Efficient Dehydration of Fructose to 5-Hydroxy-methylfurfural Catalyzed by Heteropolyacid Salts
Abstract
:1. Introduction
2. Results and Discussion
2.1. Effect of Reaction Medium on the Fructose to 5-HMF Conversion Yield
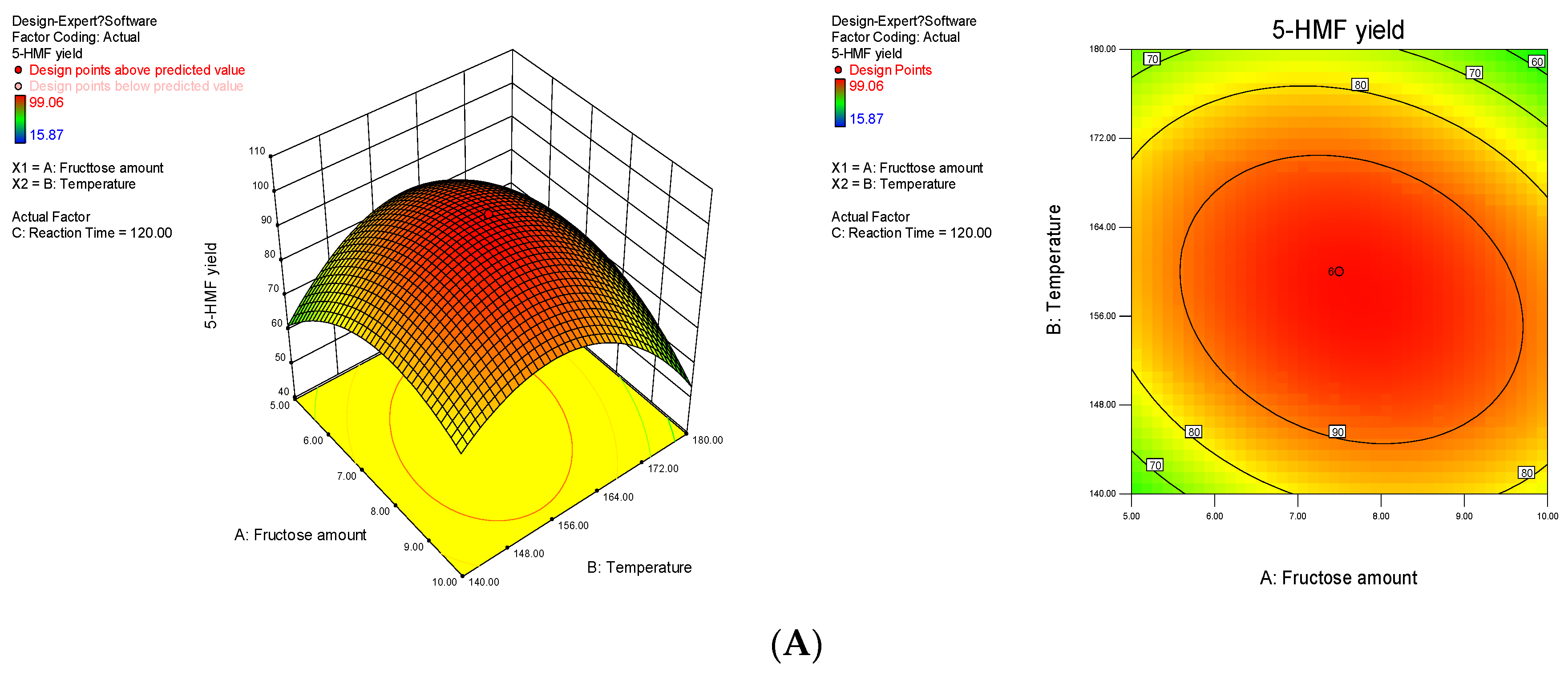
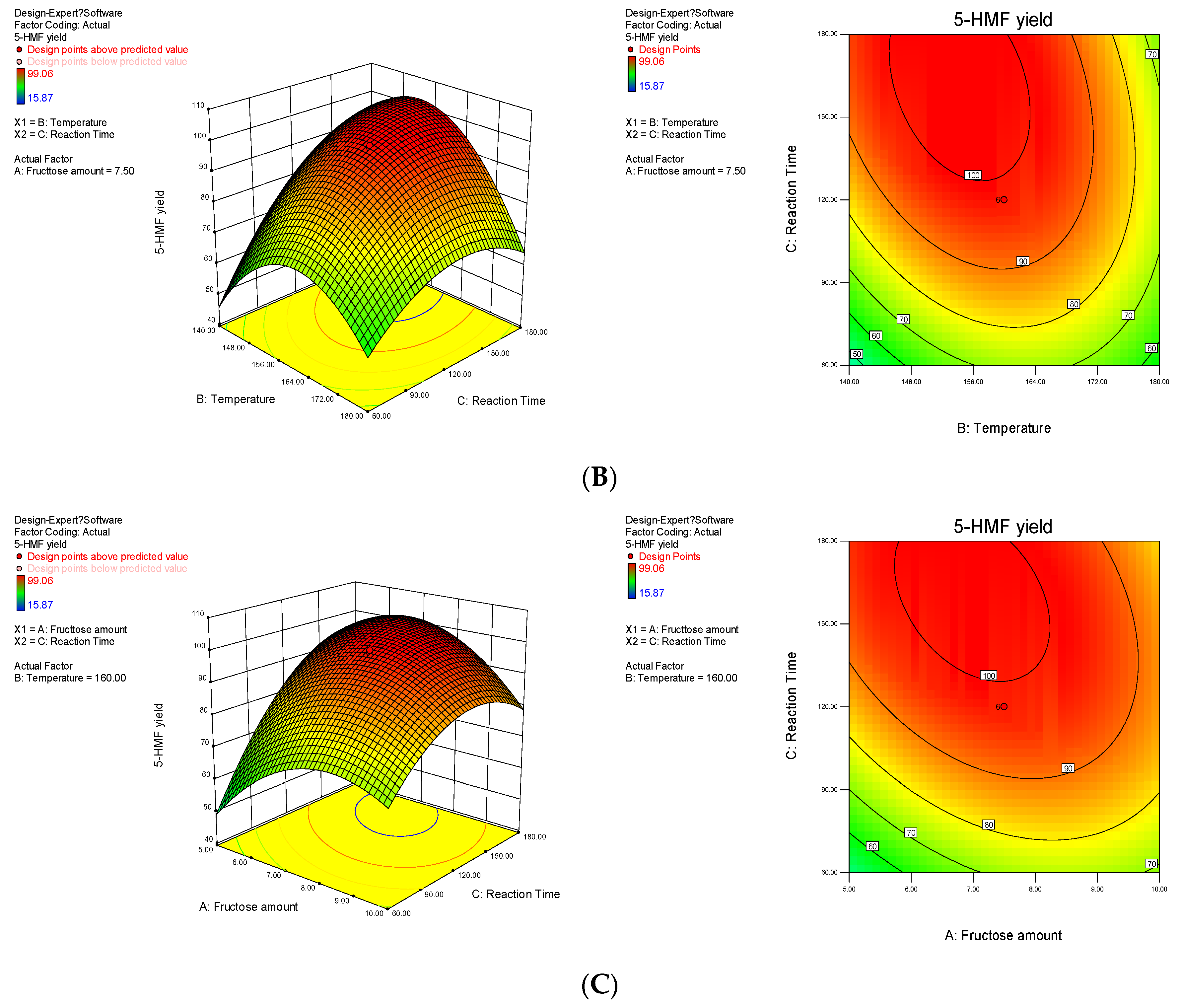
2.2. Optimization of Process Conditions
2.3. Optimisation and Confirmation Experiments
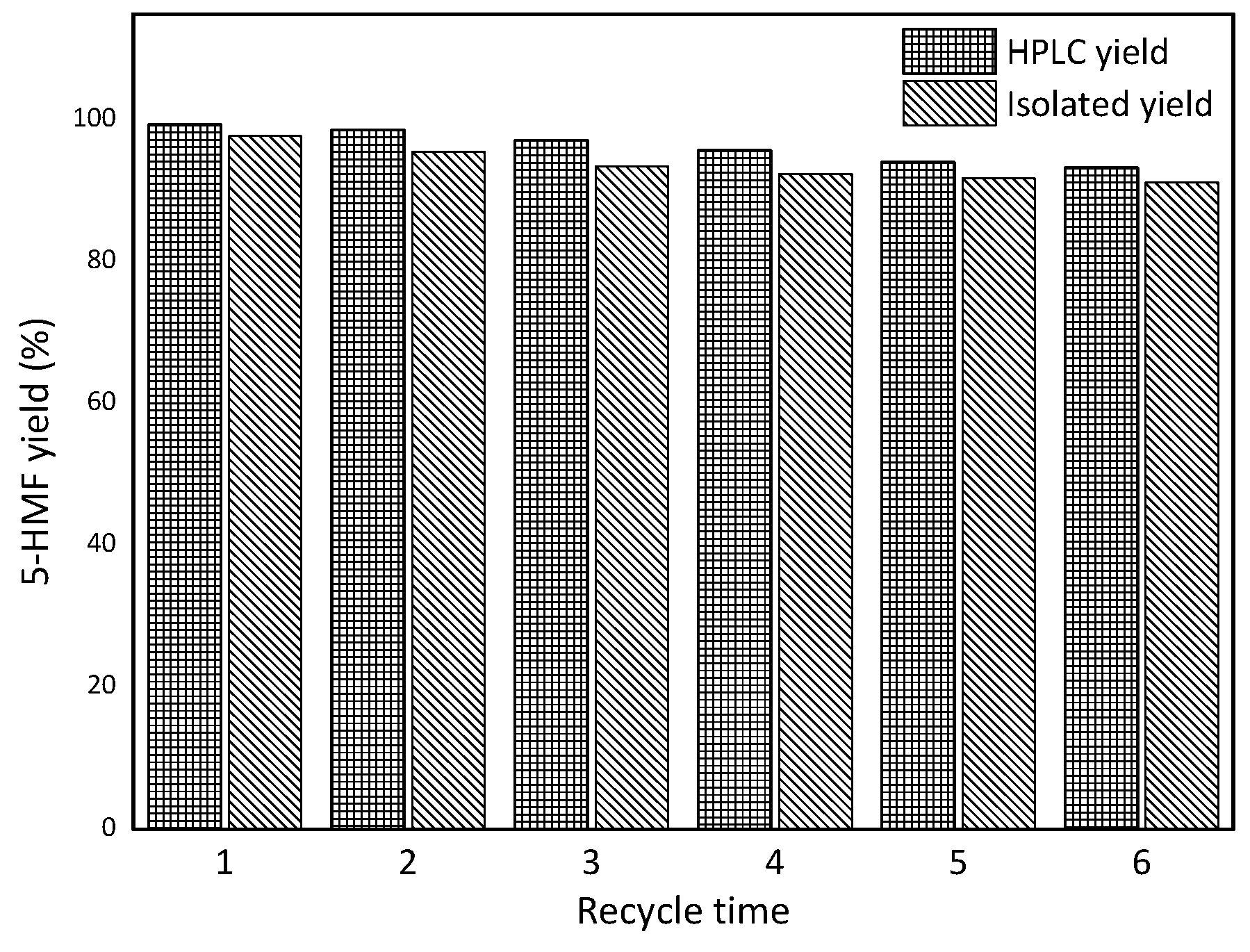
2.4. Catalyst Recycling
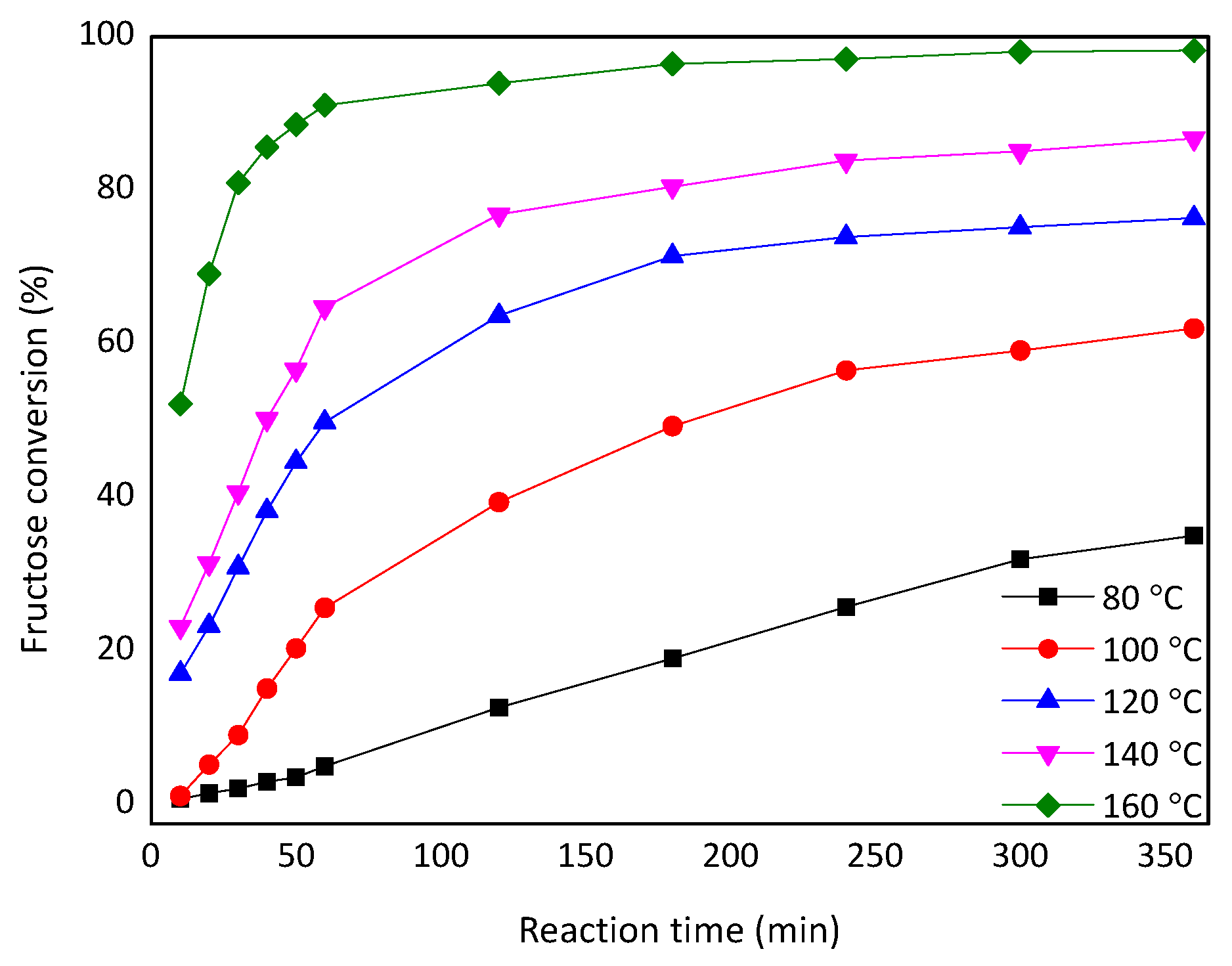
2.5. Kinetic Model
3. Experimental Section
3.1. Materials and Catalyst Preparation
3.2. General Procedure for the Conversion of Fructose to 5-HMF
3.3. Design of Experiments
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Corma, A.; Iborra, S.; Velty, A. Chemical routes for the transformation of biomass into chemicals. Chem. Rev. 2007, 107, 2411–2502. [Google Scholar] [PubMed]
- Wang, X.; Song, Y.; Huang, C.; Liang, F.; Chen, B. Lactic acid production from glucose over polymer catalysts in aqueous alkaline solution under mild conditions. Green Chem. 2014, 16, 4234–4240. [Google Scholar]
- Shi, W.; Li, S.; Jia, J.; Zhao, Y. Highly efficient conversion of cellulose to bio-oil in hot-compressed water with ultrasonic pretreatment. Ind. Eng. Chem. Res. 2013, 52, 586–593. [Google Scholar]
- Binder, J.B.; Raines, R.T. Simple chemical transformation of lignocellulosic biomass into furans for fuels and chemicals. J. Am. Chem. Soc. 2009, 131, 1979–1985. [Google Scholar] [CrossRef] [PubMed]
