Proton Exchange Membrane Fuel Cell Reversal: A Review
Abstract
:1. Introduction
2. PEMFC Cell Reversal Description
3. Causes and Consequences of Cell Reversal
3.1. Causes of Cell Reversal
3.1.1. PEMFC Anode
3.1.2. PEMFC Cathode
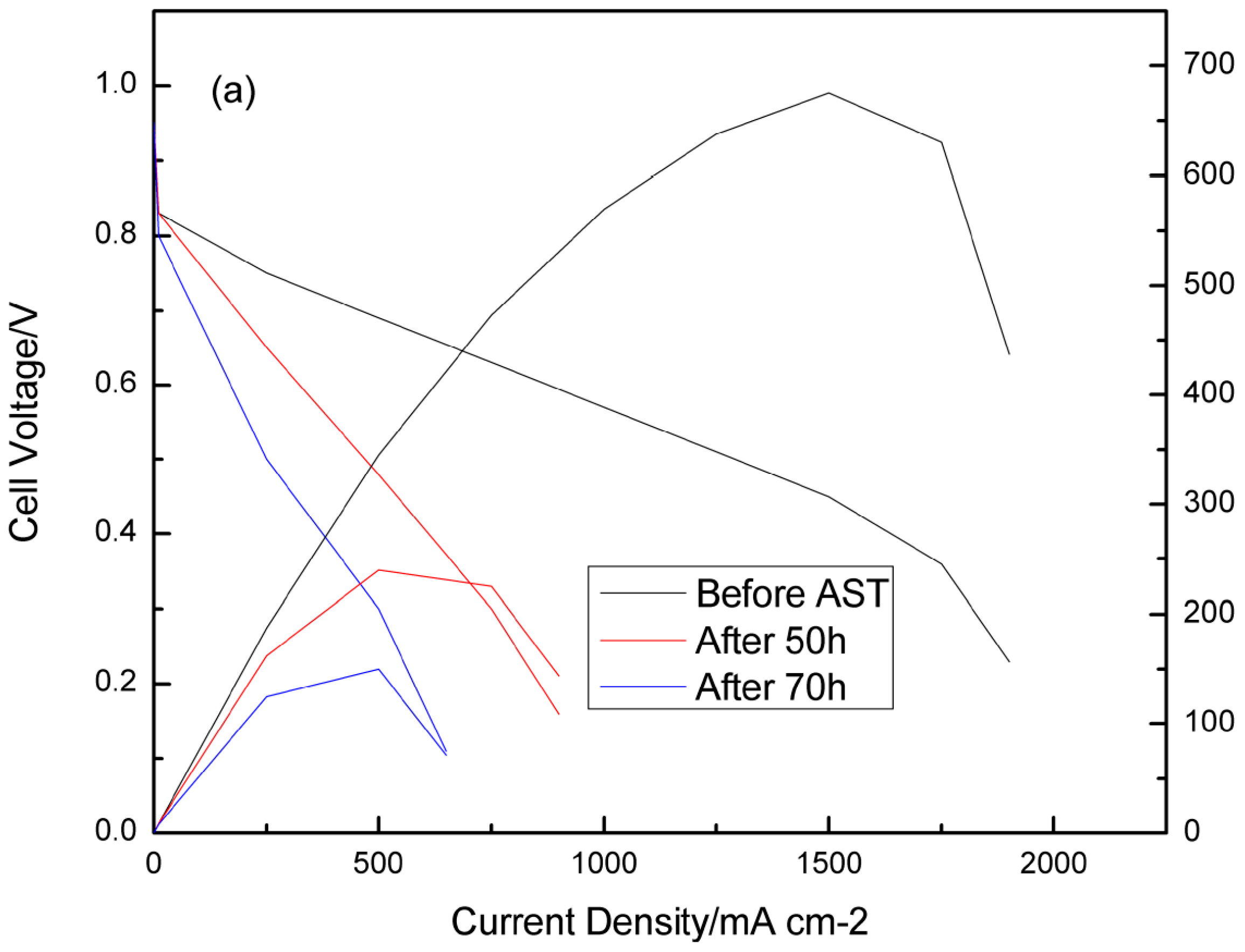
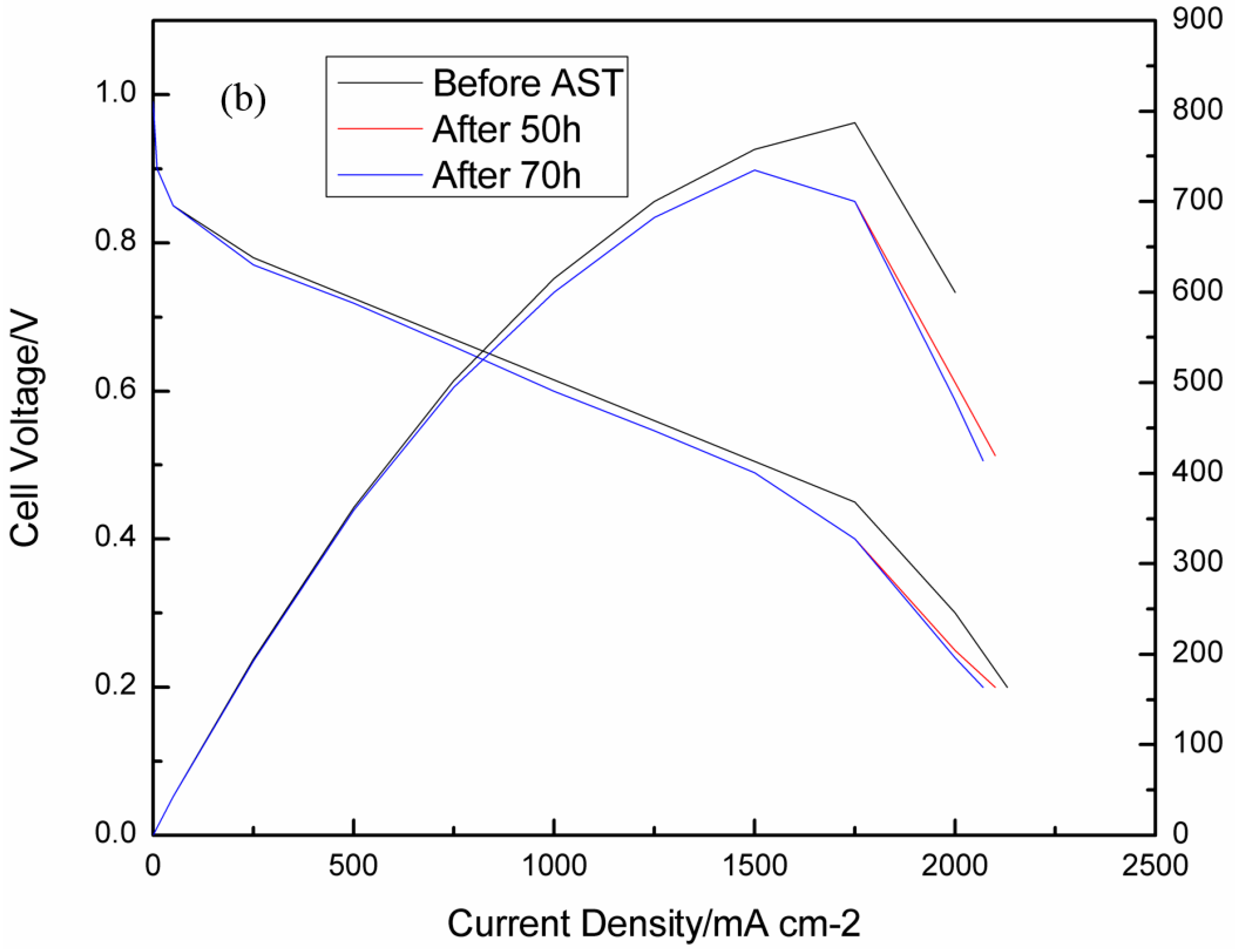
3.2. Consequences of Cell Reversal
4. Mitigation Strategies
4.1. System Management Strategies
4.2. System Material Modification
- High catalyst activity and homogenous distribution;
- Corrosion-resistant support material;
- Water oxidation catalysts to promote water oxidation instead of carbon corrosion;
- Increasing anode gas humidity.
