and Efficient Catalyst for the Sequential Aerobic Oxidation-Michael Addition of 1, 3-Dicarbonyl Compounds to Allylic Alcohols. J. Am. Chem. Soc. 2011, 133, 3095–3103. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.K.; Singh, A.K.; Aranishi, K.; Xu, Q. Noble-metal-free bimetallic nanoparticle-catalyzed selective hydrogen generation from hydrous hydrazine for chemical hydrogen storage. J. Am. Chem. Soc. 2011, 133, 19638–19641. [Google Scholar] [CrossRef] [PubMed]
- Suryanarayana, C. Mechanical alloying and milling. Prog. Mater. Sci. 2001, 46, 1–184. [Google Scholar] [CrossRef]
- Haruta, M.; Daté, M. Advances in the catalysis of Au nanoparticles. Appl. Catal. A 2001, 222, 427–437. [Google Scholar] [CrossRef]
- Pileni, M. Nanosized particles made in colloidal assemblies. Langmuir 1997, 13, 3266–3276. [Google Scholar] [CrossRef]
- Fu, X.; Wang, Y.; Wu, N.; Gui, L.; Tang, Y. Shape-selective preparation and properties of oxalate-stabilized Pt colloid. Langmuir 2002, 18, 4619–4624. [Google Scholar] [CrossRef]
- Toshima, N.; Kuriyama, M.; Yamada, Y.; Hirai, H. Colloidal platinum catalyst for light-induced hydrogen evolution from water. A particle size effect. Chem. Lett. 1981, 10, 793–806. [Google Scholar] [CrossRef]
- Lopez, N.; Janssens, T.; Clausen, B.; Xu, Y.; Mavrikakis, M.; Bligaard, T. On the origin of the catalytic activity of gold nanoparticles for low-temperature CO oxidation. J. Catal. 2004, 223, 232–235. [Google Scholar] [CrossRef]