- Climent, M.J.; Corma, A.; Iborra, S. Converting carbohydrates to bulk chemicals and fine chemicals over heterogeneous catalysts. Green Chem. 2011, 13, 520–540. [Google Scholar] [CrossRef]
- Su, Y.; Brown, H.M.; Huang, X.; Zhou, X.D.; Amonette, J.E.; Zhang, Z.C. Single-step conversion of cellulose to 5-hydroxymethylfurfural (HMF), a versatile platform chemical. Appl. Catal. A 2009, 361, 117–122. [Google Scholar] [CrossRef]
- Lv, X.N.; Li, G.; Yang, F.; Gao, P.; Liu, Z.H.; Meng, L.; Yu, X.Q. Homogeneous degradation of cotton cellulose into furan derivatives in ZnCl2 solution by integration technology of reaction and extraction. Ind. Eng. Chem. Res. 2013, 52, 297–302. [Google Scholar]
- Rosatella, A.A.; Simeonov, S.P.; Fradea, R.F.; Afonso, C.A. 5-Hydroxymethylfurfural (HMF) as a building block platform: Biological properties, synthesis and synthetic applications. Green Chem. 2011, 13, 754–793. [Google Scholar] [CrossRef]
- Agirrezaba-Tellería, I.; Gandarias, I.; Arias, P.L. Heterogeneous acid-catalysts for the production of furan-derived compounds (furfural and hydroxymethylfurfural) from renewable carbohydrates: A review. Catal. Today 2014, 234, 43–58. [Google Scholar]
- Tong, X.L.; Ma, Y.; Li, Y.D. Biomass into chemicals: Conversion of sugars to furan derivatives by catalytic processes. Appl. Catal. A 2010, 385, 1–13. [Google Scholar] [CrossRef]
- Zakrzewska, M.E.; Bogel-Łukasik, E.; Bogel-Łukasik, R. Ionic liquid-mediated formation of 5-hydroxymethylfurfural: A promising biomass-derived building block. Chem. Rev. 2011, 111, 397–417. [Google Scholar] [CrossRef] [PubMed]