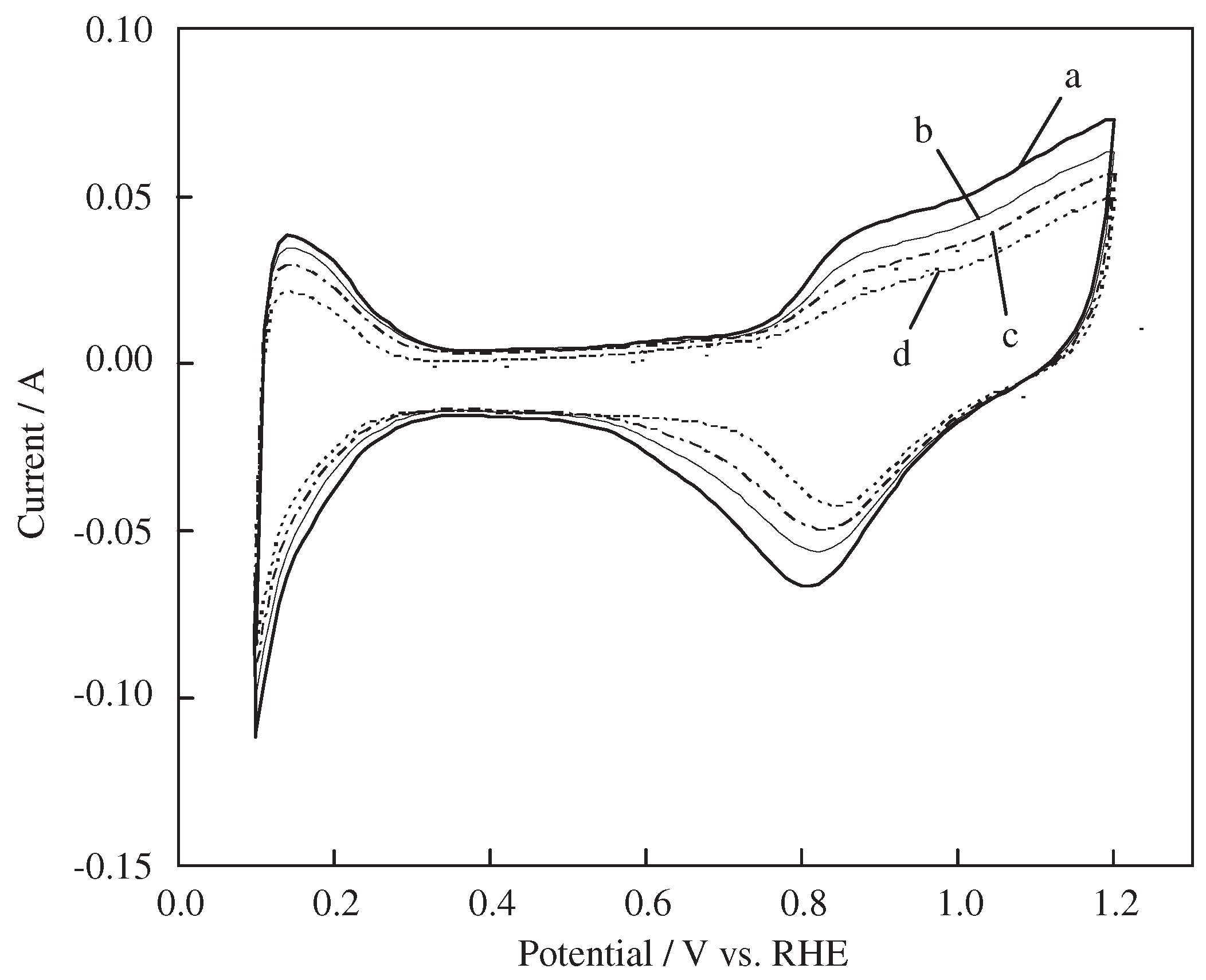
4.2.1. High-Performance Catalyst
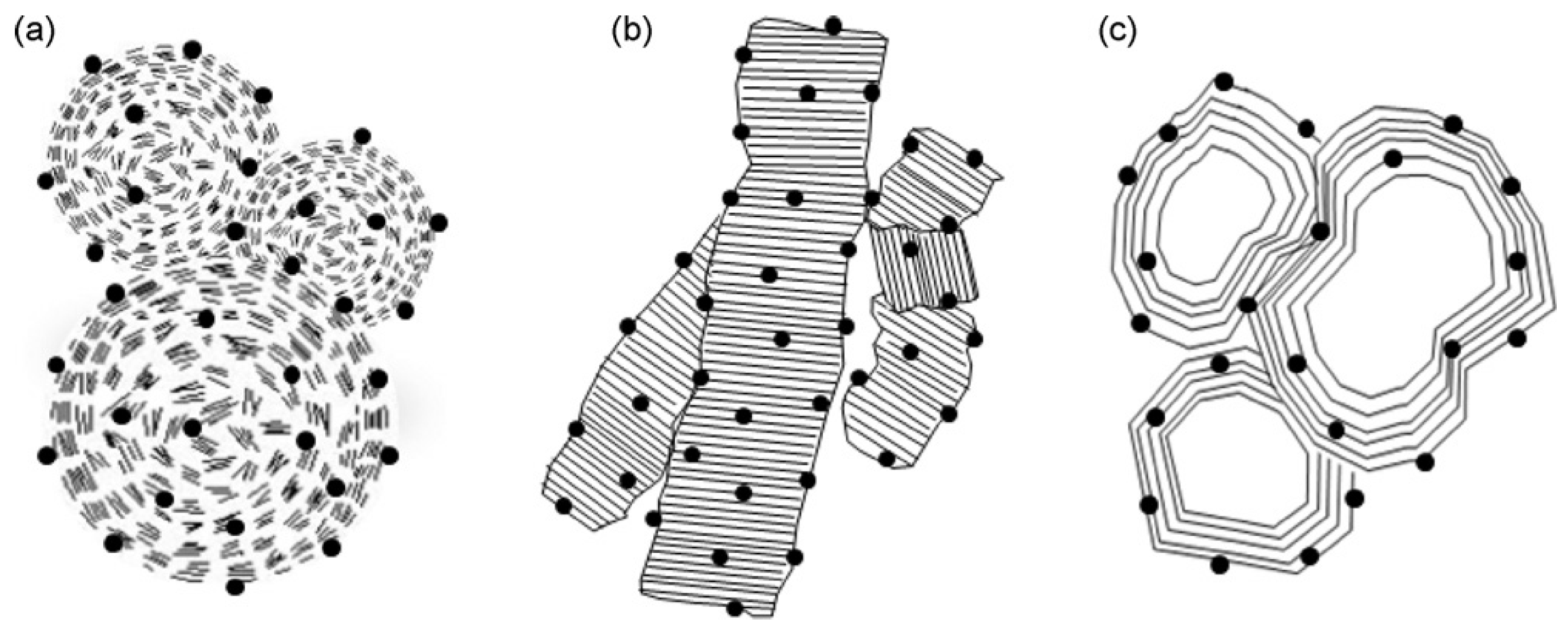
4.2.2. Carbon Support Material
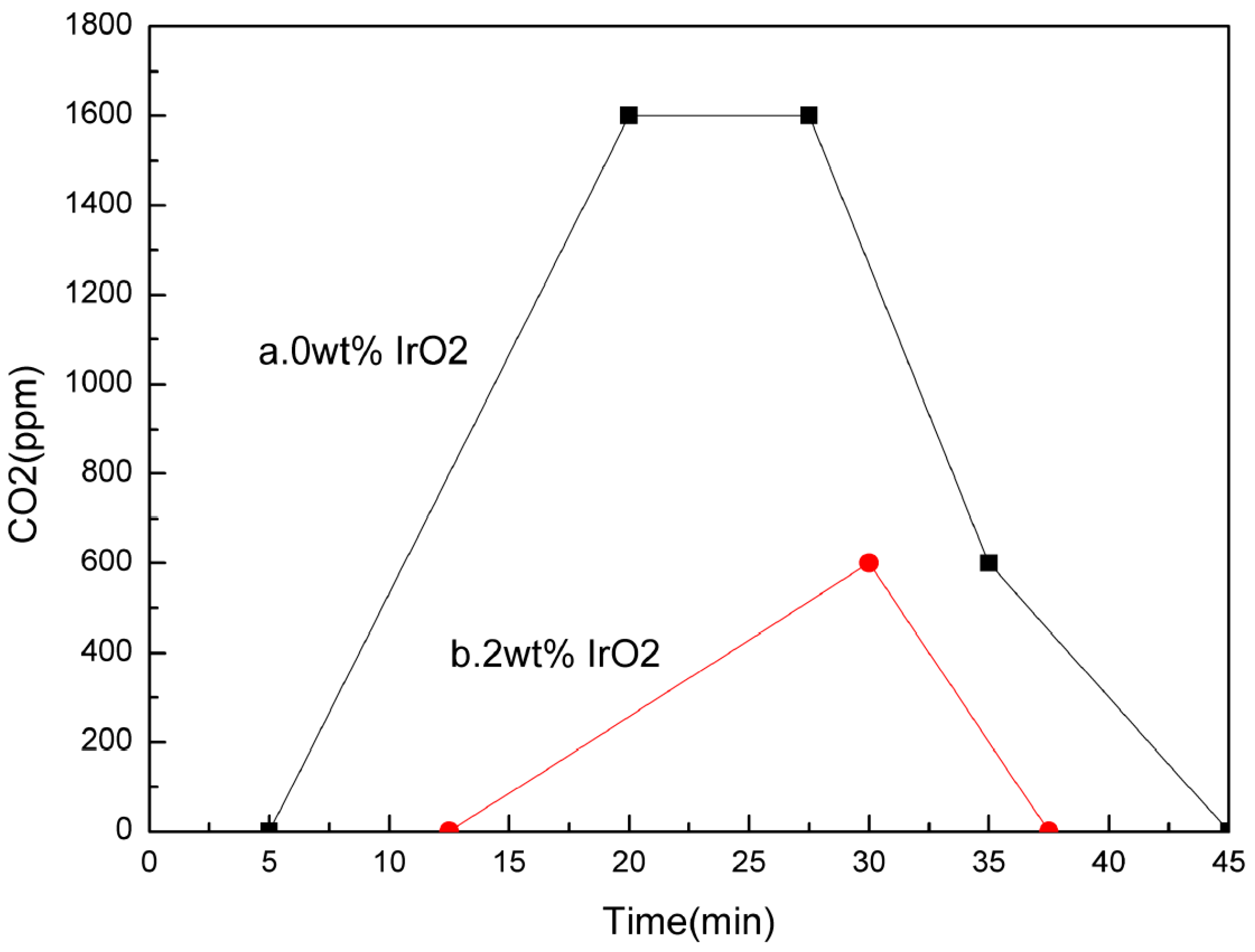
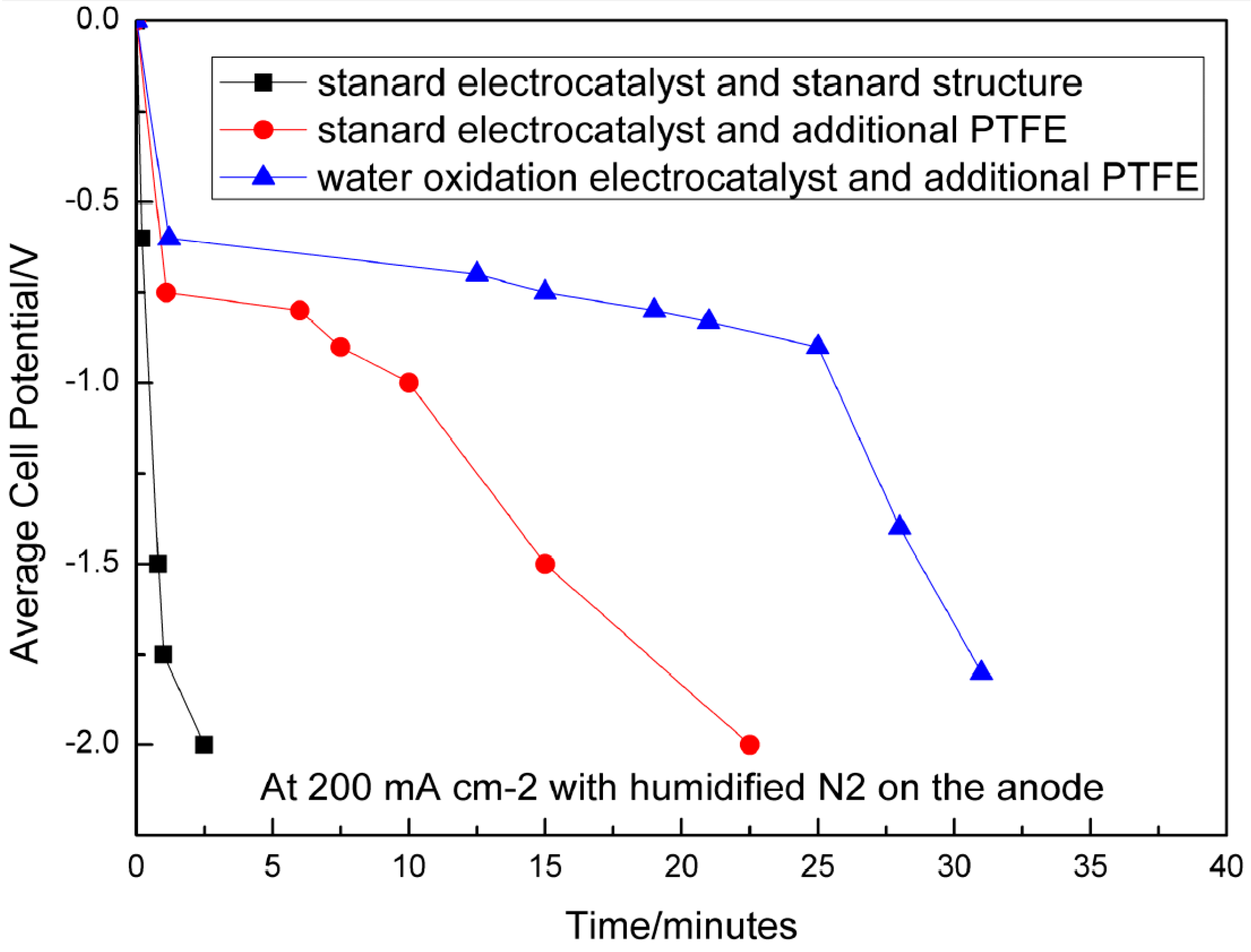
4.2.3. Water Oxidation Reaction
Water Oxidation Catalyst
Control Humidity and Holding Water Volume
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Yi, B. Fuel Cell-Principle, Technology and Application; Chemical Industry: Beijing, China, 2003; p. 41. [Google Scholar]
- Rajashekara, K.; Rathore, A.K. Power conversion and control for fuel cell systems in transportation and stationary power generation. Electr. Power Compon. Syst. 2015, 43, 1376–1387. [Google Scholar] [CrossRef]
- Popovich, N. US Department of Energy Hydrogen and Fuel Cells Program 2015 Annual Merit Review and Peer Evaluation Report: June 8–12, 2015, Arlington, Virginia; NREL (National Renewable Energy Laboratory (NREL): Golden, CO, USA, 2015. [Google Scholar]
- Zhou, F.; Andreasen, S.J.; Kær, S.K. Experimental study of cell reversal of a high temperature polymer electrolyte membrane fuel cell caused by H2 starvation. Int. J. Hydrogen Energy 2015, 40, 6672–6680. [Google Scholar] [CrossRef]
- Taniguchi, A.; Akita, T.; Yasuda, K.; Miyazaki, Y. Analysis of degradation in PEMFC caused by cell reversal during air starvation. Int. J. Hydrogen Energy 2008, 33, 2323–2329. [Google Scholar] [CrossRef]
- Taniguchi, A.; Akita, T.; Yasuda, K.; Miyazaki, Y. Analysis of electrocatalyst degradation in PEMFC caused by cell reversal during fuel starvation. J. Power Sources 2004, 130, 42–49. [Google Scholar] [CrossRef]
- Zhang, G.; Shen, S.; Guo, L.; Liu, H. Dynamic characteristics of local current densities and temperatures in proton exchange membrane fuel cells during reactant starvations. Int. J. Hydrogen Energy 2012, 37, 1884–1892. [Google Scholar] [CrossRef]
- Yu, Y.; Li, H.; Wang, H.; Yuan, X.-Z.; Wang, G.; Pan, M. A review on performance degradation of proton exchange membrane fuel cells during startup and shutdown processes: Causes, consequences, and mitigation strategies. J. Power Sources 2012, 205, 10–23. [Google Scholar] [CrossRef]
- Dou, M.; Hou, M.; Liang, D.; Shen, Q.; Zhang, H.; Lu, W.; Shao, Z.; Yi, B. Behaviors of proton exchange membrane fuel cells under oxidant starvation. J. Power Sources 2011, 196, 2759–2762. [Google Scholar] [CrossRef]