- Friebel, D.; Miller, D.J.; Nordlund, D.; Ogasawara, H.; Nilsson, A. Degradation of Bimetallic Model Electrocatalysts: An In Situ X-Ray Absorption Spectroscopy Study. Angew. Chem. Int. Ed. 2011, 50, 10190–10192. [Google Scholar] [CrossRef] [PubMed]
- Ono, L.; Sudfeld, D.; Cuenya, B.R. In situ gas-phase catalytic properties of TiC-supported size-selected gold nanoparticles synthesized by diblock copolymer encapsulation. Surf. Sci. 2006, 600, 5041–5050. [Google Scholar] [CrossRef]
- Cuenya, B.R. Synthesis and catalytic properties of metal nanoparticles: Size, shape, support, composition, and oxidation state effects. Thin Solid Films 2010, 518, 3127–3150. [Google Scholar] [CrossRef]
- Molenbroek, A.M.; Nørskov, J.K.; Clausen, B.S. Structure and reactivity of Ni-Au nanoparticle catalysts. J. Phys. Chem. B 2001, 105, 5450–5458. [Google Scholar] [CrossRef]
- Besenbacher, F.; Chorkendorff, I.I.; Clausen, B.S.; Hammer, B.; Molenbroek, A.M.; Nørskov, J.K.; Stensgaard, I.I. Design of a surface alloy catalyst for steam reforming. Science 1998, 279, 1913–1918. [Google Scholar] [CrossRef] [PubMed]
- Meille, V. Review on methods to deposit catalysts on structured surfaces. Appl. Catal. A 2006, 315, 1–17. [Google Scholar] [CrossRef]
- Chen, M.; Goodman, D. The structure of catalytically active gold on titania. Science 2004, 306, 252–257. [Google Scholar] [CrossRef] [PubMed]