- Zhao, H.; Holladay, J.E.; Brown, H.; Zhang, Z. Metal chlorides in ionic liquid solvents convert sugars to 5-hydroxymethylfurfural. Science 2007, 316, 1597–1600. [Google Scholar] [CrossRef] [PubMed]
- Román-Leshkov, Y.; Chheda, J.N.; Dumesic, J.A. Phase modifiers promote efficient production of hydroxymethylfurfural from fructose. Science 2006, 312, 1933–1937. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.; Nolte, M.W.; Shanks, B.H. Catalytic dehydration of C6 carbohydrates for the production of hydroxymethylfurfural (HMF) as a versatile platform chemical. Green Chem. 2014, 16, 548–572. [Google Scholar] [CrossRef]
- Qi, X.; Guo, H.; Li, L. Efficient conversion of fructose to 5-hydroxymethylfurfural catalyzed by sulfated zirconia in ionic liquids. Ind. Eng. Chem. Res. 2011, 50, 7985–7989. [Google Scholar] [CrossRef]
- Moreau, C.; Finiels, A.; Vanoye, L. Dehydration of fructose and sucrose into 5-hydroxymethylfurfural in the presence of 1-H-3-methyl imidazolium chloride acting both as solvent and catalyst. J. Mol. Catal. A 2006, 253, 165–169. [Google Scholar] [CrossRef]
- Hu, S.; Zhang, Z.; Song, J.; Zhou, Y.; Han, B. Efficient conversion of glucose into 5-hydroxymethylfurfural catalyzed by a common Lewis acid SnCl4 in an ionic liquid. Green Chem. 2009, 11, 1746–1749. [Google Scholar] [CrossRef]
- Hu, L.; Sun, Y.; Lin, L. Efficient conversion of glucose into 5-hydroxymethylfurfural by chromium (III) chloride in inexpensive ionic liquid. Ind. Eng. Chem. Res. 2012, 51, 1099–1104. [Google Scholar] [CrossRef]