- Castanheira, L.; Silva, W.O.; Lima, F.H.; Crisci, A.; Dubau, L.; Maillard, F.D.R. Carbon corrosion in proton-exchange membrane fuel cells: Effect of the carbon structure, the degradation protocol, and the gas atmosphere. ACS Catal. 2015, 5, 2184–2194. [Google Scholar] [CrossRef]
- Knights, S.D.; Colbow, K.M.; St-Pierre, J.; Wilkinson, D.P. Aging mechanisms and lifetime of PEFC and DMFC. J. Power Sources 2004, 127, 127–134. [Google Scholar] [CrossRef]
- Zhou, F.; Andreasen, S.J.; Kær, S.K.; Yu, D. Analysis of accelerated degradation of a HT-PEM fuel cell caused by cell reversal in fuel starvation condition. Int. J. Hydrogen Energy 2015, 40, 2833–2839. [Google Scholar] [CrossRef]
- Zhang, S.; Yuan, X.-Z.; Hin, J.N.C.; Wang, H.; Friedrich, K.A.; Schulze, M. A review of platinum-based catalyst layer degradation in proton exchange membrane fuel cells. J. Power Sources 2009, 194, 588–600. [Google Scholar] [CrossRef]
- Kang, J.; Jung, D.W.; Park, S.; Lee, J.-H.; Ko, J.; Kim, J. Accelerated test analysis of reversal potential caused by fuel starvation during PEMFCs operation. Int. J. Hydrogen Energy 2010, 35, 3727–3735. [Google Scholar] [CrossRef]
- Yousfi-Steiner, N.; Moçotéguy, P.; Candusso, D.; Hissel, D. A review on polymer electrolyte membrane fuel cell catalyst degradation and starvation issues: Causes, consequences and diagnostic for mitigation. J. Power Sources 2009, 194, 130–145. [Google Scholar] [CrossRef]
- Ralph, T.R.; Hudson, S.; Wilkinson, D.P. Electrocatalyst stability in PEMFCs and the role of fuel starvation and cell reversal tolerant anodes. ECS Trans. 2006, 1, 67–84. [Google Scholar]
- Yang, X.-G.; Ye, Q.; Cheng, P. Hydrogen pumping effect induced by fuel starvation in a single cell of a PEM fuel cell stack at galvanostatic operation. Int. J. Hydrogen Energy 2012, 37, 14439–14453. [Google Scholar] [CrossRef]
- Li, B.; Higgins, D.C.; Xiao, Q.; Yang, D.; Zhng, C.; Cai, M.; Chen, Z.; Ma, J. The durability of carbon supported Pt nanowire as novel cathode catalyst for a 1.5 kW PEMFC stack. Appl. Catal. B 2015, 162, 133–140. [Google Scholar] [CrossRef]
- Sakamoto, S.; Karakane, M.; Maeda, H.; Miyake, Y.; Susai, T.; Isono, T. Study of the Factors Affecting PEFC Life Characteristic; Ecology and Energy Systems Research Center, SANYO Electric Co., Ltd.: Tochigi, Japan, 2000; pp. 326–8534. [Google Scholar]