- Prati, L.; Villa, A. The art of manufacturing gold catalysts. Catalysts 2011, 2, 24–37. [Google Scholar] [CrossRef]
- Nishimura, S.; Takagaki, A.; Ebitani, K. Characterization, synthesis and catalysis of hydrotalcite-related materials for highly efficient materials transformations. Green Chem. 2013, 15, 2026–2042. [Google Scholar] [CrossRef]
- Debecker, D.P.; Mutin, P.H. Non-hydrolytic sol-gel routes to heterogeneous catalysts. Chem. Soc. Rev. 2012, 41, 3624–3650. [Google Scholar] [CrossRef] [PubMed]
- Ciriminna, R.; Fidalgo, A.; Pandarus, V.; Béland, F.; Ilharco, L.M.; Pagliaro, M. The sol-gel route to advanced silica-based materials and recent applications. Chem. Rev. 2013, 113, 6592–6620. [Google Scholar] [CrossRef] [PubMed]
- Esparza, R.; Téllez-Vázquez, O.; Ángeles-Pascual, A.; Pérez, R. Synthesis and characterization of bimetallic nanoparticles by cs-corrected scanning transmission electron microscopy. In Materials Characterization; Springer: Basel, Switzerland, 2015; pp. 35–42. [Google Scholar]
- Fedlheim, D.L.; Foss, C.A. Metal Nanoparticles: Synthesis, Characterization, and Applications; CRC Press: New York, NY, USA, 2001. [Google Scholar]
- Gontard, L.C.; Chang, L.Y.; Hetherington, C.J.; Kirkland, A.I.; Ozkaya, D.; Dunin-Borkowski, R.E. Aberration-Corrected Imaging of Active Sites on Industrial Catalyst Nanoparticles. Angew. Chem. Int. Ed. 2007, 119, 3757–3759. [Google Scholar] [CrossRef]