- Wei, Z.; Li, Y.; Thushara, D.; Liu, Y.; Ren, Q. Novel dehydration of carbohydrates to 5-hydroxymethylfurfural catalyzed by Ir and Au chlorides in ionic liquids. J. Taiwan Inst. Chem. E 2011, 42, 363–370. [Google Scholar] [CrossRef]
- Carlini, C.; Patrono, P.; Galletti, A.M.R.; Sbrana, G. Heterogeneous catalysts based on vanadyl phosphate for fructose dehydration to 5-hydroxymethyl-2-furaldehyde. Appl. Catal. A 2004, 275, 111–118. [Google Scholar] [CrossRef]
- Moreau, C.; Durand, R.; Razigade, S.; Duhamet, J.; Faugeras, P.; Rivalier, P. Dehydration of fructose to 5-hydroxymethylfurfural over H-mordenites. Appl. Catal. A 1996, 145, 211–224. [Google Scholar] [CrossRef]
- Armaroli, T.; Busca, G.; Carlini, C.; Giuttari, M.; Galletti, A.M.R.; Sbrana, G. Acid sites characterization of niobium phosphate catalysts and their activity in fructose dehydration to 5-hydroxymethyl-2-furaldehyd. J. Mol. Catal. A 2000, 151, 233–243. [Google Scholar] [CrossRef]
- Qi, X.; Watanabe, M.; Aida, T.M.; Smith, R.L., Jr. Catalytical conversion of fructose and glucose into 5-hydroxymethylfurfural in hot compressed water by microwave heating. Catal. Commun. 2008, 9, 2244–2249. [Google Scholar] [CrossRef]
- Shimizu, K.; Uozumi, R.; Satsuma, A. Enhanced production of hydrox ymethylfurfural from fructose with solid acid catalysts by simple water removal methods. Catal. Commun. 2009, 10, 1849–1853. [Google Scholar] [CrossRef]
- Qu, J.P.; Zhou, Y.H.; Liu, X.Y.; Qiu, J.J.; Qu, L.H. A method to produce 5-hydroxymethylfurfural. Chinese Patent 10, CN 101289435, 22 October 2008. [Google Scholar]
- Zhao, Q.; Wang, L.; Zhao, S.; Wang, X.; Wang, S. High selective production of 5-hydroymethylfurfural from fructose by a solid heteropolyacid catalyst. Fuel 2011, 90, 2289–2293. [Google Scholar] [CrossRef]