- Kim, S.; Shimpalee, S.; van Zee, J. The effect of stoichiometry on dynamic behavior of a proton exchange membrane fuel cell (PEMFC) during load change. J. Power Sources 2004, 135, 110–121. [Google Scholar] [CrossRef]
- Rao, R.M.; Rengaswamy, R. A distributed dynamic model for chronoamperometry, chronopotentiometry and gas starvation studies in PEM fuel cell cathode. Chem. Eng. Sci. 2006, 61, 7393–7409. [Google Scholar]
- Shen, Q.; Hou, M.; Yan, X.; Liang, D.; Zang, Z.; Hao, L.; Shao, Z.; Hou, Z.; Ming, P.; Yi, B. The voltage characteristics of proton exchange membrane fuel cell (PEMFC) under steady and transient states. J. Power Sources 2008, 179, 292–296. [Google Scholar] [CrossRef]
- Liu, Z.; Yang, L.; Mao, Z.; Zhuge, W.; Zhang, Y.; Wang, L. Behavior of PEMFC in starvation. J. Power Sources 2006, 157, 166–176. [Google Scholar] [CrossRef]
- Büchi, F.N.; Inaba, M.; Schmidt, T.J. Polymer Electrolyte Fuel Cell Durability; Springer: New York, NY, USA, 2009. [Google Scholar]
- Ofstad, A.; Davey, J.; Sunde, S.; Borup, R.L. Carbon corrosion of a PEMFC during shut-down/start-up when using an air purge procedure. ECS Trans. 2008, 16, 1301–1311. [Google Scholar]
- Hu, J.; Sui, P.; Kumar, S.; Djilali, N. Modelling and simulations of carbon corrosion during operation of a polymer electrolyte membrane fuel cell. Electrochim. Acta 2009, 54, 5583–5592. [Google Scholar] [CrossRef]
- Patterson, T.W.; Darling, R.M. Damage to the cathode catalyst of a PEM fuel cell caused by localized fuel starvation. Electrochem. Solid-State Lett. 2006, 9, A183–A185. [Google Scholar] [CrossRef]
- Lim, K.H.; Oh, H.-S.; Jang, S.-E.; Ko, Y.-J.; Kim, H.-J.; Kim, H. Effect of operating conditions on carbon corrosion in polymer electrolyte membrane fuel cells. J. Power Sources 2009, 193, 575–579. [Google Scholar] [CrossRef]
- Ralph, T.; Hogarth, M. Catalysis for low temperature fuel cells. Platin. Met. Rev. 2002, 46, 117–135. [Google Scholar]
- Liang, D.; Shen, Q.; Hou, M.; Shao, Z.; Yi, B. Study of the cell reversal process of large area proton exchange membrane fuel cells under fuel starvation. J. Power Sources 2009, 194, 847–853. [Google Scholar] [CrossRef]
- Liang, D.; Dou, M.; Hou, M.; Shen, Q.; Shao, Z.; Yi, B. Behavior of a unit proton exchange membrane fuel cell in a stack under fuel starvation. J. Power Sources 2011, 196, 5595–5598. [Google Scholar] [CrossRef]