- Zhou, W.; Wachs, I.E.; Kiely, C.J. Nanostructural and chemical characterization of supported metal oxide catalysts by aberration corrected analytical electron microscopy. Curr. Opin. Solid State Mater. Sci. 2012, 16, 10–22. [Google Scholar] [CrossRef]
- Akita, T.; Kohyama, M.; Haruta, M. Electron microscopy study of gold nanoparticles deposited on transition metal oxides. Acc. Chem. Res. 2013, 46, 1773–1782. [Google Scholar] [CrossRef] [PubMed]
- Tozzola, G.; Mantegazza, M.; Ranghino, G.; Petrini, G.; Bordiga, S.; Ricchiardi, G. On the structure of the active site of Ti-silicalite in reactions with hydrogen peroxide: A vibrational and computational study. J. Catal. 1998, 179, 64–71. [Google Scholar] [CrossRef]
- Lee, H.; Habas, S.E.; Kweskin, S.; Butcher, D.; Somorjai, G.A.; Yang, P. Morphological control of catalytically active platinum nanocrystals. Angew. Chem. Int. Ed. 2006, 118, 7988–7992. [Google Scholar] [CrossRef]
- Zhang, W.; Luo, X.-J.; Niu, L.-N.; Yang, H.-Y.; Yiu, C.K.; Zhou, L.-Q. Biomimetic Intrafibrillar Mineralization of Type I Collagen with Intermediate Precursors-loaded Mesoporous Carriers. Sci. Rep. 2015, 5, 11199–11210. [Google Scholar] [CrossRef] [PubMed]
- Jackson, S.D.; Hargreaves, J.S. Metal oxide catalysis; Wiley Online Library: Berlin, Germany, 2009. [Google Scholar]