- Qi, X.H.; Watanabe, M.; Aida, T.M.; Smith, R.L. Selective conversion of D-fructose to 5-hydroxymethylfurfural by ion-exchange resin in acetone/dimethyl sulfoxide solvent mixtures. Ind. Eng. Chem. Res. 2008, 47, 9234–9239. [Google Scholar] [CrossRef]
- Qi, X.; Watanabe, M.; Aida, T.M.; Smith, R.L. Fast Transformation of Glucose and Di-/Polysaccharides into 5-Hydroxymethylfurfural by Microwave Heating in an Ionic Liquid/Catalyst System. ChemSusChem 2010, 3, 1071–1077. [Google Scholar] [CrossRef] [PubMed]
- Bicker, M.; Hirth, J.; Vogel, H. Dehydration of fructose to 5-hydroxymethylfurfural in sub-and supercritical acetone. Green Chem. 2003, 5, 280–284. [Google Scholar] [CrossRef]
- Qu, Y.S.; Huang, C.P.; Song, Y.L.; Zhang, J.; Chen, B.H. Efficient dehydration of glucose to 5-hydroxymethylfurfural catalyzed by the ionic liquid, 1-hydroxyethyl-3-methylimidazolium tetrafluoroborate. Bioresour. Technol. 2012, 121, 462–466. [Google Scholar] [CrossRef] [PubMed]
- Gan, J.; Yuan, W.Q. Operating condition optimization of corncob hydrothermal conversion for bio-oil production. Appl. Energy 2013, 103, 350–357. [Google Scholar] [CrossRef]
- Means, N.E.; Starbuck, C.J.; Kremer, R.J.; Jett, L.W. Effects of a food waste-based soil conditioner on soil properties and plant growth. Compost Sci. Util. 2005, 13, 116–121. [Google Scholar] [CrossRef]
- Utami, S.P.; Amin, N.A.S. Optimization of glucose conversion to 5-hydroxymethylfulfural using [BMIM] Cl with ytterbium triflate. Ind. Crops Prod. 2013, 41, 64–70. [Google Scholar] [CrossRef]