- Tüber, K.; Pócza, D.; Hebling, C. Visualization of water buildup in the cathode of a transparent PEM fuel cell. J. Power Sources 2003, 124, 403–414. [Google Scholar] [CrossRef]
- Natarajan, D.; van Nguyen, T. Current distribution in PEM fuel cells. Part 2: Air operation and temperature effect. AIChE J. 2005, 51, 2599–2608. [Google Scholar] [CrossRef]
- Rangel, C.; Travassos, M.A.; Fernandes, V.R.; Silva, R.; Paiva, T. Fuel Starvation: Irreversible Degradation Mechanisms in PEM Fuel Cells; WHEC—World Hydrogen Energy Convention: Essen, Germany, 2010. [Google Scholar]
- Zhang, J.; Yang, H.; Fang, J.; Zou, S. Synthesis and oxygen reduction activity of shape-controlled Pt3Ni nanopolyhedra. Nano Lett. 2010, 10, 638–644. [Google Scholar] [CrossRef] [PubMed]
- Yu, X.; Ye, S. Recent advances in activity and durability enhancement of Pt/C catalytic cathode in PEMFC: Part II: Degradation mechanism and durability enhancement of carbon supported platinum catalyst. J. Power Sources 2007, 172, 145–154. [Google Scholar] [CrossRef]
- Wilkinson, D.; St-Pierre, J.; Vielstich, W.; Gasteiger, H.; Lamm, A. Handbook of fuel cells—Fundamentals, technology and applications. Fuel Cell Technol. Appl. 2003, 3, 611. [Google Scholar]
- Maass, S.; Finsterwalder, F.; Frank, G.; Hartmann, R.; Merten, C. Carbon support oxidation in PEM fuel cell cathodes. J. Power Sources 2008, 176, 444–451. [Google Scholar] [CrossRef]
- Barton, R.H. Preventing Voltage Reversal Conditions in Solid Polymer Electrolyte Cells; Heating Resistor and Rectifier in Series; Sensing Thermistor; Detecting a Low Voltage in a Fuel Cell. U.S. Patent 6,724,194, 20 April 2004. [Google Scholar]
- Baumgartner, W.; Parz, P.; Fraser, S.; Wallnöfer, E.; Hacker, V. Polarization study of a PEMFC with four reference electrodes at hydrogen starvation conditions. J. Power Sources 2008, 182, 413–421. [Google Scholar] [CrossRef]
- Perry, M.L.; Patterson, T.; Reiser, C. Systems strategies to mitigate carbon corrosion in fuel cells. ECS Trans. 2006, 3, 783–795. [Google Scholar]
- Vahidi, A.; Stefanopoulou, A.; Peng, H. Model predictive control for starvation prevention in a hybrid fuel cell system. In Proceedings of the 2004 American Control Conference, 30 June–2 July 2004; IEEE: Boston, MA, USA; pp. 834–839.