- Schoonheydt, R.A. UV-VIS-NIR spectroscopy and microscopy of heterogeneous catalysts. Chem. Soc. Rev. 2010, 39, 5051–5066. [Google Scholar] [CrossRef] [PubMed]
- Che, M.; Védrine, J.C. Characterization of Solid Materials and Heterogeneous Catalysts: From structure to surface reactivity; John Wiley & Sons: Hoboken, NJ, USA, 2012. [Google Scholar] [CrossRef]
- Busca, G. The Use of Infrared Spectroscopic Methods in the Field of Heterogeneous Catalysis by Metal Oxides. In Metal Oxide Catalysis; WILEY-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2009; pp. 95–175. [Google Scholar] [CrossRef]
- Pan, X.; Fan, Z.; Chen, W.; Ding, Y.; Luo, H.; Bao, X. Enhanced ethanol production inside carbon-nanotube reactors containing catalytic particles. Nat. Mater. 2007, 6, 507–511. [Google Scholar] [CrossRef] [PubMed]
- Borodko, Y.; Habas, S.E.; Koebel, M.; Yang, P.; Frei, H.; Somorjai, G.A. Probing the interaction of poly (vinylpyrrolidone) with platinum nanocrystals by UV-Raman and FTIR. J. Phys. Chem. B 2006, 110, 23052–23059. [Google Scholar] [CrossRef] [PubMed]
- Rivière, J.C.; Myhra, S. Handbook of Surface and Interface Analysis: Methods for Problem-Solving; CRC Press: Boca Raton, FL, USA, 2009. [Google Scholar] [CrossRef]
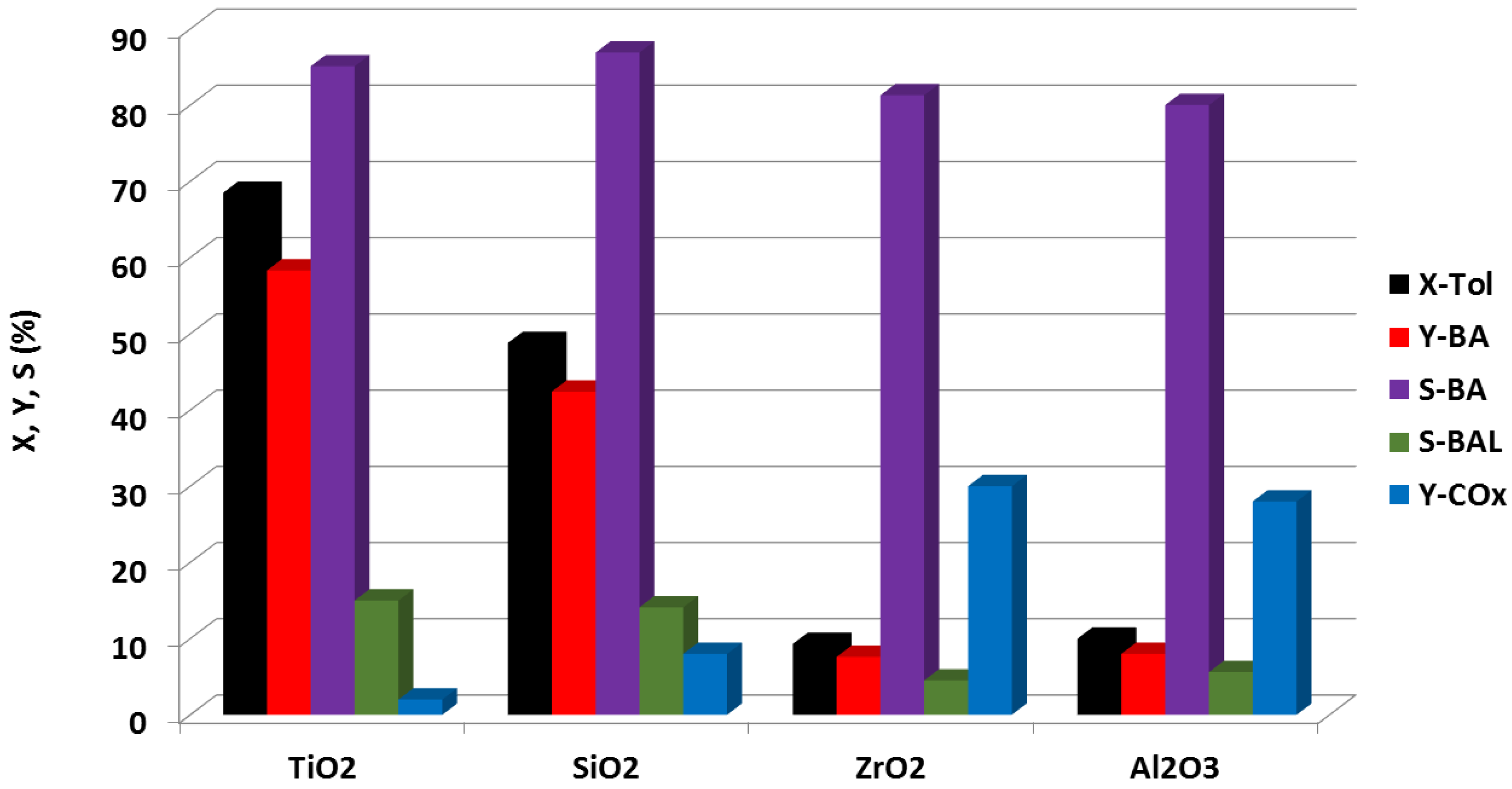