- Liu, J.; Yan, M.; Zhang, Y.K.; Du, K.F. Study of glutamate-modified cellulose beads for Cr (III) adsorption by response surface methodology. Ind. Eng. Chem. Res. 2011, 50, 10784–10791. [Google Scholar] [CrossRef]
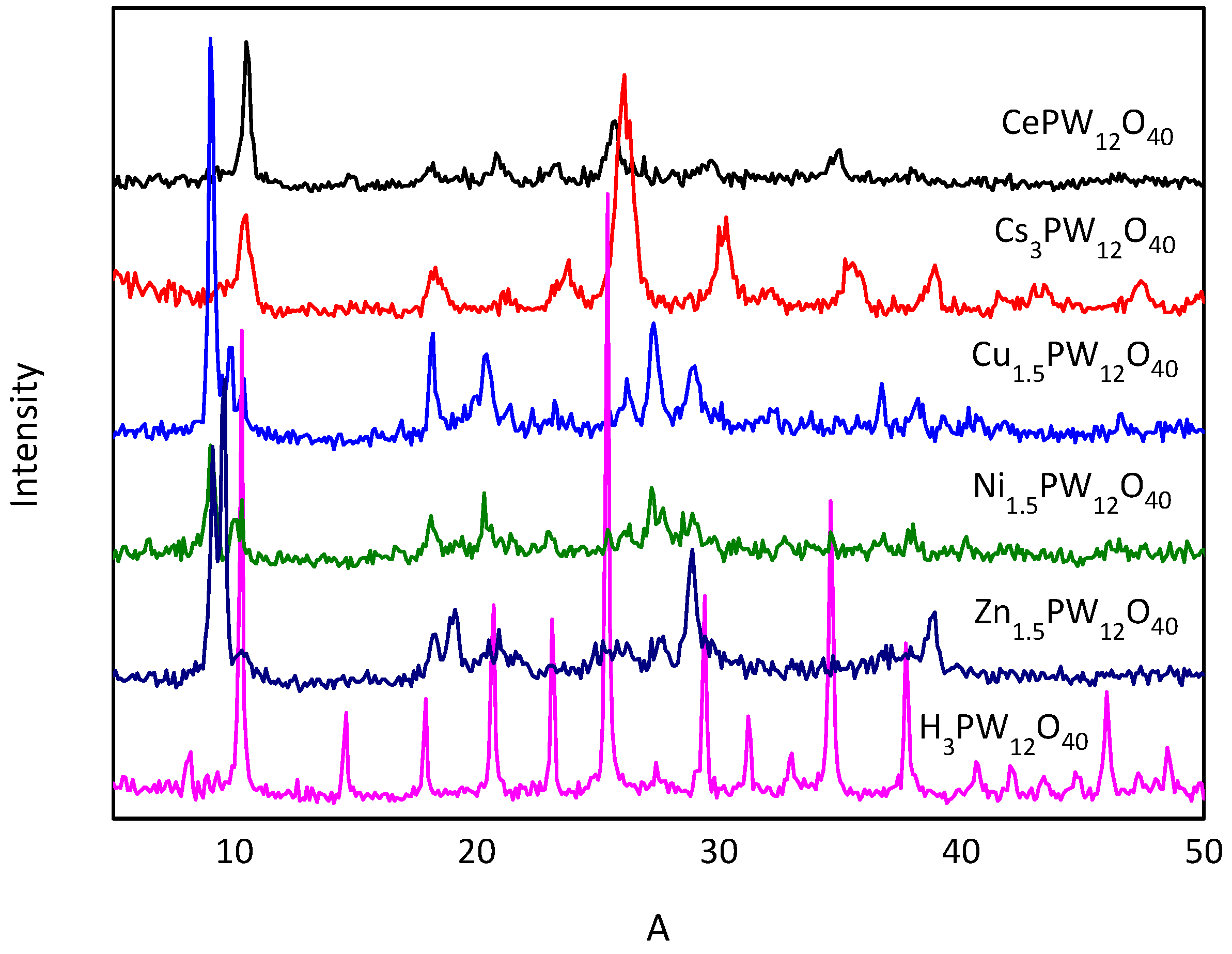
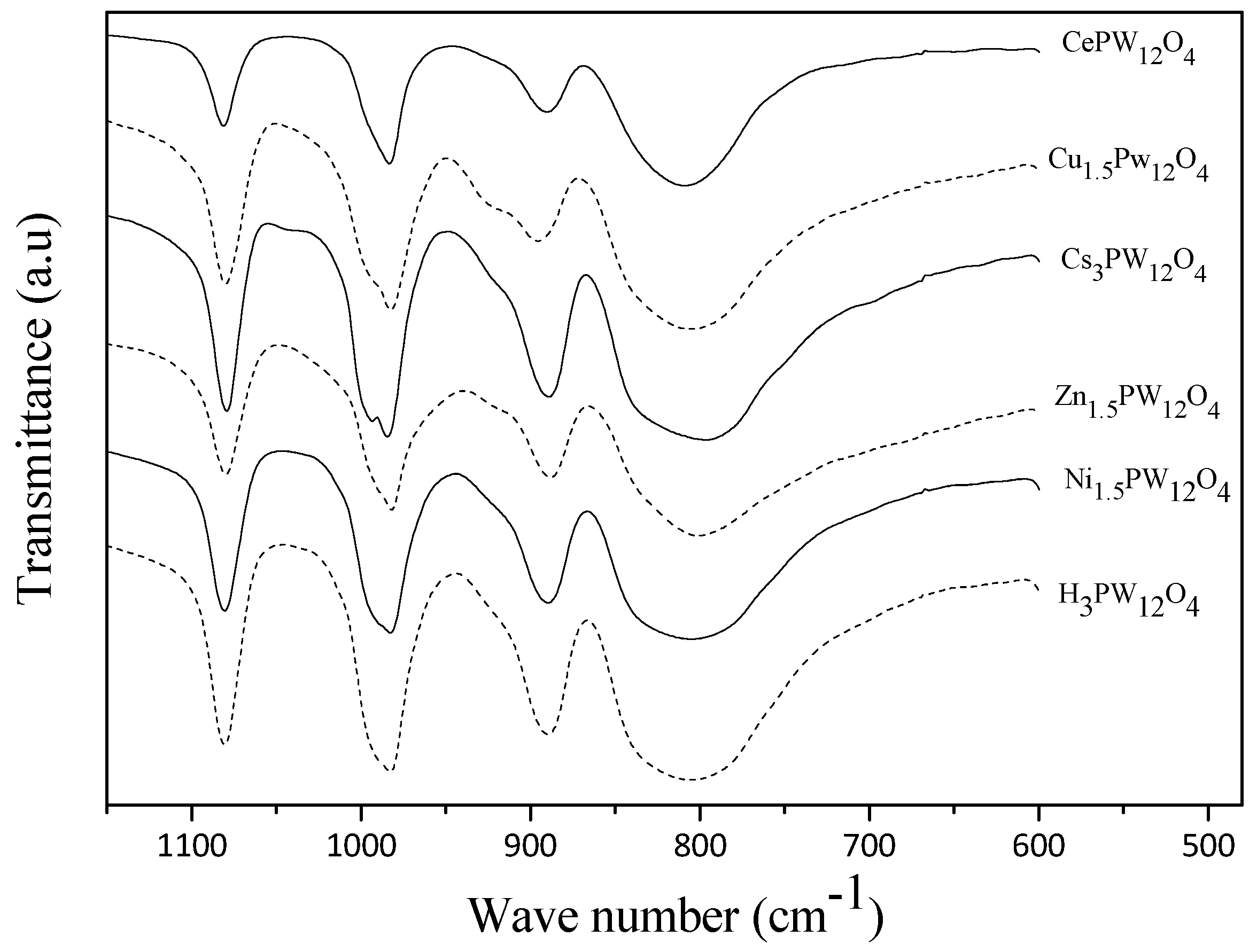

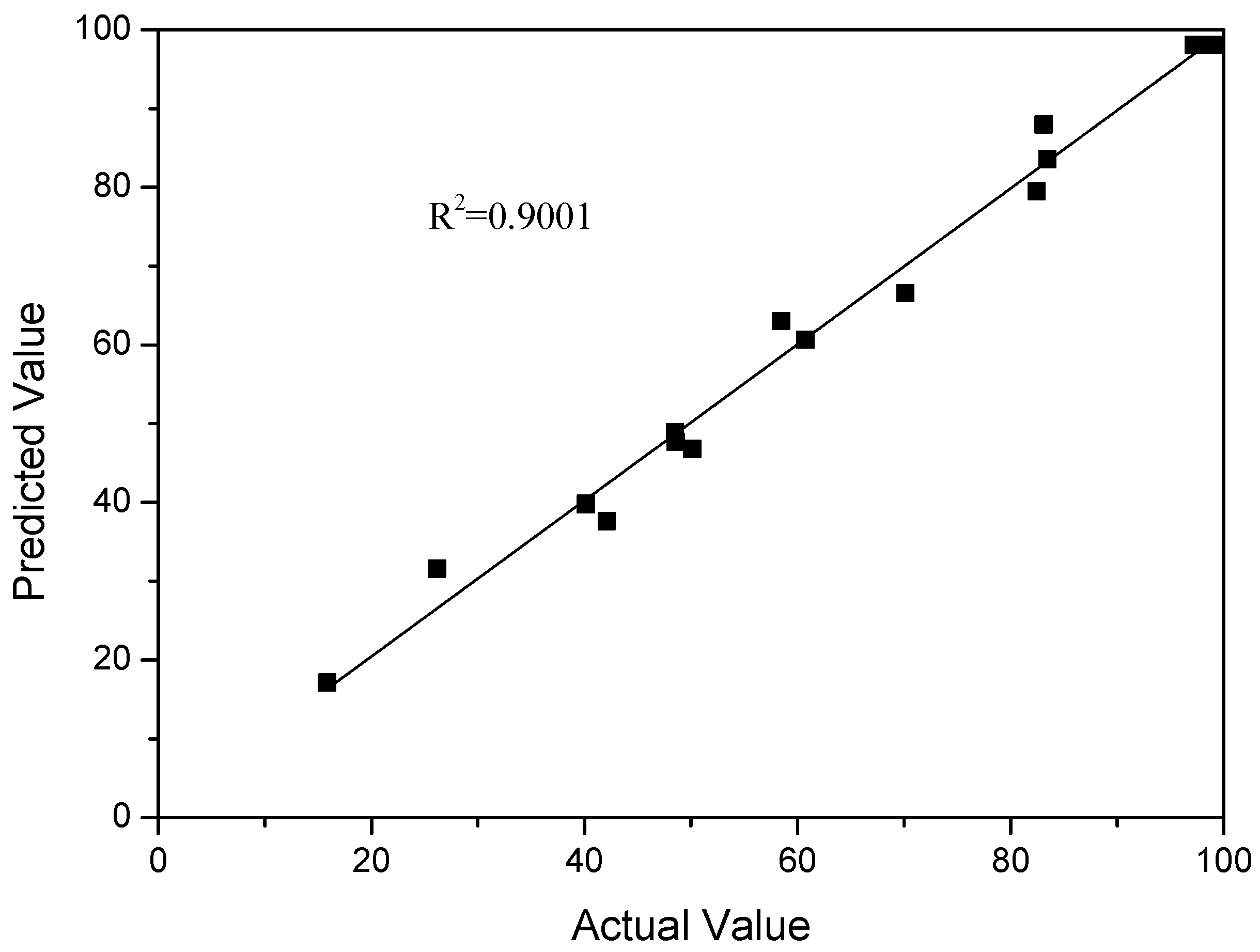
| Entry | Solvent | Catalyst | Reaction Time/min | Conv. % | Yield % |
|---|---|---|---|---|---|
| 1 | DMSO | Blank | 60 | 18.11 | 17.53 |
| 2 | n-Butanol | Blank | 60 | 1.56 | 0.58 |
| 3 | sec-Butanol | Blank | 60 | 2.15 | 1.04 |
| 4 | sec-Butanol | K3PW12O40 | 60 | 75.85 | 71.43 |
| 5 | DMSO | Zn1.5PW12O40 | 60 | 27.52 | 26.22 |
| 6 | DMSO | Ni1.5PW12O40 | 60 | 62.42 | 60.67 |
| 7 | DMSO | Cs3PW12O40 | 60 | 75.73 | 73.59 |
| 8 | DMSO | Cu1.5PW12O40 | 60 | 85.68 | 82.71 |
| 9 | DMSO | CePW12O40 | 60 | 89.61 | 88.78 |
| 10 | DMSO | Cu1.5PW12O40 | 120 | 93.79 | 86.22 |
| 11 | DMSO | CePW12O40 | 120 | 96.23 | 93.86 |
| 12 | DMAc | CePW12O40 | 120 | 91.35 | 90.13 |
| 13 | MIBK | CePW12O40 | 120 | 88.92 | 86.74 |
| 14 | n-Butanol | CePW12O40 | 120 | 98.56 | 97.48 |
| 15 | sec-Butanol | CePW12O40 | 120 | 99.10 | 98.15 |
| Source | Sum of Squares | DF * | Mean Square | F-Value | Probability > F | |
|---|---|---|---|---|---|---|
| Model | 14047.75 | 13 | 1080.60 | 1275.81 | <0.0001 | Significant |
| A | 2.62205 | 1 | 2.62 | 3.10 | 0.1290 | - |
| B | 250.6561 | 1 | 250.66 | 295.94 | <0.0001 | - |
| C | 923.6402 | 1 | 923.64 | 1090.50 | <0.0001 | - |