- Jung, N.; Chung, D.Y.; Ryu, J.; Yoo, S.J.; Sung, Y.-E. Pt-based nanoarchitecture and catalyst design for fuel cell applications. Nano Today 2014, 9, 433–456. [Google Scholar] [CrossRef]
- Wang, J.; Yin, G.; Shao, Y.; Zhang, S.; Wang, Z.; Gao, Y. Effect of carbon black support corrosion on the durability of Pt/C catalyst. J. Power Sources 2007, 171, 331–339. [Google Scholar] [CrossRef]
- Luo, Z.; Li, D.; Tang, H.; Pan, M.; Ruan, R. Degradation behavior of membrane–electrode-assembly materials in 10-cell PEMFC stack. Int. J. Hydrogen Energy 2006, 31, 1831–1837. [Google Scholar] [CrossRef]
- Yoda, T.; Uchida, H.; Watanabe, M. Effects of operating potential and temperature on degradation of electrocatalyst layer for PEFCs. Electrochim. Acta 2007, 52, 5997–6005. [Google Scholar] [CrossRef]
- Colón-Mercado, H.R.; Popov, B.N. Stability of platinum based alloy cathode catalysts in PEM fuel cells. J. Power Sources 2006, 155, 253–263. [Google Scholar] [CrossRef]
- Gasteiger, H.A.; Kocha, S.S.; Sompalli, B.; Wagner, F.T. Activity benchmarks and requirements for Pt, Pt–alloy, and non-Pt oxygen reduction catalysts for PEMFCs. Appl. Catal. B 2005, 56, 9–35. [Google Scholar] [CrossRef]
- Wan, L.J.; Moriyama, T.; Ito, M.; Uchida, H.; Watanabe, M. In situ STM imaging of surface dissolution and rearrangement of a Pt–Fe alloy electrocatalyst in electrolyte solution. Chem. Commun. 2002. [Google Scholar] [CrossRef]
- Paulus, U.; Wokaun, A.; Scherer, G.; Schmidt, T.; Stamenkovic, V.; Radmilovic, V.; Markovic, N.; Ross, P. Oxygen reduction on carbon-supported Pt–Ni and Pt–Co alloy catalysts. J. Phys. Chem. B 2002, 106, 4181–4191. [Google Scholar] [CrossRef]
- Yu, P.; Pemberton, M.; Plasse, P. PtCo/C cathode catalyst for improved durability in PEMFCs. J. Power Sources 2005, 144, 11–20. [Google Scholar] [CrossRef]
- Zhang, J.; Sasaki, K.; Sutter, E.; Adzic, R. Stabilization of platinum oxygen-reduction electrocatalysts using gold clusters. Science 2007, 315, 220–222. [Google Scholar] [CrossRef] [PubMed]
- Stevens, D.; Hicks, M.; Haugen, G.; Dahn, J. Ex situ and in situ stability studies of PEMFC catalysts effect of carbon type and humidification on degradation of the carbon. J. Electrochem. Soc. 2005, 152, A2309–A2315. [Google Scholar] [CrossRef]
- Kim, M.; Jung, N.; Eom, K.; Yoo, S.J.; Kim, J.Y.; Jang, J.H.; Kim, H.-J.; Hong, B.K.; Cho, E. Effects of anode flooding on the performance degradation of polymer electrolyte membrane fuel cells. J. Power Sources 2014, 266, 332–340. [Google Scholar] [CrossRef]
- Antolini, E. Graphene as a new carbon support for low-temperature fuel cell catalysts. Appl. Catal. B 2012, 123, 52–68. [Google Scholar] [CrossRef]
- Guo, S.; Sun, S. FePt nanoparticles assembled on graphene as enhanced catalyst for oxygen reduction reaction. J. Am. Chem. Soc. 2012, 134, 2492–2495. [Google Scholar] [CrossRef] [PubMed]
- Artyushkova, K.; Atanassov, P.; Dutta, M.; Wessel, S.; Colbow, V. Structural correlations: Design levers for performance and durability of catalyst layers. J. Power Sources 2015, 284, 631–641. [Google Scholar] [CrossRef]