- Benhmid, A.; Narayana, K.V.; Martin, A.; Lücke, B.; Pohl, M.-M. Highly Active and Selective Pd-Cu-TiO2 catalyst for the direct synthesis of benzyl acetate by gas phase acetoxylation of toluene. Chem. Lett. 2004, 33, 1238–1239. [Google Scholar] [CrossRef]
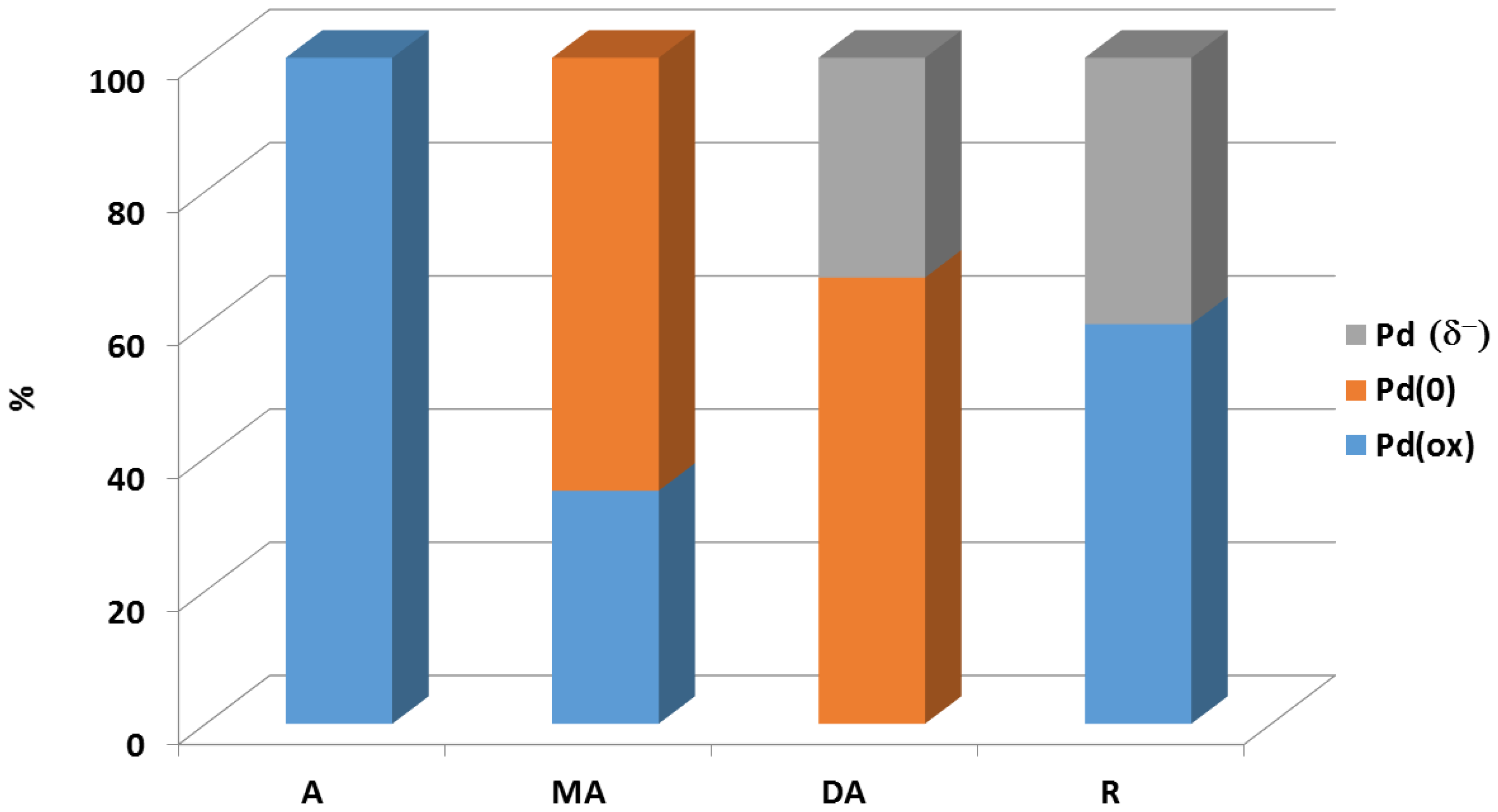
- Radnik, J.; Benhmid, A.; Kalevaru, V.N.; Pohl, M.M.; Martin, A.; Lücke, B.; Dingerdissen, U. Deactivation of Pd acetoxylation catalysts: Direct observations by XPS investigations. Angew. Chem. Int. Ed. 2005, 44, 6771–6774. [Google Scholar] [CrossRef] [PubMed]
- Radnik, J.; Pohl, M.-M.; Kalevaru, V.N.; Martin, A. First knowledge on the formation of novel core-shell structures in PdCu catalysts and their influence on the prevention of catalyst deactivation. J. Phys. Chem. C 2007, 111, 10166–10169. [Google Scholar] [CrossRef]
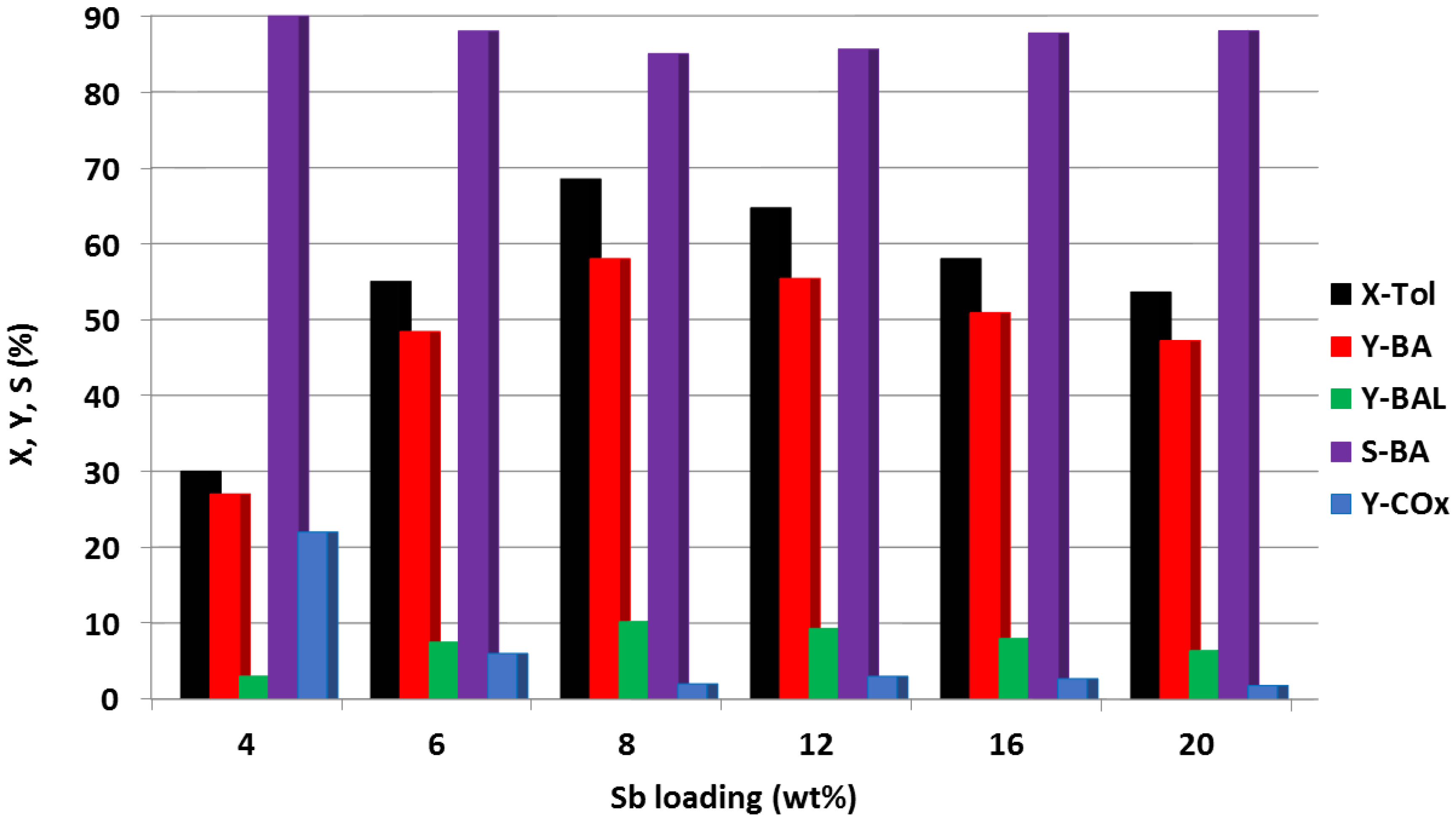
- Benhmid, A.; Narayana, K.V.; Martin, A.; Lücke, B.; Bischoff, S.; Pohl, M.-M.; Radnik, J.; Schneider, M. Highly efficient Pd-Sb-TiO2 catalysts for the vapour phase acetoxylation of toluene to benzyl acetate. J. Catal. 2005, 230, 420–435. [Google Scholar] [CrossRef]
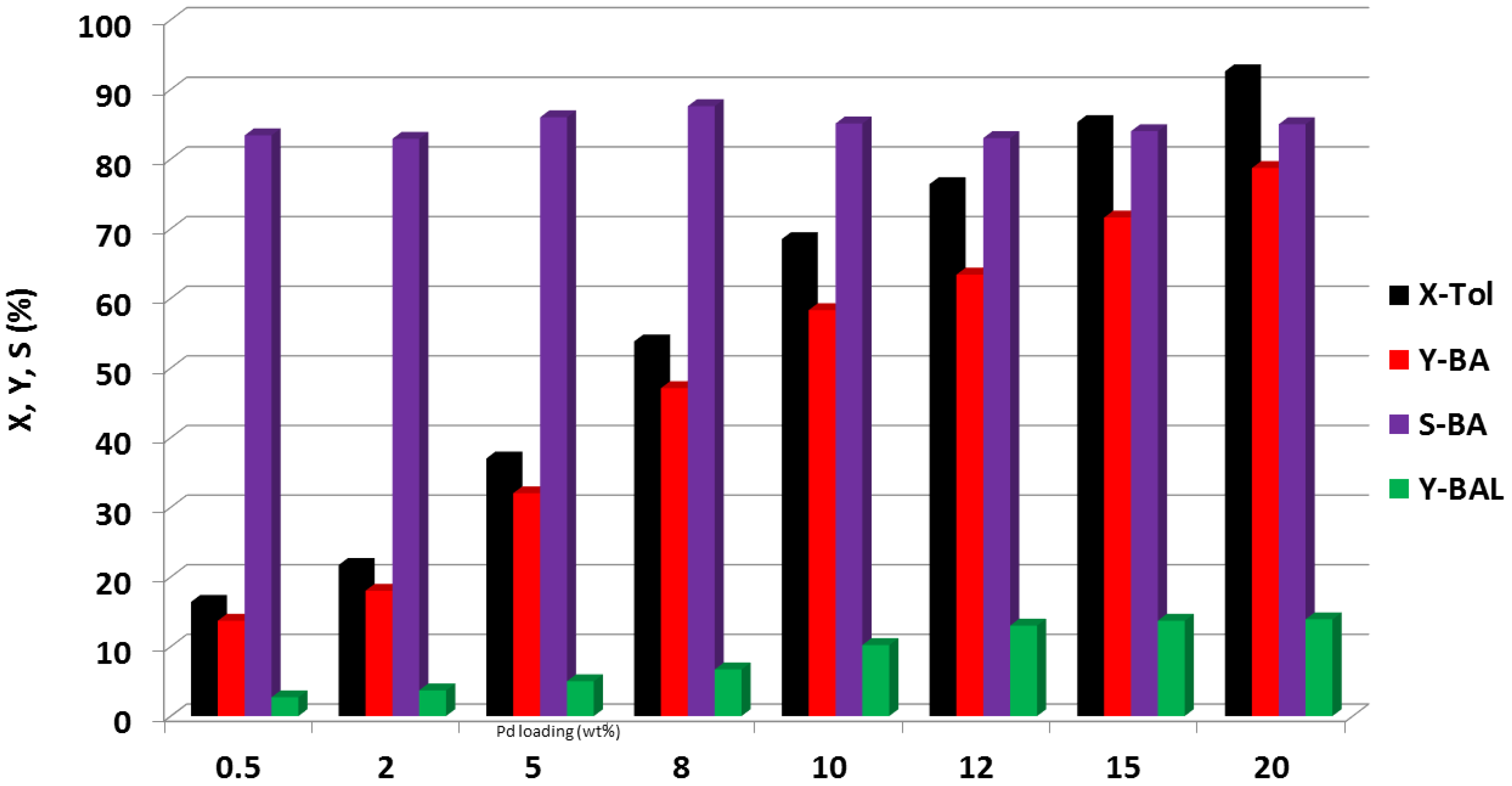
- Benhmid, A.; Narayana, K.V.; Radnik, J.; Martin, A.; Lücke, B. Effect of Sb loading on Pd nanoparticles and its influence on the catalytic performance of Sb-Pd/TiO2 solids for acetoxylation of toluene. J. Catal. 2006, 243, 25–35. [Google Scholar] [CrossRef]