| AB | 308.7613 | 1 | 308.76 | 364.54 | <0.0001 | - |
| AC | 641.8945 | 1 | 641.89 | 757.85 | <0.0001 | - |
| BC | 790.8265 | 1 | 790.83 | 933.69 | <0.0001 | - |
| A2 | 2237.45 | 1 | 2237.45 | 2641.65 | <0.0001 | - |
| B2 | 5554.388 | 1 | 5554.39 | 6557.81 | <0.0001 | - |
| C2 | 2010.072 | 1 | 2010.07 | 2373.20 | <0.0001 | - |
| A2B | 33.64851 | 1 | 33.65 | 39.73 | 0.0007 | - |
| A2C | 23.48121 | 1 | 23.48 | 27.72 | 0.0019 | - |
| AB2 | 18.70431 | 1 | 18.70 | 22.08 | 0.0033 | |
| A2B2 | 50.90518 | 1 | 50.91 | 60.10 | 0.0002 | - |
| Residual | 5.081933 | 6 | 0.85 | - | - | - |
| Lack of Fit | 2.42 | 1 | 2.42 | 4.55 | 0.0862 | Not significant |
| Pure Error | 2.661933 | 5 | 0.53 | - | - | - |
| Cor Total | 14052.83 | 19 | - | - | - | - |
| Entry | Temperature (°C) | k (min−1) | Correlation Coefficient |
|---|---|---|---|
| 1 | 80 | 0.001131 ± 9.1531 × 10−5 | 0.9963 |
| 2 | 100 | 0.001761 ± 4.0587 × 10−5 | 0.9934 |
| 3 | 120 | 0.002755 ± 3.3841 × 10−4 | 0.9959 |
| 4 | 140 | 0.004421 ± 2.4758 × 10−4 | 0.9872 |
| 5 | 160 | 0.006069 ± 1.1199 × 10−4 | 0.9973 |
| Kinetic Parameters | Value |
|---|---|
| Reaction order, n | 1.0 |
| Activation energy, Ea (kJ/mol) | 27.21 |
| Pre-exponential factor, A (min−1) | 11.72 |
| Correlation coefficient | 0.9869 |
| Variable | Unit | Symbol | Range and Level | ||
|---|---|---|---|---|---|
| −1 | 0 | 1 | |||
| Fructose loading | mg/mL | x1 | 5.0 | 7.5 | 10 |
| Temperature | °C | x2 | 140 | 160 | 180 |
| Reaction time | min | x3 | 60 | 120 | 180 |
| Run | Experimental Variable | Conv.% | 5-HMF Yield (% Mole) | ||
|---|---|---|---|---|---|