- Artyushkova, K.; Pylypenko, S.; Dowlapalli, M.; Atanassov, P. Structure-to-property relationships in fuel cell catalyst supports: Correlation of surface chemistry and morphology with oxidation resistance of carbon blacks. J. Power Sources 2012, 214, 303–313. [Google Scholar] [CrossRef]
- Wlodarczyk, R. Porous carbon materials for elements in low-temperature fuel cells. Arch. Metall. Mater. 2015, 60, 117–120. [Google Scholar] [CrossRef]
- Oh, H.-S.; Lim, K.H.; Roh, B.; Hwang, I.; Kim, H. Corrosion resistance and sintering effect of carbon supports in polymer electrolyte membrane fuel cells. Electrochim. Acta 2009, 54, 6515–6521. [Google Scholar] [CrossRef]
- Dicks, A.L. The role of carbon in fuel cells. J. Power Sources 2006, 156, 128–141. [Google Scholar]
- Yang, W.; Wang, Y.; Li, J.; Yang, X. Polymer wrapping technique: An effective route to prepare Pt nanoflower/carbon nanotube hybrids and application in oxygen reduction. Energy Environ. Sci. 2010, 3, 144–149. [Google Scholar] [CrossRef]
- Oh, H.S.; Kim, H. Efficient synthesis of Pt nanoparticles supported on hydrophobic graphitized carbon nanofibers for electrocatalysts using noncovalent functionalization. Adv. Funct. Mater. 2011, 21, 3954–3960. [Google Scholar] [CrossRef]
- Lim, K.H.; Oh, H.-S.; Kim, H. Use of a carbon nanocage as a catalyst support in polymer electrolyte membrane fuel cells. Electrochem. Commun. 2009, 11, 1131–1134. [Google Scholar] [CrossRef]
- Vinod Selvaganesh, S.; Selvarani, G.; Sridhar, P.; Pitchumani, S.; Shukla, A. Graphitic carbon as durable cathode-catalyst support for PEFCs. Fuel Cells 2011, 11, 372–384. [Google Scholar] [CrossRef]
- Zhao, X.; Hayashi, A.; Noda, Z.; Kimijima, K.i.; Yagi, I.; Sasaki, K. Evaluation of change in nanostructure through the heat treatment of carbon materials and their durability for the start/stop operation of polymer electrolyte fuel cells. Electrochim. Acta 2013, 97, 33–41. [Google Scholar] [CrossRef]
- Jang, S.-E.; Kim, H. Effect of water electrolysis catalysts on carbon corrosion in polymer electrolyte membrane fuel cells. J. Am. Chem. Soc. 2010, 132, 14700–14701. [Google Scholar] [CrossRef] [PubMed]
- Jang, I.; Hwang, I.; Tak, Y. Attenuated degradation of a PEMFC cathode during fuel starvation by using carbon-supported IrO2. Electrochim. Acta 2013, 90, 148–156. [Google Scholar] [CrossRef]
- Reiser, C.A. Preventing Fuel Starvation of a Fuel Cell Stack. U.S. Patent 7,807,302, 5 October 2010. [Google Scholar]
- Wilkinson, D.P.; Chow, C.Y.; Allan, D.E.; Johannes, E.P.; Roberts, J.A.; St-Pierre, J.; Longley, C.J.; Chan, J.K. Method and Apparatus for Operating an Electrochemical Fuel Cell with Periodic Fuel Starvation at the Anode. U.S. Patent 6,096,448, 1 August 2000. [Google Scholar]
- Park, S.; Lee, J.-W.; Popov, B.N. A review of gas diffusion layer in PEM fuel cells: Materials and designs. Int. J. Hydrogen Energy 2012, 37, 5850–5865. [Google Scholar] [CrossRef]
- Knights, S.D.; Taylor, J.L.; Wilkinson, D.P.; Wainwright, D.S. Fuel Cell Anode Structures for Voltage Reversal Tolerance. U.S. Patent 6,517,962, 11 February 2003. [Google Scholar]