- Gatla, S.; Radnik, J.; Madaan, N.; Pohl, M.-M.; Mathon, O.; Rogalev, A.; Kalevaru, V.N.; Martin, A.; Pascarelli, S.; Brueckner, A. New insights into the nature of co-components and their impact on Pd-structure: X-ray absorption studies on toluene acetoxylation studies. Chem. Eur. J. 2015, 21, 15280–15289. [Google Scholar] [CrossRef] [PubMed]
- Shu, Q.; Wang, X.; Ren, S.; Shi, W. Gas-phase oxidative acetoxylation of toluene over Pd-Sn-K/SiO2 catalyst. Cuihua Xuebao 2005, 26, 869–873. [Google Scholar]
- Komatsu, T.; Inaba, K.; Uezono, T.; Onda, A.; Yashima, T. Nano-size particles of palladium intermetallic compounds as catalysts for oxidative acetoxylation. Appl. Catal. A 2003, 251, 315–326. [Google Scholar] [CrossRef]
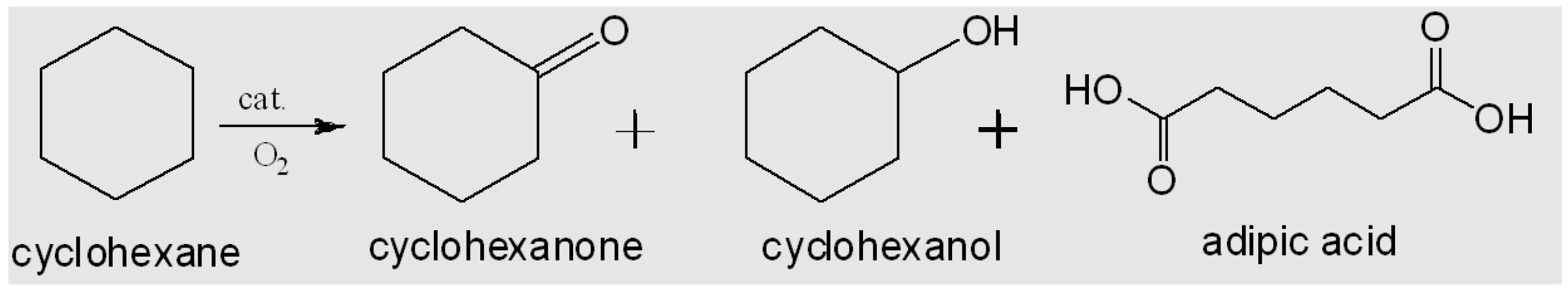
- Alshammari, A.; Koeckritz, A.; Kalevaru, V.N.; Bagabas, A.; Martin, A. Potential of Supported Gold Bimetallic Catalysts for Green Synthesis of Adipic Acid from Cyclohexane. Top. Catal. 2015, 58, 1069–1076. [Google Scholar] [CrossRef]
- Alshammari, A.; Koeckritz, A.; Kalevaru, V.N.; Bagabas, A.; Martin, A. Significant Formation of Adipic Acid by Direct Oxidation of Cyclohexane Using Supported Nano-Gold Catalysts. Chem. Cat. Chem. 2012, 4, 1330–1336. [Google Scholar] [CrossRef]
- Hereijgers, B.P.C.; Weckhuysen, B.M. Aerobic oxidation of cyclohexane by gold-based catalysts: New mechanistic insight by thorough product analysis. J. Catal. 2010, 270. [Google Scholar] [CrossRef]