| A (mg/mL) | B (°C) | C (min) | |||
| 1 | 5.00 | 140.00 | 180.00 | 85.61 | 83.45 |
| 2 | 5.00 | 180.00 | 60.00 | 78.23 | 75.81 |
| 3 | 7.50 | 160.00 | 120.00 | 99.63 | 99.06 |
| 4 | 7.50 | 193.64 | 120.00 | 30.31 | 26.17 |
| 5 | 10.00 | 140.00 | 60.00 | 26.87 | 24.31 |
| 6 | 5.00 | 140.00 | 60.00 | 37.25 | 35.69 |
| 7 | 10.00 | 180.00 | 180.00 | 93.92 | 42.10 |
| 8 | 10.00 | 180.00 | 60.00 | 53.45 | 50.12 |
| 9 | 7.50 | 160.00 | 180.00 | 88.32 | 79.10 |
| 10 | 5.00 | 180.00 | 180.00 | 95.64 | 70.15 |
| 11 | 10.00 | 140.00 | 180.00 | 83.59 | 82.45 |
| 12 | 7.50 | 140.00 | 120.00 | 50.71 | 47.32 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Song, Y.; Wang, X.; Qu, Y.; Huang, C.; Li, Y.; Chen, B. Efficient Dehydration of Fructose to 5-Hydroxy-methylfurfural Catalyzed by Heteropolyacid Salts. Catalysts 2016, 6, 49. https://doi.org/10.3390/catal6040049
Song Y, Wang X, Qu Y, Huang C, Li Y, Chen B. Efficient Dehydration of Fructose to 5-Hydroxy-methylfurfural Catalyzed by Heteropolyacid Salts. Catalysts. 2016; 6(4):49. https://doi.org/10.3390/catal6040049
Chicago/Turabian StyleSong, Yanlei, Xincheng Wang, Yongshui Qu, Chongpin Huang, Yingxia Li, and Biaohua Chen. 2016. "Efficient Dehydration of Fructose to 5-Hydroxy-methylfurfural Catalyzed by Heteropolyacid Salts" Catalysts 6, no. 4: 49. https://doi.org/10.3390/catal6040049
APA StyleSong, Y., Wang, X., Qu, Y., Huang, C., Li, Y., & Chen, B. (2016). Efficient Dehydration of Fructose to 5-Hydroxy-methylfurfural Catalyzed by Heteropolyacid Salts. Catalysts, 6(4), 49. https://doi.org/10.3390/catal6040049