- Lim, C.; Wang, C. Effects of hydrophobic polymer content in GDL on power performance of a PEM fuel cell. Electrochimica Acta 2004, 49, 4149–4156. [Google Scholar] [CrossRef]
- Park, S.; Popov, B.N. Effect of hydrophobicity and pore geometry in cathode GDL on PEM fuel cell performance. Electrochimica Acta 2009, 54, 3473–3479. [Google Scholar] [CrossRef]
- Ismail, M.; Hughes, K.; Ingham, D.; Ma, L.; Pourkashanian, M. Effect of PtFe loading of gas diffusion layers on the performance of proton exchange membrane fuel cells running at high-efficiency operating conditions. Int. J. Energy Res. 2013, 37, 1592–1599. [Google Scholar] [CrossRef]
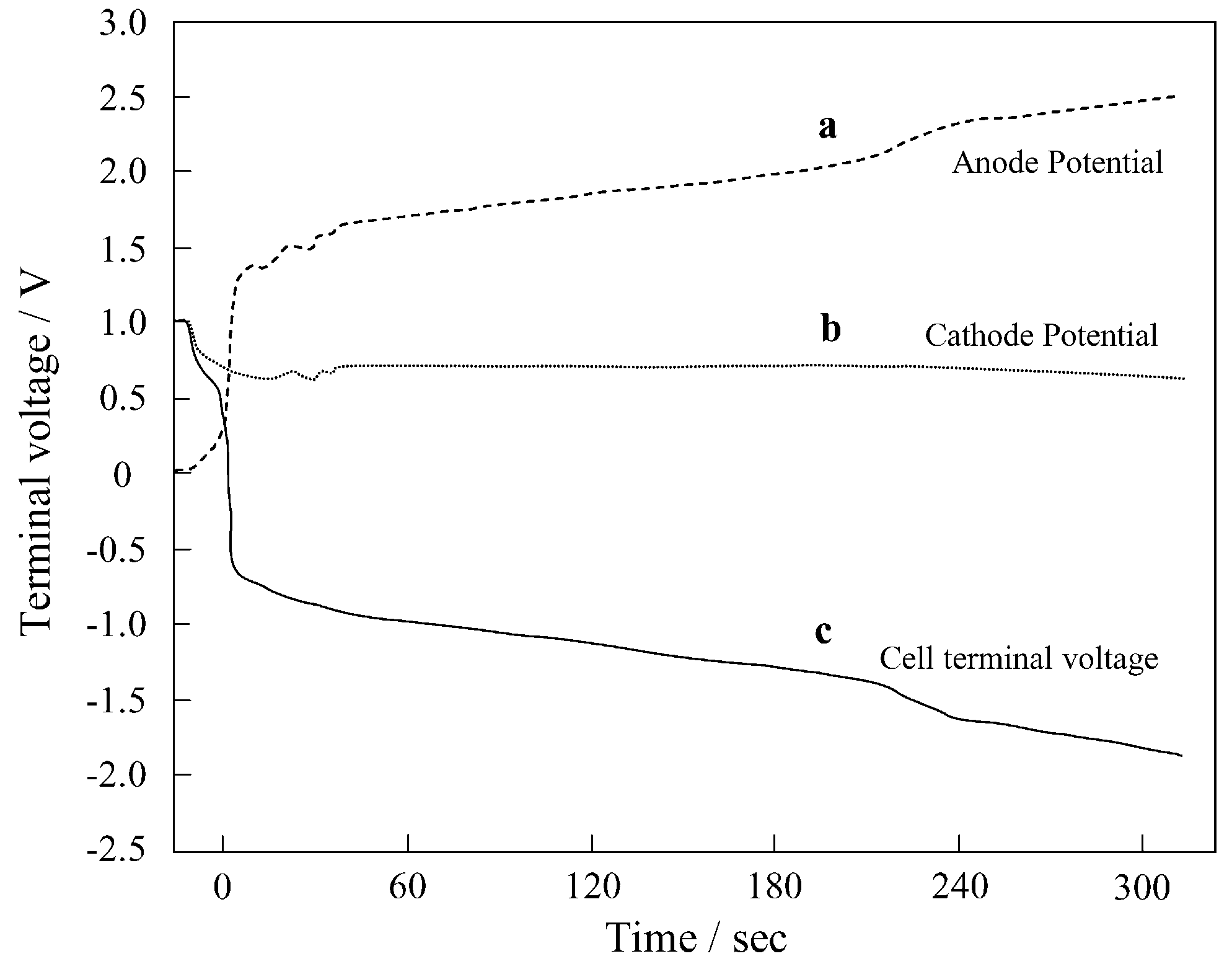
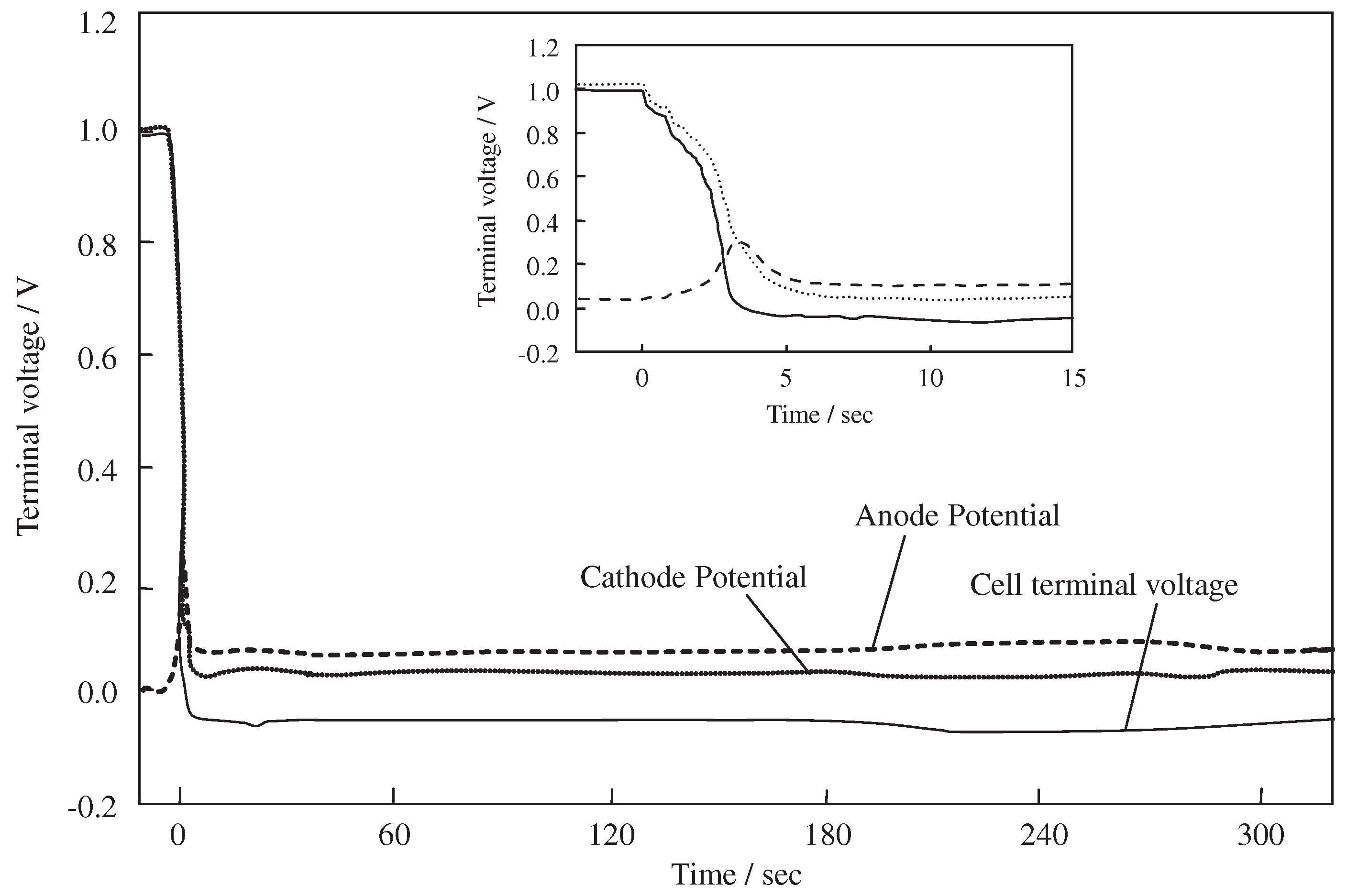
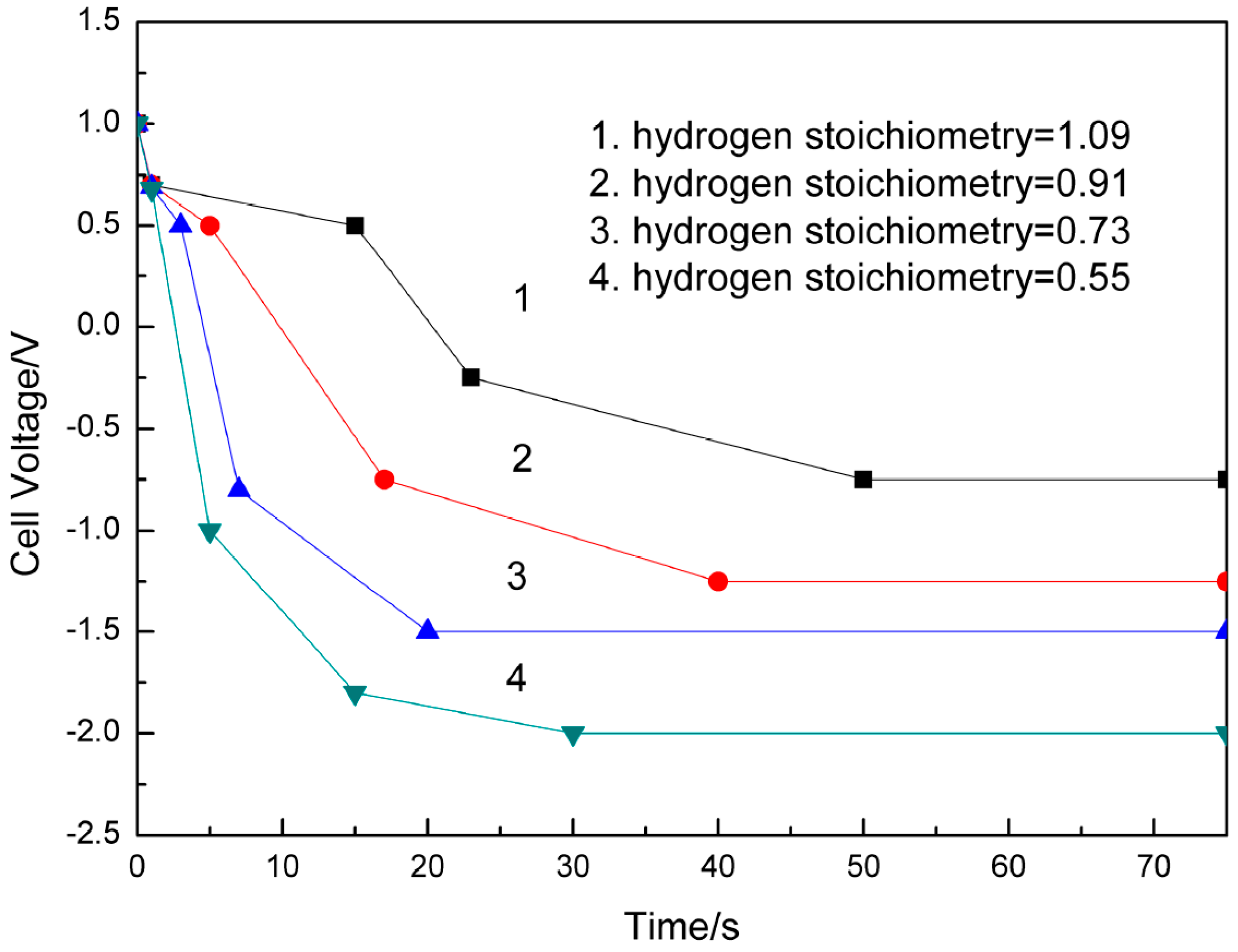
| Hydrogen Stoichiometry | Time for Cell Reversal (s) | Cell Voltage (V) | Anode Potential (V) | Reference |
|---|---|---|---|---|
| 0.8 | Experiment started | About −0.7 | 1.3 | [6] |
| 0.8 | About 300 | −2.0 | 2.5 | |
| 1.09 | 26 | −0.718 | 0.955 | [30] |
| 0.91 | 10 | −1.125 | 1.313 | |
| 0.73 | 6 | −1.689 | 1.821 | |
| 0.55 | 4 | −1.951 | 2.058 |
| Condition | Active Surface Area (m2·g−1) | Membrane Resistance | Charge Transfer Resistance | CO2 Emission (μL) | Reference | |||
|---|---|---|---|---|---|---|---|---|
| Before | After | Before | After | Before | After | 30 min@1.4 V | ||
| Pt/XC-72 (1200 cycle) | 62.2 | 40.0 | – | – | – | – | – | [44] |
| Pt/BP2000 | 59.9 | 26.7 | – | – | – | – | – | – |
| Pt/Carbon Black (up to 4000 cycle) | 41.7 | 15.2 | 0.016 | 0.025 | 0.039 | 0.328 | 651 | [60] |
| Pt/CNF | 30.1 | 27.7 | 0.016 | 0.016 | 0.054 | 0.058 | 49 | – |
| Pt/CNC | 33.6 | 32.9 | 0.016 | 0.016 | 0.04 | 0.041 | 4 | – |
| Anode Design with Different Catalyst | Time (min) |
|---|---|
| 20% Pt, 10% Ru/Shawinigan anode catalyst | 0.25 |
| Anode Catalyst* + RuO2/Shawinigan | 4.5 |
| Anode Catalyst* + RuO2–TiO2 (90:10 atomic ratio Ru/Ti)/Shawinigan | 24 |
| Anode Catalyst* + RuO2–IrO2 (90:10 atomic ratio Ru/Ir)/Shawinigan | 48 |
| 20% Pt, 10% Ru/Shawinigan+ RuO2–IrO2 (90:10 atomic ratio Ru/Ir) | 74 |
| 40% Pt, 20% Ru/Shawinigan | 167 |
| 40% Pt, 20% Ru/Shawinigan + RuO2–IrO2 (90:10 atomic ratio Ru/Ir) | 1630 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Qin, C.; Wang, J.; Yang, D.; Li, B.; Zhang, C. Proton Exchange Membrane Fuel Cell Reversal: A Review. Catalysts 2016, 6, 197. https://doi.org/10.3390/catal6120197
Qin C, Wang J, Yang D, Li B, Zhang C. Proton Exchange Membrane Fuel Cell Reversal: A Review. Catalysts. 2016; 6(12):197. https://doi.org/10.3390/catal6120197
Chicago/Turabian StyleQin, Congwei, Jue Wang, Daijun Yang, Bing Li, and Cunman Zhang. 2016. "Proton Exchange Membrane Fuel Cell Reversal: A Review" Catalysts 6, no. 12: 197. https://doi.org/10.3390/catal6120197
APA StyleQin, C., Wang, J., Yang, D., Li, B., & Zhang, C. (2016). Proton Exchange Membrane Fuel Cell Reversal: A Review. Catalysts, 6(12), 197. https://doi.org/10.3390/catal6120197