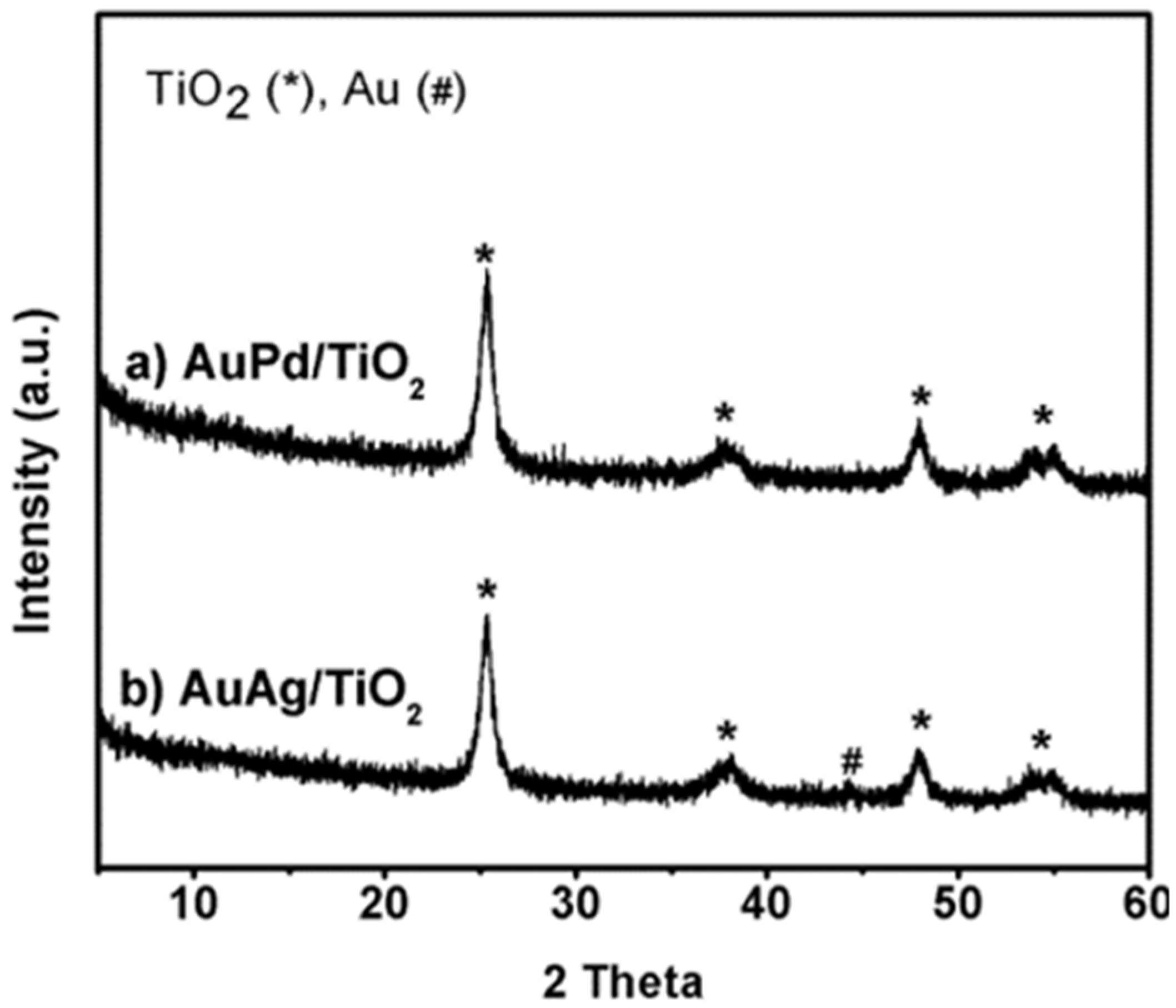
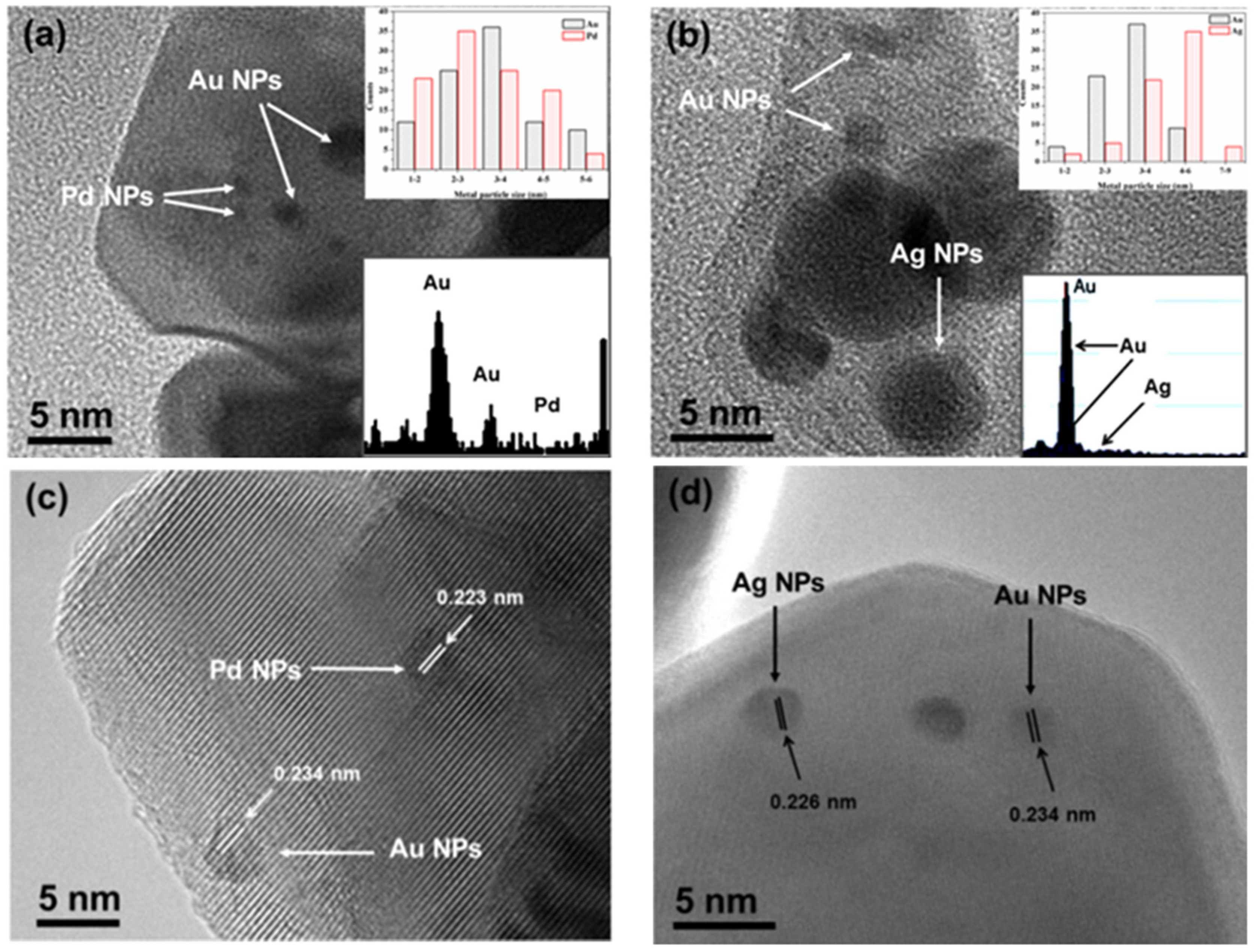
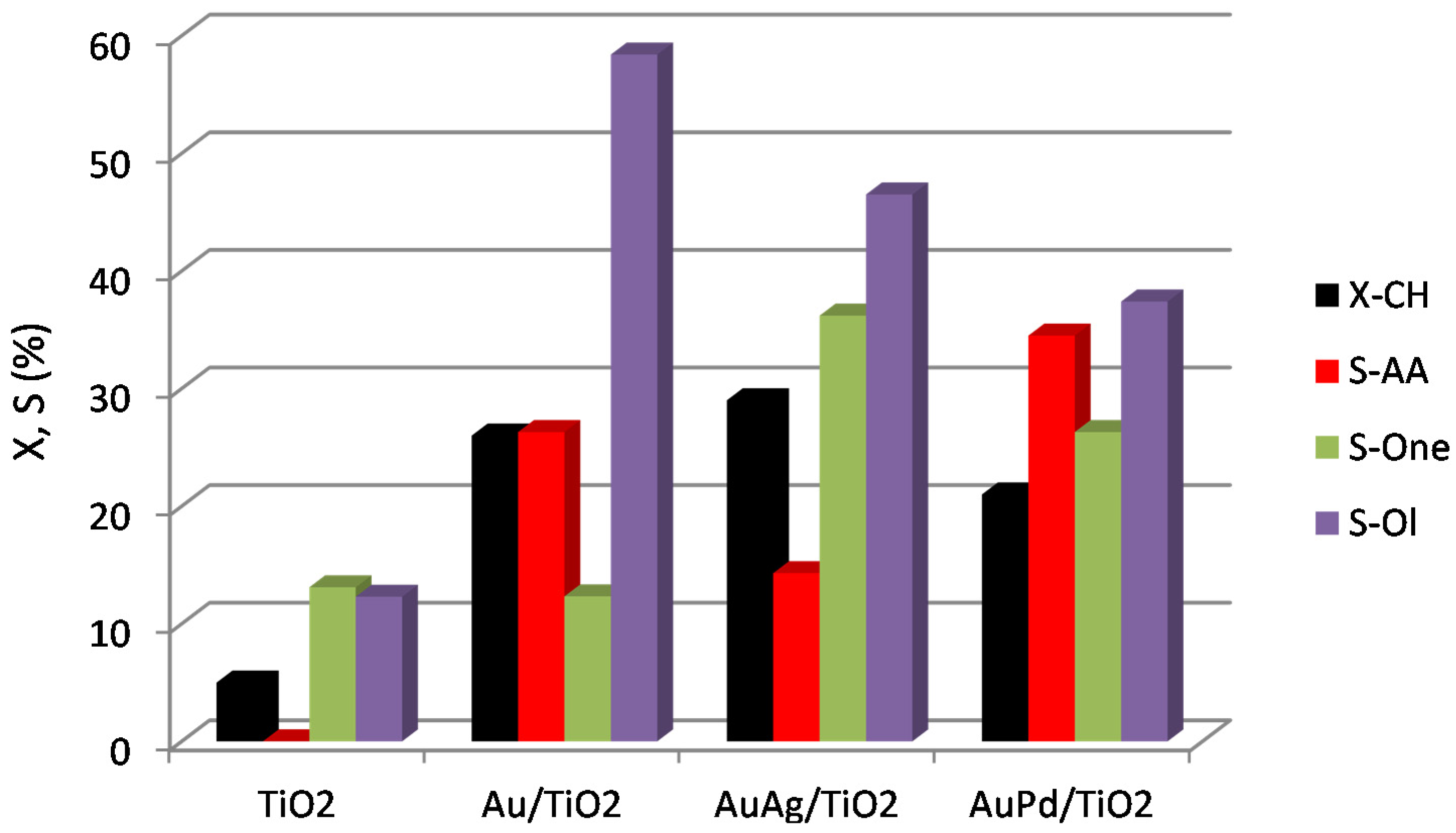
| Entry | Catalyst | Catalyst Composition (ICP) | BET-SA (m2/g) | Pore Volume (cm3/g) | |
|---|---|---|---|---|---|
| Au (wt %) | Pd/Ag (wt %) | ||||
| 1 | Au/TiO2 | 0.9 | - | 43 | 0.12 |
| 2 | Pd/TiO2 | - | 1.0 | 55 | 0.80 |
| 3 | Ag/TiO2 | - | 1.0 | 39 | 0.90 |
| 4 | AuPd/TiO2 | 1.1 | 1.2/- | 38 | 0.071 |
| 5 | AuAg/TiO2 | 1.2 | -/1.2 | 31 | 0.057 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alshammari, A.; Kalevaru, V.N.; Martin, A. Bimetallic Catalysts Containing Gold and Palladium for Environmentally Important Reactions. Catalysts 2016, 6, 97. https://doi.org/10.3390/catal6070097
Alshammari A, Kalevaru VN, Martin A. Bimetallic Catalysts Containing Gold and Palladium for Environmentally Important Reactions. Catalysts. 2016; 6(7):97. https://doi.org/10.3390/catal6070097
Chicago/Turabian StyleAlshammari, Ahmad, V. Narayana Kalevaru, and Andreas Martin. 2016. "Bimetallic Catalysts Containing Gold and Palladium for Environmentally Important Reactions" Catalysts 6, no. 7: 97. https://doi.org/10.3390/catal6070097
APA StyleAlshammari, A., Kalevaru, V. N., & Martin, A. (2016). Bimetallic Catalysts Containing Gold and Palladium for Environmentally Important Reactions. Catalysts, 6(7), 97. https://doi.org/10.3390/catal6070097








